Rapid Evaluation Methods for Quality of Trout (Oncorhynchus mykiss) Fresh Fillet Preserved in an Active Edible Coating
Abstract
1. Introduction
2. Materials and Methods
2.1. Trout Fillets Preparation
2.2. Coating-Forming Solutions Preparation and Treatments
2.3. Peroxide Value Measurement
2.4. Thiobarbituric Acid Reactive Substances (TBARS) Determination
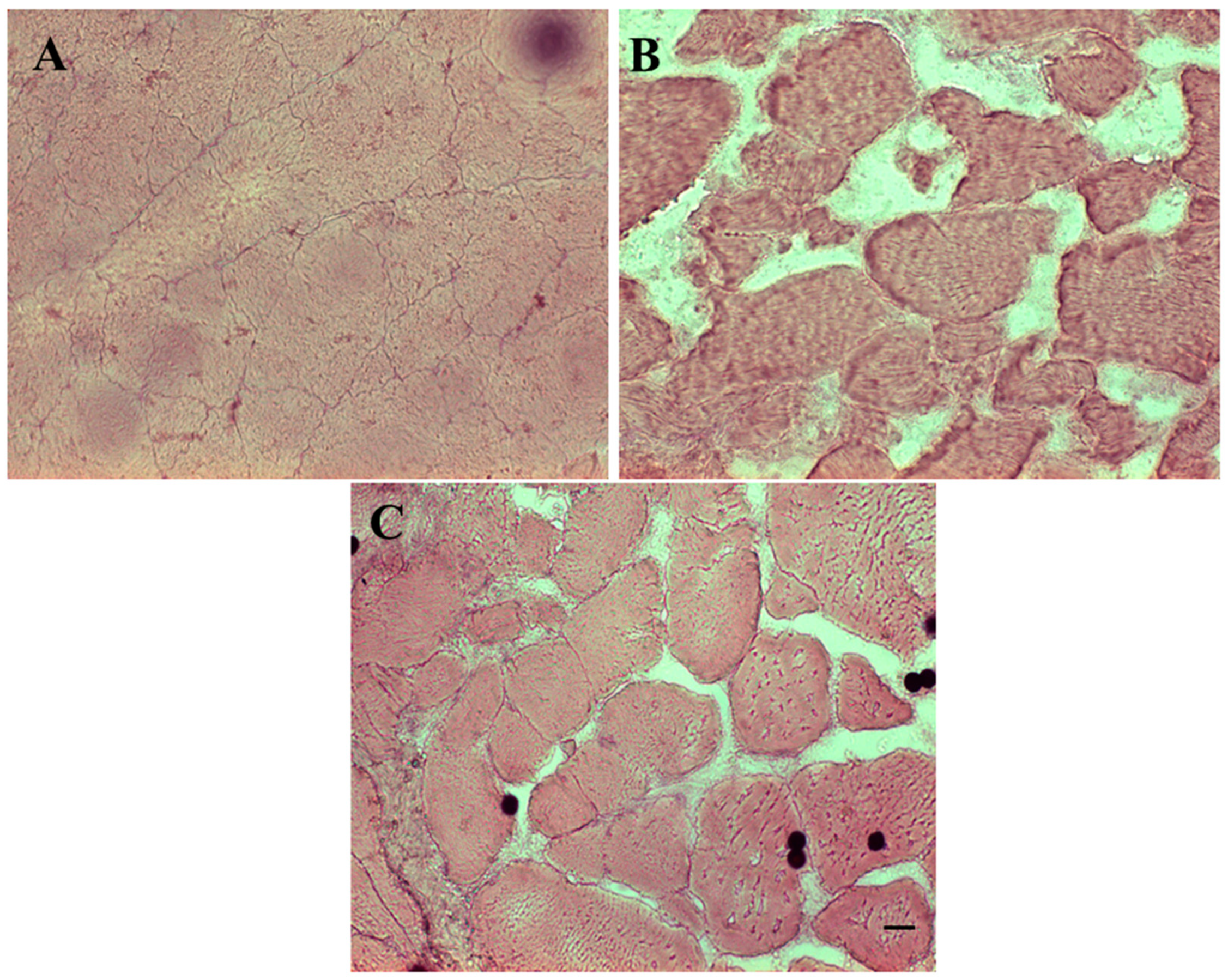
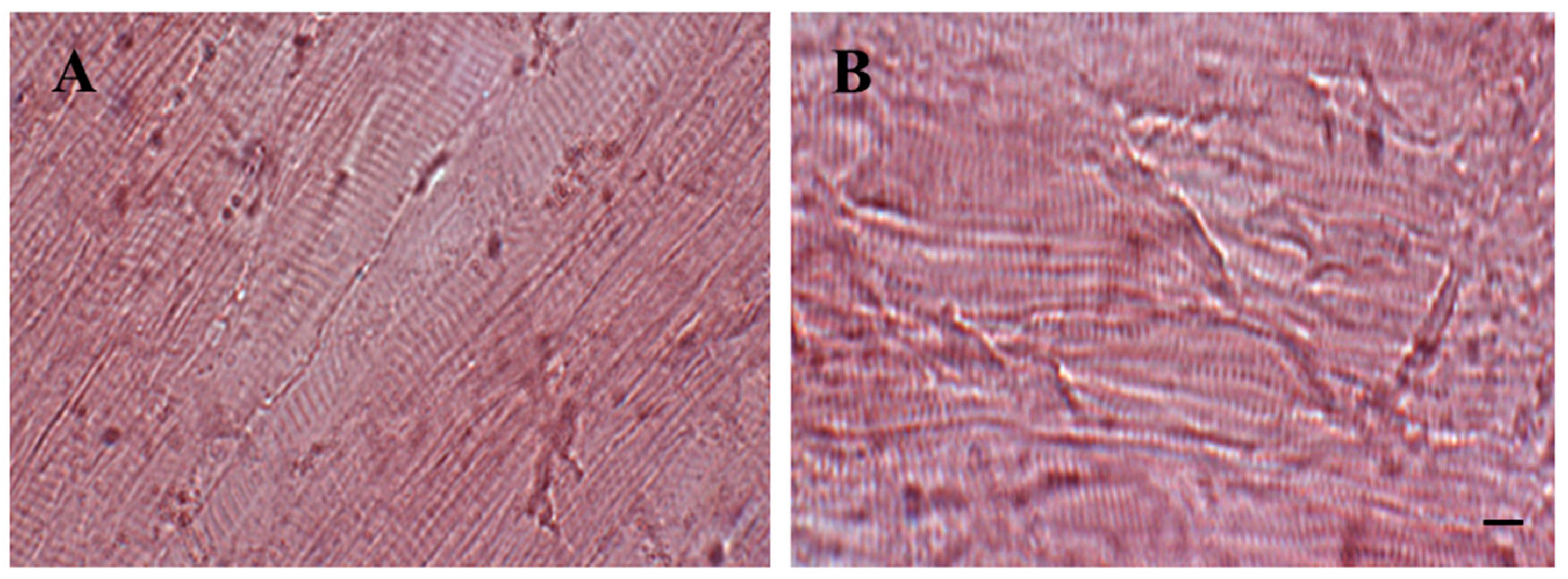
2.5. Histology
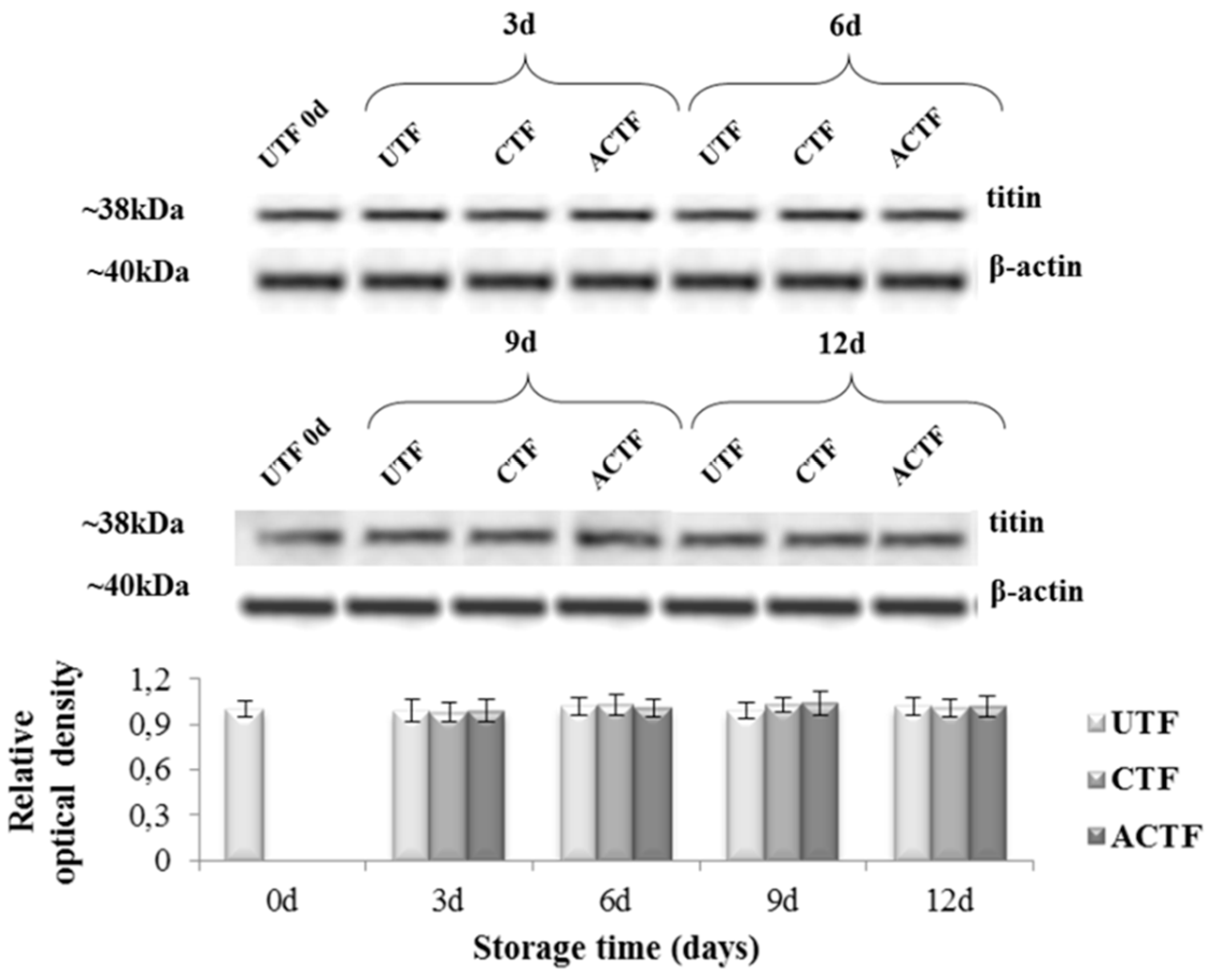
2.6. Electrophoresis and Western Blot Analysis
2.7. Electronic Nose (EN) Analysis
2.8. ATR-FTIR Spectroscopy
2.9. Statistical Analysis
3. Results and Discussion
3.1. Lipid Peroxidation
3.2. Histological and Western Blot Analysis
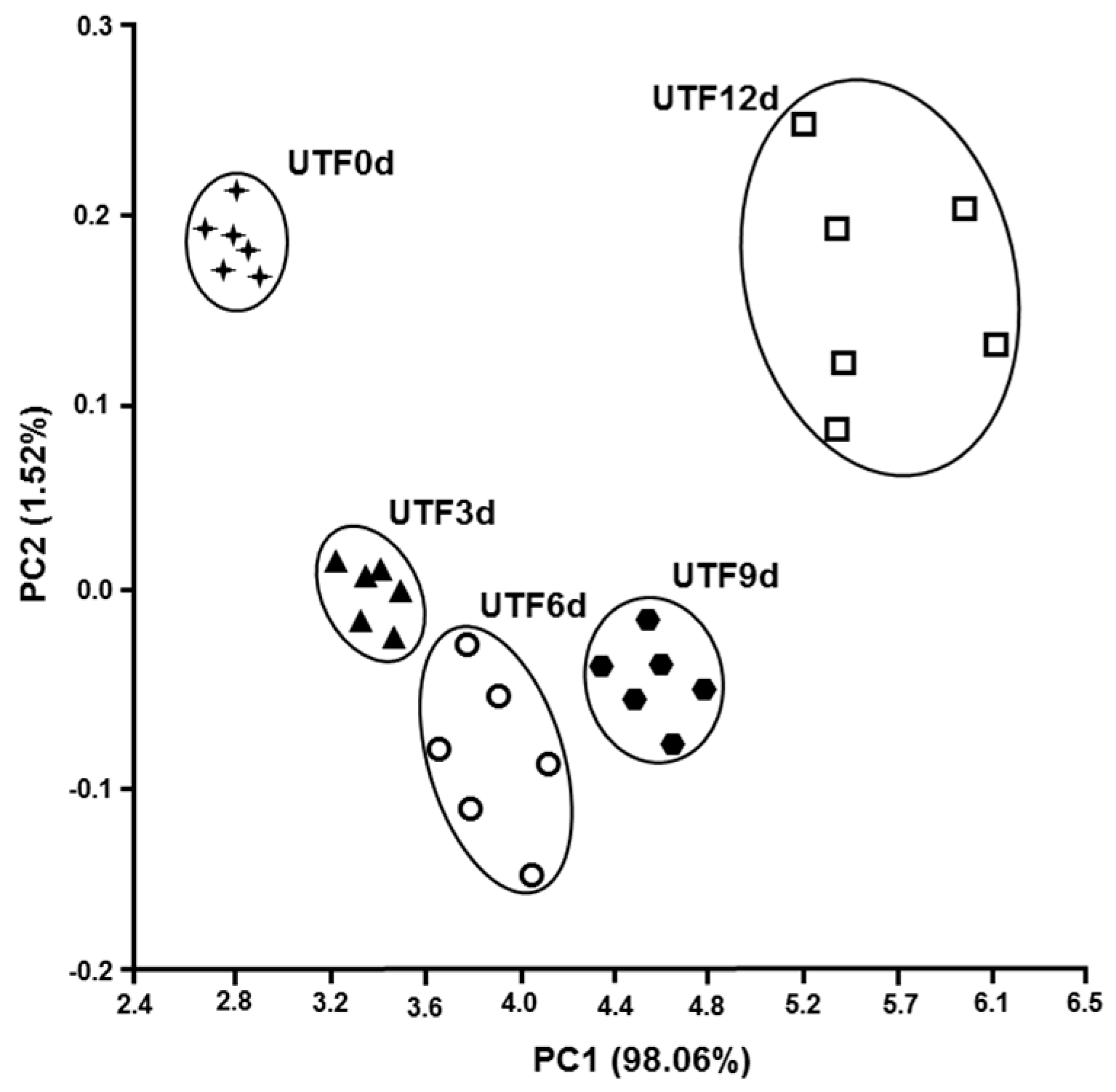
3.3. Olfactory Analysis
3.4. ATR-FTIR Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ahmad, M.; Benjakul, S.; Sumpavapol, P.; Nirmal, N.P. Quality changes of sea bass slices wrapped with gelatin film incorporated with lemongrass essential oil. Int. J. Food Microbiol. 2012, 155, 171–178. [Google Scholar] [CrossRef]
- Ul Hasan, N.; Ejaz, N.; Ejaz, W.; Kim, H.S. Meat and fish freshness inspection system based on odor sensing. Sensors 2012, 12, 1542–1557. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, X.; Liu, Y.; Xu, X.; Zhou, G. Species discrimination among three kinds of puffer fish using an electronic nose combined with olfactory sensory evaluation. Sensors 2012, 12, 12562–12571. [Google Scholar] [CrossRef]
- Perez-Alonso, F.; Arias, C.; Aubourg, S.P. Lipid deterioration during chilled storage of Atlantic pomfret (Brama brama). Eur. J. Lipid Sci. Tech. 2003, 105, 661–667. [Google Scholar] [CrossRef]
- Ando, M.; Toyohara, H.; Shimizu, Y.; Sakaguchi, M. Post-mortem tenderization of fish muscle proceeds independently of resolution of rigor mortis. Nippon Suisan Gakk. 1991, 57, 1165–1169. [Google Scholar] [CrossRef]
- Steen, D.; Claeys, E.; Uytterhaegen, L.; De Smet, S.; Demeyer, D. Early post-mortem conditions and the calpain/calpastatin system in relation to tenderness of double-muscled beef. Meat Sci. 1997, 45, 307–319. [Google Scholar] [CrossRef]
- Taylor, R.; Fjaera, S.; Skjervold, P. Salmon fillet texture is determined by myofiber-myofiber and myofiber-myocommata attachment. J. Food Sci. 2002, 67, 2067–2071. [Google Scholar] [CrossRef]
- Busconi, L.; Folco, E.J.; Martone, CB.; Sanchez, J.J. Postmortem changes in cytoskeletal elements of fish muscle. J. Food Biochem. 1989, 13, 443–451. [Google Scholar] [CrossRef]
- Ofstad, R.; Egelendsdal, B.; Kidman, S.; Myklebust, R.; Olsen, R.L.; Hermansson, A.M. Liquid loss as effected by post mortem ultrastructural changes in fish muscle; cod (Gadus morhua L.) and salmon (Salmo salar). J. Sci. Food Agric. 1996, 71, 301–312. [Google Scholar] [CrossRef]
- Seki, N.; Watanabe, T. Connectin content and its port-mortem changes in fish muscle. J. Biochem. 1984, 95, 1161–1167. [Google Scholar] [CrossRef]
- Astier, C.; Labbe, J.P.; Roustan, C.; Benyamin, Y. Sarcomeric disorganization in post-mortem fish muscles. Comp. Biochem. Physiol. B 1991, 100, 459–465. [Google Scholar] [CrossRef]
- Papa, I.; Taylor, R.G.; Astier, C.; Ventre, F.; Lebart, M.C.; Roustan, C.; Ouali, A.; Benyamin, Y. Dystrophin cleavage and sarcolemma detachment are early post mortem changes on bass (Dicentrarchus labrax) white muscle. J. Food Sci. 1997, 62, 917–921. [Google Scholar] [CrossRef]
- Tsuchiya, H.; Seki, N. Action of calpain on α-actinin within and isolated from carp myofibrils. Nippon Suisan Gakk. 1991, 57, 1133–1139. [Google Scholar] [CrossRef]
- Chéret, R.; Delbarre-Ladrat, C.; Verrez-Bagnis, V.; De Lamballerie, M. Effect of high pressure on the calpain-calpastatin system in fish muscle. J. Food Sci. 2007, 72, 313–316. [Google Scholar] [CrossRef]
- Reddy, N.R.; Villanueva, M.; Kautter, D.A. Shelf life of modified-atmosphere-packaged fresh tilapia fillets stored under refrigeration and temperature-abuse conditions. J. Food Prot. 1995, 58, 908–914. [Google Scholar] [CrossRef]
- Velu, S.; Abu Bakar, F.; Mahyudin, N.A.; Saari, N.; Zaman, M.Z. Effect of modified atmosphere packaging on microbial flora changes in fishery products. Int. Food Res. J. 2013, 20, 17–26. [Google Scholar]
- Cyprian, O.; Lauzon, H.L.; Jóhannsson, R.; Sveinsdóttir, K.; Arason, S.; Martinsdóttir, E. Shelf life of air and modified atmosphere-packaged fresh tilapia (Oreochromis niloticus) fillets stored under chilled and superchilled conditions. Food Sci. Nutr. 2013, 1, 130–140. [Google Scholar] [CrossRef]
- Volpe, M.G.; Maliconico, M.; Varricchio, E.; Paolucci, M. Polysaccharides as Biopolymers for Food Shelf-Life Extention: Recent Pat. Food Nutr. Agric. 2010, 2, 129–139. [Google Scholar]
- Volpe, M.G.; Siano, F.; Paolucci, M.; Sacco, A.; Sorrentino, A.; Malinconico, M.; Varricchio, E. Active edible coating effectiveness in shelf-life enhancement of trout (Oncorhynchus mykiss) fillets. LWT-Food Sci. Technol. 2015, 60, 615–622. [Google Scholar] [CrossRef]
- Dehghani, S.; Hosseini, S.V.; Regenstein, J.M. Edible films and coatings in seafood preservation: A review. Food Chem. 2018, 240, 505–513. [Google Scholar] [CrossRef]
- Socaciu, M.I.; Semeniuc, C.A.; Vodnar, D.C. Edible Films and Coatings for Fresh Fish Packaging: Focus on Quality Changes and Shelf-life Extension. Coatings 2018, 8, 366. [Google Scholar] [CrossRef]
- Borski, R.J.; Hodson, R.G. Fish Research and the Institutional Animal Care and Use Committee. Ilar J. 2003, 44, 286–294. [Google Scholar] [CrossRef]
- Meer, W. Agar. In Handbook of Water-soluble Gums and Resins; Davidson, R.L., Ed.; McGraw-Hill: New York, NY, USA, 1980; pp. 7.2–7.14. [Google Scholar]
- Erkan, N.; Tosun, S.Y.; Ulusoy, S.; Üretener, G. The use of thyme and laurel essential oil treatments to extend the shelf life of blue fish (Pomatomus saltatrix) during storage in ice. J. Verbr. Lebensm. 2011, 6, 39–48. [Google Scholar] [CrossRef]
- Thiansilakul, Y.; Benjakul, S.; Richards, M.P. Effect of myoglobin from Eastern little tuna muscle on lipid oxidation of washed Asian seabass mince at different pH conditions. J. Food Sci. 2011, 76, 242–249. [Google Scholar] [CrossRef]
- Laurienzo, P.; Cammarota, G.; Di Stasio, M.; Gentile, G.; Laurino, C.; Volpe, M.G. Microstructure and olfactory quality of apples de-hydrated by innovative technologies. J. Food Eng. 2013, 116, 689–694. [Google Scholar] [CrossRef]
- Volpe, M.G.; De Cunzo, F.; Siano, F.; Paolucci, M.; Barbarisi, C.; Cammarota, G. Influence of extraction techniques on physical-chemical characteristics and volatile compounds of extra virgin olive oil. J. Oleo Sci. 2014, 63, 875–883. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Kamil, J.Y.V.A.; Shahaidi, F. Chitosan as an edible invisible film for quality preservation of herring and atlantic cod. J. Agric. Food Chem. 2002, 50, 5167–5178. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Mohamad, S.; Hosseini, H. Effect of chitosan coatings enriched with cinnamon oil on the quality of refrigerated rainbow trout. Food Chem. 2010, 120, 193–198. [Google Scholar] [CrossRef]
- Benjakul, S.; Visessanguan, W.; Phongkanpai, V.; Tanaka, M. Antioxidative activity of caramelisation products and their preventive effect on lipid oxidation in fish mince. Food Chem. 2005, 90, 231–239. [Google Scholar] [CrossRef]
- Perumalla, A.V.S.; Hettiarachchy, N.S. Green tea and grape seed extracts—Potential applications in food safety and quality. Food Res. Int. 2011, 44, 827–839. [Google Scholar] [CrossRef]
- Giménez, B.; Roncalés, P.; Beltran, J.A. The effects of natural antioxidants and lighting conditions on the quality characteristics of gilthead sea bream fillets (Sparus aurata) packaged in a modified atmosphere. J. Sci. Food Agric. 2004, 84, 1052–1060. [Google Scholar] [CrossRef]
- Giménez, B.; Roncalés, P.; Beltran, J.A. The effects of natural antioxidants and lighting conditions on the quality characteristics of salmon (Salmo salar) packaged in modified atmosphere. J. Sci. Food Agric. 2005, 85, 1033–1040. [Google Scholar] [CrossRef]
- Pereira de Abreu, D.; Paseiro-Losada, P.; Maroto, J.; Cruz, J.M. Evaluation of the effectiveness of a new active packaging film containing natural antioxidants from barley husks that retard lipid damage in frozen Atlantic salmon (Salmo salar). Food Res. Int. 2010, 43, 1277–1282. [Google Scholar] [CrossRef]
- Gómez-Estaca, J.; Montero, P.; Giménez, B.; Gómez-Guillén, M.C. Effect of functional edible films and high pressure processing on microbial and oxidative spoilage in cold-smoked sardine (Sardina pilchardus). Food Chem. 2007, 105, 511–520. [Google Scholar] [CrossRef]
- Bremner, H.A. Fish flesh structure and the role of collagen—Its post-mortem aspects and implications for fish processing. In Quality Assurance in the Fish Industry; Huss, H.H., Jakobsen, M., Liston, J., Eds.; Elsevier Science: Amsterdam, The Netherlands, 1992; pp. 39–62. [Google Scholar]
- Sato, K.; Ando, M.; Kubota, S.; Origasa, K.; Kawase, H.; Toyohara, H.; Sakaguchi, M.; Nakagawa, T.; Makinodan, Y.; Ohtsuki, K.; et al. Involvement of type V collagen in softening of fish muscle during short-term chilled storage. J. Agric. Food Chem. 1997, 45, 343–348. [Google Scholar] [CrossRef]
- Ando, M.; Toyohara, H.; Sakaguchi, M. Post-mortem tenderization of rainbow trout muscle caused by disintegration of collagen fibers in the pericellular connective tissue. Bull. Jpn. Soc. Sci. Fish. 1992, 58, 567–570. [Google Scholar] [CrossRef]
- Horowits, R.; Podolsky, R.J. The positional stability of thick filaments in activated skeletal muscle depends on sarcomere length: Evidence for the role of titin filaments. J. Cell. Biol. 1987, 105, 2217–2223. [Google Scholar] [CrossRef]
- Hernández-Herrero, M.M.; Duflos, G.; Malle, P.; Bouqueletc, S. Collagenase activity and protein hydrolysis as related tospoilage of iced cod (Gadus morhua). Food Res. Int. 2003, 36, 141–147. [Google Scholar] [CrossRef]
- Bonilla, J.; Atares, L.; Vargas, M.; Chiralt, A. Edible films and coatings to prevent the detrimental effect of oxygen on food quality: Possibilities and limitations. J. Food Engin. 2012, 110, 208–213. [Google Scholar] [CrossRef]
- Meyer, R.C.; Winter, A.R.; Weiser, H.H. Edible Protective Coatings for Extending the Shelf Life of Poultry. Food Technol. 1959, 13, 146–148. [Google Scholar]
- Delbarre-Ladrat, C.; Chéret, R.; Taylor, R.; Verrez-Bagnis, V. Trends in postmortem aging in fish: Understanding of proteolysis and disorganization of the myofibrillar structure. Crit. Rev. Food Sci. 2006, 46, 409–421. [Google Scholar] [CrossRef]
- Falasconi, M.; Concina, I.; Gobbi, E.; Sberveglieri, V.; Pulvirenti, A.; Sberveglieri, G. Electronic nose for microbiological quality control of food products. Int. J. Electrochem. 2012. Article ID 715763. [Google Scholar] [CrossRef]
- Persaud, K.; Dodd, G. Analysis of discrimination mechanisms in the mammalian olfactory system using a model nose. Nature 1982, 299, 352–355. [Google Scholar] [CrossRef]
- Wilson, A.D.; Baietto, M. Applications and advances in electronic-nose technologies. Sensors 2009, 9, 5099–5148. [Google Scholar] [CrossRef]
- Capelli, L.; Sironi, S.; Del Rosso, R. Electronic noses for environmental monitoring applications. Sensors 2014, 14, 19979–20007. [Google Scholar] [CrossRef]
- Di Natale, C.; Davide, F.A.M.; D’Amico, A.; Sberveglieri, G.; Nelli, P.; Faglia, G.; Perego, C. Complex chemical pattern recognition with sensor array: The discrimination of vintage years of wine. Sens. Actuat. 1995, 25, 801–804. [Google Scholar] [CrossRef]
- Olafsdottir, G.; Chanie, E.; Westad, F.; Jonsdottir, R.; Thalmann, C.R.; Bazzo, S.; Labreche, S.; Marcq, P.; Lunby, F.; Haugen, J.E. Prediction of microbial and sensory quality of cold smoked Atlantic salmon (Salmo salar) by electronic nose. J. Food Sci. 2005, 70, S563–S574. [Google Scholar] [CrossRef]
- Baker, M.J.; Trevisan, J.; Bassan, P.; Bhargava, R.; Butler, H.J.; Dorling, K.M.; Fielden, P.M.; Fogarty, S.W. Using Fourier transform IR spectroscopy to analyze biological materials. Nat. Protoc. 2014, 9, 1771–1791. [Google Scholar] [CrossRef]
- Tasbozan, O.; Emre, Y.; Gokce, M.A.; Erbas, C.; Ozcan, F.; Kıvrak, E. The effects of different cycles of starvation and re-feeding on growth and body composition in rainbow trout (Oncorhynchus mykiss, Walbaum, 1792). J. Appl. Ichthyol. 2016, 32, 583–588. [Google Scholar] [CrossRef]
- Malek, K.; Wood, B.R.; Bambery, K.R. FTIR Imaging of Tissues: Techniques and Methods of Analysis. In Optical Spectroscopy and Computational Methods in Biology and Medicine, Challenges and Advances in Computational Chemistry and Physics; Baranska, M., Ed.; Springer Science+Business Media Dordrecht: Berlin, Germany, 2014; Volume 14. [Google Scholar] [CrossRef]
- Movasaghi, Z.; Rehman, S.; ur Rehman, I. Fourier Transform Infrared (FTIR) Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- De Campos Vidal, B.; Mello, M.L.S. Collagen type I amide I band infrared spectroscopy. Micron 2011, 42, 283–289. [Google Scholar] [CrossRef]
- Oleszko, A.; Nska-Janus, S.O.; Walski, T.; Grzeszczuk-KuT, K.; Bujok, J.; GaBecka, K.; Czerski, A.; Witkiewicz, W.; MaBgorzata, K. Application of FTIR-ATR Spectroscopy to Determine the Extent of Lipid Peroxidation in Plasma during Haemodialysis. BioMed. Res. Int. 2015, 245607, 8. [Google Scholar] [CrossRef] [PubMed]
- Botta, S.B.; Ana, P.A.; Santos, M.O.; Zezell, D.M.; Matos, A.B. Effect of dental tissue conditioners and matrix metalloproteinase inhibitors on type I collagen microstructure analyzed by Fourier transform infrared spectroscopy. J. Biomed. Mater. Res. Part. B 2012, 100B, 1009–1016. [Google Scholar] [CrossRef]
- Van de Voort, F.; Ismail, A.; Sedman, J.; Emo, G. Monitoring the oxidation of edible oils by FTIR spectroscopy. J. Am. Oil Chem. Soc. 1994, 71, 243–253. [Google Scholar] [CrossRef]
- Guillenn, M.D.; Cabo, N. Usefulness of the frequency data of the Fourier transform infrared spectra to evaluate the degree of oxidation of edible oils. J. Agric. Food Chem. 1999, 47, 709–719. [Google Scholar] [CrossRef]
- Fuchs, B.; Bresler, K.; Schiller, J. Oxidative changes of lipids monitored byMALDI MS. Chem. Phys. Lipids 2011, 164, 782–795. [Google Scholar] [CrossRef] [PubMed]
- Yue, B. Biology of the Extracellular Matrix: An Overview. J. Glaucoma 2014, S20–S23. [Google Scholar] [CrossRef]
- Muyonga, J.H.; Cole, C.G.B.; Duodu, K.G. Fourier transform infrared (FTIR) spectroscopic study of acid soluble collagen and gelatin from skins and bones of young and adult Nile perch (Lates niloticus). Food Chem. 2004, 86, 325–332. [Google Scholar] [CrossRef]
- Byler, D.M.; Susi, H. Examination of the Secondary Structure of Proteins by Deconvolved FTIR Spectra. Biopolymers 1986, 25, 469–487. [Google Scholar] [CrossRef]
- Haris, P.I.; Severcan, F. FTIR spectroscopic characterization of protein structure in aqueous and non-aqueous media. J. Mol. Cat. B-Enzymc. 1999, 7, 207–221. [Google Scholar] [CrossRef]
- Yang, H.; Yang, S.; Kong, J.; Dong, A.; Yu, S. Obtaining information about protein secondary structures in aqueous solution using Fourier transform IR spectroscopy. Nat. Protoc. 2015, 10, 382–396. [Google Scholar] [CrossRef]
- Kumosinski, T.F.; Farrell, H.M., Jr. Determination of the global secondary structure of proteins by Fourier transform infrared (FTIR) spectroscopy. Trends Food Sci. Technol. 1993, 4, 169–175. [Google Scholar] [CrossRef]
- Ogawa, M.; Nakamura, S.; Horimoto, Y.; An, H.; Tsuchiya, T.; Nakai, S. Raman Spectroscopic Study of Changes in Fish Actomyosin during Setting. J. Agric. Food Chem. 1999, 47, 3309–3318. [Google Scholar] [CrossRef]
- Moosavi-Nasab, M.; Alli, I.; Ismail, A.A.; Ngadi, M.O. Protein Structural Changes During Preparation and Storage of Surimi. J. Food Sci. 2005, 70, 448–453. [Google Scholar] [CrossRef]
- Wei, W.; Hu, W.; Zhang, X.-Y.; Zhang, F.-P.; Sun, S.-Q.; Liu, Y.; Xu, C.-H. Analysis of protein structure changes and quality regulation of surimi during gelation based on infrared spectroscopy and microscopic imaging. Sci. Rep. 2018, 8, 5566. [Google Scholar] [CrossRef]
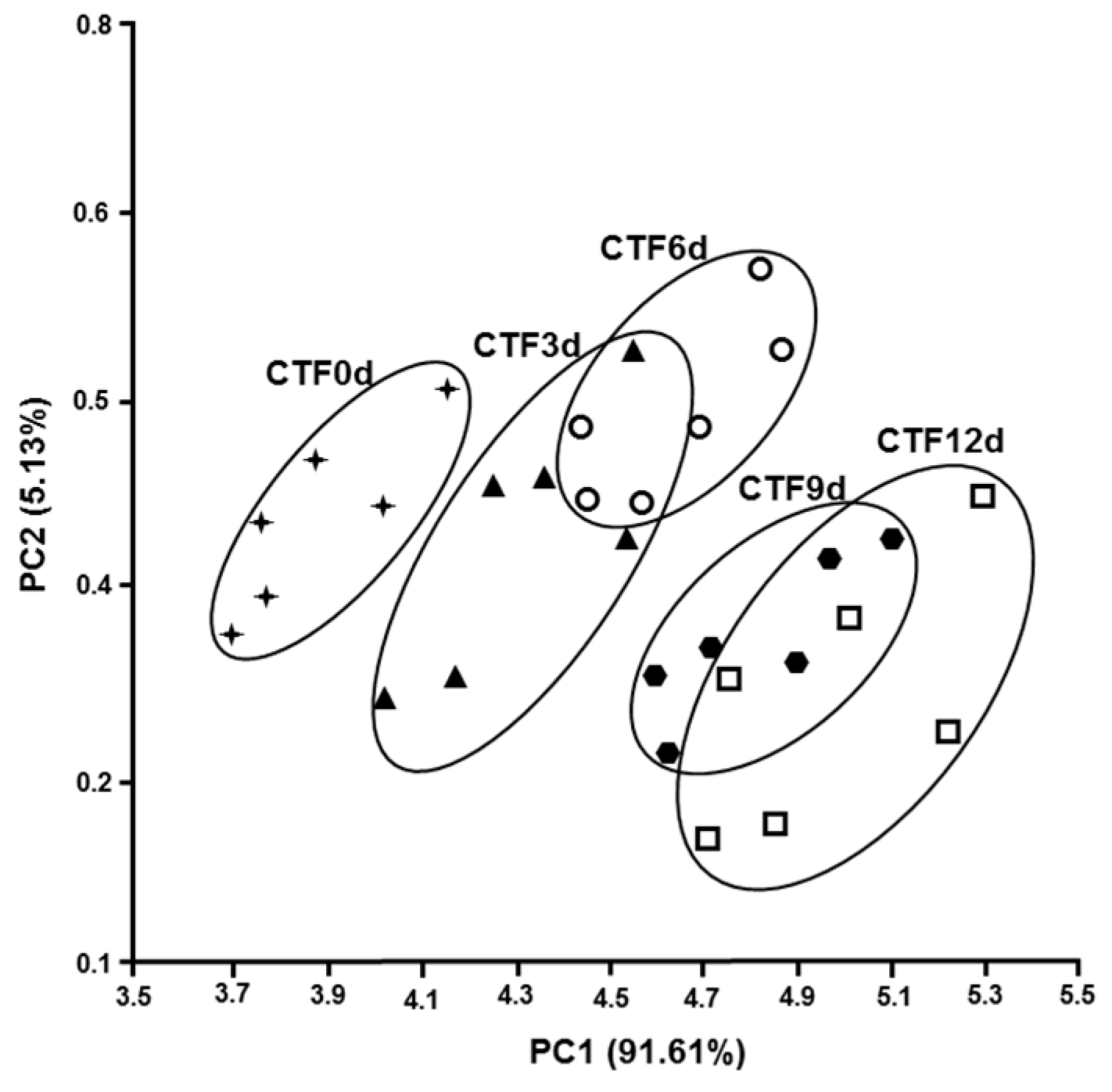
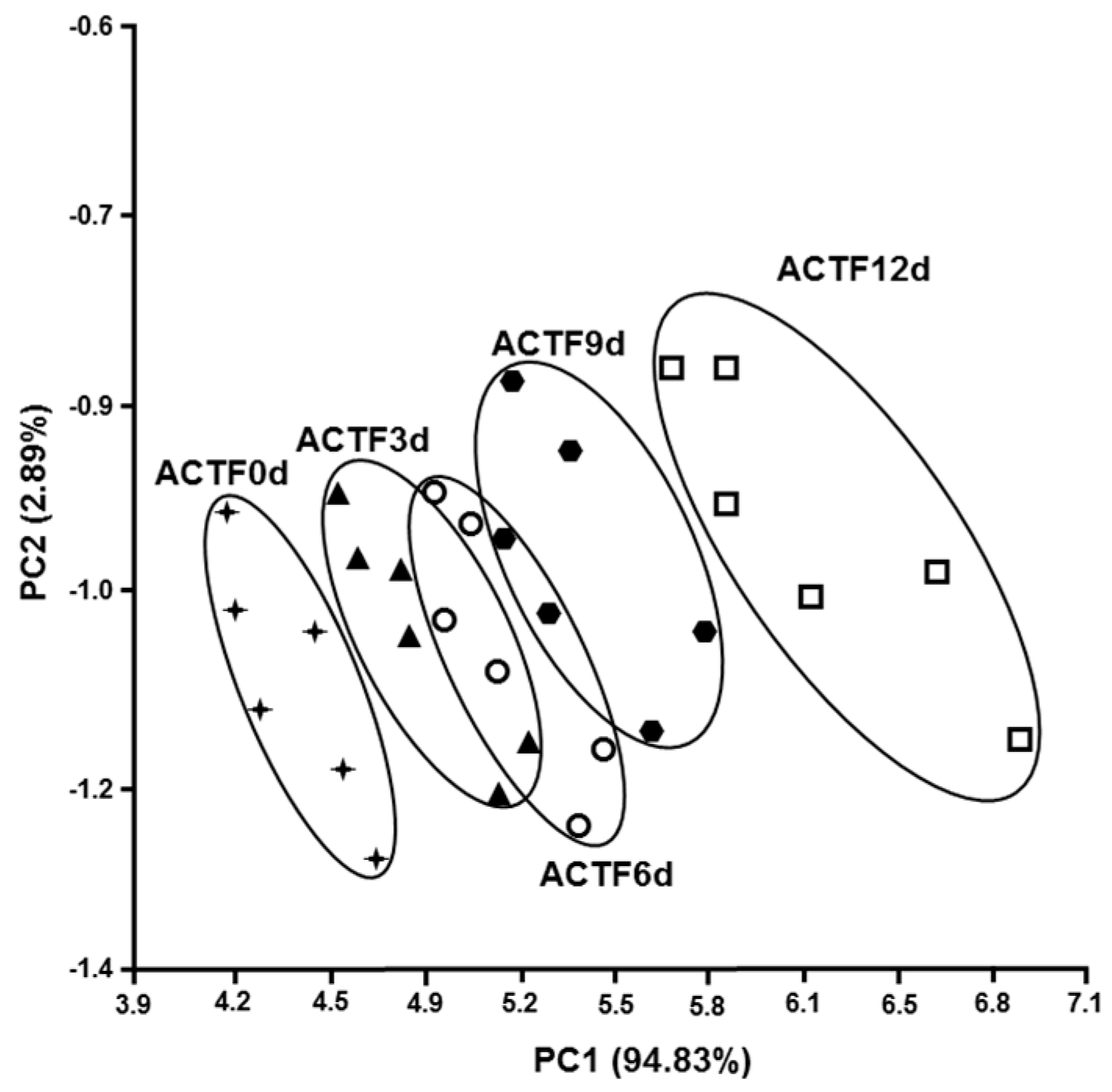
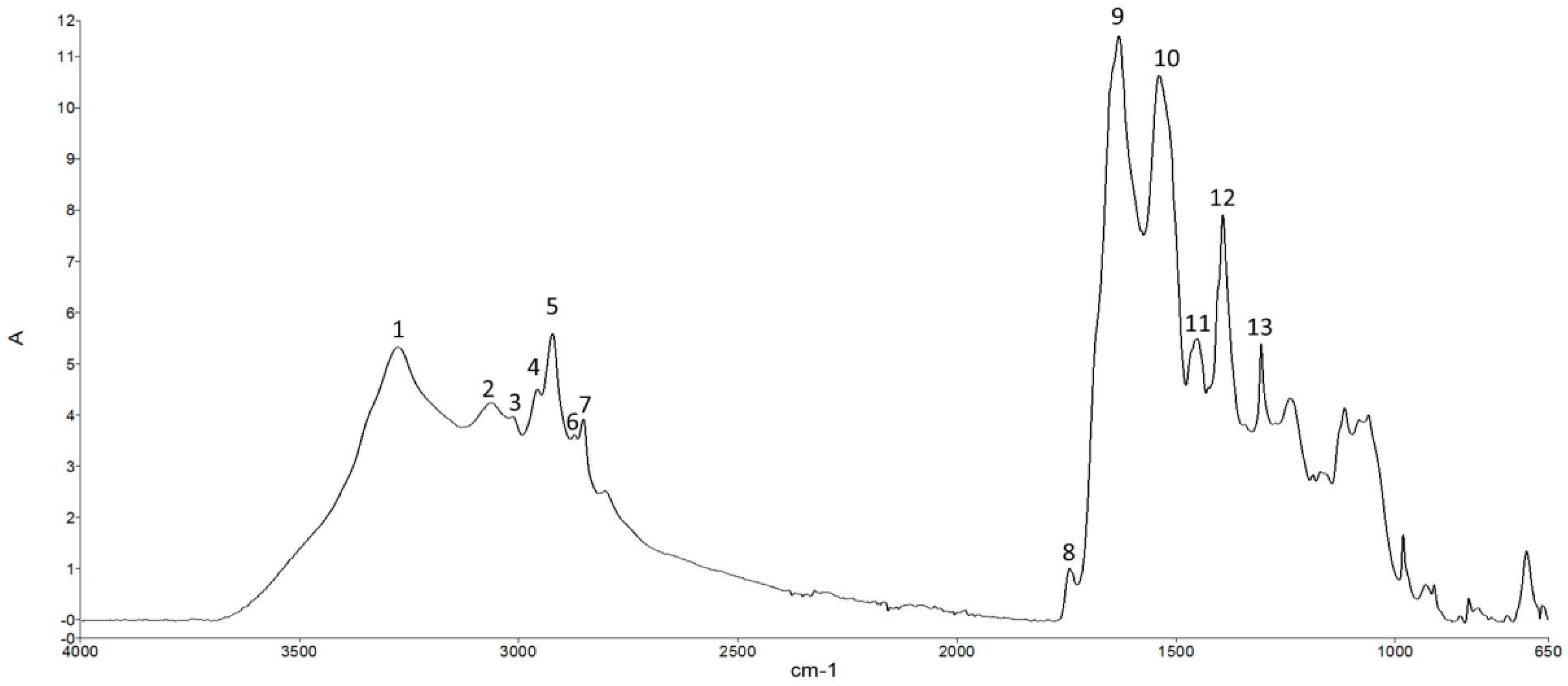
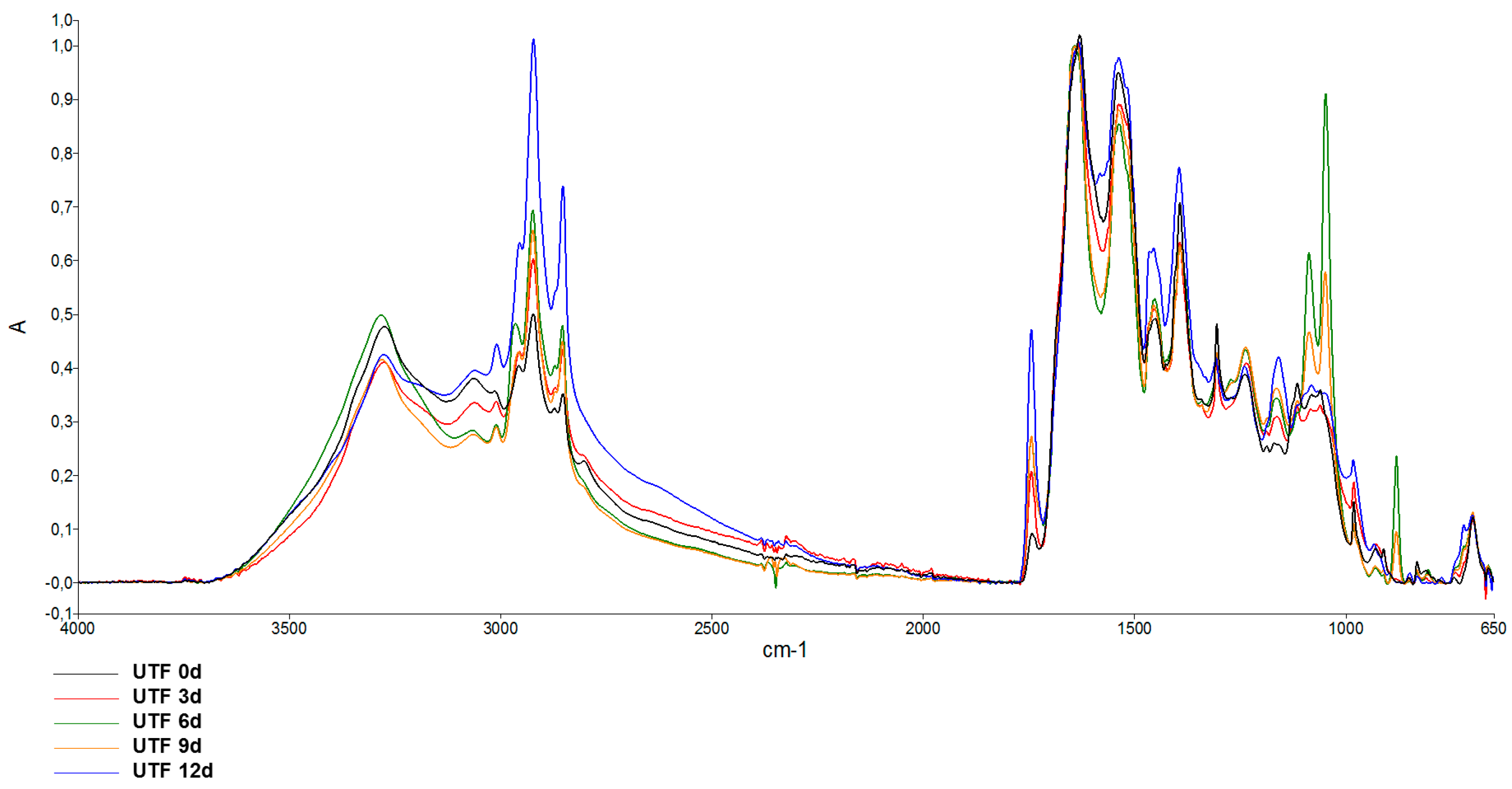
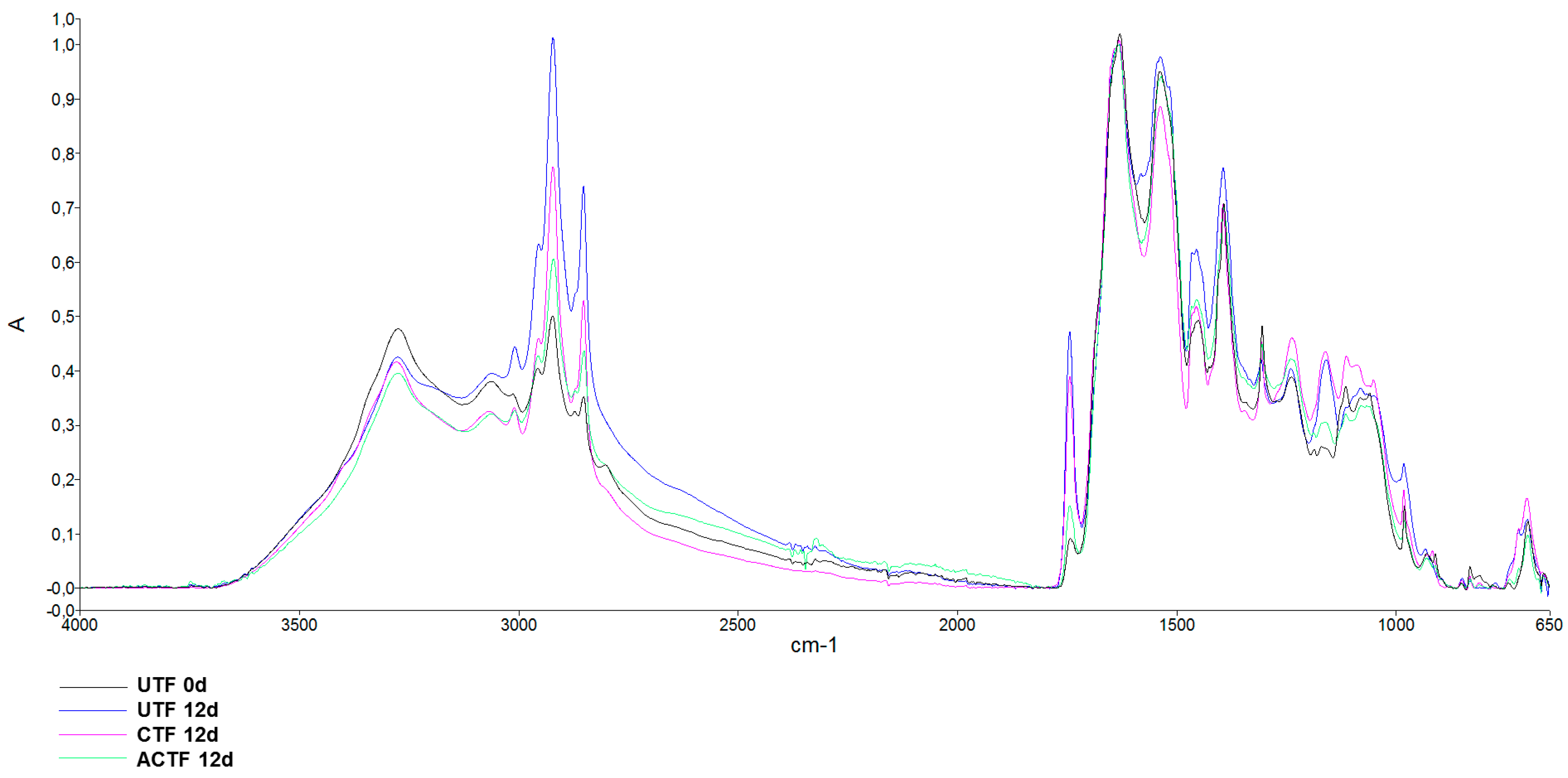
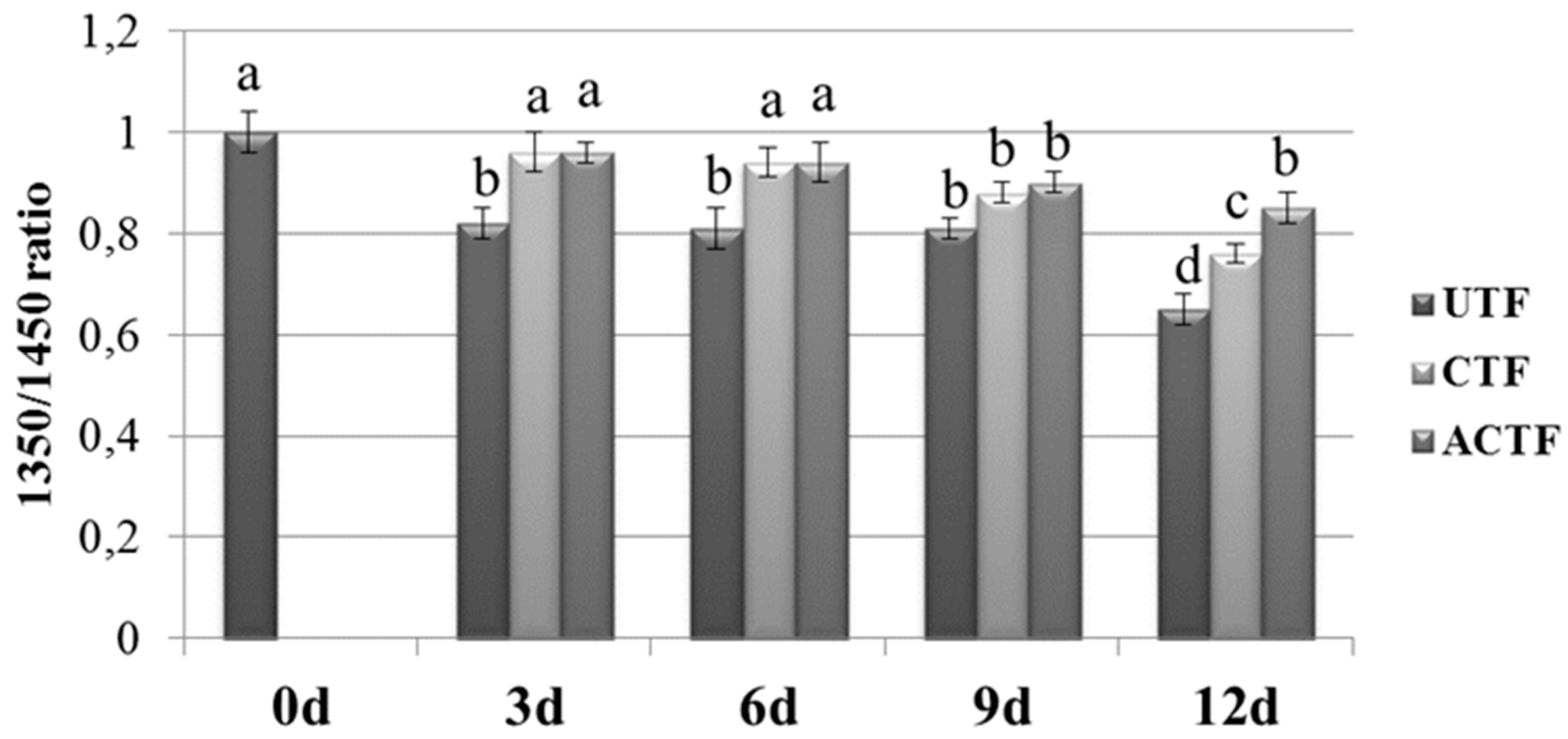
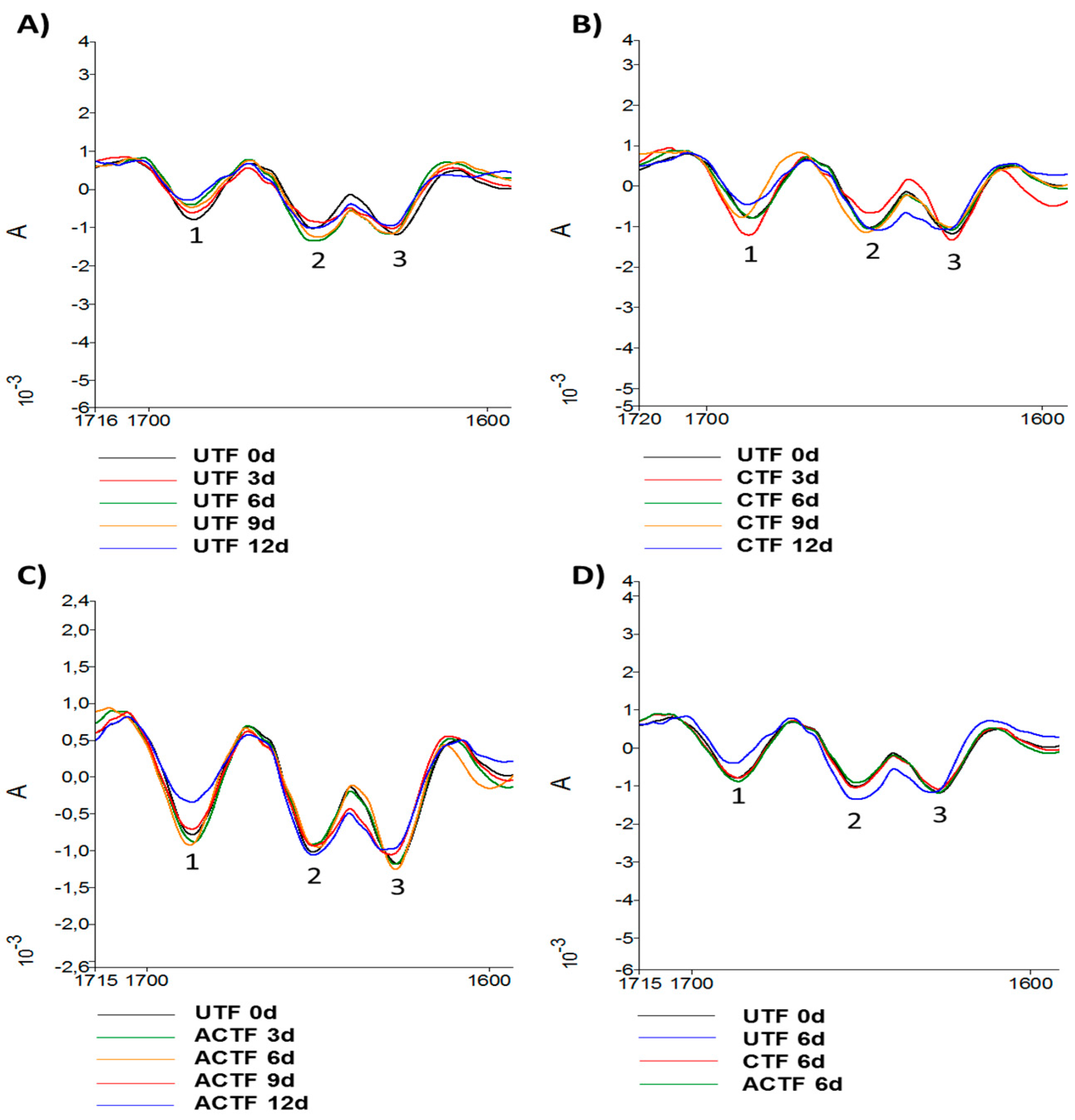
| Time (Days) | UTF | CTF | ACTF |
|---|---|---|---|
| 0 | 1.8 ± 0.02 A,a | 1.8 ± 0.05 A,a | 1.8 ± 0.07 A,a |
| 3 | 3.8 ± 0.06 A,b | 2.6 ± 0.04 B,b | 2.1 ± 0.06 C,b |
| 6 | 8.8 ± 0.04 A,c | 4.2 ± 0.08 C,c | 4.8 ± 0.03 B,c |
| 9 | 14.2 ± 0.07 A,d | 8.8 ± 0.09 B,d | 6.9 ± 0.09 C,d |
| 12 | 16.6 ± 0.02 A,e | 12.3 ± 0.10 B,e | 8.2 ± 0.03 C,e |
| Time (Days) | UTF | CTF | ACTF |
|---|---|---|---|
| 0 | 0.65 ± 0.06 A,a | 0.65 ± 0.07 A,a | 0.65 ± 0.04 A,a |
| 3 | 0.93 ± 0.08 A,b | 0.72 ± 0.09 B,b | 0.70 ± 0.03 C,b |
| 6 | 1.06 ± 0.01 A,c | 0.84 ± 0.07 B,c | 0.79 ± 0.06 C,c |
| 9 | 1.37 ± 0.07 A,d | 0.90 ± 0.05 B,d | 0.84 ± 0.08 C,d |
| 12 | 1.89 ± 0.13 A,e | 0.98 ± 0.04 B,e | 0.89 ± 0.06 C,e |
| UTF 0d | UTF 3d | UTF 6d | UTF 9d | UTF 12d | |
|---|---|---|---|---|---|
| UTF 0d | 0.000 | ||||
| UTF 3d | 0.940 a | 0.000 | |||
| UTF 6d | 0.954 b | 0.790 a | 0.000 | ||
| UTF 9d | 0.979 b | 0.946 a | 0.804 a | 0.000 | |
| UTF 12d | 0.985 b | 0.951 b | 0.911 a | 0.788 a | 0.000 |
| CTF 0d | CTF 3d | CTF 6d | CTF 9d | CTF 12d | |
|---|---|---|---|---|---|
| CTF 0d | 0.000 | ||||
| CTF 3d | 0.588 b | 0.000 | |||
| CTF 6d | 0.786 b | 0.480 a | 0.000 | ||
| CTF 9d | 0.811 b | 0.533 b | 0.611 b | 0.000 | |
| CTF 12d | 0.873 b | 0.729 b | 0.561 b | 0.247 a | 0.000 |
| ACTF 0d | ACTF 3d | ACTF 6d | ACTF 9d | ACTF 12d | |
|---|---|---|---|---|---|
| ACTF 0d | 0.000 | ||||
| ACTF 3d | 0.490 a | 0.000 | |||
| ACTF 6d | 0.632 b | 0.436 a | 0.000 | ||
| ACTF 9d | 0.820 b | 0.587 b | 0.395 a | 0.000 | |
| ACTF 12d | 0.863 b | 0.773 b | 0.715 b | 0.544 b | 0.000 |
| Peak Number | Peak Wavelength (cm−1) | Vibrational Mode | Components |
|---|---|---|---|
| 1 | 3276 | N–H stretch (Amide A) | Proteins |
| 2 | 3063 | N–H stretch (Amide B) | Proteins |
| 3 | 3011 | =CH oleficic stretch | Unsaturated lipids |
| 4 | 2957 | CH3 asymmetric stretch | Lipids and proteins |
| 5 | 2923 | CH2 asymmetric stretch. | Saturated lipids and side chains of proteins, cholesterol, phospholipids |
| 6 | 2873 | CH3 symmetric stretch | Lipids and proteins |
| 7 | 2853 | CH2 symmetric stretch. | Saturated lipids and side chains of proteins |
| 8 | 1743 | C=O stretch | Lipids, phospholipids |
| 9 | 1630 | C=O stretch + NH bend (amide I) | Proteins |
| 10 | 1539 | N–H bend + C–N stretch (amide II) | Proteins |
| 11 | 1451 | CH2 Scissoring vibrations | Pyrrolidine rings of proline and hydroxyproline |
| 12 | 1393 | COO− symmetric stretch. | Fatty acids, amino acids |
| 13 | 1305 | Amide III | Proteins |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Volpe, M.G.; Coccia, E.; Siano, F.; Di Stasio, M.; Paolucci, M. Rapid Evaluation Methods for Quality of Trout (Oncorhynchus mykiss) Fresh Fillet Preserved in an Active Edible Coating. Foods 2019, 8, 113. https://doi.org/10.3390/foods8040113
Volpe MG, Coccia E, Siano F, Di Stasio M, Paolucci M. Rapid Evaluation Methods for Quality of Trout (Oncorhynchus mykiss) Fresh Fillet Preserved in an Active Edible Coating. Foods. 2019; 8(4):113. https://doi.org/10.3390/foods8040113
Chicago/Turabian StyleVolpe, Maria Grazia, Elena Coccia, Francesco Siano, Michele Di Stasio, and Marina Paolucci. 2019. "Rapid Evaluation Methods for Quality of Trout (Oncorhynchus mykiss) Fresh Fillet Preserved in an Active Edible Coating" Foods 8, no. 4: 113. https://doi.org/10.3390/foods8040113
APA StyleVolpe, M. G., Coccia, E., Siano, F., Di Stasio, M., & Paolucci, M. (2019). Rapid Evaluation Methods for Quality of Trout (Oncorhynchus mykiss) Fresh Fillet Preserved in an Active Edible Coating. Foods, 8(4), 113. https://doi.org/10.3390/foods8040113







