Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison Between Microgreens and Baby Greens
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growth Conditions
2.2. Harvesting and Yield Assessment
2.3. Analysis
2.3.1. Total Chlorophyll and Carotenoids
2.3.2. Phenolic Index and Anthocyanins Concentration
2.3.3. Nitrate
2.3.4. Mineral Composition
2.4. Contribution to Mineral Dietary Intake and Health Risk Assessment
2.5. Statistical Analysis
3. Results
3.1. Yield
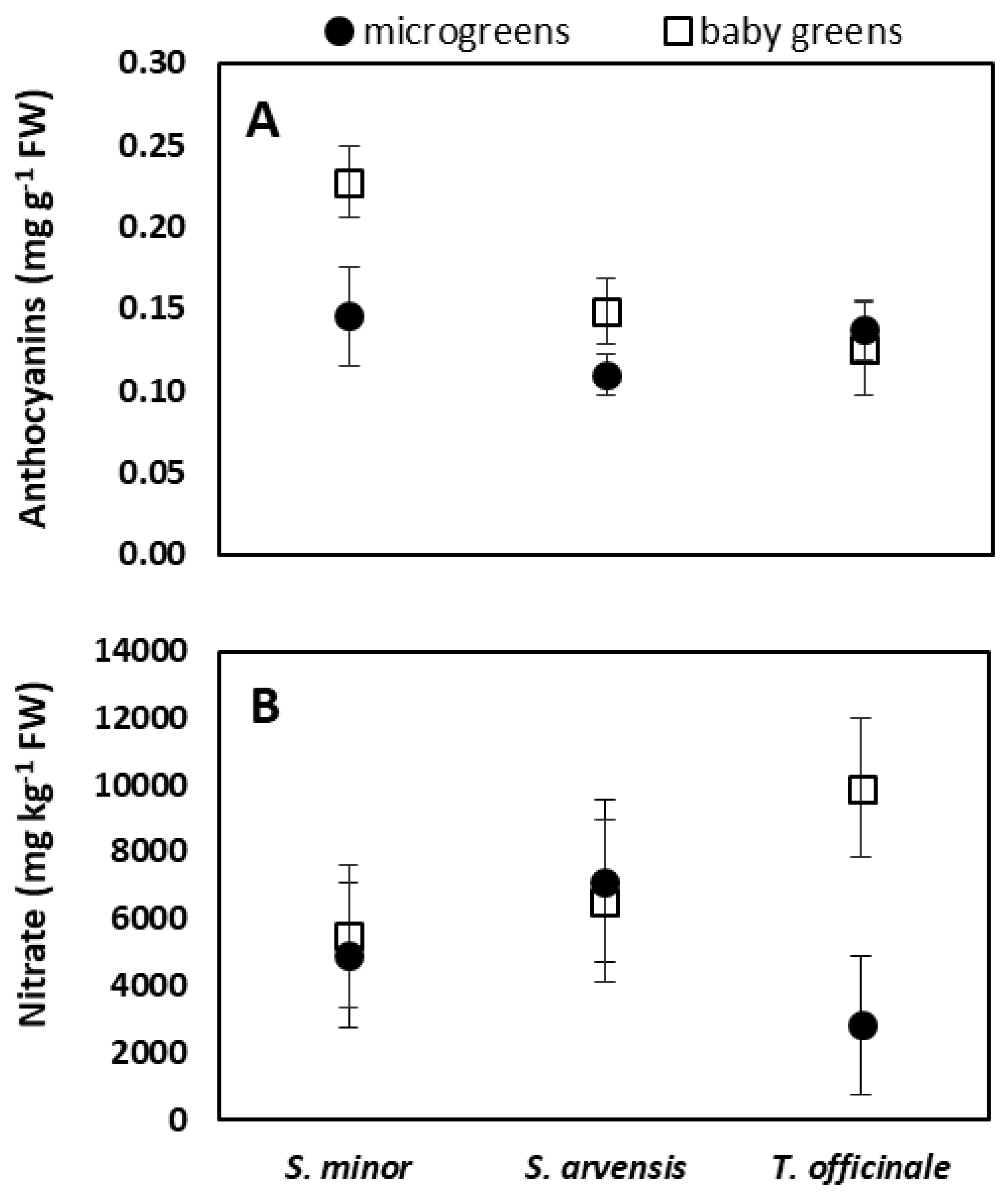
3.2. Chlorophylls, Carotenoids, Phenols, Anthocyanins and Nitrate Content
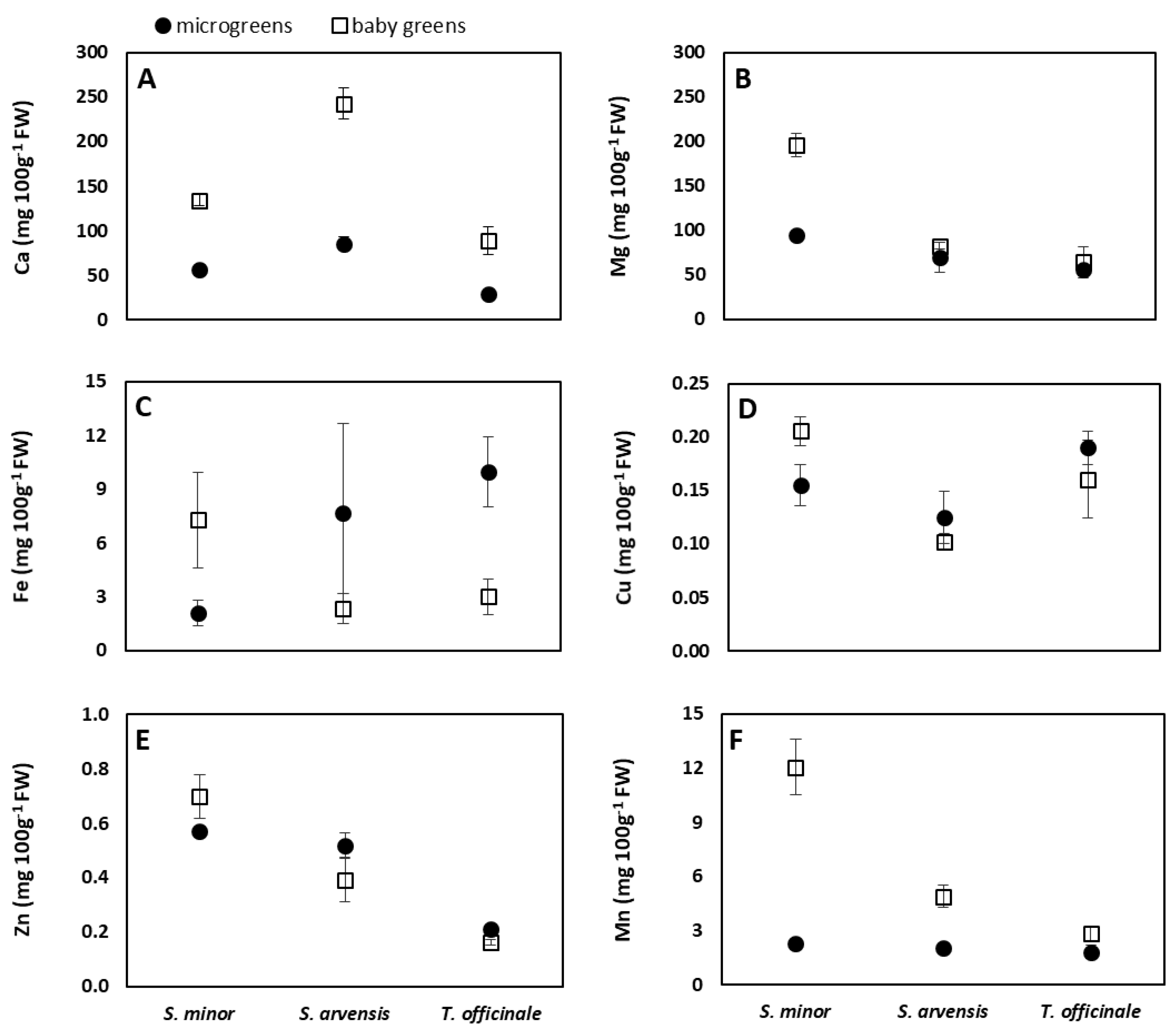
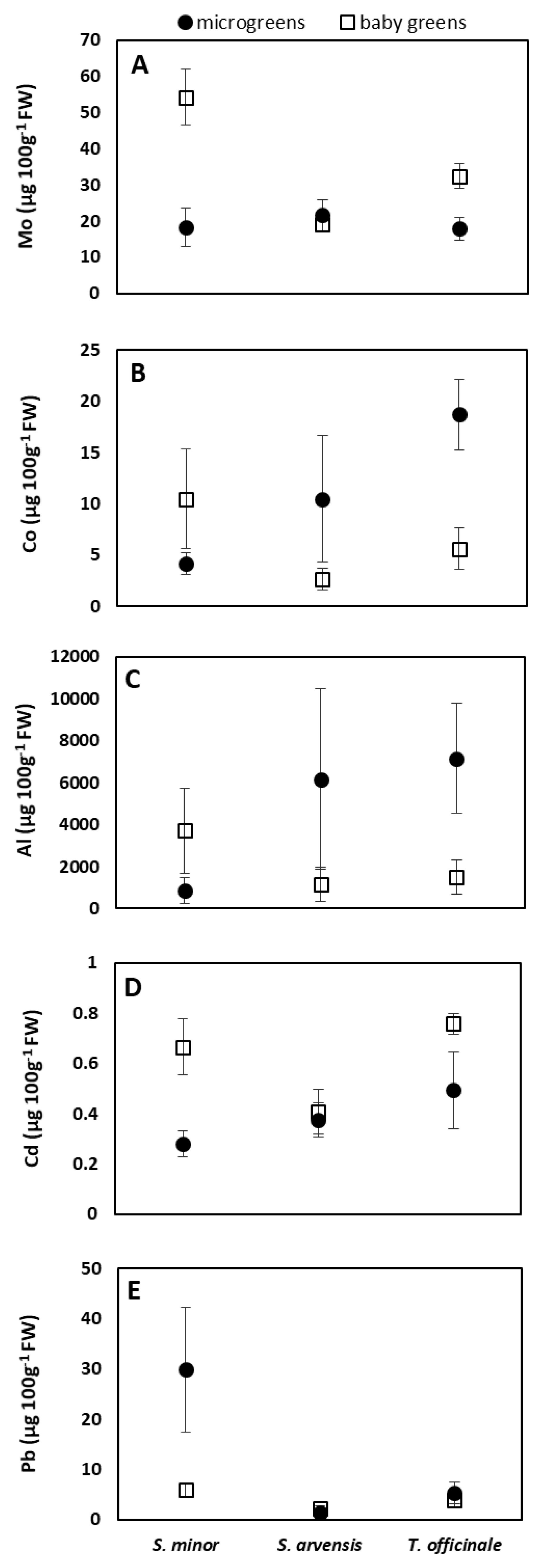
3.3. Mineral Content
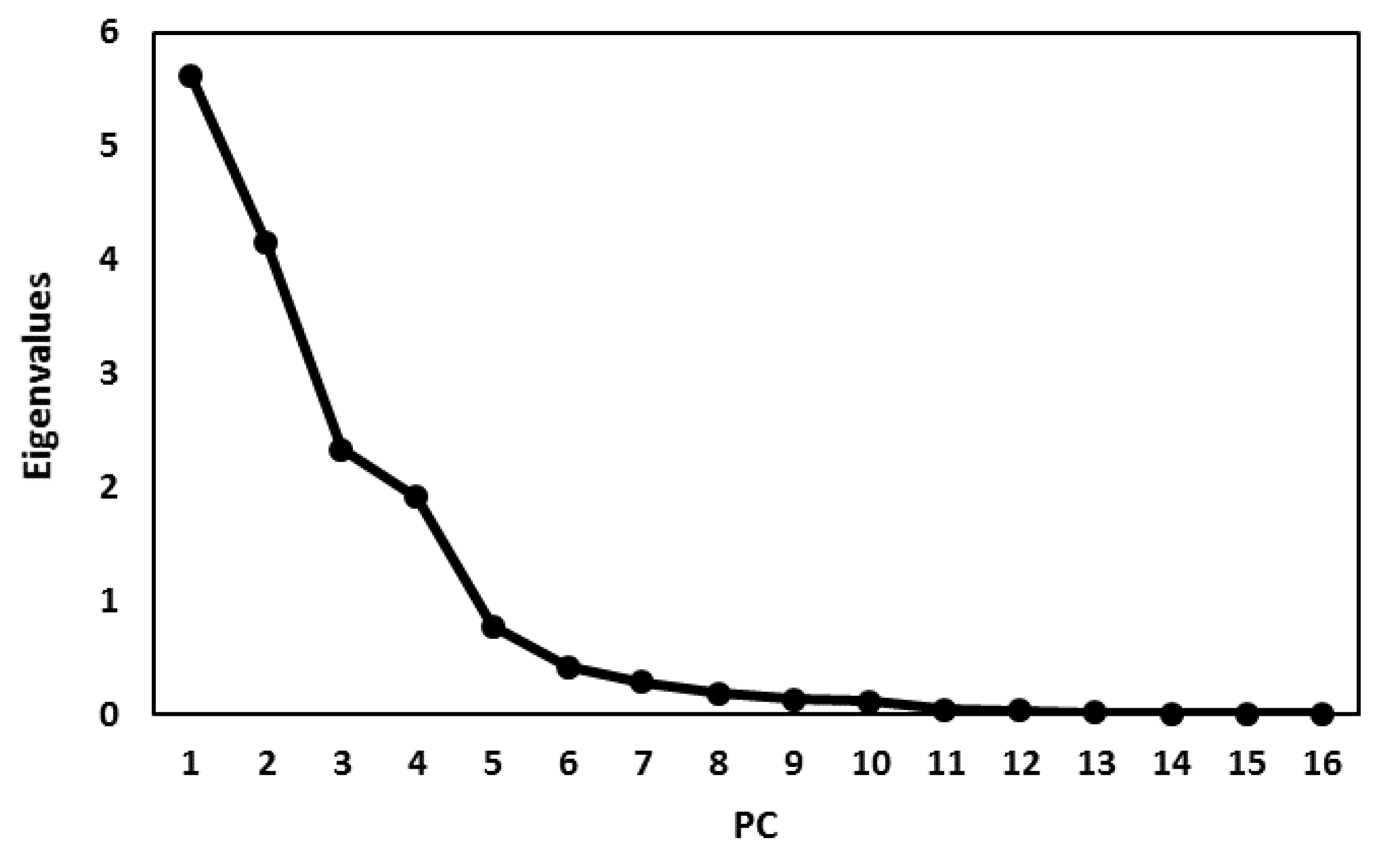
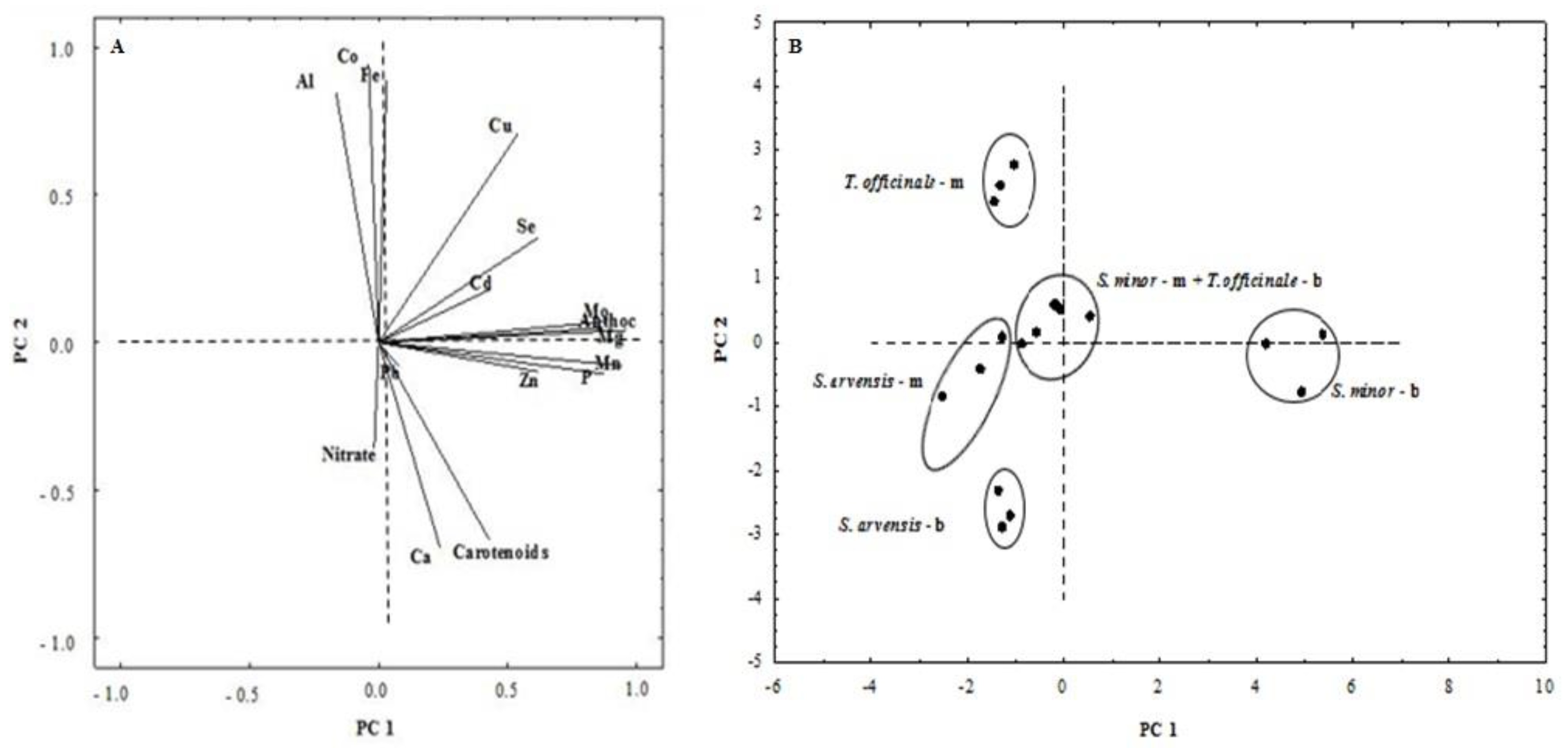
3.4. Principal Component Analysis (PCA)
3.5. Contribution to Mineral Dietary Intake and Health Risk Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Łuczaj, Ł.; Pieroni, A. Nutritional ethnobotany in Europe: From emergency foods to healthy folk cuisines and contemporary foraging trends. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 33–56. [Google Scholar] [CrossRef]
- Guil-Guerrero, J.L.; Madrid, P.C.; Isasa, M.E.T. Mineral elements determination in wild edible plants. Ecol. Food Nutr. 1999, 38, 209–222. [Google Scholar] [CrossRef]
- Sánchez-Mata, M.D.C.; Matallana-González, M.C.; Morales, P. The contribution of wild plants to dietary intakes of micronutrients (I): Vitamins. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 111–139. [Google Scholar] [CrossRef]
- García-Herrera, P.; Sánchez-Mata, M.D.C. The contribution of wild plants to dietary intakes of micronutrients (II): Mineral Elements. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 141–171. [Google Scholar] [CrossRef]
- Barros, L.; Morales, P.; Carvalho, A.M.; Ferreira, I.C.F.R. Antioxidant potential of wild plant foods. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 209–232. [Google Scholar] [CrossRef]
- Di Venere, D.; Gatto, M.A.; Ippolito, A.; Bianco, V.V. Antimicrobial potential of wild edible herbaceous species. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 233–252. [Google Scholar] [CrossRef]
- Heinrich, M.; Kerrouche, S.; Bharij, K.S. Recent advances in research on wild food plants and their biological–pharmacological activity. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 253–269. [Google Scholar] [CrossRef]
- Sánchez-Mata, D.; Morales, R. The Mediterranean landscape and wild edible plants. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 15–31. [Google Scholar] [CrossRef]
- Santamaria, P. Nitrate in vegetables: Toxicity, content, intake and EC regulation. J. Sci. Food Agric. 2006, 86, 10–17. [Google Scholar] [CrossRef]
- Bryan, N.S.; Alexander, D.D.; Coughlin, J.R.; Milkowski, A.L.; Boffetta, P. Ingested nitrate and nitrite and stomach cancer risk: An updated review. Food Chem. Toxicol. 2012, 50, 3646–3665. [Google Scholar] [CrossRef] [PubMed]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef]
- Brunetti, G.; Soler-Rovira, P.; Farrag, K.; Senesi, N. Tolerance and accumulation of heavy metals by wild plant species grown in contaminated soils in Apulia region, Southern Italy. Plant Soil 2009, 318, 285–298. [Google Scholar] [CrossRef]
- Bandiera, M.; Dal Cortivo, C.; Barion, G.; Mosca, G.; Vamerali, T. Phytoremediation opportunities with alimurgic species in metal-contaminated environments. Sustainability 2016, 8, 357. [Google Scholar] [CrossRef]
- Guarrera, P.M. Usi e Tradizioni della Flora Italiana. Medicina Popolare ed Etnobotanica; Aracne: Rome, Italy, 2006. [Google Scholar]
- Ebert, A.W. Sprouts, microgreens, and edible flowers: The potential for high value specialty produce in Asia. In High Value Vegetables in Southeast Asia: Production, Supply and Demand, Proceedings of the SEAVEG 2012, Chiang Mai, Thailand, 24–26 January 2012; AVRDC-The World Vegetable Center: Tainan, Taiwan, 2013; pp. 216–227. [Google Scholar]
- Treadwell, D.; Hochmuth, R.; Landrum, L.; Laughlin, W. Microgreens: A New Specialty Crop; University of Florida IFAS Extension: Gainesville, FL, USA, 2010. [Google Scholar]
- Saini, R.K.; Ko, E.Y.; Keum, Y.-S. Minimally processed ready-to-eat baby-leaf vegetables: Production, processing, storage, microbial safety, and nutritional potential. Food Rev. Int. 2017, 33, 644–663. [Google Scholar] [CrossRef]
- Baselice, A.; Colantuoni, F.; Lass, D.A.; Nardone, G.; Stasi, A. Trends in EU consumers’ attitude towards fresh-cut fruit and vegetables. Food Qual. Prefer. 2017, 59, 87–96. [Google Scholar] [CrossRef]
- Grahn, C.; Benedict, C.; Thornton, T.; Miles, C. Production of Baby-leaf Salad Greens in the Spring and Fall Seasons of Northwest Washington. HortScience 2015, 50, 1467–1471. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Mir, S.A.; Shah, M.A.; Mir, M.M. Microgreens: Production, shelf life, and bioactive components. Crit. Rev. Food Sci. Nutr. 2017, 57, 2730–2736. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, P.; Sharma, S. Microgreens: A Nutrient Rich Crop that can Diversify Food System. Int. J. Pure Appl. Biosci. 2018, 6, 182–186. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Wang, Q. Assessment of Vitamin and Carotenoid Concentrations of Emerging Food Products: Edible Microgreens. J. Agric. Food Chem. 2012, 60, 7644–7651. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Codling, E.E.; Luo, Y.; Nou, X.; Lester, G.E.; Wang, Q. Microgreens of Brassicaceae: Mineral composition and content of 30 varieties. J. Food Compos. Anal. 2016, 49, 87–93. [Google Scholar] [CrossRef]
- Xiao, Z.; Rausch, S.R.; Luo, Y.; Sun, J.; Yu, L.; Wang, Q.; Chen, P.; Yu, L.; Stommel, J.R. Microgreens of Brassicaceae: Genetic diversity of phytochemical concentrations and antioxidant capacity. LWT-Food Sci. Technol. 2019, 101, 731–737. [Google Scholar] [CrossRef]
- Samuolienė, G.; Brazaitytė, A.; Sirtautas, R.; Sakalauskienė, S.; Jankauskienė, J.; Duchovskis, P.; Novičkovas, A. The impact of supplementary short-term red LED lighting on the antioxidant properties of microgreens. Acta Hortic. 2012, 956, 649–656. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; El-Nakhel, C.; Graziani, G.; Pannico, A.; Soteriou, G.A.; Giordano, M.; Ritieni, A.; De Pascale, S.; Rouphael, Y. Functional quality in novel food sources: Genotypic variation in the nutritive and phytochemical composition of thirteen microgreens species. Food Chem. 2019, 277, 107–118. [Google Scholar] [CrossRef]
- Colonna, E.; Rouphael, Y.; Barbieri, G.; De Pascale, S. Nutritional quality of ten leafy vegetables harvested at two light intensities. Food Chem. 2016, 199, 702–710. [Google Scholar] [CrossRef]
- Egea-Gilabert, C.; Niñirola, D.; Conesa, E.; Candela, M.E.; Fernández, J.A. Agronomical use as baby leaf salad of Silene vulgaris based on morphological, biochemical and molecular traits. Sci. Hortic. 2013, 152, 35–43. [Google Scholar] [CrossRef]
- Ebert, A.W.; Wu, T.H.; Yang, R.Y. Amaranth sprouts and microgreens—A homestead vegetable production option to enhance food and nutrition security in the rural-urban continuum. In Proceedings of the Regional Symposium on Sustaining Small-Scale Vegetable Production and Marketing Systems for Food and Nutrition Security (SEAVEG 2014), Bangkok, Thailand, 25–27 February 2014; pp. 233–244. [Google Scholar] [CrossRef]
- Butkutė, B.; Taujenis, L.; Norkevičienė, E. Small-Seeded Legumes as a Novel Food Source. Variation of Nutritional, Mineral and Phytochemical Profiles in the Chain: Raw Seeds-Sprouted Seeds-Microgreens. Molecules 2018, 24, 133. [Google Scholar] [CrossRef]
- Zhao, X.; Iwamoto, T.; Carey, E.E. Antioxidant capacity of leafy vegetables as affected by high tunnel environment, fertilisation and growth stage. J. Sci. Food Agric. 2007, 87, 2692–2699. [Google Scholar] [CrossRef] [PubMed]
- López, A.; Javier, G.-A.; Fenoll, J.; Hellín, P.; Flores, P. Chemical composition and antioxidant capacity of lettuce: Comparative study of regular-sized (Romaine) and baby-sized (Little Gem and Mini Romaine) types. J. Food Compos. Anal. 2014, 33, 39–48. [Google Scholar] [CrossRef]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M.P.L.V.O. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Choe, U.; Yu, L.L.; Wang, T.T.Y. The Science behind Microgreens as an Exciting New Food for the 21st Century. J. Agric. Food Chem. 2018, 66, 11519–11530. [Google Scholar] [CrossRef] [PubMed]
- Waterland, N.L.; Youyoun, M.; Janet, C.T.; Moo Jung, K.; Eugenia, M.P.-Y.; Suejin, P. Mineral Content Differs among Microgreen, Baby Leaf, and Adult Stages in Three Cultivars of Kale. HortScience 2017, 52, 566–571. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1987; Volume 148, pp. 350–382. [Google Scholar]
- Ferrante, A.; Incrocci, L.; Maggini, R.; Serra, G.; Tognoni, F. Colour changes of fresh-cut leafy vegetables during storage. J. Food Agric. Environ. 2004, 2, 40–44. [Google Scholar] [CrossRef]
- Klein, A.O.; Hagen, C.W. Anthocyanin production in detached petals of Impatiens balsamina L. Plant Physiol. 1961, 36, 1–9. [Google Scholar] [CrossRef]
- Cataldo, D.A.; Maroon, M.; Schrader, L.E.; Youngs, V.L. Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun. Soil Sci. Plant Anal. 1975, 6, 71–80. [Google Scholar] [CrossRef]
- SINU. Livelli di Assunzione di Riferimento di Nutrienti ed Energia, IV Revisione. Società Italiana di Nutrizione Umana. Available online: http://www.sinu.it/html/pag/12-MINERALI.asp (accessed on 14 February 2019).
- US-EPA IRIS. Reference Dose (RfD): Description and Use in Health risk Assessments, Background Document 1A, Integrated Risk Information System (IRIS). Available online: http://www.epa.gov/iris/rfd.htm (accessed on 14 February 2019).
- Khan, S.; Cao, Q.; Zheng, Y.M.; Huang, Y.Z.; Zhu, Y.G. Health risks of heavy metals in contaminated soils and food crops irrigated with wastewater in Beijing, China. Environ. Pollut. 2008, 152, 686–692. [Google Scholar] [CrossRef]
- EFSA. Safety of aluminium from dietary intake—Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Food Contact Materials (AFC). EFSA J. 2008, 6, 754. [Google Scholar] [CrossRef]
- FAO/WHO. Codex Alimentarius: International Food Standards. Rome: World Health Organization & Food and Agriculture Organization of the United Nations. Available online: http://www.codexalimentarius.org/ (accessed on 14 February 2019).
- Bulgari, R.; Baldi, A.; Ferrante, A.; Lenzi, A. Yield and quality of basil, Swiss chard, and rocket microgreens grown in a hydroponic system. N. Z. J. Crop Hortic. Sci. 2017, 45, 119–129. [Google Scholar] [CrossRef]
- Paradiso, V.M.; Castellino, M.; Renna, M.; Gattullo, C.E.; Calasso, M.; Terzano, R.; Allegretta, I.; Leoni, B.; Caponio, F.; Santamaria, P. Nutritional characterization and shelf-life of packaged microgreens. Food Funct. 2018, 9, 5629–5640. [Google Scholar] [CrossRef] [PubMed]
- Renna, M.; Castellino, M.; Leoni, B.; Paradiso, M.V.; Santamaria, P. Microgreens Production with Low Potassium Content for Patients with Impaired Kidney Function. Nutrients 2018, 10, 675. [Google Scholar] [CrossRef] [PubMed]
- Fallovo, C.; Rouphael, Y.; Rea, E.; Battistelli, A.; Colla, G. Nutrient solution concentration and growing season affect yield and quality of Lactuca sativa L. var. acephala in floating raft culture. J. Sci. Food Agric. 2009, 89, 1682–1689. [Google Scholar] [CrossRef]
- Manzocco, L.; Foschia, M.; Tomasi, N.; Maifreni, M.; Dalla Costa, L.; Marino, M.; Cortella, G.; Cesco, S. Influence of hydroponic and soil cultivation on quality and shelf life of ready-to-eat lamb’s lettuce (Valerianella locusta L. Laterr). J. Sci. Food Agric. 2011, 91, 1373–1380. [Google Scholar] [CrossRef] [PubMed]
- Cámara, M.; Fernández-Ruiz, V.; Ruiz-Rodríguez, B.M. Wild edible plants as sources of carotenoids, fibre, phenolics and other non-nutrient bioactive compounds. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 187–205. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, J.; Tian, J.; Li, N.; Jia, L.; Shen, W.; Cui, J. Enhanced anthocyanin accumulation of immature radish microgreens by hydrogen-rich water under short wavelength light. Sci. Hortic. 2019, 247, 75–85. [Google Scholar] [CrossRef]
- Hutchings, J.B. Food Colour and Appearance; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar] [CrossRef]
- Kopsell, D.A.; Pantanizopoulos, N.I.; Sams, C.E.; Kopsell, D.E. Shoot tissue pigment levels increase in ‘Florida Broadleaf’ mustard (Brassica juncea L.) microgreens following high light treatment. Sci. Hortic. 2012, 140, 96–99. [Google Scholar] [CrossRef]
- Samuolienė, G.; Viršilė, A.; Brazaitytė, A.; Jankauskienė, J.; Sakalauskienė, S.; Vaštakaitė, V.; Novičkovas, A.; Viškelienė, A.; Sasnauskas, A.; Duchovskis, P. Blue light dosage affects carotenoids and tocopherols in microgreens. Food Chem. 2017, 228, 50–56. [Google Scholar] [CrossRef]
- Brazaitytė, A.; Sakalauskienė, S.; Samuolienė, G.; Jankauskienė, J.; Viršilė, A.; Novičkovas, A.; Sirtautas, R.; Miliauskienė, J.; Vaštakaitė, V.; Dabašinskas, L.; et al. The effects of LED illumination spectra and intensity on carotenoid content in Brassicaceae microgreens. Food Chem. 2015, 173, 600–606. [Google Scholar] [CrossRef]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, microgreens and “baby leaf” vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Yildiz, F., Wiley, R.C., Eds.; Springer: Boston, MA, USA, 2017; pp. 403–432. [Google Scholar] [CrossRef]
- Saini, R.K.; Nile, S.H.; Park, S.W. Carotenoids from fruits and vegetables: Chemistry, analysis, occurrence, bioavailability and biological activities. Food Res. Int. 2015, 76, 735–750. [Google Scholar] [CrossRef] [PubMed]
- Gorenjak, A.H.; Koležnik, U.R.; Cencič, A. Nitrate content in dandelion (Taraxacum officinale) and lettuce (Lactuca sativa) from organic and conventional origin: Intake assessment. Food Addit. Contam. Part B 2012, 5, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Butler, A. Nitrites and nitrates in the human diet: Carcinogens or beneficial hypotensive agents? J. Ethnopharmacol. 2015, 167, 105–107. [Google Scholar] [CrossRef] [PubMed]
- Omar, S.A.; Webb, A.J.; Lundberg, J.O.; Weitzberg, E. Therapeutic effects of inorganic nitrate and nitrite in cardiovascular and metabolic diseases. J. Intern. Med. 2016, 279, 315–336. [Google Scholar] [CrossRef] [PubMed]
- Stanaway, L.; Rutherfurd-Markwick, K.; Page, R.; Ali, A. Performance and Health Benefits of Dietary Nitrate Supplementation in Older Adults: A Systematic Review. Nutrients 2017, 9, 1171. [Google Scholar] [CrossRef]
- Renna, M.; Cocozza, C.; Gonnella, M.; Abdelrahman, H.; Santamaria, P. Elemental characterization of wild edible plants from countryside and urban areas. Food Chem. 2015, 177, 29–36. [Google Scholar] [CrossRef]
- Tardío, J.; Sánchez-Mata, M.D.C.; Morales, R.; Molina, M.; García-Herrera, P.; Morales, P.; Díez-Marqués, C.; Fernández-Ruiz, V.; Cámara, M.; Pardo-de-Santayana, M.; et al. Ethnobotanical and food composition monographs of selected Mediterranean wild edible plants. In Mediterranean Wild Edible Plants: Ethnobotany and Food Composition Tables; Sánchez-Mata, M.D.C., Tardío, J., Eds.; Springer: New York, NY, USA, 2016; pp. 273–470. [Google Scholar] [CrossRef]
- Weber, C.F. Broccoli Microgreens: A Mineral-Rich Crop That Can Diversify Food Systems. Front. Nutr. 2017, 4, 7. [Google Scholar] [CrossRef]
- Santos, J.; Oliva-Teles, M.T.; Delerue-Matos, C.; Oliveira, M.B.P.P. Multi-elemental analysis of ready-to-eat “baby leaf” vegetables using microwave digestion and high-resolution continuum source atomic absorption spectrometry. Food Chem. 2014, 151, 311–316. [Google Scholar] [CrossRef]
- Pinela, J.; Carvalho, A.M.; Ferreira, I.C.F.R. Wild edible plants: Nutritional and toxicological characteristics, retrieval strategies and importance for today’s society. Food Chem. Toxicol. 2017, 110, 165–188. [Google Scholar] [CrossRef]
- Giacomino, A.; Malandrino, M.; Colombo, M.L.; Miaglia, S.; Maimone, P.; Blancato, S.; Conca, E.; Abollino, O. Metal Content in Dandelion (Taraxacum officinale) Leaves: Influence of Vehicular Traffic and Safety upon Consumption as Food. J. Chem. 2016, 2016, 1–9. [Google Scholar] [CrossRef]
- Stark, P.B.; Miller, D.; Carlson, T.J.; de Vasquez, K.R. Open-source food: Nutrition, toxicology, and availability of wild edible greens in the East Bay. PLoS ONE 2019, 14, e0202450. [Google Scholar] [CrossRef] [PubMed]
- Nziguheba, G.; Smolders, E. Inputs of trace elements in agricultural soils via phosphate fertilizers in European countries. Sci. Total Environ. 2008, 390, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.; Chen, W.; Chang, A.C.; Page, A.L. Environmental risks of trace elements associated with long-term phosphate fertilizers applications: A review. Environ. Pollut. 2012, 168, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.E.; Singleton, E.; Bandli, B.R.; Lowers, H.A.; Meeker, G.P. Differentiation of commercial vermiculite based on statistical analysis of bulk chemical data: Fingerprinting vermiculite from Libby, Montana U.S.A. Am. Mineral. 2005, 90, 749–754. [Google Scholar] [CrossRef]

| Species | 1000-Seed Weight 1 g | Germination 2 (%) |
|---|---|---|
| S. minor | 7.02 ± 0.30 | 75.5 ± 3.4 |
| S. arvensis | 2.48 ± 0.13 | 61.0 ± 3.7 |
| T. officinale | 0.62 ± 0.03 | 72.0 ± 2.9 |
| Treatments | Chl a mg/g FW | Chl b mg/g FW | Chl a+b mg/g FW | Carotenoids mg/g FW | Anthocyanins 1 mg/g FW | Phenolic Index ABS320 nm/g FW | Nitrate mg/kg FW |
|---|---|---|---|---|---|---|---|
| Species | |||||||
| S. minor | 0.84 ± 0.58 | 0.66 ± 0.58 | 1.41 ± 1.17 | 0.16 ± 0.07 | 0.19 ± 0.05 a | 11.95 ± 2.85 | 5205 ± 2023 |
| S. arvensis | 1.00 ± 0.40 | 0.55 ± 0.35 | 1.55 ± 0.68 | 0.18 ± 0.08 | 0.13 ± 0.03 b | 10.98 ± 2.51 | 6833 ± 1626 |
| T. officinale | 0.90 ± 0.38 | 0.65 ± 0.31 | 1.55 ± 0.64 | 0.11 ± 0.04 | 0.13 ± 0.02 b | 10.78 ± 1.91 | 6368 ± 4100 |
| Stage of harvest | |||||||
| Microgreens | 0.76 ± 0.38 | 0.50 ± 0.50 | 1.26 ± 0.85 | 0.11 ± 0.05 b | 0.13 ± 0.02 b | 10.30 ± 2.74 | 4962 ± 2231 b |
| Baby greens | 1.07 ± 0.46 | 0.74 ± 0.26 | 1.75 ± 0.74 | 0.20 ± 0.05 a | 0.17 ± 0.05 a | 12.17 ± 1.54 | 7308 ± 2774 a |
| Significance | |||||||
| Species | ns | ns | ns | ns | *** | ns | ns |
| Stage of harvest | ns | ns | ns | ** | ** | ns | * |
| Species x stage of harvest | ns | ns | ns | ns | * | ns | * |
| Treatments | Ca | Mg | P | Fe | Cu | Zn | Mn |
|---|---|---|---|---|---|---|---|
| mg/100 g FW | |||||||
| Species | |||||||
| S. minor | 95.59 ± 42.59 b | 145.58 ± 55.71 a | 108.11 ± 13.76 a | 4.70 ± 3.32 | 0.18 ± 0.03 a | 0.63 ± 0.09 a | 7.16 ± 5.45 a |
| S. arvensis | 163.95 ± 87.09 a | 75.99 ± 13.21 b | 54.08 ± 11.56 b | 5.03 ± 4.31 | 0.11 ± 0.02 b | 0.46 ± 0.09 b | 3.48 ± 1.61 b |
| T. officinale | 59.00 ± 34.22 c | 59.79 ± 12.78 b | 61.56 ± 20.07 b | 6.48 ± 4.04 | 0.18 ± 0.03 a | 0.19 ± 0.03 c | 2.34 ± 0.67 c |
| Stage of harvest | |||||||
| Microgreens | 57.05 ± 24.60 b | 73.39 ± 19.71 b | 63.59 ± 25.16 b | 6.59 ± 4.40 | 0.16 ± 0.03 | 0.43 ± 0.17 | 2.05 ± 0.30 b |
| Baby greens | 155.31 ± 69.48 a | 114.18 ± 62.79 a | 85.57 ± 28.89 a | 4.22 ± 2.74 | 0.16 ± 0.05 | 0.42 ± 0.24 | 6.61 ± 4.27 a |
| Significance | |||||||
| Species | *** | *** | *** | ns | *** | *** | *** |
| Stage of harvest | *** | *** | ** | ns | ns | ns | *** |
| Species x stage of harvest | *** | *** | ns | ** | ** | ** | *** |
| Treatments | Cr | Se | Mo | Co | Al | Ni | As | Cd | Pb |
|---|---|---|---|---|---|---|---|---|---|
| μg/100 g FW | |||||||||
| Species | |||||||||
| S. minor | 256.4 ± 281.7 | 25.3 ± 4.2 a | 36.3 ± 20.5 a | 7.3 ± 4.7 | 2284.8 ± 2058.7 | 138.5 ± 130.2 | 2.4 ± 0.7 | 0.5 ± 0.2 b | 17.9 ± 15.3 a |
| S. arvensis | 59.3 ±5 1.1 | 10.0 ± 3.2 b | 20.3 ± 3.2 b | 6.6 ± 5.8 | 3664.8 ± 3903.1 | 32.3 ± 28.1 | 1.0 ± 0.3 | 0.4 ± 0.1 b | 1.8 ± 0.6 b |
| T. officinale | 267.3 ± 275.9 | 22.3 ± 4.1 a | 25.2 ± 8.5 | 12.2 ± 7.6 | 4331.3 ± 3546.0 | 139.8 ± 146.8 | 4.1 ± 5.7 | 0.6 ± 0.2 a | 4.6 ± 1.9 b |
| Stage of harvest | |||||||||
| Microgreens | 192.3 ± 243.0 | 18.1 ± 5.8 | 19.3 ± 4.2 b | 11.1 ± 7.3 a | 4729.6 ± 3878.4 a | 107.9 ± 125.2 | 1.3 ± 0.5 | 0.4 ± 0.1 b | 12.2 ± 14.8 a |
| Baby greens | 196.4 ± 245.6 | 20.4 ± 9.5 | 35.2 ± 16.0 a | 6.3 ± 4.3 b | 2124.3 ± 1674.4 b | 99.2 ± 120.5 | 3.6 ± 4.6 | 0.6 ± 0.2 a | 4.0 ± 1.9 b |
| Significance | |||||||||
| Species | ns | *** | *** | ns | ns | ns | ns | ** | *** |
| Stage of harvest | ns | ns | *** | * | * | ns | ns | *** | ** |
| Species x stage of harvest | ns | ns | *** | ** | * | ns | ns | * | ** |
| Mineral | RDI/AI 1 mg/day | Microgreens | Baby Greens | ||||
|---|---|---|---|---|---|---|---|
| S. minor | S. arvensis | T. officinale | S. minor | S. arvensis | T. officinale | ||
| Ca | 1000 | 1.14 | 1.7 | 0.58 | 2.69 | 4.85 | 1.78 |
| Mg | 240 | 7.94 | 5.79 | 4.61 | 16.32 | 6.88 | 5.35 |
| P | 700 | 2.76 | 1.28 | 1.41 | 3.42 | 1.81 | 2.11 |
| Fe | 10 | 4.23 | 15.39 | 19.91 | 14.56 | 4.73 | 6.02 |
| Cu | 0.9 | 3.44 | 2.78 | 4.22 | 4.56 | 2.26 | 3.57 |
| Zn | 11 | 1.04 | 0.95 | 0.39 | 1.27 | 0.71 | 0.30 |
| Mn | 2.7 | 16.82 | 15.28 | 13.39 | 89.32 | 36.30 | 21.23 |
| Cr | 0.035 | 42.27 | 54.46 | 232.98 | 250.78 | 13.33 | 72.51 |
| Se | 0.055 | 8.16 | 4.24 | 7.35 | 10.26 | 3.05 | 8.88 |
| Mo | 0.045 | 8.15 | 9.60 | 7.94 | 24.09 | 8.48 | 14.43 |
| Metal | Microgreens | Baby Greens | |||||
|---|---|---|---|---|---|---|---|
| S. minor | S. arvensis | T. officinale | S. minor | S. arvensis | T. officinale | ||
| Fe (RfD = 0.7) | EDIBW | 0.007565 | 0.027538 | 0.035613 | 0.026045 | 0.008454 | 0.010773 |
| HRI | 0.010808 | 0.039339 | 0.050876 | 0.037207 | 0.012077 | 0.015391 | |
| Cu (RfD = 0.01) | EDIBW | 0.000553 | 0.000447 | 0.00068 | 0.000735 | 0.000363 | 0.000575 |
| HRI | 0.013835 | 0.011178 | 0.016994 | 0.018383 | 0.009082 | 0.014373 | |
| Zn (RfD = 0.3) | EDIBW | 0.002037 | 0.001861 | 0.000764 | 0.002502 | 0.001398 | 0.000581 |
| HRI | 0.006791 | 0.006203 | 0.002548 | 0.008341 | 0.00466 | 0.001936 | |
| Mn (RfD = 0.14) | EDIBW | 0.008124 | 0.007378 | 0.006466 | 0.043143 | 0.017535 | 0.010253 |
| HRI | 0.058028 | 0.0527 | 0.046187 | 0.308164 | 0.125247 | 0.073234 | |
| Cr (RfD = 0.003) | EDIBW | 0.000265 | 0.000341 | 0.001459 | 0.00157 | 0.000083 | 0.000454 |
| HRI | 0.088223 | 0.113652 | 0.486246 | 0.523397 | 0.027814 | 0.151333 | |
| Se (RfD = 0.005) | EDIBW | 0.00008 | 0.000042 | 0.000072 | 0.000101 | 0.00003 | 0.000087 |
| HRI | 0.016061 | 0.00835 | 0.014472 | 0.020196 | 0.006007 | 0.017484 | |
| Mo (RfD = 0.005) | EDIBW | 0.000007 | 0.000008 | 0.000006 | 0.000019 | 0.000007 | 0.000012 |
| HRI | 0.001313 | 0.001546 | 0.001279 | 0.003879 | 0.001364 | 0.002323 | |
| Co (RfD = 0.0003) | EDIBW | 0.000015 | 0.000037 | 0.000067 | 0.000037 | 0.00001 | 0.00002 |
| HRI | 0.049521 | 0.124754 | 0.223107 | 0.12494 | 0.031681 | 0.067146 | |
| Ni (RfD = 0.02) | EDIBW | 0.000196 | 0.000192 | 0.00077 | 0.000795 | 0.00004 | 0.00023 |
| HRI | 0.009811 | 0.009592 | 0.038497 | 0.039736 | 0.001977 | 0.011521 | |
| As (RfD = 0.0003) | EDIBW | 0.000006 | 0.000003 | 0.000006 | 0.000011 | 0.000005 | 0.000024 |
| HRI | 0.020747 | 0.009076 | 0.018335 | 0.035316 | 0.01538 | 0.07893 | |
| Cd (RfD = 0.001) | EDIBW | 0.000001 | 0.000001 | 0.000002 | 0.000002 | 0.000001 | 0.000003 |
| HRI | 0.001003 | 0.001342 | 0.001767 | 0.002385 | 0.001467 | 0.002715 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenzi, A.; Orlandini, A.; Bulgari, R.; Ferrante, A.; Bruschi, P. Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison Between Microgreens and Baby Greens. Foods 2019, 8, 487. https://doi.org/10.3390/foods8100487
Lenzi A, Orlandini A, Bulgari R, Ferrante A, Bruschi P. Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison Between Microgreens and Baby Greens. Foods. 2019; 8(10):487. https://doi.org/10.3390/foods8100487
Chicago/Turabian StyleLenzi, Anna, Alessandro Orlandini, Roberta Bulgari, Antonio Ferrante, and Piero Bruschi. 2019. "Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison Between Microgreens and Baby Greens" Foods 8, no. 10: 487. https://doi.org/10.3390/foods8100487
APA StyleLenzi, A., Orlandini, A., Bulgari, R., Ferrante, A., & Bruschi, P. (2019). Antioxidant and Mineral Composition of Three Wild Leafy Species: A Comparison Between Microgreens and Baby Greens. Foods, 8(10), 487. https://doi.org/10.3390/foods8100487








