The Distinction of Chemical Profiles of Mountainous Forest Cultivated Ginseng and Garden Ginseng Based on Calcium Oxalate Crystals, Organic Acids, and Ginsenosides
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Methods
2.2.1. Standard Solutions and Sample Solutions Preparation for HPLC Determination of Non-Free Oxalic Acid
2.2.2. Sample Solutions Preparation for Microscopic Identification
2.2.3. Standard Solutions and Sample Solutions Preparation for GC-MS/MS
2.2.4. Standard Solutions and Sample Solutions Preparation for HPLC Determination of Ginsenosides
2.2.5. HPLC Conditions
2.2.6. Chromatographic and Mass Spectrometry Conditions of GC-MS/MS
2.2.7. Data Analysis
3. Results
3.1. Analysis of Non-Free Oxalic Acid
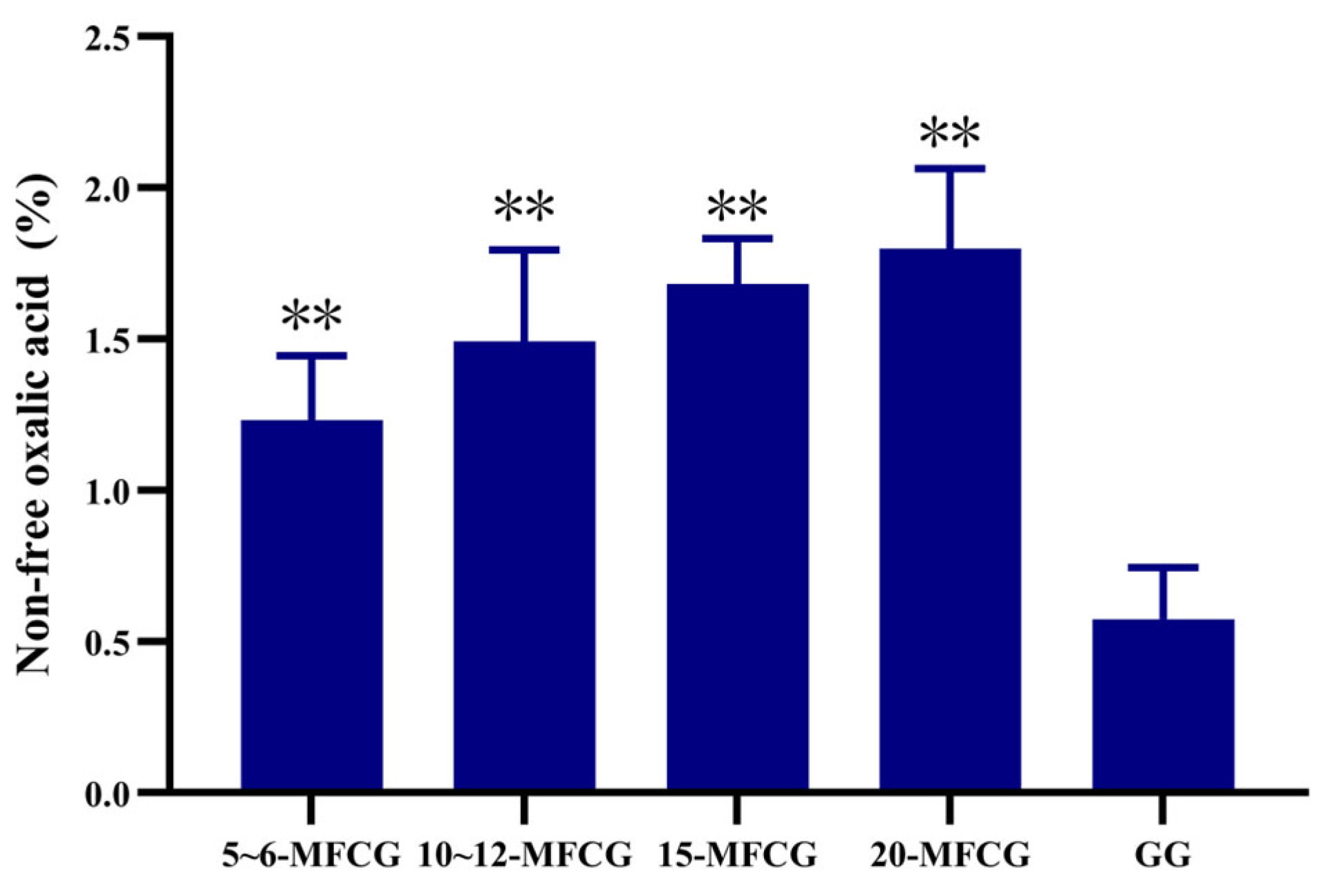
3.1.1. Determination of Non-Free Oxalic Acid in Whole GG and MFCG with Different Growth Years
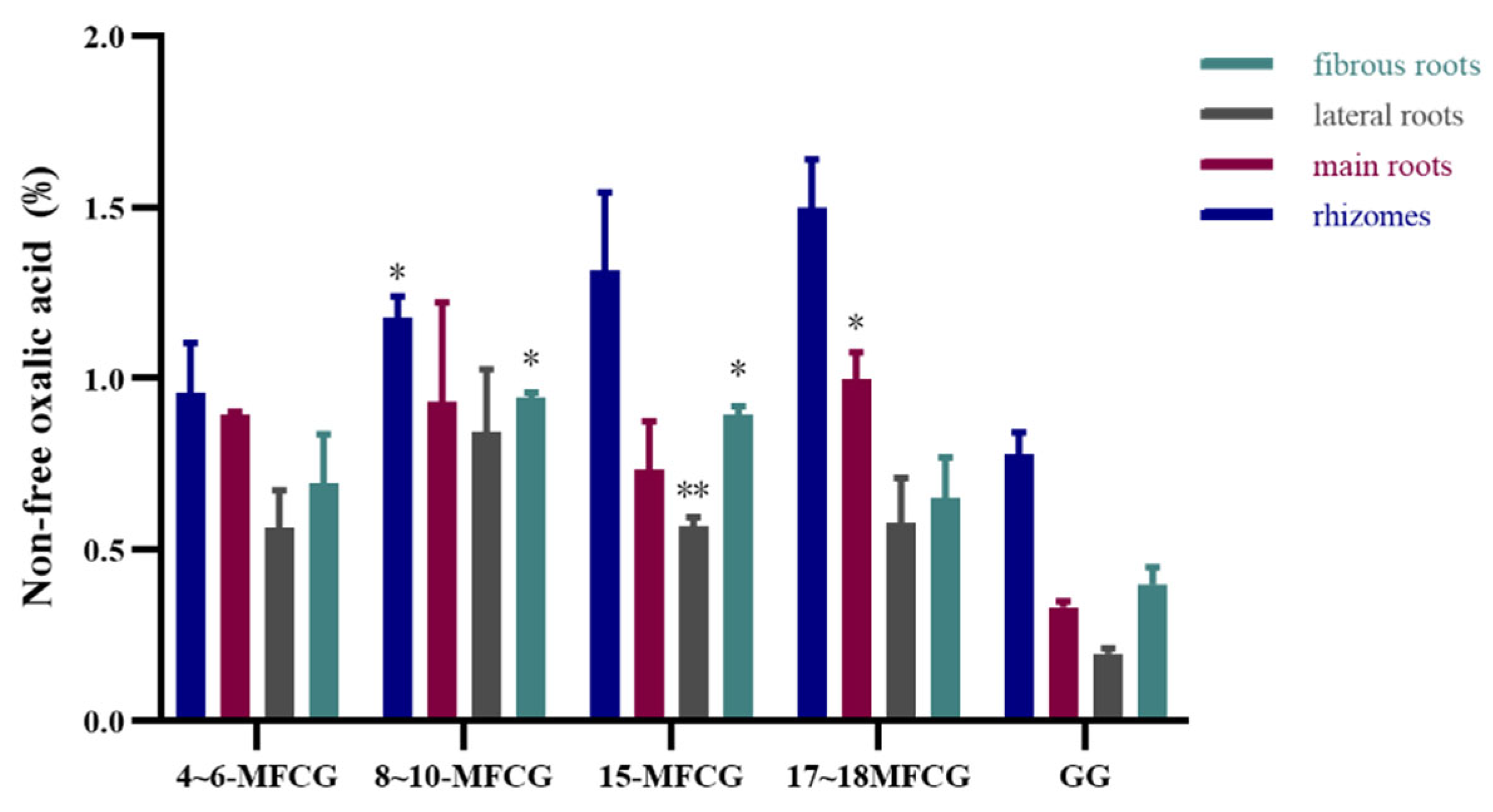
3.1.2. Determination of Non-Free Oxalic Acid in Different Parts of GG and MFCG with Different Growth Years
3.2. Analysis of Calcium Oxalate Crystals
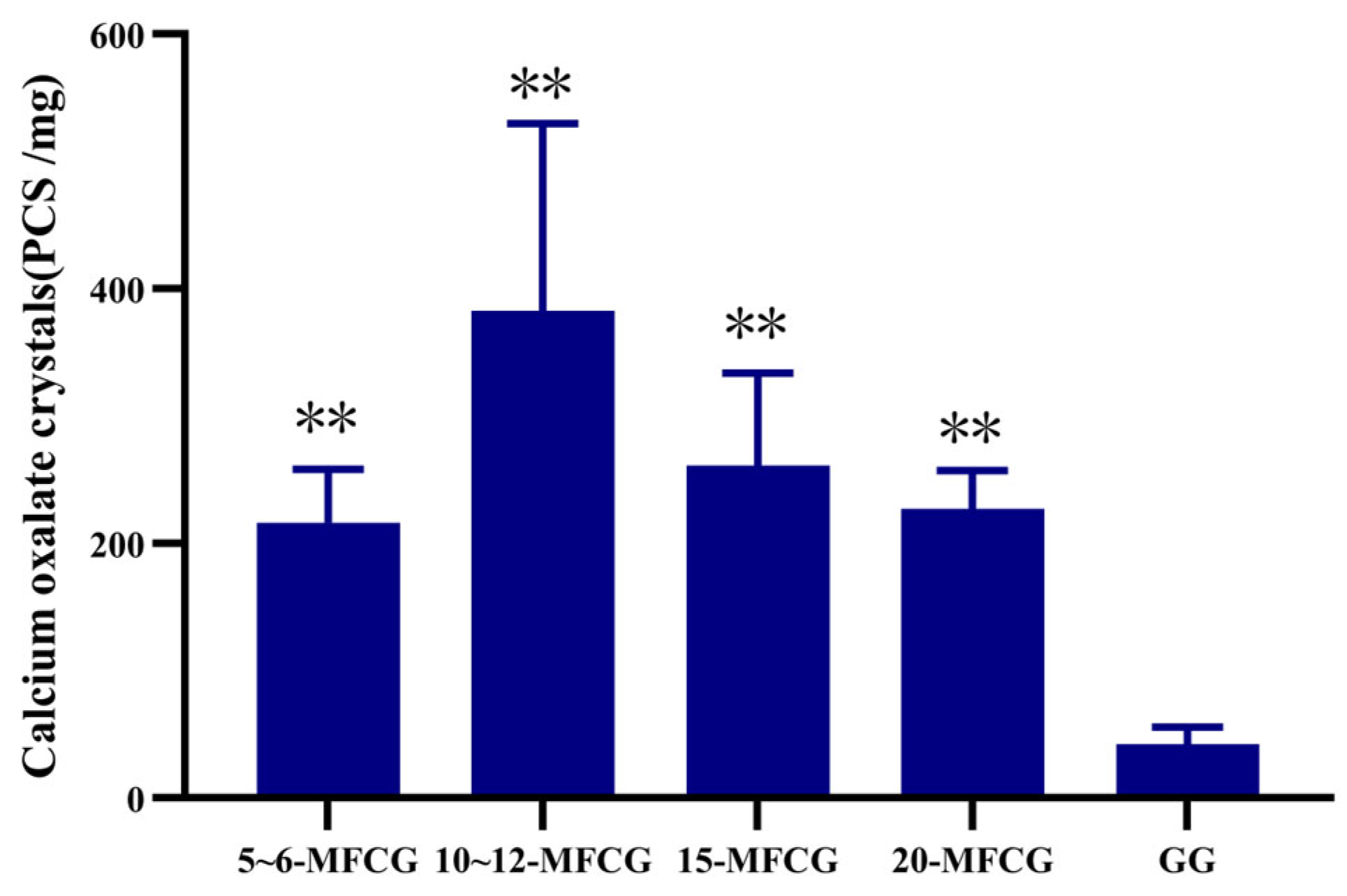
3.2.1. Determination of Calcium Oxalate Crystals in Whole GG and MFCG with Different Growth Years
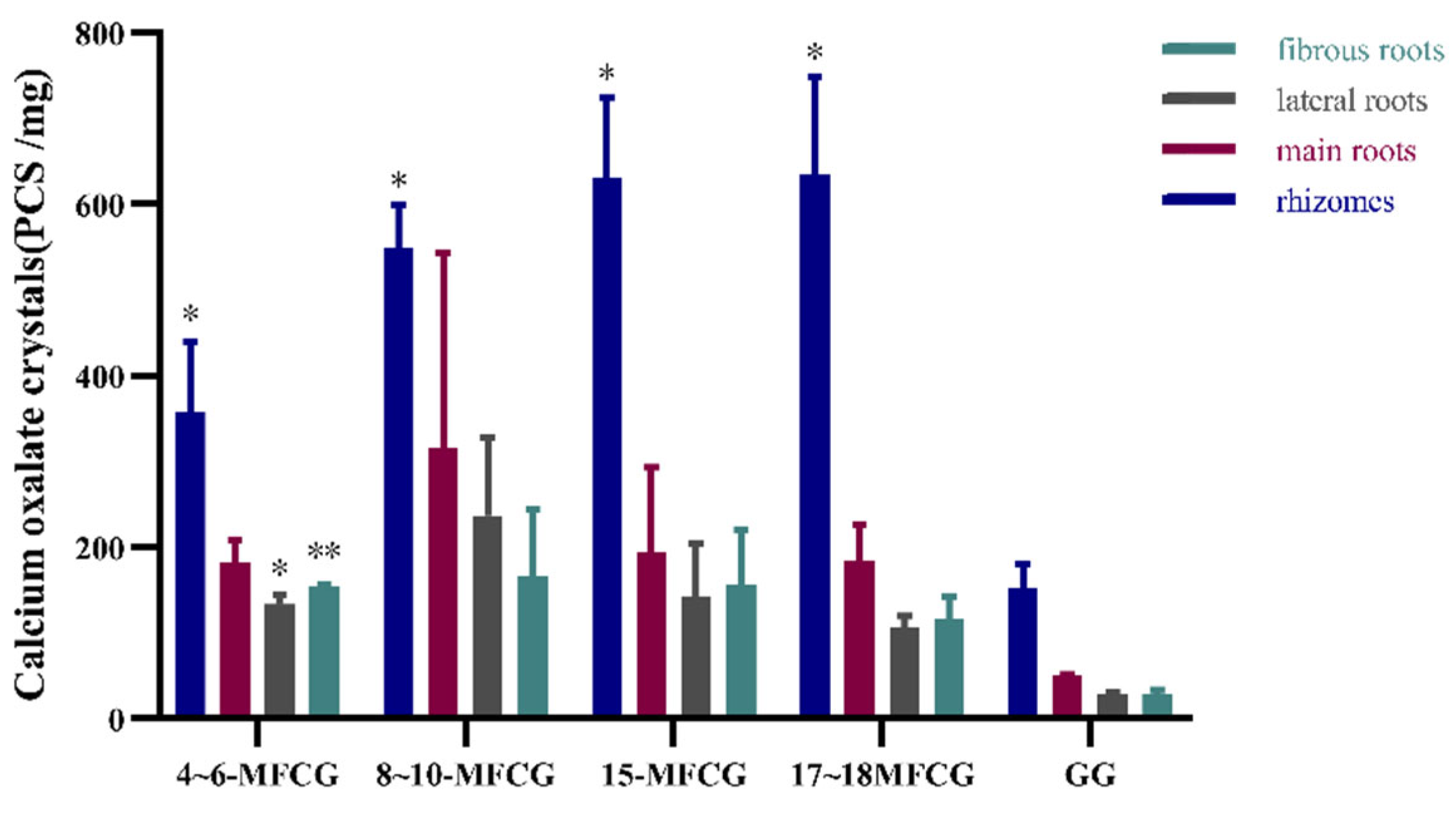
3.2.2. Determination of Calcium Oxalate Crystals in Different Parts of GG and MFCG with Different Growth Years
3.3. Analysis of Organic Acids
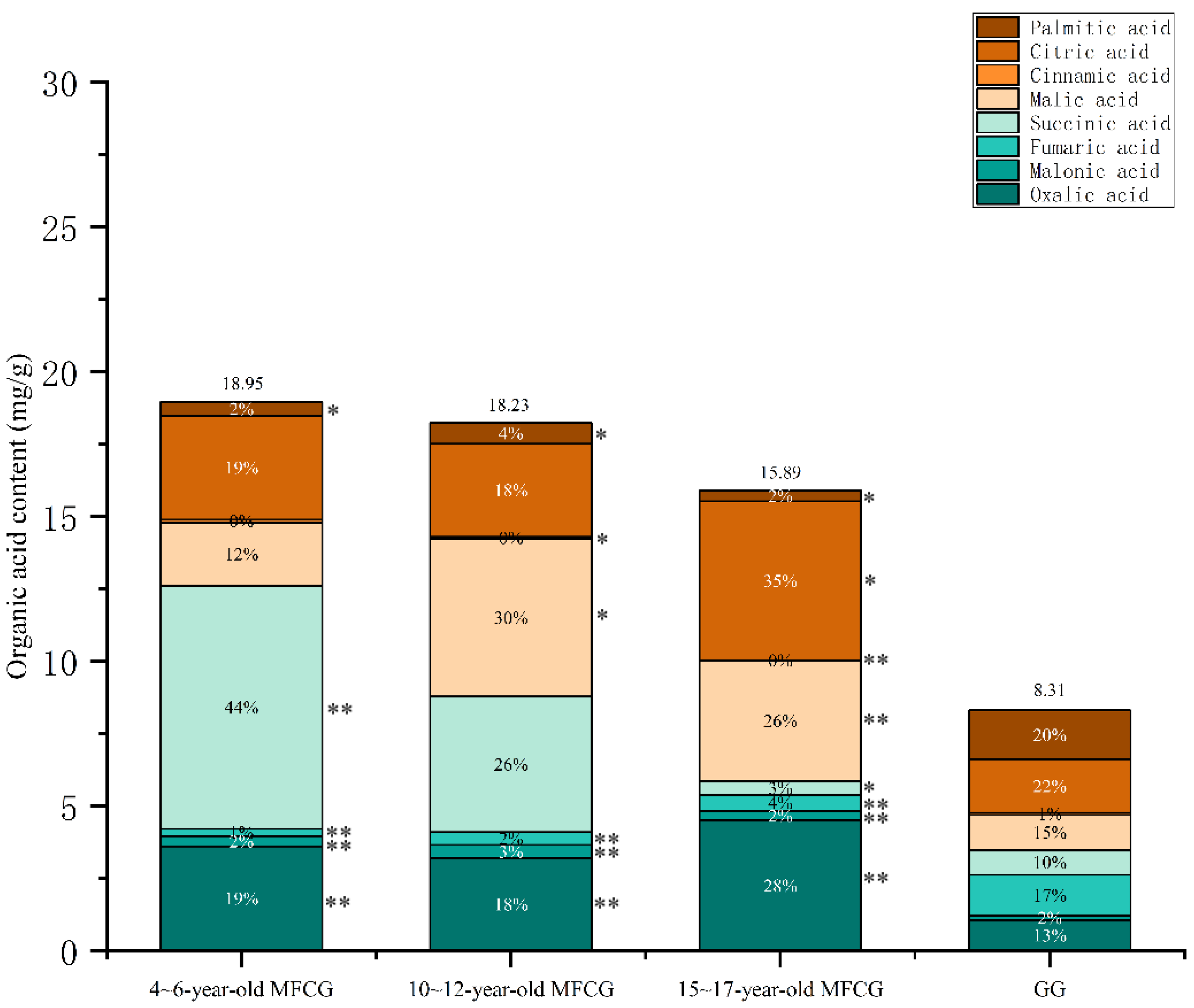
3.3.1. Determination of Organic Acids in Whole GG and MFCG with Different Growth Years
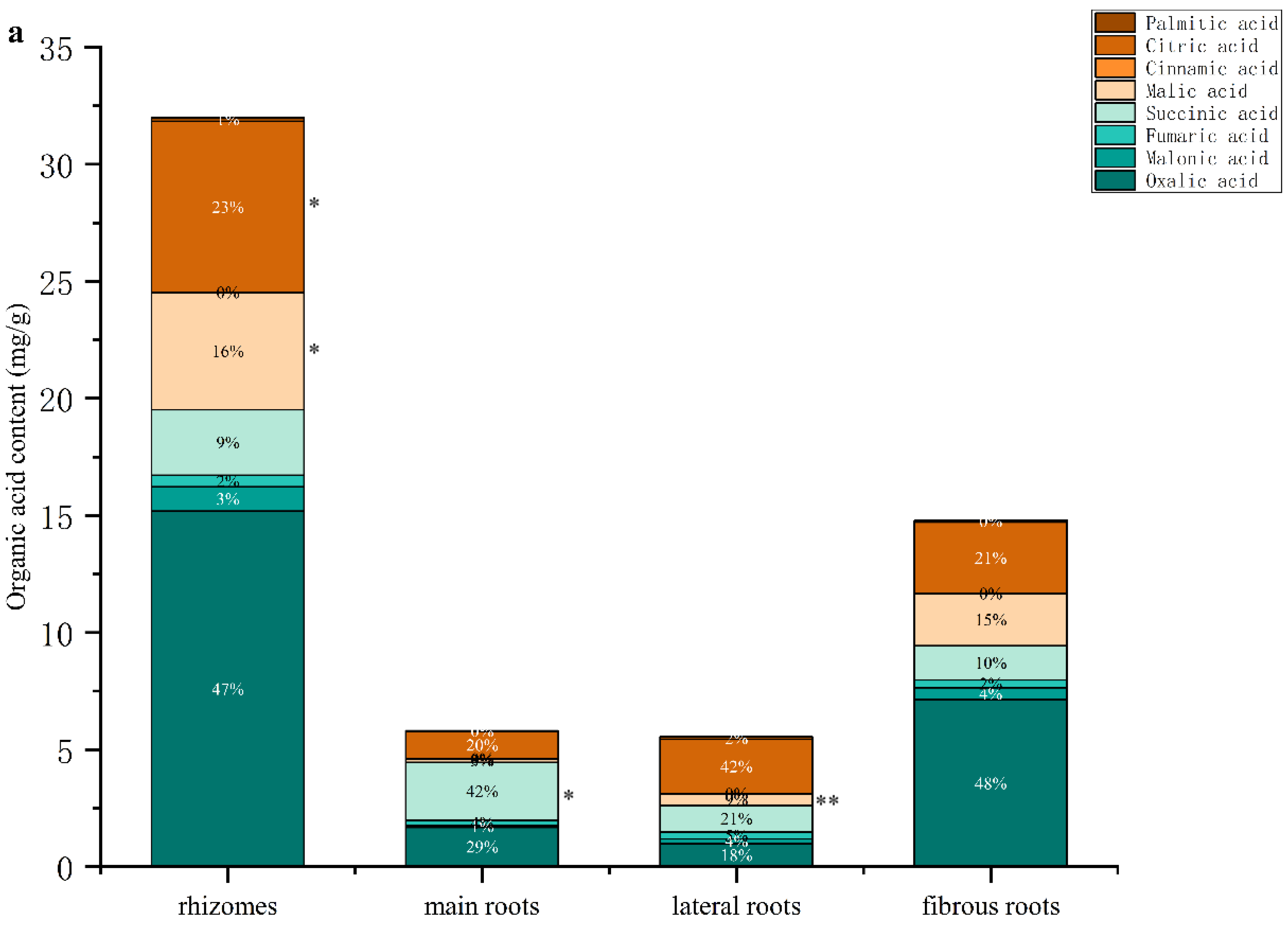
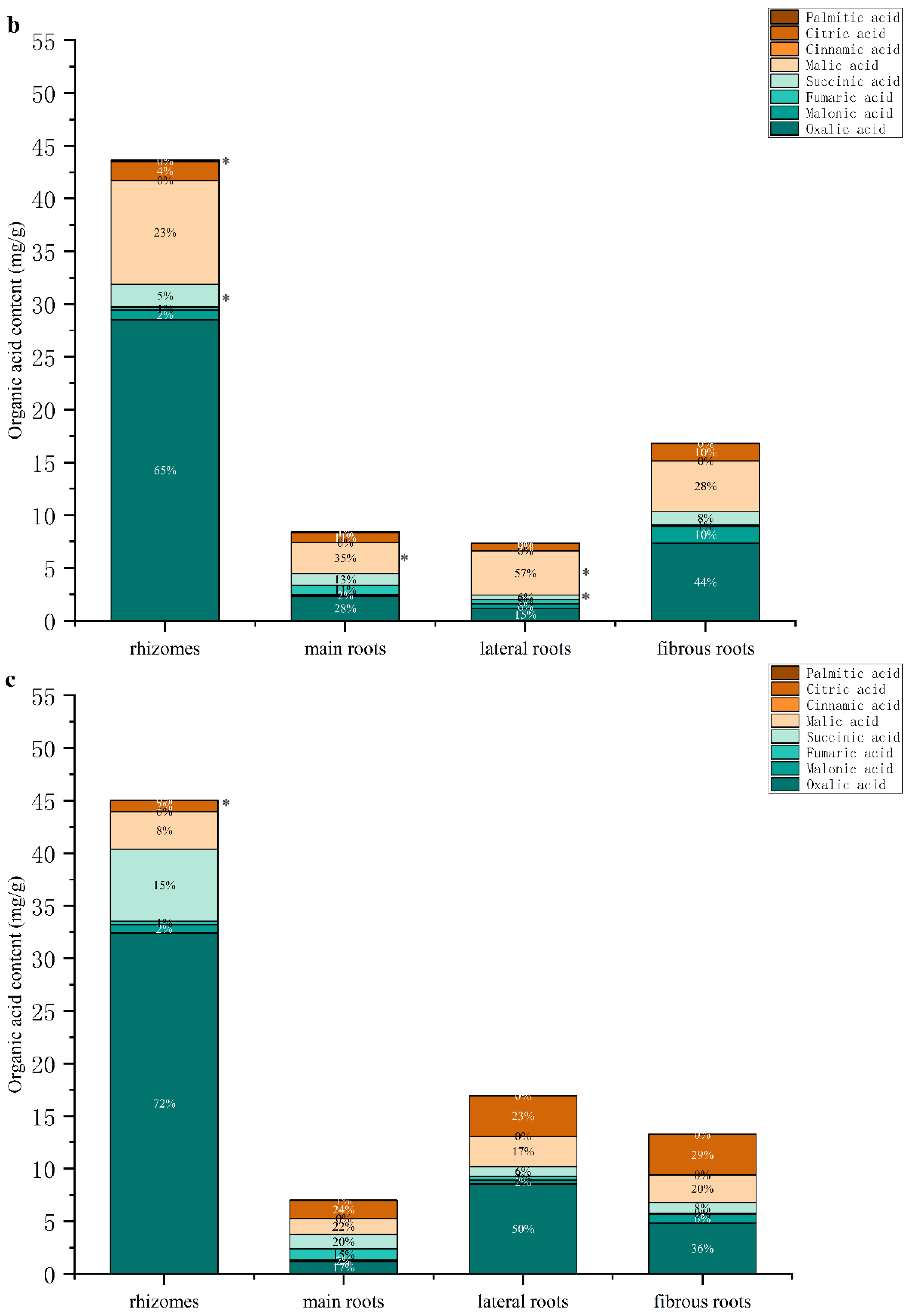
3.3.2. Determination of Organic Acids in Different Parts of GG and MFCG with Different Growth Years
3.4. Analysis of Ginsenosides
3.4.1. Determination of Ginsenosides in Whole MFCG with Different Growth Years
3.4.2. Determination of Ginsenosides in Whole GG and MFCG
3.4.3. Determination of Ginsenosides in Different Parts of GG and MFCG
3.5. Chemometric Analysis
3.5.1. Non-Free Oxalic Acid and Calcium Oxalate Crystals Chemometric Analysis of MFCG and GG
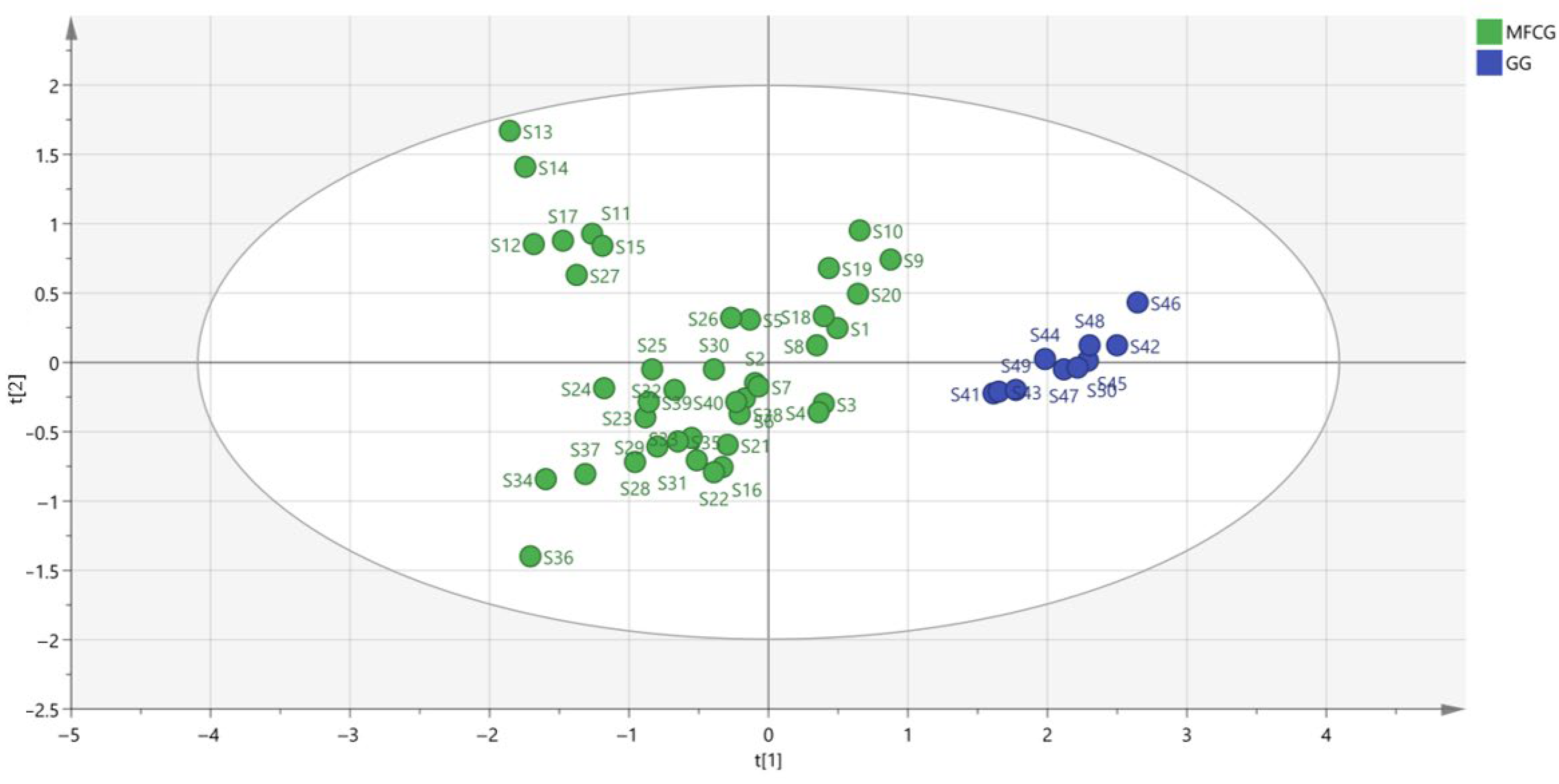
3.5.2. Organic Acid Chemometric Analysis of MFCG and GG
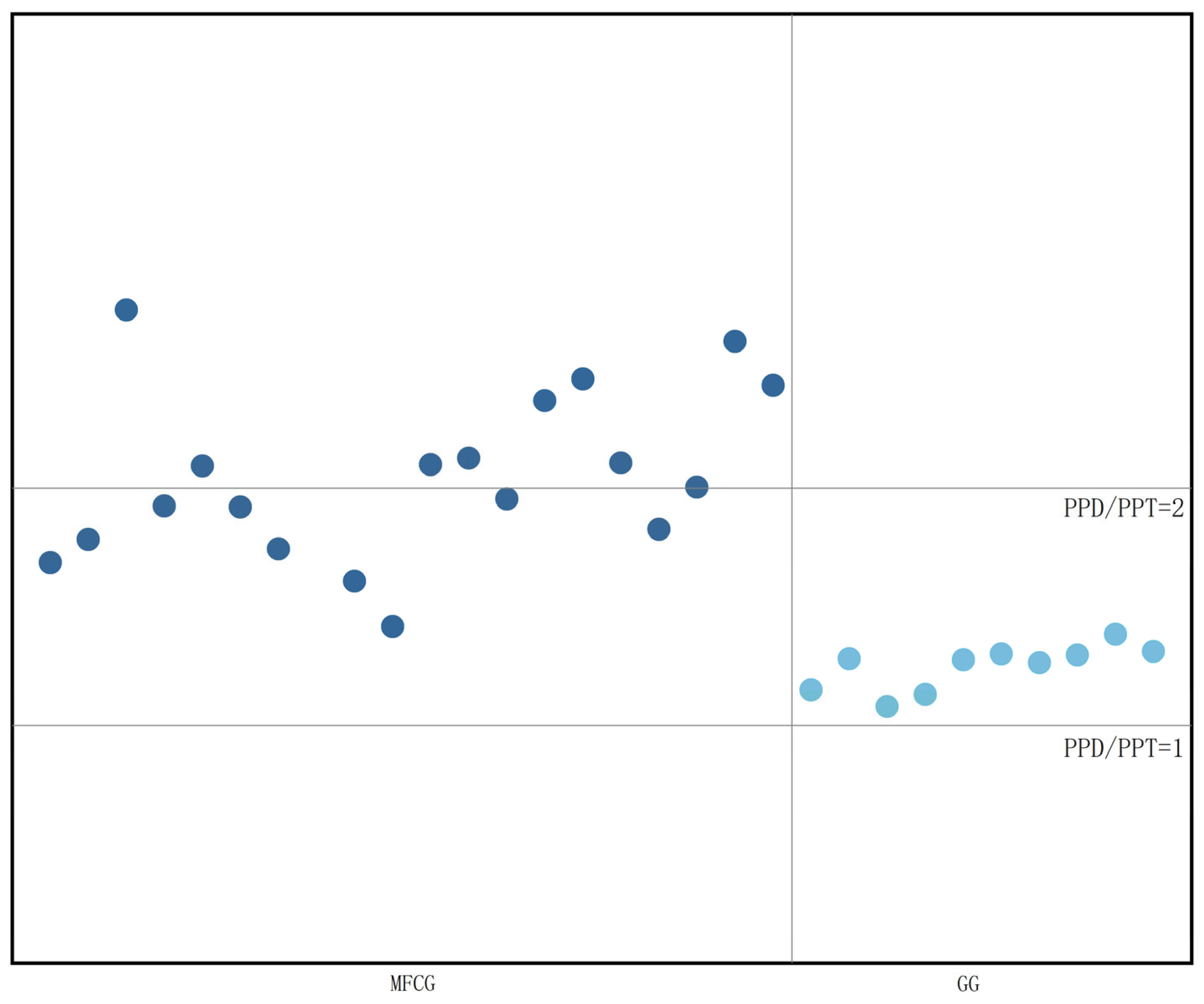
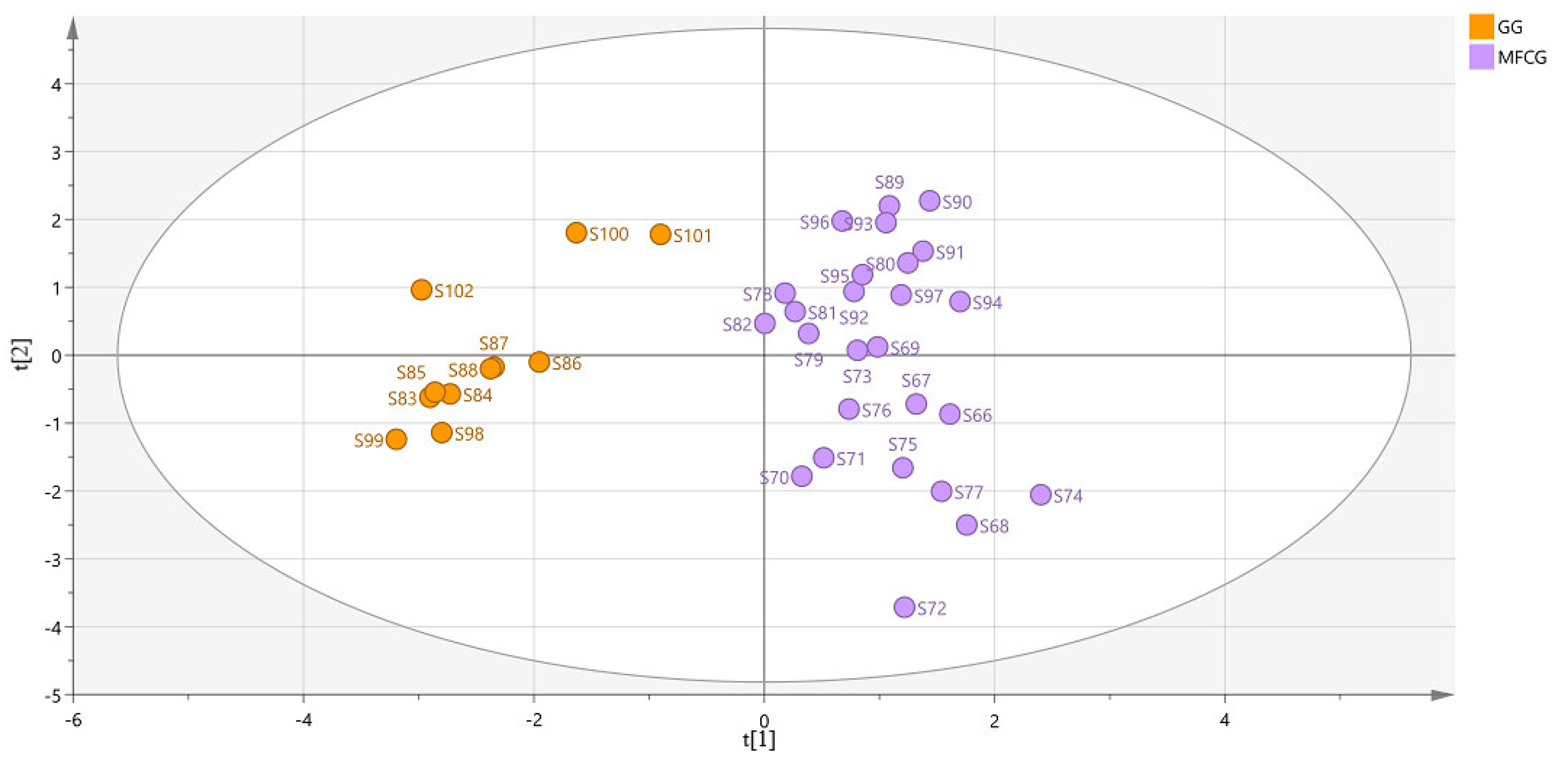
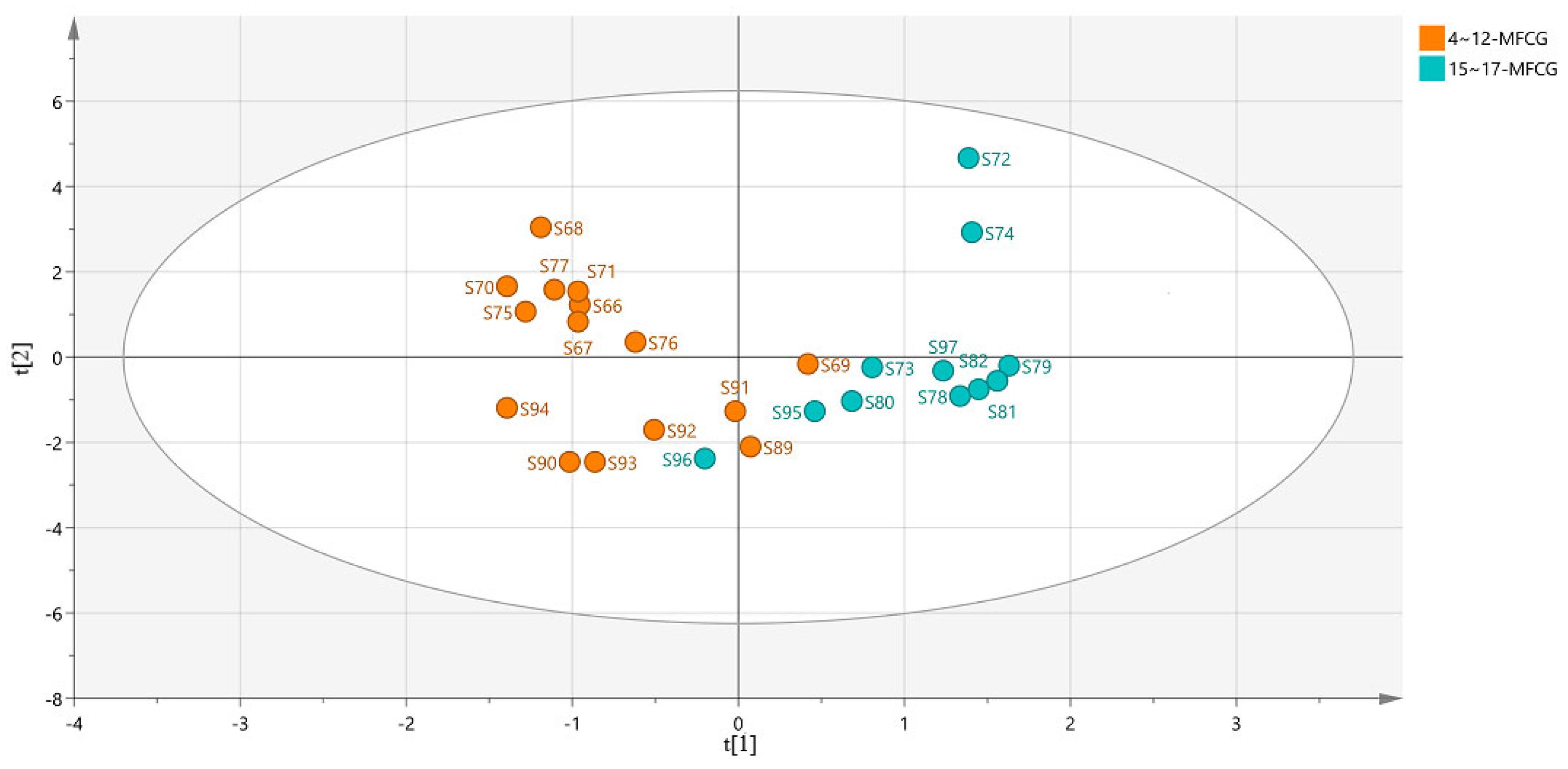
3.5.3. Organic Acid Chemometric Analysis of MFCG in Different Years
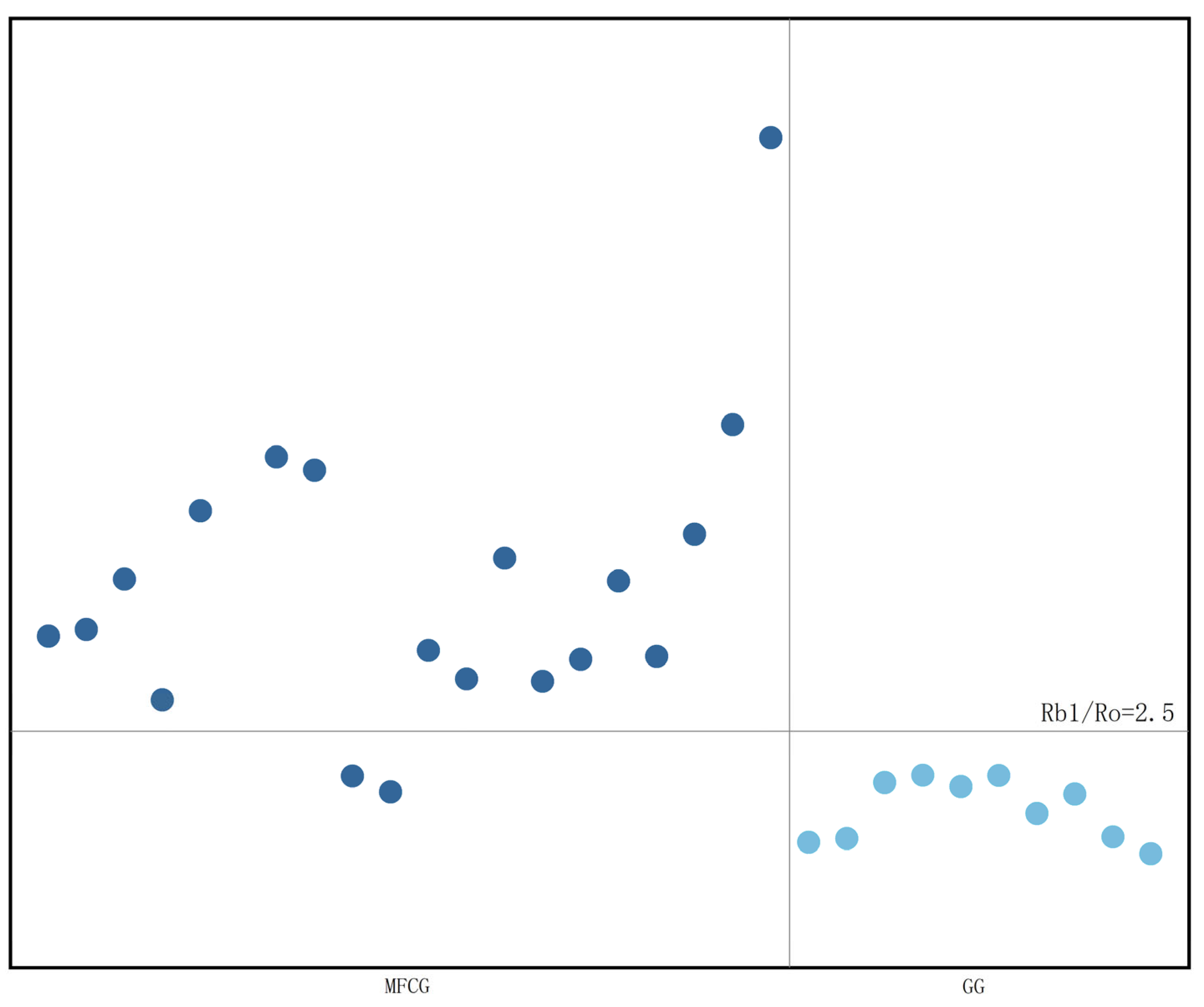
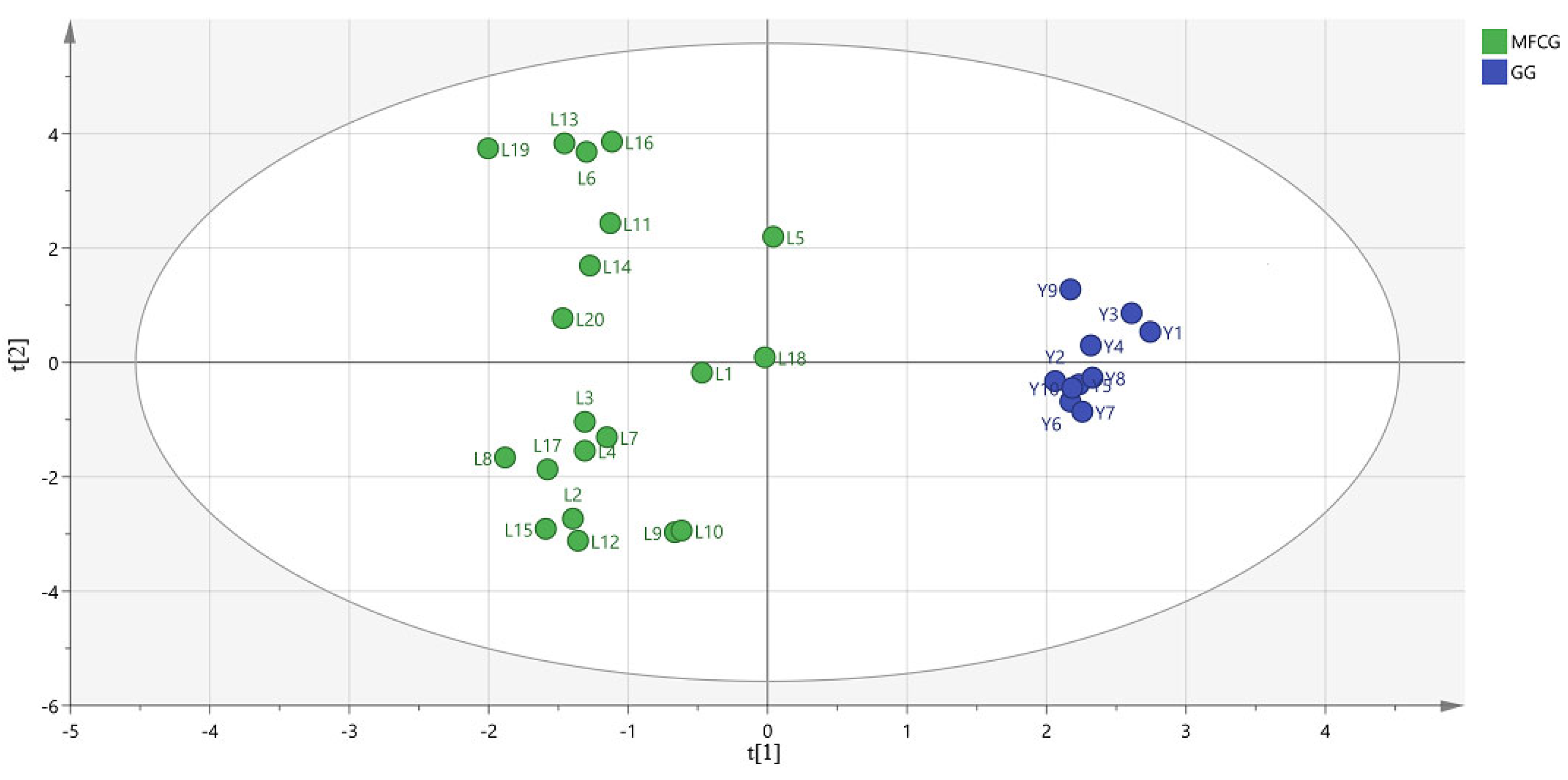
3.5.4. Ginsenosides Chemometric Analysis of MFCG and GG
3.6. Correlation Analysis of Pharmacognosy Data and Each Component
3.6.1. Correlation Analysis of Pharmacognosy Data with Non-Free Oxalic Acid and Calcium Oxalate Crystal
3.6.2. Correlation Analysis of Pharmacognosy Data and Organic Acids
3.6.3. Correlation Analysis of Pharmacognosy Data and Ginsenosides
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Grazina, L.; Mafra, I.; Monaci, L.; Amaral, J.S. Mass spectrometry-based approaches to assess the botanical authenticity of dietary supplements. Compr. Rev. Food Sci. Food Saf. 2023, 22, 3870–3909. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Luan, X.; Yuan, Y.; Dou, D.; Huang, L. The characteristics of ginsenosides and oligosaccharides in mountain-and garden-cultivated ginseng. J. Sci. Food Agric. 2021, 101, 1491–1498. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Xu, J.; Dou, P.; Dou, D.; Huang, L.J. The rhizosphere soil factors on the quality of wild-cultivated herb and its origin traceability as well as distinguishing from garden-cultivated herb: Mountainous forest cultivated ginseng for example. Ind. Crops Prod. 2021, 172, 114078. [Google Scholar] [CrossRef]
- Zhang, G.-M.; Hu, S.-Y.; Chen, G.; Huang, Q.; Gu, H.-Y.; Zheng, C.-W.; Peng, Y.; Xu, H.-X. Age identification of the root of Huanren mountain cultivated ginseng and differentiation with cultivated ginseng using terahertz spectroscopy. J. Food Compos. Anal. 2024, 125, 105790. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, Y.; Ju, Y.; Meng, X.; Yin, C. Rapid identification of rice geographical origin and adulteration by excitation-emission matrix fluorescence spectroscopy combined with chemometrics based on fluorescence probe. Food Control 2023, 146, 109547. [Google Scholar] [CrossRef]
- Biancolillo, A.; Marini, F.; Ruckebusch, C.; Vitale, R. Chemometric Strategies for Spectroscopy-Based Food Authentication. Appl. Sci. 2020, 10, 6544. [Google Scholar] [CrossRef]
- Ding, Y.; Ye, Q.; Liu, M.; Shi, Z.; Liang, Y. Reductive release of Fe mineral-associated organic matter accelerated by oxalic acid. Sci. Total Environ. 2021, 763, 142937. [Google Scholar] [CrossRef]
- Shu, L.; Wang, M.; Wang, S.; Li, Y.; Xu, H.; Qiu, Z.; Li, T. Excessive Oxalic Acid Secreted by Sparassis latifolia Inhibits the Growth of Mycelia during Its Saprophytic Process. Cells 2022, 11, 2423. [Google Scholar] [CrossRef]
- Long, B.; Liao, L.; Jia, F.; Luo, Y.; He, J.; Zhang, W.; Shi, J. Oxalic acid enhances bioremediation of Cr(VI) contaminated soil using Penicillium oxalicum SL2. Chemosphere 2023, 311, 136973. [Google Scholar] [CrossRef]
- Franceschi, V.R.; Nakata, P.A. Calcium oxalate in plants: Formation and function. Annu. Rev. Plant Biol. 2005, 56, 41–71. [Google Scholar] [CrossRef]
- Tan, R.; Tang, Q.; Xia, B.; Fu, C.; Wang, L. Organic acid treatments on citrus insoluble dietary fibers and the corresponding effects on starch in vitro digestion. Int. J. Biol. Macromol. 2024, 275, 134082. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Lee, J.W.; Jo, I.H.; Kwon, N.; Kim, D.; Chung, J.W.; Bang, K.H.; Sung, J. Volatile Compositions of Panax ginseng and Panax quinquifolium Grown for Different Cultivation Years. Foods 2022, 12, 136. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Liu, C.; Luo, Y.; Shi, H.; Li, Q.; Pinchu, C.; Li, X.; Yang, J.; Fan, W. Oxalate in Plants: Metabolism, Function, Regulation, and Application. J. Agric. Food Chem. 2022, 70, 16037–16049. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, N.; Saltz, D.W.J.F.E. Calcium Oxalate Crystals in Leaves of Pancratium sickenbergeri: Constitutive or Induced Defence? Funct. Ecol. 2002, 16, 99–105. [Google Scholar] [CrossRef]
- Vandemark, G.; Thavarajah, S.; Siva, N.; Thavarajah, D. Genotype and Environment Effects on Prebiotic Carbohydrate Concentrations in Kabuli Chickpea Cultivars and Breeding Lines Grown in the U.S. Pacific Northwest. Front. Plant Sci. 2020, 11, 112. [Google Scholar] [CrossRef]
- Oddy, J.; Elmore, J.S.; Halford, N.G. Accounting for environmental variation in the free asparagine content of wheat grain. J. Food Compos. Anal. 2023, 120, 105333. [Google Scholar] [CrossRef]
- Peng, X.Q.; Liu, Y.R.; Huang, T.; Jiang, S.; Huang, W. Interaction of gas phase oxalic acid with ammonia and its atmospheric implications. Phys. Chem. Chem. Phys. 2015, 17, 9552–9563. [Google Scholar] [CrossRef]
- Kayashima, T.; Katayama, T. Oxalic acid is available as a natural antioxidant in some systems. Biochim. Biophys. Acta 2002, 1573, 1–3. [Google Scholar] [CrossRef]
- Wolthers, B.G.; Hayer, M. The determination of oxalic acid in plasma and urine by means of capillary gas chromatography. Clin. Chim. Acta 1982, 120, 87–102. [Google Scholar] [CrossRef]
- Jin, P.; Zhu, H.; Wang, L.; Shan, T.; Zheng, Y. Oxalic acid alleviates chilling injury in peach fruit by regulating energy metabolism and fatty acid contents. Food Chem. 2014, 161, 87–93. [Google Scholar] [CrossRef]
- Xing, Y.M.; Yin, W.Q.; Liu, M.M.; Wang, C.L.; Guo, S.X. Oxalic acid and sclerotial differentiation of Polyporus umbellatus. Sci. Rep. 2015, 5, 10759. [Google Scholar] [CrossRef]
- Kauppi, A.; Tillman-Sutela, E. Calcium oxalate crystals in the mature seeds of Norway spruce, Picea abies (L.) Karst. Trees-Struct. Funct. 1999, 13, 131–137. [Google Scholar] [CrossRef]
- Fernandes, L.; Ramalhosa, E.; Pereira, J.A.; Saraiva, J.A.; Casal, S. Borage, camellia, centaurea and pansies: Nutritional, fatty acids, free sugars, vitamin E, carotenoids and organic acids characterization. Food Res. Int. 2020, 132, 109070. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, C.Z.; Zhu, X.Y.; Wan, J.Y.; Zhang, J.; Li, W.; Ruan, C.C.; Yuan, C.S. Dynamic Changes in Neutral and Acidic Ginsenosides with Different Cultivation Ages and Harvest Seasons: Identification of Chemical Characteristics for Panax ginseng Quality Control. Molecules 2017, 22, 734. [Google Scholar] [CrossRef]
- Zhang, J.K.; Gao, R.; Dou, D.Q.; Kang, T.G. The ginsenosides and carbohydrate profiles of ginseng cultivated under mountainous forest. Pharmacogn. Mag. 2013, 9, S38–S43. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Murata, K.; Takeshita, F.; Takada, K.; Samukawa, K.; Tani, T. Medicinal history and ginsenosides composition of Panax ginseng rhizome, “Rozu”. Yakushigaku Zasshi 2010, 45, 40–48. [Google Scholar]
- Pan, J.; Zheng, W.; Pang, X.; Zhang, J.; Chen, X.; Yuan, M.; Yu, K.; Guo, B.; Ma, B. Comprehensive Investigation on Ginsenosides in Different Parts of a Garden-Cultivated Ginseng Root and Rhizome. Molecules 2021, 26, 1696. [Google Scholar] [CrossRef]


| Organic Acids | Methyl Ester Derivative | Retention Time (min) | Ion Pair | Collision Energy |
|---|---|---|---|---|
| Oxalic acid | Dimethyl oxalate | 8.42 | 118.1/59.0, 59.1/43.0 | 5, 5 |
| Malonic acid | Dimethyl malonate | 11.993 | 101.1/59.0, 101.1/41.8 | 5, 25 |
| Fumaric acid | Dimethyl fumarate | 15.654 | 113.1/85.0, 113.1/53.0 | 5, 15 |
| Succinic acid | Dimethyl succinate | 16.074 | 115.1/87.1, 115.1/55.0 | 5, 15 |
| Malic acid | Dimethyl malate | 19.626 | 103.1/70.9, 103.1/61.0 | 5, 5 |
| Adipic acid | Dimethyl adipate | 23.834 | 114.1/71.0, 114.1/43.0 | 15, 15 |
| Cinnamic acid | Methyl cinnamate | 28.541 | 131.1/103.1, 131.1/77.0 | 5, 25 |
| Citric acid | Trimethyl citrate | 31.171 | 143.1/101.0, 143.1/59.0 | 5, 15 |
| Palmitic acid | Methyl palmitate | 40.986 | 74.1/43.1, 227.1/73.0 | 5, 5 |
| Ingredient | VIP Value |
|---|---|
| Non-free oxalic acid (%) | 1.01507 |
| Calcium oxalate crystals (PCS/mg) | 0.98470 |
| Ingredient | VIP Value |
|---|---|
| Fumaric acid | 1.24443 |
| Malonic acid | 1.13807 |
| Total organic acid | 1.09166 |
| Palmitic acid | 1.08190 |
| Malic acid | 0.96832 |
| Citric acid | 0.91866 |
| Oxalic acid | 0.87792 |
| Succinic acid | 0.86379 |
| Cinnamic acid | 0.70392 |
| Ingredient | VIP Value |
|---|---|
| Succinic acid | 1.50191 |
| Citric acid | 1.2061 |
| Malonic acid | 1.03052 |
| Cinnamic acid | 0.988088 |
| Fumaric acid | 0.950071 |
| Palmitic acid | 0.899554 |
| Total organic acid | 0.89132 |
| Malic acid | 0.688544 |
| Oxalic acid | 0.520494 |
| Ingredient | VIP Value |
|---|---|
| PPD/PPT | 1.38342 |
| Rb1 | 1.36364 |
| Rb1/Ro | 1.14347 |
| PPD | 1.14306 |
| Rb2 | 0.930154 |
| Rg1 | 0.921191 |
| SUM | 0.917947 |
| PPT | 0.862362 |
| Ro | 0.799999 |
| Rc | 0.772205 |
| Rd | 0.750548 |
| Re | 0.71612 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Ran, X.; Xi, Y.; Dou, D. The Distinction of Chemical Profiles of Mountainous Forest Cultivated Ginseng and Garden Ginseng Based on Calcium Oxalate Crystals, Organic Acids, and Ginsenosides. Foods 2025, 14, 3073. https://doi.org/10.3390/foods14173073
Zhang X, Ran X, Xi Y, Dou D. The Distinction of Chemical Profiles of Mountainous Forest Cultivated Ginseng and Garden Ginseng Based on Calcium Oxalate Crystals, Organic Acids, and Ginsenosides. Foods. 2025; 14(17):3073. https://doi.org/10.3390/foods14173073
Chicago/Turabian StyleZhang, Xiaotong, Xiaoku Ran, Yidan Xi, and Deqiang Dou. 2025. "The Distinction of Chemical Profiles of Mountainous Forest Cultivated Ginseng and Garden Ginseng Based on Calcium Oxalate Crystals, Organic Acids, and Ginsenosides" Foods 14, no. 17: 3073. https://doi.org/10.3390/foods14173073
APA StyleZhang, X., Ran, X., Xi, Y., & Dou, D. (2025). The Distinction of Chemical Profiles of Mountainous Forest Cultivated Ginseng and Garden Ginseng Based on Calcium Oxalate Crystals, Organic Acids, and Ginsenosides. Foods, 14(17), 3073. https://doi.org/10.3390/foods14173073






