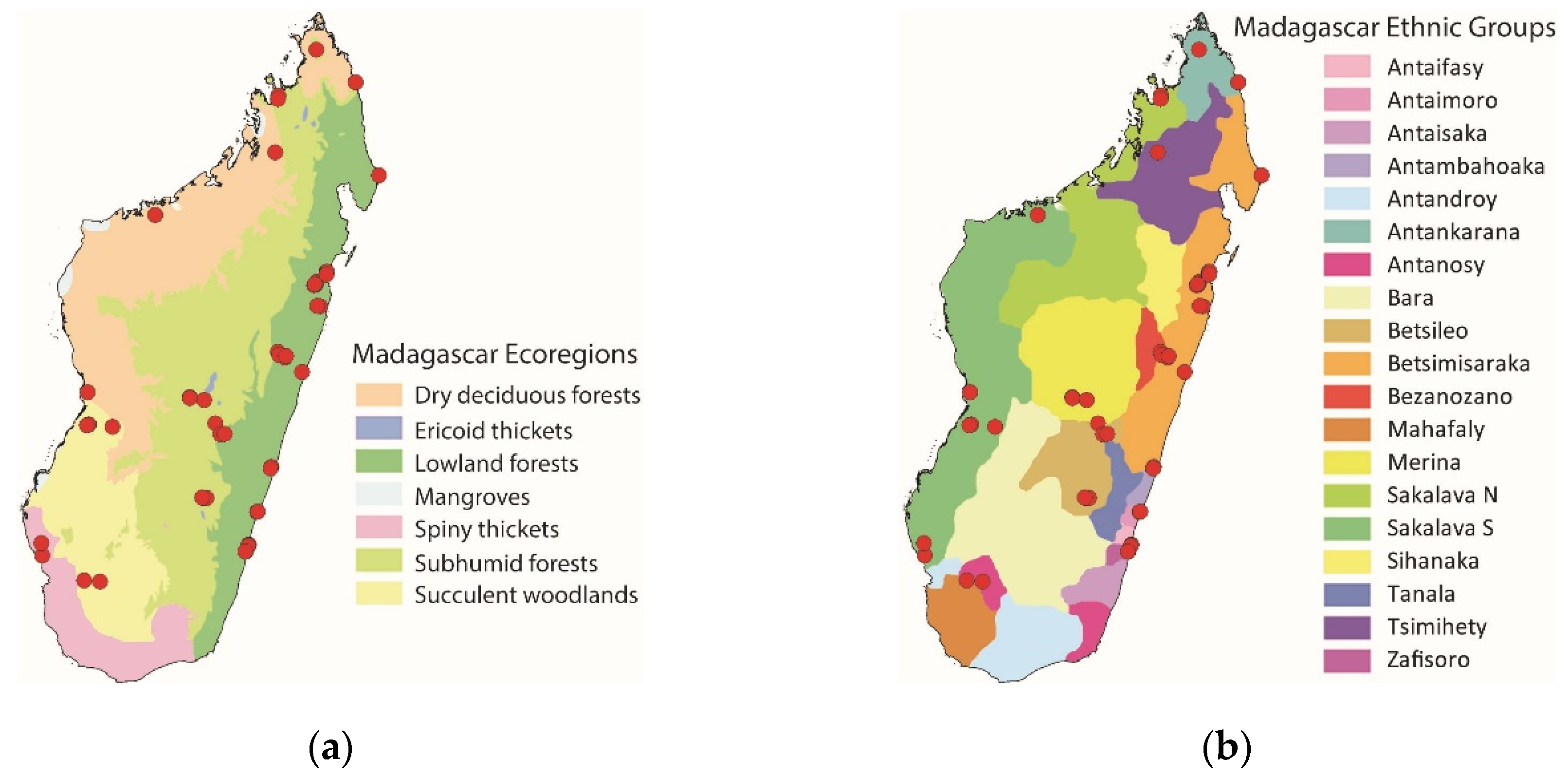
3.1. Edible Orthopteran Diversity
We recorded 37 species of edible Orthoptera, of which 31 were able to be identified to species level and six to either genus or subfamily level (
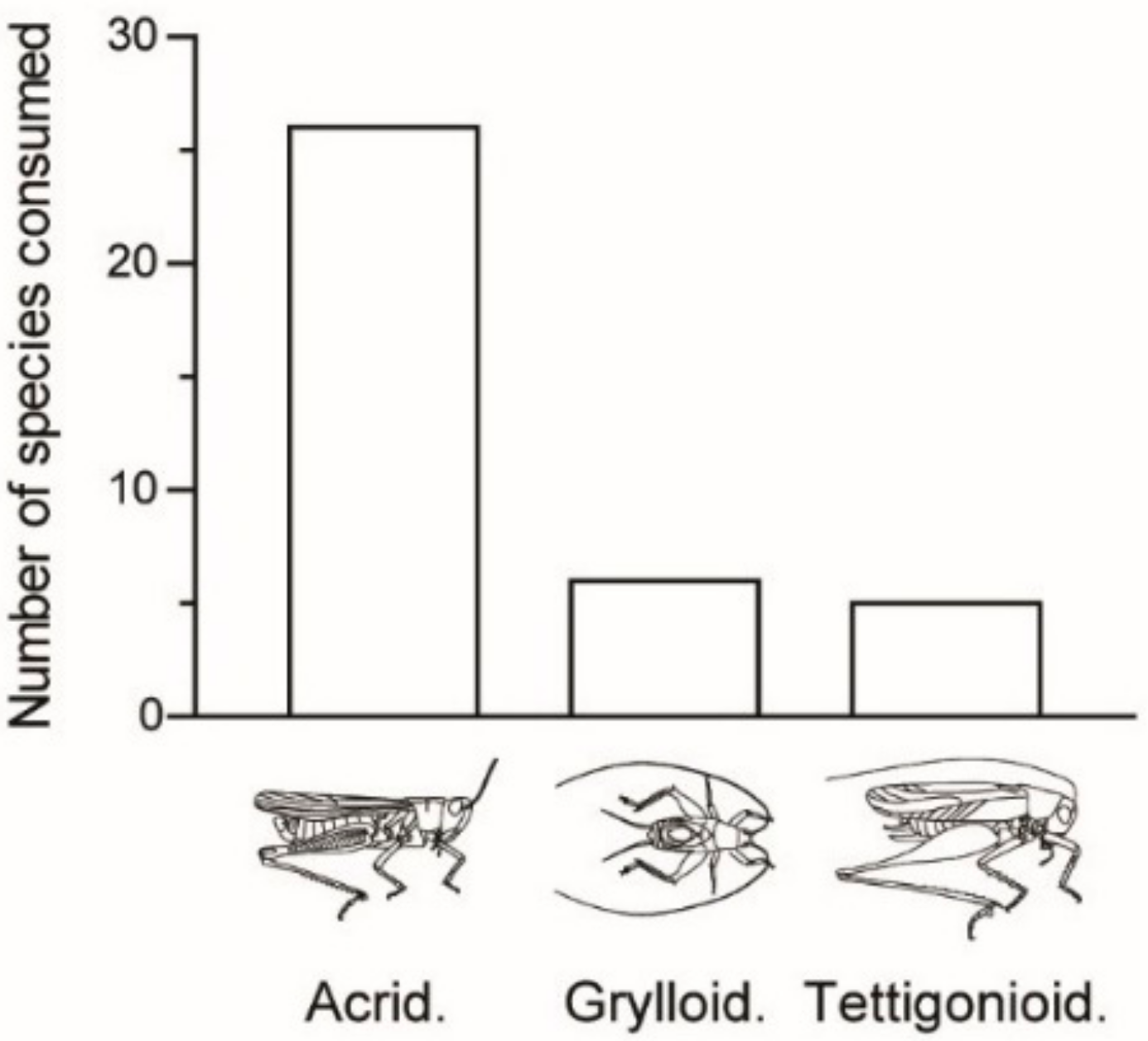
Table 1). This edible Orthopteran diversity corresponds to 3% of the recorded Orthopteran diversity in Madagascar (ca. 700 species, likely underestimated). These 37 species are comprised of 26 species of grasshoppers (Caelifera) and 11 species of crickets and katydids (Ensifera;
Figure 2). Most of the edible Caelifera belong to the Acrididae (23 out of 26 species). The edible Ensifera includes Tettigonioidea (bush crickets/katydids, six species) and Grylloidea (crickets, five species).
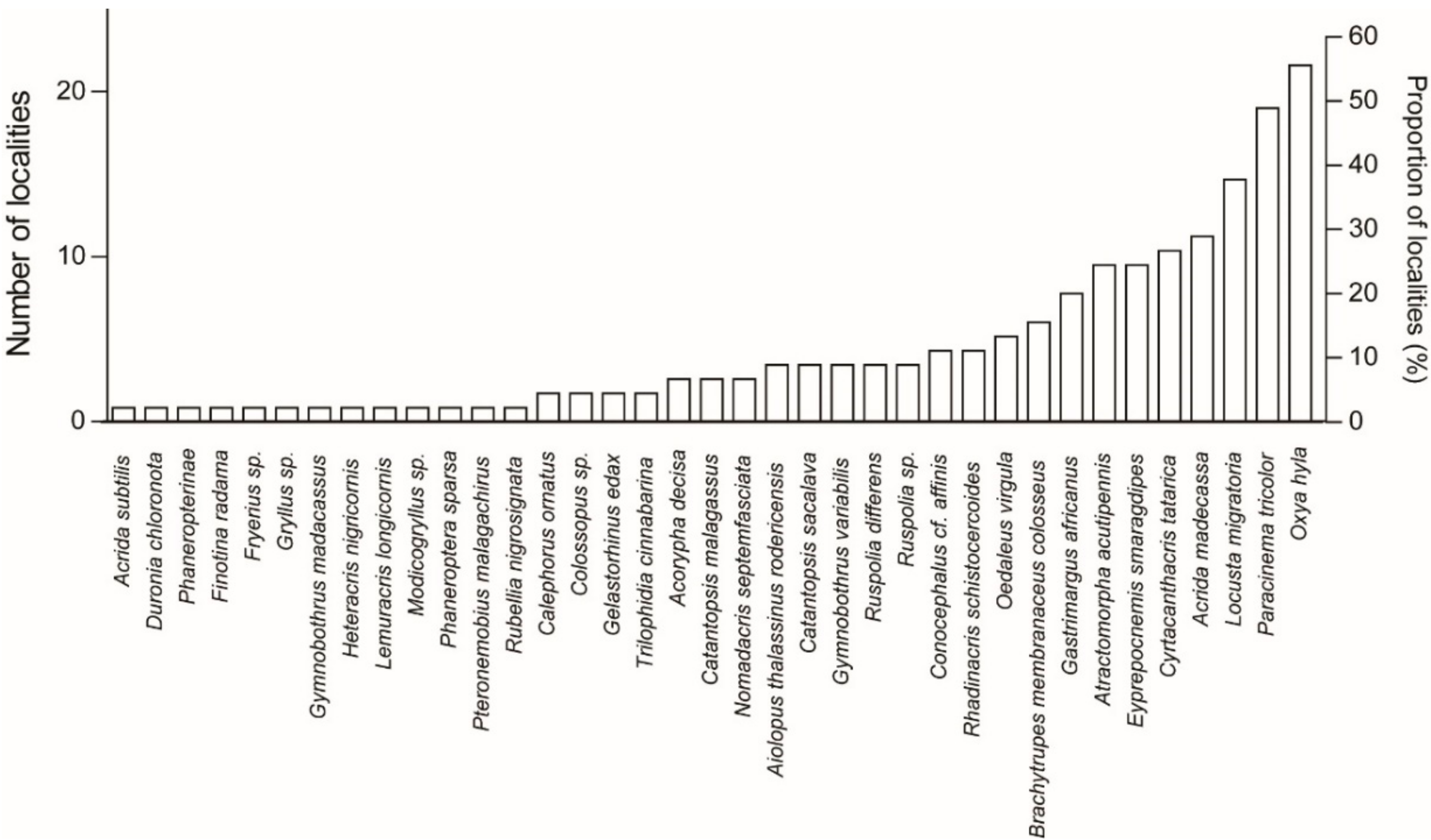
The five most recorded species were found to be
Oxya hyla (25 localities in three ecoregions: lowland forests, subhumid forests, and succulent woodlands; nine ethnic groups),
Paracinema tricolor (22 localities in three ecoregions: lowland forests, subhumid forests, and succulent woodlands; nine ethnic groups),
Locusta migratoria (17 localities in five ecoregions: lowland forests, subhumid forests, succulent woodlands, dry deciduous forests, and spiny thickets; eight ethnic groups),
Acrida madecassa (13 localities in five ecoregions: lowland forests, subhumid forests, succulent woodlands, dry deciduous forests, and spiny thickets; six ethnic groups), and
Cyrtacanthacris tatarica (12 localities in five ecoregions: lowland forests, subhumid forests, succulent woodlands, dry deciduous forests, and spiny thickets; seven ethnic groups). Of the 37 recorded species, 14 species were sampled only once (i.e., one locality per species;
Figure 3). An annotated list of edible Orthoptera is provided in
Section 3.3.
The total count of 37 species is conservative. In cases of unidentified species, all specimens belonging to a single genus or subfamily were counted as a single species. We thus have not counted Acrida sp. and Brachytrupes sp. as a different species from Acrida subtilis and A. madecassa, and from Brachytrupes membranaceus colosseus, respectively, since only juveniles were collected and could not be identified to species level. Ruspolia sp., however, was considered a separate species since some adults were collected and the specimens were definitely not Ruspolia differens. Both juveniles and adults are edible. Among the 279 species-sorted batches examined for the present work, 87% contained at least some juveniles. Juveniles are smaller than adults but cannot escape by flying.
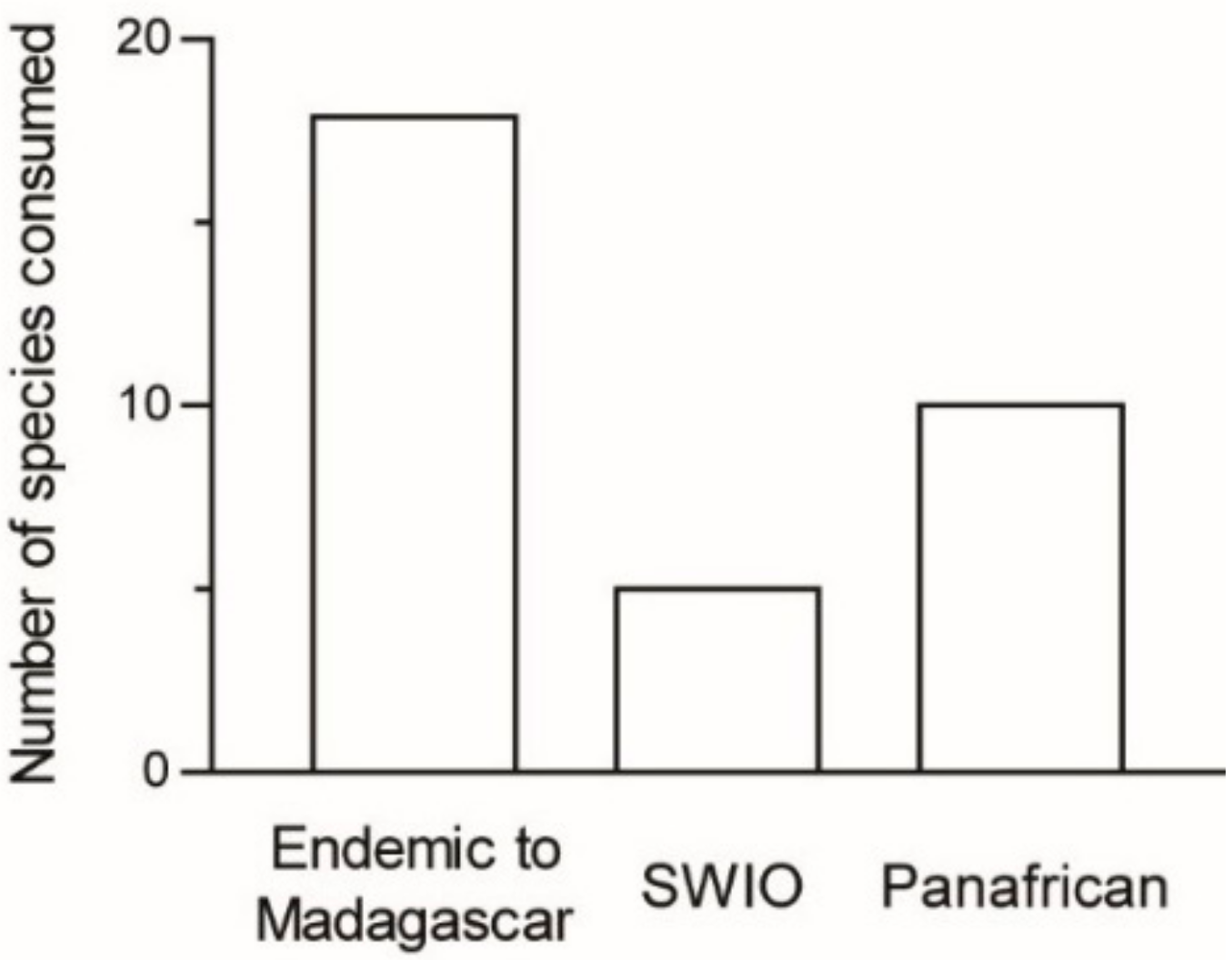
Most of the recorded species are endemic to the Malagasy area: 18 species (56%) are endemic to Madagascar and five species (16%) occur also in nearby islands, whereas nine species (28%) are widely distributed across Africa (
n = 32 species;
Figure 4).
Somewhat surprisingly, two Caelifera species, Rubellia nigrosignata and Atractomorpha acutipennis, belong to the Pyrgomorphidae, a family with many species considered to be unpleasant-tasting or -smelling, or even toxic.
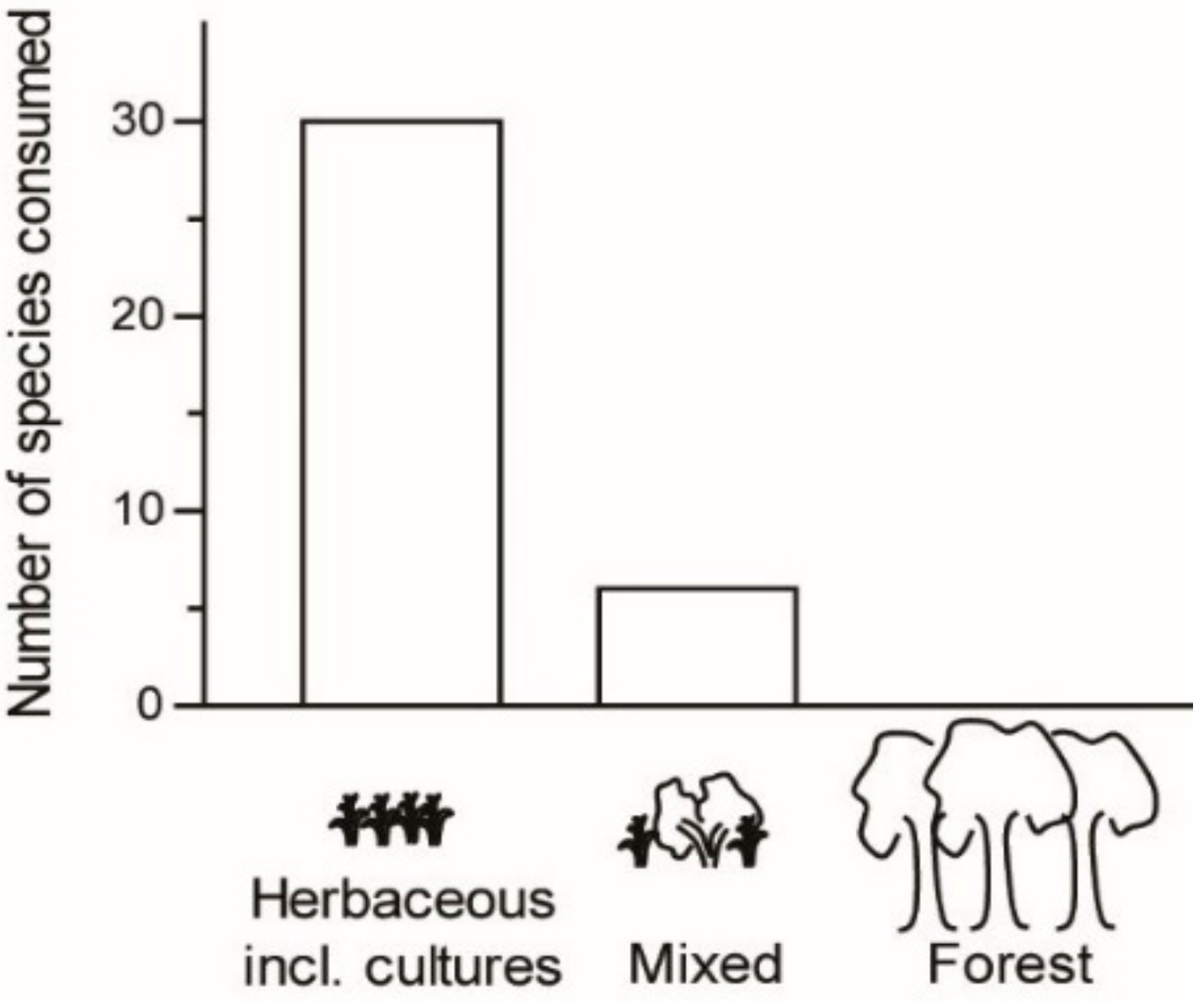
Most edible Orthoptera were found to occur in habitats with herbaceous plant cover, including farming zones (83%); few were found to occur in areas mixed with taller plant cover (17%); and none were found to occur in forest habitat (
n = 36;
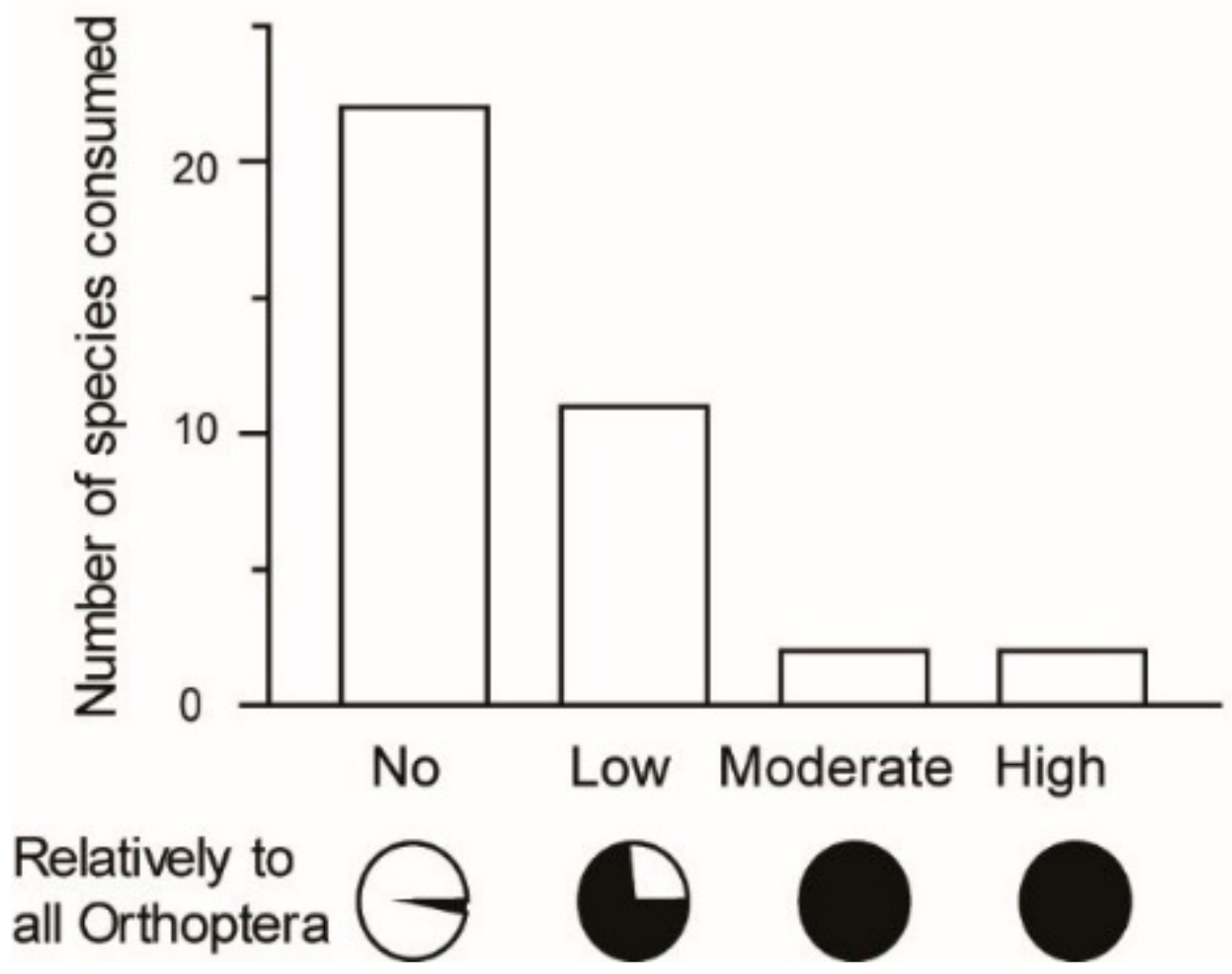
Figure 5). The majority of Orthoptera species recorded as edible were no threat to crops (22 species, 59% of recorded edible Orthoptera in the present study;
Figure 6). Fifteen species were found to be a threat to crops, of which two species posed a high threat (5%), two species a moderate threat (5%), and 11 species a low threat (30%;
n = 37 species). Our record indicates that threat level is associated with use as food: all species representing a high threat (100%) and a moderate threat (100%) to crops were reported as edible, whereas 73% of species posing a low threat were reported as edible and 3% of species posing no threat were reported as edible (
n = 37 species). Rice field ecosystems, other agricultural fields, and fallow land near agricultural fields are the main sources of the edible Orthoptera bioresource used in Madagascar.
3.2. Edible Orthoptera Uses
The highest edible Orthopteran diversity was recorded among the Merina in the subhumid forests ecoregion (22 species, 59% of recorded edible Orthoptera in the present study), the Betsileo in the subhumid forests ecoregion (20 species, 54%), and the Betsimsaraka in the lowland forest ecoregion (18 species, 49%). The lowest edible Orthopteran diversity was recorded among the Sakalava N (North) and Tsimihety, both in the dry deciduous forests ecoregion (two species each, 5%), the Bezanozano in the lowland forests ecoregion (three species, 8%), the Antankarana in the dry deciduous forests ecoregion (four species, 11%), and the Antanosy in the succulent woodlands ecoregion (four species, 11%). The most widespread used edible Orthoptera were found to be
P. tricolor and
O. hyla (nine ethnic groups each),
L. migratoria (eight ethnic groups), and
C. tatarica and
Eyprepocnemis smaragdipes (seven ethnic groups each). The most common vernacular name observed for edible Orthoptera was
valala, with some other names also mentioned (
Table 1).
Edible Orthoptera are present throughout the year with roughly two peak abundance periods from October–January and March–June. Edible Orthoptera are primarily collected casually in Madagascar. All ethnicities collect the insects only by hand. Children are the primary collectors and consumers of Orthoptera in all ethnicities, although adults may also collect and eat them. No gender differences seem to be present in the collection and consumption of Orthoptera. Collecting edible Orthoptera is a common game for children playing in the rice fields or walking home from school. The children then prepare and eat the Orthoptera themselves or have their mothers prepare the insects for them (mothers may then snack on the Orthoptera with their children). Adults mostly collect Orthoptera during their peak availability but may also collect them outside of the peak availability periods, notably while working in the rice fields. When adults collect edible Orthoptera, they may prefer to give them to their children for playing and for eating. Orthoptera are eaten as a snack and as part of a main meal. The wings, legs, head, and intestines may be removed before the insects are prepared by grilling, frying, and frying after being cooked in water. The insects may be seasoned with salt. Some recipes to prepare Orthoptera in Madagascar are provided in the
Supplementary File. We do not have clear information about collection and consumption frequencies and quantities. Orthoptera may be used as animal feed (e.g., poultry) but this seems rare. Preservation of Orthoptera is very rare. Only locusts in south Madagascar are dried when outbreaks occur.
Marketing is rare due to low numbers of Orthoptera that are collected casually and due to time constraints to increase Orthoptera yields. Some exceptions occur. Locusts are marketed during outbreaks in south and central Madagascar, and adults may then collect locusts even daily and opportunistically to eat and market them. The large-sized
B. membranaceus colosseus may be intentionally collected for marketing purposes in west Madagascar. This cricket may also be specifically sought after when meat is expensive and when no other rice accompaniments are available. The cricket is sold for 100–200 Malagasy Ariary (0.027–0.055 US
$) per individual body and for 500–1500 Malagasy Ariary (0.14–0.41 US
$) per
kapoaka, a tin can of about 400 mL commonly used as a unit of measurement for selling produce. The level of edible Orthoptera marketing seems to be lower than in some other countries with Orthoptera consumption, such as Togo [
21], Laos [
22], and Japan [
23,
24].
Malagasy Muslims, who live primarily in north-west Madagascar, do not eat any insects for religious reasons. The eating of sakoririka, a vernacular name for Orthoptera that at least includes the genus Ruspolia, is taboo among the Antaisaka ethnic group: the insect is only consumed as a medicine by infants who refrain from speech, or are incapable of speech, to help them to speak. This taboo may not be shared by all ethnicities. The taboo was mentioned by members of the Antaisaka ethnic group of southeast Madagascar who had migrated to the regions commonly associated with the Sakalava ethnic groups in west Madagascar. No other medicinal use of Orthoptera was mentioned in any surveyed localities. Mole crickets are not considered edible due to a widespread cultural belief. They are called “zazavery”, which means “lost child”. The Malagasy people regularly encounter mole crickets in wet soils, e.g., along riverbanks while cultivating vegetables or cutting wood. However, these mole crickets quickly disappear as they dig themselves into the wet soil to hide. The Malagasy liken this act of disappearing to the behavior of children who may suddenly wander off to play without notifying their parents. The large Pyrgomorphidae grasshopper Phymateus saxosus is renowned as inedible in Madagascar, as are some other Pyrgomorphidae. It has aposematic coloration and produces an unpleasant smell when disturbed. This grasshopper is named “dog’s grasshopper” (valalan’alika or valalan’amboa) and Malagasy people say that “even dogs don’t eat them”.
3.3. Annotated List of Edible Orthoptera
3.3.1. Caelifera, Acridinae
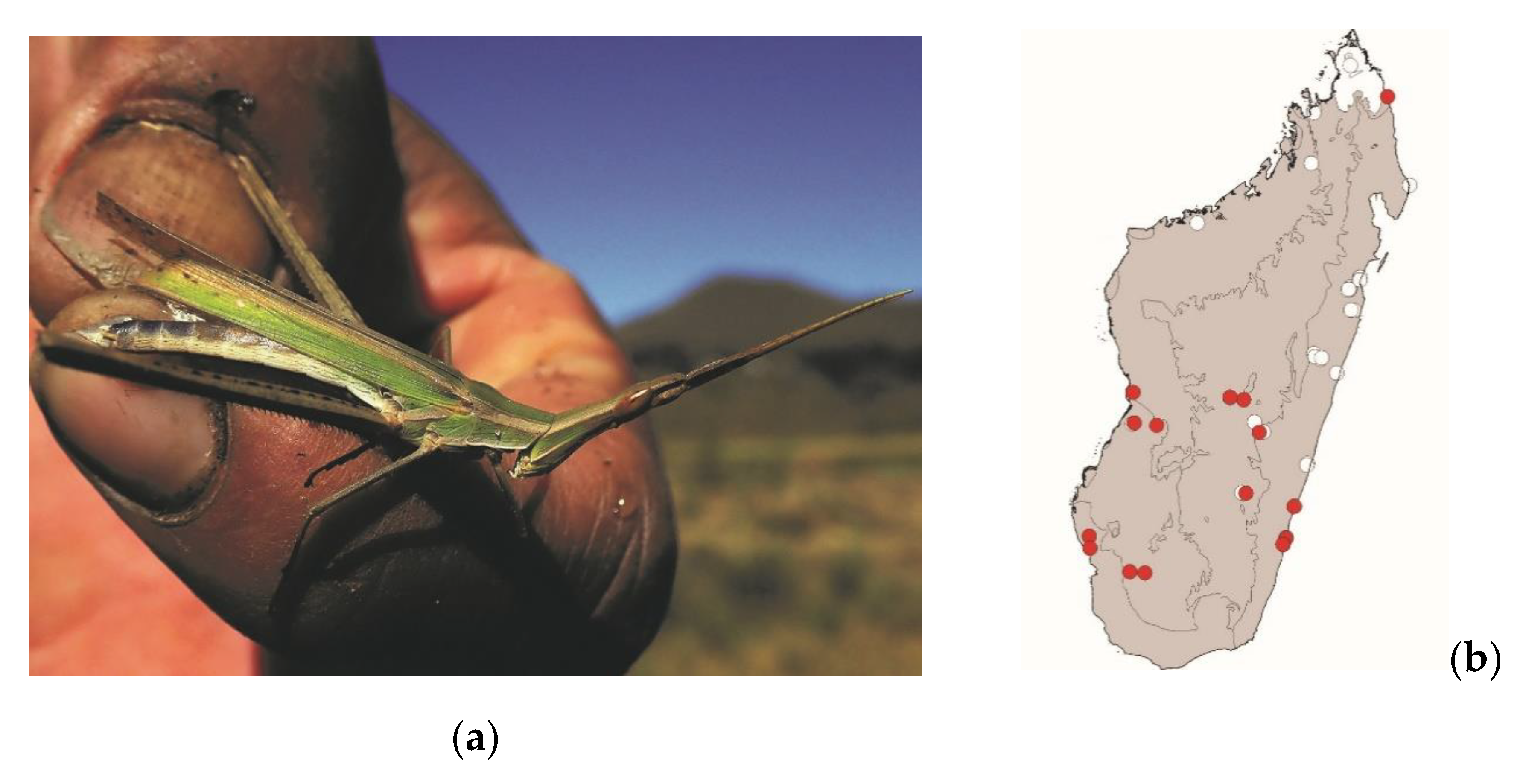
Acrida madecassa (Brancsik, 1892) (
Figure 7) and
Acrida subtilis (Burr, 1902)
These elongated grasshoppers measure 35–75 mm and are endemic to Madagascar. They are widespread in most of Madagascar but are absent from the northern tip of the island. They live in tall grassy areas, including within or near crops. These species are active by daylight and pose no threat to crops. They are consumed in the southern half of Madagascar from the coasts to the central plateau (
Figure 3). Despite their strong sexual dimorphism (males measuring 35–45 mm and females 60–75 mm), both males and females are consumed.
Aiolopus thalassinus rodericensis (Butler, 1876)
This grasshopper measures 15–25 mm. It is present in virtually every non-forested area of Madagascar except the east coast and surrounding islands. Aiolopus thalassinus rodericensis live in bare soil and grassy areas. This species is active by daylight and poses no threat to crops. It is consumed in the central part of Madagascar (from the west coast to the central plateau).
Duronia chloronota (Stål, 1876)
This grasshopper measures 20–45 mm. It is present in non-forested areas of Madagascar, except along the east coast. It lives in grassy areas. This species is active by daylight and poses no threat to crops. The consumption of this species was recorded for one single locality in west Madagascar.
Calephorus ornatus (Walker, 1870)
This small grasshopper measures 10–25 mm. It is widespread in wet grassy areas of Madagascar, particularly near rivers and lakes and in wetlands, including rice fields. This species is active by daylight and poses no threat to crops. It is consumed in the southeast of Madagascar.
Gymnobothrus madacassus (Bruner, 1910) and Gymnobothrus variabilis (Bruner, 1910)
Gymnobothrus species measure 12–25 mm. The genus Gymnobothrus is found widely across Madagascar and the Comoros. They live in bare soil and grassy areas. These species are active by daylight and pose no threat to crops. These species are consumed in central and east Madagascar.
3.3.2. Caelifera, Calliptaminae
Acorypha decisa (Walker, 1870)
This grasshopper measures 20–35 mm. It is endemic to Madagascar and widespread across most of the island save the northern tip. It lives in grassy areas. The species is active by daylight and poses no threat to crops. This species is consumed in central Madagascar between Antananarivo and Fianarantsoa.
3.3.3. Caelifera, Catantopinae
Catantopsis malagassus (Karny, 1907) and Catantopsis sacalava (Brancsik, 1892)
Catantopsis species measure 15–30 mm. The genus Catantopsis is endemic to Madagascar and the Comoros, where it is widely present. These grasshoppers live in grassy areas as well as shrublands. These species are active by daylight and pose no threat to crops. Localities where Catantopsis are consumed are scattered across Madagascar.
3.3.4. Caelifera, Cyrtacanthacridinae
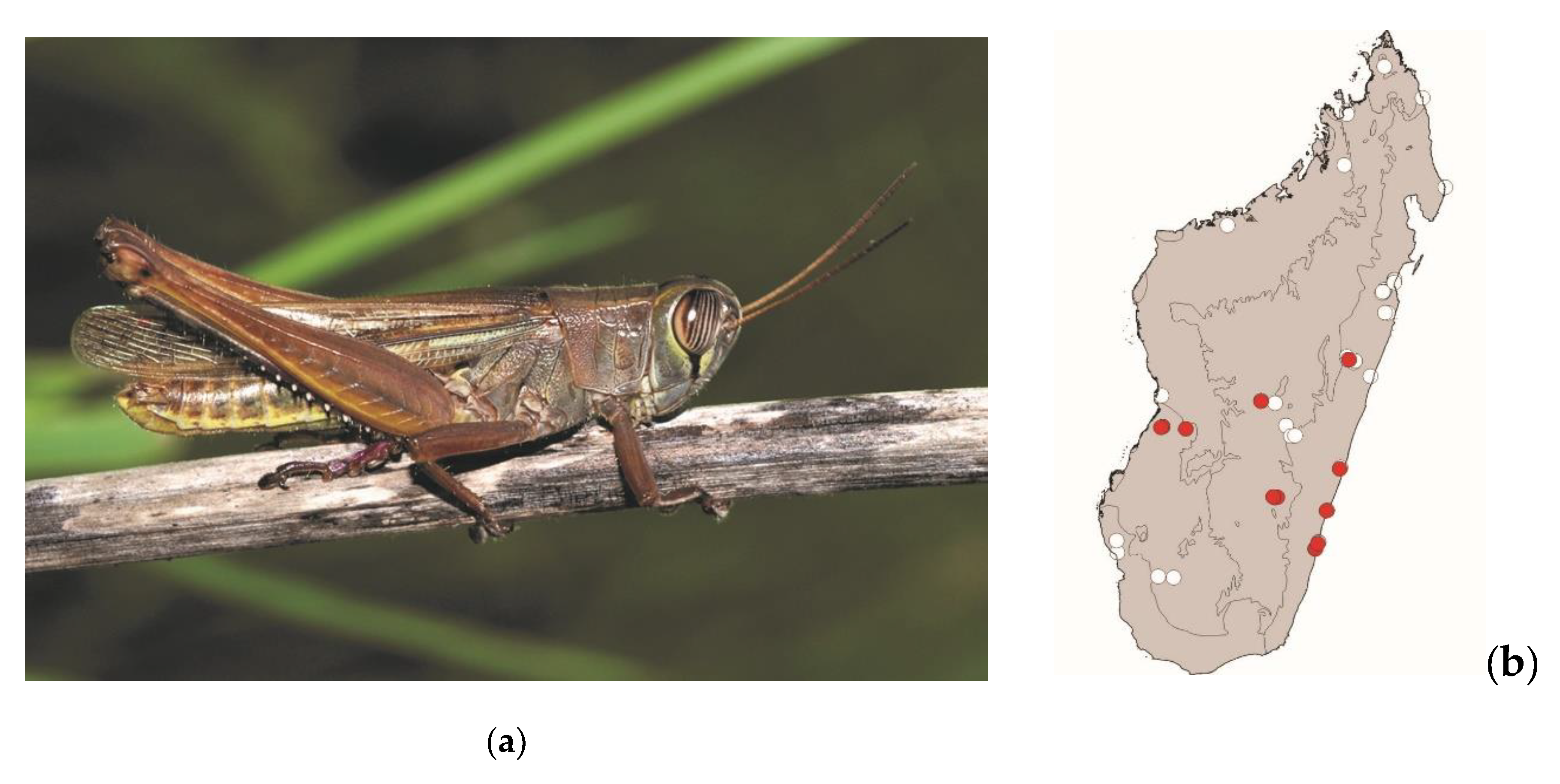
Cyrtacanthacris tatarica (Linnaeus, 1758)
This large locust measures 40–65 mm. It is widespread in Madagascar, the Comoros, and continental Africa. It lives in areas with tall grass as well as shrublands. This species is active by daylight. It represents a low threat to crops. Localities where
C. tatarica is consumed are scattered across Madagascar, in both lowlands and highlands (
Figure 8).
Rhadinacris schistocercoides (Brancsik, 1892)
This large locust measures 30–50 mm. It is endemic to Madagascar. It is widespread across the island, except along the east coast. It lives in areas with tall grass as well as shrublands. This species is active by daylight and poses no threat to crops. This species is consumed in central Madagascar, from the east to the central plateau.
Finotina radama (Brancsik, 1892)
This large locust measures 38–60 mm. It is endemic to Madagascar. It is widespread across the island except along the east coast. It lives in areas with tall grass as well as shrublands. This species is active by daylight and poses no threat to crops. The consumption of this species was recorded for one locality in central Madagascar.
Nomadacris septemfasciata (Serville, 1838)
This large plague locust measures 60–75 mm. It is widespread in continental Africa, Madagascar, and islands around Madagascar. It lives in areas with tall grass as well as shrublands. This species is active by daylight and poses a high threat to crops. Localities where N. septemfasciata is consumed are scattered across Madagascar.
3.3.5. Caelifera, Eyprepocnemidinae
Heteracris nigricornis (Saussure, 1899)
This grasshopper measures 28–50 mm. It is endemic to Madagascar. It is widespread across the island except in the north and southwest. It lives in areas with tall grass as well as shrublands. This species is active by daylight and poses no threat to crops. The consumption of this species was recorded for one locality in central Madagascar.
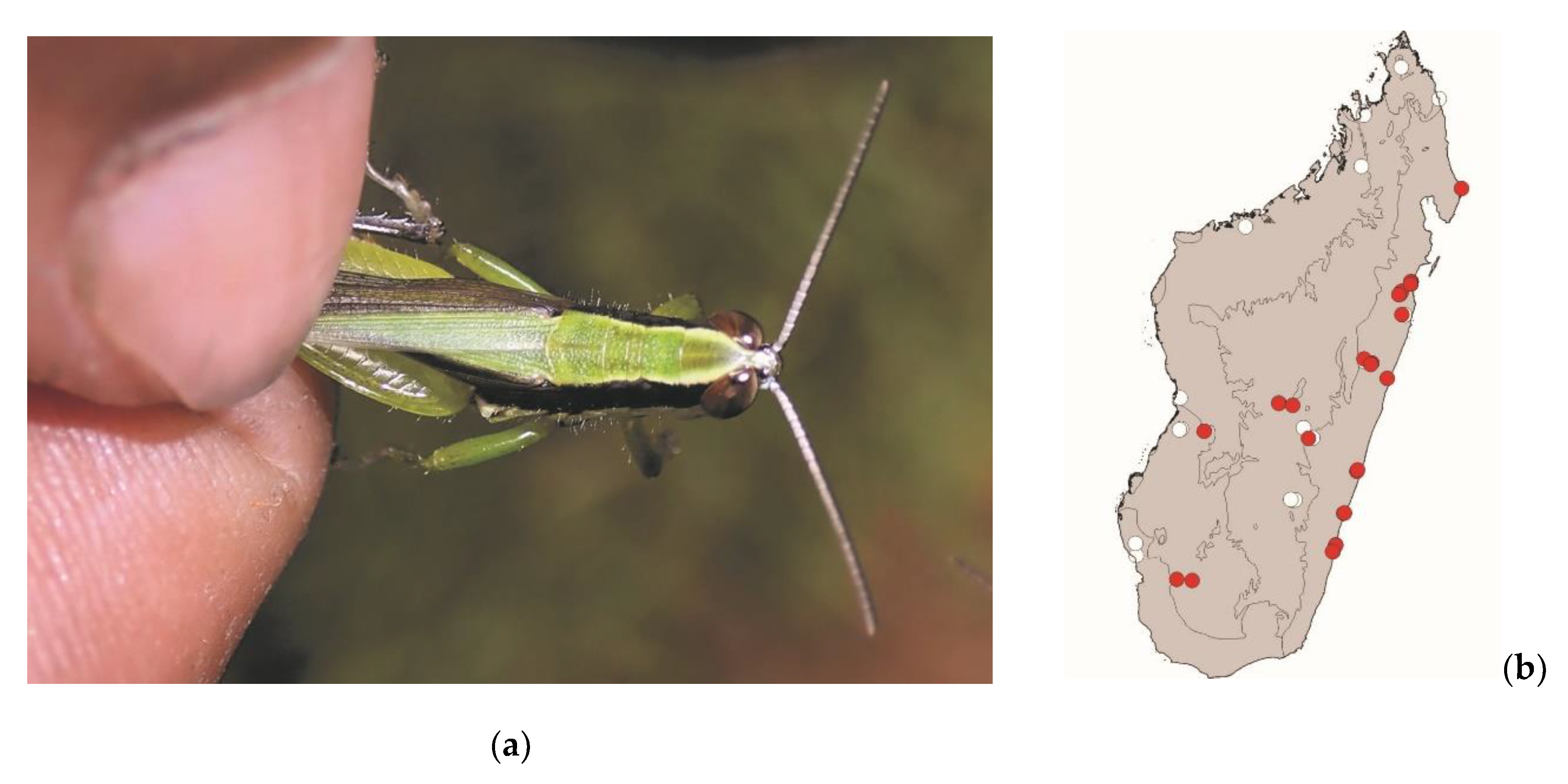
Eyprepocnemis smaragdipes (Bruner, 1910)
This grasshopper measures 20–40 mm. It is widespread in continental Africa, Madagascar, and islands around Madagascar. It lives in areas with tall grass. This species is active by daylight and poses a low threat to crops. This species is consumed in central Madagascar, from the east to the west coast (
Figure 9).
3.3.6. Caelifera, Gomphocerinae
Gelastorhinus edax (Saussure, 1888)
This grasshopper measures 22–45 mm. It is endemic and widespread in Madagascar. It lives in grassy areas. This species is active by daylight and poses no threat to crops. This species is consumed in central Madagascar, between Antananarivo and Fianarantsoa.
3.3.7. Caelifera, Oedipodinae
Paracinema tricolor (Thunberg, 1815)
This grasshopper measures 15–40 mm. It is widespread in continental Africa and Madagascar. It is among the most common grasshoppers found in wet, grassy areas of Madagascar, particularly near rivers and lakes and in wetlands, including rice fields. This species is active by daylight and poses a moderate threat to crops. This species is among the most widely consumed in Madagascar (
Figure 10).
Gastrimargus africanus (Saussure, 1888)
This relatively large locust measures 22–40 mm. It is widespread in continental Africa, Madagascar, and the southwestern Indian Ocean islands. In Madagascar it is widespread in grassy areas, including savannas maintained by fires. This species is active by daylight and poses a moderate threat to crops. Localities where
G. africanus is consumed are mostly in the central and eastern parts of Madagascar (
Figure 11).
Lemuracris longicornis (Dirsh, 1966)
This grasshopper measures > 30 mm. It is endemic to the east of Madagascar. It lives in areas with tall grass as well as shrublands. This species is active by daylight and poses no threat to crops. The consumption of this species was recorded for one locality in east Madagascar.
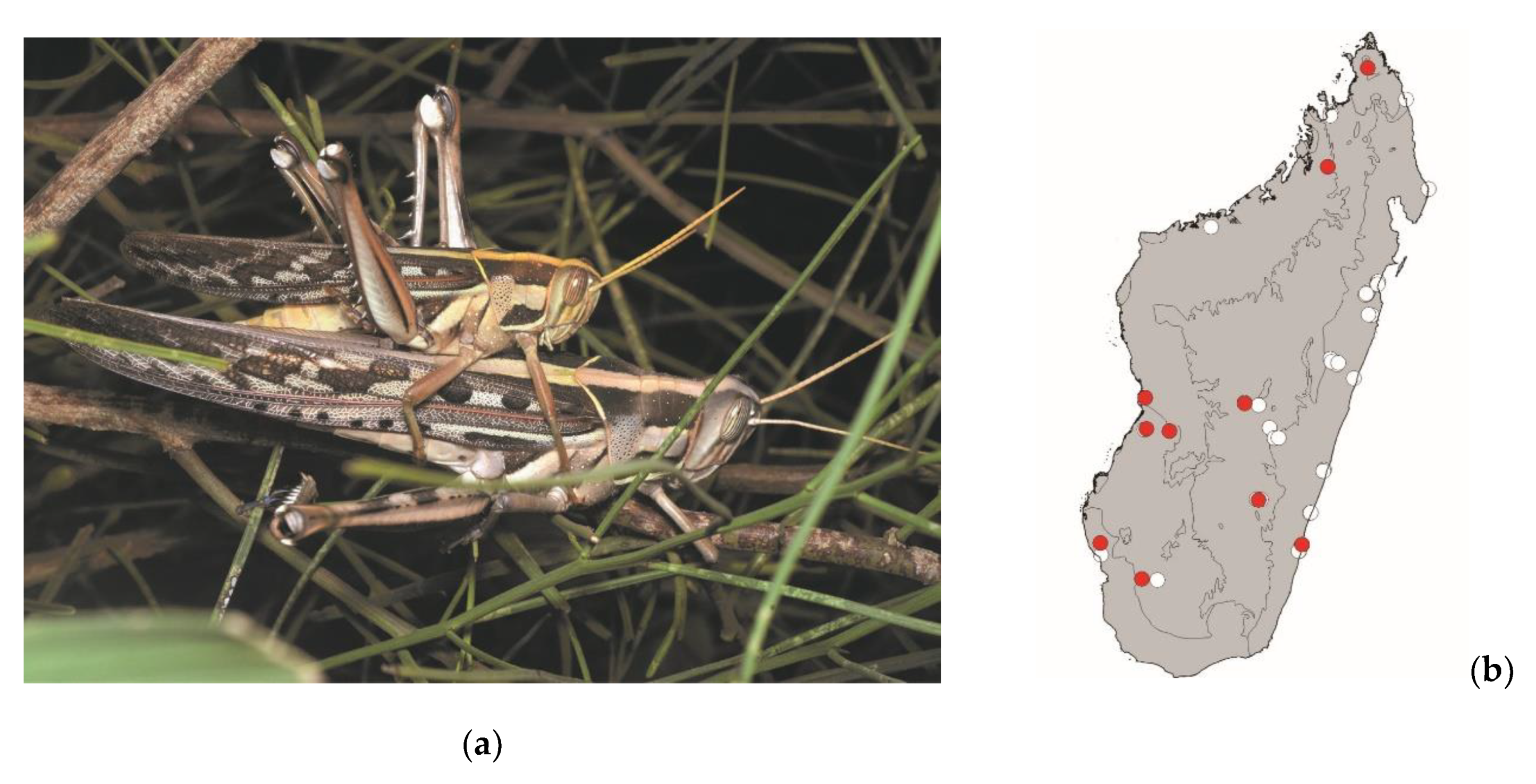
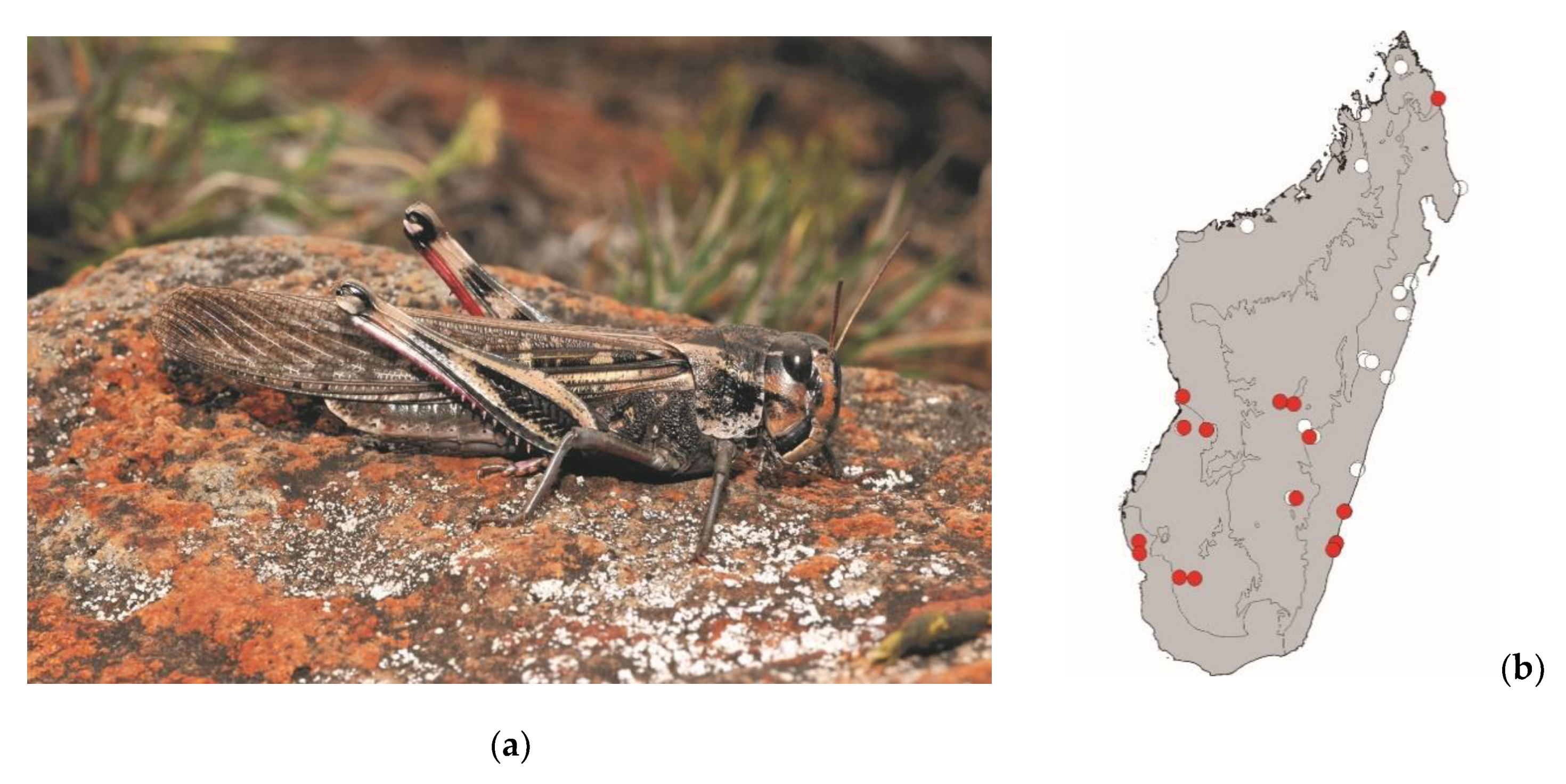
Locusta migratoria (Linnaeus, 1758)
This large plague locust measures 35–55 mm. It is widespread in continental Africa, Madagascar, and the southwestern Indian Ocean islands. It occurs in grassy areas of Madagascar and is frequently found in and around crops. This species is active by daylight and poses a high threat to crops.
Locusta migratoria is mostly consumed in the southern half of Madagascar (
Figure 12).
Oedaleus virgula (Snellen van Vollenhoven, 1869)
This grasshopper measures 13–32 mm. It is widespread in continental Africa and Madagascar. It occurs in bare soil and grassy areas of Madagascar. It is frequently found in and around crops. This species is active by daylight and poses no threat to crops. Oedaleus virgula is mostly consumed in the eastern half of Madagascar, from the central plateau to the east coast.
Trilophidia cinnabarina (Brancsik, 1892)
This small grasshopper measures 14–25 mm. It is endemic and widespread in Madagascar. It occurs in bare soil and grassy areas of Madagascar and is frequently found in and around crops. This species is active by daylight and poses no threat to crops. Consumption of T. cinnabarina was recorded for two localities in central Madagascar.
3.3.8. Caelifera, Oxyinae
Oxya hyla (Serville, 1831)
This grasshopper measures 20–31 mm. It is widespread in continental Africa and Madagascar. It is among the most common grasshoppers in wet, grassy areas of Madagascar, particularly near rivers and lakes and in wetlands, including rice fields. This species is active by daylight. It represents a low-to-moderate threat to crops. This species is consumed in the central plateau and along the east coast. This species is among the most widely consumed in Madagascar (
Figure 13).
3.3.9. Caelifera, Pyrgomorphinae
Members of Pyrgomorphidae feed on toxic plants such as Apocynaceae and sequester secondary metabolites which have significant effects on the physiology of vertebrates via cardenolide steroids and pyrrolizidine alkaloids [
25,
26,
27,
28,
29]. These Pyrgomorphidae often display aposematic coloration and some possess specialized glands or devices allowing the release of these metabolites upon disturbance.
Atractomorpha acutipennis (Guérin-Méneville, 1844)
This grasshopper measures 15-40 mm. It is widespread in continental Africa, Madagascar, and the Comoros. This grasshopper lives in areas with tall grass as well as shrublands. The species is active by daylight and poses no threat to crops. This species is very widely consumed in the central plateau and along the east coast (
Figure 14).
Atractomorpha acutipennis does not have an aposematic coloration, emits an odor less strong than that of most Pyrgomorphidae, and does not feed primarily on toxic plants.
Rubellia nigrosignata (Stål, 1875)
This grasshopper measures 15–30 mm. It is endemic to Madagascar. This grasshopper lives in areas with tall grass as well as shrublands. The species is active by daylight and poses a low threat to crops. Consumption of R. nigrosignata was recorded for one locality in east Madagascar. The insect produces an unpleasant smell when manipulated. Rubellia nigrosignata feeds on a variety of plants, though not primarily toxic ones.
3.3.10. Ensifera, Grylloidea
Gryllus sp.
The consumption of a juvenile of one large ground cricket (20 mm) was recorded for one locality on the east coast of Madagascar. These crickets are nocturnal and represent no threat to crops. Juveniles of Gryllus sp. Cannot be identified at the species level.
Modicogryllus sp.
The consumption of a medium-sized female ground cricket (10 mm) was recorded for one locality on the east coast of Madagascar. These crickets are nocturnal and represent no threat to crops. Females of Modicogryllus sp. Cannot be identified at the species level.
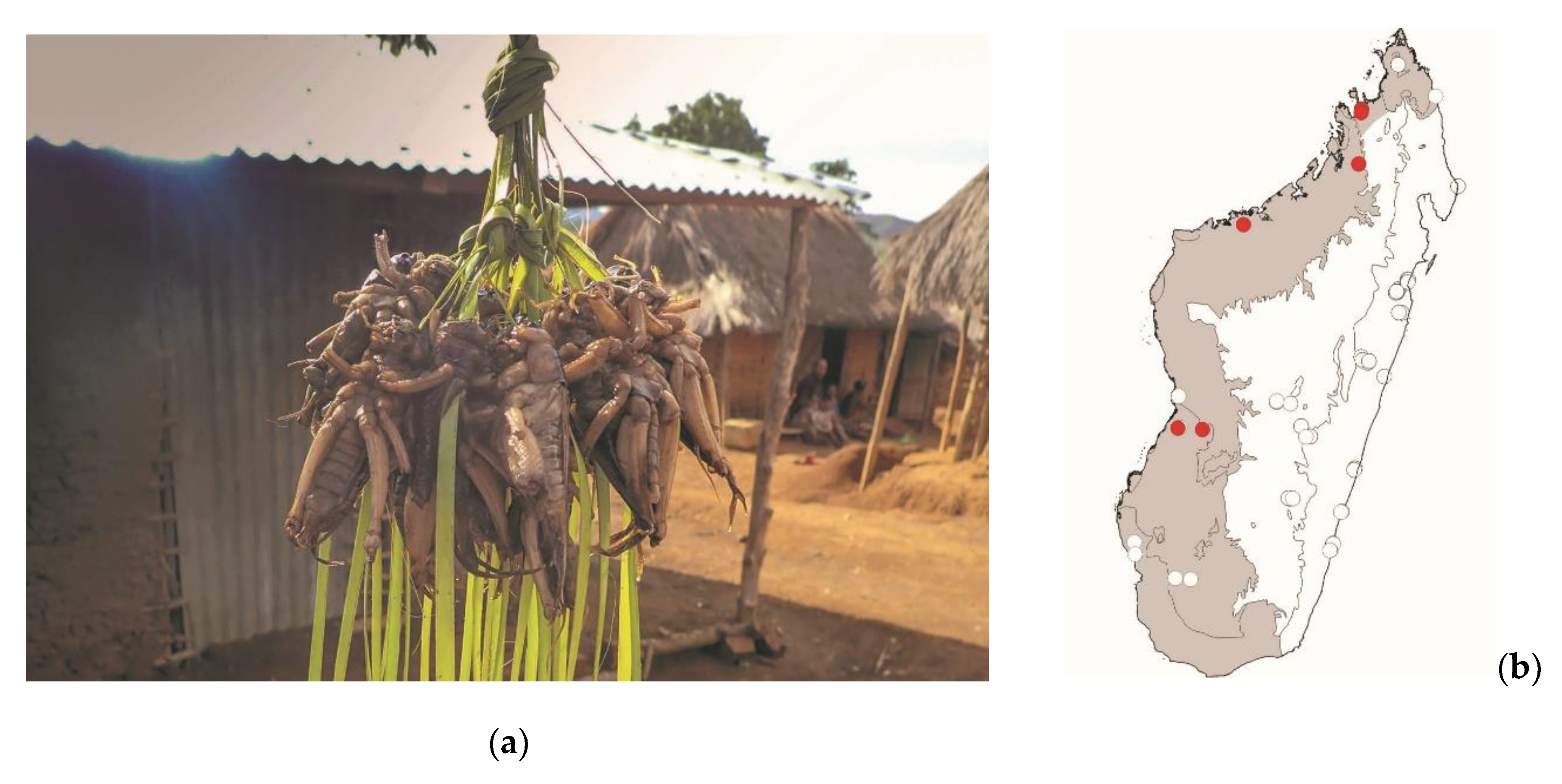
Brachytrupes membranaceus colosseus (Saussure, 1899)
These large ground crickets are widespread in sandy areas of the west of Madagascar. Several endemic species of
Brachytrupes sp. Occur in Madagascar and are in need of taxonomic review. These large crickets may represent a low threat to crops.
Brachytrupes sp. Dig burrows at the entrance of which males produce a loud call at dusk. These crickets are consumed in various localities in the west of Madagascar (
Figure 15).
Fryerius sp.
The consumption of a large female tree cricket (>20 mm) was recorded for one locality. These crickets are nocturnal and represent no threat to crops. Females of Fryerius sp. cannot be identified at the species level.
Pteronemobius malagachus (Saussure, 1877)
The consumption of a small ground cricket (>10 mm) was recorded for one locality on the east coast of Madagascar. Pteronemobius malagachus is frequently found in relatively wet grassy areas of Madagascar and its surrounding islands, including in and around rice fields. This cricket is both diurnal and nocturnal and represents no threat to crops.
3.3.11. Ensifera, Tettigoniidae
Conocephalus affinis (Redtenbacher, 1891)
This katydid measures 20–30 mm. It occurs in areas with tall grass all over Madagascar, including cultivated lands. This katydid is both diurnal and nocturnal and poses no threat to crops. Conocephalus affinis is consumed in various localities in the east of Madagascar.
Ruspolia differens and other Ruspolia sp.
Ruspolia sp. measure 30–40 mm. They occur in areas with tall grass all over Madagascar, including cultivated lands. These katydids are mostly nocturnal and may represent a low threat to crops (particularly R. differens). Ruspolia sp. are consumed in various localities from the central plateau to the east of Madagascar.
Colossopus sp.
Colossopus sp. are large nocturnal katydids living in areas with shrubs and trees. Adults can reach up to 60 mm. Some species occur in and around cultivated areas. Consumption of Colossopus sp. has been recorded in various localities from the central plateau. The collected samples were juveniles and not identifiable at the species level.
Phaneroptera sparsa (Stål, 1857) and other Phaneropterinae
Phaneroptera sparsa is a small katydid. It is widespread in tropical Africa and across Madagascar and the Comoros. It is mostly nocturnal and lives in areas with tall grass and/or shrubs, including cultivated lands. Consumption of Phaneroptera sparsa and another non-adult Phaneropterinae were recorded for two localities in the central plateau.
Odontolakis sp.
We have not recorded the consumption of
Odontolakis species in the present work, but
Odontolakis sexpunctata bush crickets have previously been recorded as edible by Randrianandrasana and Berenbaum [
17].