Chemical Fingerprinting of Seeds of Some Salvia Species in Turkey by Using GC-MS and FTIR
Abstract
:1. Introduction
2. Materials and Methods
2.1. Equipment
2.2. Sampling
2.3. Static HS-GC/MS Analysis
2.4. FTIR Spectroscopy Measurements
2.5. Chemometric Analysis
3. Results and Discussion
3.1. Static HS-GC/MS Analysis
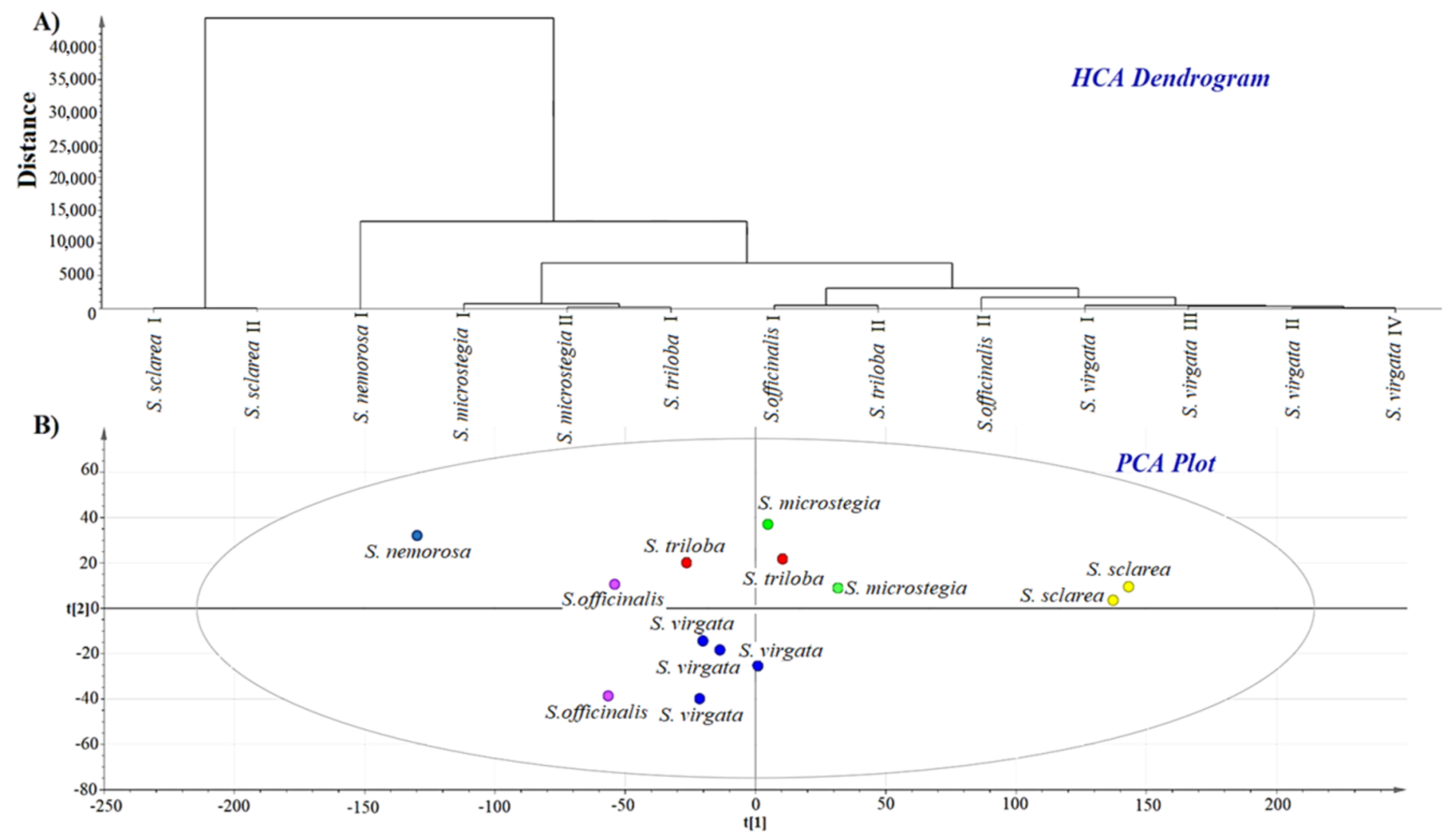
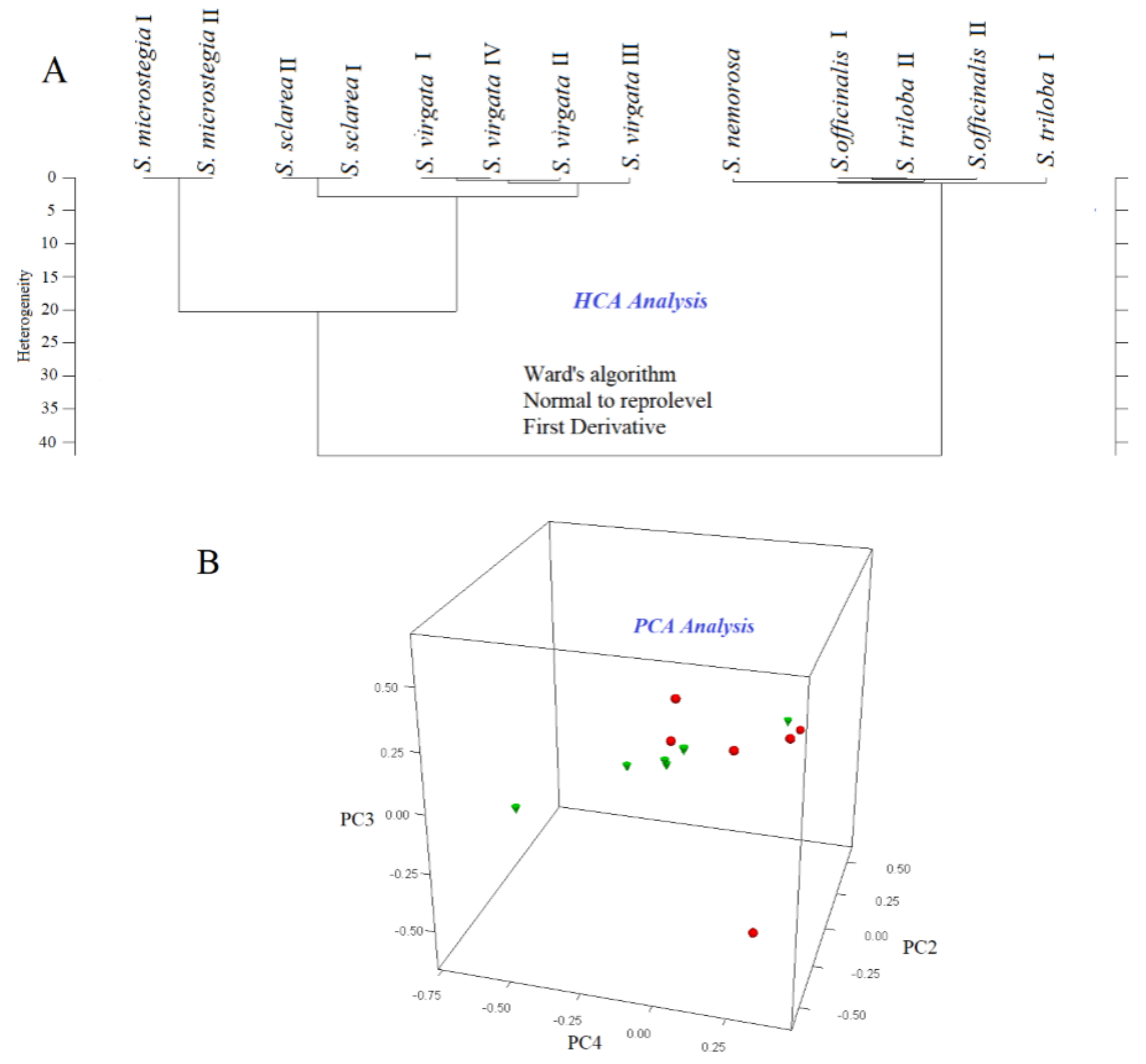
3.2. HCA and PCA of GC-MS data
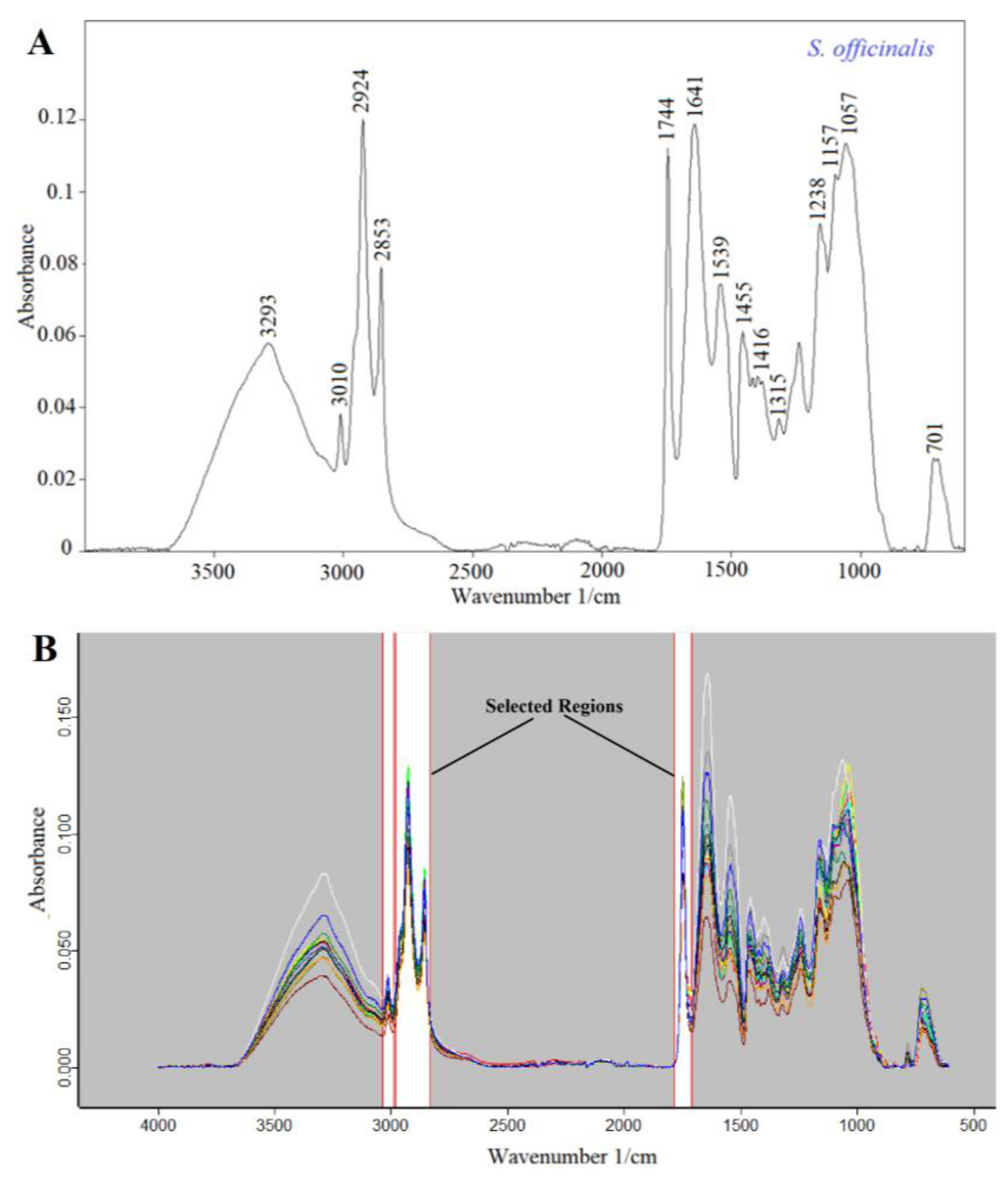
3.3. FTIR-ATR Analysis
Characterization of FTIR spectra of Salvia Seeds
3.4. Discrimination and Clustering of Salvia Seed Species Using FTIR Spectra
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Compliance with Ethics Requirements
References
- Ozkan, G.; Sagdic, O.; Gokturk, R.S.; Unal, O.; Albayrak, S. Study on chemical composition and biological activities of essential oil and extract from Salvia pisidica. LWT Food Sci. Technol. 2010, 43, 186–190. [Google Scholar] [CrossRef]
- Ulukanli, Z.; Karabörklü, S.; Cenet, M.; Sagdic, O.; Ozturk, I.; Balcilar, M. Essential oil composition, insecticidal and antibacterial activities of Salvia tomentosa Miller. Med. Chem. Res. 2013, 22, 832–840. [Google Scholar] [CrossRef]
- Yadav, A.; Joshi, A.; Kothari, S.L.; Kachhwaha, S.; Purohit, S. Medicinal, nutritional and industrial applications of Salvia species: A revisit. Int. J. Pharm. Sci. Rev. Res. 2017, 43, 27–37. [Google Scholar]
- Tulukcu, E.; Sagdic, O.; Albayrak, S.; Ekici, L.; Yetim, H. Effect of collection time on biological activity of clary sage (Salvia Sclarea). J. Appl. Bot. Food Qual. 2009, 83, 44–49. [Google Scholar]
- Yalcin, H.; Ozturk, I.; Tulukcu, E.; Sagdic, O. Effect of γ-Irradiation on Bioactivity, Fatty Acid Compositions and Volatile Compounds of Clary Sage Seed (Salvia sclarea L.). J. Food Sci. 2011, 76, 1056–1061. [Google Scholar] [CrossRef]
- Yurt, M.; Gezer, C. Chia tohumunun (Salvia hispanica) fonksiyonel özellikleri ve sağlık üzerine etkileri. Gıda J. Food 2018, 43, 446–460. [Google Scholar]
- Porras-Loaiza, P.; Jiménez-Munguía, M.T.; Sosa-Morales, M.E.; Palou, E.; López-Malo, A. Physical properties, chemical characterization and fatty acid composition of Mexican chia (Salvia hispanica L.) seeds. Int. J. Food Sci. Technol. 2014, 49, 571–577. [Google Scholar] [CrossRef]
- Rzepa, J.; Wojtal, Ł.; Staszek, D.; Grygierczyk, G.; Labe, K.; Hajnos, M.; Kowalska, T.; Waksmundzka-Hajnos, M. Fingerprint of selected Salvia species by HS-GC-MS analysis of their volatile fraction. J. Chromatogr. Sci. 2009, 47, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Marineli, R.d.S.; Moraes, É.A.; Lenquiste, S.A.; Godoy, A.T.; Eberlin, M.N.; Maróstica, M.R. Chemical characterization and antioxidant potential of Chilean chia seeds and oil (Salvia hispanica L.). LWT Food Sci. Technol. 2014, 59, 1304–1310. [Google Scholar] [CrossRef]
- Capitani, M.I.; Spotorno, V.; Nolasco, S.M.; Tomás, M.C. Physicochemical and functional characterization of by-products from chia (Salvia hispanica L.) seeds of Argentina. LWT - Food Sci. Technol. 2012, 45, 94–102. [Google Scholar] [CrossRef]
- Yilmaz, M.T.; Kutlu, G.; Tulukcu, E.; Toker, O.S.; Sagdic, O.; Karaman, S. Rheological characteristics of Salvia sclarea seed gum solutions at different hydration temperature levels: Application of three interval thixotropy test (3ITT). LWT Food Sci. Technol. 2016, 71, 391–399. [Google Scholar] [CrossRef]
- Sermakkani, M.; Thangapandian, V. GC-MS analysis of Cassia italica leaf methanol extract. Asian J. Pharm. Clin. Res. 2012, 5, 90–94. [Google Scholar]
- Chauhan, A. GC-MS Technique and its Analytical Applications in Science and Technology. J. Anal. Bioanal. Tech. 2014, 5, 222. [Google Scholar] [CrossRef]
- Sivakesava, S.; Irudayaraj, J. Prediction of inverted cane sugar adulteration of honey by Fourier transform infrared spectroscopy. J. Food Sci. 2001, 66, 972–978. [Google Scholar] [CrossRef]
- Rodriguez-Saona, L.E.; Pujolras, M.P.; Giusti, M.M. Targeted and Non-Targeted Analysis. In Analytical Separation Science; Wiley-VCH: Weinheim, Germany, 2015; pp. 1401–1436. [Google Scholar]
- Pop (Cuceu), A.-V.; Tofana, M.; Socaci, S.A.; POP, C.; Rotar, A.M.; Nagy, M.; Salanta, L. Determination of Antioxidant Capacity and Antimicrobial Activity of Selected Salvia Species. Bull. UASVM Food Sci. Technol. 2016, 73, 14–18. [Google Scholar] [CrossRef]
- Pop (Cuceu), A.-V.; Tofana, M.; Socaci, S.A.; Nagy, M.; Bors, M.-D.; Salanta, L.; Vlaic, R. Studies on Total Polyphenols Content and Antioxidant Activity of Methanolic Extractsfrom Selected Salvia Species. Bull. UASVM Food Sci. Technol. 2015, 72, 86–90. [Google Scholar]
- Pop, A.V.; Tofană, M.; Socaci, S.A.; Nagy, M.; Fărcaș, A.; Borș, M.D.; Salanță, L.; Feier, D.; Vârva, L. Comparative study regarding the chemical composition of essential oils of some Salvia species. Hop. Med. Plants 2014, 22, 79–91. [Google Scholar]
- Ehrnhöfer-Ressler, M.M.; Fricke, K.; Pignitter, M.; Walker, J.M.; Walker, J.; Rychlik, M.; Somoza, V. Identification of 1,8-cineole, borneol, camphor, and thujone as anti-inflammatory compounds in a Salvia officinalis L. infusion using human gingival fibroblasts. J. Agric. Food Chem. 2013, 61, 3451–3459. [Google Scholar] [CrossRef]
- Socaci, S.A.; Socaciu, C.; Mureşan, C.; Fǎrcaş, A.; Tofanǎ, M.; Vicaş, S.; Pintea, A. Chemometric discrimination of different tomato cultivars based on their volatile fingerprint in relation to lycopene and total phenolics content. Phytochem. Anal. 2014, 25, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, B.; Xin, Z.; Ren, D.; Yi, L. GC-MS fingerprinting combined with chemometric methods reveals key bioactive components in Acori tatarinowii rhizoma. Int. J. Mol. Sci. 2017, 18, E1342. [Google Scholar] [CrossRef]
- Cebi, N.; Yilmaz, M.T.; Sagdic, O. A rapid ATR-FTIR spectroscopic method for detection of sibutramine adulteration in tea and coffee based on hierarchical cluster and principal component analyses. Food Chem. 2017, 229, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Ordonez, A.; Prieto, M. Fourier Transform Infrared Spectroscopy in Food Microbiology; Springer: New York, NY, USA, 2012; ISBN 9781461438120. [Google Scholar]
- Grassino, A.N.; Brnčić, M.; Vikić-Topić, D.; Roca, S.; Dent, M.; Brnčić, S.R. Ultrasound assisted extraction and characterization of pectin from tomato waste. Food Chem. 2016, 198, 93–100. [Google Scholar] [CrossRef] [PubMed]
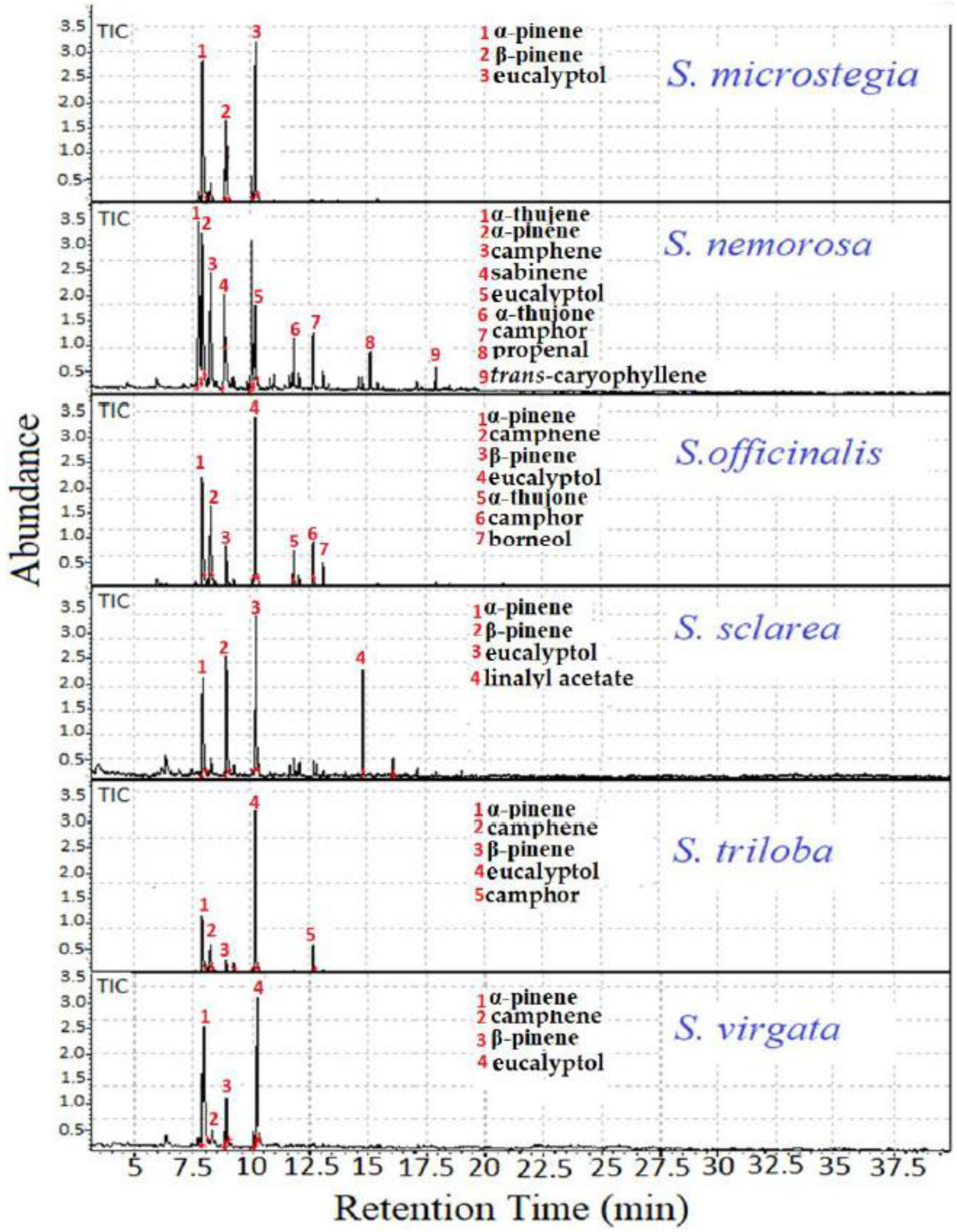
| RI | Compounds (Area %) | S. microstegia I | S. microstegia II | S. nemorosa I | S. officinalis I | S. officinalis II | S. sclarea I | S. sclarea II | S. triloba I | S. triloba II | S. virgata I | S. virgata II | S. virgata III | S. virgata IV |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 861 | n-hexanal | - | - | 3.72 ± 0.14 | 1.58 ± 0.12 | 1.97 ± 0.51 | - | - | - | - | 1.51 ± 0.75 | 16.76 ± 1.31 | 19.44 ± 1.38 | 25.47 ± 1.65 |
| 876 | n-hexanol | 0.68 ± 0.22 | - | - | 0.6 ± 0.24 | 0.29 ± 0.62 | 1.1 ± 0.34 | 3.79 ± 0.62 | 0.24 ± 0.18 | 0.3 ± 0.12 | 1.72 ± 0.42 | 3.67 ± 0.15 | 2.81 ± 0.47 | 0.87 ± 0.72 |
| 926 | tricyclene | 0.18 ± 0.16 | 0.33 ± 0.11 | - | 0.72 ± 0.31 | 0.78 ± 0.34 | - | - | 0.56 ± 0.35 | 0.6 ± 0.67 | - | - | - | - |
| 928 | α-thujene | 2.24 ± 0.73 | 2.67 ± 0.81 | 32.65 ± 1.15 | 0.08 ± 0.05 | - | - | - | - | - | - | - | - | - |
| 939 | α-pinene | 36.46 ± 1.36 | 36.06 ± 1.52 | 0.63 ± 0.17 | 27.16 ± 1.34 | 25.42 ± 1.52 | 8.8 ± 0.91 | 3.81 ± 0.34 | 22.94 ± 0.35 | 24.1 ± 1.44 | 31.29 ± 1.68 | 17.8 ± 1.13 | 14.68 ± 1.24 | 17.16 ± 1.32 |
| 952 | camphene | 3.17 ± 0.28 | 3.95 ± 0.42 | 15.20 ± 1.82 | 17.81 ± 1.17 | 18.02 ± 1.67 | 3.24 ± 0.41 | 3.25 ± 0.67 | 11.52 ± 0.71 | 13 ± 1.74 | 10.9 ± 1.12 | 6.42 ± 0.24 | 4.43 ± 0.15 | 8.2 ± 0.14 |
| 957 | 2,4(10)-thujadien | 0.48 ± 0.14 | 0.79 ± 0.13 | - | - | - | - | - | - | - | - | - | - | - |
| 978 | sabinene | 4.49 ± 0.47 | - | 2.76 ± 0.14 | - | - | - | - | - | - | - | - | 8.88 ± 0.71 | - |
| 979 | β-pinene | 16.32 ± 1.32 | 20.8 ± 1.5 | 3.05 ± 0.18 | 6 ± 0.82 | 7.59 ± 0.52 | 20.79 ± 1.62 | 21.62 ± 1.42 | 4.39 ± 0.51 | 5 ± 0.65 | 12.01 ± 0.43 | 1.66 ± 0.62 | 5.38 ± 0.25 | 9.88 ± 0.81 |
| 991 | β-myrcene | 0.6 ± 0.12 | - | - | 1.02 ± 0.17 | 0.71 ± 0.32 | - | - | 0.11 ± 0.16 | 0.8 ± 0.55 | 3.24 ± 0.27 | - | - | 5.34 ± 0.76 |
| 1026 | α-terpinene | - | - | - | 0.08 ± 0.02 | 0.1 ± 0.42 | - | - | - | - | - | - | - | - |
| 1029 | limonen | 3.09 ± 0.41 | 3.82 ± 0.53 | 28.46 ± 1.13 | 2.09 ± 0.25 | 1.94 ± 0.51 | 0.69 ± 0.61 | 1.56 ± 0.71 | 1.7 ± 0.51 | 1.2 ± 0.22 | - | 4.09 ± 0.58 | 8.55 ± 0.94 | 1.08 ± 0.75 |
| 1033 | Eucalyptol (1,8-cineole ) | 24.73 ± 1.23 | 25.17 ± 1.12 | 25.03 ± 1.11 | 30.4 ± 1.34 | 32.26 ± 1.65 | 23.55 ± 1.72 | 4.17 ± 0.48 | 46.02 ± 0.28 | 45.4 ± 1.20 | 3.61 ± 0.63 | 13.99 ± 1.52 | 3.27 ± 0.28 | 2.89 ± 0.53 |
| 1060 | γ-terpinene | - | 0.18 ± 0.51 | - | - | - | - | - | - | - | - | - | 0.51 ± 0.15 | |
| 1068 | trans-sabinene hydrate | 0.29 ± 0.22 | 0.33 ± 0.62 | - | - | - | - | - | - | - | - | - | 1.03 ± 0.64 | 0.51 ± 0.71 |
| 1100 | linalool | 0.26 ± 0.14 | 0.09 ± 0.03 | - | 0.19 ± 0.14 | 0.11 ± 0.12 | 3.095 ± 0.45 | 10.32 ± 0.56 | 0.08 ± 0.02 | 0.05 ± 0.08 | - | 1.31 ± 0.56 | 1.03 ± 0.47 | - |
| 1133 | α-thujone | - | - | - | 4.02 ± 0.71 | - | - | - | 0.1 ± 0.23 | 0.2 ± 0.31 | 3.75 ± 0.85 | - | - | - |
| 1139 | trans-pinocarveol | 0.36 ± 0.13 | 0.35 ± 0.26 | - | 5.1 ± 0.86 | 4.12 ± 0.28 | - | - | 0.28 ± 0.15 | 0.4 ± 0.25 | 1.36 ± 0.68 | 1.58 ± 0.47 | - | 0.95 ± 0.56 |
| 1143 | camphor | 0.45 ± 0.28 | - | 5.09 ± 0.72 | 5.59 ± 0.66 | - | 1.6 ± 0.19 | 5.86 ± 0.35 | 4.58 ± 1.45 | 2.94 ± 0.58 | 1.5 ± 0.34 | - | 0.97 ± 0.62 | |
| 1165 | borneol | 1.42 ± 0.62 | 0.24 ± 0.18 | - | 4.17 ± 0.56 | 2.81 ± 0.18 | 2.13 ± 0.81 | - | 0.47 ± 0.12 | 0.6 ± 0.47 | 1.47 ± 0.61 | 4 ± 0.91 | 3.22 ± 0.87 | 3.43 ± 0.48 |
| 1257 | linalyl acetate | 0.62 ± 0.33 | 0.34 ± 0.33 | 0.3 ± 0.52 | 0.3 ± 0.18 | - | 10.0 ± 0.90 | 26.4 ± 0.82 | 0.1 ± 0.14 | 0.5 ± 0.21 | - | - | 0.8 ± 0.11 | 2.5 ± 0.63 |
| 1275 | α-fenchyl acetate | 0.71 ± 0.16 | 0.39 ± 0.41 | 2.32 ± 0.84 | - | - | - | - | 0.05 ± 0.01 | 0.09 ± 0.04 | - | 1.39 ± 0.25 | 2.53 ± 0.42 | 0.63 ± 0.33 |
| 1285 | bornyl acetate | 0.39 ± 0.32 | - | 0.2 ± 0.13 | 0.5 ± 0.43 | 0.26 ± 0.31 | 0.85 ± 0.15 | 7.75 ± 0.55 | 0.08 ± 0.02 | 0.06 ± 0.03 | 3.16 ± 0.17 | 2.87 ± 0.61 | 5.12 ± 0.71 | 2.97 ± 0.82 |
| 1420 | trans-caryophyllene | - | - | 0.19 ± 0.61 | 0.31 ± 0.87 | 0.33 ± 0.42 | - | - | - | - | - | - | - | - |
| 1465 | α-humulene | - | - | 0.12 ± 0.27 | 0.17 ± 0.36 | 0.19 ± 0.23 | 0.65 ± 0.37 | 2.63 ± 0.42 | 0.05 ± 0.01 | 0.04 ± 0.03 | 0.9 ± 0.05 | 1.13 ± 0.15 | 1.79 ± 0.64 | 0.66 ± 0.28 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tulukcu, E.; Cebi, N.; Sagdic, O. Chemical Fingerprinting of Seeds of Some Salvia Species in Turkey by Using GC-MS and FTIR. Foods 2019, 8, 118. https://doi.org/10.3390/foods8040118
Tulukcu E, Cebi N, Sagdic O. Chemical Fingerprinting of Seeds of Some Salvia Species in Turkey by Using GC-MS and FTIR. Foods. 2019; 8(4):118. https://doi.org/10.3390/foods8040118
Chicago/Turabian StyleTulukcu, Eray, Nur Cebi, and Osman Sagdic. 2019. "Chemical Fingerprinting of Seeds of Some Salvia Species in Turkey by Using GC-MS and FTIR" Foods 8, no. 4: 118. https://doi.org/10.3390/foods8040118
APA StyleTulukcu, E., Cebi, N., & Sagdic, O. (2019). Chemical Fingerprinting of Seeds of Some Salvia Species in Turkey by Using GC-MS and FTIR. Foods, 8(4), 118. https://doi.org/10.3390/foods8040118