Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. GL Extract
2.3. Fermentation
2.4. Basic Chemical Analyses
2.5. Headspace Solid-Phase Micro-Extraction (HS-SPME-GC-MS)
2.6. Rate-All-That-Apply Sensory Evaluation of GL Wines
2.7. Data Analyses
3. Results and Discussion
3.1. Impact of GL Concentrations on Fermentation Kinetics
3.1.1. Small-Scale Fermentations
3.1.2. Larger-Scale Fermentations
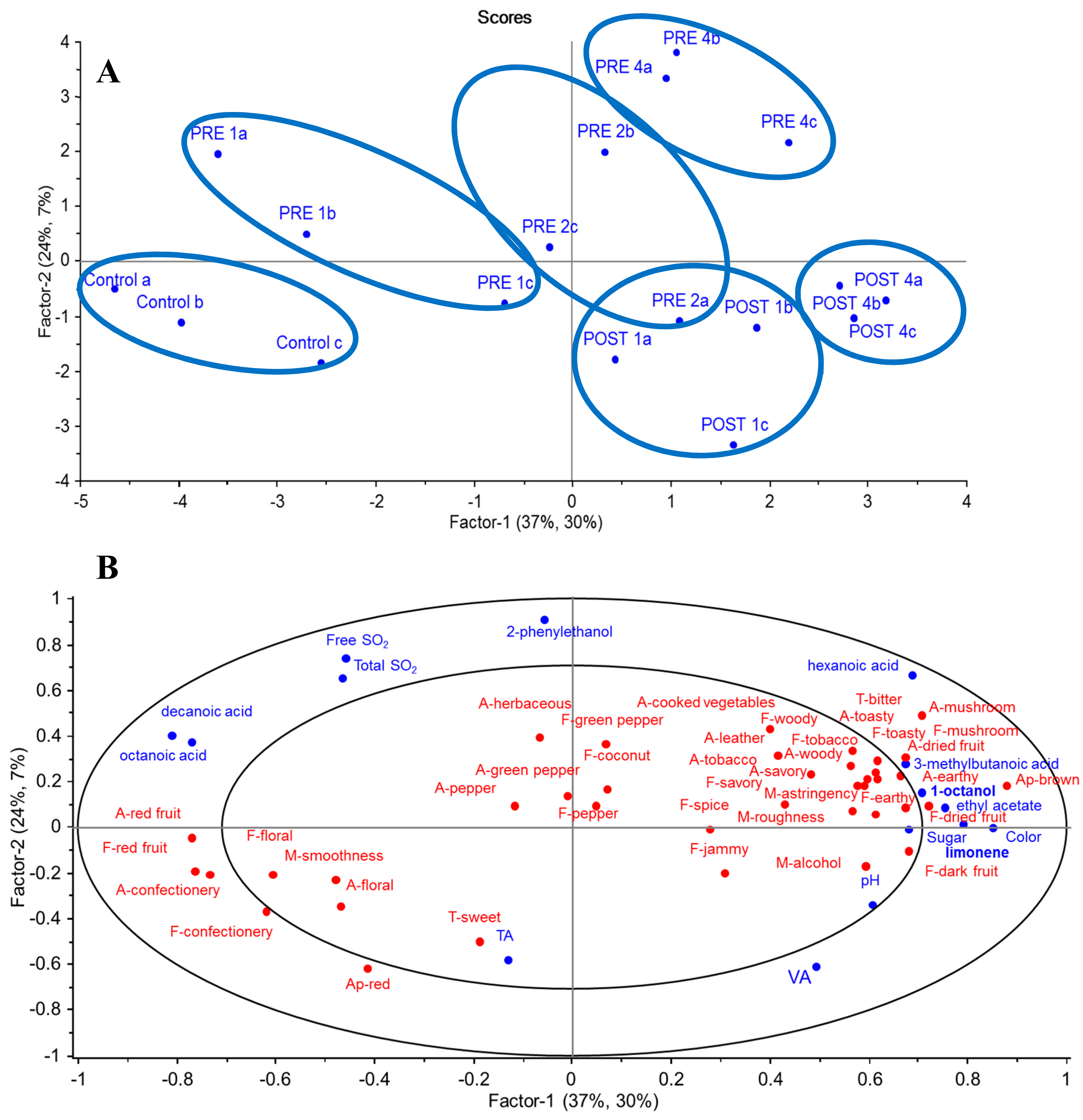
3.2. Sensory and Chemistry Profiles of GL Wines
3.3. Volatile Compounds in GL Wine
3.4. Correlation between Chemical and Sensory Data of GL Wines
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wachtel-Galor, S.; Benzie, F.F.I. Chapter 9 Ganoderma lucidum (lingzhi or reishi)—A medicinal mushroom. In Herbal Medicine: Biomolecular and Clinical Aspects, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2011; pp. 157–199. [Google Scholar]
- Moon, B.; Lo, Y.M. Conventional and novel applications of edible mushrooms in today’s food industry. J. Food Process. Preserv. 2014, 38, 2146–2153. [Google Scholar] [CrossRef]
- Jong, S.C.; Birmingham, J.M. Medicinal benefits of the mushroom Ganoderma. Adv. Appl. Microbiol. 1992, 37, 101–134. [Google Scholar] [PubMed]
- Leskosek-Cukalovic, I.; Despotovic, S.; Lakic, N.; Niksic, M.; Nedovic, V.; Tesevic, V. Ganoderma lucidum—Medical mushroom as a raw material for beer with enhanced functional properties. Food Res. Int. 2010, 43, 2262–2269. [Google Scholar] [CrossRef]
- Zhou, X.; Su, K.; Zhang, Y. Applied modern biotechnology for cultivation of Ganoderma and development of their products. Appl. Microbiol. Biotechnol. 2012, 93, 941–963. [Google Scholar] [CrossRef]
- Cheung, W.M.W.; Hui, W.S.; Chu, P.W.K.; Chiu, S.W.; Ip, N.Y. Ganoderma extract activates MAP kinases and induces the neuronal differentiation of rat pheochromocytoma PC12 cells. FEBS Lett. 2000, 486, 291–296. [Google Scholar] [CrossRef]
- Gao, Y.; Zhou, S.; Huang, M.; Xu, A. Antibacterial and antiviral value of the genus Ganoderma P. Karst. Species (Aphyllophoromycetideae): A review. Int. J. Med. Mushrooms 2003, 5, 235–246. [Google Scholar] [CrossRef]
- Chang, S.; Miles, P.G. Mushrooms: Cultivation, Nutritional Value, Medicinal, Effect, and Environmental Impact, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 357–371. [Google Scholar]
- Paterson, R.R.M. Ganoderma—A therapeutic fungal biofactory. Phytochemistry 2006, 67, 1985–2001. [Google Scholar] [CrossRef]
- Bishop, K.S.; Kao, C.H.; Xu, Y.; Glucina, M.P.; Paterson, R.R.; Ferguson, L.R. From 2000 years of Ganoderma lucidum to recent developments in nutraceuticals. Phytochemistry 2015, 114, 56–65. [Google Scholar] [CrossRef]
- Bai, W.; Guo, X.; Ma, L.; Guo, L.; Lin, J. Chemical composition and sensory evaluation of fermented tea with medicinal mushrooms. Indian J. Microbiol. 2013, 53, 70–76. [Google Scholar] [CrossRef][Green Version]
- Yang, H.; Zhang, L. Changes in some components of soymilk during fermentation with the basidiomycete Ganoderma lucidum. Food Chem. 2009, 112, 1–5. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, D.H.; Lee, S.H.; Choi, S.Y.; Lee, J.S. Effect of Ganoderma lucidum on the quality and functionality of Korean traditional rice wine, Yakju. J. Biosci. Bioeng. 2003, 97, 24–28. [Google Scholar] [CrossRef]
- Pecić, S.; Veljović, M.; Despotović, S.; Tešević, V.; Nikićević, N.; Nikšić, M. The sensory properties of special brandy with Ganoderma lucidum. In 7th International Congress of Food Technologists, Biotechnologists and Nutritionists; Medić, H., Ed.; Croatian Society of Food Technologists, Biotechnologists and Nutritionists: Opatija, Croatia, 2015; pp. 89–92. [Google Scholar]
- Nguyen, A.N.H.; Johnson, T.E.; Jeffery, D.W.; Danner, L.; Bastian, S.E.P. A cross-cultural examination of Australian, Chinese and Vietnamese consumers’ attitudes towards a new Australian wine product containing Ganoderma lucidum extract. Food Res. Int. 2018, 115, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Danner, L.; Crump, A.M.; Croker, A.; Gambetta, J.M.; Johnson, T.E.; Bastian, S.E. Comparison of Rate-All-That-Apply and descriptive analysis for the sensory profiling of wine. Am. J. Enol. Vitic. 2018, 69, 12–21. [Google Scholar] [CrossRef]
- Long, D.; Wilkinson, K.L.; Taylor, D.K.; Jiranek, V. Novel wine yeast for improved utilisation of proline during fermentation. Fermentation 2018, 4, 10. [Google Scholar] [CrossRef]
- Patynowski, R.J.; Jiranek, V.; Markides, A.J. Yeast viability during fermentation and sur lie ageing of a defined medium and subsequent growth of Oenococcus oeni. Aust. J. Grape Wine Res. 2002, 8, 62–69. [Google Scholar] [CrossRef]
- Iland, P.; Bruer, N.; Edwards, G.; Weeks, S.; Wilkes, E. Chemical Analysis of Grapes and Wine: Techniques and Concepts; Patrick Iland Wine Promotions: Campbelltown, Australia, 2004. [Google Scholar]
- Wang, J.; Capone, D.L.; Wilkinson, K.L.; Jeffery, D.W. Chemical and sensory profiles of rosé wines from Australia. Food Chem. 2016, 196, 682–693. [Google Scholar] [CrossRef]
- Copper, A.W.; Johnson, T.E.; Danner, L.; Bastian, S.E.; Collins, C. Preliminary sensory and chemical profiling of Cypriot wines made from indigenous grape varieties Xynisteri, Maratheftiko and Giannoudhi and acceptability to Australian consumers. OENO One 2019, 53. [Google Scholar] [CrossRef]
- Crump, A.M.; Johnson, T.E.; Wilkinson, K.L.; Bastian, S.E. Influence of oak maturation regimen on composition, sensory properties, quality, and consumer acceptability of Cabernet Sauvignon wines. J. Agric. Food Chem. 2015, 63, 1593–1600. [Google Scholar] [CrossRef]
- MacFie, H.J.; Bratchell, N.; Greenhoff, K.; Vallis, L.V. Designs to balance the effect of order of presentation and first-order carry-over effects in hall tests. J. Sens. Stud. 1989, 4, 129–148. [Google Scholar] [CrossRef]
- Skalicka-Wozniak, K.; Szypowski, J.; Los, R.; Siwulski, M.; Sobieralski, K.; Glowniak, K.; Malm, A. Evaluation of polysaccharides content in fruit bodies and their antimicrobial activity of four Ganoderma lucidum (W curt.: Fr.) p. Karst. Strains cultivated on different wood type substrates. Acta Soc. Botanicorum Poloniae 2012, 81, 17–21. [Google Scholar] [CrossRef]
- Heleno, S.A.; Ferreira, I.C.F.R.; Esteves, A.P.; Ćirić, A.; Glamočlija, J.; Martins, A.; Soković, M.; Queiroz, M.J.R.P. Antimicrobial and demelanizing activity of Ganoderma lucidum extract, p-hydroxybenzoic and cinnamic acids and their synthetic acetylated glucuronide methyl esters. Food Chem. Toxicol. 2013, 58, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Boh, B.; Berovic, M.; Zhang, J.; Zhi-Bin, L. Ganoderma lucidum and its pharmaceutically active compounds. Biotechnol. Ann. Rev. 2007, 13, 265–301. [Google Scholar]
- Wasser, S.P. Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl. Microbiol. Biotechnol. 2002, 60, 258–274. [Google Scholar] [PubMed]
- Sun-Waterhouse, D.; Wadhwa, S.S. Industry-relevant approaches for minimising the bitterness of bioactive compounds in functional foods: A review. Food Bioprocess Technol. 2013, 6, 607–627. [Google Scholar] [CrossRef]
- Kinghorn, A.D.; Kaneda, N.; Baek, N.I.; Kennelly, E.J.; Soejarto, D.D. Noncariogenic intense natural sweeteners. Med. Res. Rev. 1998, 18, 347–360. [Google Scholar] [CrossRef]
- Pecić, S.; Nikićević, N.; Veljović, M.; Jadranin, M.; Tešević, V.; Belović, M.; Nikšić, M. The influence of extraction parameter on physicochemical properties of special grain brandies with Ganoderma lucidum. Chem. Ind. Chem. Eng. Q. 2016, 22, 181–189. [Google Scholar] [CrossRef]
- Bastian, S.E.P.; Collins, C.; Johnson, T.E. Understanding consumer preferences for Shiraz wine and Cheddar cheese pairings. Food Qual. Prefer. 2010, 21, 668–678. [Google Scholar] [CrossRef]
- Wang, J.; Capone, D.L.; Wilkinson, K.L.; Jeffery, D.W. Rosé wine volatile composition and the preferences of Chinese wine professionals. Food Chem. 2016, 202, 507–517. [Google Scholar] [CrossRef]
- Williamson, P.O.; Robichaud, J.; Francis, I.L. Comparison of Chinese and Australian consumers’ liking responses for red wines. Aust. J. Grape Wine Res. 2012, 18, 256–267. [Google Scholar] [CrossRef]
- Kontoudakis, N.; Gonzalez, E.; Gil, M.; Esteruelas, M.; Fort, F.; Canals, J.M.; Zamora, F. Influence of wine pH on changes in color and polyphenol composition induced by micro-oxygenation. J. Agric. Food Chem. 2011, 59, 1974–1984. [Google Scholar] [CrossRef]
- Harbertson, J.F.; Keller, M. Rootstock effects on deficit-irrigated winegrapes in a dry climate: Grape and wine composition. Am. J. Enol. Vitic. 2011, 63, 40–48. [Google Scholar] [CrossRef]
- Sims, C.; Bates, R. Effects of skin fermentation time on the phenols, anthocyanins, ellagic acid sediment, and sensory characteristics of a red Vitis rotundifolia wine. Am. J. Enol. Vitic. 1994, 45, 56–62. [Google Scholar]
- Henderson, J.P. About Wine; Thompson Delmar Learning: Cifton Park, NY, USA, 2006. [Google Scholar]
- Yu, P.; Pickering, G.J. Ethanol difference thresholds in wine and the influence of mode of evaluation and wine style. Am. J. Enol. Vitic. 2008, 59, 146–152. [Google Scholar]
- García-Marino, M.; Escudero-Gilete, M.L.; Heredia, F.J.; Escribano-Bailón, M.T.; Rivas-Gonzalo, J.C. Color-copigmentation study by tristimulus colorimetry (CIELAB) in red wines obtained from Tempranillo and Graciano varieties. Food Res. Int. 2013, 51, 123–131. [Google Scholar] [CrossRef]
- Bisson, L.F. Stuck and sluggish fermentations. Am. J. Enol. Vitic. 1999, 50, 107–119. [Google Scholar]
- Chen, Z.; Yang, Z.; Gu, Z. Determination of volatile flavor compounds in Ganoderma lucidum by HS-SPME-GC-MS. Food Res. Dev. 2010, 31, 132–135. [Google Scholar]
- Taskin, H.; Kafkas, E.; Çakiroglu, Ö.; Büyükalaca, S. Determination of volatile aroma compounds of Ganoderma lucidum by gas chromatography mass spectrometry (HS-GC/MS). Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 353–355. [Google Scholar] [CrossRef]
- Darici, M.; Cabaroglu, T.; Ferreira, V.; Lopez, R. Chemical and sensory characterisation of the aroma of çalkarası rosé wine. Aust. J. Grape Wine Res. 2014, 20, 340–346. [Google Scholar] [CrossRef]
- Vilanova, M.; Genisheva, Z.; Masa, A.; Oliveira, J.M. Correlation between volatile composition and sensory properties in Spanish Albariño wines. Microchem. J. 2010, 95, 240–246. [Google Scholar] [CrossRef]
- Ferreira, V.; San Juan, F.; Escudero, A.; Culleré, L.; Fernandez-Zurbano, P.; Saenz-Navajas, M.P.; Cacho, J. Modeling quality of premium Spanish red wines from gas chromatography−olfactometry data. J. Agric. Food Chem. 2009, 57, 7490–7498. [Google Scholar] [CrossRef]
- Longo, R.; Blackman, J.W.; Antalick, G.; Torley, P.J.; Rogiers, S.Y.; Schmidtke, L.M. Volatile and sensory profiling of shiraz wine in response to alcohol management: Comparison of harvest timing versus technological approaches. Food Res. Int. 2018, 109, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.M.; Geue, J.P.; Holt, H.E.; Pearson, W.P.; Jeffery, D.W.; Francis, I.L. Characterization of the key aroma compounds in Shiraz wine by quantitation, aroma reconstitution, and omission studies. J. Agric. Food Chem. 2014, 62, 4528–4536. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Gao, J.; Qian, M.; Li, H. Characterization of the key aroma compounds in Chinese Syrah wine by gas chromatography-olfactometry-mass spectrometry and aroma reconstitution studies. Molecules 2017, 22, 1045. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Bartowsky, E.J.; Henschke, P.A.; Pretorius, I.S. Yeast and bacterial modulation of wine aroma and flavour. Aust. J. Grape Wine Res. 2005, 11, 139–173. [Google Scholar] [CrossRef]
- Robinson, A.L.; Adams, D.O.; Boss, P.K.; Heymann, H.; Solomon, P.S.; Trengove, R.D. The relationship between sensory attributes and wine composition for Australian Cabernet Sauvignon wines. Aust. J. Grape Wine Res. 2011, 17, 327–340. [Google Scholar] [CrossRef]


| Treatment Samples | pH | Titratable Acidity (g/L) | Ethanol (% v/v) | Volatile Acidity (g/L) | Free SO2 (mg/L) | Total SO2 (mg/L) | Chroma C*(D650) | a* (D650) | b* (D650) | Residual Sugar (g/L) |
|---|---|---|---|---|---|---|---|---|---|---|
| Control a | 3.91 gh | 4.63 b | 12.43 gh | 0.25 f | 48.53 f | 112.53 j | 10.49 m | 8.89 k | 5.58 m | 0.68 hi |
| Control b | 3.91 gh | 4.56 c | 12.75 f | 0.25 f | 48.53 f | 123.73 fg | 10.54 l | 8.85 l | 5.73 l | 0.61 j |
| Control c | 3.93 efg | 4.73 a | 12.79 ef | 0.32 c | 51.20 e | 121.60 h | 10.59 k | 8.81 m | 5.87 j | 0.68 hi |
| PRE 1a | 3.84 i | 4.16 lm | 12.91 e | 0.25 f | 62.40 a | 134.4 a | 11.70 i | 10.11 f | 5.89 j | 0.67 ij |
| PRE 1b | 3.90 h | 4.21 jk | 12.30 hi | 0.25 f | 51.73 de | 117.33 i | 11.67 j | 10.16 e | 5.75 l | 0.74 h |
| PRE 1c | 4.02 a | 4.40 e | 12.73 f | 0.32 c | 52.80 d | 128.53 c | 11.68 i | 10.14 ef | 5.82 k | 0.67 ij |
| PRE 2a | 3.97 b | 4.58 c | 13.32 c | 0.39 a | 48.00 f | 122.13 gh | 11.81 h | 9.80 h | 6.62 h | 0.95 g |
| PRE 2b | 3.93 efg | 4.12 m | 13.36 bc | 0.25 f | 49.06 f | 124.26 ef | 11.82 h | 9.82 gh | 6.59 hi | 1.01 g |
| PRE 2c | 3.92 efgh | 4.29 hi | 13.16 d | 0.25 f | 52.80 d | 125.86 de | 11.82 h | 9.84 g | 6.56 i | 0.98 g |
| PRE 4a | 3.92 fgh | 4.18 kl | 12.25 i | 0.25 f | 56.53 c | 127.46 cd | 12.60 e | 9.55 j | 8.22 c | 1.54 d |
| PRE 4b | 3.93 efg | 4.36 ef | 12.50 g | 0.25 f | 56.53 c | 130.66 b | 12.56 f | 9.59 ij | 8.12 d | 1.68 c |
| PRE 4c | 3.96 bc | 4.48 d | 12.76 f | 0.29 e | 58.66 b | 133.86 a | 12.52 g | 9.62 i | 8.02 e | 1.56 d |
| POST 1a | 3.96 bdc | 4.33 fgh | 13.44 bc | 0.29 e | 40.53 gh | 104.53 k | 12.62 d | 10.35 c | 7.22 f | 1.30 ef |
| POST 1b | 3.96 bcd | 4.25 ij | 13.48 b | 0.31 cd | 40.00 h | 100.80 l | 12.58 e | 10.34 cd | 7.17 g | 1.25 f |
| POST 1c | 3.94 cde | 4.63 b | 13.83 a | 0.34 b | 36.26 i | 90.66 n | 12.59 e | 10.32 d | 7.22 f | 1.32 e |
| POST 4a | 3.97 b | 4.34 fg | 13.48 b | 0.30 de | 36.26 i | 96.53 m | 14.62 b | 11.23 b | 9.37 a | 2.96 a |
| POST 4b | 3.98 b | 4.32 gh | 13.39 bc | 0.31 cd | 41.60 g | 101.86 l | 14.43 c | 11.21 b | 9.08 b | 2.77 b |
| POST 4c | 3.94 def | 4.56 b | 13.45 bc | 0.35 b | 41.60 g | 82.133 o | 14.75 a | 11.62 a | 9.09 b | 2.75 b |
| Compound | Control | PRE 1 | PRE 2 | PRE 4 | POST 1 | POST 4 | Sig | Aroma Detection Threshold (μg/L) |
|---|---|---|---|---|---|---|---|---|
| ethyl acetate | 3793.6 ± 2330.8b | 7018.5 ± 604.1ab | 8252.6 ± 1258.8a | 7755.0 ± 998.4a | 7374.0 ± 1810.3ab | 8016.4 ± 561.9a | 0.021 | 15,000 ** a |
| ethyl butanoate | 26.1 ± 4.6 | 23.9 ± 2.2 | 25.9 ± 2.1 | 26.3 ± 1.3 | 29.4 ± 2.2 | 27.9 ± 2.8 | 0.292 | 20 ** a |
| ethyl-2-methylbutanoate | 0.5 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.1 | 0.6 ± 0.1 | 0.5 ± 0.0 | 0.572 | 1 ** a |
| ethyl 3-methylbutanoate | 0.6 ± 0.1 | 0.6 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.1 | 0.7 ± 0.0 | 0.653 | 3 ** a |
| 3-methylbutyl acetate | 614.1 ± 28.3 | 609.5 ± 124.1 | 636.6 ± 37.5 | 502.2 ± 6.9 | 563.3 ± 69.0 | 539.0 ± 41.9 | 0.156 | 30 ** a |
| ethyl hexanoate | 60.3 ± 3.2 | 65.6 ± 3.2 | 60.8 ± 2.0 | 58.0 ± 6.3 | 66.2 ± 10.4 | 58.0 ± 5.6 | 0.381 | 14 ** b |
| hexyl acetate | 32.3 ± 2.4 | 30.8 ± 5.6 | 29.0 ± 6.3 | 26.5 ± 3.6 | 29.9 ± 7.4 | 25.7 ± 2.7 | 0.603 | 670 ** a |
| ethyl lactate | 5186.6 ± 494.4 | 5165.3 ± 551.4 | 5637.1 ± 565.7 | 5458.7 ± 1000.1 | 5742.9 ± 1164.8 | 5776.8 ± 366.6 | 0.845 | 146,000 ** a |
| ethyl octanoate | 15.7 ± 0.9 | 15.8 ± 1.0 | 16.8 ± 1.6 | 18.0 ± 0.2 | 16.5 ± 1.8 | 15.8 ± 1.2 | 0.235 | 20 ** b |
| ethyl decanoate | 28.5 ± 4.5 | 33.2 ± 3.9 | 41.2 ± 10.0 | 46.8 ± 6.2 | 35.3 ± 10.9 | 31.8 ± 6.2 | 0.093 | 200 ** b |
| diethyl succinate | 1.4 ± 0.0 | 1.6 ± 0.0 | 12.2 ± 1.9 | 19.0 ± 13.6 | 10.8 ± 7.4 | 11.0 ± 3.3 | 0.052 | 1,250,000 ** a |
| 2-phenylethyl acetate | 21.2 ± 1.6 | 21.2 ± 5.1 | 20.8 ± 4.3 | 20.8 ± 3.2 | 16.4 ± 1.7 | 17.0 ± 1.5 | 0.277 | 250 ** a |
| 1-propanol | 9029.4 ± 732.8 | 8357.8 ± 570.6 | 7802.2 ± 770.4 | 8692.7 ± 616.2 | 9394.5 ± 277.2 | 9215.7 ± 589.7 | 0.066 | 500 ** b |
| 2-methyl-1-propanol | 2362.3 ± 161.8 | 2258.3 ± 231.4 | 2190.4 ± 24.5 | 2102.6 ± 46.6 | 2083.8 ± 57.4 | 2136.8 ± 30.3 | 0.107 | 40,000 ** b |
| 1-butanol | 64.3 ± 10.5 | 60.3 ± 11.2 | 67.3 ± 9.6 | 50.8 ± 8.5 | 71.5 ± 8.1 | 61.1 ± 13.3 | 0.29 | 150,000 * a |
| 3-methyl-1-butanol | 17185.2 ± 765.4 | 16411.5 ± 351.2 | 17774.1 ± 515.1 | 16513.6 ± 2882.3 | 17612.4 ± 256.5 | 16733.9 ± 560.0 | 0.69 | 30,000 ** a |
| 1-hexanol | 199.5 ± 13.8 | 194.7 ± 24.7 | 199.8 ± 14.9 | 193.7 ± 14.6 | 209.7 ± 7.0 | 211.1 ± 4.3 | 0.598 | 8000 ** a |
| Linalool | 6.2 ± 0.6 | 6.7 ± 0.7 | 6.7 ± 0.3 | 7.0 ± 1.2 | 7.6 ± 0.9 | 6.8 ± 0.4 | 0.427 | 15 ** a |
| 1-octanol | 2.4 b ± 0.1 | 2.4 ± 0.2ab | 2.6 ± 0.1ab | 2.8 ± 0.2a | 2.6 ± 0.0ab | 2.6 ± 0.1ab | 0.041 | 0.7 ** a |
| α-terpineol | 5.0 ± 0.4 | 5.2 ± 0.1 | 5.6 ± 0.8 | 6.1 ± 0.9 | 5.4 ± 0.3 | 5.1 ± 0.5 | 0.267 | 250 ** b |
| benzyl alcohol | 178.3 ± 18.7 | 179.5 ± 23.3 | 173.8 ± 22.7 | 166.1 ± 18.4 | 178.2 ± 11.7 | 159.9 ± 8.9 | 0.715 | 200,000 *** a |
| 2-phenylethanol | 1443.4 ± 123.0b | 1587 ± 180.3ab | 1545.8 ± 250.5b | 1985.5 ± 162.7a | 1356.9 ± 21.6b | 1354.5 ± 26.4b | 0.002 | 14,000 ** b |
| Limonene | 0.4 ± 0.0b | 0.5 ± 0.0a | 0.6 ± 0.0a | 0.6 ± 0.0a | 0.6 ± 0.1a | 0.6 ± 0.1a | 0.034 | 15 ** a |
| Nonanal | 2.8 ± 0.0 | 2.8 ± 0.0 | 2.8 ± 0.1 | 2.9 ± 0.1 | 2.9 ± 0.1 | 2.8 ± 0.1 | 0.461 | 2.5 ** a |
| 3-methylbutanoic acid | 45.6 ± 0.6bc | 44.1 ± 2.2c | 47.3 ± 0.7bc | 50.7 ± 1.1a | 47.5 ± 0.7ab | 47.4 ± 1.2bc | 0.001 | 33 * a |
| hexanoic acid | 702.9 ± 29.9c | 788.8 ± 13.1b | 849.5 ± 46.4ab | 887.5 ± 12.0a | 779.1 ± 43.6bc | 848.0 ± 4.8ab | <0.001 | 420 ** b |
| octanoic acid | 1448.5 ± 87.3a | 1392.1 ± 185.5a | 1301.6 ± 112.0ab | 1240.0 ± 67.6ab | 1072.4 ± 71.6b | 1181.0 ± 48.5ab | 0.009 | 500 ** b |
| decanoic acid | 375.7 ± 33.1a | 362.5 ± 13.2a | 330.7 ± 28.0abc | 341.0 ± 13.1ab | 293.3 ± 24.7bc | 271.4 ± 26.2c | 0.001 | 1000 ** b |
| β-damascenone | 24.1 ± 7.4 | 20.7 ± 6.2 | 28.7 ± 2.9 | 21.8 ± 5.2 | 22.9 ± 4.0 | 25.7 ± 6.3 | 0.561 | 0.05 ** a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, A.N.H.; Capone, D.L.; Johnson, T.E.; Jeffery, D.W.; Danner, L.; Bastian, S.E.P. Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract. Foods 2019, 8, 538. https://doi.org/10.3390/foods8110538
Nguyen ANH, Capone DL, Johnson TE, Jeffery DW, Danner L, Bastian SEP. Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract. Foods. 2019; 8(11):538. https://doi.org/10.3390/foods8110538
Chicago/Turabian StyleNguyen, Anh N.H., Dimitra L. Capone, Trent E. Johnson, David W. Jeffery, Lukas Danner, and Susan E.P. Bastian. 2019. "Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract" Foods 8, no. 11: 538. https://doi.org/10.3390/foods8110538
APA StyleNguyen, A. N. H., Capone, D. L., Johnson, T. E., Jeffery, D. W., Danner, L., & Bastian, S. E. P. (2019). Volatile Composition and Sensory Profiles of a Shiraz Wine Product Made with Pre- and Post-Fermentation Additions of Ganoderma lucidum Extract. Foods, 8(11), 538. https://doi.org/10.3390/foods8110538