Processing-Induced Variations in Bamboo Leaf Powder: Effects of Fixation Methods on Color Stability, Volatile Compounds, and Sensory Profiles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fixation of Bamboo Leaves and Powder Preparation
2.3. Determination of Color Value
2.4. Chlorophyll Content Determination
2.5. Determination of POD and PPO Activities
2.6. HS-SPME-GC-MS Analysis of VOCs
2.7. Calculation of ROAV
2.8. HS-SPME-GC-O Analysis of VOCs
2.9. Sensory Evaluation
2.10. Statistical Analysis
3. Results
3.1. Effect of Fixation on POD and PPO Activities in Bamboo Leaf Powder
3.2. Effects of Fixation on Chlorophyll Content and Color of Bamboo Leaf Powder
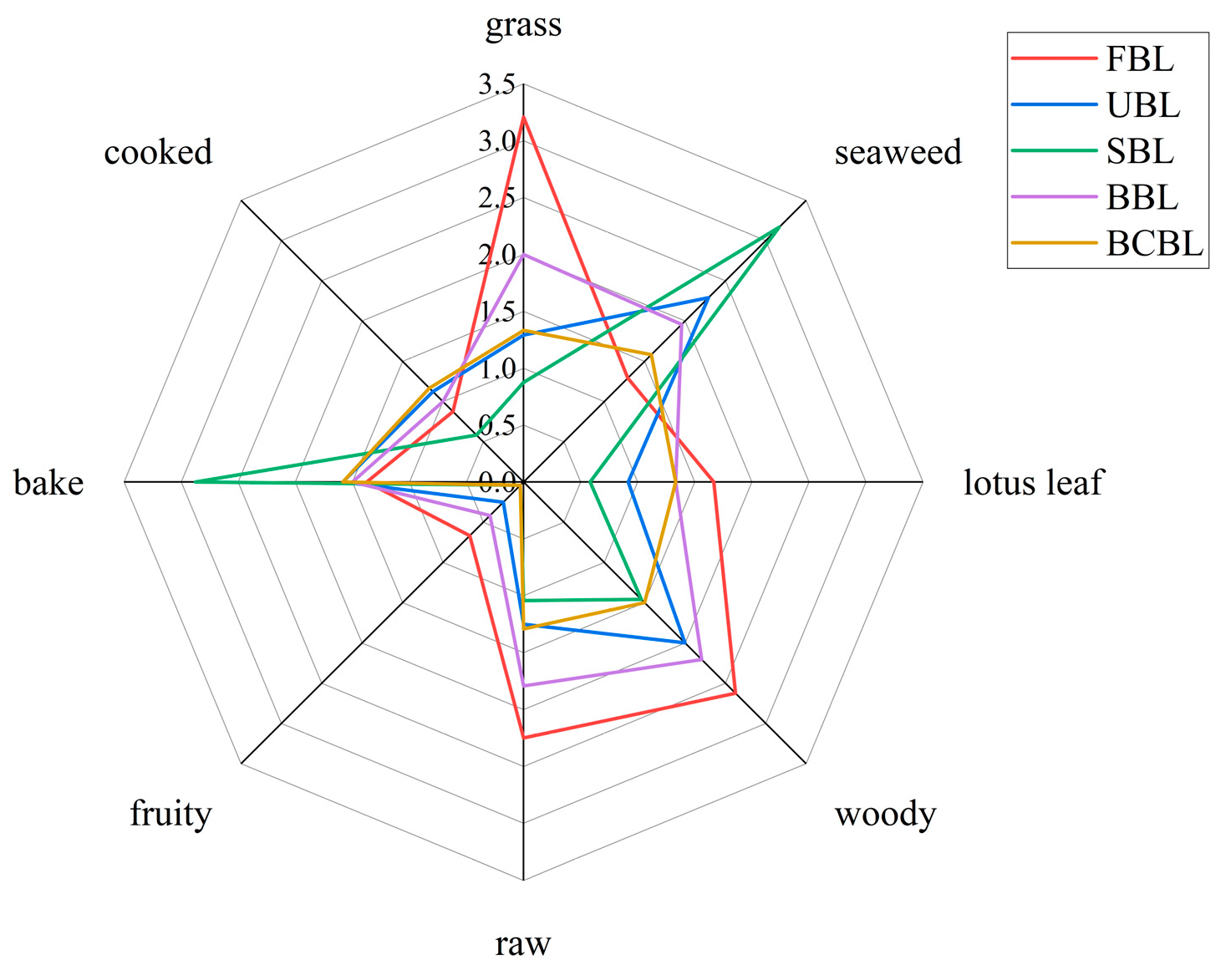
3.3. Effect of Fixation on the Aromatic Profiles of Bamboo Leaf Powder
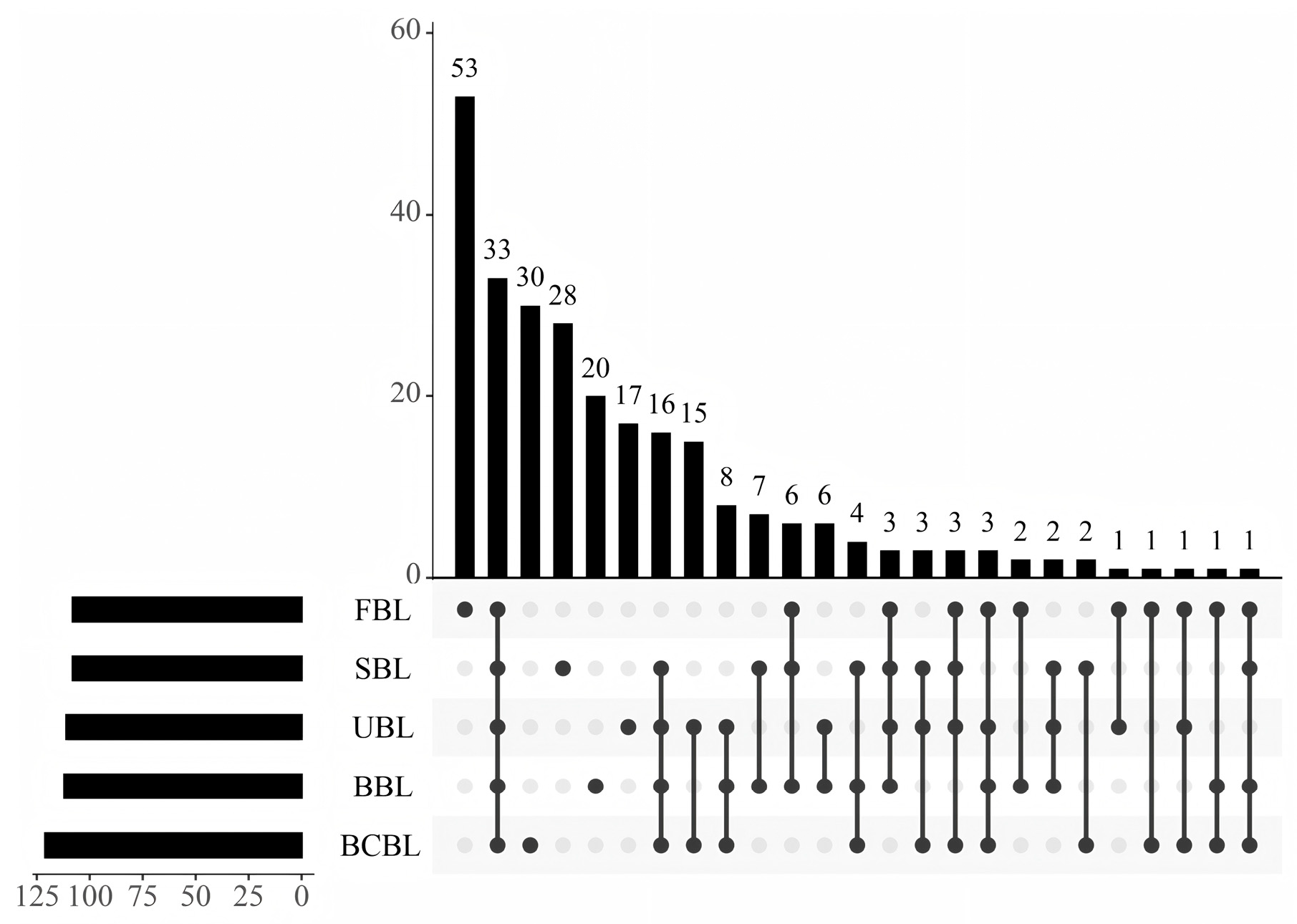
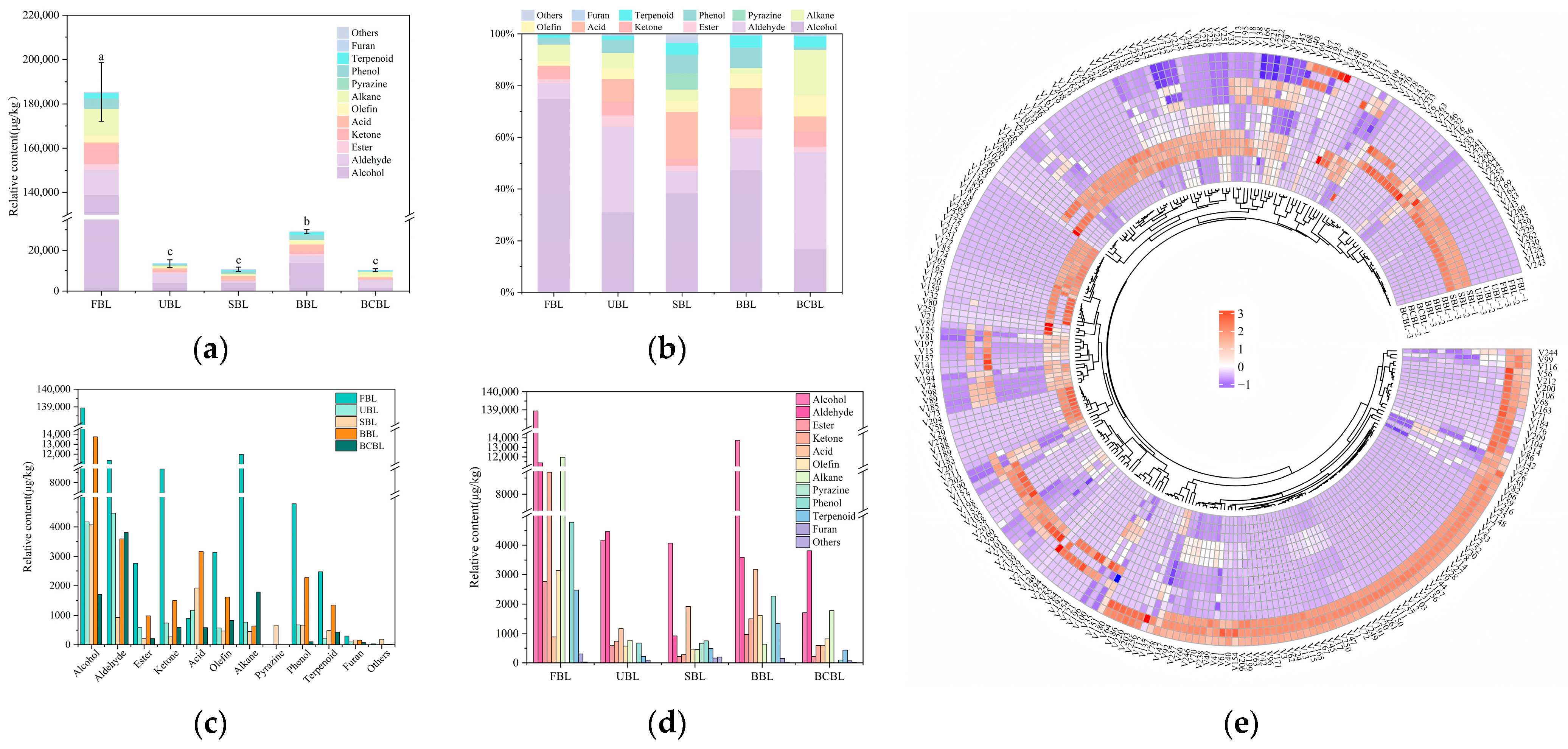
3.4. Effect of Fixation on the Volatile Compound Composition of Bamboo Leaf Powder
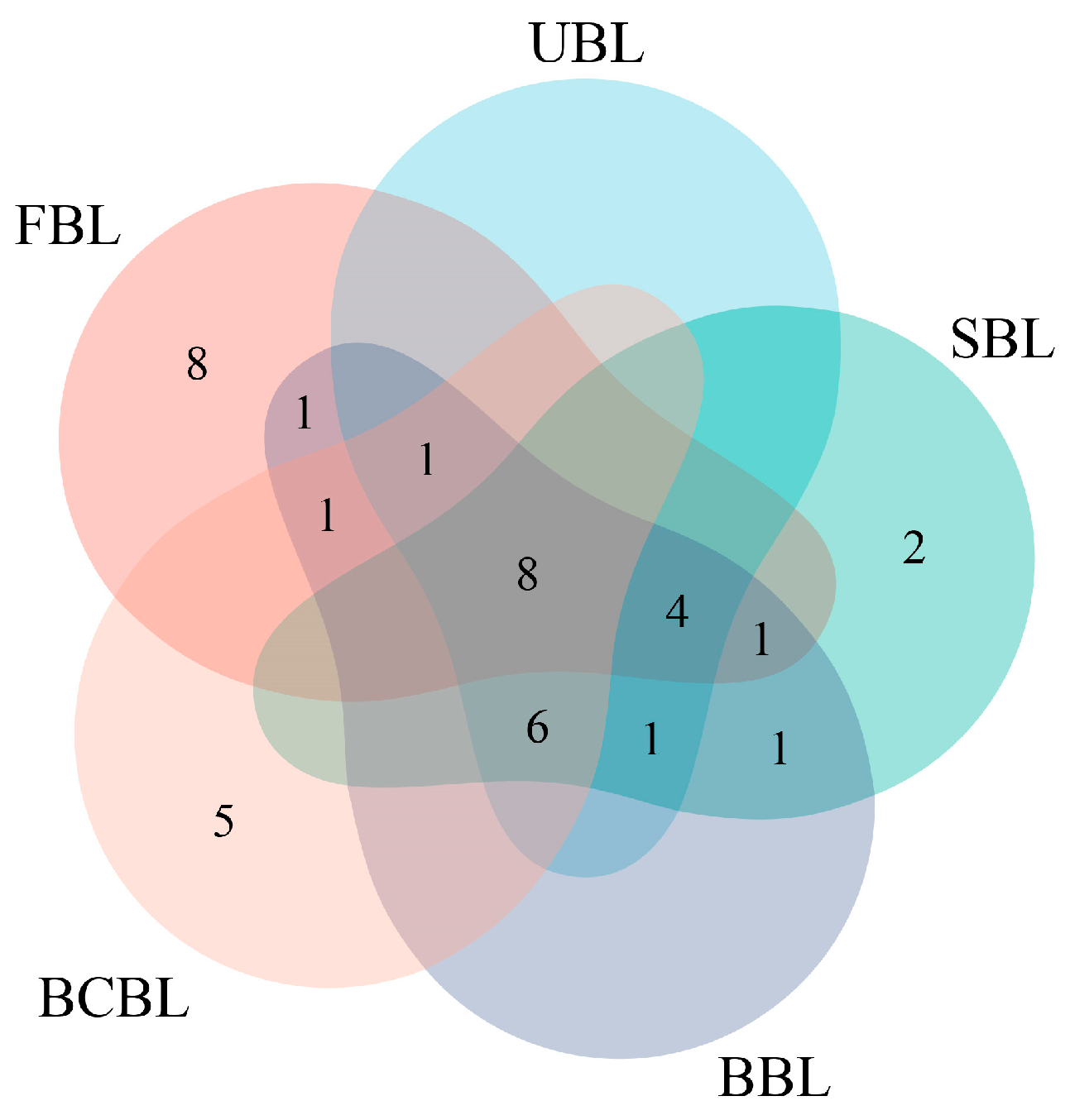
3.5. Screening of Aroma-Active Compounds in Bamboo Leaf Samples Using ROAVs
3.6. Effects of Fixation on the Key Aroma Compounds in Bamboo Leaf Powder
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| NO. | Time (min) | Compounds | Class | CAS | Content (μg/kg) | ||||
|---|---|---|---|---|---|---|---|---|---|
| FBL | UBL | SBL | BBL | BCBL | |||||
| 1 | 5.98 | 1-Propanol, 2-methyl- | Alcohol | 78-83-1 | 2136.37 ± 659.63 | ND | 49.14 ± 4.50 | 120.05 ± 1.93 | ND |
| 2 | 6.306 | 3-Pentanol | Alcohol | 584-2-1 | 5606.38 ± 531.21 | ND | ND | ND | ND |
| 3 | 6.475 | (R)-[51]-2-Pentanol | Alcohol | 31087-44-2 | 4284.21 ± 412.97 | ND | ND | ND | ND |
| 4 | 6.508 | 2-Pentanol | Alcohol | 6032-29-7 | ND | ND | ND | 58.43 ± 11.76 | ND |
| 5 | 6.517 | (S)-(+)-2-Pentanol | Alcohol | 26184-62-3 | ND | ND | 40.40 ± 28.84 | ND | ND |
| 6 | 6.851 | 1-Butanol | Alcohol | 71-36-3 | 171.17 ± 87.69 | ND | ND | ND | ND |
| 7 | 7.144 | 1-Penten-3-ol | Alcohol | 616-25-1 | 996.73 ± 193.38 | 148.69 ± 6.02 | 92.07 ± 12.05 | 506.95 ± 47.03 | 41.09 ± 3.50 |
| 8 | 7.901 | 1-Butanol, 3-methyl- | Alcohol | 123-51-3 | 12,768.25 ± 1393.07 | 29.37 ± 2.67 | 74.41 ± 8.01 | 382.70 ± 15.73 | 8.19 ± 2.66 |
| 9 | 8.57 | 1-Pentanol | Alcohol | 71-41-0 | 6114.44 ± 563.15 | ND | ND | 142.52 ± 3.69 | ND |
| 10 | 9.237 | 1-Heptanol, 2,4-dimethyl-, | Alcohol | 98982-97-9 | 345.70 ± 38.47 | ND | ND | ND | ND |
| 11 | 9.499 | Cyclobutanemethanol | Alcohol | 4415-82-1 | ND | ND | 12.73 ± 9.01 | 78.78 ± 6.25 | ND |
| 12 | 9.502 | Cyclopentanol | Alcohol | 96-41-3 | 231.86 ± 41.57 | ND | ND | ND | ND |
| 13 | 9.602 | 2-Penten-1-ol, (Z)- | Alcohol | 1576-95-0 | 10,583.87 ± 325.07 | 277.72 ± 12.79 | 282.30 ± 33.21 | 1326.91 ± 46.81 | ND |
| 14 | 10.134 | 1-Hexanol | Alcohol | 111-27-3 | 24,602.32 ± 1893.33 | 144.04 ± 7.32 | 150.09 ± 46.20 | 729.89 ± 16.14 | ND |
| 15 | 10.173 | n-Pentadecanol | Alcohol | 629-76-5 | ND | ND | ND | ND | 42.15 ± 3.64 |
| 16 | 10.254 | 3-Hexen-1-ol, (Z)- | Alcohol | 928-96-1 | 2091.24 ± 177.69 | ND | 316.82 ± 179.99 | 157.21 ± 4.69 | ND |
| 17 | 10.396 | 1-Octanol, 2-butyl- | Alcohol | 3913-2-8 | ND | 159.87 ± 128.15 | ND | ND | 561.55 ± 373.95 |
| 18 | 10.54 | 4-Penten-1-ol, 3-methyl- | Alcohol | 51174-44-8 | ND | 283.14 ± 3.08 | ND | ND | ND |
| 19 | 10.548 | 3-Hexen-1-ol, (E)- | Alcohol | 928-97-2 | 11,704.47 ± 934.65 | ND | ND | 1198.39 ± 45.55 | ND |
| 20 | 10.712 | 3-Octanol | Alcohol | 589-98-0 | 3161.28 ± 301.18 | ND | 65.39 ± 8.62 | 281.87 ± 17.67 | ND |
| 21 | 10.846 | 2-Hexen-1-ol, (Z)- | Alcohol | 928-94-9 | 776.90 ± 75.26 | ND | ND | ND | ND |
| 22 | 10.851 | 2-Hexen-1-ol, (E)- | Alcohol | 928-95-0 | ND | ND | ND | 112.12 ± 4.87 | ND |
| 23 | 10.91 | 4-Hexen-1-ol, (E)- | Alcohol | 928-92-7 | 706.36 ± 56.41 | ND | ND | ND | ND |
| 24 | 10.961 | 6-Hepten-3-ol, 4-methyl- | Alcohol | 53907-71-4 | 1354.93 ± 101.92 | ND | ND | ND | ND |
| 25 | 11.062 | 2-Octanol | Alcohol | 123-96-6 | 1043.08 ± 92.22 | ND | ND | ND | ND |
| 26 | 11.476 | 1-Octen-3-ol | Alcohol | 3391-86-4 | ND | ND | ND | 62.37 ± 88.21 | ND |
| 27 | 11.476 | 1-Dodecen-3-ol | Alcohol | 4048-42-4 | ND | ND | ND | 127.72 ± 90.74 | ND |
| 28 | 11.525 | 1-Heptanol | Alcohol | 111-70-6 | 2439.38 ± 185.33 | ND | ND | ND | ND |
| 29 | 11.778 | trans-Linalool oxide (furanoid) | Alcohol | 34995-77-2 | 4983.17 ± 401.25 | ND | ND | ND | ND |
| 30 | 12.029 | cis-Hept-4-enol | Alcohol | 6191-71-5 | 5078.22 ± 1101.43 | ND | ND | ND | ND |
| 31 | 12.508 | 2,3-Butanediol, [R- (R*, R*)]- | Alcohol | 24347-58-8 | 2441.91 ± 214.11 | 285.88 ± 26.81 | 1055.03 ± 233.37 | 1804.42 ± 64.44 | 111.79 ± 8.45 |
| 32 | 12.649 | Bicyclo [4.1.0] heptan-2-ol, 3,7,7-trimethyl-, (1α,2α,3β,6α)- | Alcohol | 90026-62-3 | 1007.54 ± 104.34 | ND | ND | ND | ND |
| 33 | 12.735 | Linalool | Alcohol | 78-70-6 | 4392.55 ± 369.60 | 533.54 ± 40.12 | 663.62 ± 69.98 | 2361.89 ± 80.64 | 468.23 ± 22.90 |
| 34 | 12.858 | 1-Octanol | Alcohol | 111-87-5 | 3541.67 ± 303.73 | 100.28 ± 9.15 | 56.68 ± 6.38 | ND | 95.08 ± 6.49 |
| 35 | 12.958 | 2,3-Butanediol | Alcohol | 513-85-9 | ND | 401.26 ± 140.61 | ND | ND | 112.06 ± 12.51 |
| 36 | 13.119 | Ethanol, 2-(tetradecyloxy)- | Alcohol | 2136-70-1 | ND | ND | ND | ND | 12.86 ± 14.70 |
| 37 | 13.127 | 2-Octanol, (R)- | Alcohol | 5978-70-1 | ND | ND | ND | 20.61 ± 16.15 | ND |
| 38 | 13.128 | Propylene Glycol | Alcohol | 57-55-6 | ND | ND | 11.94 ± 1.88 | ND | ND |
| 39 | 13.427 | Cyclohexanol, 2,6-dimethyl- | Alcohol | 5337-72-4 | 387.19 ± 41.48 | 132.71 ± 20.94 | 28.49 ± 20.14 | 165.04 ± 9.73 | 89.30 ± 6.55 |
| 40 | 13.561 | 5-Octen-1-ol, (Z)- | Alcohol | 64275-73-6 | 639.98 ± 48.26 | ND | ND | ND | ND |
| 41 | 13.569 | 2-Octen-1-ol, (E)- | Alcohol | 18409-17-1 | ND | ND | ND | 79.42 ± 2.59 | ND |
| 42 | 13.897 | Cyclohexanol, 5-methyl-2-(1-methylethyl)- | Alcohol | 1490-4-6 | ND | 139.83 ± 24.91 | ND | ND | 101.26 ± 3.33 |
| 43 | 14.113 | 1-Nonanol | Alcohol | 143-8-8 | 682.04 ± 53.48 | ND | ND | ND | ND |
| 44 | 14.171 | (5-Ethylcyclopent-1-enyl) methanol | Alcohol | 36431-59-1 | 233.87 ± 16.84 | ND | ND | ND | ND |
| 45 | 14.358 | 3-Cyclohexene-1-ethanol | Alcohol | 18240-10-3 | ND | ND | ND | ND | 10.43 ± 0.39 |
| 46 | 14.396 | 3-Nonen-1-ol, (Z)- | Alcohol | 10340-23-5 | 136.46 ± 10.32 | ND | ND | ND | ND |
| 47 | 14.4 | 1,9-Nonanediol | Alcohol | 3937-56-2 | ND | 12.39 ± 0.15 | ND | 12.81 ± 1.03 | ND |
| 48 | 14.556 | L-α-Terpineol | Alcohol | 10482-56-1 | ND | 576.54 ± 73.37 | ND | 1387.4 ± 60.83 | 91.93 ± 130.00 |
| 49 | 14.556 | α-Terpineol | Alcohol | 98-55-5 | 5575.53 ± 161.68 | 560.19 ± 0.00 | 438.00 ± 50.22 | ND | 208.11 ± 147.45 |
| 50 | 14.76 | (6Z)-Nonen-1-ol | Alcohol | 35854-86-5 | 155.52 ± 12.61 | ND | ND | ND | ND |
| 51 | 14.805 | 2-Cyclohexene-1-methanol, 2,6,6-trimethyl- | Alcohol | 6627-74-3 | 1238.19 ± 103.15 | ND | ND | ND | ND |
| 52 | 14.898 | 1,3-Butanediol | Alcohol | 107-88-0 | ND | ND | 12.69 ± 8.97 | ND | ND |
| 53 | 15.285 | (3R,6S)-2,2,6-Trimethyl-6-vinyltetrahydro-2H-pyran-3-ol | Alcohol | 39028-58-5 | 288.30 ± 33.37 | ND | ND | ND | ND |
| 54 | 15.712 | 2,6-Octadien-1-ol, 3,7-dimethyl-, (Z)- | Alcohol | 106-25-2 | 2259.80 ± 200.85 | ND | ND | ND | ND |
| 55 | 15.803 | Benzenemethanol, α-methyl- | Alcohol | 98-85-1 | 226.89 ± 22.47 | 31.31 ± 5.41 | 23.31 ± 2.67 | 70.17 ± 3.83 | 8.19 ± 1.57 |
| 56 | 16.278 | 1-Octanol, 5,7,7-trimethyl-2-(1,3,3-trimethylbutyl)- | Alcohol | 36400-98-3 | ND | ND | ND | ND | 30.74 ± 22.17 |
| 57 | 16.476 | Benzyl alcohol | Alcohol | 100-51-6 | 8971.43 ± 891.56 | 754.23 ± 92.37 | 402.45 ± 52.56 | 1514.09 ± 76.02 | 85.27 ± 3.30 |
| 58 | 16.555 | 1,2-Hexanediol | Alcohol | 6920-22-5 | ND | ND | 24.75 ± 1.08 | ND | ND |
| 59 | 16.863 | Phenylethyl Alcohol | Alcohol | 60-12-8 | 5521.27 ± 546.45 | 537.9 ± 78.74 | 246.63 ± 30.54 | 971.84 ± 45.13 | 74.53 ± 14.35 |
| 60 | 17.146 | 2,6,10,10-Tetramethyl-1-oxaspiro [4.5] decan-6-ol | Alcohol | 77981-89-6 | 81.42 ± 15.08 | ND | ND | ND | ND |
| 61 | 17.509 | 1-Cyclohexene-1-propanol, α,2,6,6-tetramethyl- | Alcohol | 3293-47-8 | 545.33 ± 46.16 | ND | ND | ND | ND |
| 62 | 17.522 | 1-Dodecanol | Alcohol | 112-53-8 | ND | 39.70 ± 3.69 | ND | ND | 8.10 ± 5.77 |
| 63 | 17.65 | β-Ethylphenethyl alcohol | Alcohol | 2035-94-1 | 45.93 ± 5.57 | ND | ND | ND | ND |
| 64 | 18.233 | 3-Phenylpropanol | Alcohol | 122-97-4 | 1301.93 ± 158.72 | ND | ND | ND | ND |
| 65 | 18.274 | 1,6,10-Dodecatrien-3-ol, 3,7,11-trimethyl-, (E)- | Alcohol | 40716-66-3 | ND | 12.74 ± 3.92 | 4.82 ± 0.18 | 8.35 ± 0.93 | 1.88 ± 1.36 |
| 66 | 19.1 | Cedrol | Alcohol | 77-53-2 | ND | ND | ND | ND | 2.62 ± 1.91 |
| 67 | 19.966 | α-Bisabolol | Alcohol | 515-69-5 | 283.44 ± 50.87 | 17.31 ± 2.16 | 11.32 ± 1.11 | 41.47 ± 5.05 | 13.58 ± 2.96 |
| 68 | 20.465 | 2-Propen-1-ol, 3-phenyl- | Alcohol | 104-54-1 | 22.18 ± 1.44 | ND | ND | ND | ND |
| 69 | 20.677 | Isophytol | Alcohol | 505-32-8 | 14.13 ± 2.72 | ND | 4.01 ± 3.22 | 28.89 ± 4.44 | 6.55 ± 1.55 |
| 70 | 21.213 | 3-Buten-2-ol, 3-methyl-4-(2,6,6-trimethyl-2-cyclohexen-1-yl)- | Alcohol | 70172-0-8 | ND | 5.46 ± 0.07 | ND | ND | ND |
| 71 | 23.687 | Phytol | Alcohol | 150-86-7 | ND | ND | 2.61 ± 1.12 | ND | 0.88 ± 0.62 |
| 72 | 4.13 | Butanal, 3-methyl- | Aldehyde | 590-86-3 | ND | 38.58 ± 0.00 | ND | ND | 12.59 ± 8.92 |
| 73 | 5.859 | Hexanal | Aldehyde | 66-25-1 | 1899.74 ± 245.83 | 787.68 ± 67.18 | 50.71 ± 5.15 | 351.57 ± 13.58 | 625.44 ± 6.17 |
| 74 | 6.567 | 2-Pentenal, (E)- | Aldehyde | 1576-87-0 | ND | 197.22 ± 5.93 | ND | 127.21 ± 13.25 | 105.57 ± 11.62 |
| 75 | 8.054 | 2-Hexenal | Aldehyde | 505-57-7 | ND | 1084.96 ± 15.02 | ND | ND | ND |
| 76 | 8.059 | 2-Hexenal, (E)- | Aldehyde | 6728-26-3 | ND | 1126.97 ± 0.00 | ND | 460.49 ± 27.8 | ND |
| 77 | 8.474 | 4-Heptenal, (Z)- | Alcohol | 6728-31-0 | ND | 34.55 ± 2.37 | ND | ND | 14.54 ± 10.28 |
| 78 | 9.236 | Octanal | Aldehyde | 124-13-0 | 292.28 ± 0.00 | 93.22 ± 2.00 | 26.76 ± 2.39 | 75.75 ± 20.82 | 139.03 ± 4.87 |
| 79 | 10.765 | Nonanal | Aldehyde | 124-19-6 | 1291.52 ± 249.42 | 774.66 ± 77.68 | 203.68 ± 21.36 | 423.41 ± 25.49 | 1274.44 ± 71.51 |
| 80 | 10.976 | Hexanal, 4-methyl- | Aldehyde | 41065-97-8 | ND | ND | ND | 160.08 ± 113.3 | ND |
| 81 | 11.042 | 5-Ethylcyclopent-1-enecarboxaldehyde | Aldehyde | 36431-60-4 | ND | ND | ND | 53.93 ± 38.15 | ND |
| 82 | 11.207 | 2-Octenal, (E)- | Aldehyde | 2548-87-0 | 462.21 ± 36.93 | 94.72 ± 9.44 | 15.07 ± 1.21 | 69.99 ± 4.04 | 71.16 ± 3.97 |
| 83 | 11.646 | 2,4-Heptadienal, (E, E)- | Aldehyde | 4313-3-5 | ND | 131.23 ± 38.61 | 25.09 ± 1.91 | 125.13 ± 8.29 | 119.13 ± 6.43 |
| 84 | 12.188 | Decanal | Aldehyde | 112-31-2 | ND | 188.97 ± 22.76 | ND | ND | 226.01 ± 21.73 |
| 85 | 12.406 | Benzaldehyde | Aldehyde | 100-52-7 | 6861.91 ± 761.44 | 452.46 ± 61.79 | 148.48 ± 20.71 | 967.35 ± 48.79 | 162.35 ± 8.80 |
| 86 | 12.622 | 2-Nonenal, (E)- | Aldehyde | 18829-56-6 | ND | 209.61 ± 19.46 | 44.07 ± 4.42 | 55.94 ± 7.42 | 167.85 ± 16.2 |
| 87 | 13.246 | 2,6-Nonadienal, (E, Z)- | Aldehyde | 557-48-2 | ND | 121.26 ± 14.96 | ND | 18.71 ± 9.74 | 54.58 ± 4.31 |
| 88 | 13.527 | Undecanal | Aldehyde | 112-44-7 | ND | 31.39 ± 4.84 | ND | ND | 33.36 ± 6.05 |
| 89 | 13.701 | 1-Cyclohexene-1-carboxaldehyde, 2,6,6-trimethyl- | Aldehyde | 432-25-7 | 333.79 ± 33.93 | 86.84 ± 13.16 | 57.64 ± 6.85 | 157.10 ± 6.33 | 75.73 ± 6.10 |
| 90 | 13.856 | Benzeneacetaldehyde | Aldehyde | 122-78-1 | 369.76 ± 46.13 | 134.94 ± 11.77 | 180.23 ± 23.90 | 249.68 ± 20.32 | 92.09 ± 6.97 |
| 91 | 13.967 | 2-Decenal, (E)- | Aldehyde | 3913-81-3 | ND | 98.75 ± 4.30 | ND | ND | 84.38 ± 17.94 |
| 92 | 13.998 | 1,3-Cyclohexadiene-1-carboxaldehyde, 2,6,6-trimethyl- | Aldehyde | 116-26-7 | ND | ND | 96.52 ± 10.49 | 198.20 ± 9.08 | ND |
| 93 | 14.481 | 1-Cyclohexene-1-carboxaldehyde, 5,5-dimethyl-3-oxo- | Aldehyde | 56621-35-3 | 136.63 ± 12.08 | ND | ND | ND | ND |
| 94 | 14.62 | 2,4-Nonadienal, (E, E)- | Aldehyde | 5910-87-2 | ND | 19.76 ± 2.59 | ND | ND | 14.18 ± 1.66 |
| 95 | 15.193 | 4-Oxohex-2-enal | Aldehyde | 20697-55-6 | 79.57 ± 4.37 | 39.27 ± 5.43 | 36.80 ± 4.83 | 43.48 ± 3.32 | 16.13 ± 1.39 |
| 96 | 15.237 | 2-Undecenal | Aldehyde | 2463-77-6 | ND | 39.84 ± 0.40 | ND | ND | ND |
| 97 | 15.35 | 2,4-Decadienal, (E, E)- | Aldehyde | 25152-84-5 | ND | ND | ND | ND | 32.70 ± 6.57 |
| 98 | 17.119 | Benzeneacetaldehyde, α-ethylidene- | Aldehyde | 4411-89-6 | ND | ND | 27.74 ± 4.62 | 42.37 ± 3.48 | ND |
| 99 | 18.227 | Pentadecanal- | Aldehyde | 2765-11-9 | ND | 7.09 ± 0.85 | ND | ND | 9.97 ± 7.79 |
| 100 | 18.605 | 5-Methyl-2-phenyl-2-hexenal | Aldehyde | 21834-92-4 | ND | ND | 15.76 ± 2.23 | 11.4 ± 1.31 | ND |
| 101 | 22.385 | 3,5-di-tert-Butyl-4-hydroxybenzaldehyde | Aldehyde | 1620-98-0 | ND | ND | ND | ND | 1.00 ± 0.82 |
| 102 | 7.639 | Hexanoic acid, methyl ester | Ester | 106-70-7 | ND | 143.89 ± 11.32 | ND | ND | ND |
| 103 | 8.245 | 2-Penten-1-ol, acetate, (Z)- | Ester | 42125-10-0 | ND | ND | ND | 2.77 ± 0.28 | ND |
| 104 | 8.495 | Hexanoic acid, ethyl ester | Ester | 123-66-0 | 726.72 ± 0.00 | ND | ND | ND | ND |
| 105 | 8.767 | 4-Hexenoic acid, methyl ester | Ester | 2396-79-4 | ND | 125.86 ± 22.74 | ND | ND | ND |
| 106 | 9.019 | Acetic acid, hexyl ester | Ester | 142-92-7 | 1526.70 ± 183.09 | ND | ND | ND | ND |
| 107 | 9.441 | 3-Hexenoic acid, ethyl ester | Ester | 2396-83-0 | ND | ND | ND | 38.14 ± 1.54 | ND |
| 108 | 9.745 | n-Caproic acid vinyl ester | Ester | 3050-69-9 | ND | ND | ND | 16.73 ± 0.68 | ND |
| 109 | 11.449 | Hexanoic acid, 3,7-dimethyl-6-octenyl ester | Ester | 10580-25-3 | 834.84 ± 36.28 | ND | ND | ND | ND |
| 110 | 12.115 | Nonanoic acid, methyl ester | Ester | 1731-84-6 | ND | 56.46 ± 12.29 | ND | 22.45 ± 2.70 | 11.32 ± 8.17 |
| 111 | 13.659 | Butanoic acid, 4-hydroxy- | Ester | 591-81-1 | ND | ND | 25.96 ± 4.36 | ND | ND |
| 112 | 14.841 | Cyclohexanol, 2,2-dimethyl-, acetate | Ester | 65322-50-1 | ND | ND | ND | ND | 29.64 ± 1.76 |
| 113 | 14.89 | Hexanoic acid, 1-methylethyl ester | Ester | 2311-46-8 | ND | ND | ND | ND | 8.47 ± 6.24 |
| 114 | 15.475 | Methyl salicylate | Ester | 119-36-8 | 12.48 ± 15.90 | ND | ND | 14.63 ± 1.25 | 2.23 ± 1.58 |
| 115 | 15.845 | Dodecanoic acid, methyl ester | Ester | 111-82-0 | 37.68 ± 10.73 | ND | ND | ND | ND |
| 116 | 15.885 | Benzoic acid, 2-hydroxy-, ethyl ester | Ester | 118-61-6 | 103.13 ± 0.30 | ND | 26.92 ± 3.03 | 136.46 ± 11.07 | ND |
| 117 | 16.669 | 2,2,4-Trimethyl-1,3-pentanediol diisobutyrate | Ester | 6846-50-0 | 20.38 ± 5.42 | 13.22 ± 2.38 | ND | ND | 13.27 ± 2.60 |
| 118 | 16.693 | Benzenepropanoic acid, ethyl ester | Ester | 2021-28-5 | ND | ND | ND | 29.69 ± 1.98 | ND |
| 119 | 18.05 | Methyl tetradecanoate | Ester | 124-10-7 | ND | 15.07 ± 2.50 | ND | 17.43 ± 2.11 | ND |
| 120 | 18.11 | 2(3H)-Furanone, dihydro-5-pentyl- | Ester | 104-61-0 | 77.11 ± 9.58 | ND | ND | ND | ND |
| 121 | 18.452 | Tetradecanoic acid, ethyl ester | Ester | 124-6-1 | ND | 9.75 ± 1.05 | 6.86 ± 0.19 | 19.95 ± 0.13 | 8.35 ± 1.14 |
| 122 | 18.76 | Nonanoic acid, 9-oxo-, ethyl ester | Ester | 3433-16-7 | ND | 35.92 ± 3.29 | ND | 11.47 ± 1.34 | 21.14 ± 1.09 |
| 123 | 19.076 | Pentadecanoic acid, methyl ester | Ester | 7132-64-1 | ND | 23.25 ± 1.47 | ND | 16.47 ± 12.29 | ND |
| 124 | 19.271 | 2-Hexadecen-1-ol, 3,7,11,15-tetramethyl-, acetate, [R- [R*, R*-(E)]]- | Ester | 10236-16-5 | ND | ND | ND | 2.57 ± 1.82 | ND |
| 125 | 19.561 | (Z)-Ethyl pentadec-9-enoate | Ester | 56219-9-1 | ND | 16.62 ± 3.63 | 9.39 ± 0.35 | 27.29 ± 2.28 | 9.80 ± 1.75 |
| 126 | 20.013 | Benzoic acid, 2,5-dihydroxy-, ethyl ester | Ester | 3943-91-7 | ND | 8.33 ± 1.23 | 4.16 ± 0.47 | 22.02 ± 0.99 | ND |
| 127 | 20.062 | Hexadecanoic acid, methyl ester | Ester | 112-39-0 | 29.14 ± 1.94 | 63.26 ± 19.90 | 16.91 ± 4.98 | 134.91 ± 13.94 | 11.96 ± 2.50 |
| 128 | 20.235 | Ethanol, 2-(dodecyloxy)- | Ester | 4536-30-5 | ND | 22.74 ± 9.21 | ND | ND | ND |
| 129 | 20.248 | Hexanedioic acid, mono(2-ethylhexyl) ester | Ester | 4337-65-9 | ND | ND | 17.93 ± 1.35 | ND | ND |
| 130 | 20.419 | Hexadecanoic acid, ethyl ester | Ester | 628-97-7 | 79.28 ± 10.06 | 72.78 ± 16.8 | 49.66 ± 7.53 | 191.24 ± 10.27 | 68.53 ± 12.10 |
| 131 | 20.842 | Ethyl 9-hexadecenoate | Ester | 54546-22-4 | ND | 3.50 ± 0.28 | 2.24 ± 0.26 | 4.74 ± 0.64 | 2.21 ± 0.38 |
| 132 | 21.089 | 2(4H)-Benzofuranone, 5,6,7,7a-tetrahydro-4,4,7a-trimethyl-, (R)- | Ester | 17092-92-1 | 76.96 ± 8.67 | 26.8 ± 3.65 | 10.6 ± 0.88 | 19.29 ± 2.37 | 10.63 ± 0.87 |
| 133 | 22.102 | 9-Octadecenoic acid, methyl ester, (E)- | Ester | 1937-62-8 | ND | 7.83 ± 1.46 | 2.59 ± 1.08 | 8.24 ± 5.83 | 1.74 ± 0.5 |
| 134 | 22.418 | (E)-9-Octadecenoic acid ethyl ester | Ester | 6114-18-7 | ND | 3.27 ± 0.11 | 3.92 ± 1.11 | 11.02 ± 1.73 | 2.19 ± 1.77 |
| 135 | 22.525 | 9,12-Octadecadienoic acid (Z, Z)-, methyl ester | Ester | 112-63-0 | ND | 6.60 ± 2.51 | 4.42 ± 2.05 | 27.08 ± 3.04 | 2.54 ± 0.91 |
| 136 | 22.87 | Linoleic acid ethyl ester | Ester | 544-35-4 | ND | ND | ND | 64.94 ± 4.52 | ND |
| 137 | 22.875 | 1,2-Benzenedicarboxylic acid, bis(2-methylpropyl) ester | Ester | 84-69-5 | ND | 30.71 ± 9.41 | ND | ND | ND |
| 138 | 23.177 | 9,12,15-Octadecatrienoic acid, methyl ester, (Z, Z, Z)- | Ester | 301-0-8 | ND | 6.57 ± 4.27 | 5.43 ± 1.33 | 36.06 ± 5.66 | ND |
| 139 | 23.554 | 9,12,15-Octadecatrienoic acid, ethyl ester, (Z, Z, Z)- | Ester | 1191-41-9 | ND | 4.30 ± 1.62 | 19.81 ± 4.69 | 79.04 ± 11.63 | 5.14 ± 3.66 |
| 140 | 24.622 | Dibutyl phthalate | Ester | 84-74-2 | ND | 26.87 ± 5.65 | 10.72 ± 2.70 | 29.11 ± 9.12 | 13.35 ± 3.33 |
| 141 | 4.11 | 3-Heptanone, 5-ethyl-4-methyl- | Ketone | 27607-63-2 | 427.04 ± 148.89 | ND | ND | ND | ND |
| 142 | 4.826 | 1-Penten-3-one | Ketone | 1629-58-9 | ND | 67.06 ± 0.00 | ND | ND | ND |
| 143 | 9.046 | Acetoin | Ketone | 513-86-0 | ND | ND | ND | 58.92 ± 0.9 | ND |
| 144 | 9.149 | 2-Decanone | Ketone | 693-54-9 | 209.66 ± 40.73 | ND | ND | ND | ND |
| 145 | 9.15 | Cyclooctanone | Ketone | 502-49-8 | ND | ND | ND | ND | 8.06 ± 5.70 |
| 146 | 9.164 | 2-Octanone | Ketone | 111-13-7 | ND | ND | 12.33 ± 5.30 | ND | ND |
| 147 | 9.355 | 3-Hepten-2-one | Ketone | 1119-44-4 | ND | ND | 23.14 ± 2.19 | ND | ND |
| 148 | 9.851 | 6-Octen-2-one, (Z)- | Ketone | 74810-53-0 | ND | ND | 95.39 ± 10.89 | ND | 11.11 ± 0.22 |
| 149 | 9.919 | 5-Hepten-2-one, 6-methyl- | Ketone | 110-93-0 | 268.52 ± 124.89 | 103.45 ± 8.50 | ND | 68.42 ± 3.78 | 100.91 ± 3.15 |
| 150 | 12.786 | 1,13-Tetradecadien-3-one | Ketone | 58879-40-6 | ND | ND | ND | ND | 1.05 ± 0.76 |
| 151 | 13.021 | 3,5-Octadien-2-one, (E, E)- | Ketone | 30086-2-3 | ND | 41.86 ± 5.51 | ND | ND | ND |
| 152 | 13.038 | 3-Undecanone | Ketone | 2216-87-7 | ND | ND | ND | ND | 35.28 ± 2.59 |
| 153 | 13.315 | Isophorone | Ketone | 78-59-1 | 1788.28 ± 141.75 | 75.77 ± 7.31 | 96.07 ± 11.07 | 234.98 ± 16.41 | 68.28 ± 4.65 |
| 154 | 13.987 | Acetophenone | Ketone | 98-86-2 | 902.04 ± 87.73 | ND | ND | ND | ND |
| 155 | 14.487 | 2,6,6-Trimethyl-2-cyclohexene-1,4-dione | Ketone | 1125-21-9 | ND | 27.56 ± 5.69 | ND | 30.88 ± 2.77 | 15.18 ± 1.16 |
| 156 | 15.98 | 2-Butanone, 4-(2,6,6-trimethyl-2-cyclohexen-1-yl)- | Ketone | 31499-72-6 | 670.96 ± 68.51 | ND | 2.52 ± 1.78 | 5.95 ± 0.84 | ND |
| 157 | 16.148 | 2-Butanone, 4-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | Ketone | 17283-81-7 | 845.75 ± 112.45 | ND | ND | ND | ND |
| 158 | 16.333 | α-Ionone | Ketone | 127-41-3 | 2371.64 ± 209.46 | 406.52 ± 21.47 | ND | 1047.11 ± 37.32 | 206.02 ± 24.15 |
| 159 | 16.379 | 5,9-Undecadien-2-one, 6,10-dimethyl-, (E)- | Ketone | 3796-70-1 | ND | ND | ND | ND | 54.02 ± 6.97 |
| 160 | 17.259 | β-Ionone | Ketone | 14901-7-6 | 1118.78 ± 93.39 | ND | ND | ND | ND |
| 161 | 17.695 | 7,8-Epoxy-α-ionone | Ketone | 37079-64-4 | ND | 35.46 ± 6.04 | ND | ND | 12.76 ± 2.18 |
| 162 | 17.899 | 4-(2,6,6-Trimethylcyclohexa-1,3-dienyl) but-3-en-2-one | Ketone | 1203-8-3 | ND | ND | 20.77 ± 15.07 | ND | ND |
| 163 | 18.828 | 2,6-Di-tert-butyl-4-hydroxy-4-methylcyclohexa-2,5-dien-1-one | Ketone | 10396-80-2 | ND | 23.6 ± 11.46 | ND | ND | 7.62 ± 3.05 |
| 164 | 19.193 | 2-Pentadecanone, 6,10,14-trimethyl- | Ketone | 502-69-2 | 47.10 ± 9.83 | 133.61 ± 17.95 | 22.07 ± 0.83 | 43.93 ± 3.74 | 65.58 ± 8.02 |
| 165 | 20.34 | 1H-Pyrrole-2,5-dione, 3-ethyl-4-methyl- | Ketone | 20189-42-8 | 20.31 ± 0.70 | 27.34 ± 4.25 | 8.31 ± 1.64 | 19.30 ± 3.15 | 5.70 ± 0.74 |
| 166 | 21.574 | 1H-Pyrrole-2,5-dione, 3-ethenyl-4-methyl- | Ketone | 21494-57-5 | ND | 4.13 ± 0.9 | ND | ND | ND |
| 167 | 25.062 | 7,9-Di-tert-butyl-1-oxaspiro (4,5) deca-6,9-diene-2,8-dione | Ketone | 82304-66-3 | ND | 15.15 ± 2.80 | 7.07 ± 1.82 | 11.54 ± 4.07 | 8.00 ± 0.74 |
| 168 | 4.615 | Dodecane | Alkane | 112-40-3 | ND | ND | 257.56 ± 25.57 | 251.09 ± 37.15 | 291.86 ± 411.65 |
| 169 | 4.616 | Octane, 2,4,6-trimethyl- | Alkane | 62016-37-9 | ND | 5.59 ± 1.2 | ND | ND | ND |
| 170 | 4.64 | Undecane, 4,6-dimethyl- | Alkane | 17312-82-2 | 450.14 ± 57.87 | ND | ND | ND | ND |
| 171 | 4.817 | Nonane, 4-methyl-5-propyl- | Alkane | 62185-55-1 | 675.10 ± 467.30 | ND | ND | ND | ND |
| 172 | 5.234 | Dodecane, 4,6-dimethyl- | Alkane | 61141-72-8 | 1198.39 ± 149.13 | ND | ND | ND | ND |
| 173 | 6.135 | Heptadecane, 2,6,10,15-tetramethyl- | Alkane | 54833-48-6 | 794.25 ± 49.13 | ND | ND | ND | ND |
| 174 | 6.35 | Undecane | Alkane | 1120-21-4 | ND | ND | 38.79 ± 2.84 | ND | ND |
| 175 | 6.365 | Undecane, 3,6-dimethyl- | Alkane | 17301-28-9 | ND | ND | ND | ND | 125.99 ± 14.83 |
| 176 | 6.95 | 5-Ethyldecane | Alkane | 17302-36-2 | ND | ND | 2.93 ± 2.20 | ND | ND |
| 177 | 6.962 | Tridecane, 2,5-dimethyl- | Alkane | 56292-66-1 | ND | 37.41 ± 8.31 | 26.16 ± 2.47 | ND | 28.75 ± 2.53 |
| 178 | 7.46 | Undecane, 3-methyl- | Alkane | 1002-43-3 | ND | 90 ± 0.43 | ND | 98.86 ± 1.80 | 68.18 ± 5.72 |
| 179 | 7.46 | Heptane, 2,2,3,3,5,6,6-heptamethyl- | Alkane | 7225-67-4 | 500.19 ± 25.78 | ND | ND | ND | ND |
| 180 | 8.015 | Hexadecane | Alkane | 544-76-3 | 9011.18 ± 683.35 | ND | ND | ND | 392.50 ± 23.97 |
| 181 | 8.555 | Undecane, 3-methylene- | Alkane | 71138-64-2 | ND | 59.77 ± 1.40 | 45.32 ± 3.83 | ND | 31.23 ± 22.10 |
| 182 | 8.76 | Heptadecane, 8-methyl- | Alkane | 13287-23-5 | 1737.39 ± 122.43 | ND | ND | ND | ND |
| 183 | 8.889 | Nonane, 5-methyl-5-propyl- | Alkane | 17312-75-3 | 193.12 ± 84.87 | ND | ND | ND | ND |
| 184 | 8.89 | Dodecane, 2,6,11-trimethyl- | Alkane | 31295-56-4 | 314.87 ± 55.15 | ND | ND | ND | ND |
| 185 | 9.551 | Heptadecane | Alkane | 629-78-7 | ND | 143.91 ± 27.1 | 64.57 ± 9.45 | 105.42 ± 74.66 | 31.09 ± 22.34 |
| 186 | 9.706 | Cyclopentane, 1,1,3-trimethyl- | Alkane | 4516-69-2 | ND | 93.53 ± 1.81 | ND | 7.94 ± 1.45 | 94.99 ± 4.76 |
| 187 | 9.768 | Cyclohexane, ethyl- | Alkane | 1678-91-7 | 424.70 ± 253.46 | ND | ND | ND | ND |
| 188 | 10.253 | Nonadecane, 9-methyl- | Alkane | 13287-24-6 | ND | ND | ND | ND | 46.13 ± 32.96 |
| 189 | 10.413 | Hexadecane, 2,6,10,14-tetramethyl- | Alkane | 638-36-8 | 362.81 ± 50.8 | ND | 15.08 ± 1.46 | 27.15 ± 19.25 | ND |
| 190 | 10.462 | Heneicosane | Alkane | 629-94-7 | ND | 103.81 ± 58.12 | ND | ND | 123.51 ± 29.93 |
| 191 | 10.855 | Cyclopentane, 1,1′-(1,4-butandiyl) bis- | Alkane | 2980-70-3 | ND | ND | ND | ND | 37.36 ± 26.42 |
| 192 | 10.985 | Tetradecane | Alkane | 629-59-4 | ND | 216.64 ± 2.04 | 51.74 ± 37.26 | ND | 300.52 ± 62.27 |
| 193 | 11.731 | Tetradecane, 3-methyl- | Alkane | 18435-22-8 | ND | ND | ND | ND | 53.48 ± 2.46 |
| 194 | 11.835 | Hexadecane, 2-methyl- | Alkane | 1560-92-5 | ND | ND | ND | ND | 36.08 ± 5.68 |
| 195 | 12.329 | Pentadecane | Alkane | 629-62-9 | ND | 43.48 ± 6.12 | ND | 47.39 ± 3.18 | ND |
| 196 | 12.585 | Triacontane | Alkane | 638-68-6 | 66.54 ± 15.3 | 11.65 ± 1.24 | ND | ND | ND |
| 197 | 12.863 | Cyclopropane, octyl- | Alkane | 1472-9-9 | ND | ND | ND | 99.61 ± 70.96 | ND |
| 198 | 12.9 | Tritetracontane | Alkane | 7098-21-7 | ND | 26.41 ± 7.91 | ND | ND | ND |
| 199 | 13.122 | Pentadecane, 1-methoxy-13-methyl- | Alkane | 56196-9-9 | ND | ND | ND | ND | 17.52 ± 12.48 |
| 200 | 13.181 | Pentadecane, 3-methyl- | Alkane | 2882-96-4 | ND | ND | ND | ND | 63.42 ± 5.82 |
| 201 | 14.4 | 2-Methyltetracosane | Alkane | 1560-78-7 | ND | ND | ND | ND | 39.65 ± 11.31 |
| 202 | 14.79 | Hexacosane | Alkane | 630-1-3 | ND | 49.23 ± 5.89 | ND | ND | ND |
| 203 | 17.178 | 1H-3a,7-Methanoazulene, 2,3,6,7,8,8a-hexahydro-1,4,9,9-tetramethyl-, (1α,3aα,7α,8aβ)- | Alkane | 560-32-7 | ND | ND | ND | ND | 31.66 ± 19.55 |
| 204 | 10.33 | Tridecane, 3-methylene- | Olefin | 19780-34-8 | ND | ND | ND | ND | 22.81 ± 8.52 |
| 205 | 10.34 | 1-Decene, 3,3,4-trimethyl- | Olefin | 49622-17-5 | 77.17 ± 28.39 | ND | ND | ND | ND |
| 206 | 10.629 | 2,4,4,6,6,8,8-Heptamethyl-1-nonene | Olefin | 15796-4-0 | ND | ND | ND | ND | 13.09 ± 9.41 |
| 207 | 11.095 | Nonane, 5-methylene- | Olefin | 6795-79-5 | ND | ND | ND | ND | 22.96 ± 2.97 |
| 208 | 11.47 | 1-Hexene, 3,5,5-trimethyl- | Olefin | 4316-65-8 | ND | 59.24 ± 33.84 | ND | ND | 77.18 ± 54.71 |
| 209 | 11.934 | 2-Hexene, 3,5,5-trimethyl- | Olefin | 26456-76-8 | ND | ND | ND | 38.37 ± 2.16 | ND |
| 210 | 12.03 | Cyclohexene, 3-methyl- | Olefin | 591-48-0 | 2716.23 ± 87.14 | ND | ND | ND | ND |
| 211 | 13.02 | Bicyclo [3.2.1] oct-2-ene, 3-(1,1-dimethylethoxy)- | Olefin | 37609-41-9 | ND | ND | ND | 15.64 ± 19.81 | ND |
| 212 | 13.174 | cis-α-Bergamotene | Olefin | 18252-46-5 | 1002.97 ± 129.23 | 20.97 ± 3.62 | 30.66 ± 17.36 | 138.95 ± 17.11 | 12.16 ± 8.62 |
| 213 | 13.5 | 1,7-Octadiene | Olefin | 3710-30-3 | 180.94 ± 2.17 | ND | ND | ND | ND |
| 214 | 14.62 | 1,4-Methano-1H-indene, octahydro-1,7a-dimethyl-4-(1-methylethenyl)-, [1S-(1α,3aβ,4α,7aβ)]- | Olefin | 87064-18-4 | 125.07 ± 7.81 | ND | ND | ND | ND |
| 215 | 14.999 | trans-α-Bergamotene | Olefin | 13474-59-4 | ND | ND | ND | ND | 88.00 ± 64.01 |
| 216 | 17.252 | Neophytadiene | Olefin | 504-96-1 | ND | 778.38 ± 113.42 | 392.06 ± 49.05 | 1426.64 ± 87.07 | 561.49 ± 77.93 |
| 217 | 11.309 | Acetic acid | Acid | 64-19-7 | 786.62 ± 72.16 | 632.90 ± 58.03 | 1525.83 ± 181.75 | 2575.75 ± 304.82 | 371.19 ± 27.02 |
| 218 | 14.142 | Butanoic acid, 2-methyl- | Acid | 116-53-0 | ND | ND | 140.91 ± 15.5 | 200.07 ± 11.81 | ND |
| 219 | 14.663 | Butanoic acid, 2-ethyl-, 1,2,3-propanetriyl ester | Acid | 56554-54-2 | ND | ND | 29.61 ± 5.1 | 67.17 ± 11.55 | 29.48 ± 7.13 |
| 220 | 14.844 | 2-Hexenoic acid, 3,4,4-trimethyl-5-oxo-, (Z)- | Acid | 14919-56-3 | ND | 54.45 ± 9.51 | ND | 110.23 ± 14.92 | ND |
| 221 | 15.437 | cis-13,16-Docasadienoic acid | Acid | 7370-49-2 | ND | ND | ND | ND | 8.19 ± 6.06 |
| 222 | 16.109 | Hexanoic acid | Acid | 142-62-1 | ND | 189.88 ± 18.55 | 114.98 ± 11.05 | 159.88 ± 9.98 | 54.09 ± 9.81 |
| 223 | 17.13 | Undecylenic acid | Acid | 112-38-9 | ND | 16.22 ± 3.27 | ND | ND | ND |
| 224 | 17.363 | 2-Hexenoic acid, (E)- | Acid | 13419-69-7 | ND | 61.25 ± 10.92 | 32.02 ± 4.59 | 32.92 ± 8.30 | 15.13 ± 11.00 |
| 225 | 17.928 | 3-Heptenoic acid | Acid | 29901-85-7 | ND | 32.38 ± 0.70 | ND | ND | ND |
| 226 | 18.339 | Octanoic acid | Acid | 124-7-2 | 59.52 ± 0.78 | ND | ND | ND | ND |
| 227 | 19.378 | Nonanoic acid | Acid | 112-5-0 | 69.87 ± 39.40 | 217.61 ± 34.9 | 74.06 ± 10.39 | 19.66 ± 18.97 | 107.71 ± 17.99 |
| 228 | 20.38 | n-Decanoic acid | Acid | 334-48-5 | ND | ND | 3.90 ± 2.86 | ND | ND |
| 229 | 7.815 | D-Limonene | Triterpenoid | 5989-27-5 | ND | ND | ND | ND | 2.86 ± 4.05 |
| 230 | 8.714 | 1,3,6-Octatriene, 3,7-dimethyl-, (Z)- | Triterpenoid | 3338-55-4 | ND | ND | 161.20 ± 13.41 | 429.44 ± 15.03 | 148.57 ± 8.24 |
| 231 | 12.207 | Copaene | Triterpenoid | 3856-25-5 | 770.2 ± 0 | ND | ND | ND | ND |
| 232 | 14.309 | (E)-β-Famesene | Triterpenoid | 18794-84-8 | 309.54 ± 28.32 | 54.65 ± 8.29 | 40.50 ± 4.87 | 106.07 ± 4.43 | 33.96 ± 2.59 |
| 233 | 15.041 | β-Bisabolene | Triterpenoid | 495-61-4 | 951.06 ± 81.43 | 64.72 ± 8.38 | 75.00 ± 11.84 | 229.62 ± 6.71 | 64.8 ± 4.57 |
| 234 | 15.086 | (Z)-1-Methyl-4-(6-methylhept-5-en-2-ylidene) cyclohex-1-ene | Triterpenoid | 13062-0-5 | 394.35 ± 113.51 | 59.03 ± 9.64 | 177.04 ± 10.98 | 436.27 ± 20.12 | 78.04 ± 6.76 |
| 235 | 15.262 | α-Farnesene | Triterpenoid | 502-61-4 | ND | ND | 32.53 ± 2.62 | 75.57 ± 4.10 | 40.70 ± 3.84 |
| 236 | 15.397 | Naphthalene, 1,2,3,5,6,8a-hexahydro-4,7-dimethyl-1-(1-methylethyl)-, (1S-cis)- | Triterpenoid | 483-76-1 | 559.67 ± 43 | 56.09 ± 10.59 | ND | 71.93 ± 2.38 | 17.94 ± 13.17 |
| 237 | 15.563 | Bicyclo [3.1.0] hexan-3-ol, 4-methyl-1-(1-methylethyl)- | Triterpenoid | 513-23-5 | ND | ND | ND | ND | 47.78 ± 6.13 |
| 238 | 8.767 | Pyrazine, methyl- | Pyrazine | 109-8-0 | ND | ND | 215.40 ± 17.42 | ND | ND |
| 239 | 9.737 | Pyrazine, 2,6-dimethyl- | Pyrazine | 108-50-9 | ND | ND | 95.44 ± 10.94 | ND | ND |
| 240 | 10.672 | Pyrazine, 2-ethyl-5-methyl- | Pyrazine | 13360-64-0 | ND | ND | 20.99 ± 15.17 | ND | ND |
| 241 | 10.843 | Pyrazine, 2-ethyl-3-methyl- | Pyrazine | 15707-23-0 | ND | ND | 112.33 ± 14.32 | ND | ND |
| 242 | 11.429 | Pyrazine, 3-ethyl-2,5-dimethyl- | Pyrazine | 13360-65-1 | ND | ND | 79.17 ± 57.18 | ND | ND |
| 243 | 11.643 | Pyrazine, 2,6-diethyl- | Pyrazine | 13067-27-1 | ND | ND | 80.11 ± 45.24 | ND | ND |
| 244 | 11.988 | Pyrazine, 2-ethenyl-6-methyl- | Pyrazine | 13925-9-2 | ND | ND | 34.98 ± 6.74 | ND | ND |
| 245 | 14.383 | 1-(6-Methyl-2-pyrazinyl)-1-ethanone | Pyrazine | 22047-26-3 | ND | ND | 30.11 ± 21.47 | ND | ND |
| 246 | 16.321 | Phenol, 2-methoxy- | Phenol | 90-05-1 | ND | ND | 239.59 ± 25.46 | ND | ND |
| 247 | 17.774 | Phenol | Phenol | 108-95-2 | 2868.72 ± 268.39 | 405.75 ± 53.38 | 240.47 ± 33.04 | 1310.53 ± 57.17 | ND |
| 248 | 18.108 | Phenol, 4-ethyl-2-methoxy- | Phenol | 2785-89-9 | ND | 32.39 ± 4.91 | 75.93 ± 10.34 | 284.16 ± 22.69 | 20.58 ± 0.75 |
| 249 | 19.451 | Phenol, 4-ethyl- | Phenol | 123-7-9 | 1830.97 ± 194.64 | 204.15 ± 25.37 | 105.88 ± 14.46 | 616.01 ± 33.17 | 63.54 ± 2.57 |
| 250 | 19.679 | 2-Methoxy-4-vinylphenol | Phenol | 7786-61-0 | ND | ND | 61.56 ± 10.30 | 34.25 ± 5.97 | ND |
| 251 | 19.854 | Thymol | Phenol | 89-83-8 | ND | ND | ND | 5.57 ± 1.44 | ND |
| 252 | 20.767 | 2,4-Di-tert-butylphenol | Phenol | 96-76-4 | 78.56 ± 7.27 | 43.42 ± 12.54 | 22.84 ± 13.01 | 16.73 ± 9.05 | 15.38 ± 3.66 |
| 253 | 22.143 | Phenol, 3,4,5-trimethoxy- | Phenol | 642-71-7 | ND | ND | 4.06 ± 0.75 | 6.71 ± 0.52 | ND |
| 254 | 3.777 | Furan, 2-ethyl- | Furan | 3208-16-0 | ND | 7.87 ± 1.76 | ND | ND | ND |
| 255 | 8.38 | Furan, 2-pentyl- | Furan | 3777-69-3 | 425.14 ± 34.84 | 60.46 ± 4.71 | 75.88 ± 54.03 | ND | 62.78 ± 13.59 |
| 256 | 11.886 | 5-Isopropyl-3,3-dimethyl-2-methylene-2,3-dihydrofuran | Furan | 81250-44-4 | ND | ND | ND | 9.52 ± 6.74 | ND |
| 257 | 18.029 | Furaneol | Furan | 3658-77-3 | ND | ND | 45.92 ± 8.14 | ND | ND |
| 258 | 21.381 | Benzofuran, 2,3-dihydro- | Furan | 496-16-2 | 23.21 ± 2.07 | 26.75 ± 5.06 | 44.20 ± 7.43 | 141.74 ± 9.84 | 9.04 ± 1.09 |
| 259 | 13.612 | 1H-Pyrrole, 2-ethyl- | Others | 1551-6-0 | ND | ND | 120.67 ± 13.03 | ND | ND |
| 260 | 15.398 | 1-Isopropyl-4,7-dimethyl-1,2,3,5,6,8a-hexahydronaphthalene | Others | 16729-1-4 | ND | ND | 38.16 ± 27.16 | ND | ND |
| 261 | 16.226 | 3-Nonen-5-yne, 4-ethyl- | Others | 74685-67-9 | ND | ND | ND | ND | 14.04 ± 1.86 |
| 262 | 16.748 | 10,11-Epoxycalamenene | Others | 143785-42-6 | 27.31 ± 3.69 | ND | ND | ND | ND |
| 263 | 17.061 | Benzyl nitrile | Others | 140-29-4 | ND | ND | 7.01 ± 1.03 | ND | ND |
| 264 | 17.462 | Ethanone, 1-(1H-pyrrol-2-yl)- | Others | 1072-83-9 | ND | ND | 14.31 ± 1.93 | ND | ND |
| 265 | 20.296 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | Others | 28564-83-2 | ND | ND | 9.55 ± 2.20 | ND | ND |
| 266 | 21.155 | Ethosuximide | Others | 77-67-8 | ND | ND | 0.93 ± 0.09 | ND | ND |
References
- Li, L.; Zhu, L.; Zhu, P. Analysis of the Current Situation of Bamboo Resources and Bamboo Industry Development in China. South China Agric. 2017, 11, 4. [Google Scholar] [CrossRef]
- Xia, Y.; Dong, J.; Zhong, G. The Development Status of Bamboo Leavf Extract. China Food Addit. 2009, 2, 77–81+148. [Google Scholar] [CrossRef]
- Dai, X.; Xu, C.; Dai, Q. Bamboo Resources and Research Progress. Shandong For. Sci. Technol. 2009, 39, 5. [Google Scholar] [CrossRef]
- Huang, L. Research on the Effects and Mechanism of Matzhu on the Quality and Storage Stability of Chiffon Cakes. Master’s Thesis, South China University of Technology, Guangzhou, China, 2023. [Google Scholar]
- Xia, D.Z.; Zhang, J.X.; Xiong, Z.C.; Huang, W.J.; Wei, Y.M.; Feng, W.Z.; Huang, J.L.; Ning, J.M. Effects of different fixation methods on the aroma quality of Anjibai tea. LWT Food Sci. Technol. 2024, 204, 9. [Google Scholar] [CrossRef]
- Zhang, Y.; Shao, C.; Lv, H.; Llin, Z.; Yu, L.; Zhu, Y. Comparative Analysis of Volatile Components and Key Aroma-Active Components in Baked Green Tea Harvested in Different Seasons. Food Sci. 2024, 45, 213–221. [Google Scholar] [CrossRef]
- You, Q.; Shi, Y.; Zhu, Y.; Yang, G.; Yan, H.; Lin, Z.; Lv, H. Effect of Different Processing Technologies on the Key Aroma-Active Compounds of Green Tea. Food Sci. 2023, 44, 7. [Google Scholar] [CrossRef]
- Sun, R.; Liu, X. The Development and Value of Enshi Yulu Production Technique. China Tea 2023, 45, 54–61. [Google Scholar] [CrossRef]
- Chen, B. Study on the Technology of Green Tea Powder and Instant Green Tea Powder Made from Tea Leaves. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2018. [Google Scholar]
- Ouyang, W.; Yu, Y.Y.; Wang, H.J.; Jiang, Y.W.; Hua, J.J.; Ning, J.M.; Yuan, H.B. Analysis of volatile metabolite variations in strip green tea during processing and effect of rubbing degree using untargeted and targeted metabolomics. Food Res. Int. 2022, 162, 112099. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Wang, J.J.; Kong, Y.S.; Zhu, Y.; Zhang, J.W.; Liu, H.; Wang, X.; Guo, G.Y.; Wang, G.M.; Liu, Z.H. Dynamic Changes of Volatile Compounds during the Xinyang Maojian Green Tea Manufacturing at an Industrial Scale. Foods 2022, 11, 2682. [Google Scholar] [CrossRef]
- Zhang, X. Preparation and Preliminary Application of Green High GABA Mulberry Powder. Master’s Thesis, Southwest University, Chongqing, China, 2023. [Google Scholar]
- Han, Z.; Rana, M.M.; Liu, G.; Gao, M.; Li, D.; Wu, F.; Li, X.; Wan, X.; Wei, S. Data on green tea flavor determinantes as affected by cultivars and manufacturing processes. Data Brief 2017, 10, 492–498. [Google Scholar] [CrossRef]
- Jiao, Y.F.; Song, Y.L.; Yan, Z.; Wu, Z.R.; Yu, Z.; Zhang, D.; Ni, D.J.; Chen, Y.Q. The New Insight into the Effects of Different Fixing Technology on Flavor and Bioactivities of Orange Dark Tea. Molecules 2023, 28, 1079. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.Y.; Song, H.L.; Zou, T.T.; Wan, S.Y.; Li, M.K. Characterization of odor-active compounds in moso bamboo leaf gas chromatography-ion mobility spectrometry, one- and two-dimensional gas chromatography-olfactory-mass spectrometry, and electronic nose. Food Res. Int. 2022, 152, 110916. [Google Scholar] [CrossRef]
- Wei, J.; Yin, Y.; Jiang, W.; Song, C.; Wei, L.; Liao, Z.; Han, X. Study on statistical evaluation techniques of volatile substances from seven kinds of bamboo leaves. Food Ferment. Ind. 2021, 47, 181–187. [Google Scholar] [CrossRef]
- Wang, H.L.; Wu, B.X.; Zhang, M.; Chen, L.; Liu, Y.; Li, Z.L.; Li, Z.T.; Kan, H.; Cao, C.W. Comparative analysis of drying characteristics, physicochemical properties and volatile flavor components of Dendrocalamus brandisii shoots under four drying techniques. LWT Food Sci. Technol. 2024, 205, 116509. [Google Scholar] [CrossRef]
- Qin, M.; Wang, Z.H.; Sun, D.L.; Jiang, X.W.; Yao, L.H.; Yu, M.G. Preparation of green-protected bamboo with excellent color fastness using natural sodium copper chlorophyll. J. Clean. Prod. 2022, 371, 133701. [Google Scholar] [CrossRef]
- Zhai, X.; Ban, W.; Sun, Y.; Guo, Z.; Zhang, L.; Liu, B.; Wu, K.; Chen, Z.; Zou, W. Study on the Quality Changes of Wencheng Glutinous Yam During Vacuum Freezing Storage. Agric. Prod. Process. 2025, 1–5. [Google Scholar]
- Deng, J.; Wang, B.; Shi, H. Effect of different concentrations of mulberry leaf polyphenols on the browning of fresh-cut lettuce and inhibition of Pseudomonas spp. Food Ferment. Ind. 2022, 48, 206–211. [Google Scholar] [CrossRef]
- Liu, H.; Jiang, W.B.; Cao, J.K.; Ma, L. A combination of 1-methylcyclopropene treatment and intermittent warming alleviates chilling injury and affects phenolics and antioxidant activity of peach fruit during storage. Sci. Hortic. 2018, 229, 175–181. [Google Scholar] [CrossRef]
- Wuyts, N.; De Waele, D.; Swennen, R. Extraction and partial characterization of polyphenol oxidase from banana (Grande naine) roots. Plant Physiol. Biochem. 2006, 44, 308–314. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, J.; Tang, J.; Huang, R.; Luo, W.; Tuo, Y.; Liao, N.; Zhuang, D.; Lin, J.; Zhang, Y. Study on dynamic changes in characteristic volatile compounds uncovers aroma development of Hainan Dayezhong (Camellia sinensis var. assamica) black tea. Food Chem. 2025, 477, 143578. [Google Scholar] [CrossRef]
- Geng, Q.Y.; Qin, F.; Zeng, M.M.; Wang, Z.J.; Chen, Q.M.; Zhang, W.; Zhu, B.K.; He, Z.Y.; Chen, J. Effects of heat treatment on the profile of volatile flavor components in bamboo shoots. Food Biosci. 2024, 58, 103817. [Google Scholar] [CrossRef]
- ISO 13299:2016; Sensory Analysis: Methodology: General Guidance for Establishing a Sensory Profile. International Organization for Standardization: Geneva, Switzerland, 2016.
- ISO 8586:2023; Sensory Analysis: Selection and Training of Sensory Assessors. International Organization for Standardization: Geneva, Switzerland, 2023.
- Xu, J.; Zeng, M.M.; Wang, Z.J.; Chen, Q.M.; Qin, F.; Chen, J.; He, Z.Y. Characteristic flavor compounds in soy protein isolate and their correlation with sensory attributes. Food Res. Int. 2025, 206, 116038. [Google Scholar] [CrossRef]
- Li, R.; Shang, H.; Wu, H.; Wang, M.; Duan, M.; Yang, J. Thermal inactivation kinetics and effects of drying methods on the phenolic profile and antioxidant activities of chicory (Cichorium intybus L.) leaves. Sci. Rep. 2018, 8, 9529. [Google Scholar] [CrossRef]
- Moscetti, R.; Raponi, F.; Monarca, D.; Bedini, G.; Ferri, S.; Massantini, R. Effects of hot-water and steam blanching of sliced potato on polyphenol oxidase activity. Int. J. Food Sci. Technol. 2019, 54, 403–411. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, W.; Stockmann, R.; Terefe, N.S. Effect of citric acid and high pressure thermal processing on enzyme activity and related quality attributes of pear puree. Innov. Food Sci. Emerg. Technol. 2018, 45, 196–207. [Google Scholar] [CrossRef]
- Tan, T.C.; Cheng, L.H.; Bhat, R.; Rusul, G.; Easa, A.M. Composition, physicochemical properties and thermal inactivation kinetics of polyphenol oxidase and peroxidase from coconut (Cocos nucifera) water obtained from immature, mature and overly-mature coconut. Food Chem. 2014, 142, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Feng, C.; Yao, E.; Teng, Q.; Jiang, Q.; Huang, C. Research on the Effect of Enzyme Activity during Zhaoping Tea Processing on Tea Quality. China Food Ind. 2024, 5, 110–112. [Google Scholar]
- Tanaka, A.; Ito, H. Chlorophyll Degradation and Its Physiological Function. Plant Cell Physiol. 2024, 66, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Donlao, N.; Ogawa, Y. The influence of processing conditions on catechin, caffeine and chlorophyll contents of green tea (Camelia sinensis) leaves and infusions. LWT Food Sci. Technol. 2019, 116, 108567. [Google Scholar] [CrossRef]
- Long, Y.; Lei, Y.; Ma, X.; Min, T.; Zhao, T.; Yue, J. Research Progress on the Degradation of and Preservation Techniques for Chlorophyll in Vegetables during Thermal Processing. Food Sci. 2025, 46, 285–294. [Google Scholar] [CrossRef]
- Mao, Y.L.; Wang, J.Q.; Wang, F.; Cao, Q.Q.; Yin, J.F.; Xu, Y.Q. Effect of different drying temperature settings on the color characteristics of Tencha. Food Chem. X 2024, 24, 101963. [Google Scholar] [CrossRef] [PubMed]
- Kou, Y. The Lotus Leaf Photochromic Mechanism and Preserving Green Technology. Master’s Thesis, Wuhan Polytechnic University, Wuhan, China, 2013. [Google Scholar]
- Fu, S. Research and Development of Matzhu. Master’s Thesis, Zhejiang University, Hangzhou, China, 2018. [Google Scholar]
- Qi, D.; Dai, W.; Tan, J.; Peng, Q.; Zhang, Y.; Lin, Z. Study on the Effects of the Fixation Methods on the Chemical Components and Taste Quality of Summer Green Tea. J. Tea Sci. 2016, 36, 18–26. [Google Scholar] [CrossRef]
- Pei, F.; Yang, W.J.; Ma, N.; Fang, Y.; Zhao, L.Y.; An, X.X.; Xin, Z.H.; Hu, Q.H. Effect of the two drying approaches on the volatile profiles of button mushroom (Agaricus bisporus) by headspace GC–MS and electronic nose. LWT Food Sci. Technol. 2016, 72, 343–350. [Google Scholar] [CrossRef]
- Liu, H.; Zeng, F.K.; Wang, Q.H.; Ou, S.Y.; Tan, L.H.; Gu, F.L. The effect of cryogenic grinding and hammer milling on the flavour quality of ground pepper (Piper nigrum L.). Food Chem. 2013, 141, 3402–3408. [Google Scholar] [CrossRef]
- Hu, B.B. Study on the Interactive Effect of Lipid Oxidation and the Maillard Reaction on Aroma Formationin Sesame. Master’s Thesis, Henan University of Technology, Zhengzhou, China, 2025. [Google Scholar]
- Hong, L.P.; Zhang, W.H.; Chen, F.; He, E.M.; Wu, J.W.; Wang, W.H. Transcriptomic analysis and volatile profiling reveals the regulatory mechanism of flavor volatiles in fresh-cut potatoes. Int. J. Food Prop. 2023, 26, 2795–2810. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, Z.; Rren, J.; Pan, S.; Fan, G. Study on the Effect of Pasteurization on the Bound Aroma Compounds in Orange Juice. Sci. Technol. Food Ind. 2014, 35, 49–52+56. [Google Scholar] [CrossRef]
- Tang, J.J.; Zhang, Z.X.; Zheng, S.L.; Gao, N.; Li, Z.J.; Li, K. Changes of Main Nutrient Components and Volatile Flavor Substances in Processing of Canned Bamboo Shoots. Fermentation 2021, 7, 293. [Google Scholar] [CrossRef]
- Guo, X.; Song, C.; Ho, C.T.; Wan, X. Contribution of l-theanine to the formation of 2,5-dimethylpyrazine, a key roasted peanutty flavor in Oolong tea during manufacturing processes. Food Chem. 2018, 263, 18–28. [Google Scholar] [CrossRef]
- Zheng, X.X.; Hong, X.; Jin, Y.L.; Wang, C.; Liu, Z.H.; Huang, J.A.; Li, Q. Characterization of key aroma compounds and relationship between aroma compounds and sensory attributes in different aroma types of Fu brick tea. Food Chem. X 2022, 13, 100248. [Google Scholar] [CrossRef]
- Van Gemert, L.J. Compilations of Odour Threshold Values in Air, Water and Other Media, 2nd ed.; Oliemans Punter & Partners B.V.: Zeist, The Netherlands, 2011. [Google Scholar]
- Ouyang, J.; Jiang, R.G.; Chen, H.Y.; Liu, Q.; Yi, X.Q.; Wen, S.; Huang, F.F.; Zhang, X.Y.; Li, J.; Wen, H.T.; et al. Characterization of key odorants in ‘Baimaocha’ black teas from different regions. Food Chem. X 2024, 22, 101303. [Google Scholar] [CrossRef]
- Wang, M.Q.; Ma, W.J.; Shi, J.; Zhu, Y.; Lin, Z.; Lv, H.P. Characterization of the key aroma compounds in Longjing tea using stir bar sorptive extraction (SBSE) combined with gas chromatography-mass spectrometry (GC-MS), gas chromatography-olfactometry (GC-O), odor activity value (OAV), and aroma recombination. Food Res. Int. 2020, 130, 108908. [Google Scholar] [CrossRef]
- Lewinsohn, E.; Ziv-Raz, I.; Dudai, N.; Tadmor, Y.; Lastochkin, E.; Larkov, O.; Chaimovitsh, D.; Ravid, U.; Putievsky, E.; Pichersky, E.; et al. Biosynthesis of estragole and methyl-eugenol in sweet basil (Ocimum basilicum L).: Developmental and chemotypic association of allylphenol O-methyltransferase activities. Plant Sci. 2000, 160, 27–35. [Google Scholar] [CrossRef]
- Yang, T.; Zhu, Y.; Shao, C.Y.; Zhang, Y.; Shi, J.; Lv, H.P.; Lin, Z. Enantiomeric analysis of linalool in teas using headspace solid-phase microextraction with chiral gas chromatography. Ind. Crops Prod. 2016, 83, 17–23. [Google Scholar] [CrossRef]
- Long, D.; Liu, X.; Luo, L.; Wu, Y. Aroma Enhancement of Exogenous β-Carotene in Tea Extracts. J. Food Saf. Qual. 2017, 8, 8. [Google Scholar] [CrossRef]
- Gao, M.; Cai, Y.; Wu, J.; Wang, Y.; Huang, C.; Lao, F. Research Progress on Preparation Technology and Flavor Characteristics of Cold Brew Coffee. Sci. Technol. Food Ind. 2023, 44, 14. [Google Scholar] [CrossRef]
- Zhu, J.C.; Chen, F.; Wang, L.Y.; Niu, Y.W.; Chen, H.X.; Wang, H.L.; Xiao, Z.B. Characterization of the Key Aroma Volatile Compounds in Cranberry (Vaccinium macrocarpon Ait.) Using Gas Chromatography-Olfactometry (GC-O) and Odor Activity Value (OAV). J. Agric. Food. Chem. 2016, 64, 4990–4999. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.Y.; Yan, H.; Zhu, Y.; Liu, X.; Lv, H.P.; Zhang, Y.; Dai, W.D.; Guo, L.; Tan, J.F.; Peng, Q.H.; et al. Identification and quantification of key odorants in the world’s four most famous black teas. Food Res. Int. 2019, 121, 73–83. [Google Scholar] [CrossRef]

| Samples | Chlorophyll Content mg/g | PPO Relative Activity | POD Relative Activity | L* | a* | b* | ΔE |
|---|---|---|---|---|---|---|---|
| FBL | 1.90 ± 0.03 A | 100% A | 100% A | 49.90 ± 0.67 BC | −2.04 ± 0.25 G | 9.31 ± 0.26 E | 0.00 ± 0.00 H |
| UBL | 0.90 ± 0.04 D | 97.38% ± 7.44% ABC | 97.30% ± 1.20% B | 50.29 ± 0.67 AB | −1.06 ± 0.13 CD | 9.65 ± 0.15 D | 1.22 ± 0.24 EF |
| SBL/30 s | 0.81 ± 0.04 DEb | 89.82% ± 8.13% BCa | 46.3% ± 0.82% Ea | 49.96 ± 0.41 BCb | −1.05 ± 0.07 CDb | 9.66 ± 0.24 Dc | 1.12 ± 0.04 Fd |
| SBL/60 s | 1.06 ± 0.11 Ca | 85.09% ± 6.13% CDa | 20.48% ± 0.15% Fb | 48.40 ± 0.23 EFc | −1.17 ± 0.05 Dc | 10.34 ± 0.44 Bb | 2.06 ± 0.08 Dc |
| SBL/120 s | 0.82 ± 0.05 DEb | 86.79% ± 0.9% BCDa | 5.19% ± 0.12% Hc | 50.68 ± 0.24 Aa | −1.37 ± 0.09 Eb | 11.77 ± 0.19 Aa | 2.67 ± 0.18 Cb |
| SBL/180 s | 0.73 ± 0.03 Eb | 85.70% ± 10.43% BCDa | 3.54% ± 0.64% Id | 47.07 ± 0.24 Gd | 0.54 ± 0.09 Aa | 10.00 ± 0.21 Cbc | 3.90 ± 0.11 Aa |
| BBL/30 s | 0.81 ± 0.02 DEb | 94.82% ± 2.73% ABa | 83.08% ± 1.03% Ca | 48.84 ± 0.12 DEa | −0.99 ± 0.15 Cb | 9.17 ± 0.19 EFb | 1.51 ± 0.15 Eb |
| BBL/60 s | 0.93 ± 0.08 CDa | 73.66% ± 4.13% Eb | 61.29% ± 0.76% Db | 48.91 ± 0.20 DEa | −1.44 ± 0.05 Ec | 9.28 ± 0.09 Eb | 1.16 ± 0.15 Fb |
| BBL/120 s | 0.50 ± 0.03 Fc | 51.91% ± 3.18% Gc | 0.79% ± 0.33% Jc | 47.15 ± 0.17 Gc | −1.05 ± 0.09 CDb | 10.36 ± 0.10 Ba | 3.11 ± 0.12 Ba |
| BBL/180 s | 0.59 ± 0.04 Fc | 31.48% ± 2.41% Hd | 0.45% ± 0.13% Jc | 47.14 ± 0.64 Gc | −0.44 ± 0.06 Ba | 10.51 ± 0.14 Ba | 3.43 ± 0.42 Ba |
| BCBL/15 s | 0.88 ± 0.07 Db | 77.65% ± 3.98% DEa | 13.75% ± 0.00% Ga | 49.47 ± 0.48 CDa | −1.06 ± 0.09 CDb | 9.06 ± 0.11 EFbc | 1.17 ± 0.13 Fb |
| BCBL/30 s | 1.55 ± 0.15 Ba | 63.82% ± 2.08% Fb | 0.36% ± 0.04% Jb | 49.60 ± 0.16 Ca | −1.62 ± 0.07 Fc | 9.68 ± 0.09 Da | 0.66 ± 0.02 Gc |
| BCBL/60 s | 0.93 ± 0.09 CDb | 50.20% ± 5.25% Gc | 0.26% ± 0.04% Jc | 49.87 ± 0.72 BCa | −1.01 ± 0.17 CDb | 9.32 ± 0.24 Eb | 1.20 ± 0.20 EFb |
| BCBL/120 s | 0.82 ± 0.06 DEb | 17.02% ± 6.37% Id | 0.21% ± 0.08% Jc | 48.07 ± 0.26 Fb | −0.44 ± 0.06 Ba | 8.95 ± 0.12 Fc | 2.47 ± 0.16 Ca |
| NO. | CAS | Compounds | Class | Odor Description 1 | Threshold 2 (μg/kg) | ROAV 3 | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| FBL | UBL | SBL | BBL | BCBL | ||||||
| 1 | 123-51-3 | 1-Butanol, 3-methyl- | Alcohol | fusel, oil, alcoholic, whiskey, fruity, banana | 4 | 271.87 | 1.04 | 10.05 | 24.86 | 0.18 |
| 2 | 71-41-0 | 1-Pentanol | Alcohol | fusel, oil, sweet, balsam | 120 | 4.34 | ND | ND | 0.31 | ND |
| 3 | 1576-95-0 | 2-Penten-1-ol, (Z)- | Alcohol | green, phenolic, nasturtium, ethereal, medicinal, aldehydic, cherry, narcissus, metallic, fruity | 720 | 1.25 | 0.05 | 0.21 | 0.48 | ND |
| 4 | 111-27-3 | 1-Hexanol | Alcohol | ethereal, fusel, oil, fruity, alcoholic, sweet, green | 5.6 | 374.18 | 3.65 | 14.47 | 33.86 | ND |
| 5 | 928-96-1 | 3-Hexen-1-ol, (Z)- | Alcohol | fresh, green, cut, grass, foliage, vegetable, herbal, oily | 3.9 | 45.67 | ND | 43.87 | 10.47 | ND |
| 6 | 928-97-2 | 3-Hexen-1-ol, (E)- | Alcohol | green, cortex, privet, leafy, floral, petal, oily, earthy | 110 | 9.06 | ND | ND | 2.83 | ND |
| 7 | 589-98-0 | 3-Octanol | Alcohol | earthy, mushroom, herbal, melon, citrus, woody, spicy, minty | 18 | 14.96 | ND | 1.96 | 4.07 | ND |
| 8 | 123-96-6 | 2-Octanol | Alcohol | fresh, spicy, green, woody, herbal, earthy | 7.8 | 11.39 | ND | ND | ND | ND |
| 9 | 3391-86-4 | 1-Octen-3-ol | Alcohol | mushroom, earthy, green, oily, fungal, raw, chicken | 1.5 | ND | ND | ND | 10.80 | ND |
| 10 | 111-70-6 | 1-Heptanol | Alcohol | musty, leafy, violet, herbal, green, sweet, woody, peony | 5.4 | 38.47 | ND | ND | ND | ND |
| 11 | 34995-77-2 | trans-Linalool oxide (furanoid) | Alcohol | floral | 60 | 7.07 | ND | ND | ND | ND |
| 12 | 24347-58-8 | 2,3-Butanediol, [R- (R*, R*)]- | Alcohol | buttery, sweet, fatty | 95.1 | 2.19 | 0.43 | 5.99 | 4.93 | 0.10 |
| 13 | 78-70-6 | Linalool | Alcohol | citrus, floral, sweet, bois, de, rose, woody, green, blueberry | 1 | 374.12 | 75.76 | 358.39 | 613.61 | 40.41 |
| 14 | 111-87-5 | 1-Octanol | Alcohol | waxy, green, orange, aldehydic rose, mushroom | 77.5 | 3.89 | 0.18 | 0.40 | ND | 0.11 |
| 15 | 64275-73-6 | 5-Octen-1-ol, (Z)- | Alcohol | green, melon, watery, watermelon, earthy, mushroom, violet, leaf, fishy, soapy | 6 | 9.08 | ND | ND | ND | ND |
| 16 | 18409-17-1 | 2-Octen-1-ol, (E)- | Alcohol | green, citrus, vegetable, fatty | 20 | ND | ND | ND | 1.03 | ND |
| 17 | 143-8-8 | 1-Nonanol | Alcohol | fresh, clean, fatty, floral, rose, orange, dusty, wet, oily | 42 | 1.38 | ND | ND | ND | ND |
| 18 | 98-55-5 | α-Terpineol | Alcohol | pine, terpene, lilac, citrus, woody, floral | 4.6 | 103.23 | 17.29 | 51.42 | ND | 3.90 |
| 19 | 35854-86-5 | (6Z)-Nonen-1-ol | Alcohol | fresh, green, melon, waxy, honeydew, cantaloupe, cucumber, clean | 1 | 13.25 | ND | ND | ND | ND |
| 20 | 100-51-6 | Benzyl alcohol | Alcohol | floral, rose, phenolic, balsamic | 100 | 7.64 | 1.07 | 2.17 | 3.93 | 0.07 |
| 21 | 60-12-8 | Phenylethyl Alcohol | Alcohol | floral, rose, dried, rose, flower, rose, water | 60 | 7.84 | 1.27 | 2.22 | 4.21 | 0.11 |
| 22 | 590-86-3 | Butanal, 3-methyl- | Aldehyde | ethereal, aldehydic, chocolate, peach, fatty | 5 | ND | 1.10 | ND | ND | 0.22 |
| 23 | 66-25-1 | Hexanal | Aldehyde | fresh, green, fatty, aldehydic, grass, leafy, fruity, sweaty | 4.5 | 35.96 | 24.86 | 6.09 | 20.30 | 12.00 |
| 24 | 505-57-7 | 2-Hexenal | Aldehyde | sweet, almond, fruity, green, leafy, apple, plum, vegetable | 30 | ND | 5.14 | ND | ND | ND |
| 25 | 6728-26-3 | 2-Hexenal, (E)- | Aldehyde | green, banana, aldehydic, fatty, cheesy | 10 | ND | 16.00 | ND | 11.96 | ND |
| 26 | 6728-31-0 | 4-Heptenal, (Z)- | Aldehyde | oily, fatty, green, dairy, milky, creamy | 0.2 | ND | 24.53 | ND | ND | 6.28 |
| 27 | 124-13-0 | Octanal | Aldehyde | aldehydic, waxy, citrus, orange, peel, green, herbal, fresh, fatty | 0.8 | 31.12 | 16.55 | 18.07 | 24.60 | 15.00 |
| 28 | 124-19-6 | Nonanal | Aldehyde | waxy, aldehydic, rose, fresh, orris, orange, peel, fatty, peely | 1.1 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| 29 | 2548-87-0 | 2-Octenal, (E)- | Aldehyde | fresh, cucumber, fatty, green, herbal, banana, waxy, green, leaf | 0.34 | 115.78 | 39.56 | 23.94 | 53.48 | 18.07 |
| 30 | 4313-3-5 | 2,4-Heptadienal, (E, E)- | Aldehyde | fatty, green, oily, aldehydic, vegetable, cake, cinnamon | 15.4 | ND | 1.21 | 0.88 | 2.11 | 0.67 |
| 31 | 112-31-2 | Decanal | Aldehyde | sweet, aldehydic, waxy, orange, peel, citrus, floral | 3 | ND | 8.94 | ND | ND | 6.50 |
| 32 | 100-52-7 | Benzaldehyde | Aldehyde | strong, sharp, sweet, bitter, almond, cherry | 24 | 24.35 | 2.68 | 3.34 | 10.47 | 0.58 |
| 33 | 18829-56-6 | 2-Nonenal, (E)- | Aldehyde | fatty, green, cucumber, aldehydic, citrus | 0.19 | ND | 156.66 | 125.27 | 76.50 | 76.25 |
| 34 | 557-48-2 | 2,6-Nonadienal, (E, Z)- | Aldehyde | green, fatty, dry, cucumber, violet, leaf | 0.02 | ND | 860.91 | ND | 243.08 | 235.54 |
| 35 | 432-25-7 | 1-Cyclohexene-1-carboxaldehyde, 2,6,6-trimethyl- | Aldehyde | tropical, saffron, herbal, clean, rose, oxide, sweet, tobacco, damascone, fruity | 3 | 9.48 | 4.11 | 10.38 | 13.61 | 2.18 |
| 36 | 122-78-1 | Benzeneacetaldehyde | Aldehyde | green, sweet, floral, hyacinth, clover, honey, cocoa | 2 | 15.75 | 9.58 | 48.67 | 32.43 | 3.97 |
| 37 | 3913-81-3 | 2-Decenal, (E)- | Aldehyde | waxy, fatty, earthy, green, cilantro, mushroom, aldehydic, fried, chicken, fat, tallow | 0.4 | ND | 35.06 | ND | ND | 18.21 |
| 38 | 5910-87-2 | 2,4-Nonadienal, (E, E)- | Aldehyde | fatty, melon, waxy, green, violet, leaf, cucumber, tropical, fruit, chicken, fat | 0.1 | ND | 28.06 | ND | ND | 12.24 |
| 39 | 25152-84-5 | 2,4-Decadienal, (E, E)- | Aldehyde | oily, cucumber, melon, citrus, pumpkin, nut, meat | 0.2 | ND | ND | ND | ND | 14.11 |
| 40 | 123-66-0 | Hexanoic acid, ethyl ester | Ester | sweet, fruity, pineapple, waxy, green, banana, | 1 | 61.90 | ND | ND | ND | ND |
| 41 | 142-92-7 | Acetic acid, hexyl ester | Ester | fruity, green, apple, banana, sweet | 11 | 11.82 | ND | ND | ND | ND |
| 42 | 1629-58-9 | 1-Penten-3-one | Ketone | pungent, peppery, mustard, garlic, onion | 0.9 | ND | 10.58 | ND | ND | ND |
| 43 | 513-86-0 | Acetoin | Ketone | sweet, buttery, creamy, dairy, milky, fatty | 14 | ND | ND | ND | 1.09 | ND |
| 44 | 693-54-9 | 2-Decanone | Ketone | orange, floral, fatty, peach | 3 | 5.95 | ND | ND | ND | ND |
| 45 | 111-13-7 | 2-Octanone | Ketone | earthy, weedy, natural, woody, herbal | 5 | ND | ND | 1.33 | ND | ND |
| 46 | 98-86-2 | Acetophenone | Ketone | sweet, pungent, hawthorn, mimosa, almond, acacia, chemical | 36 | 2.13 | ND | ND | ND | ND |
| 47 | 17283-81-7 | 2-Butanone, 4-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | Ketone | earthy, woody, mahogany, orris, dry, amber | 1 | 72.03 | ND | ND | ND | ND |
| 48 | 127-41-3 | α-Ionone | Ketone | sweet, woody, floral, violet, orris, tropical, fruity | 0.4 | 504.99 | 144.31 | ND | 680.09 | 44.45 |
| 49 | 14901-7-6 | β-Ionone | Ketone | floral, woody, sweet, fruity, berry, tropical, beeswax | 0.007 | 13,612.49 | ND | ND | ND | ND |
| 50 | 142-62-1 | Hexanoic acid | Acid | sour, fatty, sweat, cheese | 36 | ND | 0.75 | 1.72 | 1.15 | 0.13 |
| 51 | 3338-55-4 | 1,3,6-Octatriene, 3,7-dimethyl-, (Z)- | Triterpenoid | warm, floral, herb, flower, sweet | 34 | ND | ND | 2.56 | 3.28 | 0.38 |
| 52 | 3856-25-5 | Copaene | Triterpenoid | woody, spicy, honey | 6 | 10.93 | ND | ND | ND | ND |
| 53 | 13360-65-1 | Pyrazine, 3-ethyl-2,5-dimethyl- | Pyrazine | potato, cocoa, roasted, nutty | 0.4 | ND | ND | 106.90 | ND | ND |
| 54 | 13067-27-1 | Pyrazine, 2,6-diethyl- | Pyrazine | nutty, hazelnut | 6 | ND | ND | 7.21 | ND | ND |
| 55 | 90-05-1 | Phenol, 2-methoxy- | Phenol | phenolic, smoke, spice, vanilla, woody | 1.6 | ND | ND | 80.87 | ND | ND |
| 56 | 108-95-2 | Phenol | Phenol | phenolic, plastic, rubber | 31 | 7.88 | 1.86 | 4.19 | 10.98 | ND |
| 57 | 2785-89-9 | Phenol, 4-ethyl-2-methoxy- | Phenol | spicy, smoky, bacon, phenolic, clove | 16 | ND | 0.29 | 2.56 | 4.61 | 0.11 |
| 58 | 123-7-9 | Phenol, 4-ethyl- | Phenol | phenolic, castoreum, smoke, guaiacol | 13 | 12.00 | 2.23 | 4.40 | 12.31 | 0.42 |
| 59 | 7786-61-0 | 2-Methoxy-4-vinylphenol | Phenol | sweet, spicy, clove, carnation, phenolic, peppery, smoky, woody, powdery | 3 | ND | ND | 11.08 | 2.97 | ND |
| 60 | 3777-69-3 | Furan, 2-pentyl- | Furan | fruity, green, earthy, beany, vegetable, metallic | 5.8 | 6.24 | 1.48 | 7.07 | ND | 0.93 |
| NO. | CAS | Compounds | Class | Odor Description 1 | Aroma Intensity 2 | ||||
|---|---|---|---|---|---|---|---|---|---|
| FBL | UBL | SBL | BBL | BCBL | |||||
| 1 | 123-51-3 | 1-Butanol, 3-methyl- | Alcohol | oil, alcoholic, fruity | 1.67 | ND | ND | ND | ND |
| 2 | 71-41-0 | 1-Pentanol | Alcohol | oil, sweet, balsam | 2.67 | ND | ND | ND | ND |
| 3 | 1576-95-0 | 2-Penten-1-ol, (Z)- | Alcohol | green, medicinal, mushroom | 1.83 | ND | ND | ND | ND |
| 4 | 111-27-3 | 1-Hexanol | Alcohol | fruity, sweet, rice | 1.33 | 2.33 | 3.67 | 2.33 | 2.50 |
| 5 | 928-96-1 | 3-Hexen-1-ol, (Z)- | Alcohol | fresh, green, grass, foliage | 2.00 | 3.33 | 2.00 | 3.33 | 3.00 |
| 6 | 589-98-0 | 3-Octanol | Alcohol | herbal, spicy, minty | 1.50 | 3.00 | 2.00 | 3.00 | 3.00 |
| 7 | 111-70-6 | 1-Heptanol | Alcohol | green, sweet, woody, potato | 2.50 | ND | ND | ND | ND |
| 8 | 3391-86-4 | 1-Octen-3-ol | Alcohol | mushroom, earthy, herbal, woody | ND | 3.00 | 3.00 | 2.00 | 3.50 |
| 9 | 78-70-6 | Linalool | Alcohol | floral, sweet, fresh | 3.00 | 3.33 | 3.33 | 3.33 | ND |
| 10 | 24347-58-8 | 2,3-Butanediol, [R- (R*, R*)]- | Alcohol | green, cucumber | ND | 3.00 | 3.33 | 2.50 | ND |
| 11 | 98-55-5 | α-Terpineol | Alcohol | woody, floral | 2.00 | ND | ND | 3.33 | 3.00 |
| 12 | 100-51-6 | Benzyl alcohol | Alcohol | floral, rose | 1.67 | 2.67 | 3.00 | 3.33 | ND |
| 13 | 60-12-8 | Phenylethyl Alcohol | Alcohol | floral, rose | 2.00 | 2.33 | 3.00 | 1.50 | ND |
| 14 | 590-86-3 | Butanal, 3-methyl- | Aldehyde | woody, grass, earthy | ND | 3.00 | 2.00 | 2.00 | 2.50 |
| 15 | 66-25-1 | Hexanal | Aldehyde | grass | 2.67 | 3.00 | 2.50 | 2.00 | 2.67 |
| 16 | 6728-26-3 | 2-Hexenal, (E)- | Aldehyde | grass, fresh, green | 1.67 | 3.00 | 3.00 | 2.67 | 2.00 |
| 17 | 111-71-7 | Heptanal | Aldehyde | minty, fresh, green | ND | ND | ND | ND | 3.50 |
| 18 | 124-13-0 | Octanal | Aldehyde | green, fresh, lemon | ND | 2.67 | 3.67 | 2.33 | 3.00 |
| 19 | 2548-87-0 | 2-Octenal, (E)- | Aldehyde | fresh, cucumber, green | 2.50 | 3.00 | 2.50 | 3.00 | 3.00 |
| 20 | 124-19-6 | Nonanal | Aldehyde | fresh, green, minty | ND | 3.00 | 2.00 | 2.67 | 3.33 |
| 21 | 100-52-7 | Benzaldehyde | Aldehyde | fresh, sweet, minty | 1.67 | 3.00 | 3.00 | 3.00 | ND |
| 22 | 4313-3-5 | 2,4-Heptadienal, (E, E)- | Aldehyde | green, cucumber, lemon, potato | ND | 3.00 | 3.33 | 3.33 | 3.00 |
| 23 | 112-31-2 | Decanal | Aldehyde | sweet, floral | ND | ND | ND | ND | 2.50 |
| 24 | 18829-56-6 | 2-Nonenal, (E)- | Aldehyde | green, cucumber | ND | ND | ND | ND | 4.00 |
| 25 | 432-25-7 | 1-Cyclohexene-1-carboxaldehyde, 2,6,6-trimethyl- | Aldehyde | herbal, clean, fruity | 3.00 | ND | ND | 2.67 | ND |
| 26 | 122-78-1 | Benzeneacetaldehyde | Aldehyde | sweet, floral | 2.33 | 3.33 | ND | 2.00 | 2.00 |
| 27 | 6750-03-4 | 2,4-Nonadienal | Aldehyde | green, cucumber | ND | ND | ND | ND | 2.00 |
| 28 | 4411-89-6 | Benzeneacetaldehyde, α-ethylidene- | Aldehyde | sweet | ND | ND | 3.00 | ND | ND |
| 29 | 110-93-0 | 5-Hepten-2-one, 6-methyl- | Ketone | fruity, sweet, rice | 2.67 | 2.67 | 2.33 | 2.00 | 3.00 |
| 30 | 4312-99-6 | 1-Octen-3-one | Ketone | herbal, mushroom, earthy | ND | ND | ND | ND | 2.50 |
| 31 | 98-86-2 | Acetophenone | Ketone | pungent | 2.33 | ND | ND | ND | ND |
| 32 | 127-41-3 | α-Ionone | Ketone | sweet, floral, violet | 1.67 | 3.33 | 2.67 | 3.00 | 2.67 |
| 33 | 79-77-6 | β-Ionone | Ketone | floral, violet | ND | 3.67 | 3.50 | 2.50 | 3.50 |
| 34 | 123-66-0 | Hexanoic acid, ethyl ester | Ester | sweet, fruity | 2.67 | ND | ND | ND | ND |
| 35 | 3856-25-5 | Copaene | Triterpenoid | honey, potato | 1.50 | ND | ND | ND | ND |
| 36 | 140-67-0 | Estragole | Phenol | spice, foul | 3.33 | ND | ND | ND | ND |
| 37 | 90-05-1 | Phenol, 2-methoxy- | Phenol | spice, woody | ND | ND | 3.33 | ND | ND |
| 38 | 108-95-2 | Phenol | Phenol | foul | 3.00 | ND | 2.33 | 2.33 | ND |
| 39 | 2785-89-9 | Phenol, 4-ethyl-2-methoxy- | Phenol | spicy, clove | ND | ND | 2.33 | 3.00 | ND |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Wang, Z.; Chen, Q.; Zeng, M.; Chen, J.; Adhikari, B.; Guo, F.; He, Z. Processing-Induced Variations in Bamboo Leaf Powder: Effects of Fixation Methods on Color Stability, Volatile Compounds, and Sensory Profiles. Foods 2025, 14, 3898. https://doi.org/10.3390/foods14223898
Wang Q, Wang Z, Chen Q, Zeng M, Chen J, Adhikari B, Guo F, He Z. Processing-Induced Variations in Bamboo Leaf Powder: Effects of Fixation Methods on Color Stability, Volatile Compounds, and Sensory Profiles. Foods. 2025; 14(22):3898. https://doi.org/10.3390/foods14223898
Chicago/Turabian StyleWang, Qi, Zhaojun Wang, Qiuming Chen, Maomao Zeng, Jie Chen, Benu Adhikari, Fengxian Guo, and Zhiyong He. 2025. "Processing-Induced Variations in Bamboo Leaf Powder: Effects of Fixation Methods on Color Stability, Volatile Compounds, and Sensory Profiles" Foods 14, no. 22: 3898. https://doi.org/10.3390/foods14223898
APA StyleWang, Q., Wang, Z., Chen, Q., Zeng, M., Chen, J., Adhikari, B., Guo, F., & He, Z. (2025). Processing-Induced Variations in Bamboo Leaf Powder: Effects of Fixation Methods on Color Stability, Volatile Compounds, and Sensory Profiles. Foods, 14(22), 3898. https://doi.org/10.3390/foods14223898








