Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview
Abstract
1. Introduction
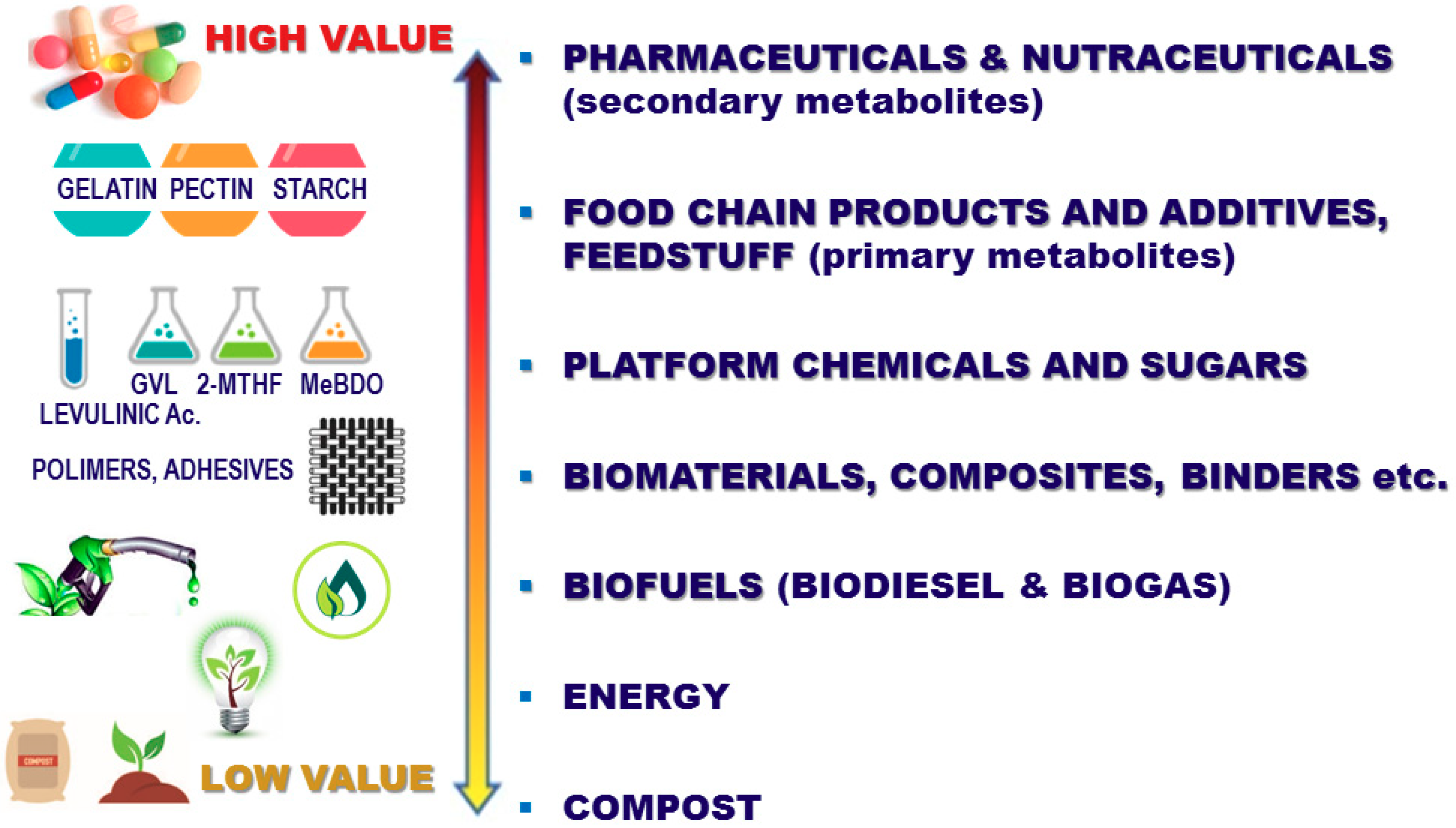
2. The State of the Art in Agro-Food Extraction
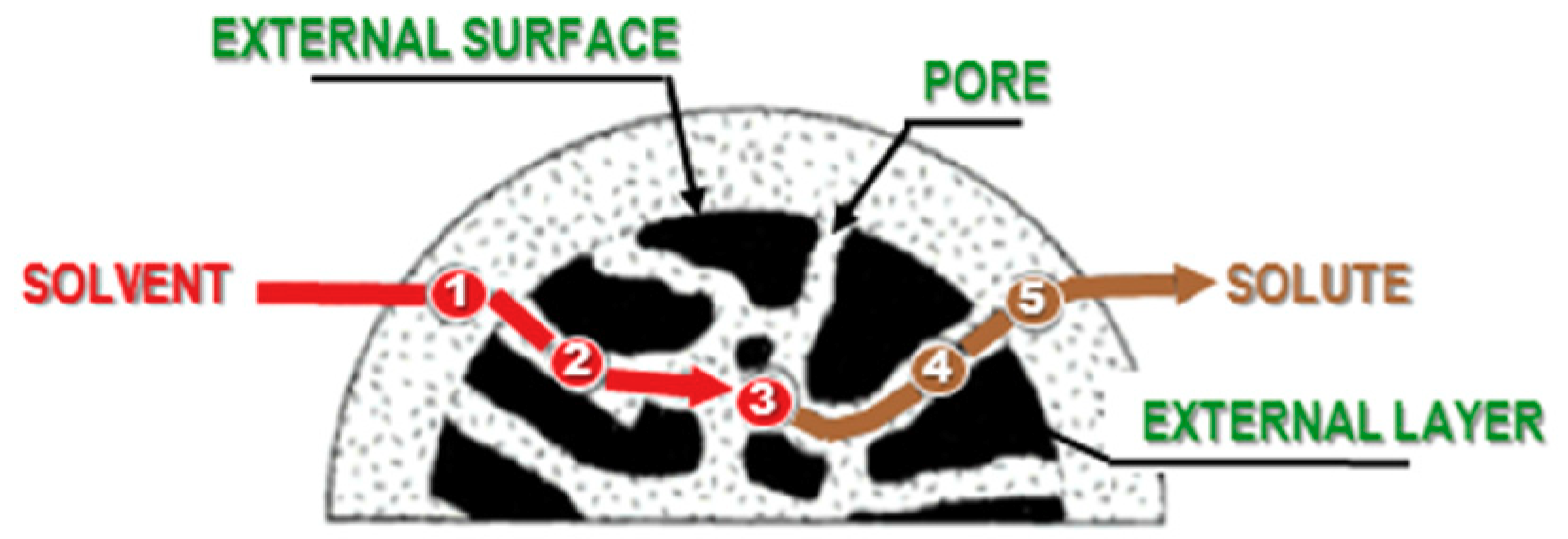
3. Improved Extractions under Ultrasound and Hydrodynamic Cavitation
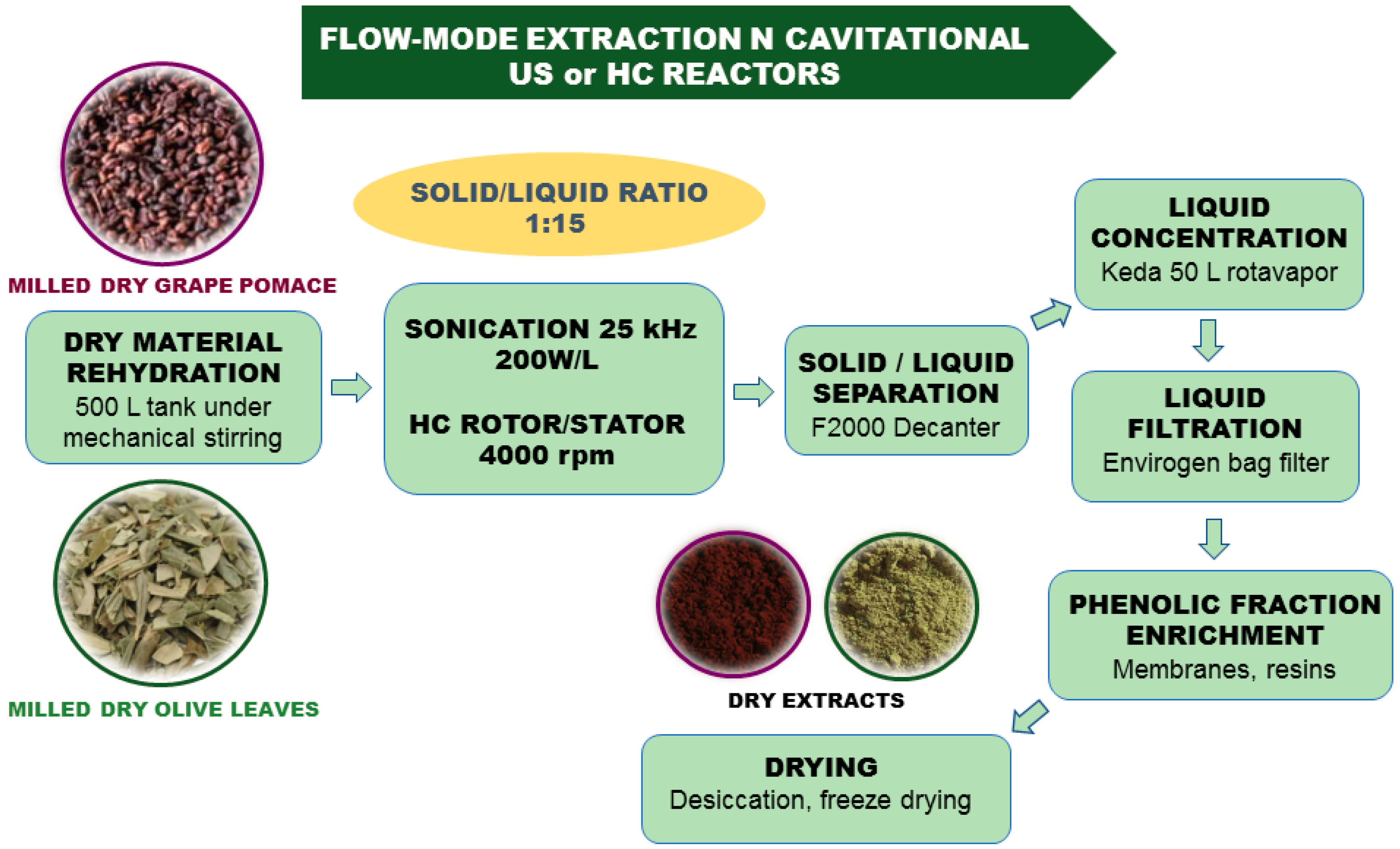
4. Flow-Mode Extraction in Pilot Scale
5. Main Features of a Pilot Scale Green Technologies Development Platform
- (i)
- The efficient and selective extraction of the primary and secondary metabolites (e.g., starches, cellulose, pectins, polyphenols, anthocyans, flavonoids, etc.) from agro-food matrices and production waste, in particular from grape pomace and olive leaves.
- (ii)
- Multi-stage processing in the food, nutraceutical, pharmaceutical, and flavouring fields, including a transition from lab-scale data to pilot-scale preparation, and the subsequent design of potential large-scale production plants.
6. Features of Grape Pomace and Olive Leaves Polyphenols
6.1. Polyphenols
6.2. Grape Polyphenols
6.3. Olive Leaf Polyphenols
6.4. Flavonoids
6.5. The Polyphenol Market
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Innovative Food Processing Technologies. Extraction, separation, component modification and process intensification. In Woodhead Publishing Series in Food Science, Technology and Nutrition; Knoerzer, K., Juliano, P., Smithers, G., Eds.; Woodhead Publishing: Cambridge, UK, 2016; ISBN 9780081002940. [Google Scholar]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Roohinejad, S.; Nikmaram, N.; Brahim, M.; Kouba, M.; Khelfa, A.; Greiner, R. Chapter 16: Potential of novel technologies for aqueous extraction of plant bioactives. In Water Extraction of Bioactive Compounds; González, H.D., Ed.; Elsevier: New York, NY, USA, 2017; pp. 399–419. ISBN 978-0-12-809380-1. [Google Scholar]
- Zhu, Z.; He, J.; Liu, G.; Barba, F.J.; Koubaa, M.; Ding, L.; Bals, O.; Grimi, N.; Vorobiev, E. Recent insights for the green recovery of inulin from plant food materials using non-conventional extraction technologies: A review. Innov. Food Sci. Emerg. Technol. 2016, 33, 1–9. [Google Scholar] [CrossRef]
- Roohinejad, S.; Kouba, M.; Barba, F.J.; Greiner, R.; Orlien, V.; Lebovka, N.I. Negative pressure cavitation extraction: A novel method for extraction of food bioactive compounds from plant materials. Trends Food Sci. Technol. 2016, 52, 98–108. [Google Scholar] [CrossRef]
- Cabeza, L.F.; Taylor, M.M.; Di Maio, G.L.; Brown, E.M.; Marmer, W.N.; Carrió, R.; Celma, P.J.; Cotc, J. Processing of leather waste: Pilot scale studies on chrome shavings. Isolation of potentially valuable protein products and chromium. Waste Manag. 1998, 18, 211–218. [Google Scholar] [CrossRef]
- Herrero, M.; Cifuentes, A.; Ibañez, E. Sub-and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae: A review. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhang, R.; Zhan, S.; He, J.; Barba, F.J.; Cravotto, G.; Wu, W.; Li, S. Recovery of oil with unsaturated fatty acids and polyphenols from Chaenomelessinensis (Thouin) Koehne: Process optimization of pilot-scale subcritical fluid assisted extraction. Molecules 2017, 22, 1788. [Google Scholar] [CrossRef] [PubMed]
- Filly, A.; Fernandez, X.; Minuti, M.; Visinoni, F.; Cravotto, G.; Chemat, F. Solvent free microwave extraction of essential oil from aromatic herbs. From laboratory to pilot and industrial scale. Food Chem. 2014, 150, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Plateforme d’Eco-Extraction de Valreas. Available online: http://www.peev.fr/ (accessed on 6 August 2018).
- Chemat, F.; Abert-Vian, M.; Cravotto, G. Review: Green Extraction of Natural Products: Concept and Principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Cintas, P. Extraction of flavourings from natural sources. In Modifying Flavour in Food; Taylor, A., Hort, J., Eds.; Woodhead Publishing: Cambridge, UK, 2007; pp. 41–63. [Google Scholar]
- Baiano, A.; Del Nobile, M.A. Antioxidant compounds from vegetable matrices: Biosynthesis, occurrence, and extraction systems. Crit. Rev. Food Sci. Nutr. 2016, 56, 2053–2068. [Google Scholar] [CrossRef] [PubMed]
- Prado, J.M.; Veggi, P.C.; Meireles, M.A.A. Extraction methods for obtaining carotenoids from vegetables—Review. Curr. Anal. Chem. 2013, 10, 29–66. [Google Scholar] [CrossRef]
- Talmaciu, A.I.; Volf, I.; Popa, V.I. A comparative analysis of the “green” techniques applied for polyphenols extraction from bioresources. Chem. Biodivers. 2015, 12, 1635–1651. [Google Scholar] [CrossRef] [PubMed]
- Pasqualone, A.; Gambacorta, G.; Summo, C.; Caponio, F.; Di Miceli, G.; Flagella, Z.; Marrese, P.P.; Piro, G.; Perrotta, C.; De Bellis, L.; et al. Functional, textural and sensory properties of dry pasta supplemented with lyophilized tomato matrix or with durum wheat bran extracts produced by supercritical carbon dioxide or ultrasound. Food Chem. 2016, 213, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Pasqualone, A.; Punzi, R.; Trani, A.; Summo, C.; Paradiso, V.M.; Caponio, F.; Gambacorta, G. Enrichment of fresh pasta with antioxidant extracts obtained from artichoke canning by-products by ultrasound-assisted technology and quality characterisation of the end product. Int. J. Food Sci. Technol. 2017, 52, 2078–2087. [Google Scholar] [CrossRef]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef] [PubMed]
- Busto, M.; Tarifa, E.E.; Vera, C.R. Coupling solvent extraction units to cyclic adsorption units. Int. J. Chem. Eng. 2018. [Google Scholar] [CrossRef]
- Ilghami, A.; Ghanbarzadeh, S.; Hamishehkar, H. Optimization of the ultrasonic-assisted extraction of phenolic compounds, ferric reducing activity and antioxidant activity of the beta vulgaris using response surface methodology. Pharm. Sci. 2015, 21, 46–50. [Google Scholar] [CrossRef]
- Falleh, H.; Ksouri, R.; Lucchessi, M.-E.; Abdelly, C.; Magné, C. Ultrasound-assisted extraction: Effect of extraction time and solvent power on the levels of polyphenols and antioxidant activity of Mesembryanthemum edule L. aizoaceae shoots. Trop. J. Pharm. Res. 2012, 11, 243–249. [Google Scholar] [CrossRef]
- Feng, H.; Barbosa-Canovas, G.; Weiss, J. (Eds.) Ultrasound technologies for food and bioprocessing. In Food Engineering Series; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7471-6. [Google Scholar]
- Villamiel, M.; Garcia-Perez, J.V.; Montilla, A.; Carcel, J.A.; Benedito, J. (Eds.) Ultrasound in food processing: Recent advances. In IFST Advances in Food Science; John Wiley & Son: West Sussex, UK, 2017; ISBN 9781118964163. [Google Scholar]
- Lohani, U.C.; Muthukumarappan, K.; Meletharayil, G.H. Application of hydrodynamic cavitation to improve antioxidant activity in sorghum flour and apple pomace. Food Bioprod. Process. 2016, 100, 335–343. [Google Scholar] [CrossRef]
- Lee, A.K.; Lewis, D.M.; Ashman, P.J. Disruption of microalgal cells for the extraction of lipids for biofuels: Processes and specific energy requirements. Biomass Bioenergy 2012, 46, 89–101. [Google Scholar] [CrossRef]
- Chemat, F.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Gogate, P.R. Hydrodynamic cavitation for food and water processing. Food Bioprocess Technol. 2011, 4, 996–1011. [Google Scholar] [CrossRef]
- Daghero, P.; Cravotto, G. Plant Composition Comprising the Phytocomplex of A plant Species and Process for Preparation. EP 2520182A1, 3 May 2011. [Google Scholar]
- Crudo, D.; Bosco, V.; Cavaglià, G.; Grillo, G.; Mantegna, S.; Cravotto, G. Process intensification in biodiesel production with a rotor-stator type generator of hydrodynamic cavitation. Ultrason. Sonochem. 2016, 33, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Omiccioli, G.; Stevanato, L. An Improved Sonochemical Reactor. Ultrason. Sonochem. 2005, 12, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Cintas, P.; Mantegna, S.; Calcio Gaudino, E.; Cravotto, G. A new pilot flow-reactor for high-intensity ultrasound irradiation. Application to the synthesis of biodiesel. Ultrason. Sonochem. 2010, 17, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Cintas, P. Power ultrasound in organic synthesis: Moving cavitational chemistry from academia to innovative and large-scale applications. Chem. Soc. Rev. 2006, 35, 180–196. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, J.; Badve, M.; Rajoriya, S.; George, S.; Saharan, V.K.; Pandit, A.B. Hydrodynamic cavitation: An emerging technology for the intensification of various chemical and physical processes in a chemical process industry. Rev. Chem. Eng. 2017, 33, 433–468. [Google Scholar] [CrossRef]
- Dular, M. Hydrodynamic cavitation damage in water at elevated temperatures. Wear 2016, 346/347, 78–86. [Google Scholar] [CrossRef]
- BOB Service Srl; Cavaglià, G. Apparatus and Method for Enhancing Phase Contact. PCT/IB2018/050850, 10 February 2017. [Google Scholar]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.-H. Green extraction methods for polyphenols from plant matrices and their byproducts: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef]
- Cravotto, G.; Binello, A.; Orio, L. Green extraction techniques for high-quality natural products. AgroFOOD Ind. Hi-Tech 2011, 22, 24–36. [Google Scholar]
- Alexandru, L.; Cravottom, G.; Giordanam, L.; Binellom, A.; Chematm, F. Ultrasound-assisted extraction of clove buds using batch- and flow-reactors: A comparative study on a pilot scale. Innov. Food Sci. Emerg. Technol. 2013, 20, 167–172. [Google Scholar] [CrossRef]
- Rinaldi, L.; Wu, Z.; Giovando, S.; Bracco, M.; Crudo, D.; Bosco, V.; Cravotto, G. Oxidative polymerization of waste cooking oil with air under hydrodynamic cavitation. Green Process Synth. 2017, 6, 425–432. [Google Scholar] [CrossRef]
- Crudo, D.; Bosco, V.; Cavaglià, G.; Mantegna, S.; Battaglia, L.; Cravotto, G. Process intensification in food industry: Hydrodynamic and acoustic cavitation for fresh milk treatment. AgroFOOD Ind. Hi-Tech 2014, 25, 55–59. [Google Scholar]
- Albanese, L.; Ciriminna, R.; Meneguzzo, F.; Pagliaro, M. Beer-brewing powered by controlled hydrodynamic cavitation: Theory and real-scale experiments. J. Clean. Prod. 2017, 142, 1457–1470. [Google Scholar] [CrossRef]
- Petkovšek, M.; Mlakar, M.; Levstek, M.; Stražar, M.; Širok, B.; Dular, M. A novel rotation generator of hydrodynamic cavitation for waste-activated sludge disintegration. Ultrason. Sonochem. 2015, 26, 408–414. [Google Scholar] [CrossRef] [PubMed]
- ANDRITZ Group. Available online: https://www.andritz.com/group-en (accessed on 6 August 2018).
- Hydro Air Research Italia. Available online: https://www.hydair.com/ (accessed on 6 August 2018).
- Tecnoimpianti Water Treatment. Available online: https://www.tecnoimp.com/ (accessed on 6 August 2018).
- Tsibranska, I.; Tylkowski, B. Concentration of polyphenols by integrated membrane operations. Integr. Membr. Oper. Food Prod. 2014, 269–293. [Google Scholar] [CrossRef]
- Uyttebroek, M.; Vandezande, P.; Van Dael, M.; Vloemans, S.; Noten, B.; Bongers, B.; Porto-Carrero, W.; Muniz Unamunzaga, M.; Bulut, M.; Lemmens, B. Concentration of phenolic compounds from apple pomace extracts by nanofiltration at lab and pilot scale with a techno-economic assessment. J. Food Process Eng. 2018, 41, e12629. [Google Scholar] [CrossRef]
- Kammerer, D.R.; Carle, R.; Stanley, R.A.; Saleh, Z.S. Pilot-scale resin adsorption as a means to recover and fractionate apple polyphenols. J. Agric. Food Chem. 2010, 58, 6787–6796. [Google Scholar] [CrossRef] [PubMed]
- Soccio, M.; Laus, M.N.; Alfarano, M.; Dalfino, G.; Panunzio, M.F.; Pastore, D. Antioxidant/oxidant balance as a novel approach to evaluate the effect on serum of long-term intake of plant antioxidant-rich foods. J. Funct. Foods 2018, 40, 778–784. [Google Scholar] [CrossRef]
- El Gharras, H. Polyphenols: Food sources, properties and applications—A review. Int. J. Food Sci. Technol. 2009, 44, 2512–2518. [Google Scholar] [CrossRef]
- Shi, J.; Yu, J.; Pohorly, J.E.; Kakuda, Y. Polyphenolics in grape seeds, biochemistry and functionality. J. Med. Food. 2004, 6. [Google Scholar] [CrossRef] [PubMed]
- El, S.N.; Karakaya, S. Olive tree (Olea europaea) leaves: Potential beneficial effects on human health. Nutr. Rev. 2009, 67, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Meneguzzo, F.; Delisi, R.; Pagliaro, M. Olive biophenols as new antioxidant additives in food and beverage. Chem. Sel. 2017, 2, 1360–1365. [Google Scholar] [CrossRef]
- Grand View Research Inc. Available online: www.grandviewresearch.com (accessed on 6 August 2018).







© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cravotto, G.; Mariatti, F.; Gunjevic, V.; Secondo, M.; Villa, M.; Parolin, J.; Cavaglià, G. Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview. Foods 2018, 7, 130. https://doi.org/10.3390/foods7090130
Cravotto G, Mariatti F, Gunjevic V, Secondo M, Villa M, Parolin J, Cavaglià G. Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview. Foods. 2018; 7(9):130. https://doi.org/10.3390/foods7090130
Chicago/Turabian StyleCravotto, Giancarlo, Francesco Mariatti, Veronika Gunjevic, Massimo Secondo, Matteo Villa, Jacopo Parolin, and Giuliano Cavaglià. 2018. "Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview" Foods 7, no. 9: 130. https://doi.org/10.3390/foods7090130
APA StyleCravotto, G., Mariatti, F., Gunjevic, V., Secondo, M., Villa, M., Parolin, J., & Cavaglià, G. (2018). Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview. Foods, 7(9), 130. https://doi.org/10.3390/foods7090130





