Characterization of Retail Conventional, Organic, and Grass Full-Fat Butters by Their Fat Contents, Free Fatty Acid Contents, and Triglyceride and Fatty Acid Profiling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Chemicals
2.3. Analytical Methods
2.3.1. Fat Content
2.3.2. Free Fatty Acid Content
2.3.3. Triglyceride Analysis
2.3.4. Fatty Acid Analysis
2.4. Data Analysis
3. Results
3.1. Fat Content and Free Fatty Acids
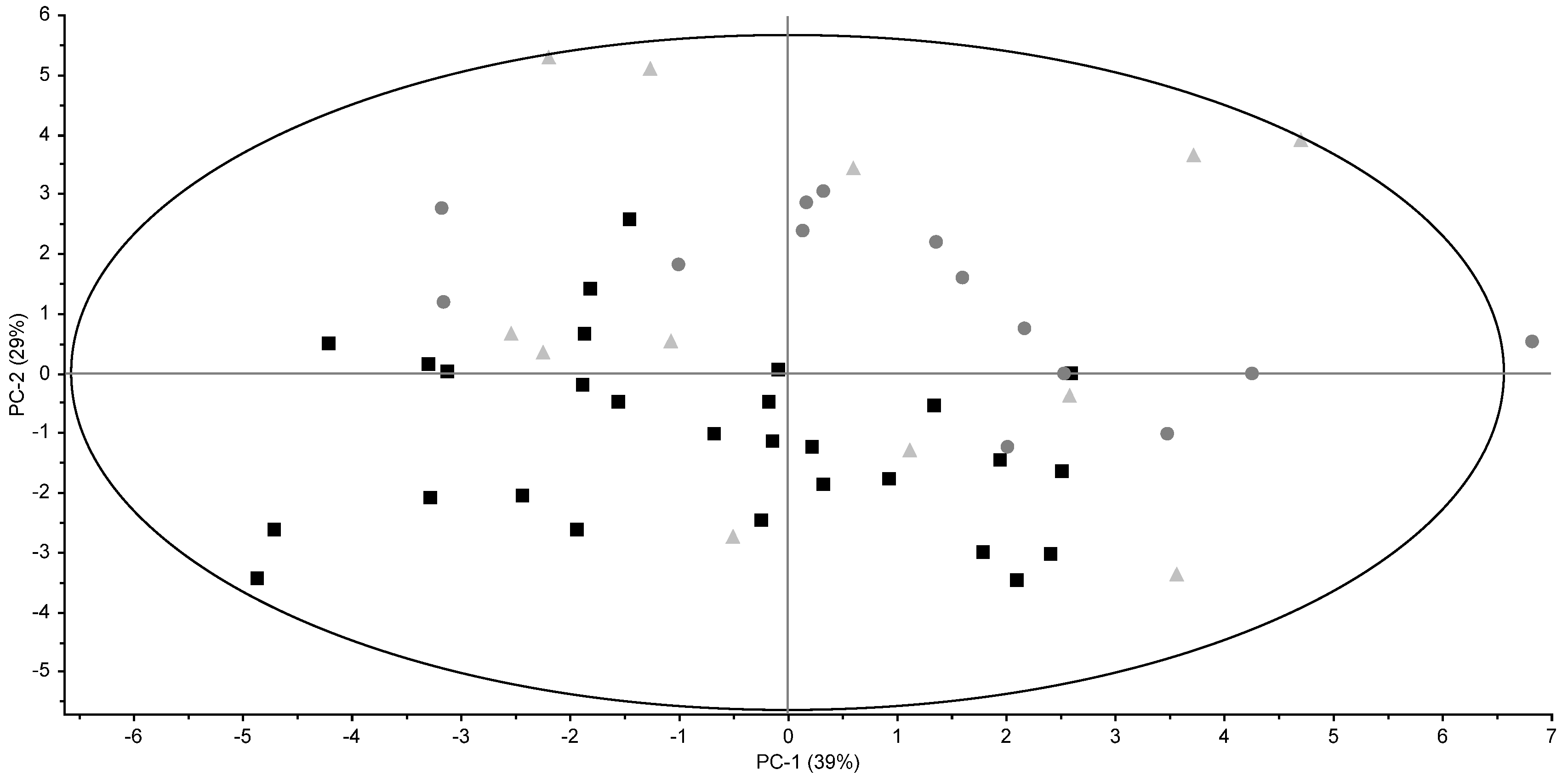
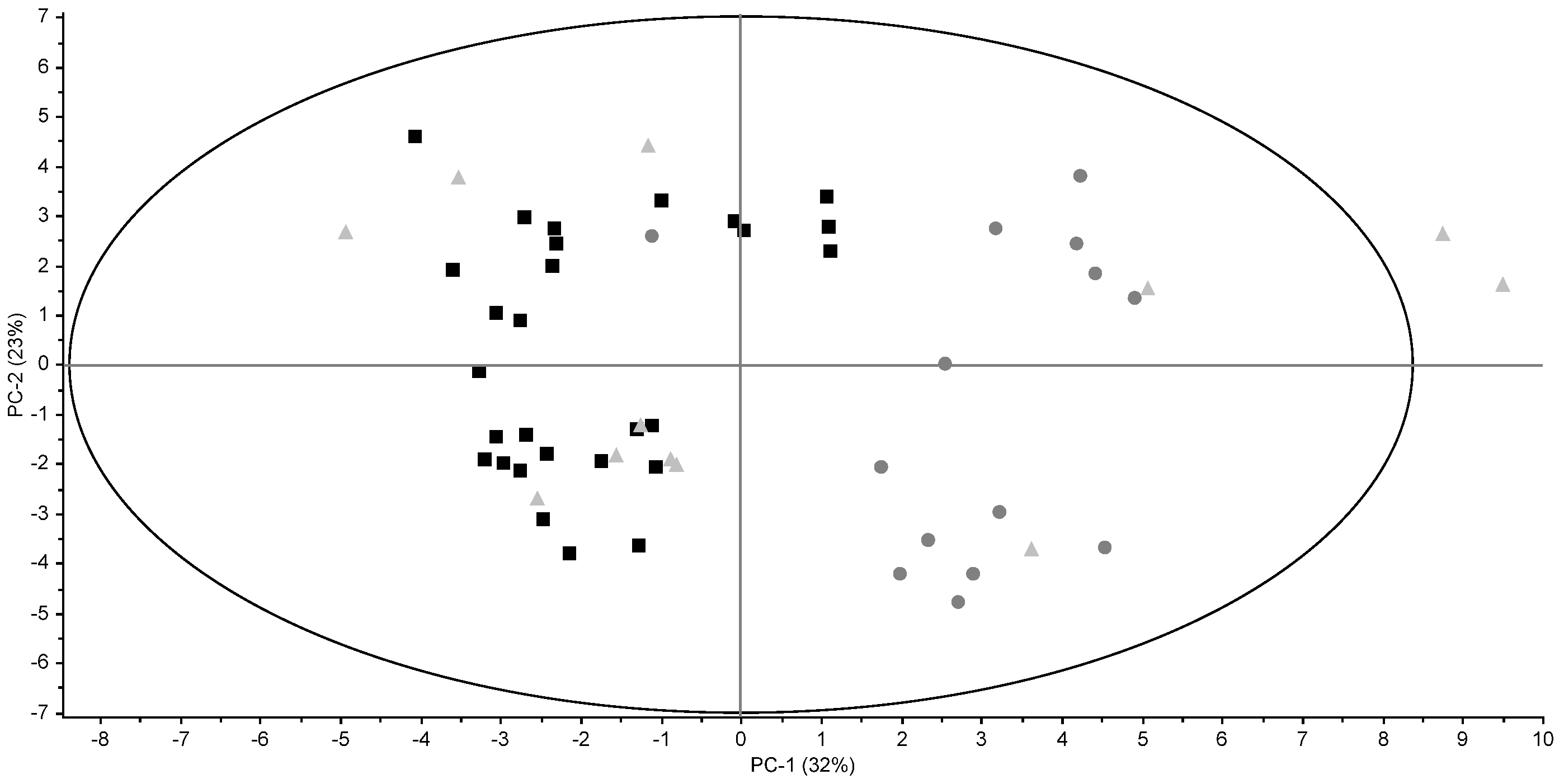
3.2. Triglyceride Profiles
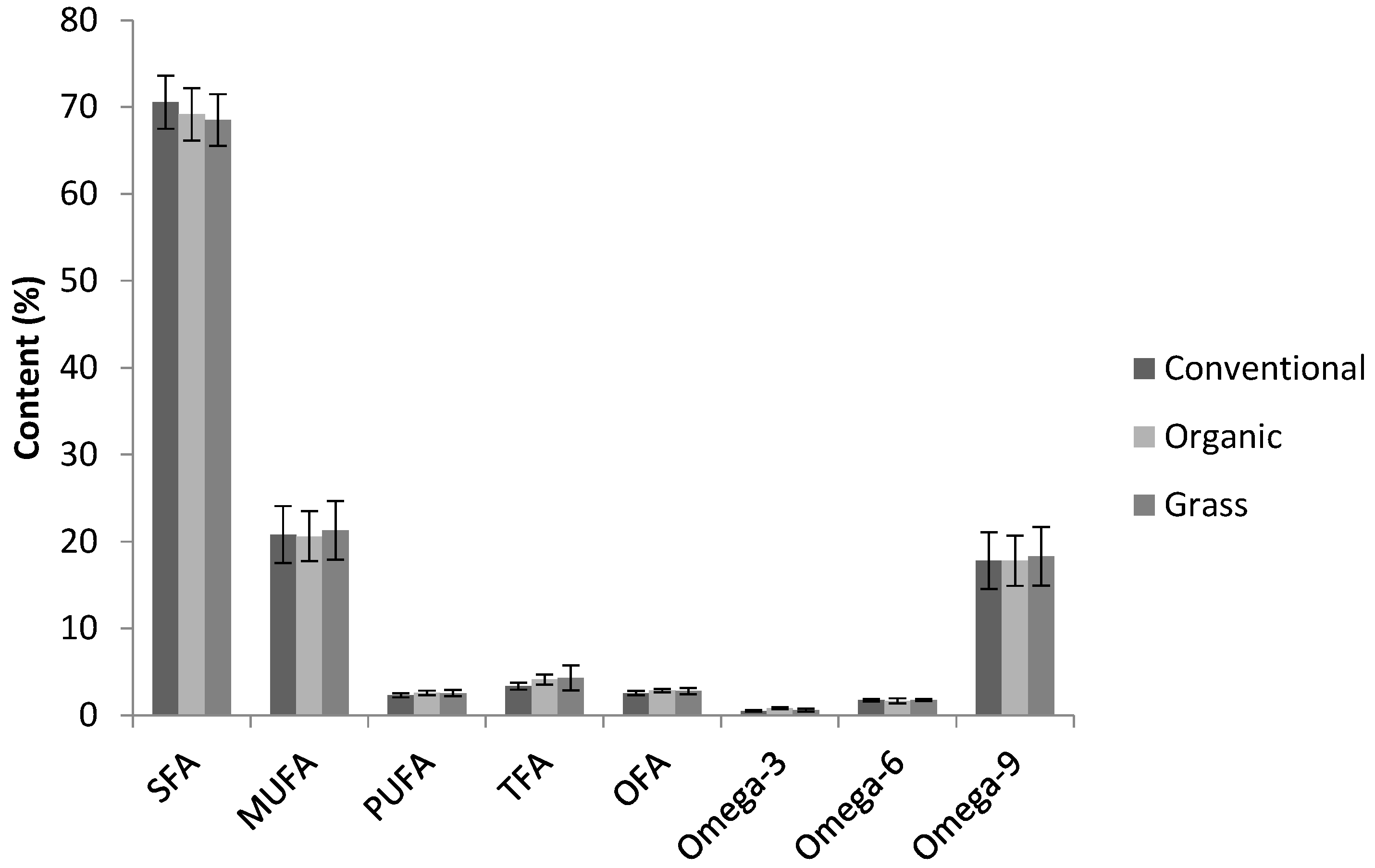
3.3. Fatty Acid Profiles
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Commission Regulation (EC) No 889/2008 of 5 September 2008 Laying Down Detailed Rules for the Implementation of Council Regulation (EC) No 834/2007 on Organic Production and Labelling of Organic Products with Regard to Organic Production, Labelling and Control. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2008.250.01.0001.01.ENG&toc=OJ:L:2008:250:TOC (accessed on 21 December 2016).
- Bergamo, P.; Fedele, E.; Iannibelli, L.; Marzillo, G. Fat-soluble vitamin contents and fatty acid composition in organic and conventional Italian dairy products. Food Chem. 2003, 82, 625–631. [Google Scholar] [CrossRef]
- Slots, T.; Sørensen, J.; Nielsen, J.H. Tocopherol, carotenoids and fatty acid composition in organic and conventional milk. Milchwissenschaft 2008, 63, 352–355. [Google Scholar]
- Molkentin, J. Authentication of organic milk using δ13C and the α-linolenic acid content of milk fat. J. Agric. Food Chem. 2009, 57, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Molkentin, J.; Giesemann, A. Follow-up of stable isotope analysis of organic versus conventional milk. Anal. Bioanal. Chem. 2010, 398, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
- Erich, S.; Schill, S.; Annweiler, E.; Waiblinger, H.U.; Kuballa, T.; Lachenmeier, D.W.; Monakhova, Y.B. Combined chemometric analysis of 1H NMR, 13C NMR and stable isotope data to differentiate organic and conventional milk. Food Chem. 2015, 188, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Capuano, E.; Boerrigter-Eenling, R.; Elgersma, A.; van Ruth, S.M. Effect of fresh grass feeding, pasture grazing and organic/biodynamic farming on bovine milk triglyceride profile and implications for authentication. Eur. Food Res. Technol. 2014, 238, 573–580. [Google Scholar] [CrossRef]
- Capuano, E.; Gravink, R.; Boerrigter-Eenling, R.; van Ruth, S.M. Fatty acid and triglycerides profiling of retail organic, conventional and pasture milk: Implications for health and authenticity. Int. Dairy J. 2015, 42, 58–63. [Google Scholar] [CrossRef]
- Capuano, E.; van der Veer, G.; Boerrigter-Eenling, R.; Elgersma, A.; Rademaker, J.; Sterian, A.; van Ruth, S.M. Verification of fresh grass feeding, pasture grazing and organic farming by cows farm milk fatty acid profile. Food Chem. 2014, 164, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Nozière, P.; Grauleta, B.; Lucasa, A.; Martina, B.; Grolierb, P.; Doreaua, M. Carotenoids for ruminants: From forages to dairy products. Anim. Feed Sci. Technol. 2006, 131, 418–450. [Google Scholar] [CrossRef]
- Plans Pujolras, M.; Ayvaz, H.; Shotts, M.-L.; Pittman, R.A., Jr.; Herringshaw, S.; Rodriguez-Saona, L.E. Portable Infrared Spectrometer to Characterize and Differentiate Between Organic and Conventional Bovine Butter. J. Am. Oil Chem. Soc. 2015, 92, 175–184. [Google Scholar] [CrossRef]
- Cullinane, N.; Aherne, S.; Connolly, J.F.; Phelan, J.A. Seasonal Variation in the Triglyceride and Fatty Acid Composition of Irish Butter. Irish J. Food Sci. Technol. 1984, 8, 1–12. [Google Scholar]
- Tomaszewska-Gras, J. Multivariate analysis of seasonal variation in the composition and thermal properties of butterfat with an emphasis on authenticity assessment. Grasas Y Aceites 2016, 67, 167. [Google Scholar] [CrossRef]
- Heck, J.M.L.; van Valenberg, H.J.; Dijkstra, J.; van Hooijdonk, A.C. Seasonal variation in the Dutch bovine raw milk composition. J. Dairy Sci. 2009, 92, 4745–4755. [Google Scholar] [CrossRef] [PubMed]
- Pabai, F.; Kermasha, S.; Morin, A. Lipase from Pseudomonas fragi CRDA 323: Partial purification, characterization and interesterification of butter fat. Appl. Microbiol. Biotechnol. 1995, 43, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Kaylegian, K.E.; Hartel, R.W.; Lindsay, R.C. Applications of Modified Milk Fat in Food Products. J. Dairy Sci. 1993, 76, 1782–1796. [Google Scholar] [CrossRef]
- International Organization for Standardization. Butter, Edible Oil Emulsions and Spreadable Fats—Determination of Fat Content; NEN-EN-ISO 17189:2003 | IDF 194; ISO: Geneva, Switzerland, 2003. [Google Scholar]
- International Organization for Standardization. Milk Fat Products and Butter—Determination of Fat Acidity (Reference Method); NEN-ISO 1740:2004 | IDF 6; ISO: Geneva, Switzerland, 2004. [Google Scholar]
- International Organization for Standardization. Milk and Milk Products—Determination of Milk Fat Purity by Gas Chromatographic Analysis of Triglycerides (Reference Method); ISO 17678 | IDF 202; ISO: Geneva, Switzerland, 2010. [Google Scholar]
- International Organization for Standardization. Milk, Milk Products, Infant Formula and Adult Nutritionals—Determination of Fatty Acids Composition—Capillary Gas Chromatographic Method; ISO 16958; ISO: Geneva, Switzerland, 2015. [Google Scholar]
- Commission Regulation (EC) No 1898/2005 of 9 November 2005 Laying Down Detailed Rules for Implementing Council Regulation (EC) No 1255/1999 as Regards Measures for the Disposal of Cream, Butter and Concentrated Butter on the Community Market. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1490871272954&uri=CELEX:32005R1898 (accessed on 21 December 2016).
- Grazing versus Indoor Feeding: Effects on Milk Quality. Available online: http://edepot.wur.nl/571 (accessed on 6 January 2017).
- Molkentin, J. Applicability of organic milk indicators to the authentication of processed products. Food Chem. 2013, 137, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Precht, D.; Molkentin, J. Effect of feeding on conjugated cis delta 9, trans delta 11-octadecadienoic acid and other isomers of linoleic acid in bovine milk fats. Mol. Nutr. Food Res. 1997, 41, 330–335. [Google Scholar]
- Delgadillo-Puga, C.; Sánchez-Muñoz, B.; Nahed-Toral, J.; Cuchillo-Hilario, M.; Díaz-Martínez, M.; Solis-Zabaleta, R.; Reyes-Hernández, A.; Castillo-Domíguez, R.M. Fatty acid content, health and risk indices, physicochemical composition, and somatic cell counts of milk from organic and conventional farming systems in tropical south-eastern Mexico. Trop. Anim. Health Prod. 2014, 46, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Elgersma, A. Grazing increases the unsaturated fatty acid concentration of milk from grass-fed cows: A review of the contributing factors, challenges and future perspectives. Eur. J. Lipid Sci Technol. 2015, 117, 1345–1369. [Google Scholar] [CrossRef]
| C (n = 28) | O (n = 14) | G (n = 12) | p-Value | |
|---|---|---|---|---|
| Fat content (m/m %) | 82.33 ± 0.56 a | 83.16 ± 0.68 b | 82.74 ± 0.48 ab | 0.001 |
| Free fatty acids (% oleic acid) | 0.148 ± 0.017 a | 0.164 ± 0.036 ab | 0.166 ± 0.020 b | 0.035 |
| TG | C (n = 28) | O (n = 13) | G (n = 12) | p-Value |
|---|---|---|---|---|
| CN24 | 0.06 ± 0.02 a | 0.09 ± 0.03 b | 0.06 ± 0.02 a | 0.007 |
| Cholesterol | 0.29 ± 0.05 a | 0.32 ± 0.02 a | 0.31 ± 0.04 a | 0.155 |
| CN26 | 0.33 ± 0.02 a | 0.34 ± 0.02 a | 0.33 ± 0.03 a | 0.144 |
| CN28 | 0.73 ± 0.04 a | 0.78 ± 0.04 b | 0.77 ± 0.07 ab | 0.006 |
| CN30 | 1.39 ± 0.08 a | 1.43 ± 0.10 a | 1.45 ± 0.12 a | 0.240 |
| CN32 | 2.86 ± 0.12 a | 2.86 ± 0.15 a | 2.93 ± 0.19 a | 0.543 |
| CN34 | 6.30 ± 0.19 a | 6.13 ± 0.25 a | 6.25 ± 0.28 a | 0.132 |
| CN36 | 11.11 ± 0.25 a | 10.91 ± 0.29 a | 10.88 ± 0.47 a | 0.225 |
| CN38 | 12.23 ± 0.17 a | 12.27 ± 0.14 a | 12.36 ± 0.27 a | 0.285 |
| CN40 | 9.58 ± 0.13 a | 9.91 ± 0.17 b | 9.92 ± 0.39 b | <0.001 |
| CN42 | 7.08 ± 0.19 a | 7.11 ± 0.17 a | 7.05 ± 0.17 a | 0.777 |
| CN44 | 6.91 ± 0.21 a | 6.68 ± 0.24 b | 6.69 ± 0.14 b | 0.002 |
| CN46 | 7.68 ± 0.28 a | 7.34 ± 0.23 b | 7.38 ± 0.27 b | 0.001 |
| CN48 | 9.19 ± 0.18 a | 8.79 ± 0.20 b | 8.84 ± 0.39 b | <0.001 |
| CN50 | 11.01 ± 0.26 a | 10.93 ± 0.31 a | 10.77 ± 0.34 a | 0.089 |
| CN52 | 9.18 ± 0.52 a | 9.44 ± 0.56 a | 9.40 ± 0.50 a | 0.361 |
| CN54 | 4.08 ± 0.45 a | 4.67 ± 0.56 b | 4.60 ± 0.84 ab | 0.007 |
| Fatty Acid | C (n = 28) | O (n = 14) | G (n = 12) | p-Value |
|---|---|---|---|---|
| C4:0 | 3.99 ± 0.61 a | 4.02 ± 0.53 a | 3.99 ± 0.49 a | 0.970 |
| C6:0 | 2.33 ± 0.13 a | 2.39 ± 0.11 a | 2.35 ± 0.11 a | 0.255 |
| C8:0 | 1.36 ± 0.05 a | 1.40 ± 0.04 b | 1.40 ± 0.06 b | 0.031 |
| C10:0 | 3.13 ± 0.18 a | 3.19 ± 0.20 a | 3.25 ± 0.27 a | 0.407 |
| C12:0 | 4.05 ± 0.24 a | 3.78 ± 0.23 b | 4.03 ± 0.30 a | 0.008 |
| C14:0 | 12.00 ± 0.54 a | 11.82 ± 0.36 a | 11.75 ± 0.45 a | 0.377 |
| C14:1n5 | 1.08 ± 0.07 a | 1.03 ± 0.07 a | 1.07 ± 0.13 a | 0.274 |
| C15:0 | 1.16 ± 0.06 a | 1.25 ± 0.05 b | 1.19 ± 0.10 ab | 0.001 |
| C15:1n5 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 1.000 |
| C16:0 | 31.77 ± 1.29 a | 30.19 ± 1.94 b | 29.66 ± 2.55 b | 0.002 |
| C16:1n7 | 1.72 ± 0.08 a | 1.56 ± 0.08 b | 1.68 ± 0.23 ab | 0.001 |
| C17:0 | 0.50 ± 0.05 a | 0.55 ± 0.04 b | 0.51 ± 0.06 ab | 0.009 |
| C17:1n8 | 0.21 ± 0.03 a | 0.23 ± 0.03 ab | 0.25 ± 0.03 b | 0.006 |
| C18:0 | 10.11 ± 0.53 a | 10.33 ± 0.65 a | 10.24 ± 0.73 a | 0.550 |
| trans C18:1 | 2.89 ± 0.35 a | 3.58 ± 0.54 b | 3.72 ± 1.22 b | <0.001 |
| C18:1 | 17.75 ± 3.27 a | 17.72 ± 2.88 a | 18.24 ± 3.36 a | 0.550 |
| trans C18:2 | 0.45 ± 0.06 a | 0.52 ± 0.09 b | 0.57 ± 0.22 b | 0.008 |
| C18:2n6 | 1.49 ± 0.14 a | 1.45 ± 0.27 a | 1.51 ± 0.15 a | 0.508 |
| C18:2 CLA | 0.54 ± 0.13 a | 0.86 ± 0.19 b | 0.78 ± 0.36 b | <0.001 |
| C18:3n6 | 0.03 ± 0.05 a | 0.01 ± 0.03 a | 0.05 ± 0.06 a | 0.069 |
| trans C18:3 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.021 |
| C18:3n3 | 0.43 ± 0.09 a | 0.73 ± 0.11 b | 0.54 ± 0.15 c | <0.001 |
| C20:0 | 0.10 ± 0.06 a | 0.13 ± 0.03 b | 0.07 ± 0.06 a | 0.001 |
| C20:1n9 | 0.04 ± 0.01 a | 0.04 ± 0.01 a | 0.04 ± 0.01 a | 0.536 |
| C20:2n6 | 0.01 ± 0.00 a | 0.02 ± 0.00 a | 0.01 ± 0.01 a | 0.232 |
| C20:3n6 | 0.06 ± 0.01 a | 0.05 ± 0.01 b | 0.05 ± 0.01 ab | <0.001 |
| C20:3n3 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.003 |
| C20:4n6 | 0.11 ± 0.01 a | 0.09 ± 0.01 b | 0.10 ± 0.01 a | 0.028 |
| C20:5n3 | 0.04 ± 0.01 a | 0.06 ± 0.01 b | 0.05 ± 0.02 a | <0.001 |
| C22:0 | 0.05 ± 0.01 a | 0.06 ± 0.01 b | 0.05 ± 0.01 a | 0.003 |
| C22:1n9 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.511 |
| C22:2n6 | 0.03 ± 0.01 a | 0.04 ± 0.00 b | 0.04 ± 0.02 ab | <0.001 |
| C22:6 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 1.000 |
| C24:0 | 0.03 ± 0.01 a | 0.04 ± 0.00 b | 0.02 ± 0.01 a | 0.001 |
| C24:1n9 | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.00 ± 0.00 a | 0.928 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pustjens, A.M.; Boerrigter-Eenling, R.; Koot, A.H.; Rozijn, M.; Van Ruth, S.M. Characterization of Retail Conventional, Organic, and Grass Full-Fat Butters by Their Fat Contents, Free Fatty Acid Contents, and Triglyceride and Fatty Acid Profiling. Foods 2017, 6, 26. https://doi.org/10.3390/foods6040026
Pustjens AM, Boerrigter-Eenling R, Koot AH, Rozijn M, Van Ruth SM. Characterization of Retail Conventional, Organic, and Grass Full-Fat Butters by Their Fat Contents, Free Fatty Acid Contents, and Triglyceride and Fatty Acid Profiling. Foods. 2017; 6(4):26. https://doi.org/10.3390/foods6040026
Chicago/Turabian StylePustjens, Annemieke M., Rita Boerrigter-Eenling, Alex H. Koot, Maikel Rozijn, and Saskia M. Van Ruth. 2017. "Characterization of Retail Conventional, Organic, and Grass Full-Fat Butters by Their Fat Contents, Free Fatty Acid Contents, and Triglyceride and Fatty Acid Profiling" Foods 6, no. 4: 26. https://doi.org/10.3390/foods6040026
APA StylePustjens, A. M., Boerrigter-Eenling, R., Koot, A. H., Rozijn, M., & Van Ruth, S. M. (2017). Characterization of Retail Conventional, Organic, and Grass Full-Fat Butters by Their Fat Contents, Free Fatty Acid Contents, and Triglyceride and Fatty Acid Profiling. Foods, 6(4), 26. https://doi.org/10.3390/foods6040026





