Effects and Mechanisms of Long-Term Lycium barbarum Water Consumption on Skeletal Muscle Function in Aged Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of LBW
2.3. Antioxidant Properties
2.4. Component Analysis of LBW
2.5. Animal Experiment
2.6. Section of Skeletal Muscles and Hematoxylin and Eosin-Straining
2.7. Mice Grip Strength Test
2.8. Mice Rotarod Fatigue Test
2.9. Mice Treadmill Exercise Test
2.10. Measurement of Expression Levels of Aging-Related Factors
2.11. Measurement of Oxidative Stress and Inflammatory Levels
2.12. Quantitative Real-Time PCR Analysis (qRT-PCR)
2.13. Western Blot
2.14. Unsupervised Metabolomics Analysis
2.15. Statistical Analysis
3. Result
3.1. Optimization of LBW Preparation
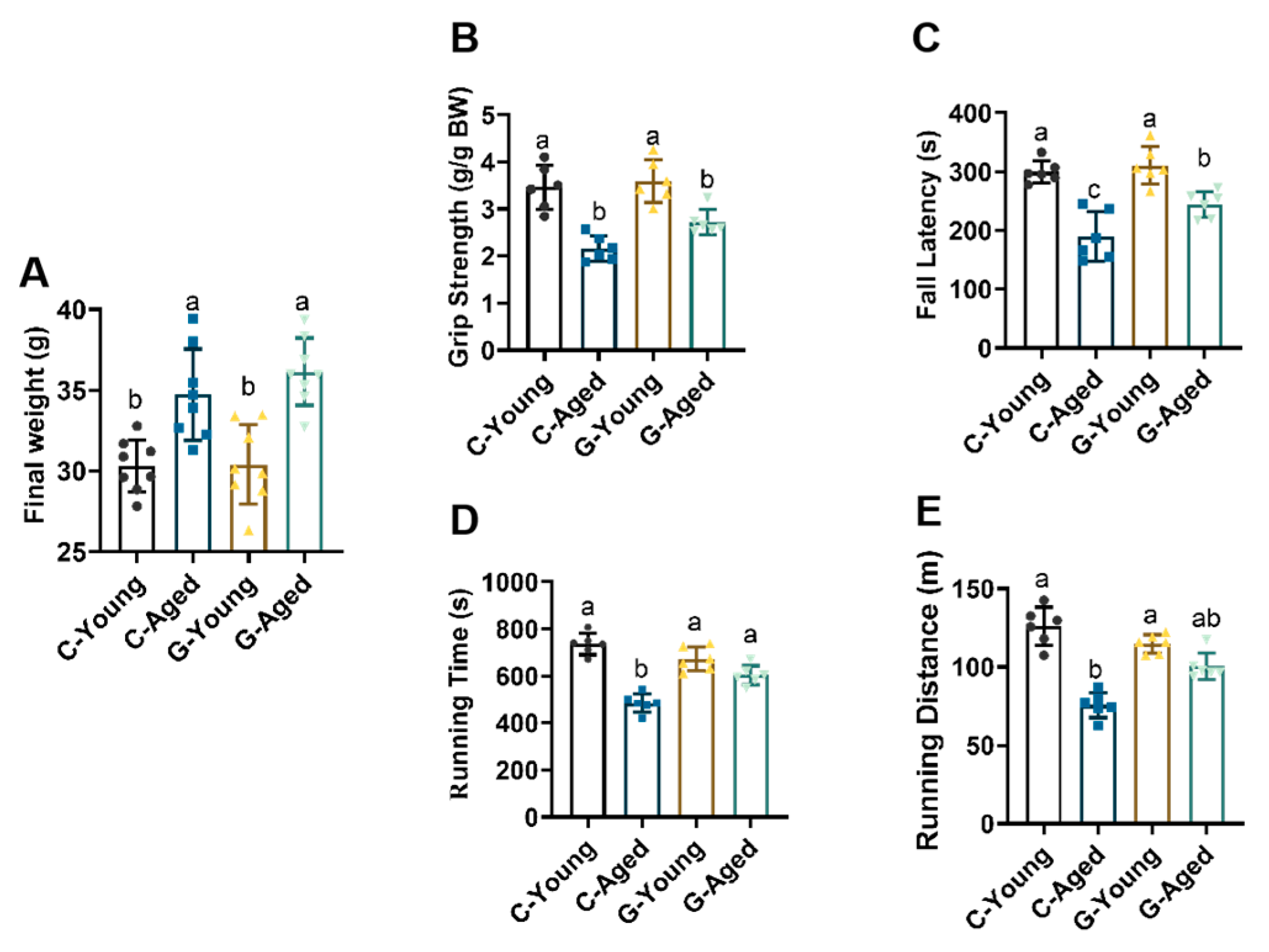
3.2. Effects of Long-Term LBW Consumption on Mice Motor Function and Body Weight
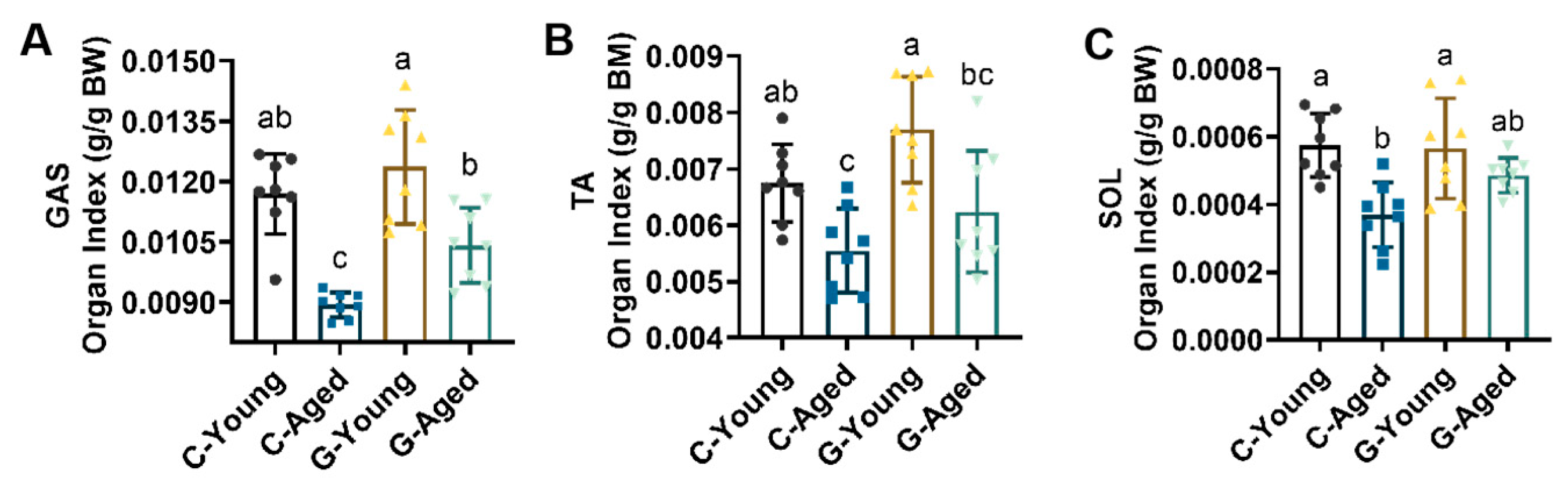
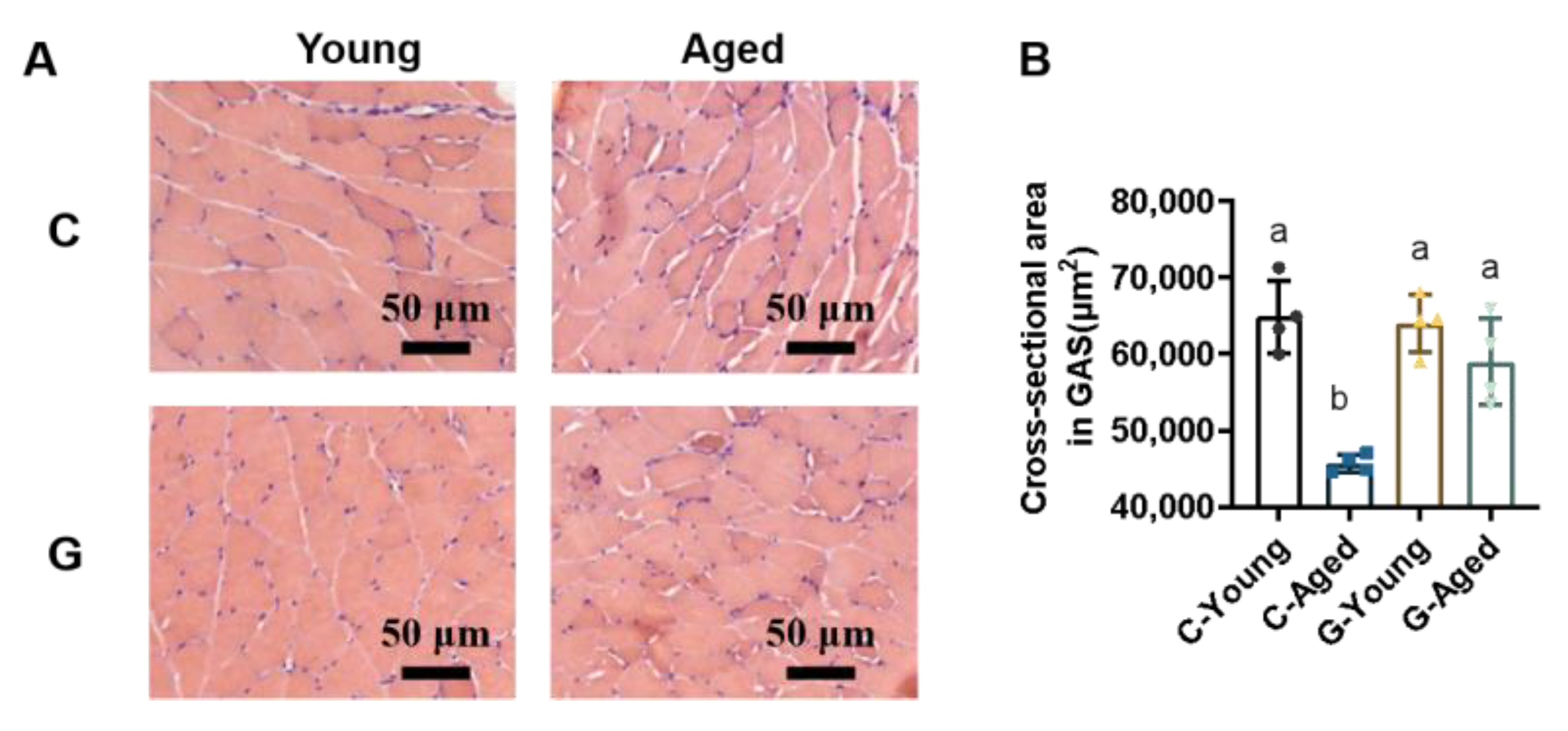
3.3. Effects of Long-Term LBW Consumption on Skeletal Muscle Mass in Mice
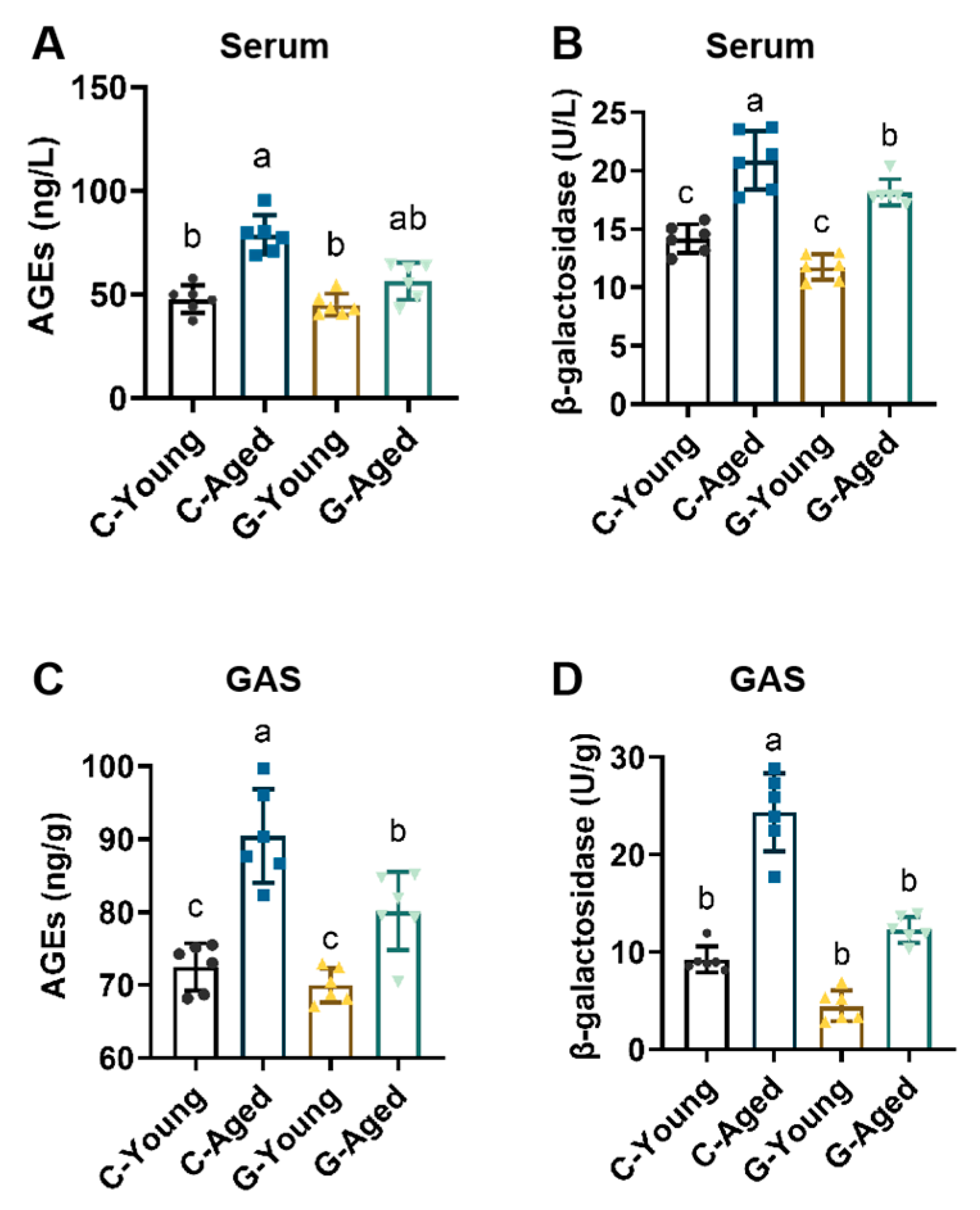
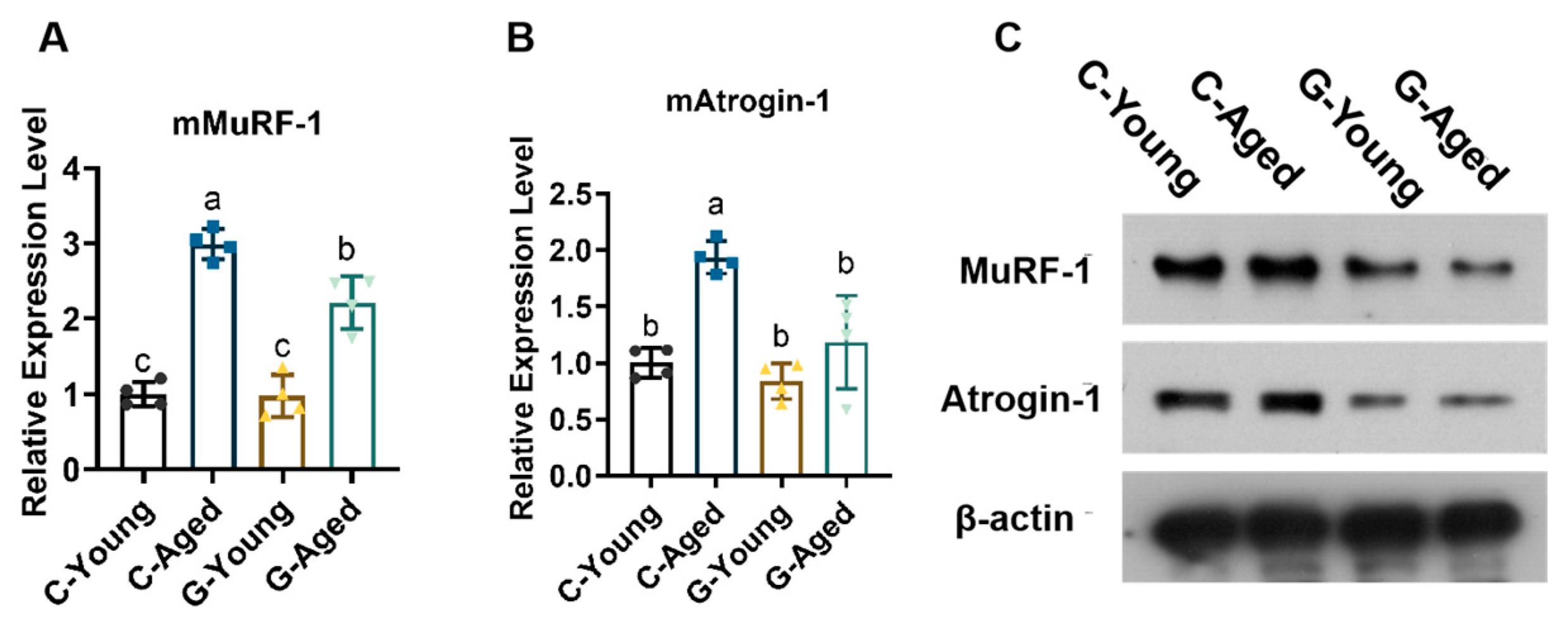
3.4. Mechanisms Underlying Effects on Aging-Related Factors
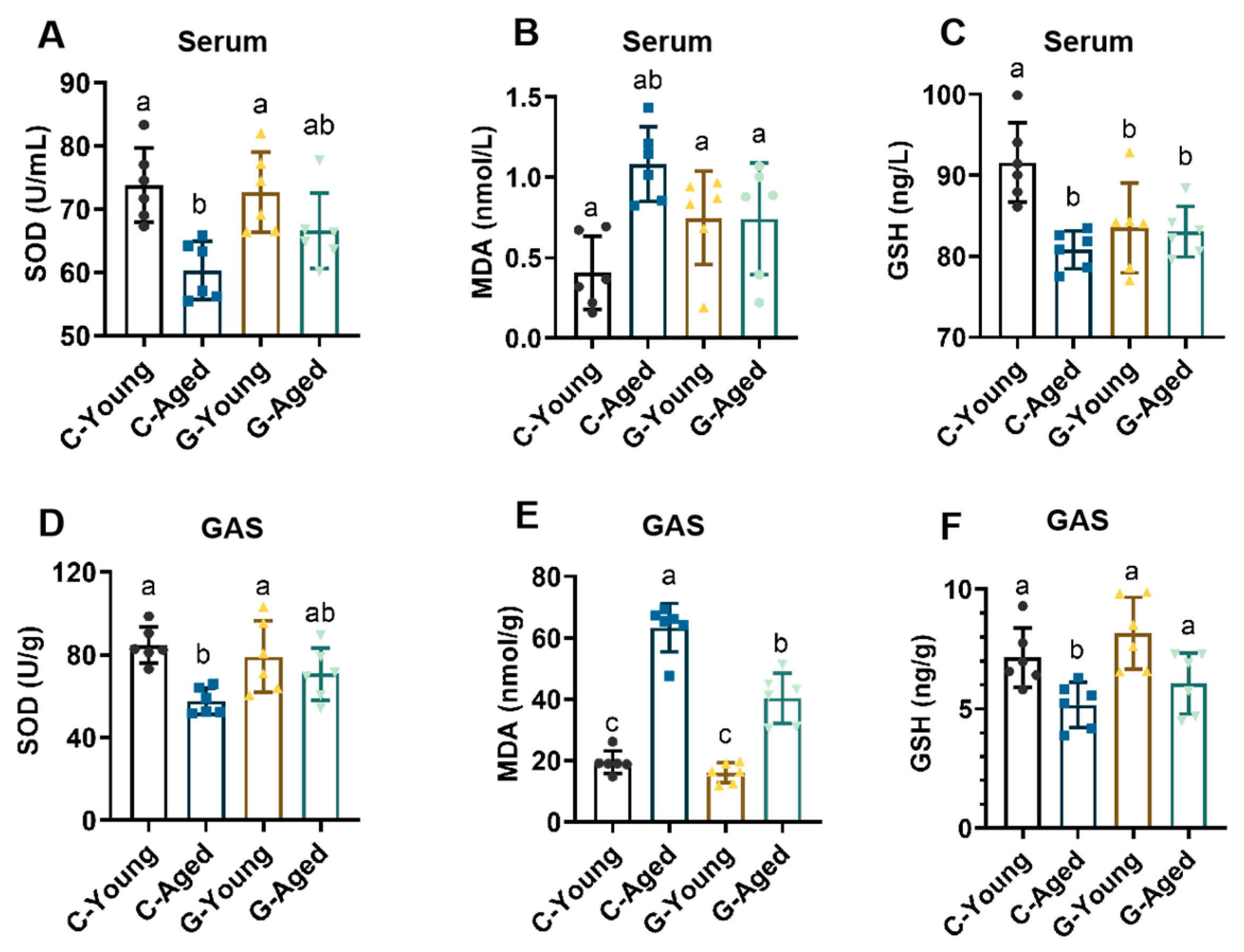
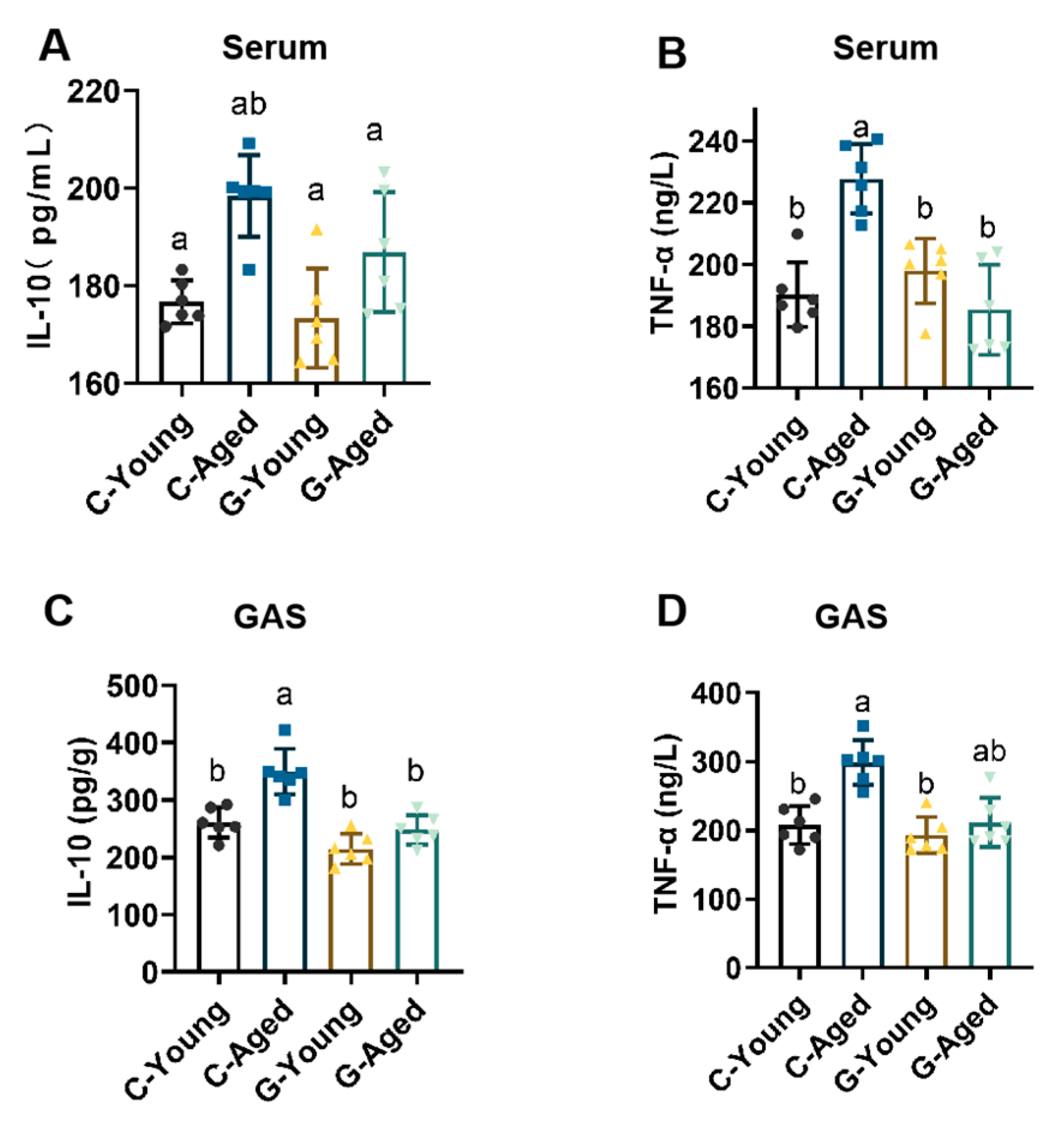
3.5. Mechanisms Underlying Effects on Oxidative Stress and Inflammation Levels
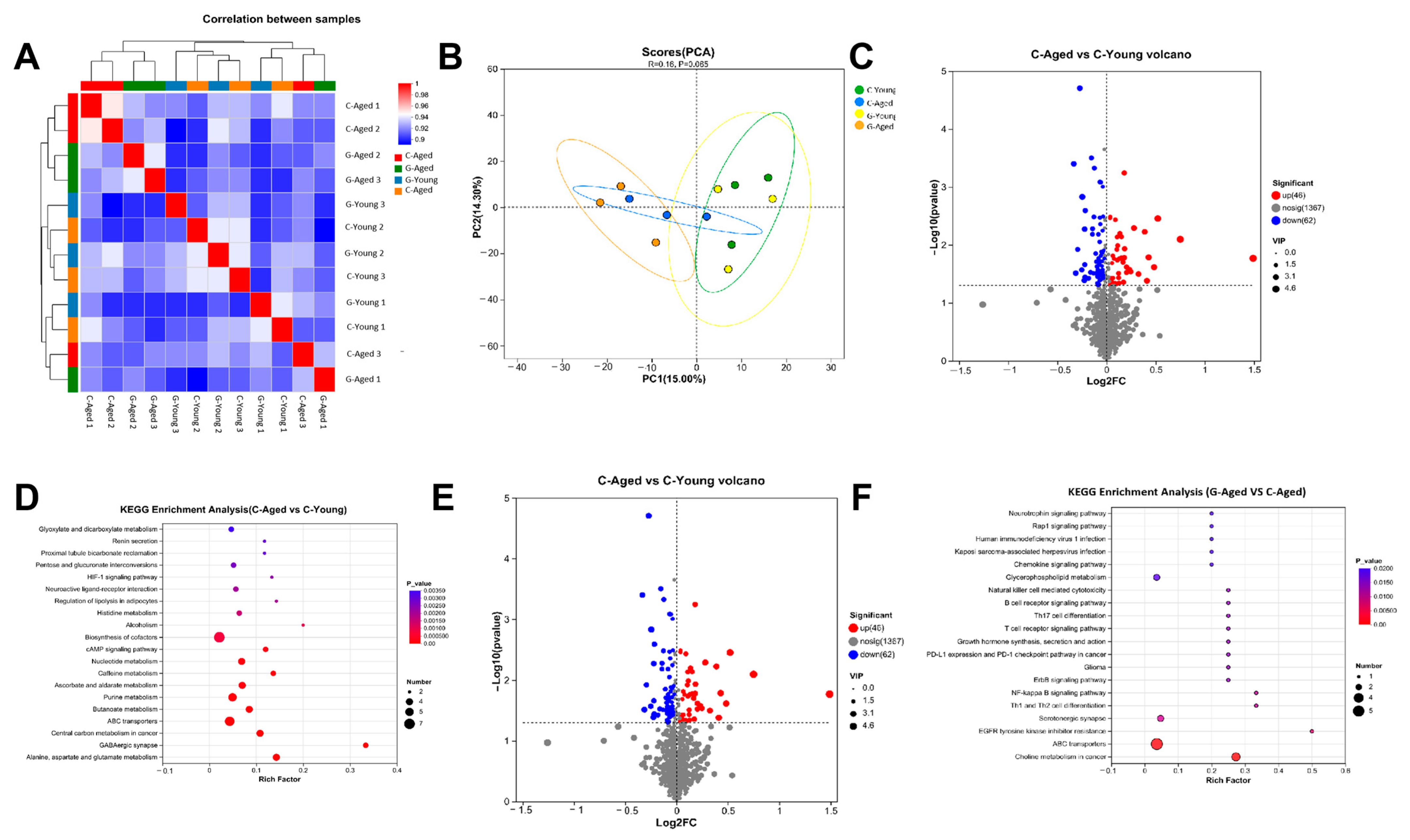
3.6. Metabolomics Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- da Costa, J.P.; Vitorino, R.; Silva, G.M.; Vogel, C.; Duarte, A.C.; Rocha-Santos, T. A synopsis on aging—Theories, mechanisms and future prospects. Ageing Res. Rev. 2016, 29, 90–112. [Google Scholar] [CrossRef]
- Navaneetham, K.; Arunachalam, D. Global Population Aging, 1950–2050. In Handbook of Aging, Health and Public Policy; Springer: Singapore, 2023; pp. 1–18. [Google Scholar] [CrossRef]
- Joo, S.K.; Kim, W. Interaction between sarcopenia and nonalcoholic fatty liver disease. Clin. Mol. Hepatol. 2023, 29, 68–78. [Google Scholar] [CrossRef]
- Chen, W.J.; Lin, I.H.; Lee, C.W.; Chen, Y.F. Aged skeletal muscle retains the ability to remodel extracellular matrix for degradation of collagen deposition after muscle injury. Int. J. Mol. Sci. 2021, 22, 2123. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.J.; Gries, K.J.; Marcotte, G.R.; Ryan, Z.; Strub, M.D.; Kunz, H.E.; Arendt, B.K.; Dasari, S.; Ebert, S.M.; Adams, C.M.; et al. Human myofiber-enriched aging-induced lncRNA FRAIL1 promotes loss of skeletal muscle function. Aging Cell 2024, 23, e14097. [Google Scholar] [CrossRef]
- Kedlian, V.R.; Wang, Y.; Liu, T.; Chen, X.; Bolt, L.; Tudor, C.; Shen, Z.; Fasouli, E.S.; Prigmore, E.; Kleshchevnikov, V.; et al. Human skeletal muscle aging atlas. Nat. Aging 2024, 4, 727–744. [Google Scholar] [CrossRef]
- Fang, Z.; Meng, Q.; Xu, J.; Wang, W.; Zhang, B.; Liu, J.; Liang, C.; Hua, J.; Zhao, Y.; Yu, X.; et al. Signaling pathways in cancer-associated fibroblasts: Recent advances and future perspectives. Cancer Commun. 2023, 43, 3–41. [Google Scholar] [CrossRef] [PubMed]
- Du, S.; Zheng, H. Role of FoxO transcription factors in aging and age-related metabolic and neurodegenerative diseases. Cell Biosci. 2021, 11, 188. [Google Scholar] [CrossRef]
- Petermann-Rocha, F.; Balntzi, V.; Gray, S.R.; Lara, J.; Ho, F.K.; Pell, J.P.; Celis-Morale, C. Global prevalence of sarcopenia and severe sarcopenia: A systematic review and meta-analysis. Cachexia Sarcopenia Muscle 2022, 13, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.K.; Lee, J.Y.; Gil, C.R.; Kim, M.K. Prevalence of sarcopenia in community-dwelling older adults according to simplified algorithms for sarcopenia consensus based on Asian working group for sarcopenia. Clin. Interv. Aging 2020, 15, 2291–2299. [Google Scholar] [CrossRef]
- Wang, F.; Li, W.; Chen, H.; Weil, R.R.; Zhu, L.; Nan, X.X. Forage Radish Cover Crops Improve Soil Quality and Fruit Yield of Lycium barbarum L. in an Arid Area of Northwest China. Agronomy 2023, 13, 1634. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Martorell, M.; Quetglas-Llabrés, M.M.; Sureda, A.; Mardones, L.; Villagran, M.; Gürer, E.S.; Živković, J.; Ezzat, S.M.; Zayed, A.; et al. Supercharging metabolic health with Lycium barbarum L.: A review of the therapeutic potential of this functional food for managing metabolic syndrome. Food Front. 2024, 5, 420–434. [Google Scholar] [CrossRef]
- Ma, R.H.; Zhang, X.X.; Thakur, K.; Zhang, J.G.; Wei, Z.J. Research progress of Lycium barbarum L. as functional food: Phytochemical composition and health benefits. Curr. Opin. Food Sci. 2022, 47, 100871. [Google Scholar] [CrossRef]
- Toh, D.W.K.; Zhou, H.; Cazenave-Gassiot, A.; Choi, H.; Burla, B.; Bendt, A.K.; Wenk, M.R.; Ling, L.H.; Kim, J.E. Effects of wolfberry (Lycium barbarum) consumption on the human plasma lipidome and its association with cardiovascular disease risk factors: A randomized controlled trial of middle-aged and older adults. Front. Nutr. 2024, 11, 1258570. [Google Scholar] [CrossRef]
- Zhou, X.L.; Xu, S.Y.; Zhang, Z.X.; Tang, M.M.; Meng, Z.T.; Peng, Z.; Liao, Y.X.; Yang, X.F.; Nüssler, A.K.; Liu, L.G.; et al. Gouqi-derived nanovesicles (GqDNVs) inhibited dexamethasone-induced muscle atrophy associating with AMPK/SIRT1/PGC1α signaling pathway. J. Nanobiotechnology 2024, 22, 276. [Google Scholar] [CrossRef]
- Peng, Y.F.; Zhao, L.L.; Hu, K.; Yang, Y.J.; Ma, J.; Zhai, Y.Q.; Jiang, Y.; Zhang, D.J. Anti-fatigue effects of Lycium barbarum polysaccharide and effervescent tablet by regulating oxidative stress and energy metabolism in rats. Int. J. Mol. Sci. 2022, 23, 10920. [Google Scholar] [CrossRef]
- Yang, C.; Yang, J.; Tan, L.; Tang, P.; Pen, T.; Gao, T.; Liu, S.; Guo, J. A novel formula comprising wolfberry, figs, white lentils, raspberries, and maca (WFWRM) induced antifatigue effects in a forced exercise mouse model. Evid. Based Complement. Altern. Med. 2022, 3784580. [Google Scholar] [CrossRef]
- Qi, Y.; Duan, G.; Fan, G.; Peng, N. Effect of Lycium barbarum polysaccharides on cell signal transduction pathways. Biomed. Pharmacother. 2022, 147, 112620. [Google Scholar] [CrossRef] [PubMed]
- He, N.W.; Yang, X.B.; Jiao, Y.D.; Tian, L.M.; Zhao, Y. Characterisation of antioxidant and antiproliferative acidic polysaccharides from Chinese Lycium barbarum fruits. Food Chem. 2012, 133, 978–989. [Google Scholar] [CrossRef]
- Zhao, C.; Zhao, H.; Zhang, C.C.; Yang, X.H.; Chen, K.; Xue, Y.; Li, Q.; Deng, S.Y.; Cai, H.Z. Impact of Lycium barbarum polysaccharide on the expression of glucagon-like peptide 1 in vitro and in vivo. Int. J. Biol. Macromol. 2023, 224, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Rajkowska, K.; Simińska, D.; Kunicka-Styczyńska, A. Bioactivities and Microbial Quality of Lycium Fruits (Goji) Extracts Derived by Various Solvents and Green Extraction Methods. Molecules 2022, 27, 7856. [Google Scholar] [CrossRef]
- Zhou, S.; Rahman, A.; Li, J.; Wei, C.; Chen, J.L.; Linhardt, R.J.; Ye, X.; Chen, S. Extraction methods affect the structure of goji (Lycium barbarum) polysaccharides. Molecules 2020, 25, 936. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Li, Z.; Huo, J.J.; Li, J.D.; Liu, F.H.; Yin, P. Effects of Chinese wolfberry and Astragalus extract on the antioxidant capacity of Tibetan pig liver. PLoS ONE 2021, 16, e0245749. [Google Scholar] [CrossRef] [PubMed]
- Bharti, R.; Thakral, A.; Thakur, A.; Sahu, B.; Verma, M.; Sharma, R.; Narwal, P.; Gupta, A.; Sharma, V.; Bansal, R. Innovative green synthesis and multifunctional applications of pyrimidopyrimidine derivatives. Curr. Org. Synth. 2025, 94, 769–789. [Google Scholar] [CrossRef]
- Ren, R.; Li, Y.; Chen, H.; Wang, Y.; Yang, L.; Su, C.; Zhao, X.; Chen, J.; Ma, X. Carotenoid contents of Lycium barbarum: A novel QAMS analyses, geographical origins discriminant evaluation, and storage stability assessment. Molecules 2021, 26, 5374. [Google Scholar] [CrossRef]
- Mitchell, S.J.; Madrigal-Matute, J.; Scheibye-Knudsen, M.; Fang, E.; Aon, M.; González-Reyes, J.A.; Cortassa, S.; Kaushik, S.; Gonzalez-Freire, M.; Patel, B.; et al. Effects of sex, strain, and energy intake on hallmarks of aging in mice. Cell Metab. 2016, 23, 1093–1112. [Google Scholar] [CrossRef]
- Jacobs, R.A.; Díaz, V.; Meinild, A.-K.; Gassmann, M.; Lundby, C. The C57Bl/6 mouse serves as a suitable model of human skeletal muscle mitochondrial function. Exp. Physiol. 2013, 98, 908–921. [Google Scholar] [CrossRef] [PubMed]
- Onishi, S.; Ishino, M.; Kitazawa, H.; Yoto, A.; Shimba, Y.; Mochizuki, Y.; Unno, K.; Meguro, S.; Tokimitsu, I.; Miura, S. Green tea extracts ameliorate high-fat diet–induced muscle atrophy in senescence-accelerated mouse prone-8 mice. PLoS ONE 2018, 13, e0195753. [Google Scholar] [CrossRef]
- Cutler, A.A.; Pawlikowski, B.; Wheeler, J.R.; Dalla Betta, N.; Elston, T.; O’Rourke, R.; Jones, K.; Olwin, B.B. The regenerating skeletal muscle niche drives satellite cell return to quiescence. iScience 2022, 25, 104444. [Google Scholar] [CrossRef] [PubMed]
- Geifman, N.; Rubin, E. The mouse age phenome knowledgebase and disease-specific inter-species age mapping. PLoS ONE 2013, 8, e81114. [Google Scholar] [CrossRef]
- Xia, Z.; Cholewa, J.; Zhao, Y.; Yang, Y.Q.; Shang, H.Y.; Guimarães-Ferreira, L.; Naimo, M.A.; Su, Q.S.; Zanchi, N.E. Hypertrophy-promoting effects of leucine supplementation and moderate intensity aerobic exercise in pre-senescent mice. Nutrients 2016, 8, 246. [Google Scholar] [CrossRef]
- Xie, W.Q.; He, M.; Yu, D.J.; Wu, Y.X.; Wang, X.H.; Lv, S.; Xiao, W.F.; Li, Y.S. Mouse models of sarcopenia: Classification and evaluation. Cachexia Sarcopenia Muscle 2021, 12, 538–554. [Google Scholar] [CrossRef]
- Lauretani, F.; Russo, C.R.; Bandinelli, S.; Bartali, B.; Cavazzini, C.; Di Iorio, A.; Corsi, A.M.; Rantanen, T.; Guralnik, J.M.; Ferrucci, L. Age-associated changes in skeletal muscles and their effect on mobility: An operational diagnosis of sarcopenia. J. Appl. Physiol. 2003, 95, 1851–1860. [Google Scholar] [CrossRef]
- Widjaja, J.H.; Sloan, D.C.; Hauger, J.A.; Muntean, B.S. Customizable open-source rotating rod (rotarod) enables robust low-cost assessment of motor performance in mice. eNeuro 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.W.; Sun, Q.R.; Fang, J.X.; Wang, C.T.; Wang, D.D.; Li, M. The anti-aging activity of Lycium barbarum polysaccharide extracted by yeast fermentation: In vivo and in vitro studies. Int. J. Biol. Macromol. 2022, 209, 2032–2041. [Google Scholar] [CrossRef]
- Pan, H.; Li, Y.; Qian, H.; Qi, X.; Wu, G.; Zhang, H.; Xu, M.; Rao, Z.; Li, J.L.; Wang, L.; et al. Effects of Geniposide from Gardenia Fruit Pomace on Skeletal-Muscle Fibrosis. J. Agric. Food Chem. 2018, 66, 5802–5811. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.Y.; Yu, J.; Zhang, Z.H.; Su, X.L.; Li, L.; Li, L.J. Controlling preanalytical process in high-coverage quantitative metabolomics: Spot-sample collection for mouse urine and fecal metabolome profiling. Anal. Chem. 2019, 91, 4958–4963. [Google Scholar] [CrossRef]
- Masci, A.; Carradori, S.; Casadei, M.A.; Paolicelli, P.; Petralito, S.; Ragno, R.; Cesa, S. Lycium barbarum polysaccharides: Extraction, purification, structural characterisation and evidence about hypoglycaemic and hypolipidaemic effects. A review. Food Chem. 2018, 245, 1234–1245. [Google Scholar] [CrossRef] [PubMed]
- Islam, H.; Chamberlain, T.C.; Mui, A.L. Elevated Interleukin-10 Levels in COVID-19: Potentiation of Pro-Inflammatory Responses or Impaired Anti-Inflammatory Action? Front. Immunol. 2021, 12, 677008. [Google Scholar] [CrossRef]
- Bae, T.; Hallis, S.P.; Kwak, M.K. Hypoxia, oxidative stress, and the interplay of HIFs and NRF2 signaling in cancer. Exp. Mol. Med. 2024, 56, 501–514. [Google Scholar] [CrossRef]
- Guo, Q.; Jin, Y.; Chen, X.; Ye, X.; Shen, X.; Lin, M.; Zeng, C.; Zhou, T.; Zhang, J. NF-κB in biology and targeted therapy: New insights and translational implications. Signal Transduct. Target. Ther. 2024, 9, 53. [Google Scholar] [CrossRef]
- McGettrick, A.F.; O’Neill, L.A.J. The role of HIF in immunity and inflammation. Cell Metab. 2020, 32, 524–536. [Google Scholar] [CrossRef]
- Ou, Y.W.; Huang, Y.H.; Yang, X.; Li, L.; Mei, R.S.; Yu, Z.X.; So, K.F.; Chen, J.S.; Ma, J.H.; Tang, S.B. Lycium barbarum glycopeptide alleviates retinal inflammation by suppressing microglial M1 polarization via NF-κB/MAPK pathways. Exp. Eye Res. 2025, 247, 110452. [Google Scholar] [CrossRef]
- Yi, M.; Li, T.; Niu, M.; Zhang, H.; Wu, Y.; Wu, K.; Dai, Z. Targeting cytokine and chemokine signaling pathways for cancer therapy. Signal Transduct. Target. Ther. 2024, 9, 176. [Google Scholar] [CrossRef]
- Demontis, F.; Piccirillo, R.; Goldberg, A.L.; Perrimon, N. The influence of skeletal muscle on systemic aging and lifespan. Aging Cell 2013, 12, 943–949. [Google Scholar] [CrossRef]
- Laumonier, T.; Menetrey, J. Muscle injuries and strategies for improving their repair. Exp. Orthop. 2016, 3, 15. [Google Scholar] [CrossRef]
- Orchard, J.W.; Jomaa, M.C.; Orchard, J.J.; Rae, K.; Hoffman, D.T.; Reddin, T.; Driscoll, T. Fifteen-week window for recurrent muscle strains in football: A prospective cohort of 3600 muscle strains over 23 years in professional Australian rules football. Br. J. Sports Med. 2020, 54, 1103–1107. [Google Scholar] [CrossRef] [PubMed]
- Lucas, L.; Cooper, T.A. Insights into Cell-Specific Functions of Microtubules in Skeletal Muscle Development and Homeostasis. Int. J. Mol. Sci. 2023, 24, 2903. [Google Scholar] [CrossRef] [PubMed]
- Penniman, C.M.; Bhardwaj, G.; Nowers, C.J.; Brown, C.U.; Junck, T.L.; Boyer, C.K.; Jena, J.; Fuqua, J.D.; Lira, V.A.; O’Neill, B.T. Loss of FoxOs in muscle increases strength and mitochondrial function during aging. J. Cachexia Sarcopenia Muscle 2022, 13, 1825–1840. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.; Kausar, M.A.; Singh, R.; Siddiqui, A.J.; Akhter, A. The role of glyoxalase in glycation and carbonyl stress induced metabolic disorders. Curr. Protein Pept. Sci. 2020, 21, 846–859. [Google Scholar] [CrossRef]
- García-Prat, L.; Martínez-Vicente, M.; Perdiguero, E.; Ortet, L.; Rodríguez-Ubreva, J.; Rebollo, E.; Ruiz-Bonilla, V.; Gutarra, S.; Ballestar, E.; Serrano, A.L.; et al. Autophagy maintains stemness by preventing senescence. Nature 2021, 591, 45–51. [Google Scholar] [CrossRef][Green Version]
- Maldonado, E.; Morales-Pison, S.; Urbina, F.; Solari, A. Aging hallmarks and the role of oxidative stress. Antioxidants 2023, 12, 651. [Google Scholar] [CrossRef] [PubMed]
- Pawelec, G.; Goldeck, D.; Derhovanessian, E. Inflammation, ageing and chronic disease. Curr. Opin. Immunol. 2014, 29, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Dang, B.R.; Hu, L.M.; Zhu, S.; Liu, Z.H.; Cao, X.H.; Li, Z.; Wang, C.M.; Lin, C.J. Lycium barbarum polysaccharide inhibits blue-light-induced skin oxidative damage with the involvement of mitophagy. Phytother. Res. 2023, 37, 4055–4067. [Google Scholar] [CrossRef] [PubMed]
| Group | Age | Intervention Methods | Numbers of Animals |
|---|---|---|---|
| C-Young | 8 Weeks | Drinking Water | 8 |
| C-Aged | ≥18 Months | Drinking Water | 8 |
| G-Young | 8 Weeks | LBW | 8 |
| G-Aged | ≥18 Months | LBW | 8 |
| Gene | Forward Primer (5′-3′) | Reverse Primer (5′-3′) |
|---|---|---|
| 18S | ACCGCAGCTAGGAATAATGGA | CAAATGCTTTCGCTCTGGTC |
| mAtrogin-1 | CGCCACTCCGGGACATAG | GAAGTCGTCTGCTGTCTCAAAGG |
| mMuRF-1 | GAGACATCCCCCTATTTCTACCA | GCTCAGTCCGCTCATAGCC |
| Free Radical | 1:2 | 1:5 | 1:10 | 1:20 |
|---|---|---|---|---|
| DPPH clearance activity (%) | 42.67 ± 5.59 de | 57.57 ± 3.43 c | 74.94 ± 3.29 a | 78.25 ± 6.82 a |
| ABTS free radical scavenging activity (%) | 37.78 ± 2.40 e | 42.73 ± 3.29 e | 46.27 ± 4.99 d | 48.27 ± 5.01 d |
| Hydroxyl radical scavenging clearance (%) | 35.40 ± 4.01 e | 54.82 ± 6.77 cd | 63.22 ± 4.21 bc | 71.75 ± 6.82 ab |
| Free Radical | 40 °C | 60 °C | 80 °C | 100 °C |
|---|---|---|---|---|
| DPPH clearance activity (%) | 63.60 ± 4.26 b | 66.90 ± 6.11 ab | 72.79 ± 3.16 a | 50.76 ± 6.35 c |
| ABTS free radical scavenging activity (%) | 47.30 ± 1.32 d | 50.30 ± 6.19 cd | 63.38 ± 2.15 b | 46.58 ± 1.91 d |
| Hydroxyl radical clearance (%) | 39.70 ± 3.11 e | 61.85 ± 4.57 b | 67.40 ± 4.39 a | 59.70 ± 3.27b c |
| Component Name | LBW |
|---|---|
| Polysaccharide content (mg GLU/g) | 71.81 ± 2.76 |
| Anthocyanin content (mg/100 g) | 21.13 ± 3.52 |
| Total phenol content (mg GAE/g) | 18.80 ± 1.14 |
| Total flavonoid content (mg CAE/g) | 2.37 ± 0.39 |
| Carotenoid content (mg/100 g) | 9.08 ± 1.22 |
| Betaine content (mg/100 g) | 41.31 ± 6.82 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, Y.; Zheng, Q.; Wang, J.; Fan, M.; Qian, H.; Wang, L.; Li, Y. Effects and Mechanisms of Long-Term Lycium barbarum Water Consumption on Skeletal Muscle Function in Aged Mice. Foods 2025, 14, 3049. https://doi.org/10.3390/foods14173049
Tang Y, Zheng Q, Wang J, Fan M, Qian H, Wang L, Li Y. Effects and Mechanisms of Long-Term Lycium barbarum Water Consumption on Skeletal Muscle Function in Aged Mice. Foods. 2025; 14(17):3049. https://doi.org/10.3390/foods14173049
Chicago/Turabian StyleTang, Yundi, Qingwei Zheng, Jinyi Wang, Mingcong Fan, Haifeng Qian, Li Wang, and Yan Li. 2025. "Effects and Mechanisms of Long-Term Lycium barbarum Water Consumption on Skeletal Muscle Function in Aged Mice" Foods 14, no. 17: 3049. https://doi.org/10.3390/foods14173049
APA StyleTang, Y., Zheng, Q., Wang, J., Fan, M., Qian, H., Wang, L., & Li, Y. (2025). Effects and Mechanisms of Long-Term Lycium barbarum Water Consumption on Skeletal Muscle Function in Aged Mice. Foods, 14(17), 3049. https://doi.org/10.3390/foods14173049








