Behavior of Salmonella and Listeria monocytogenes in Raw Yellowfin Tuna during Cold Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Target Pathogen Strains and Culture Preparation
2.2. Tuna Samples and Inoculation of Target Pathogens
2.3. Determination of Aerobic Plate Counts (APC) and Psychrotrophic Bacterial Counts (PBC)
2.4. Enumeration of Target Pathogens in Inoculated Tuna
2.5. Data Analysis
3. Results and Discussion
3.1. Changes of Aerobic Plate Counts (APC) and Psychrotrophic Bacterial Counts (PBC) in Tuna during Refrigerated and Frozen Storage
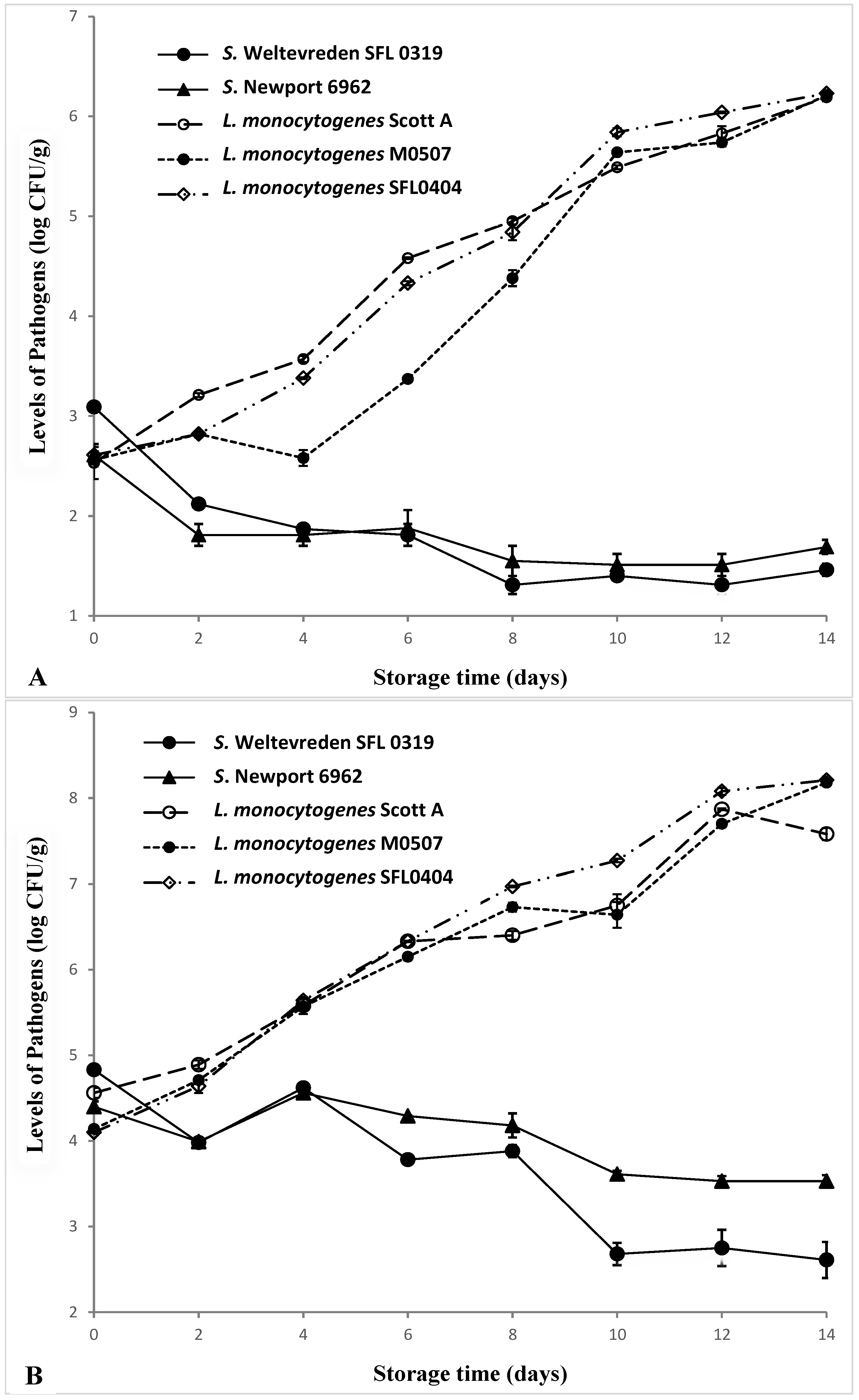
3.2. Behavior of Salmonella and Listeria monocytogenes in Tuna during Refrigeration Storage
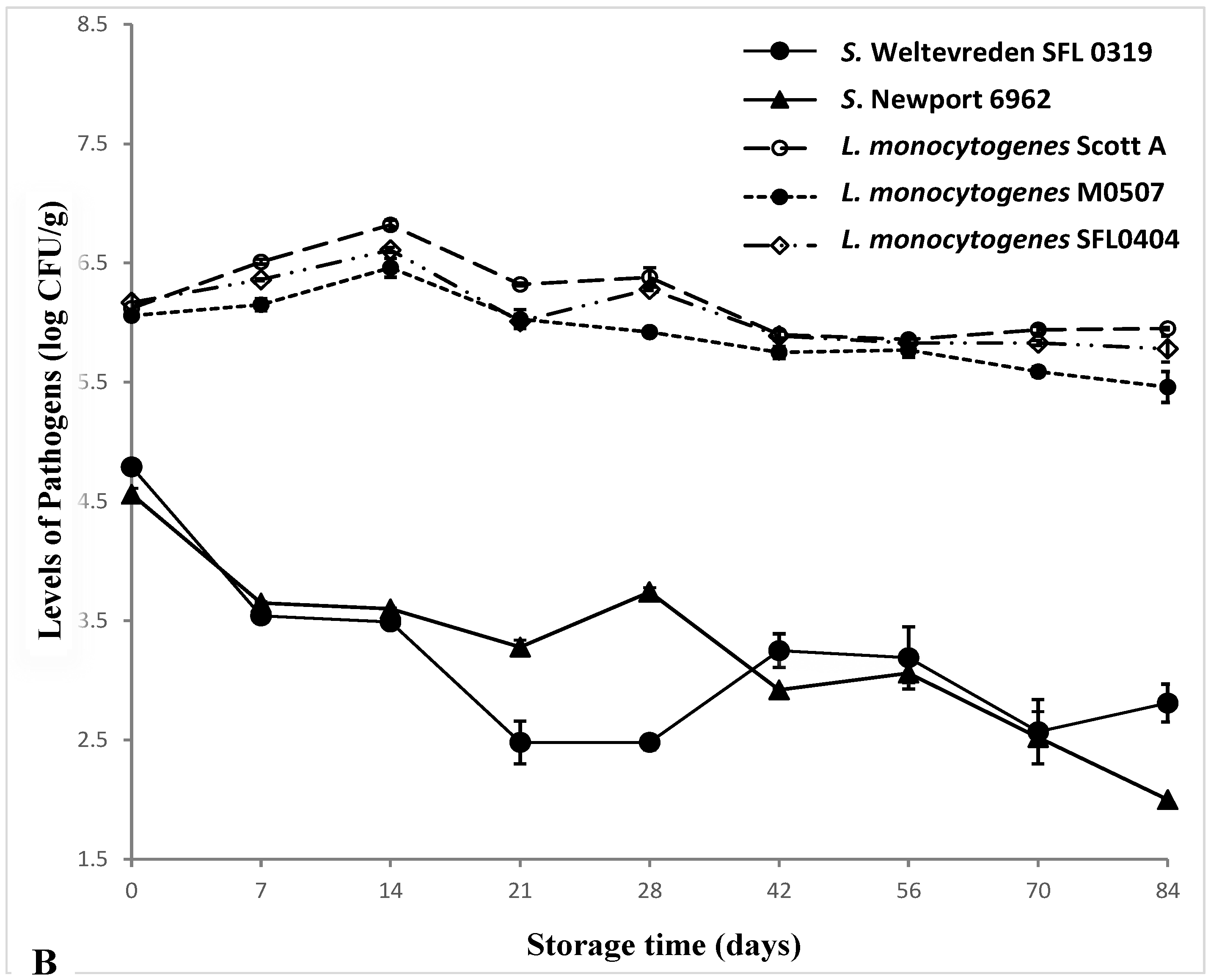
3.3. Survival of Salmonella and L. monocytogenes in Tuna during Frozen Storage
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Centers for Diseases Control and Prevention (CDC). Foodborne Outbreak Online Database (FOOD Tool). Available online: http://wwwn.cdc.gov/foodborneoutbreaks/Default.aspx (accessed on 3 January 2016).
- Center for Science in the Public Interest (CSPI). Outbreak Alert! 2014. Available online: http://cspinet.org/reports/outbreakalert2014.pdf (accessed on 3 January 2016).
- Wan Norhana, M.N.; Poole, S.E.; Deeth, H.C.; Dykes, G.A. Prevalence, persistence and control of Salmonella and Listeria in shrimp and shrimp products: A review. Food Control 2010, 21, 343–361. [Google Scholar] [CrossRef]
- Amagliani, G.; Brandi, G.; Schiavano, G.F. Incidence and role of Salmonella in seafood safety. Food Res. Int. 2012, 45, 780–788. [Google Scholar] [CrossRef]
- Ben Embarek, P.K. Presence, detection and growth of Listeria monocytogenes in seafoods: A review. Int. J. Food Microbiol. 1994, 23, 17–34. [Google Scholar] [CrossRef]
- Salihu, M.D.; Junaidu, A.U.; Manga, S.B.; Gulumbe, M.L.; Magaji, A.A.; Ahmed, A.; Adamu, A.Y.; Shittu, A.; Balarabe, I. Occurrence of Listeria monocytogenes in smoked fish in Sokoto, Nigeria. Afr. J. Biotechnol. 2008, 7, 3082–3084. [Google Scholar]
- Miya, S.; Takahashi, H.; Ishikawa, T.; Fujii, T.; Kimura, B. Risk of Listeria monocytogenes contamination of raw ready-to-eat seafood products available at retail outlets in Japan. Appl. Environ. Microbiol. 2010, 76, 3383–3386. [Google Scholar] [CrossRef] [PubMed]
- Jami, M.; Ghanbari, M.; Zunabovic, M.; Domig, K.J.; Kneifel, W. Listeria monocytogenes in Aquatic Food Products—A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 798–813. [Google Scholar] [CrossRef]
- Heinitz, M.L.; Ruble, R.D.; Wagner, D.E.; Tatini, S.R. Incidence of Salmonella in Fish and Seafood. J. Food Prot. 2000, 63, 579–592. [Google Scholar] [PubMed]
- Handa, S.; Kimura, B.; Takahashi, H.; Koda, T.; Hisa, K.; Fujii, T. Incidence of Listeria monocytogenes in raw seafood products in Japanese retail stores. J. Food Prot. 2005, 68, 411–415. [Google Scholar] [PubMed]
- Centers for Diseases Control and Prevention. Multistate Outbreak of Salmonella Bareilly and Salmonella Nchanga Infections Associated with a Raw Scraped Ground Tuna Product (Final Update). Available online: http://www.cdc.gov/salmonella/bareilly-04-12/index.html (accessed on 3 January 2016).
- Centers for Diseases Control and Prevention. Multistate Outbreak of Salmonella Paratyphi B variant L(+) tartrate(+) and Salmonella Weltevreden Infections Linked to Frozen Raw Tuna (Final Update). Available online: http://www.cdc.gov/salmonella/paratyphi-b-05-15/index.html (accessed on 3 January 2016).
- Friesema, I.H.; Jong, A.E.; James, I.A.; Heck, M.E.; Kerkhof, J.H.; Notermans, D.W.; Pelt, W.; Hofhuis, A. Outbreak of Salmonella Thompson in the Netherlands since July 2012. Available online: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=20303 (accessed on 3 January 2016).
- Ericsson, H.; Eklöw, A.; Danielsson-Tham, M.-L.; Loncarevic, S.; Mentzing, L.-O.; Persson, I.; Unnerstad, H.; Tham, W. An outbreak of listeriosis suspected to have been caused by rainbow trout. J. Clin. Microbiol. 1997, 35, 2904–2907. [Google Scholar] [PubMed]
- Miettinen, M.K.; Siitonen, A.; Heiskanen, P.; Haajanen, H.; Björkroth, K.J.; Korkeala, H.J. Molecular epidemiology of an outbreak of febrile gastroenteritis caused by Listeria monocytogenes in cold-smoked rainbow trout. J. Clin. Microbiol. 1999, 37, 2358–2360. [Google Scholar] [PubMed]
- Cousin, M.A.; Jay, J.M.; Vasavada, P.C. Psychrotrophic microorganisms. In Compendium of Methods for the Microbiological Examination of Foods; Downes, I., Ed.; American Public Health Association: Washington, DC, USA, 2001; pp. 159–160. [Google Scholar]
- Rahn, K.; Grandis, S.A.; Clarke, R.C.; McEwen, S.A.; Galan, J.E.; Ginocchio, C.; Curtiss, R.; Gyles, C.L. Amplification of an invA gene sequence of Salmonella typhimurium by polymerase chain reaction as a specific method of detection Salmonella. Mol. Cell. Probes 1992, 6, 271–279. [Google Scholar] [CrossRef]
- Chen, Y.; Knabel, S.J. Multiplex PCR for simultaneous detection of bacteria of the genus Listeria, Listeria monocytogenes, and major serotypes and epidemic clones of L. monocytogenes. Appl. Environ. Microbiol. 2007, 73, 6299–6304. [Google Scholar]
- Ercolini, D.; Russo, F.; Nasi, A.; Ferranti, P.; Villani, F. Mesophilic and psychrotrophic bacteria from meat and their spoilage potential in vireo and in beef. Appl. Environ. Microbiol. 2009, 75, 1990–2001. [Google Scholar] [CrossRef] [PubMed]
- Amarita, F. Methods for Microbiological Quality Assessment of Seafood. Available online: http://valid.aztionline.com/index.php?idioma=en&seccion=6&ctipo=2&contenido=320&menu=136&submenu=142 (accessed on 16 January 2016).
- International Commission on Microbiological Specifications for Foods (ICMSF). Microorganisms in Foods 2: Sampling for Microbiological Analysis: Principles and Specific Applications, 2nd ed.; University of Toronto Press: Toronto, ON, Canada, 1986. [Google Scholar]
- Fish and Fishery Products Hazards and Controls Guidance, Fourth Edition—April 2011. Available online: http://www.fda.gov/downloads/Food/GuidanceComplianceRegulatoryInformation/GuidanceDocuments/Seafood/UCM251970.pdf (accessed on 16 January 2016).
- Gill, C.O.; Reichel, M.P. Growth of the cold-tolerant pathogens Yersinia enterocolitica, Aeromonas hydrophila, and Listeria monocytogenes on high-pH beef packaged under vacuum or carbon dioxide. Food Microbiol. 1989, 6, 223–240. [Google Scholar] [CrossRef]
- Francis, G.A.; O’Beirne, D. Effects of vegetable type, package atmosphere and storage temperature on growth and survival of Escherichia coli O157:H7 and Listeria monocytogenes. J. Ind. Microbiol. Biotechnol. 2001, 27, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.A.; Tamplin, M.L. The influence of mayonnaise pH and storage temperature on the growth of Listeria monocytogenes in seafood salad. Int. J. Food Microbiol. 2005, 102, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.A.; Mott, S.J. Growoth of Listeria monocytogenes, Aeromonas hydrophila and Yersinia enterocolitica on cold-smoked salmon under refrigeration and mild temperature abuse. Food Microbiol. 1993, 10, 61–68. [Google Scholar] [CrossRef]
- Digirolamo, R.; Liston, J.; Matches, J. The effects of freezing on the survival of Salmonella and E. coli in pacific oysters. J. Food Sci. 1970, 35, 13–16. [Google Scholar] [CrossRef]
- Iyer, T.S. Incidence and low temperature survival of Salmonella in fishery products. Fish. Technol. 1989, 26, 39–42. [Google Scholar]
- Harrison, M.A.; Huang, Y.W.; Chao, C.H.; Shineman, T. Fate of Listeria monocytogenes on packaged, refrigerated, and frozen seafood. J. Food Prot. 1991, 54, 524–527. [Google Scholar]
- Miladi, H.; Chaieb, K.; Bakhrouf, A.; Elmnasser, N.; Ammar, E. Freezing effects on survival of Listeria monocytogenes in artificially contaminated cold fresh-salmon. Ann. Microbiol. 2008, 58, 471–476. [Google Scholar] [CrossRef]

| Pathogen | Gene | Primer | Sequence (5′–3′) | Size (bp) |
|---|---|---|---|---|
| Salmonella | invA | invAF | GTGAAATTATCGCCACGTTCGGGCAA | 284 |
| invAR | TCATCGCACCGTCAAAGGAACC | |||
| L. monocytogenes | lmo2234 | lmoF | TGTCCAGTTCCATTTTTAACT | 420 |
| lmoR | TTGTTGTTCTGCTGTACGA |
| Bacterial Counts | Day 0 | Day 2 | Day 4 | Day 6 | Day 8 | Day 10 | Day 12 |
|---|---|---|---|---|---|---|---|
| APC | 3.69 ± 0.06 a | 3.80 ± 0.09 a | 4.95 ± 0.05 b | 7.36 ± 0.02 c | 8.03 ± 0.08 d | 8.53 ± 0.17 e | 8.75 ± 0.08 e |
| PBC | 3.98 ± 0.02 a | 3.98 ± 0.06 a | 5.57 ± 0.14 b | 7.61 ± 0.01 c | 8.73 ± 0.05 d | 9.11 ± 0.06 e | 9.18 ± 0.01 e |
| Bacterial Counts | Day 0 | Day 14 | Day 28 | Day 42 | Day 56 | Day 70 | Day 84 |
|---|---|---|---|---|---|---|---|
| APC | 4.89 ± 0.06 a | 5.00 ± 0.03 a | 4.91 ± 0.05 a | 4.96 ± 0.05 a | 5.07 ± 0.03 a | 5.08 ± 0.04 a | 5.09 ± 0.01 a |
| PBC | 5.30 ± 0.02 a | 4.93 ± 0.18 a | 5.01 ± 0.03 a | 5.13 ± 0.02 a | 5.13 ± 0.01 a | 5.17 ± 0.02 a | 5.12 ± 0.01 a |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.; Mou, J.; Su, Y.-C. Behavior of Salmonella and Listeria monocytogenes in Raw Yellowfin Tuna during Cold Storage. Foods 2016, 5, 16. https://doi.org/10.3390/foods5010016
Liu C, Mou J, Su Y-C. Behavior of Salmonella and Listeria monocytogenes in Raw Yellowfin Tuna during Cold Storage. Foods. 2016; 5(1):16. https://doi.org/10.3390/foods5010016
Chicago/Turabian StyleLiu, Chengchu, Jing Mou, and Yi-Cheng Su. 2016. "Behavior of Salmonella and Listeria monocytogenes in Raw Yellowfin Tuna during Cold Storage" Foods 5, no. 1: 16. https://doi.org/10.3390/foods5010016
APA StyleLiu, C., Mou, J., & Su, Y.-C. (2016). Behavior of Salmonella and Listeria monocytogenes in Raw Yellowfin Tuna during Cold Storage. Foods, 5(1), 16. https://doi.org/10.3390/foods5010016





