Platinum and Rhodium in Potato Samples by Using Voltammetric Techniques
Abstract
:1. Introduction
2. Materials and Methods
2.1. Instrumentation
2.2. Reagents
2.3. Quality Assurance
2.4. Samples
2.5. Mineralization Procedure
2.6. Analytical Methods
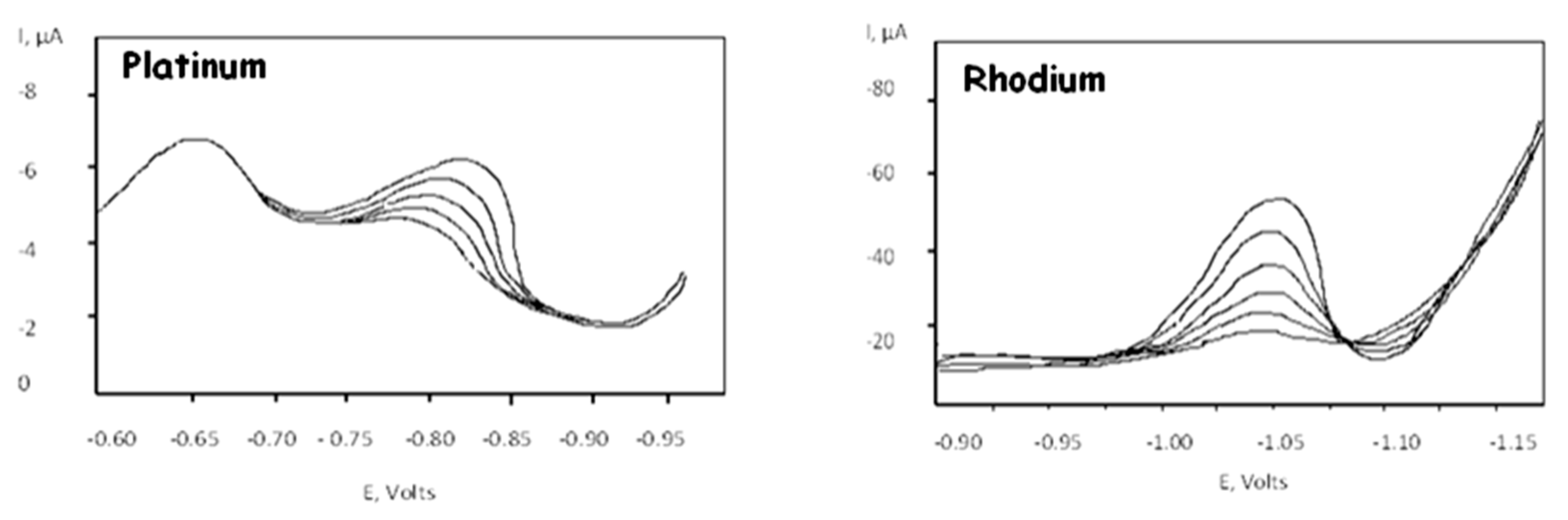
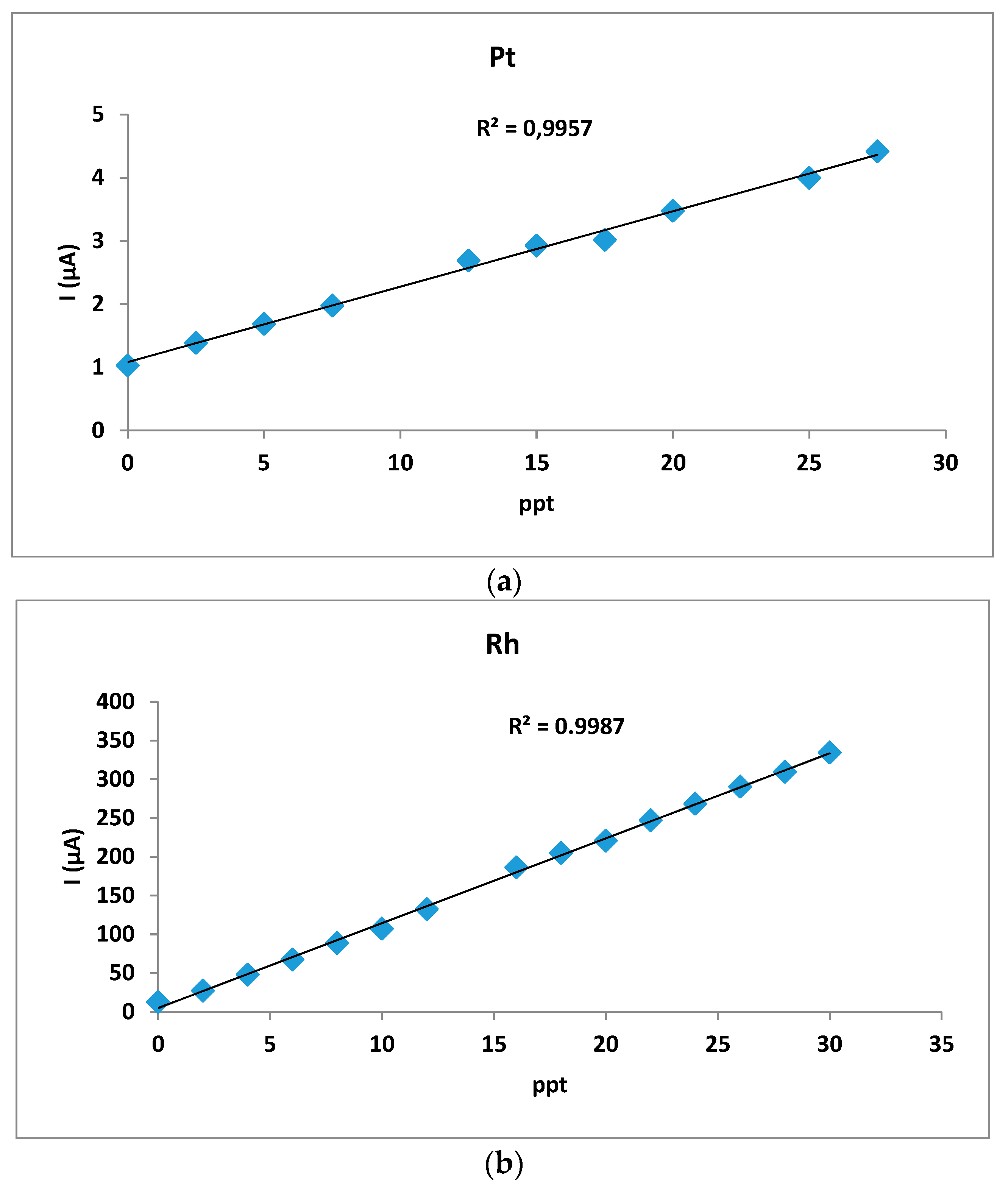
3. Results and Discussion
4. Daily Intake and Health Risk
5. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Helmers, E.; Mergel, N. Platinum and rhodium in a polluted environment: Studying the emissions of automobile catalysts with emphasis on the application of CSV rhodium analysis. Fresenius J. Anal. Chem. 1998, 362, 522–528. [Google Scholar] [CrossRef]
- Schafer, J.; Pulchet, H. Platinum group metals (PGM) emitted from automobile catalytic converters and their distribution in roadside soil. J. Geochem. Explor. 1998, 64, 307–314. [Google Scholar] [CrossRef]
- Schlogl, R.; Indlekofer, G.; Oelhafen, P. Emission of micro particles from automotive sources–X-ray photoelectron-spectroscopy in environmental analysis. Angew. Chem. Int. Ed. 1987, 26, 309–319. [Google Scholar] [CrossRef]
- Lustig, S.; Zang, S.; Michalke, B.; Schramel, P.; Beck, W. Transformation behavior of different platinum compounds in clay humic soil: Speciation investigation. Sci. Total Environ. 1996, 188, 195–204. [Google Scholar] [CrossRef]
- Rosenberg, B.; van Camp, L.; Grimley, E.B.; Thomson, A.J. The inhibition of growth or cell division in Escherichia coli by different ionic species of platinum(IV) complexes. J. Biol. Chem. 1967, 242, 1347–1352. [Google Scholar]
- Kelland, L.R.; Farrell, N.P. Platinum-Based Drugs in Cancer Therapy; Humana Press: Totowa, NJ, USA, 2000. [Google Scholar]
- Amatori, S.; Ambrosi, G.; Provenzano, A.E.; Fanelli, M.; Formica, M.; Fusi, V.; Giorgi, L.; Macedi, E.; Micheloni, M.; Paoli, P.; et al. PdII and PtII complexes with a thio-aza macrocycle ligand containing an intercalating fragment: Structural and antitumor activity studies. J. Inorg. Biochem. 2016, 162, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Orecchio, S.; Amorello, D. Platinum levels in urban soils from Palermo (Italy); Analytical method using voltammetry. Microchem. J. 2011, 99, 283–288. [Google Scholar] [CrossRef]
- Orecchio, S.; Amorello, D. Voltammetric analysis of platinum in environment. In Platinum Metals in the Environment; Zerein, F., Wiseman, C.L.S., Eds.; Springer: Berlin, Germany, 2015; pp. 79–86. [Google Scholar]
- Liu, Q.T.; Diamond, M.E.; Gingrich, S.E.; Ondov, J.M.; Maciejczyk, P.; Gary, A.S. Accumulation of metals, trace elements and semi volatile organic compounds on exterior windows surfaces in Baltimore. Environ. Pollut. 2003, 122, 51–61. [Google Scholar] [CrossRef]
- Wichmann, H.; Anquandah, G.A.K.; Schmidt, C.; Zachmann, D.; Bahadir, A.M. Increase of platinum group element concentrations in soils and airborne dust in an urban area in Germany. Sci. Total Environ. 2007, 388, 121–127. [Google Scholar] [CrossRef]
- Amorello, D.; Barreca, S.; Gulli, E.; Orecchio, S. Platinum and Rhodium in wine samples by using voltammetric techniques. Microchem. J. 2017, 130, 229–235. [Google Scholar] [CrossRef]
- Peng, Y.; Yang, R.; Jin, T.; Chen, J.; Zhang, J. Risk assessment for potentially toxic metal(loid)s in potatoes in the indigenous zinc smelting area of northwestern Guizhou Province, China. Food Chem. Toxicol. 2018, 120, 328–339. [Google Scholar] [CrossRef] [PubMed]
- Krachler, M.; Alimonti, A.; Petrucci, F.; Irgolic, K.J.; Caroli, S. Analytical problem in the determination of platinum group metals in urine by quadruple and magnetic sector field inductively coupled plasma mass spectrometry. Anal. Chim. Acta 1998, 1, 363–369. [Google Scholar]
- Zimmermann, S.; Menzel, C.M.; Berner, Z.; Eckhardt, J.D.; Stüben, D.; Alt, F.; Messerschmidt, J.; Taraschewski, H.; Sures, B. Trace analysis of platinum in biological samples: A comparison between sector field ICP-MS and adsorptive cathodic stripping voltammetry following different digestion procedures. Anal. Chim. Acta 2001, 439, 203–209. [Google Scholar] [CrossRef]
- Vlasankova, R.; Otruba, V.; Bendl, J.; Fisera, M.; Kanicky, V. Preconcentration of platinum group metals on modified silica gel and their determination by inductively coupled plasma atomic emission spectrometry and inductively coupled plasma mass spectrometry in airborne particulates. Talanta 1999, 48, 839–846. [Google Scholar] [CrossRef]
- Panahi, H.A.; Kalal, H.S.; Moniri, E.; Nezhati, M.N.; Menderjani, M.T.; Kelahrodi, S.R.; Mahmoudi, F. Amberlite XAD-4 functionalized with m-phenylendiamine: Synthesis, characterization and applications as extractant for preconcentration and determination of rhodium (III) in water samples by inductive couple plasma atomic emission spectroscopy (ICP-AES). Microchem. J. 2009, 93, 49–54. [Google Scholar] [CrossRef]
- Piech, R.; Baśa, B.; Kubiaka, W.W. The cyclic renewable mercury film silver based electrode for determination of molybdenum(VI) traces using adsorptive stripping voltammetry. Talanta 2008, 76, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Celik, U.; Oehlenschlager, J. Determination of zinc and copper in fish samples collected from Northeast Atlantic by DPSAV. Food Chem. 2004, 87, 343–347. [Google Scholar] [CrossRef]
- Stoica, A.I.; Peltea, M.; Baiulescu, G.E.; Ionica, M. Determination of cobalt in pharmaceutical products. J. Pharm. Biomed. Anal. 2004, 36, 653–656. [Google Scholar] [CrossRef]
- Locatelli, C. Simultaneous square wave stripping voltammetric determination of platinum group metals (PGMs) and lead at trace and ultra trace concentration level, application to surface water. Anal. Chim. Acta 2006, 557, 70–77. [Google Scholar] [CrossRef]
- Amorello, D.; Orecchio, S. Micro determination of dithiocarbamates in pesticide formulations using voltammetry. Microchem. J. 2013, 110, 334–339. [Google Scholar] [CrossRef]
- Leon, C.; Emons, H.; Ostapczuk, P.; Hoppstock, K. Simultaneous ultra trace determination of platinum and rhodium by cathodic stripping voltammetry. Anal. Chim. Acta 1997, 356, 99–104. [Google Scholar] [CrossRef]
- Orecchio, S.; Amorello, D.; Carollo, C. Voltammetric determination of platinum in perfusate and blood: Preliminary data on pharmacokinetic study of arterial infusion with oxaliplatin. Microchem. J. 2012, 100, 72–76. [Google Scholar] [CrossRef]
- Orecchio, S.; Amorello, D. Platinum and rhodium associated with the leaves of Nerium oleander L.; Analytical method using voltammetry; Assessment of air quality in the Palermo (Italy) area. J. Hazard. Mater. 2010, 174, 720–727. [Google Scholar] [CrossRef]
- Helmers, E.; Mergel, N. Platinum and rhodium in the polluted environment: Studying the emissions of automobile catalysts with emphasis on the application of CSV rhodium analysis. Fresenius J. Anal. Chem. 2002, 362, 522–528. [Google Scholar] [CrossRef]
- Vaughan, G.T.; Florence, T.M. Platinum in the human diet, blood, hair and excreta. Sci. Total Environ. 1992, 111, 47–58. [Google Scholar] [CrossRef]
- Helmers, E.; Schwarzer, M.; Schuster, M. Comparison of palladium and platinum in environmental matrices: Palladium pollution by automobile emissions. Environ. Sci. Pollut. Res. 1998, 5, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Nolting, R.F.; Ramkema, A.; Everaarts, J.M. The geochemistry of Cu, Cd, Zn, Ni and Pb in sediment cores from the continental slope of the banc d’Arguin (Mauritania). Cont. Shelf Res. 1999, 19, 665–691. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements; Pergamon Press: Oxford, UK, 1985. [Google Scholar]
- European Medicines Agency Pre-authorization Evaluation of Medicines for Human Use, Doc. Ref. CPMP/SWP/QWP/4446/00 corr, 2007 (London). Available online: https://www.ema.europa.eu/documents/work-programme/work-programme-european-medicines-agency-2008_en.pdf (accessed on 19 February 2019).
- Hoppstock, K.; Sures, B. Platinum group metals. In Elements and Their Compounds in the Environment; Merian, E., Anke, M., Ihnat, M., Stoeppler, M., Eds.; Wiley-VCH: Weinheim, Germany, 2004; pp. 1047–1086. [Google Scholar]
- Potatoes: Health Benefits, Risks & Nutrition Facts. Available online: https://www.livescience.com/45838-potato-nutrition.html (accessed on 19 February 2019).

| Analytes | Tecniques | Electrolytes | Reagent |
|---|---|---|---|
| Pt | DPV/a | H2SO4 1 M | (N2H4SO4) = 1.2 mmol L−1, (H2CO) = 0.6 mmol L−1 |
| Rh | DPSAV | HCl 0.42 M | (H2CO) = 0.02 mol L−1 |
| Parameter | Pt | Rh |
|---|---|---|
| Initial potential (mV) | −300 | −900 |
| Final potential (mV) | −1000 | −1200 |
| Current range | Automatic | Automatic |
| Potential scan rate (mV s−1) | 50 | 10 |
| Potential of deposition (mV) | - | −700 |
| Cycle n | 1 | 1 |
| Deposition time (s) | - | 30 |
| Stirring rate (r.p.m.) | 300 | 300 |
| Size of the drop (a.u.) | 60 | 60 |
| Delay time before potential sweep (s) | 10 | 10 |
| Working electrode | Hanging mercury drop electrode | |
| Auxiliary electrode | Glassy carbon | |
| Reference electrode | Ag/AgCl/KCl (sat) | |
| Flowing gas | Nitrogen (99.998%) | |
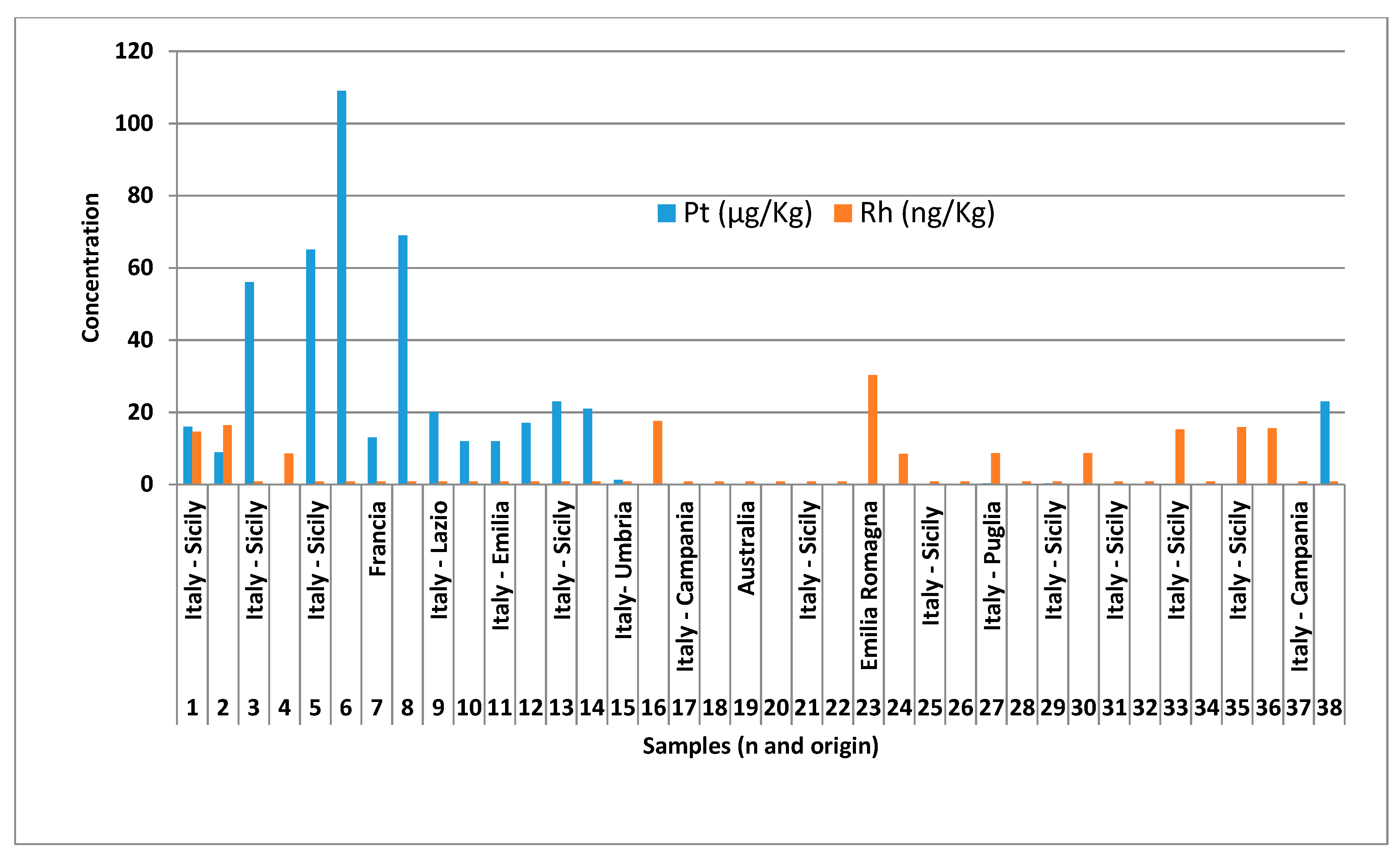
| Sample | Origin | Pt (μg/kg) | R.S.D. % ± | Rh (μg/kg) | R.S.D. % ± |
|---|---|---|---|---|---|
| 1 | Italy - Sicily | 16 | 2.2 | 0.015 | 5.4 |
| 2 | Italy - Sicily | 8.9 | 6.5 | 0.016 | 6.5 |
| 3 | Italy - Sicily | 56 | 2.4 | 0.0008 | 11 |
| 4 | Italy - Sicily | 6.7 | 6.1 | 0.0086 | 4.2 |
| 5 | Italy - Sicily | 65 | 1.0 | 0.0008 | 10 |
| 6 | Italy - Sicily | 109 | 0.5 | 0.0008 | 11 |
| 7 | Francia | 13 | 6.2 | 0.0008 | 20 |
| 8 | Italy - Veneto | 69 | 4.0 | 0.0008 | 12 |
| 9 | Italy - Lazio | 20 | 3.5 | 0.0008 | 9.9 |
| 10 | Italy - Marche | 12 | 5.2 | 0.0008 | 16 |
| 11 | Italy - Emilia | 12 | 4.7 | 0.0008 | 4.9 |
| 12 | Italy - Emilia | 17 | 2.3 | 0.0008 | 14 |
| 13 | Italy - Sicily | 23 | 2.4 | 0.0008 | 13 |
| 14 | Francia | 21 | 1.0 | 0.0008 | 13 |
| 15 | Italy - Umbria | 1.3 | 5.6 | 0.0008 | 5.6 |
| 16 | Belgio | 0.007 | 8.0 | 0.017 | 8.0 |
| 17 | Italy - Campania | 0.007 | 11 | 0.0008 | 21 |
| 18 | Italy - Puglia | 0.007 | 10 | 0.0008 | 25 |
| 19 | Australia | 0.007 | 12 | 0.0008 | 12 |
| 20 | Italy - Abruzzo | 0.007 | 16 | 0.0008 | 15 |
| 21 | Italy - Sicily | 0.007 | 12 | 0.0008 | 16 |
| 22 | Italy - Sicily | 0.21 | 3.2 | 0.0008 | 3.2 |
| 23 | Emilia Romagna | 0.007 | 12 | 0.0303 | 6.1 |
| 24 | Italy - Sicily | 0.007 | 13 | 0.0085 | 2.4 |
| 25 | Italy - Sicily | 0.007 | 11 | 0.0008 | 25 |
| 26 | Italy - Sicily | 0.007 | 18 | 0.0008 | 22 |
| 27 | Italy - Puglia | 0.288 | 12 | 0.0087 | 11 |
| 28 | Italy - Abruzzo | 0.233 | 15 | 0.0008 | 24 |
| 29 | Italy - Sicily | 0.326 | 15 | 0.0008 | 16 |
| 30 | Italy - Sicily | 0.007 | 10 | 0.0087 | 10 |
| 31 | Italy - Sicily | 0.007 | 17 | 0.0008 | 21 |
| 32 | Italy - Sicily | 0.007 | 14 | 0.0008 | 22 |
| 33 | Italy - Sicily | 0.007 | 15 | 0.015 | 0.88 |
| 34 | Italy - Sicily | 0.007 | 16 | 0.0008 | 24 |
| 35 | Italy - Sicily | 0.007 | 18 | 0.016 | 7.6 |
| 36 | Italy - Sicily | 0.007 | 25 | 0.016 | 7.2 |
| 37 | Italy - Campania | 0.16 | 14 | 0.0008 | 14 |
| 38 | Italy - Sicily | 23 | 1.2 | 0.0008 | 33 |
| Place | Pt (μg/kg) | Rh (μg/kg) | Sample |
|---|---|---|---|
| Stuttgart | 2.9 4.6 | - - | Roadside grass (1993) Roadside grass (0.2 m) (1994) |
| Gent (Belgio) | 1.4–1.7 | - | Roadside grass |
| Germania | 3.61 | 0.65 | Roadside grass (1994) |
| 10.6 | 1.54 | Roadside grass (1997) | |
| ≤0.03 | ≤0.03 | Area uncontaminated (1997) | |
| Sheffield | 0.07–5.4 | - | Bark |
| Bialystok (Polonia) | 8.63 | 0.65 | Roadside grass (1 m) |
| San Francisco | 38 | - | Bark |
| EF < 2 | Deficiency to Minimal Enrichment |
|---|---|
| EF 2–5 | Moderate enrichment |
| EF 5–20 | Significant enrichment |
| EF 20–40 | Very high enrichment |
| EF N 40 | Extremely high enrichment |
| Class | Index | Significance |
|---|---|---|
| 0 | <0 | Practically uncontaminated |
| 1 | 0–1 | Uncontaminated to moderately contaminated |
| 2 | 1–2 | Moderately contaminated |
| 3 | 2–3 | Moderately to heavily contaminated |
| 4 | 3–4 | Heavily contaminated |
| 5 | 4–5 | Heavily to extremely contaminated |
| 6 | 5 | Extremely contaminated |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Orecchio, S.; Amorello, D. Platinum and Rhodium in Potato Samples by Using Voltammetric Techniques. Foods 2019, 8, 59. https://doi.org/10.3390/foods8020059
Orecchio S, Amorello D. Platinum and Rhodium in Potato Samples by Using Voltammetric Techniques. Foods. 2019; 8(2):59. https://doi.org/10.3390/foods8020059
Chicago/Turabian StyleOrecchio, Santino, and Diana Amorello. 2019. "Platinum and Rhodium in Potato Samples by Using Voltammetric Techniques" Foods 8, no. 2: 59. https://doi.org/10.3390/foods8020059
APA StyleOrecchio, S., & Amorello, D. (2019). Platinum and Rhodium in Potato Samples by Using Voltammetric Techniques. Foods, 8(2), 59. https://doi.org/10.3390/foods8020059





