1. Introduction
Escherichia coli O157:H7 can generate shiga toxins that can cause, with as few as 10 cells, severe hemolytic uremic syndrome in infected humans [
1].
E. coli O157:H7 has been considered an adulterant of raw, non-intact beef products since 1999 [
2]. The United States Department of Agriculture, Food Safety and Inspection Service (USDA-FSIS) defines non-intact beef products as products that have gone through treatments, such as grinding, restructuring, or mechanical tenderization processes, including cubing, needling, and pounding [
3]. These non-intact beef products have been involved in several
E. coli O157:H7 outbreaks in the United States since 2000 [
4]. During non-intact beef production, pathogen cells, such as those of
E. coli O157:H7, on the meat surface may be translocated and trapped in sterile internal tissues, thus protected from thermal destruction if the meat is undercooked. A recent survey showed that 40–58% of US consumers ordered beefsteaks at medium rare (60–62.8 °C) to rare (54.4–57.2 °C), which could potentially put consumers at a high risk from
E. coli O157:H7 contaminated non-intact veal meat if consumers order the same way as beefsteaks [
5].
Thermal processing includes using high temperature to inactive spoilage and foodborne pathogens is one of the most effective and widely used technologies for meat products preservation [
6]. The effectiveness of cooking in inactivating
E. coli O157:H7 contaminated non-intact beef has been documented in numerous studies [
4,
7,
8] indicating that the cooking effectiveness on pathogen inactivation increased in the order of broiling > grilling > frying, the thicker the products the higher reduction achieved, and lower fat content increased thermal inactivation activity.
Veal, which originated from Europe, is the meat from 16–18-week-old calves. In the past 10 years, 25% of American households purchased veal products in restaurants or retail stores at least once every three months [
9]. Different veal cuts, such as cutlet, loin, rib, breast, and shank, are more popular to restaurant consumers due to their unique tenderness and flavor. Moreover, the nutrition of veal products matches the dietary guidelines that are recommended by the American Heart Association, the American Dietetic Association, and the USDA. The veal market generates approximately
$1.5 billion sales each year in the US [
10]. Although veal products have not been implicated in
E. coli outbreaks in the US, since 2009 there have been multiple recalls of veal products amounting to 14,600 lb (ca. 6649 kg) due to possible
E. coli O157:H7 and STEC contamination [
11]. According to the USDA-FSIS, in May 2017, a large veal processor recalled over 5000 lb of ground veal, pork, and beef due to possible non O157:H7 shiga toxin producing
E. coli contamination [
12]. According to the USDA-FSIS, there is a greater prevalence of STEC in veal products than in other beef products. For example, in 2013, the USDA-FSIS in their testing of raw ground beef component samples in federal meat-processing factories discovered 0 (0%) of 733 samples to be positive for
E. coli O157:H7 and three (0.24%) of 1232 samples to be positive for STEC in beef; in contrast, in veal, three (3.49%) of 86 samples were positive for
E. coli O157:H7 and 4 (4.00%) of 100 samples were positive for STEC [
13]. The difference in confirmed STEC-positive samples of veal compared to those of beef is striking and raises the question of whether the consumption of veal poses a greater risk to public health than that of beef. Currently, only two studies have reported the thermal inactivation of
E. coli O157:H7 strains in non-intact veal products [
11,
14].
The safety of beef and veal products is important to the industry and to consumers, but consumers tend to identify the quality of products based on appearance. Cornforth and Jayasingh [
15] stated that color is one of the most important characteristics regarding consumers’ purchasing decisions, even though color is sometimes poorly related to meat quality. Fresh beef or veal meat is often displayed in styrofoam trays and covered with poly-vinyl chloride (PVC) oxygen-permeable films, which allow the rapid development of the desirable bright cherry-red (beef) or light pink color (veal), respectively, due to rapid pigment oxygenation. However, discoloration often occurs within 1 week of shelf time. Currently, the number of studies that focus on the quality changes in veal products during processing, storage and cooking in terms of factors such as water activity, pH, moisture, fat content and color change is very limited.
The objective of this study is to investigate the quality variances, including color variation in non-intact coarse ground beef and veal patties during aerobic storage and cooking and to evaluate the thermal inactivation of E. coli O157:H7 in coarse ground beef and veal patties. We hypothesize that (1) beef and veal patties have similar tendencies in quality change throughout storage and cooking and (2) a higher internal temperature with a longer rest time will increase the inactivation of E. coli O157:H7 in beef and veal patties. The novelty of this study are (1) a detailed side-by-side comparison study of quality attributes and thermal inactivation activity of E. coli O157:H7 between beef and veal and (2) the thermal kinetics study was conducted in a commercial size patties cooked on a griller instead of using small amount of meat heated in water bath.
2. Materials and Methods
2.1. Preparation of Bacterial Strains and Inoculum
Escherichia coli O157:H7 strains ATCC 43895, ATCC 43888, and ATCC 43889 (kindly provided by Beth Whittam, Michigan State University, East Lansing, MI, USA) were cultured and sub-cultured individually in 10 mL of tryptic soy broth (TSB) at 35 °C for 24 h. The three cultures were then mixed and centrifuged (Eppendorf model 5810R, Brinkmann Instruments Inc., Westbury, NY, USA) at 4629× g for 15 min at 4 °C. The harvested cells were washed twice with 10 mL of phosphate-buffered saline (PBS), centrifuged as described above, and re-suspended in 30 mL of fresh PBS. The washed pathogen cells were 10-fold diluted in PBS to obtain an initial inoculum level of 8 log CFU/mL, and then 40 mL of this prepared inoculum was added into 2 kg of coarse ground beef or veal to reach the inoculation level of ~6 log CFU/g.
2.2. Preparation of Non-Intact Ground Veal and Beef Patties
Fresh beef knuckles and veal round top were purchased from a local meat retailer for each replicate. The meat was manually cut into trimmings and then coarse ground in a meat grinder (Gander Mountain #5 Electric Meat Grinder, Saint Paul, MN, USA) with a kidney plate (0.95 cm diameter). The ground meat was then mixed with 40 mL of the aforementioned E. coli O157:H7 inoculum cocktail in a bowl-lift stand mixer (Kitchen Aid Professional 600, Benton Harbor, MI, USA) at medium speed for two minutes to ensure an even distribution of the inoculum into the sample, which simulates E. coli O157:H7 contamination during the preparation of non-intact beef or veal products. A manual hamburger patty maker (Mainstays 6-ounce-patty maker, Walmart, Bentonville, AR) was then used to make beef or veal patties with 180 g of grounded meat. The beef/veal patties (2.1 cm thick and 12.4 cm diameter) were packaged aerobically in foam trays (20 × 25 cm, Pactiv, Lake Forest, IL, USA) with the absorbent pads, covered using air-permeable plastic film (Omni-film, Pliant Corporation, OH, USA) and stored at 4.0 °C for four days.
2.3. Cooking Beef or Veal Patty Samples
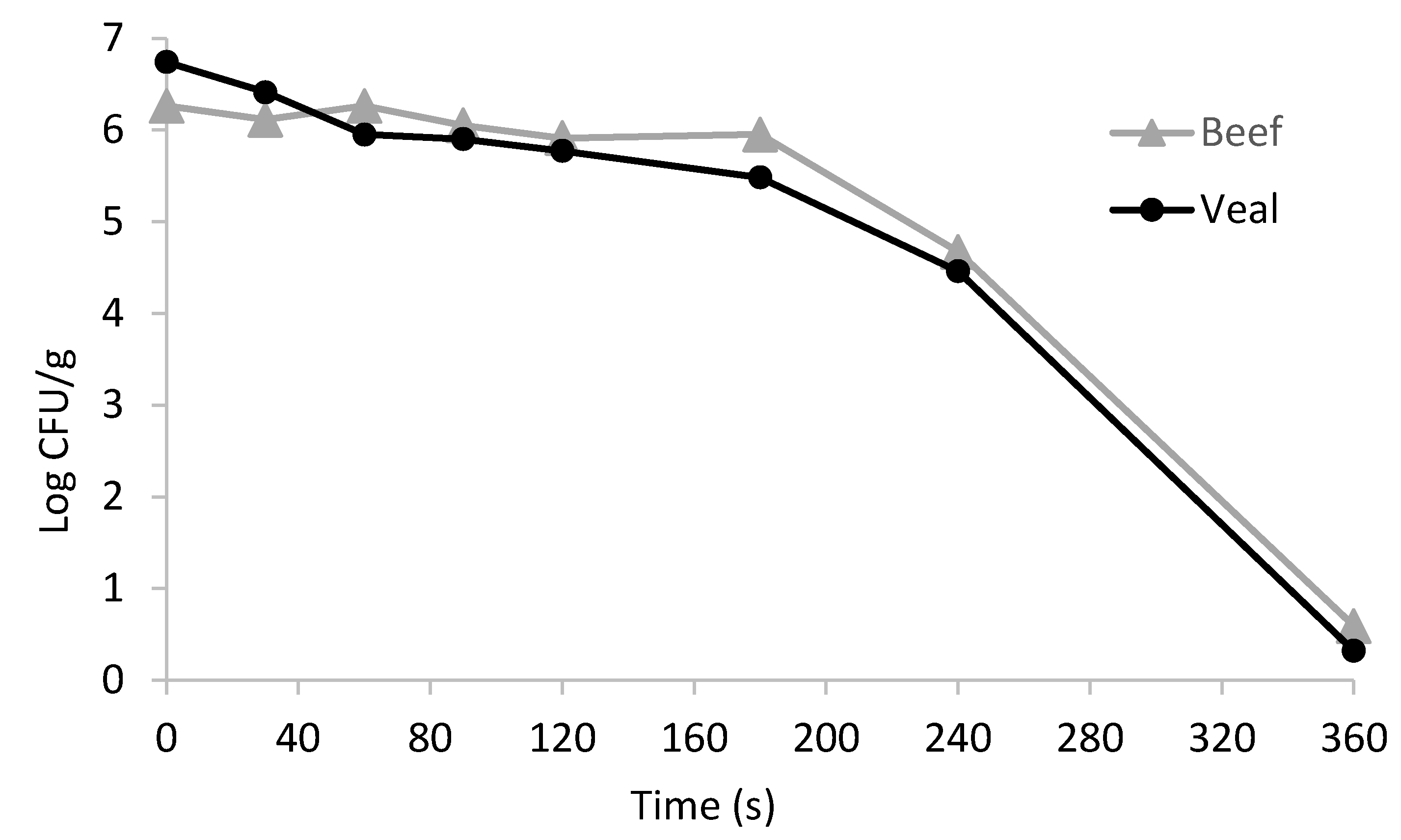
After four days of storage, the beef or veal patties were removed from their packages, weighed, and double pan-broiled in a Farberware grill (Farberware 4-in-1 Grill, Fairfield, CA, USA) with a set-up temperature of 177 °C (or 350 °F) (1) for 0, 30, 60, 90, 120, 180, 240, and 360 s with 0-min rest to determine the thermal dynamic parameters (i.e., D-value, “shoulder”, α) (2) to an internal geometric target temperature of 55, 62.5, 71.1, or 76 °C, followed by a 0.5- or 3.5-min rest. The cooked patties were allowed to rest on the tray after cooking without any cover. Double broiling, also known as contact grilling, is when the food (usually meat, especially burger patties, chicken, and steaks) is cooked on both sides simultaneously by applying two cooking surfaces, from both the bottom and the top, greatly reducing the cooking time. A type-K thermocouple was attached to the geometric center of the patty to monitor the internal temperature throughout cooking using PicoLog (Pico Technology Ltd., Cambridge, UK), a real-time data-recording software [
4,
7,
8]. The cooked meat rest on the tray were also monitored the internal temperature using the same type-K thermocouple. The meat quality test including cooking losses, color, pH, water activity, moisture, and fat content were conducted in a separate study using the uninoculated beef and veal samples with the same storage and cooking treatments and cooled to room temperature after cooking.
2.4. Color Measurement
The objective color of non-intact beef or veal patties was measured on each day of storage and after cooking to 55, 62.5, 71.1, or 76 °C (internal and external parts) using a portable spectrophotometer (HunterLab MiniScan EZ, Reston, VA, USA), with full spectral data being obtained as L* (lightness), a* (redness), and b* (yellowness), along with reflectance data [
16]. For the external surface color measurement, an average value for L*, a*, and b* was determined from the mean of three random readings on the surface from three pieces of each treatment that was used for the color analysis. To measure the internal color of the cooked samples, the beef or veal patties were split transversely across the longitudinal axis to expose the center portion with three random readings from three pieces of each treatment.
2.5. Physical, Chemical and Microbiological Analyses
Cooking losses were determined by measuring the difference in patty weight before cooking and after cooking when the samples had cooled to room temperature. The pH of the meat homogenate was measured after microbial analysis using a digital pH meter (Fisher Scientific, Fair Lawn, NY, USA). The water activity (aw) indicates the availability of water for bacterial growth. The water activity of the uncooked and cooked samples was measured using an AquaLab water activity meter (model series 3, Decagon Devices Inc., Pullman, WA, USA). All of the samples were tested for fat and moisture content at the Meat Science Lab of the University of Illinois at Urbana– Champaign. For microbiological analysis, the individual uncooked or cooked beef or veal samples were transferred to a Whirl-Pak filter bag (1627 mL, 19 × 30 cm, Nasco, Modesto, CA, USA) with a 1:1 ratio of nutrient broth by weight and homogenized (Masticator, IUL Instruments, Barcelona, Spain) for 2 min. Serial 10-fold dilutions of each sample in PBS were surface-plated onto tryptic soy agar (Acumedia, Lansing, MI, USA) supplemented with 0.1% sodium pyruvate (Fisher Scientific, Fair Lawn, NY; TSAP) and MacConkey agar (Acumedia, Lansing, MI, USA) for the enumeration of total bacterial populations and E. coli O157:H7, respectively. Colonies were counted manually after incubation at 35 °C for 48 h. The samples below the detect limit of spread-plating were enriched at 35 °C for 48 h and streak-plated onto MacConkey agar to enrich any cells that were not recovered.
2.6. Data Analysis
The experiment was repeated twice, with three samples in each replicate in quality and microbial thermal inactivation studies. The quality parameters of beef and veal samples, including cooling losses, pH, water activity, fat and moisture content, were analyzed with a one-way ANOVA of SAS. All of the comparisons were performed with
p = 0.05. Microbial populations (log CFU/g) were analyzed using the PROC MIXED procedure of Statistical Analysis System (SAS; version 9.3, SAS Institute Inc., Cary, NC, USA), with independent variables including beef or veal, cooked internal temperatures, rest time, and interactions between two or three variables. USDA-Integrated-Predictive-Modeling-Program software [
17], provided by Dr. Lihan Huang, was used to estimate parameters of the survival of the pathogen cells in ground beef and veal samples during thermal processing with various heating time. The means and standard deviations were calculated, and the mean differences between treatments were determined using the Least Significant Difference (LSD) function for multiple comparisons at a significance level of α = 0.05.