Carcass Quality, Meat Quality and Sensory Properties of the Dual-Purpose Chicken Lohmann Dual
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Declaration
2.2. Materials
Animals and Sample Collection
2.3. Methods
2.3.1. Carcass-Characteristics and Meat Quality Traits
2.3.2. Descriptive Sensory Analysis
2.4. Chemicals
2.5. Data Analysis
3. Results and Discussion
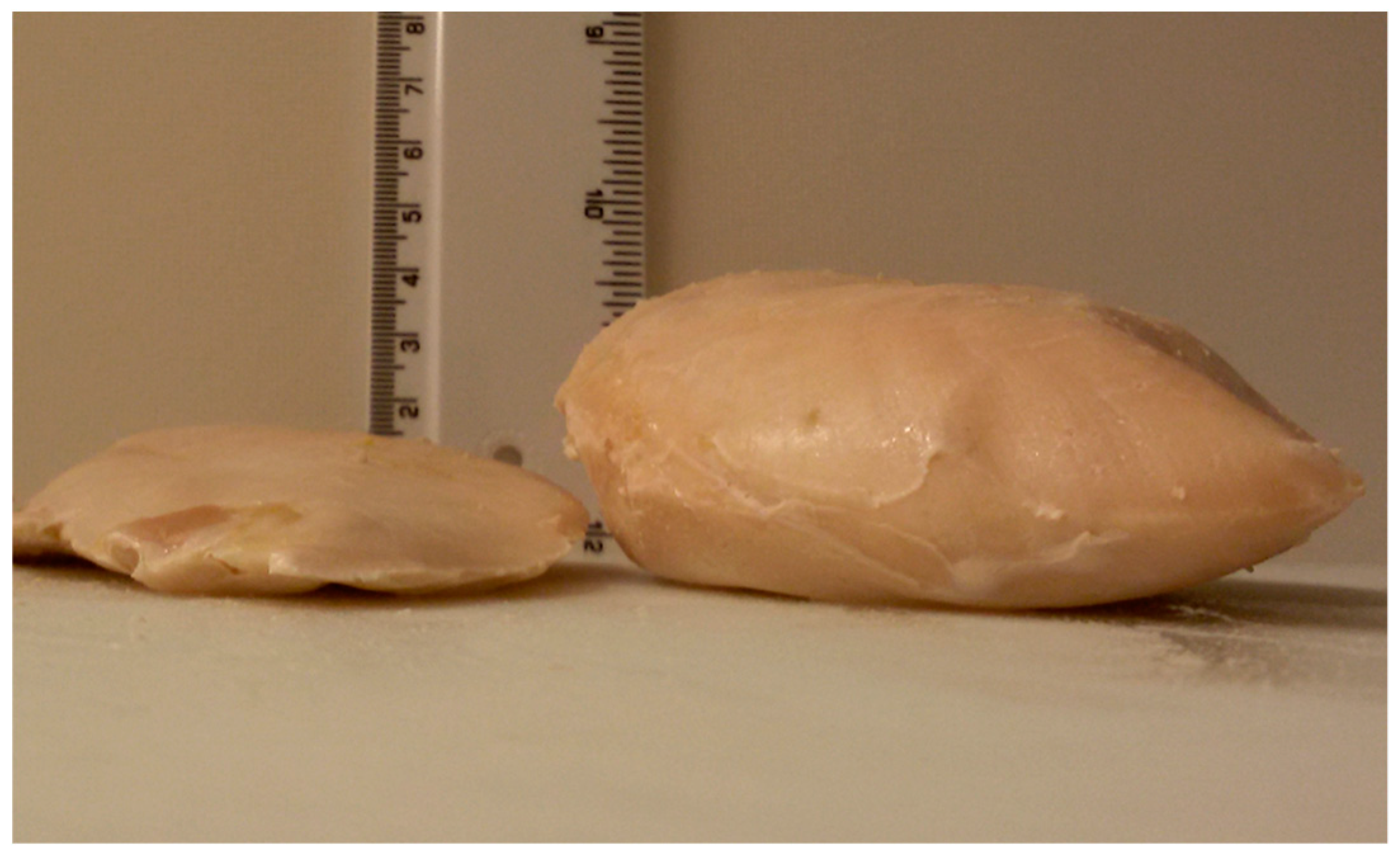
3.1. Carcass-Characteristics and Meat Quality Traits
3.2. Objective Sensory Characteristics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Koenig, M.; Hahn, G.; Damme, K.; Schmutz, M. Untersuchungen zur Mastleistung und Schlachtkörperzusammensetzung von Stubenküken aus Masthybriden und verschiedenen Legehybridherkünften. Züchtungskunde 2012, 84, 511–522. [Google Scholar]
- Bruijnis, M.R.N.; Blok, V.; Stassen, E.N.; Gremmen, H.G.J. Moral “lock-in” in responsible innovation, the ethical and social aspects of killing day-old chicks and its alternatives. J. Agric. Environ. Ethics 2015, 28, 939–960. [Google Scholar] [CrossRef]
- Damme, K.; Ristic, M. Fattening performance, meat yield and economic aspects of meat and layer type hybrids. Worlds Poult. Sci. J. 2003, 59, 50–53. [Google Scholar]
- Gerken, M.; Jaenecke, D.; Kreuzer, M. Growth, behaviour and carcass characteristics of egg-type cockerels compared to male broilers. Worlds Poult. Sci. J. 2003, 59, 46–49. [Google Scholar]
- Murawska, D.; Bochno, R. Comparison of the slaughter quality of layer-type cockerels and broiler chickens. J. Poult. Sci. 2007, 44, 105–110. [Google Scholar] [CrossRef]
- Ellendorff, F.; Klein, S. Current knowledge on sex determination and sex diagnosis: Potential solutions. Worlds Poult. Sci. J. 2003, 59, 7. [Google Scholar] [CrossRef]
- Galli, R.; Preusse, G.; Uckermann, O.; Bartels, T.; Krautwald-Junghanns, M.E.; Koch, E.; Steiner, G. In ovo sexing of domestic chicken eggs by Raman spectroscopy. Anal. Chem. 2016, 88, 8657–8663. [Google Scholar] [CrossRef] [PubMed]
- Weissmann, A.; Reitemeier, S.; Hahn, A.; Gottschalk, J.; Einspanier, A. Sexing domestic chicken before hatch: A new method for in ovo gender identification. Theriogenology 2013, 80, 199–205. [Google Scholar] [CrossRef]
- Weissmann, A.; Gottschalk, J.; Reitemeier, S.; Preisinger, R.; Einspanier, A. In ovo-gender identification in laying hen hybrids: Effects on hatching and production performance. Europ. Poult. Sci. 2014, 78, 1–12. [Google Scholar]
- Göhler, D.; Fischer, B.; Meissner, S. In-ovo sexing of 14-day-old chicken embryos by pattern analysis in hyperspectral images (VIS/NIR spectra): A non-destructive method for layer lines with gender-specific down feather color. Poult. Sci. 2017, 96, 1–4. [Google Scholar] [CrossRef]
- Icken, W.; Schmutz, M.; Cavero, D.; Preisinger, R. Dual Purpose Chickens: The Breeder’s Answer to the Culling of Day-Old Male Layers. In Proceedings of the IX European Symposium on Poultry Welfare, Uppsala, Sweden, 2013; Available online: https://www.ltz.de/de-wAssets/docs/dual/IX-European-symposium-on-poultry-welfare.pdf (accessed on 22 September 2018).
- Quentin, M.; Bouvarel, I.; Berri, C.; Le Bihan-Duval, E.; Baeza, E.; Jégo, Y.; Picard, M. Growth, carcass composition and meat quality response to dietary concentrations in fast-, medium- and slow-growing commercial broilers. Anim. Res. 2003, 52, 65–77. [Google Scholar] [CrossRef]
- Koreleski, J.; Swiatkiewicz, S.; Arczewska-Wlosek, A. Dual-purpose type of slow-growing chickens for meat production. Ann. Anim. Sci. 2008, 8, 91–97. [Google Scholar]
- Castellini, C.; Berri, C.; Le Bihan-Duval, E.; Martino, G. Qualitative attributes and consumer perception of organic and free-range poultry meat. Worlds Poult. Sci. J. 2008, 64, 500–512. [Google Scholar] [CrossRef]
- Chumngoen, W.; Tan, F.J. Relationships between descriptive sensory attributes and physicochemical analysis of broiler and Taiwan native chicken breast meat. Asian-Australas. J. Anim. Sci. 2015, 28, 1028–1037. [Google Scholar] [CrossRef] [PubMed]
- Horsted, K.; Allesen-Holm, B.H.; Hermansen, J.E.; Kongsted, A.G. Sensory profiles of breast meat from broilers reared in an organic niche production system and conventional standard broilers. J. Sci. Food Agric. 2011, 92, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Kokoszynski, D.; Bernacki, Z.; Korytkowska, H.; Krajewski, K.; Skrobiszewska, L. Carcass composition and physicochemical and sensory properties of meat from broiler chickens of different origin. J. Cent. Eur. Agric. 2013, 14, 781–793. [Google Scholar] [CrossRef]
- Molette, C.; Rémignon, H.; Babilé, R. Maintaining muscles at a high post-mortem temperature induces PSE-like meat in turkey. Meat Sci. 2003, 63, 525–532. [Google Scholar] [CrossRef]
- Xiong, R.; Cavitt, L.C.; Meullenet, J.F.; Owens, C.M. Comparison of Allo-Kramer, Warner-Bratzler and razor blade shears for predicting sensory tenderness of broiler breast meat. J. Texture Stud. 2006, 37, 179–199. [Google Scholar] [CrossRef]
- Stone, H.; Sidel, J.L.; Bloomquist, J. Quantitative descriptive analysis. Cereal Food World 1980, 25, 642–644. [Google Scholar]
- Lawless, H.T. Appendix A: Overview of sensory evaluation. In Quantitative Sensory Analysis: Psychophysics, Models and Intelligent Design; Wiley–Blackwell: Chichester, UK, 2013; pp. 361–376. ISBN 978-970-470-67346-1. [Google Scholar]
- International Organization for Standardization. ISO 8586:2012 Sensory Analysis—General Guidelines for the Selection, Training and Monitoring of Selected Assessors and Expert Sensory Assessors. 1993. Available online: https://www.iso.org/obp/ui/#iso:std:iso:8586:ed-1:v2:en (accessed on 1 August 2016).
- International Organization for Standardization. ISO 8589:2007 Sensory Analysis—General Guidance for the Design of Test Rooms. 2007. Available online: https://www.iso.org/standard/36385.html (accessed on 1 August 2016).
- Farmer, L.J.; Perry, G.C.; Lewis, P.D.; Nute, G.R.; Piggott, J.R.; Patterson, R.L.S. Responses of two genotypes of chicken to the diets and stocking densities typical of UK and “Label Rouge” production systems: II. Sensory attributes. Meat Sci. 1997, 47, 77–93. [Google Scholar] [CrossRef]
- Fanatico, A.C.; Pillai, P.B.; Cavitt, L.C.; Owens, C.M.; Emmert, J.L. Evaluation of slower-growing broiler genotypes grown with and without outdoor access: Growth performance and carcass yield. Poult. Sci. 2005, 84, 1321–1327. [Google Scholar] [CrossRef]
- Mueller, S.; Kreuzer, M.; Siegrist, M.; Mannale, K.; Messikommer, R.E.; Gangnat, I.D.M. Carcass and meat quality of dual-purpose chickens (Lohmann Dual, Belgian Malines, Schweizerhuhn) in comparison to broiler and layer chicken types. Poult. Sci. 2018, 97, 3325–3336. [Google Scholar] [CrossRef] [PubMed]
- Habig, C.; Beyerbach, M.; Kemper, N. Comparative analyses of layer males, dual purpose males and mixed sex broilers kept for fattening purposes regarding their floor space covering, weight-gain and several animal health traits. Europ. Poult. Sci. 2016, 80, 1–10. [Google Scholar]
- Janisch, S.; Krischek, C.; Wicke, M. Color values and other meat quality characteristics of breast muscles collected from 3 broiler genetic lines slaughtered at 2 ages. Poult. Sci. 2011, 90, 1774–1781. [Google Scholar] [CrossRef] [PubMed]
- Aviagen. Broiler Ross 308 Performance Objectives. Performance Objectives. 2014, pp. 2–16. Available online: http://eu.aviagen.com/assets/Tech_Center/Ross_Broiler/Ross-308-Broiler-PO-2014-EN.pdf (accessed on 1 March 2016).
- Nielsen, B.L.; Thomsen, M.G.; Sørensen, J.P.; Young, J.F. Feed and strain effects on the use of outdoor areas by broilers. Br. Poult. Sci. 2003, 44, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Fanatico, A.C.; Pillai, P.B.; Hester, P.Y.; Falcone, C.; Mench, J.A.; Owens, C.A.; Emmert, J.L. Performance, livability, and carcass yield of slow- and fast-growing chicken genotypes fed low-nutrient or standard diets and raised indoors or with outdoor access. Poult. Sci. 2008, 87, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.P.; Chen, J.L.; Zhao, G.P.; Zheng, M.Q.; Jiang, R.R.; Wen, J. Live performance, carcass composition, and blood metabolite responses to dietary nutrient density in two distinct broiler breeds of male chickens. Poult. Sci. 2009, 88, 2575–2584. [Google Scholar] [CrossRef] [PubMed]
- Mikulski, D.; Celej, J.; Jankowski, J.; Majewska, T.; Mikulska, M. Growth performance, carcass traits and meat quality of slower-growing and fast-growing chickens raised with and without outdoor access. Asian-Aust. J. Anim. Sci. 2011, 24, 1407–1416. [Google Scholar] [CrossRef]
- Duclos, M.J.; Berri, C.; Le Bihan-Duval, E. Muscle growth and meat quality. J. Appl. Poult. Res. 2007, 16, 107–112. [Google Scholar] [CrossRef]
- Wattanachant, S.; Benjakul, S.; Ledward, D.A. Composition, color, and texture of Thai indigenous and broiler chicken muscles. Poult. Sci. 2004, 83, 123–128. [Google Scholar] [CrossRef]
- Debut, M.; Baeza, E.; Sellier, N.; Arnould, C.; Guémené, D.; Jehl, N.; Boutten, B.; Jégo, Y.; Beaumont, C.; Le Bihan-Duval, E. Variation of chicken technological meat quality in relation to genotype and preslaughter stress conditions. Poult. Sci. 2003, 82, 1829–1838. [Google Scholar] [CrossRef] [PubMed]
- Debut, M.; Berri, C.; Arnould, C.; Guémené, D.; Santé-Lhoutellier, V.; Sellier, N.; Baéza, E.; Jehl, N.; Jégo, Y.; Beaumont, C.; et al. Behavioural and physiological responses of three chicken breeds to pre-slaughter shackling and acute heat stress. Br. Poult. Sci. 2005, 46, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Berri, C.; Debut, M.; Santé-Lhoutellier, V.; Arnould, C.; Boutten, B.; Sellier, N.; Baéza, E.; Jehl, N.; Jégo, Y.; Duclos, M.J.; et al. Variations in chicken breast meat quality: Implications of struggle and muscle glycogen content at death. Br. Poult. Sci. 2005, 46, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Chabault, M.; Baéza, E.; Gigaud, V.; Chartrin, P.; Chapuis, H.; Boulay, M.; Arnauld, C.; D’Abbadie, F.; Berri, C.; Le Bihan-Duval, E. Analysis of a slow-growing line reveals wide genetic variability of carcass and meat quality-related traits. BMC Genet. 2012, 13, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, D.L. Poultry meat quality. Worlds Poult. Sci. J. 2002, 58, 131–145. [Google Scholar] [CrossRef]
- Berri, C.; Wacrenier, N.; Millet, N.; Le Bihan-Duval, E. Effect of selection for improved body composition on muscle and meat characteristics of broilers from experimental and commercial lines. Poult. Sci. 2001, 80, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.P.; Zhao, G.P.; Jiang, R.R.; Zheng, M.Q.; Chen, J.L.; Liu, R.R.; Wen, J. Effects of diet-induced differences in growth rate on metabolic, histological, and meat-quality properties of 2 muscles in male chickens of 2 distinct broiler breeds. Poult. Sci. 2012, 91, 237–247. [Google Scholar] [CrossRef]
- Le Bihan-Duval, E.; Millet, N.; Remignon, H. Broiler meat quality: Effect of selection for increased carcass quality and estimates of genetic parameters. Poult. Sci. 1999, 78, 822–826. [Google Scholar] [CrossRef]
- Santos, A.L.; Sakomura, N.K.; Freitas, E.R.; Sá Fortes, C.M.L.; Carrilho, E.N.V.M.; Fernandes, J.B.K. Growth, performance, carcass yield and meat quality of three broiler chickens strains. Rev. Bras. Zootec. 2005, 34, 1589–1598. [Google Scholar] [CrossRef]
- Fanatico, A.C.; Pillai, P.B.; Emmert, J.L.; Owens, C.M. Meat quality of slow- and fast-growing chicken genotypes fed low-nutrient or standard diets and raised indoors or with outdoor access. Poult. Sci. 2007, 86, 2245–2255. [Google Scholar] [CrossRef]
- Berri, C.; Le Bihan-Duval, E.; Debut, M.; Santé-Lhoutellier, V.; Baéza, E.; Gigaud, V.; Jégo, Y.; Duclos, M.J. Consequence of muscle hypertrophy on characteristics of pectoralis major muscle and breast meat quality of broiler chickens. J. Anim. Sci. 2007, 85, 2005–2011. [Google Scholar] [CrossRef] [PubMed]
- Honikel, K.O. How to measure the water-holding capacity of meat? Recommendation of standardized methods. In Evaluation and Control of Meat Quality in Pigs, CEC Agricultural Research Programme; Martinus Nijhoff: Leiden, Belgium, 1987; pp. 129–130. [Google Scholar]
- Berri, C.; Le Bihan-Duval, E.; Baéza, E.; Chartrin, P.; Picgirard, L.; Jehl, N.; Quentin, M.; Picard, M.; Duclos, M.J. Further processing characteristics of breast and leg meat from fast-, medium-and slow-growing commercial chickens. Anim. Res. 2005, 54, 123–134. [Google Scholar] [CrossRef]
- Nakamura, R.; Sekoguchi, S.; Sato, Y. The contribution of intramuscular collagen to the tenderness of meat from chickens with different ages. Poult. Sci. 1975, 54, 1604–1612. [Google Scholar] [CrossRef]
- Sonaiya, E.B.; Ristic, M.; Klein, F.W. Effect of environmental temperature, dietary energy, age and sex on broiler carcase portions and palatability. Br. Poult. Sci. 1990, 31, 121–128. [Google Scholar] [CrossRef]
- Krischek, C.; Gerken, M.; Wicke, M. Effects of a higher incubation temperature between embryonic day 9 and 12 on growth and meat quality characteristics of turkeys. Br. Poult. Sci. 2013, 54, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Bouton, P.E.; Harris, P.V. A comparison of some objective methods used to assess meat tenderness. J. Food Sci. 1972, 37, 218–221. [Google Scholar] [CrossRef]
- Oshima, I.; Iwamoto, H.; Tabata, S.; Ono, Y.; Ishibashi, A.; Shiba, N.; Miyachi, M.; Gotoh, T.; Nishimura, S. Comparative observations on the growth changes of the histochemical property and collagen architecture of the musculus pectoralis from Silky, layer-type and meat-type cockerels. Anim. Sci. J. 2007, 78, 619–630. [Google Scholar] [CrossRef]
- Fernandez, X.; Sante, V.; Baeza, E.; Le Bihan-Duval, E.; Berri, C.; Remignon, H.; Babile, R.; Le Pottier, G.; Millet, N.; Berge, P.; et al. Post mortem muscle metabolism and meat quality in three genetic types of turkey. Br. Poult. Sci. 2001, 42, 462–469. [Google Scholar] [CrossRef]
- Nakamura, Y.N.; Iwamoto, H.; Shiba, N.; Miyachi, H.; Tabata, S.; Nishimura, S. Growth changes of the collagen content and architecture in the pectoralis and iliotibialis lateralis muscles of cockerels. Br. Poult. Sci. 2004, 45, 37–41. [Google Scholar] [CrossRef]
- Glitsch, K. Consumer perceptions of fresh meat quality: Cross-national comparison. Br. Food J. 2000, 102, 177–194. [Google Scholar] [CrossRef]
- Jahan, K.; Paterson, A.; Piggott, J.R. Sensory quality in retailed organic, free range and corn-fed chicken breast. Food Res. Int. 2005, 38, 495–503. [Google Scholar] [CrossRef]
- Castellini, C.; Mugnai, C.; Dal Bosco, A. Effect of organic production system on broiler carcass and meat quality. Meat Sci. 2002, 60, 219–225. [Google Scholar] [CrossRef]
- Sandercock, D.A.; Nute, G.R.; Hocking, P.M. Quantifying the effects of genetic selection and genetic variation for body size, carcass composition, and meat quality in the domestic fowl (Gallus domesticus). Poult. Sci. 2009, 88, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Neta, E.R.D.; Johanningsmeier, S.D.; Drake, M.A.; McFeeters, R.F. Effects of pH adjustment and sodium ions on sour taste intensity of organic acids. J. Food Sci. 2009, 74, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, T.; Kato, H. Taste of free amino acids and peptides. Food Rev. Int. 2009, 2, 175–194. [Google Scholar] [CrossRef]
- Wattanachant, S.; Benjakul, S.; Ledward, D.A. Microstructure and thermal characteristics of Thai indigenous and broiler chicken muscles. Poult. Sci. 2005, 84, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Lyon, C.E.; Hamm, D.; Thomson, J.E. pH and tenderness of broiler breast meat deboned various times after chilling. Poult. Sci. 1985, 64, 307–310. [Google Scholar] [CrossRef]
- Allen, C.D.; Russell, S.M.; Fletcher, D.L. The relationship of broiler breast meat color and pH to shelf-life and odor development. Poult. Sci. 1997, 76, 1042–1046. [Google Scholar] [CrossRef]


| Name of Attribute | Definition | How to Determine | Reference Material | Minimal and Maximal Specifications |
|---|---|---|---|---|
| Dimension: Appearance | ||||
| Color intensity | intensity of color on the surface | color intensity on the surface | bright/pale to dark | |
| Fibrousness | size of visible fibers on the surface | evaluation on the surface | single hair (minimum), bundles on the picture (7), rough bundles (maximum) | no visible (hair) to clearly visible fibers bundles (rough bundles) |
| Elasticity/springiness | ability to recover into original form after being pressed with a fork | press the entire surface with the fork | soft cheese (minimum), toast (middle), wine gum (maximum) | inelastic (shows imprints like soft cheese) to toast to highly elastic (springs back like wine gum) |
| Dimension: Smell | ||||
| Intensity of smell | overall intensity of smell | fork the sample and evaluate the smell on the bottom side | weak to very intensive | |
| ‘Stable-like’ smell | intensity of smell like dung/animals/housing | skatole, NH3 | not perceivable to highly perceivable | |
| ‘Metallic/blood-like’ smell | intensity of smell like metal or blood | blood, iron, old money | not perceivable to highly perceivable | |
| ‘Meaty’ smell (cooked chicken) | intensity of smell like cooked meat, especially poultry meat | chicken broth, bouillon | not perceivable to highly perceivable | |
| Dimension: Taste and Flavor | ||||
| Intensity of taste | overall intensity of taste | evaluation after seven to ten chews with closed mouth | weak to very intensive | |
| Sweet taste | intensity of sweetness | 2.1 g saccharose (middle); 4.2 g saccharose (maximum) | not perceivable to highly perceivable | |
| Sour taste | intensity of sour taste | 0.14 g citric acid; 0.28 g citric (high, 80%) | not perceivable to highly perceivable | |
| Bitter taste | intensity of bitterness | 0.115 g caffeine (middle); 0.21 g caffeine (maximum) | not perceivable to highly perceivable | |
| ‘Metallic, blood-like’ taste | intensity of metallic or blood-like taste (fresh drip loss) | iron sulfate, blood, ‘Kräuterblut’ | not perceivable to highly perceivable | |
| ‘Meaty’ taste (cooked chicken) | intensity of meaty taste (like cooked chicken) | chicken broth | not perceivable to highly perceivable | |
| Dimension: Aftertaste | ||||
| Intensity of aftertaste | allover-intensity of aftertaste | evaluation 5 s after swallowing | not perceivable to highly perceivable | |
| Dimension: Texture | ||||
| Firmness/consistency | power, to divide the sample with the incisors | use your incisors and start biting on the surface to bite a 1 cm-piece off | soft cheese (minimum), Gouda cheese (middle), carrot/Werthers Original Bonbon (maximum) | soft (incisors cut easily through the sample) to firm (sample crushes) |
| Juiciness | amount of juice while chewing | chewing with the molars for two to three times | Banana (minimum), cucumber (middle), mandarin (maximum) | not juicy to highly juicy |
| Adhesiveness | coherence while chewing and adhesion to the molars | chewing with the molars for two to three times | cucumber (minimum), gouda cheese (middle), toffee-bonbon/dry fruit (maximum) | not adhesive to highly adhesive |
| Crumbliness | amount of particles existing before swallowing and rate for coherence | evaluation of the particles/fibers | mash (minimum), gouda cheese (middle), shortbread (maximum) | not crumbly to crumbly |
| Tenderness | power needed to chew | chewing until swallowing | jerky meat (minimum), seasoned apricot (maximum | chewy to tender |
| Ross (n = 30) | Lohmann Dual (n = 30) | df | p-Values | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Carcass weight (g) | 2182.5 | 131.3 | 1415.7 | 126.7 | 58.0 | <0.0001 |
| Breast weight (g) | 549.1 | 50.2 | 179.6 | 20.6 | 38.6 | <0.0001 |
| Leg weight (g) | 596.9 | 38.6 | 440.9 | 41.3 | 50.1 | <0.0001 |
| Breast yield (%) | 25.1 | 1.3 | 12.7 | 0.8 | 58.0 | <0.0001 |
| Leg yield (%) | 27.4 | 1.2 | 31.2 | 1.1 | 58.0 | <0.0001 |
| Ross (n = 30) | Lohmann Dual (n = 30) | df | p-Values | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| pH 24 h p.m. * | 5.8 | 0.2 | 5.7 | 0.3 | 32.8 | 0.0079 |
| L* 24 h p.m. | 61.1 | 2.1 | 57.0 | 2.9 | 58.0 | <0.0001 |
| a* 24 h p.m. * | 1.9 | 0.8 | 3.1 | 1.1 | 58.0 | <0.0001 |
| b* 24 h p.m. | 5.1 | 1.4 | 1.9 | 1.1 | 58.0 | <0.0001 |
| Drip loss, DL (%) | 0.6 | 0.3 | 1.3 | 0.5 | 44.4 | <0.0001 |
| Thawing loss, TL (%) | 2.2 | 1.0 | 6.9 | 1.7 | 47.0 | <0.0001 |
| Cooking loss, CL (%) * | 25.0 | 3.0 | 22.8 | 2.8 | 58.0 | 0.0045 |
| Shear force, SF (N)* | 5.2 | 0.9 | 7.1 | 1.1 | 58.0 | <0.0001 |
| Shear energy, SF (N x mm) | 11.2 | 2.3 | 15.7 | 3.8 | 48.1 | <0.0001 |
| Ross (n = 24) | LD (n = 48) | df | p-Values | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Dimension: Appearance | ||||||
| Color intensity | 24.0 | 13.5 | 24.0 | 13.3 | 9 | 0.9771 |
| Fibrousness | 35.6 | 22.6 | 28.0 | 18.4 | 9 | 0.0032 |
| Elasticity/springiness | 60.6 | 19.2 | 61.3 | 18.2 | 9 | 0.6624 |
| Dimension: Smell | ||||||
| Intensity of smell | 58.7 | 17.1 | 63.7 | 12.9 | 9 | 0.0540 |
| ‘Stable-like’ smell | 16.8 | 11.9 | 23.2 | 18.2 | 9 | 0.0349 |
| ‘Metallic/blood-like’ smell | 15.5 | 9.6 | 16.6 | 10.6 | 9 | 0.3172 |
| ‘Meaty’ smell (cooked chicken) | 62.4 | 12.6 | 61.0 | 11.3 | 9 | 0.3682 |
| Dimension: Taste | ||||||
| Intensity of taste | 59.6 | 13.1 | 60.4 | 11.8 | 9 | 0.6919 |
| Sweet taste | 28.2 | 17.4 | 18.9 | 17.8 | 9 | 0.0053 |
| Sour taste | 26.8 | 18.2 | 43.6 | 21.7 | 9 | 0.0003 |
| Bitter taste | 9.9 | 8.6 | 13.9 | 13.1 | 9 | 0.0844 |
| ‘Metallic/blood-like’ taste | 14.6 | 9.2 | 14.9 | 10.0 | 9 | 0.8452 |
| ‘Meaty’ taste (cooked chicken) | 60.3 | 13.7 | 58.4 | 12.5 | 9 | 0.3298 |
| Dimension: Aftertaste | ||||||
| Intensity of aftertaste | 42.7 | 16.8 | 41.5 | 17.6 | 9 | 0.3755 |
| Dimension: Texture | ||||||
| Firmness/consistency | 35.8 | 17.0 | 53.2 | 15.7 | 9 | 0.0005 |
| Juiciness | 44.4 | 14.0 | 48.6 | 14.7 | 9 | 0.0501 |
| Adhesiveness | 48.8 | 19.8 | 44.2 | 19.4 | 9 | 0.2918 |
| Crumbliness | 54.4 | 16.8 | 50.9 | 17.5 | 9 | 0.1089 |
| Tenderness | 70.2 | 14.6 | 49.7 | 19.8 | 9 | <0.0001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siekmann, L.; Meier-Dinkel, L.; Janisch, S.; Altmann, B.; Kaltwasser, C.; Sürie, C.; Krischek, C. Carcass Quality, Meat Quality and Sensory Properties of the Dual-Purpose Chicken Lohmann Dual. Foods 2018, 7, 156. https://doi.org/10.3390/foods7100156
Siekmann L, Meier-Dinkel L, Janisch S, Altmann B, Kaltwasser C, Sürie C, Krischek C. Carcass Quality, Meat Quality and Sensory Properties of the Dual-Purpose Chicken Lohmann Dual. Foods. 2018; 7(10):156. https://doi.org/10.3390/foods7100156
Chicago/Turabian StyleSiekmann, Lisa, Lisa Meier-Dinkel, Sabine Janisch, Brianne Altmann, Claudia Kaltwasser, Christian Sürie, and Carsten Krischek. 2018. "Carcass Quality, Meat Quality and Sensory Properties of the Dual-Purpose Chicken Lohmann Dual" Foods 7, no. 10: 156. https://doi.org/10.3390/foods7100156
APA StyleSiekmann, L., Meier-Dinkel, L., Janisch, S., Altmann, B., Kaltwasser, C., Sürie, C., & Krischek, C. (2018). Carcass Quality, Meat Quality and Sensory Properties of the Dual-Purpose Chicken Lohmann Dual. Foods, 7(10), 156. https://doi.org/10.3390/foods7100156





