Impacts of Six Methods of Extraction on Physicochemical Properties, Structural Characteristics and Bioactivities of Polysaccharides from Pholiota nameko Residue
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
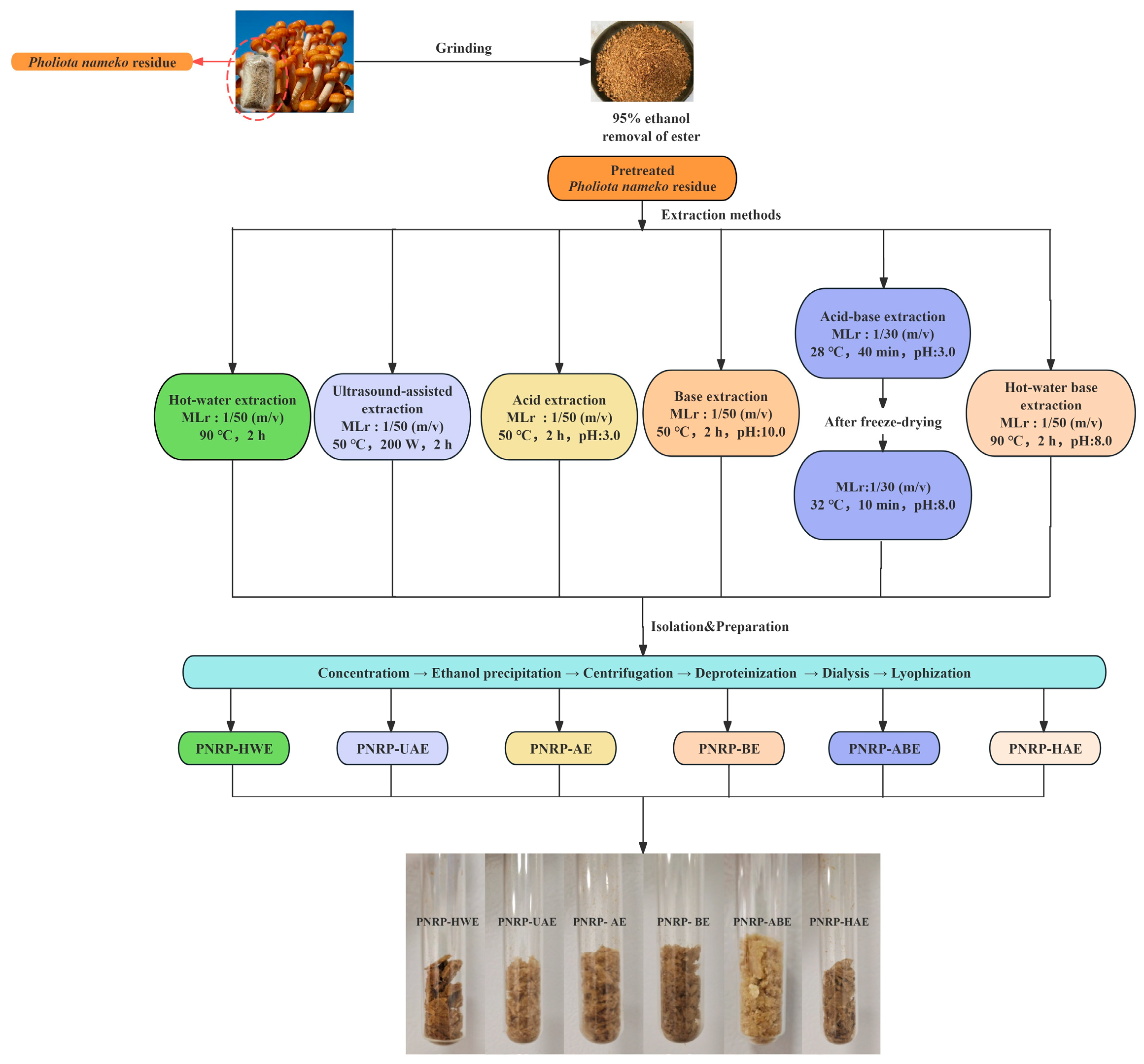
2.2. Extraction Procedure
2.2.1. Hot Water Extraction (HWE)
2.2.2. Ultrasonic-Assisted Extraction (UAE)
2.2.3. Acid-Assisted Extraction (AE)
2.2.4. Base-Assisted Extraction (BE)
2.2.5. Acid–Base Extraction (ABE)
2.2.6. Hot Water–Alkaline-Assisted Extraction (HAE)
2.2.7. The Extraction Yield of PNRPs
2.3. Physicochemical Properties and Structure Characterization
2.3.1. Physicochemical Properties’ Analysis of PNRPs
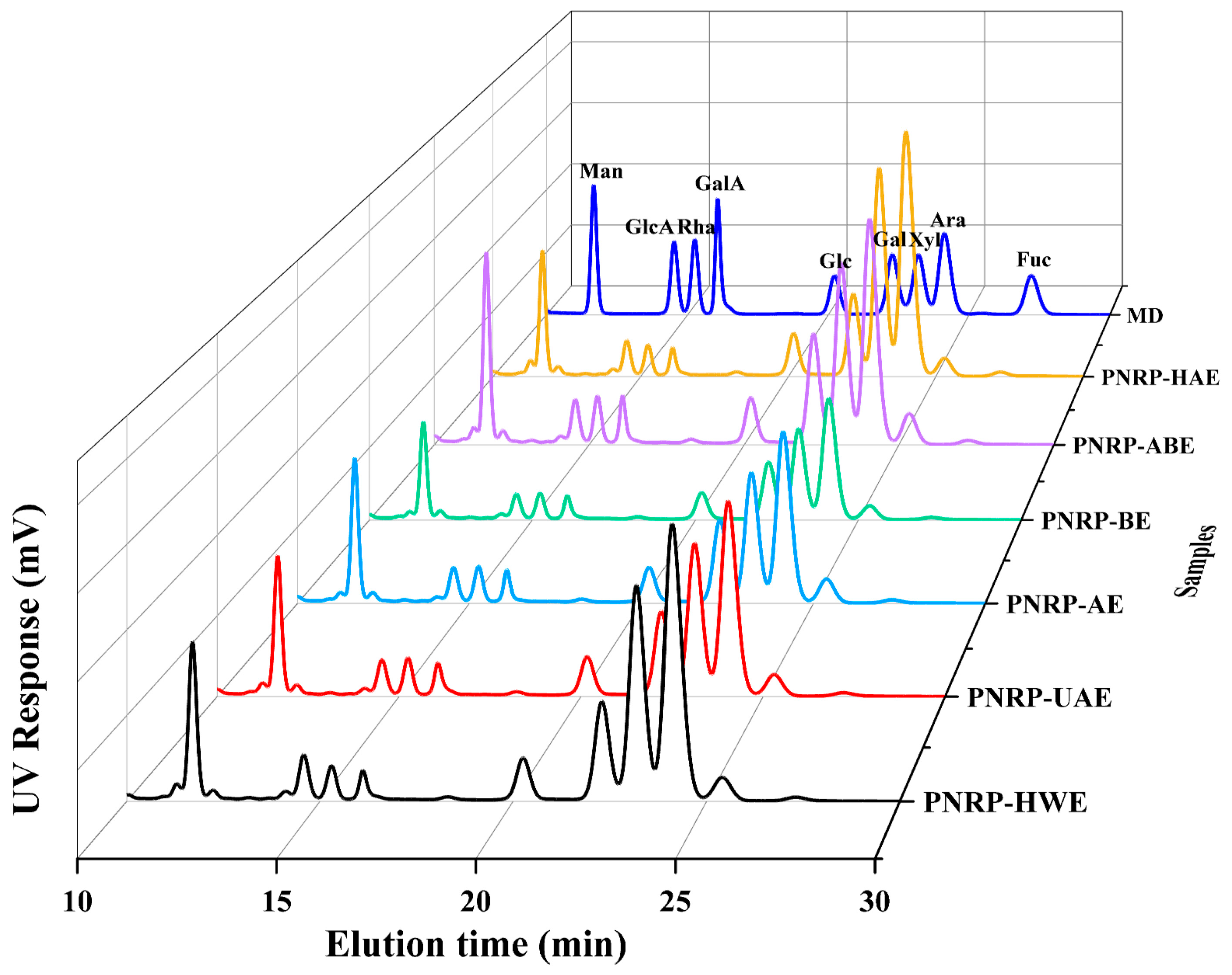
2.3.2. Monosaccharide Composition Determination of PNRPs
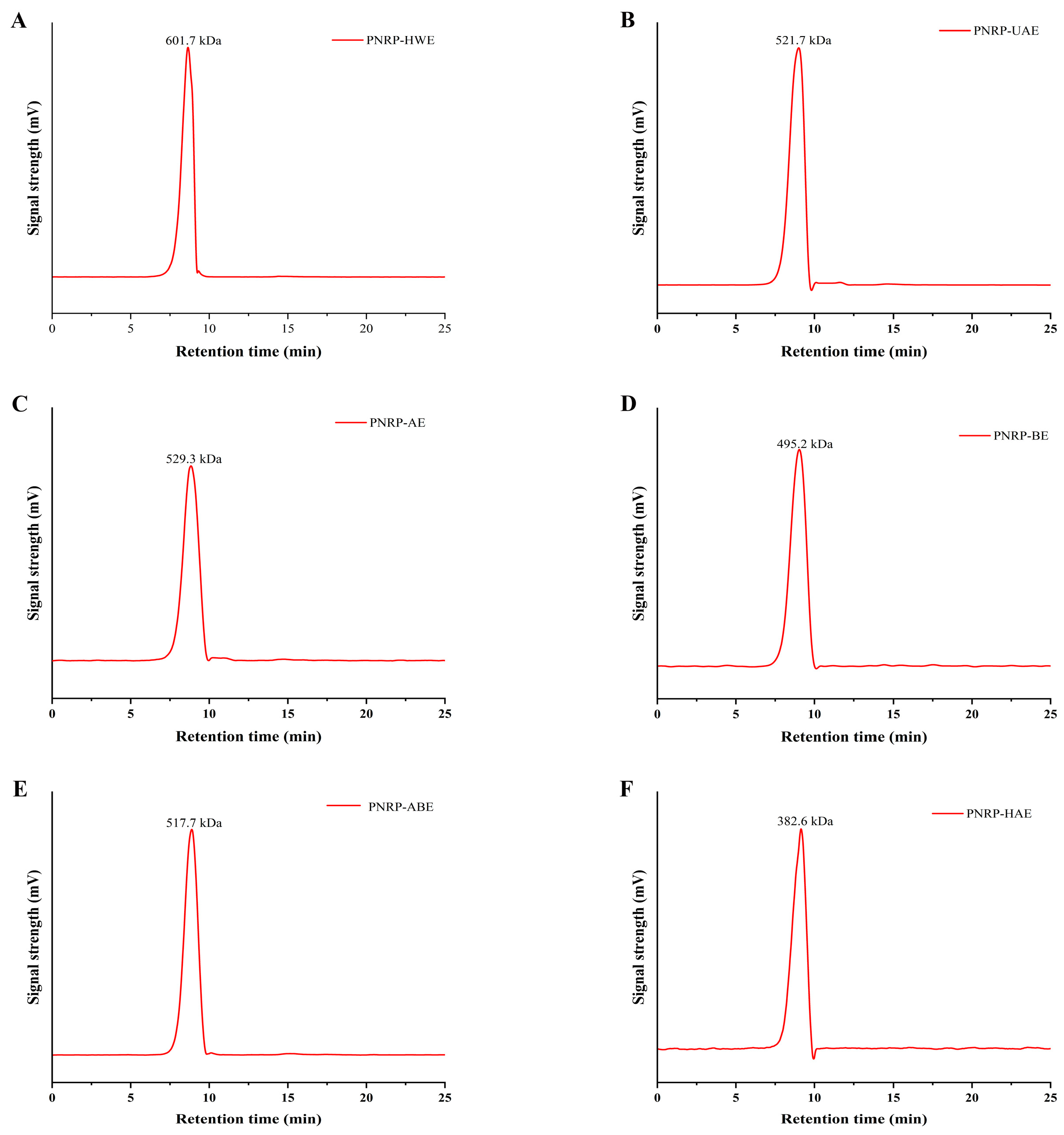
2.3.3. Molecular Weight (Mw) Analysis of PNRPs
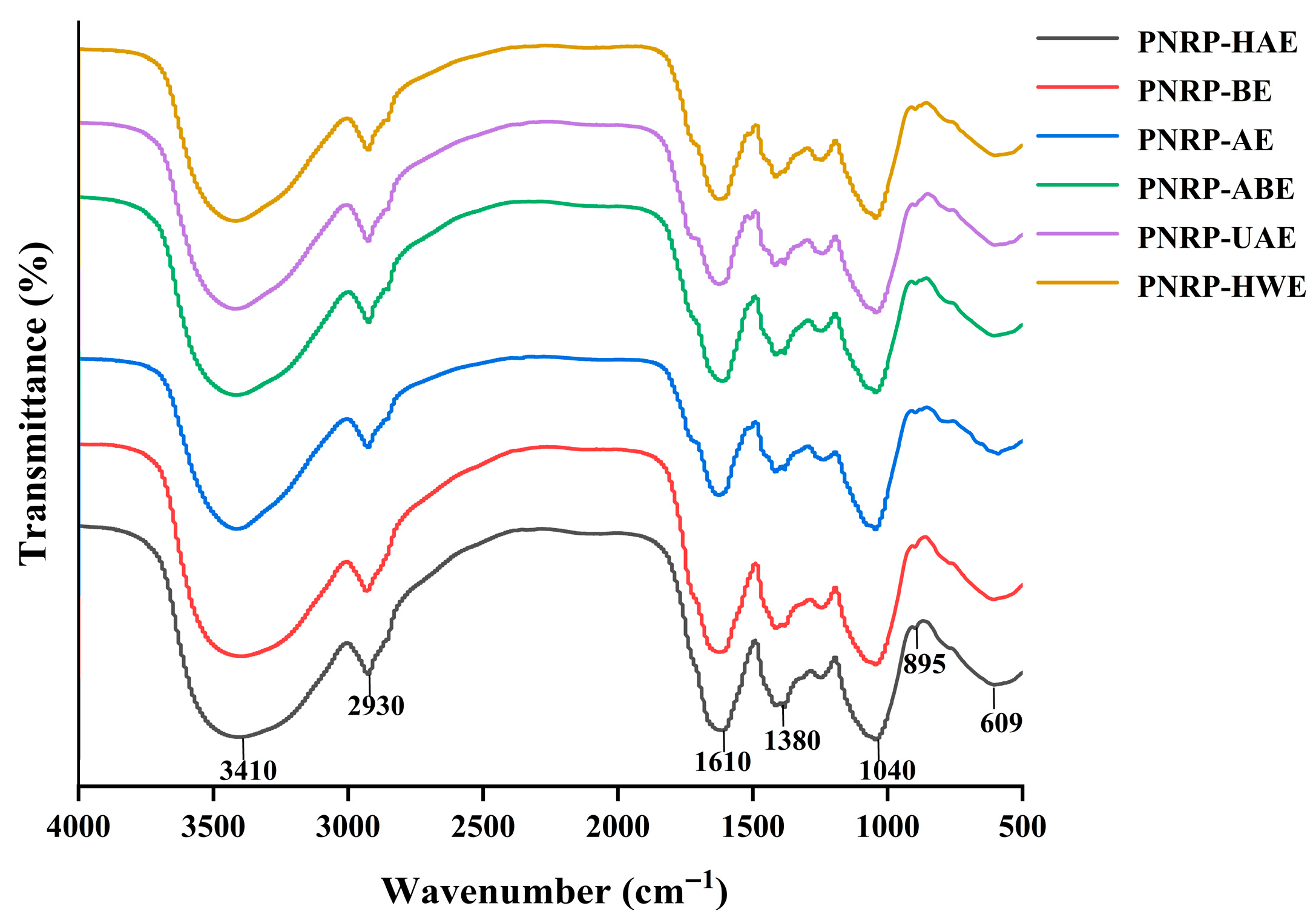
2.3.4. Fourier Transform-Infrared Spectroscopy (FT-IR) Analysis of PNRPs
2.4. Characterization of Surface Morphology of PNRPs
2.5. Chain Conformation Analysis of PNRPs
2.5.1. Congo Red Examination
2.5.2. I2-KI Assay
2.6. Thermal Characteristic Identification
2.7. Functional Characteristics analyse
2.7.1. Water-Holding Capacity (WHC) and Oil-Holding Capacity (OHC)
2.7.2. Emulsifying Activity Index (EAI) and Emulsifying Stability Index (ESI)
2.7.3. Foaming Capacity (FC) and Foaming Stability (FS)
2.8. In Vitro Antioxidant Abilities Analysis of PNRPs
2.9. In Vitro Immunomodulatory Activity of PNRPs
2.10. Statistical Analysis
3. Results and Discussion
3.1. Extraction Yield and Chemical Components of PNRPs
3.1.1. Monosaccharide Composition of PNRPs
3.1.2. Mw of PNRPs
3.1.3. FT-IR Analysis
3.2. Surface Morphological Characterization
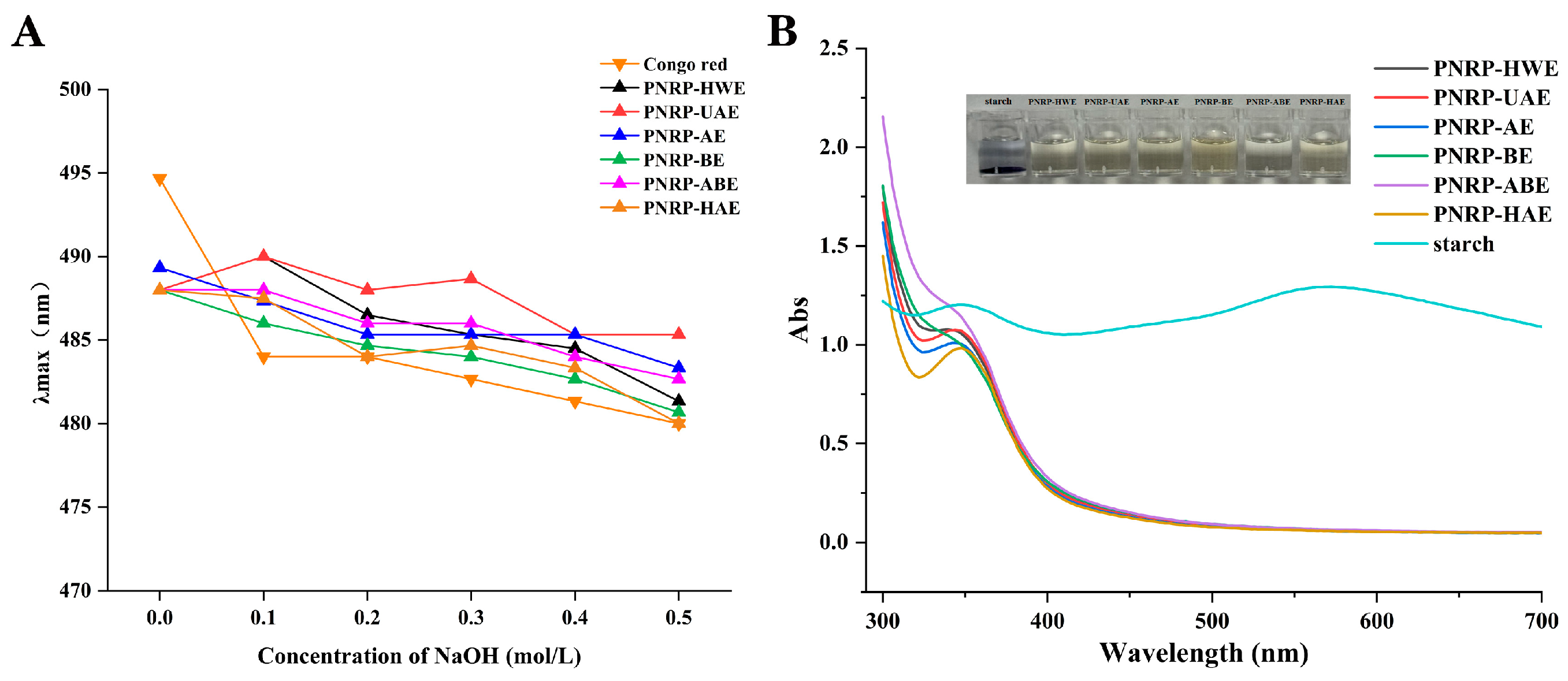
3.3. Congo Red Assay
3.4. I2-KI Analysis
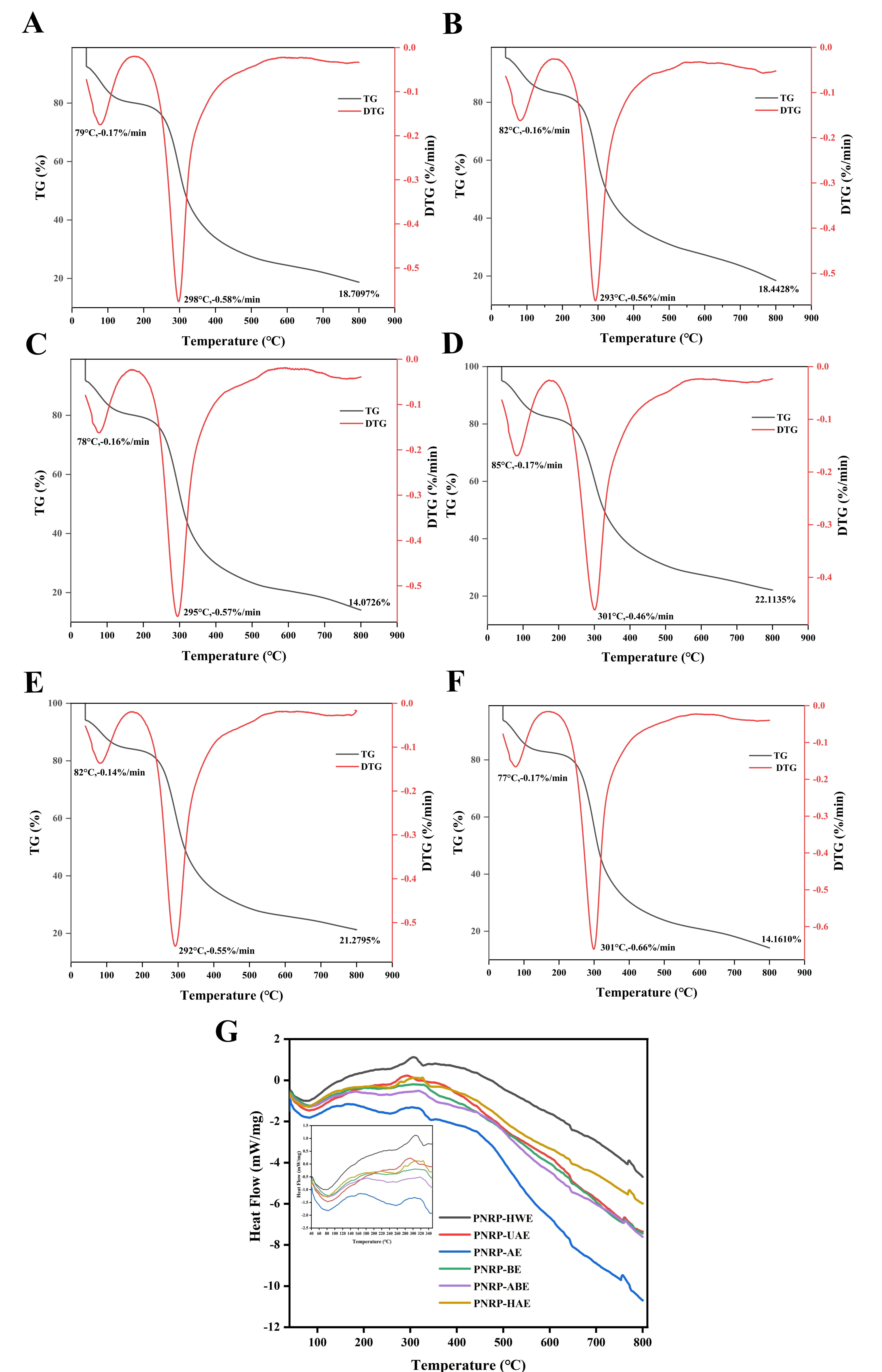
3.5. Thermal Stability Determination of PNRPs
3.6. Function Characteristics of PNRPs
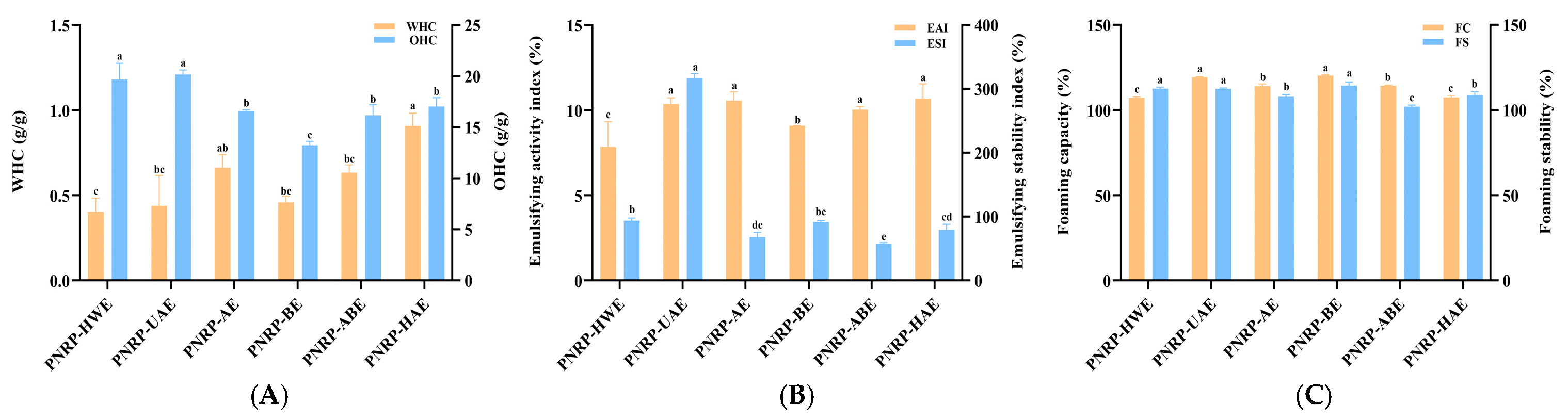
3.6.1. WHC and OHC
3.6.2. EAI and ESI
3.6.3. FC and FS
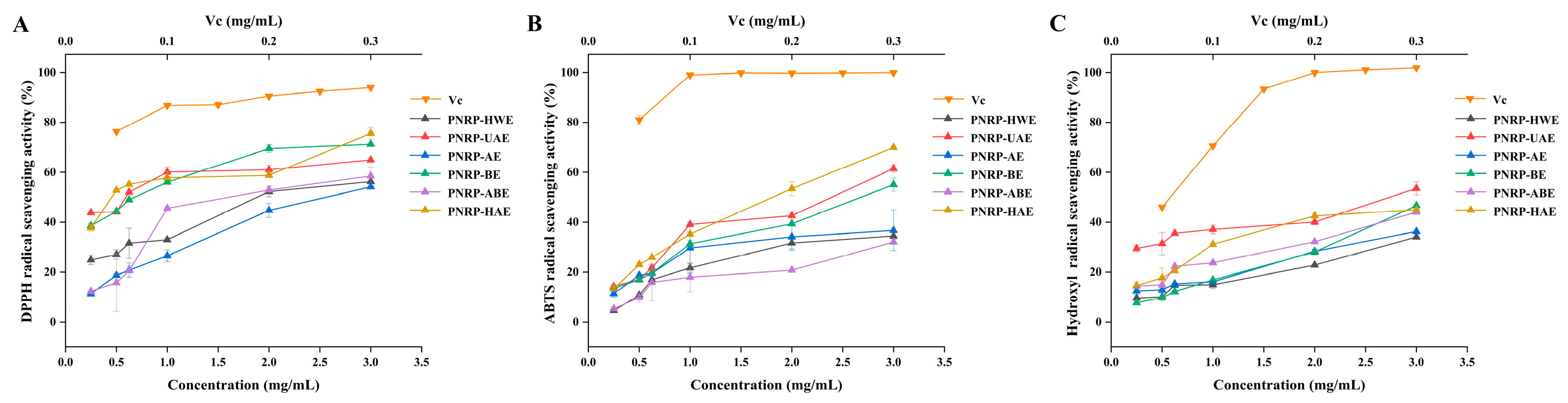
3.7. In Vitro Antioxidant Abilities of PNRPs
3.8. In Vitro Immunoregulatory Activity of PNRPs
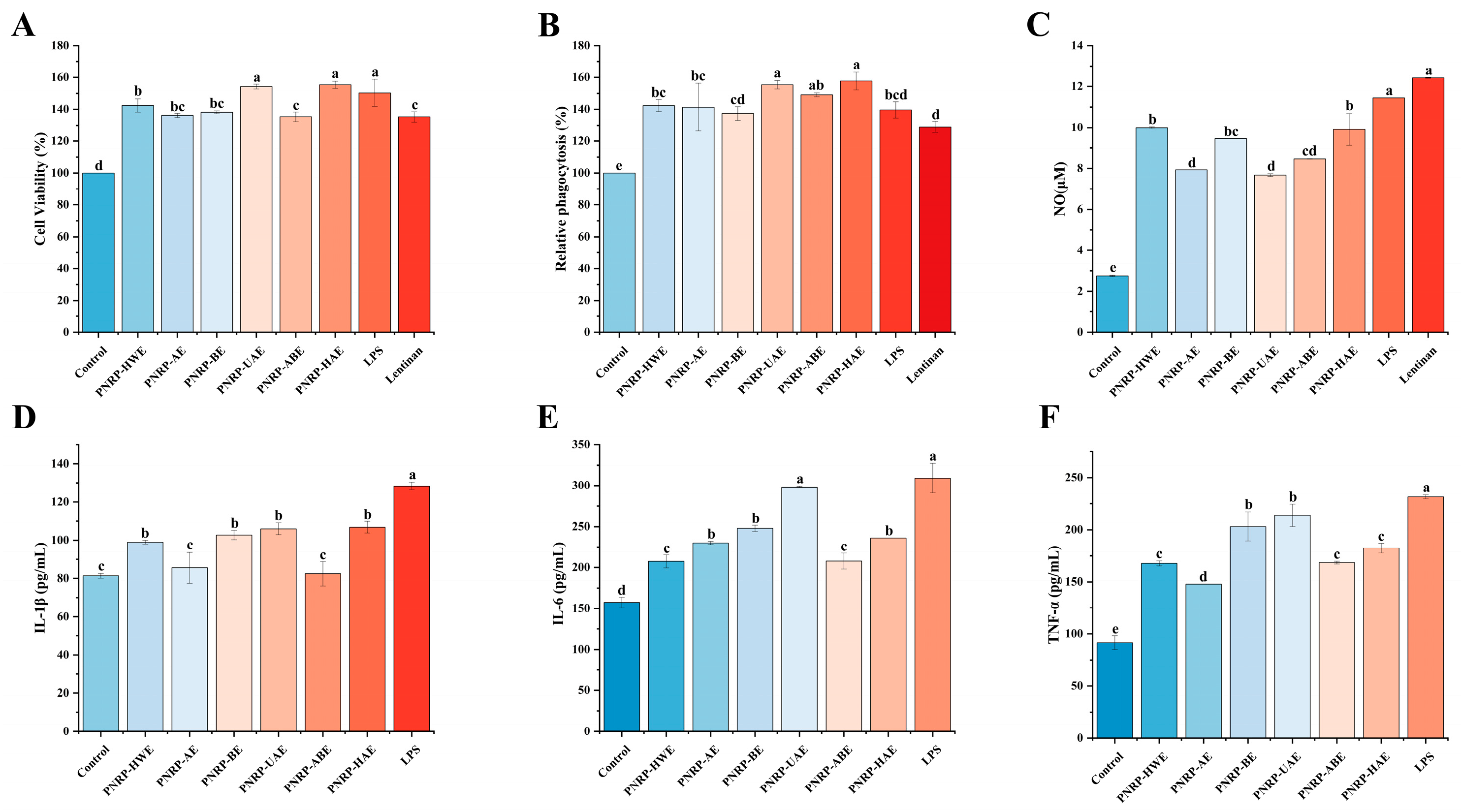
3.8.1. The Effect of PNRPs on the Viability of RAW264.7 Cells
3.8.2. The Effect of PNRPs on the Phagocytic Ability of RAW 264.7 Cells
3.8.3. The Effect of PNRPs on the Secretion of NO and Cytokines in RAW264.7 Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, Y.; Tian, L.; Xu, M.H.; Wang, B.; Song, B.; Li, Y. Research progress in comprehensive utilization of spent mushroom substrates. Microbiol. China 2020, 47, 3658–3670. [Google Scholar]
- Wang, L.; Zhang, B.; Xiao, J.; Huang, Q.; Li, C.; Fu, X. Physicochemical, functional, and biological properties of water-soluble polysaccharides from Rosa roxburghii Tratt fruit. Food Chem. 2018, 249, 127–135. [Google Scholar] [CrossRef]
- Li, X.; Zhang, D.; Sheng, F.; Qing, H. Adsorption characteristics of Copper (II), Zinc (II) and Mercury (II) by four kinds of immobilized fungi residues. Ecotoxicol. Environ. Saf. 2018, 147, 357–366. [Google Scholar] [CrossRef]
- Bao, H.N.D.; Shinomiya, Y.; Ikeda, H.; Ohshima, T. Preventing discoloration and lipid oxidation in dark muscle of yellowtail by feeding an extract prepared from mushroom (Flammulina velutipes) cultured medium. Aquaculture 2009, 295, 243–249. [Google Scholar] [CrossRef]
- Ren, Z.; Liu, W.; Song, X.; Qi, Y.; Zhang, C.; Gao, Z.; Zhang, J.; Jia, L. Antioxidant and anti-inflammation of enzymatic-hydrolysis residue polysaccharides by Lentinula edodes. Int. J. Biol. Macromol. 2018, 120, 811–822. [Google Scholar] [CrossRef]
- Wang, L.; Xu, N.; Zhang, J.; Zhao, H.; Lin, L.; Jia, S.; Jia, L. Antihyperlipidemic and hepatoprotective activities of residue polysaccharide from Cordyceps militaris SU-12. Carbohydr. Polym. 2015, 131, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Song, X.; Cui, W.; Yang, Q. Antioxidant and anti-ageing effects of enzymatic polysaccharide from Pleurotus eryngii residue. Int. J. Biol. Macromol. 2021, 173, 341–350. [Google Scholar] [CrossRef]
- Liu, Y.; Li, H.; Ren, P.; Che, Y.; Zhou, J.; Wang, W.; Yang, Y.; Guan, L. Polysaccharide from Flammulina velutipes residues protects mice from Pb poisoning by activating Akt/GSK3beta/Nrf-2/HO-1 signaling pathway and modulating gut microbiota. Int. J. Biol. Macromol. 2023, 230, 123154. [Google Scholar] [CrossRef]
- Li, H.; Tao, Y.; Zhao, P.; Huai, L.; Zhi, D.; Liu, J.; Li, G.; Dang, C.; Xu, Y. Effects of Pholiota nameko polysaccharide on NF-kappaB pathway of murine bone marrow-derived dendritic cells. Int. J. Biol. Macromol. 2015, 77, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, Y.; Gao, W.; Zhou, R.; Liu, F.; Ng, T.B. A novel antitumor protein from the mushroom Pholiota nameko induces apoptosis of human breast adenocarcinoma MCF-7 cells in vivo and modulates cytokine secretion in mice bearing MCF-7 xenografts. Int. J. Biol. Macromol. 2020, 164, 3171–3178. [Google Scholar] [CrossRef]
- Sung, T.J.; Wang, Y.Y.; Liu, K.L.; Chou, C.H.; Lai, P.S.; Hsieh, C.W. Pholiota nameko Polysaccharides Promotes Cell Proliferation and Migration and Reduces ROS Content in H(2)O(2)-Induced L929 Cells. Antioxidants 2020, 9, 65. [Google Scholar] [CrossRef]
- Fan, Y.J.; Chun, Z.; Wang, G.; Pu, S.R.; Pan, Y.Z.; Ma, J.; Miao, R.S.; Luo, A.X. Isolation, structural characteristics, and in vitro and in vivo antioxidant activity of the acid polysaccharide isolated from Pholiota nameko. Pharmacogn. Mag. 2020, 16, 738–744. [Google Scholar] [CrossRef]
- Zheng, L.; Ma, Y.; Zhang, Y.; Meng, Q.; Yang, J.; Wang, B.; Liu, Q.; Cai, L.; Gong, W.; Yang, Y.; et al. Increased antioxidant activity and improved structural characterization of sulfuric acid-treated stepwise degraded polysaccharides from Pholiota nameko PN-01. Int. J. Biol. Macromol. 2021, 166, 1220–1229. [Google Scholar] [CrossRef]
- Liu, D.; Wang, D.P. Study on Structure and Properties of PKAA-K/WMCPN Composite Superabsorbent under Microwave Irradiation. Henan Sci. Technol. 2020, 48–50. (In Chinese) [Google Scholar]
- Dong, H.; Lin, S.; Zhang, Q.; Chen, H.; Lan, W.; Li, H.; He, J.; Qin, W. Effect of extraction methods on the properties and antioxidant activities of Chuanminshen violaceum polysaccharides. Int. J. Biol. Macromol. 2016, 93, 179–185. [Google Scholar] [CrossRef]
- Chen, H.; Zeng, J.; Wang, B.; Cheng, Z.; Xu, J.; Gao, W.; Chen, K. Structural characterization and antioxidant activities of Bletilla striata polysaccharide extracted by different methods. Carbohydr. Polym. 2021, 266, 118149. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.Q.; Cao, J.J.; Liu, R.; Chen, H.Q. Structural characterization, alpha-amylase and alpha-glucosidase inhibitory activities of polysaccharides from wheat bran. Food Chem. 2021, 341, 128218. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liu, X.; Wang, L.; Song, G.; Jiang, W.; Mu, L.; Li, J. Ficus carica polysaccharide extraction via ultrasound-assisted technique: Structure characterization, antioxidant, hypoglycemic and immunomodulatory activities. Ultrason. Sonochemistry 2023, 101, 106680. [Google Scholar] [CrossRef]
- Wang, J.; Li, J.; Zeng, D.; Zhou, Z.; Zhang, Y.; Ye, G.; Hu, S.; Xin, N.; Li, R.; Cifuentes, A.; et al. Comprehensive evaluation of extraction process, physicochemical properties and bioactivities of hawk tea polysaccharides: Comparison of ultrasonic and hot water extraction. Ultrason. Sonochemistry 2025, 120, 107477. [Google Scholar] [CrossRef]
- Lin, X.; Ji, X.; Wang, M.; Yin, S.; Peng, Q. An alkali-extracted polysaccharide from Zizyphus jujuba cv. Muzao: Structural characterizations and antioxidant activities. Int. J. Biol. Macromol. 2019, 136, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Leong, Y.K.; Yang, F.C.; Chang, J.S. Extraction of polysaccharides from edible mushrooms: Emerging technologies and recent advances. Carbohydr. Polym. 2021, 251, 117006. [Google Scholar] [CrossRef]
- Xu, T.R.; Zhang, H.M.; Wang, S.G.; Xiang, Z.D.; Kong, H.W.; Xue, Q.; He, M.Y.; Yu, X.J.; Li, Y.N.; Sun, D.J.; et al. A review on the advances in the extraction methods and structure elucidation of Poria cocos polysaccharide and its pharmacological activities and drug carrier applications. Int. J. Biol. Macromol. 2022, 217, 536–551. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, M.; Sun, J.; Qin, S.; Diao, T.; Bai, J.; Li, Y. Microwave-assisted aqueous two-phase extraction of polysaccharides from Hippophae rhamnoide L.: Modeling, characterization and hypoglycemic activity. Int. J. Biol. Macromol. 2024, 254, 127626. [Google Scholar] [CrossRef]
- Guoxia, W.; Yilin, N.; Futing, N.; Yingying, C. Effects of Different Extraction Methods on Physicochemical Properties and Antioxidant Activity of Polysaccharides from Lonicerae japonicae Flos. Sci. Technol. Food Ind. 2024, 45, 196–203. [Google Scholar]
- Geng, X.; Guo, D.; Wu, B.; Wang, W.; Zhang, D.; Hou, S.; Bau, T.; Lei, J.; Xu, L.; Cheng, Y.; et al. Effects of different extraction methods on the physico-chemical characteristics and biological activities of polysaccharides from Clitocybe squamulosa. Int. J. Biol. Macromol. 2024, 259, 129234. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chen, J.; Li, J.; Yan, L.; Li, S.; Ye, X.; Liu, D.; Ding, T.; Linhardt, R.J.; Orfila, C.; et al. Extraction and characterization of RG-I enriched pectic polysaccharides from mandarin citrus peel. Food Hydrocoll 2018, 79, 579–586. [Google Scholar] [CrossRef]
- Tang, Y.; Wei, Z.; He, X.; Ling, D.; Qin, M.; Yi, P.; Liu, G.; Li, L.; Li, C.; Sun, J. A comparison study on polysaccharides extracted from banana flower using different methods: Physicochemical characterization, and antioxidant and antihyperglycemic activities. Int. J. Biol. Macromol. 2024, 264, 130459. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, J.; Cao, Y.; Huang, J.; Lin, H.; Zhao, T.; Liu, L.; Shen, P.; Julian McClements, D.; Chen, J.; et al. Extraction and characterization of pectic polysaccharides from Choerospondias axillaris peels: Comparison of hot water and ultrasound-assisted extraction methods. Food Chem. 2023, 401, 134156. [Google Scholar] [CrossRef]
- Liu, J.; Wu, S.; Chen, L.; Li, Q.; Shen, Y.; Jin, L.; Zhang, X.; Chen, P.; Wu, M.; Choi, J.; et al. Different extraction methods bring about distinct physicochemical properties and antioxidant activities of Sargassum fusiforme fucoidans. Int. J. Biol. Macromol. 2020, 155, 1385–1392. [Google Scholar] [CrossRef]
- Chen, S.; Qin, L.; Xie, L.; Yu, Q.; Chen, Y.; Chen, T.; Lu, H.; Xie, J. Physicochemical characterization, rheological and antioxidant properties of three alkali-extracted polysaccharides from mung bean skin. Food Hydrocoll. 2022, 132, 107867. [Google Scholar] [CrossRef]
- Jiao, X.; Li, F.; Zhao, J.; Wei, Y.L.; Zhang, L.Y.; Wang, H.J.; Yu, W.J.; Li, Q.H. Structural diversity and physicochemical properties of polysaccharides isolated from pumpkin (Cucurbita moschata) by different methods. Food Res. Int. 2023, 163, 112157. [Google Scholar] [CrossRef]
- Huang, L.; Shen, M.; Zhang, X.; Jiang, L.; Song, Q.; Xie, J. Effect of high-pressure microfluidization treatment on the physicochemical properties and antioxidant activities of polysaccharide from Mesona chinensis Benth. Carbohydr. Polym. 2018, 200, 191–199. [Google Scholar] [CrossRef]
- Filisetti-Cozzi, T.M.; Carpita, N.C. Measurement of uronic acids without interference from neutral sugars. Anal. Biochem. 1991, 197, 157–162. [Google Scholar] [CrossRef]
- Chen, L.Y.; Cheng, C.W.; Liang, J.Y. Effect of esterification condensation on the Folin-Ciocalteu method for the quantitative measurement of total phenols. Food Chem. 2015, 170, 10–15. [Google Scholar] [CrossRef]
- Li, H.; Liu, S.; Liu, Y.; Li, W.; Niu, A.; Ren, P.; Liu, Y.; Jiang, C.; Inam, M.; Guan, L. Effects of in vitro digestion and fermentation of Nostoc commune Vauch. polysaccharides on properties and gut microbiota. Carbohydr. Polym. 2022, 281, 119055. [Google Scholar] [CrossRef]
- Yu, X.; Luo, Y.; Zhang, X. Determination Method of Relative Molecular Mass and Distribution of Bovine Serum Albumin by High Performance Size Exclusion Chromatography. Chem. Anal. Meterage. 2013, 22, 20–23. [Google Scholar]
- Chen, G.T.; Fu, Y.X.; Yang, W.J.; Hu, Q.H.; Zhao, L.Y. Effects of polysaccharides from the base of Flammulina Velutipes stipe on growth of murine RAW264.7, B16F10 and L929 cells. Int. J. Biol. Macromol. 2018, 107, 2150–2156. [Google Scholar] [CrossRef]
- Mutailifu, P.; Nuerxiati, R.; Lu, C.F.; Huojiaaihemaiti, H.; Abuduwaili, A.; Yili, A. Extraction, purification, and characterization of polysaccharides from Alhagi pseudoalhagi with antioxidant and hypoglycemic activities. Process Biochem. 2022, 121, 339–348. [Google Scholar] [CrossRef]
- Lv, Y.Y.; Yao, L.; Qiu, M.J.; Li, L.; Qiu, S.Y.; Liu, Y.Y.; Wei, C.Y. Physicochemical properties, structural characteristics and bioactivities of Pyracantha fortuneana polysaccharides prepared by six methods. Ind. Crop. Prod. 2024, 208, 117933. [Google Scholar] [CrossRef]
- Abarghoei, M.; Goli, M.; Shahi, S. Investigation of cold atmospheric plasma effects on functional and physicochemical properties of wheat germ protein isolate. Food Sci. Technol. 2023, 177, 114585. [Google Scholar] [CrossRef]
- Bing, S.; Chen, X.; Zhong, X.; Li, Y.; Sun, G.; Wang, C.; Liang, Y.; Zhao, X.; Hua, D.; Chen, L.; et al. Structural, functional and antioxidant properties of Lentinus edodes protein hydrolysates prepared by five enzymes. Food Chem. 2024, 437, 137805. [Google Scholar] [CrossRef]
- Hu, Y.N.; Sung, T.J.; Chou, C.H.; Liu, K.L.; Hsieh, L.P.; Hsieh, C.W. Characterization and Antioxidant Activities of Yellow Strain Flammulina velutipes (Jinhua Mushroom) Polysaccharides and Their Effects on ROS Content in L929 Cell. Antioxidants 2019, 8, 298. [Google Scholar] [CrossRef]
- Yin, C.; Fan, X.; Fan, Z.; Shi, D.; Gao, H. Optimization of enzymes-microwave-ultrasound assisted extraction of Lentinus edodes polysaccharides and determination of its antioxidant activity. Int. J. Biol. Macromol. 2018, 111, 446–454. [Google Scholar] [CrossRef]
- Zhang, H.; Zou, P.; Zhao, H.; Qiu, J.; Regenstein, J.M.; Yang, X. Isolation, purification, structure and antioxidant activity of polysaccharide from pinecones of Pinus koraiensis. Carbohydr. Polym. 2021, 251, 117078. [Google Scholar] [CrossRef]
- Yang, Y.; Ren, P.; Sun, Y.; Li, J.; Zhou, X.; Zhang, H.; He, C.; Dai, H.; Guan, L. Structure elucidation and molecular mechanism of an immunomodulatory polysaccharide from Nostoc commune. Int. J. Biol. Macromol. 2024, 283, 137435. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.M.; Castro, R.; Arranz, J.A.; Lamas, J. Immunomodulating activities of acidic sulphated polysaccharides obtained from the seaweed Ulva rigida C. Agardh. Int. Immunopharmacol. 2007, 7, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; He, X.; Liu, G.; Wei, Z.; Sheng, J.; Sun, J.; Li, C.; Xin, M.; Li, L.; Yi, P. Effects of different extraction methods on the structural, antioxidant and hypoglycemic properties of red pitaya stem polysaccharide. Food Chem. 2023, 405, 134804. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, H.; Du, W.; Qian, L.; Xu, Y.; Huang, Y.; Xiong, Q.; Li, H.; Yuan, J. Comparison of different extraction methods for polysaccharides from Crataeguspinnatifida Bunge. Int. J. Biol. Macromol. 2020, 150, 1011–1019. [Google Scholar] [CrossRef]
- Li, H.; Tao, Y.; Zhao, P.; Zhi, D.; Gao, X.; Zhao, X.; Li, M. Effect of ultrasound-assisted extraction on physicochemical properties and TLR2-affinity binding of the polysaccharides from Pholiota nameko. Int. J. Biol. Macromol. 2019, 135, 1020–1027. [Google Scholar] [CrossRef]
- Wang, H.; Wang, N.L.; Ma, L.; Pei, D.; Di, D.L.; Liu, J.F. Polysaccharides from different cultivars of wolfberry: Physicochemical characteristics and protection against oxidative stress. Ind. Crop. Prod. 2023, 197, 116548. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, F.; Wang, M.; Ma, L.; Ye, H.; Zeng, X. A comparative study of the neutral and acidic polysaccharides from Allium macrostemon Bunge. Carbohydr. Polym. 2015, 117, 980–987. [Google Scholar] [CrossRef]
- Wang, X.; Yu, A.; Zhang, Z.; Wang, J.; Bi, H.; Wang, M. Research progress on extraction and purification, structural characteristics, pharmacological activities, structure-activity relationships, and applications of Chrysanthemum indicum L. polysaccharides: A review. Int. J. Biol. Macromol. 2025, 315, 144689. [Google Scholar] [CrossRef]
- Pu, J.B.; Xia, B.H.; Hu, Y.J.; Zhang, H.J.; Chen, J.; Zhou, J.; Liang, W.Q.; Xu, P. Multi-Optimization of Ultrasonic-Assisted Enzymatic Extraction of Atratylodes macrocephala Polysaccharides and Antioxidants Using Response Surface Methodology and Desirability Function Approach. Molecules 2015, 20, 22220–22235. [Google Scholar] [CrossRef]
- Reichembach, L.H.; de Oliveira Petkowicz, C.L. New findings on acid-extractable pectins from soy hull. Carbohydr. Polym. 2022, 294, 119831. [Google Scholar] [CrossRef] [PubMed]
- Constant, S.; Barakat, A.; Robitzer, M.; Di Renzo, F.; Dumas, C.; Quignard, F. Composition, texture and methane potential of cellulosic residues from Lewis acids organosolv pulping of wheat straw. Bioresour. Technol. 2016, 216, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, L.; Zhu, T.; Liu, Y.; Chu, J.; Chen, N. Structural characterization of polysaccharides of marine origin: A review. Int. J. Biol. Macromol. 2025, 317, 144797. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Xu, H.; Xie, L.; Sun, J.; Sun, T.; Wu, X.; Fu, Q. Purification, characterization and in vitro anticoagulant activity of polysaccharides from Gentiana scabra Bunge roots. Carbohydr. Polym. 2016, 140, 308–313. [Google Scholar] [CrossRef]
- Kan, L.; Chai, Y.; Li, X.; Zhao, M. Structural analysis and potential anti-tumor activity of Sporisorium reilianum (Fries) polysaccharide. Int. J. Biol. Macromol. 2020, 153, 986–994. [Google Scholar] [CrossRef]
- Yang, W.J.; Fang, Y.; Liang, J.; Hu, Q.H. Optimization of ultrasonic extraction of Flammulina velutipes polysaccharides and evaluation of its acetylcholinesterase inhibitory activity. Food Res. Int. 2011, 44, 1269–1275. [Google Scholar] [CrossRef]
- Kačuráková, M.; Capek, P.; Sasinková, V.; Wellner, N.; Ebringerová, A. FT-IR study of plant cell wall model compounds: Pectic polysaccharides and hemicelluloses. Carbohydr. Polym. 2000, 43, 195–203. [Google Scholar] [CrossRef]
- Rozi, P.; Abuduwaili, A.; Mutailifu, P.; Gao, Y.; Rakhmanberdieva, R.; Aisa, H.A.; Yili, A. Sequential extraction, characterization and antioxidant activity of polysaccharides from Fritillaria pallidiflora Schrenk. Int. J. Biol. Macromol. 2019, 131, 97–106. [Google Scholar] [CrossRef]
- Gan, J.P.; Huang, Z.Y.; Yu, Q.; Peng, G.Y.; Chen, Y.; Xie, J.H.; Nie, S.P.; Xie, M.Y. Microwave assisted extraction with three modifications on structural and functional properties of soluble dietary fibers from grapefruit peel. Food Hydrocoll. 2020, 101, 105549. [Google Scholar] [CrossRef]
- Abuduwaili, A.; Rozi, P.; Mutailifu, P.; Gao, Y.H.; Nuerxiati, R.; Aisa, H.A.; Yili, A. Effects of different extraction techniques on physicochemical properties and biological activities of polysaccharides from Fritillaria pallidiflora Schrenk. Process Biochem. 2019, 83, 189–197. [Google Scholar] [CrossRef]
- Gong, L.L.; Meng, F.J.; Hou, Y.C.; Liu, Y.; Xu, J.J.; Zhang, W.N.; Chen, Y. Purification, characterization, and bioactivity of two new polysaccharide fractions from Thelephora ganbajun mushroom. J. Food Biochem. 2020, 44, e13092. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.X.; Yu, C.; Han, Z.; Chen, Z.Y.; Wei, X.L.; Wang, Y.F. Comparative analysis of existence form for selenium and structural characteristics in artificial selenium-enriched and synthetic selenized green tea polysaccharides. Int. J. Biol. Macromol. 2020, 154, 1408–1418. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, Z.; Zhao, H.; Liu, M.; Lin, C.; Li, L.; Ma, B. Pectin polysaccharide from Flos Magnoliae (Xin Yi, Magnolia biondii Pamp. flower buds): Hot-compressed water extraction, purification and partial structural characterization. Food Hydrocoll. 2022, 122, 107061. [Google Scholar] [CrossRef]
- Einhorn-Stoll, U.; Kunzek, H.; Dongowski, G. Thermal analysis of chemically and mechanically modified pectins. Food Hydrocoll. 2007, 21, 1101–1112. [Google Scholar] [CrossRef]
- Shi, Z.J.; Xiao, L.P.; Deng, J.; Feng, X.; Sun, R.C. Isolation and characterization of solublepolysaccharides solublepolysaccharides of dendrocalamus brandisii: A high-ylelding bamboo species. BioRes 2011, 6, 5151–5166. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, S.; Shen, M.; Jiang, L.; Ren, Y.; Luo, Y.; Wen, H.; Xie, J. Physicochemical, rheological and thermal properties of Mesona chinensis polysaccharides obtained by sodium carbonate assisted and cellulase assisted extraction. Int. J. Biol. Macromol. 2019, 126, 30–36. [Google Scholar] [CrossRef]
- de Oliveira, A.M.; de Oliveira Junior, E.N.; Franco, T.T.; Yu, L. Physicochemical Characterization of Thermally Treated Chitosans and Chitosans Obtained by Alkaline Deacetylation. Int. J. Polym. Sci. 2014, 2014, 174–182. [Google Scholar] [CrossRef]
- Kang, Y.; Chang, Y.H. Structural characterization and prebiotic activity of rhamnogalacturonan-I rich pumpkin pectic polysaccharide extracted by alkaline solution. Int. J. Biol. Macromol. 2024, 270, 132311. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.A. Physicochemical characteristics of food and the digestion of starch and dietary fibre during gut transit. Proc. Nutr. Soc. 1988, 47, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Spinei, M.; Oroian, M. Structural, functional and physicochemical properties of pectin from grape pomace as affected by different extraction techniques. Int. J. Biol. Macromol. 2023, 224, 739–753. [Google Scholar] [CrossRef]
- Wang, Z.; Li, L.; Jia, F.; Wu, J.; Jin, W.; Zhao, W.; Cao, J.; Cheng, Y.; Shi, L.; Yun, S.; et al. Exploring the effect of pH-shifting on the gel properties and interaction of heat-induced Flammulina velutipes polysaccharide-porcine myofibrillar protein for improving the quality of Flammulina velutipes-pork patties. Food Chem. 2025, 465, 142187. [Google Scholar] [CrossRef]
- Liang, X.; Ran, J.; Sun, J.; Wang, T.; Jiao, Z.; He, H.; Zhu, M. Steam-explosion-modified optimization of soluble dietary fiber extraction from apple pomace using response surface methodology. Cyta J. Food 2018, 16, 20–26. [Google Scholar] [CrossRef]
- Carvalho, A.; Portela, M.; Sousa, M.B.; Martins, F.S.; Rocha, F.C.; Farias, D.F.; Feitosa, J. Physiological and physico-chemical characterization of dietary fibre from the green seaweed Ulva fasciata Delile. Braz. J. Biol. 2009, 69, 969–977. [Google Scholar] [CrossRef]
- Ahmadi, S.; Sheikh-Zeinoddin, M.; Soleimanian-Zad, S.; Alihosseini, F.; Yadav, H. Effects of different drying methods on the physicochemical properties and antioxidant activities of isolated acorn polysaccharides. Food Sci. Technol. 2019, 100, 1–9. [Google Scholar] [CrossRef]
- Chen, R.; Luo, S.; Wang, C.; Bai, H.; Lu, J.; Tian, L.; Gao, M.; Wu, J.; Bai, C.; Sun, H. Effects of ultra-high pressure enzyme extraction on characteristics and functional properties of red pitaya (Hylocereus polyrhizus) peel pectic polysaccharides. Food Hydrocoll. 2021, 121, 107016. [Google Scholar] [CrossRef]
- Yang, M.D. Study on the Functional Properties, Structural Characterizationon Auricularia auricula and Its Hypoglycmic Activity. Master’s Thesis, Nanjing Agricultural University, Nanjing, China, 2023. [Google Scholar]
- Tian, R.; Han, X.; Tian, B.; Li, G.; Sun, L.; Tian, S.; Qin, L.; Wang, S. Effects of covalent binding of different polyphenols on structure, rheology and functional properties of whey protein isolate. Food Sci. Technol. 2023, 184, 114968. [Google Scholar] [CrossRef]
- Naji-Tabasi, S.; Razavi, S.M.A. New studies on basil (Ocimum bacilicum L.) seed gum: Part II—Emulsifying and foaming characterization. Carbohydr. Polym. 2016, 149, 140–150. [Google Scholar] [CrossRef]
- Chen, X.; Wang, Z.; Kan, J. Polysaccharides from ginger stems and leaves: Effects of dual and triple frequency ultrasound assisted extraction on structural characteristics and biological activities. Food Biosci. 2021, 42, 101166. [Google Scholar] [CrossRef]
- Genc, B.; Tunç, M.T.; Adiguzel, A. Characterization of Water-Soluble Extracellular Polysaccharide from Aeribacillus pallidus IM17. Indian J. Microbiol. 2024, 64, 973–982. [Google Scholar] [CrossRef]
- Leal, M.R.S.; Albuquerque, P.B.S.; Rodrigues, N.E.R.; Silva, P.M.D.S.; de Oliveira, W.F.; Correia, M.T.D.S.; Kennedy, J.F.; Coelho, L.C.B.B. A review on the use of polysaccharides as thickeners in yogurts. Carbohydr. Polym. Technol. Appl. 2024, 8, 100547. [Google Scholar] [CrossRef]
- Liu, W.; Jiang, Y.; Shi, J. Effects of selenylation on Chinese yam polysaccharides: Structure, antioxidant, and digestive properties. Food Chem. X 2025, 27, 102435. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, X.; Zhang, J.; Wang, T.; Liu, S.; Ma, H.; Inam, M.; Guan, L. Green ultrasonic-assisted enzymatic extraction of polysaccharides from Flammulina velutipes residues by response surface methodology. Sustain. Chem. Pharm. 2024, 41, 101690. [Google Scholar]
- Bingwu, L.; Huihua, H. Structural characterization of a novel polysaccharide from Hericium erinaceus and its protective effects against H2O2-induced injury in human gastric epithelium cells. J. Funct. Foods. 2019, 56, 265–275. [Google Scholar]
- Wang, J.; Hu, S.; Nie, S.; Yu, Q.; Xie, M. Reviews on Mechanisms of In Vitro Antioxidant Activity of Polysaccharides. Oxidative Med. Cell. Longev. 2016, 2016, 5692852. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, H.; Wei, M.; Zhu, C.H. Effects of enzymatic treatment on the physicochemical properties and antioxidant activity of hawthorn pectin. Mater. Today Commun. 2022, 30, 103225. [Google Scholar] [CrossRef]
- Xue, H.; Wang, W.; Bian, J.; Gao, Y.; Hao, Z.; Tan, J. Recent advances in medicinal and edible homologous polysaccharides: Extraction, purification, structure, modification, and biological activities. Int. J. Biol. Macromol. 2022, 222, 1110–1126. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.Y.; Zou, B.; WU, J.J. Structural characterization, antioxidant, and prebiotic activity of wampee polysaccharides. Food Ferment. Ind. 2024, 40, 156–164. [Google Scholar]
- Sun, Y.; Zhang, M.; Jiang, X.; Peng, K.; Yi, Y.; Meng, Y.; Wang, H. Structural characterization and immunoregulatory mechanism of a low-molecular-weight polysaccharide from lotus root. Int. J. Biol. Macromol. 2024, 280, 135957. [Google Scholar] [CrossRef]
- Xu, G.; Yu, Z.; Zhao, W. Immunomodulation effects of isochlorogenic acid a from apple on RAW264.7 cells via modulation of TLR2 and TLR4 target proteins. Food Biosci. 2024, 58, 103773. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef]
- Xu, X.; Yan, H.; Tang, J.; Chen, J.; Zhang, X. Polysaccharides in Lentinus edodes: Isolation, Structure, Immunomodulating Activity and Future Prospective. Crit. Rev. Food Sci. Nutr. 2014, 54, 474–487. [Google Scholar] [CrossRef]
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef]
- Trivedi, S.; Patel, K.; Belgamwar, V.; Wadher, K. Functional polysaccharide lentinan: Role in anti-cancer therapies and management of carcinomas. Pharmacological Research. Mod. Chin. Med. 2022, 2, 100045. [Google Scholar]
- Ferreira, S.S.; Passos, C.P.; Madureira, P.; Vilanova, M.; Coimbra, M.A. Structure–function relationships of immunostimulatory polysaccharides: A review. Carbohydr. Polym. 2015, 132, 378–396. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Liu, Q.; Fu, Y.; Luo, X.; Hu, M.; Ma, F.; Wang, Q.; Lai, X.; Zhou, L. Effects of Lycium barbarum polysaccharides with different molecular weights on function of RAW264.7 macrophages. Food Agric. Immunol. 2018, 29, 808–820. [Google Scholar] [CrossRef]
- Zhang, J.; Pan, W.; Cui, J.; Lin, Z.; Gao, J.; Lin, J. Purified polysaccharides from Plantago asiatica L.: Preparation, characterization and immune-activating effects. Int. J. Biol. Macromol. 2025, 318, 144771. [Google Scholar] [CrossRef] [PubMed]
- Lü, L.; Li, X.; Zou, Q.; He, J.; Xing, H.; Miyashita, K.; Yang, Y.; Du, L. EPA-enriched sulfoquinovosyldiacylglycerol attenuates inflammation in lipopolysaccharide-stimulated RAW264.7 macrophages. J. Funct. Foods. 2025, 129, 106888. [Google Scholar] [CrossRef]
- Lee, K.Y.; Jeon, Y.J. Macrophage activation by polysaccharide isolated from Astragalus membranaceus. Int. Immunopharmacol. 2005, 5, 1225–1233. [Google Scholar] [CrossRef]
- Lin, Y.; Guan, Q.; Li, L.; Tang, Z.; Zhang, Q.; Zhao, X. In Vitro Immuno-Modulatory Potentials of Purslane (Portulaca oleracea L.) Polysaccharides with a Chemical Selenylation. Foods 2021, 11, 14. [Google Scholar] [CrossRef]
- Idriss, H.T.; Naismith, J.H. TNF α and the TNF receptor superfamily: Structure-function relationship(s). Microsc. Res. Tech. 2000, 50, 184–195. [Google Scholar] [CrossRef]

| Extraction Yield (%, w/w) | Polysaccharides (%, w/w) | Total Uronic Acids (%, w/w) | Total Proteins (%, w/w) | Total Phenols (mg GAE/g) | |
|---|---|---|---|---|---|
| PNRP-HWE | 4.24 ± 0.22 a | 92.56 ± 1.17 a | 5.17 ± 0.08 f | 0.18 ± 0.004 b | 0.03 ± 0.0002 a |
| PNRP-UAE | 3.08 ± 0.53 b | 90.33 ± 0.09 b | 7.67 ± 0.04 a | 0.14 ± 0.006 e | 0.03 ± 0.0001 a |
| PNRP-AE | 3.01 ± 0.43 b | 75.68 ± 0.12 f | 7.43 ± 0.06 b | 0.17 ± 0.003 c | 0.03 ± 0.0001 a |
| PNRP-BE | 2.57 ± 0.67 b | 77.89 ± 0.78 e | 5.37 ± 0.01 e | 0.19 ± 0.001 a | 0.02 ± 0.0001 b |
| PNRP-ABE | 1.09 ± 0.22 c | 81.54 ± 0.05 d | 6.66 ± 0.11 c | 0.15 ± 0.001 d | 0.02 ± 0.0001 b |
| PNRP-HAE | 3.92 ± 0.24 a | 85.12 ± 0.05 c | 6.06 ± 0.14 d | 0.18 ± 0.002 b | 0.03 ± 0.0001 a |
| Variety | IC50 Values for DPPH (mg/mL) | IC50 Values for ABTS (mg/mL) | IC50 Values for Hydroxyl (mg/mL) |
|---|---|---|---|
| Vc | 0.07 ± 0.0075 d | 0.05 ± 0.0003 e | 0.17 ± 0.0013 f |
| PNRP-HWE | 1.91 ± 0.0695 b | 5.86 ± 0.1160 ab | 8.37 ± 0.3410 a |
| PNRP-UAE | 0.58 ± 0.0202 c | 2.00 ± 0.0505 d | 3.00 ± 0.1920 e |
| PNRP-AE | 2.60 ± 0.0735 a | 5.20 ± 0.8065 b | 6.71 ± 0.0980 b |
| PNRP-BE | 0.57 ± 0.0015 c | 3.42 ± 0.0295 c | 3.72 ± 0.0710 d |
| PNRP-ABE | 1.89 ± 0.1590 b | 7.12 ± 0.3180 a | 5.55 ± 0.0655 c |
| PNRP-HAE | 0.59 ± 0.0164 c | 1.57 ± 0.0290 d | 3.97 ± 0.1145 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Bai, Y.; Zhang, X.; Wang, Y.; Li, Z.; He, C.; Guan, L. Impacts of Six Methods of Extraction on Physicochemical Properties, Structural Characteristics and Bioactivities of Polysaccharides from Pholiota nameko Residue. Foods 2025, 14, 3071. https://doi.org/10.3390/foods14173071
Zhang J, Bai Y, Zhang X, Wang Y, Li Z, He C, Guan L. Impacts of Six Methods of Extraction on Physicochemical Properties, Structural Characteristics and Bioactivities of Polysaccharides from Pholiota nameko Residue. Foods. 2025; 14(17):3071. https://doi.org/10.3390/foods14173071
Chicago/Turabian StyleZhang, Jingbo, Yajing Bai, Xiaoxue Zhang, Yiyao Wang, Zongshu Li, Chengguang He, and Lili Guan. 2025. "Impacts of Six Methods of Extraction on Physicochemical Properties, Structural Characteristics and Bioactivities of Polysaccharides from Pholiota nameko Residue" Foods 14, no. 17: 3071. https://doi.org/10.3390/foods14173071
APA StyleZhang, J., Bai, Y., Zhang, X., Wang, Y., Li, Z., He, C., & Guan, L. (2025). Impacts of Six Methods of Extraction on Physicochemical Properties, Structural Characteristics and Bioactivities of Polysaccharides from Pholiota nameko Residue. Foods, 14(17), 3071. https://doi.org/10.3390/foods14173071






