Improvement of Quality of Sour Camel Milk by Extract of Sparassis crispa: Physicochemical Properties, Sensory Quality and Metabolic Changes
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain Activation
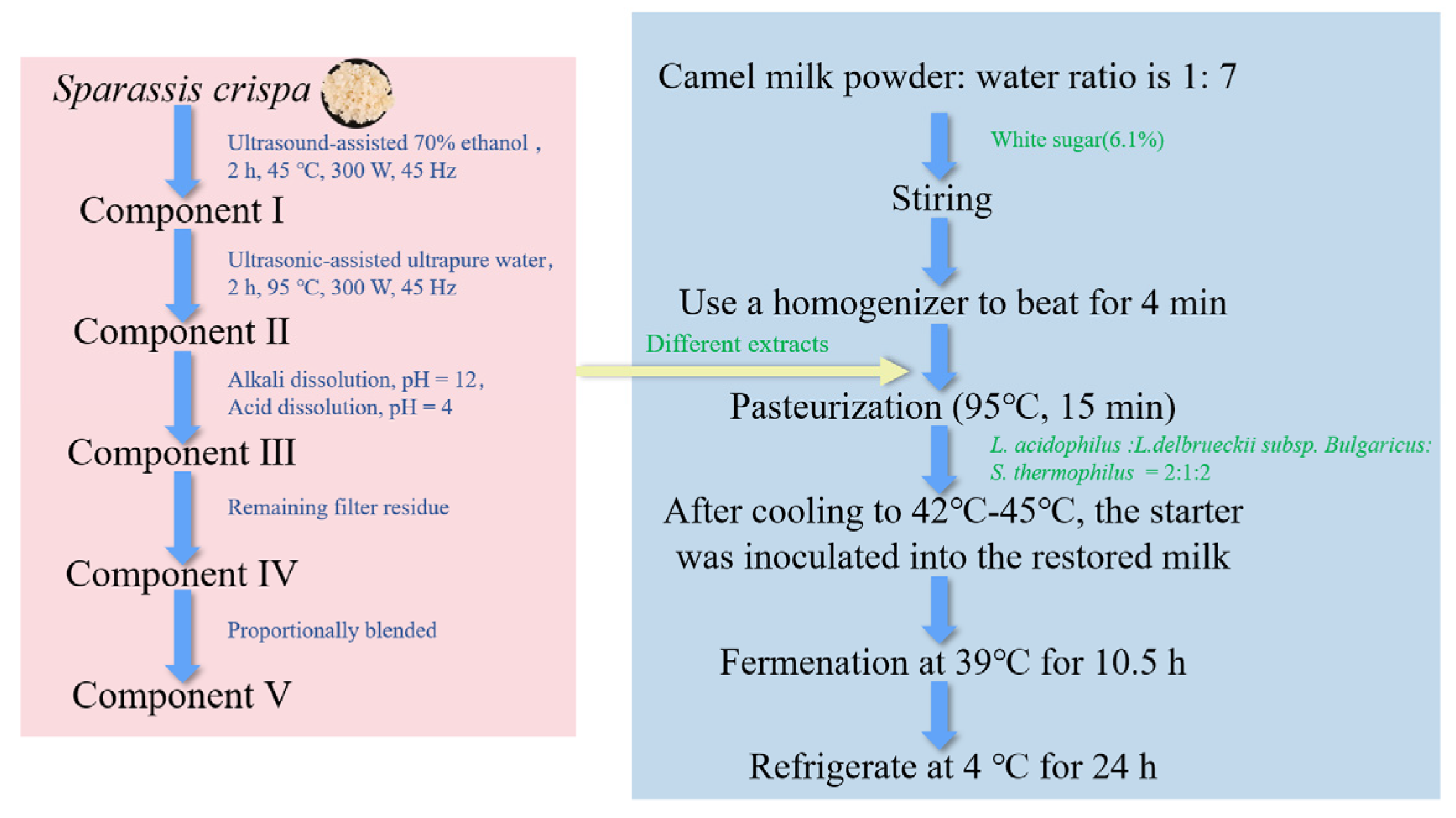
2.2. Preparation of Sour Camel Milk
2.3. Determination of Cell Counts
2.4. Determination of TA, pH, and WHC
2.5. Determination of Nutritional Quality
2.6. Textural Analysis
2.7. Rheological Analysis
2.8. Sensory Evaluation
2.9. Identification of Component Ⅰ Substances
2.10. Determination of Volatile Flavor Compounds
2.11. Determination of Non-Volatile Flavor Compounds
2.12. Statistical Analysis
3. Results
3.1. Physicochemical Properties of Sour Camel Milk
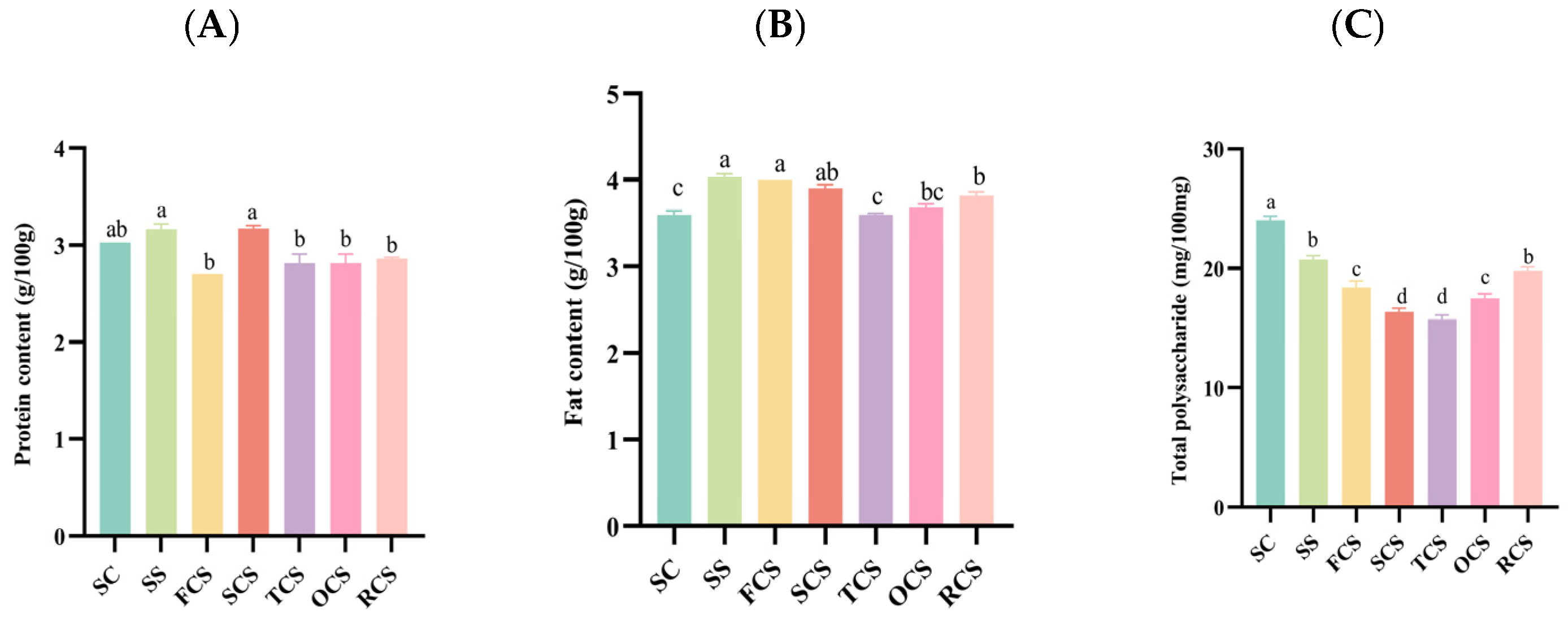
3.2. Nutritional Quality Analysis of Sour Camel Milk
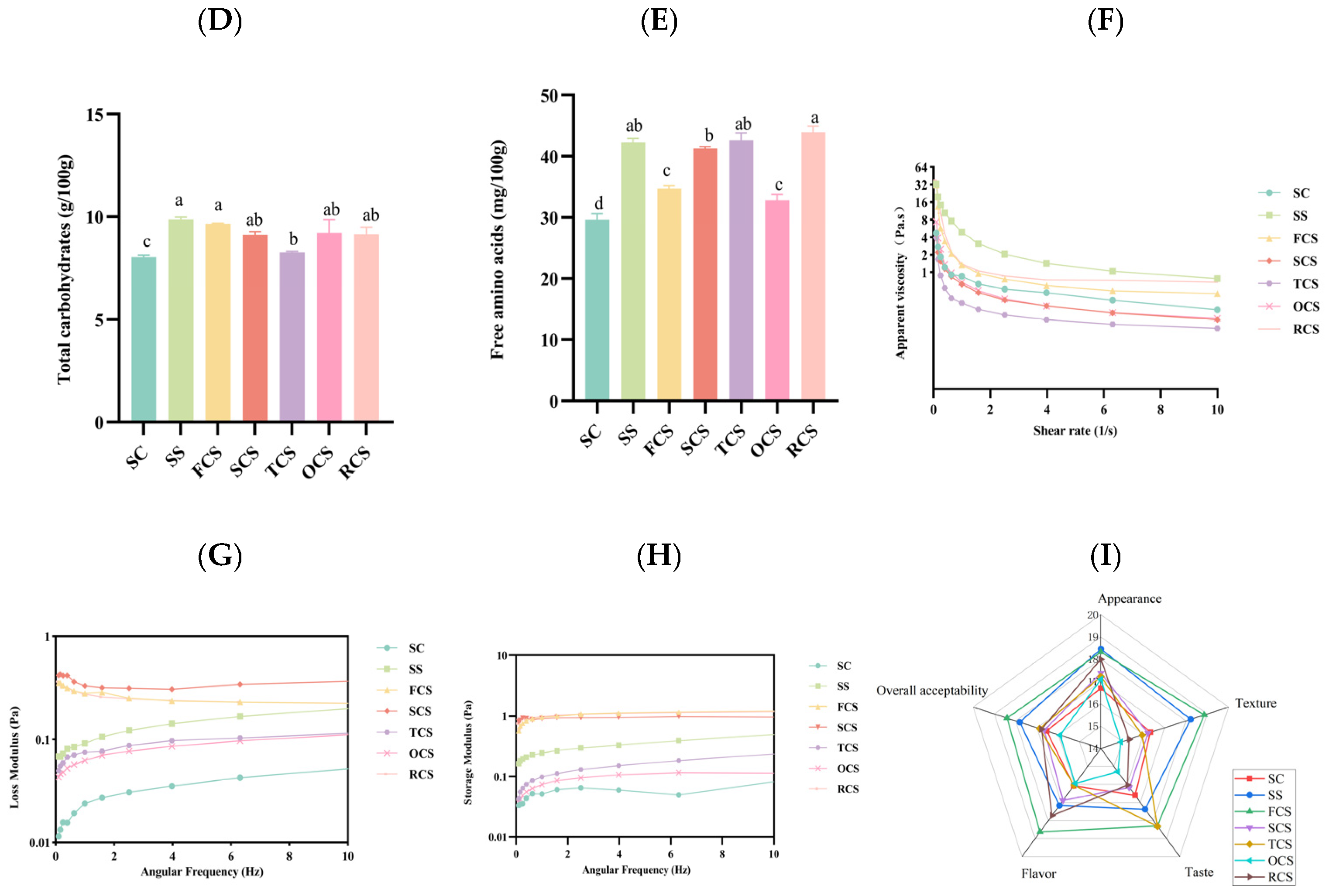
3.3. Analysis of the Textural Composition of Sour Camel Milk
3.4. Rheological Compositional Analysis of Sour Camel Milk
3.5. Sensory Score Analysis of Sour Camel Milk
3.6. Component I Substance Identification Analysis
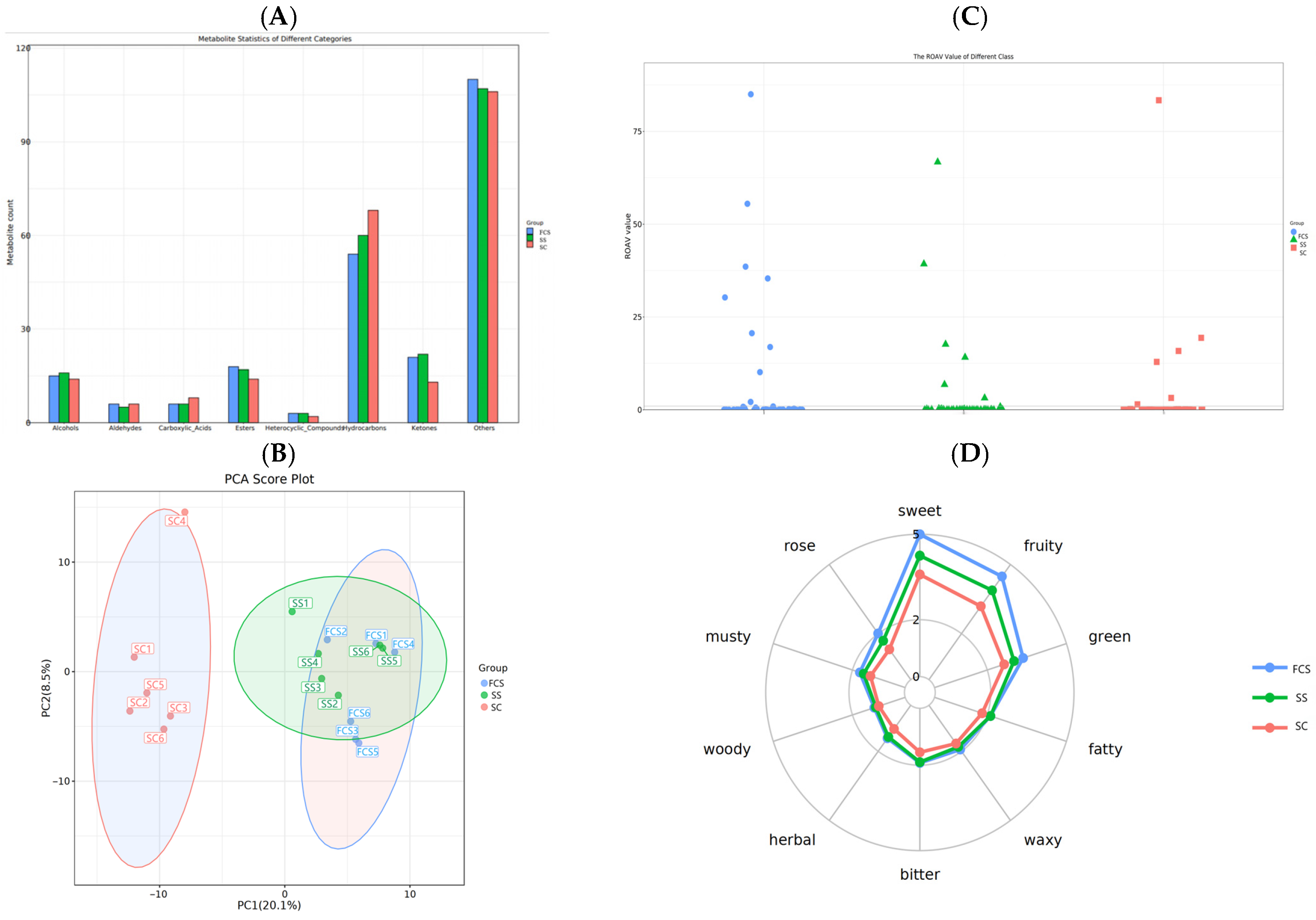
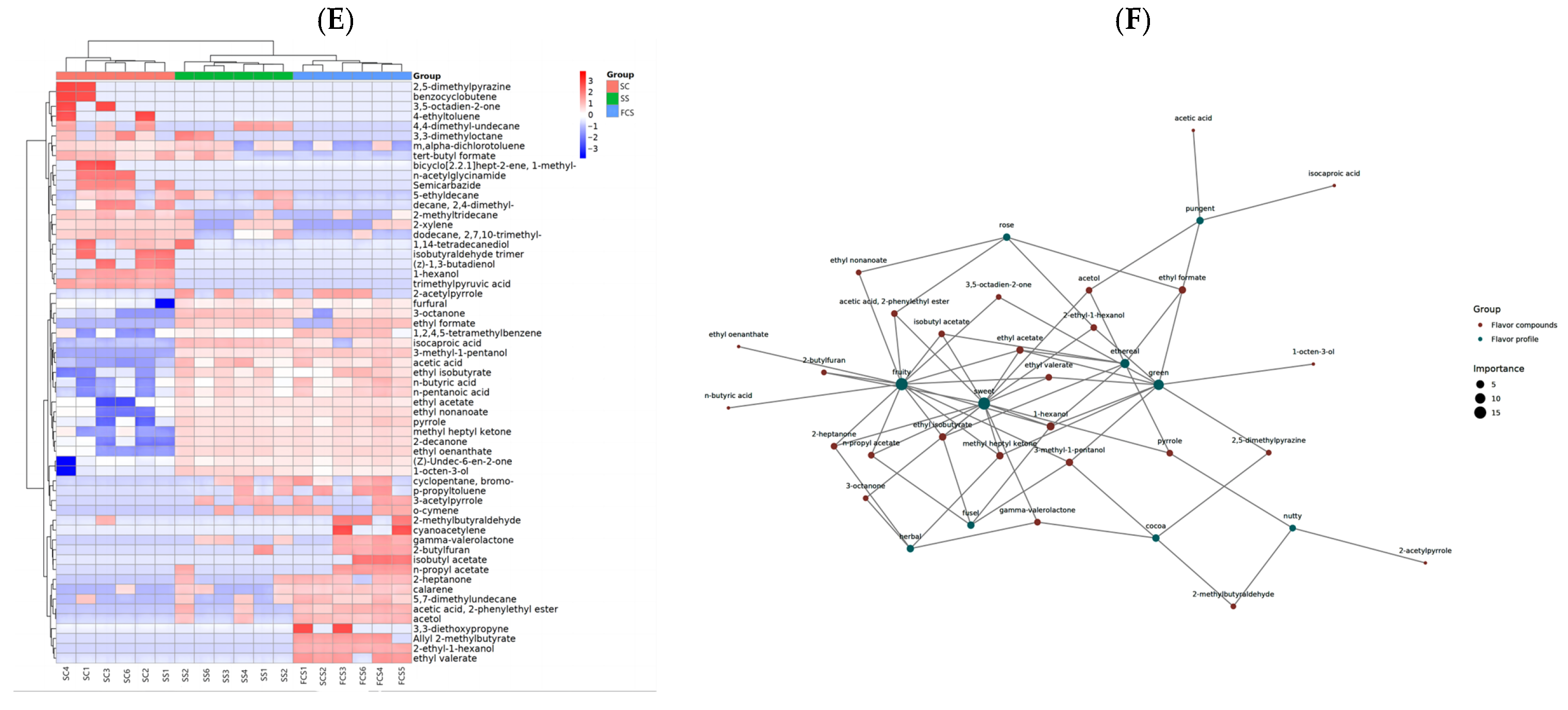
3.7. Analysis of the Flavor Metabolites in Sour Camel Milk
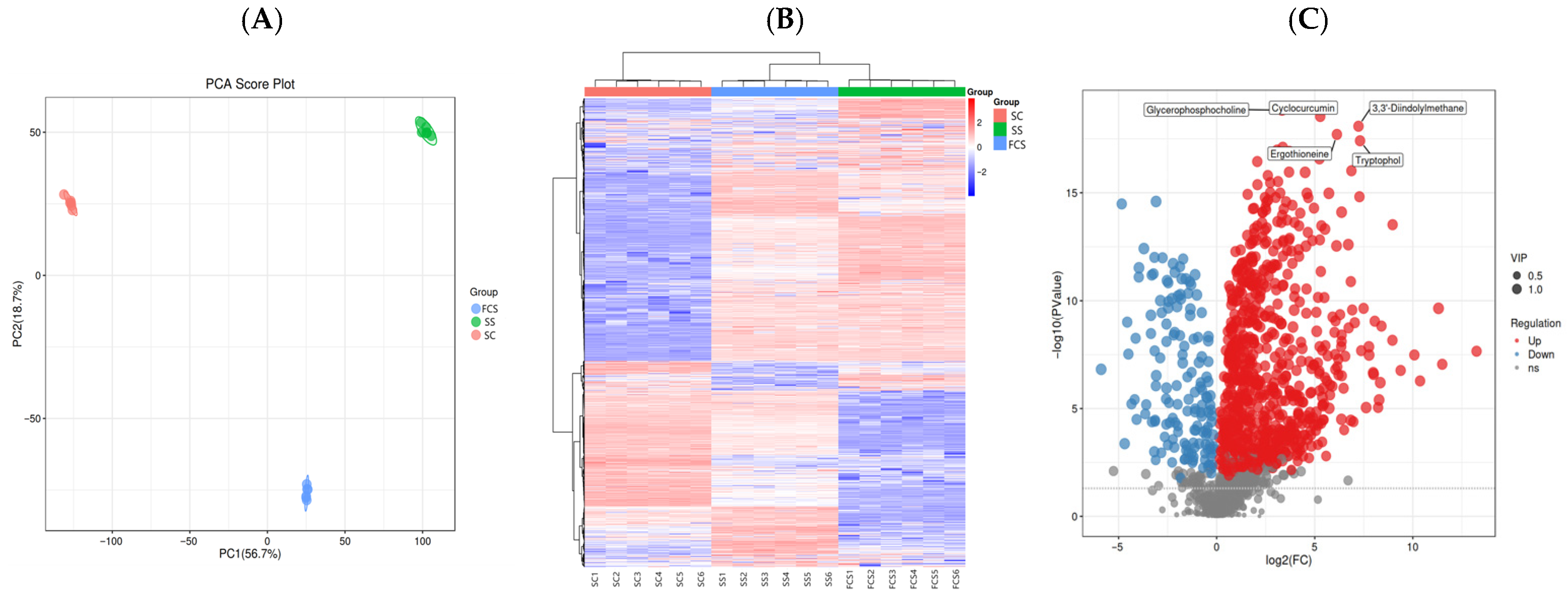
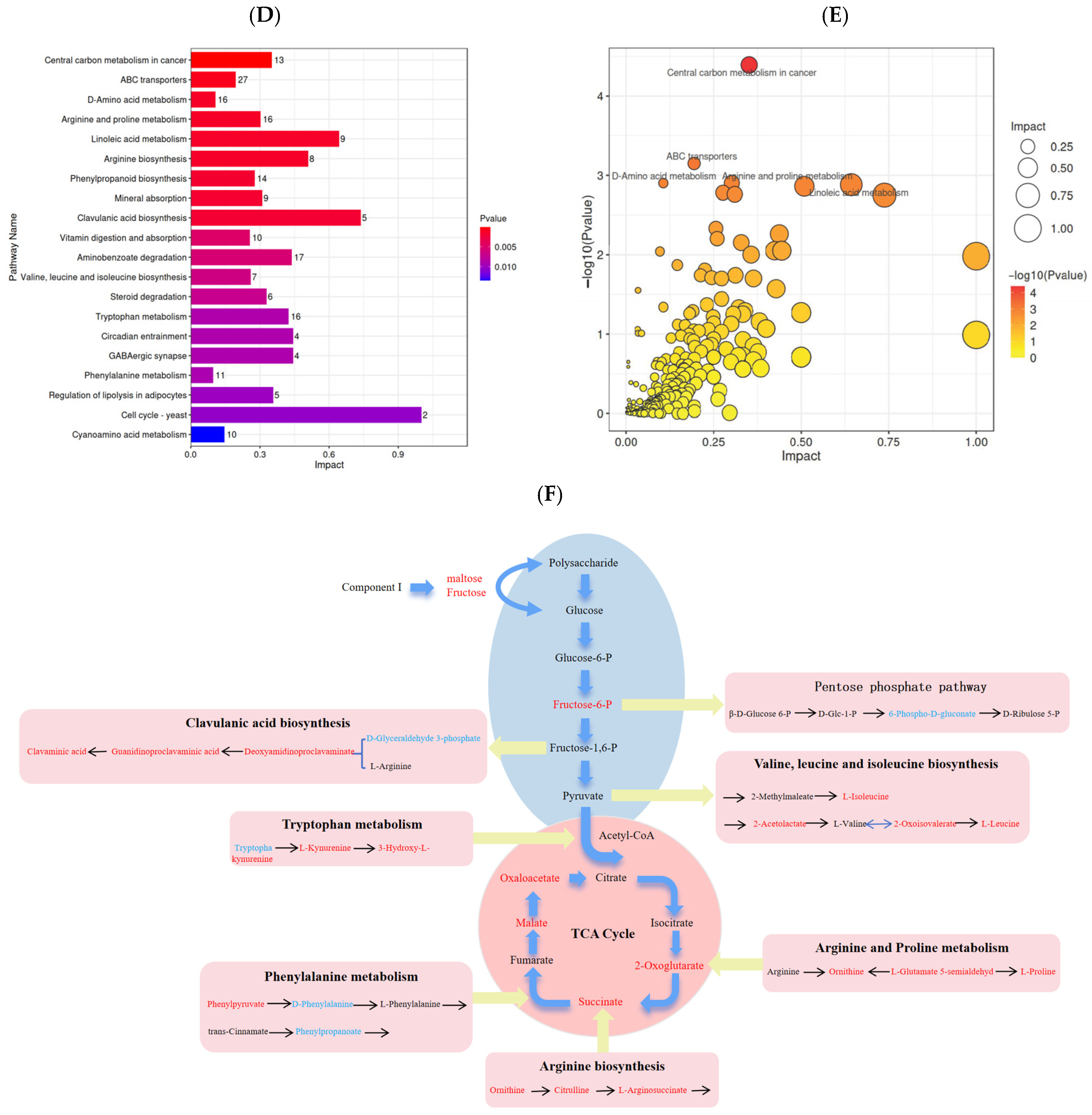
3.8. Analysis of the Non-Volatile Flavor Metabolites in Sour Camel Milk
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Xu, W.; Dong, Q.; Zhao, G.; Han, B. Analysis of metabolites of bactrain camel milk in Alxa of China before and after fermentation with fermenting agent TR1 based on untargeted LC-MS/MS based metabolomics. Heliyon 2023, 9, e18522. [Google Scholar] [CrossRef] [PubMed]
- Aili, T.; Xu, Z.; Liu, C.; Yang, J.; Yue, H. Camel milk and D-allulose synergistically improved camel dairy flavor and alleviated insulin resistance of human HepG2 cells. Heliyon 2025, 11, e41825. [Google Scholar] [CrossRef] [PubMed]
- Nongonierma, A.B.; Paolella, S.; Mudgil, P.; Maqsood, S.; FitzGerald, R.J. Identification of novel dipeptidyl peptidase IV (DPP-IV) inhibitory peptides in camel milk protein hydrolysates. Food Chem. 2018, 244, 340–348. [Google Scholar] [CrossRef] [PubMed]
- Bilal, Z.; Akhmetsadykova, S.; Baubekova, A.; Tormo, H.; Faye, B.; Konuspayeva, G. The Main Features and Microbiota Diversity of Fermented Camel Milk. Foods 2024, 13, 1985. [Google Scholar] [CrossRef]
- Mbye, M.; Ali, A.H.; Kamal-Eldin, A.; Banat, F. The impact of camel milk and its products on diabetes mellitus management: A review of bioactive components and therapeutic potential. NFS J. 2025, 38, 100204. [Google Scholar] [CrossRef]
- Khalifa, A.; Ibrahim, H.I.M.; Sheikh, A.; Khalil, H.E. Probiotic-Fermented Camel Milk Attenuates Neurodegenerative Symptoms via SOX5/miR-218 Axis Orchestration in Mouse Models. Pharmaceuticals 2023, 16, 357. [Google Scholar] [CrossRef]
- Singh, S.; Mann, S.; Kalsi, R.; Singh, S.; Taneja, N.K.; Oberoi, H.S.; Chauhan, K.; Taneja, P. Exploring the therapeutic and nutritional potential of camel milk: Challenges and prospects: A comprehensive review. Appl. Food Res. 2024, 4, 100622. [Google Scholar] [CrossRef]
- Sobti, B.; Mustapha, M.; Huda, A.; Alya, A.; Aysha, A.; Muna, A.; Haleimah, S. Tholkappiyan Ramachandran, Fathalla Hamed, and Afaf Kamal-Eldin. Rheological characteristics and consumer acceptance of camel milk yogurts as affected by bovine proteins and hydrocolloids. Int. J. Food Prop. 2020, 23, 1347–1360. [Google Scholar] [CrossRef]
- Liu, C.; Liu, L.-X.; Yang, J.; Liu, Y.-G. Exploration and analysis of the composition and mechanism of efficacy of camel milk. Food Biosci. 2023, 53, 102564. [Google Scholar] [CrossRef]
- Elkot, W.F.; Rayan, A.M.; Malek, G.; Alsulami, T.; Elmahdy, A.; AL-Farga, A.; Abu-Taha, H.L.; Tantawy, A.A. Assessment of using heart of date palm as a new source of protein and carbohydrate on the quality of low-fat bio fermented camel milk and its potential anticancer properties. Int. J. Biol. Macromol. 2023, 282, 137174. [Google Scholar] [CrossRef] [PubMed]
- Törős, G.; Béni, Á.; Peles, F.; Gulyás, G.; Prokisch, J. Comparative Analysis of Freeze-Dried Pleurotus ostreatus Mushroom Powders on Probiotic and Harmful Bacteria and Its Bioactive Compounds. J. Fungi. 2024, 11, 1. [Google Scholar] [CrossRef]
- Kumar, H.; Bhardwaj, K.; Sharma, R.; Nepovimova, E.; Cruz-Martins, N.; Dhanjal, D.S.; Singh, R.; Chopra, C.; Verma, R.; Abd-Elsalam, K.A.; et al. Potential Usage of Edible Mushrooms and Their Residues to Retrieve Valuable Supplies for Industrial Applications. J. Fungi. 2021, 7, 427. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, D.; Ning, Q.; Wang, J. Growth characteristics and metabonomics analysis of Lactobacillus rhamnosus GG in Ganoderma lucidum aqueous extract medium. Food Biosci. 2023, 53, 102486. [Google Scholar] [CrossRef]
- Akin, Z.; Ozcan, T. Functional properties of fermented milk produced with plant proteins. LWT 2017, 86, 25–30. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, C.; Wang, B.; Ge, Y.; Jiang, G.; Tang, Z.; Song, Y. Research on the Fermentation Cultivation of Long-type Bifidobacterium by Polyene Polysaccharide from Hydrangium japonicum. Food Eng. 2024, 3, 76–80. [Google Scholar]
- Wei, X.; Cheng, F.; Liu, J.; Cheng, Y.; Yun, S.; Meng, J.; Chang, M.; Geng, X.; Xu, L.; Cao, J.; et al. Sparassis latifolia polysaccharides inhibit colon cancer in mice by modulating gut microbiota and metabolism. Int. J. Biol. Macromol. 2023, 232, 123299. [Google Scholar] [CrossRef]
- Lan, M.; Weng, M.; Lin, Z.; Wang, J.; Zhao, F.; Qiu, B. Metabolomic analysis of antimicrobial mechanism of polysaccharides from Sparassis crispa based on HPLC-Q-TOF/MS. Carbohydr. Res. 2021, 503, 108299. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Nishioka, J.; Suzuki, K. Innate immune activation and antitumor effects of Lactobacillus-fermented Sparassis crispa extract in mice. J. Funct. Foods 2020, 75, 104215. [Google Scholar] [CrossRef]
- Aoki, R.; Watanabe, Y.; Sakai, Y.; Furukawa, M.; Shigetomi, T.; Jiun Rong, C.; Izumo, N. Wood cauliflower mushroom (Sparassis crispa) suppresses the body weight and visceral fat increased by ovariectomy in mice. Curr. Res. Food Sci. 2024, 8, 100713. [Google Scholar] [CrossRef] [PubMed]
- GB 4789.35-2016; National Food Safety Standard—Microbiological Examination of Food—Examination of Lactic Acid Bacteria. China Standards Press: Beijing, China, 2016.
- GB 5009.239-2016; National Food Safety Standard—Determination of Acidity in Food. Standards Press of China: Beijing, China, 2016.
- Martin, K.R.; Moré, M.; Hipólito, J.; Charlemagne, S.; Schlumpberger, B.O.; Raguso, R.A. Spatial and temporal variation in volatile composition suggests olfactory division of labor within the trap flowers of Aristolochia gigantea. Flora 2017, 232, 153–168. [Google Scholar] [CrossRef]
- Santos, G.B.M.; Dionísio, A.P.; Magalhães, H.C.R.; de Abreu, F.A.P.; Lira, S.M.; de Lima, A.C.V.; da Silva, G.S.; Guedes, J.A.C.; Araujo, I.M.d.S.; Artur, A.G.; et al. Effects of processing on the chemical, physicochemical, enzymatic, and volatile metabolic composition of pitaya. Food Res. Int. 2020, 127, 108710. [Google Scholar] [CrossRef] [PubMed]
- Dunn, W.B.; Broadhurst, D.; Begley, P.; Zelena, E.; Francis-McIntyre, S.; Anderson, N.; Brown, M.; Knowles, J.D.; Halsall, A.; Haselden, J.N.; et al. Human Serum Metabolome (HUSERMET) Consortium. Procedures for large-scale metabolic profiling of serum and plasma using gas chromatography and liquid chromatography coupled to mass spectrometry. Nat. Protoc. 2011, 6, 1060–1083. [Google Scholar] [CrossRef] [PubMed]
- Zelena, E.; Dunn, W.B.; Broadhurst, D.; Francis-McIntyre, S.; Carroll, K.M.; Begley, P.; O’Hagan, S.; Knowles, J.D.; Halsall, A.; Wilson, I.D.; et al. Development of a robust and repeatable UPLC-MS method for the long-term metabolomic study of human serum. Anal. Chem. 2009, 81, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Want, E.J.; Masson, P.; Michopoulos, F.; Wilson, I.D.; Theodoridis, G.; Plumb, R.S.; Shockcor, J.; Loftus, N.; Holmes, E.; Nicholson, J.K. Global metabolic profiling of animal and human tissues via UPLC-MS. Nat. Protoc. 2013, 8, 17–32. [Google Scholar] [CrossRef]
- Peng, C.; Yao, G.; Sun, Y.; Guo, S.; Wang, J.; Mu, X.; Sun, Z.; Zhang, H. Comparative effects of the single and binary probiotics of Lacticaseibacillus casei Zhang and Bifidobacterium lactis V9 on the growth and metabolomic profiles in yogurts. Food Res. Int. 2022, 152, 110603. [Google Scholar] [CrossRef]
- Aida, F.M.N.A.; Shuhaimi, M.; Yazid, M.; Maaruf, A.G. Mushroom as a potential source of prebiotics: A review. Trends Food Sci. Technol. 2009, 20, 567–575. [Google Scholar] [CrossRef]
- MacDonald, A.; Singh, R.H.; Rocha, J.C.; van Spronsen, F.J. Optimising amino acid absorption: Essential to improve nitrogen balance and metabolic control in phenylketonuria. Nutr. Res. Rev. 2019, 32, 70–78. [Google Scholar] [CrossRef]
- Liao, Y.; Chen, Q.; Liu, L.; Huang, H.; Sun, J.; Bai, X.; Jin, C.; Li, H.; Sun, F.; Xiao, X.; et al. Amino acid is a major carbon source for hepatic lipogenesis. Cell Metab. 2024, 36, 2437–2448. [Google Scholar] [CrossRef]
- Li, S.; Zeng, H.; Fan, J.; Wang, F.; Xu, C.; Li, Y.; Tu, J.; Nephew, K.P.; Long, X. Glutamine metabolism in breast cancer and possible therapeutic targets. Biochem. Pharmacol. 2023, 210, 115464. [Google Scholar] [CrossRef]
- Zhang, G.-F.; Jensen, M.V.; Gray, S.M.; El, K.; Wang, Y.; Lu, D.; Becker, T.C.; Campbell, J.E.; Newgard, C.B. Reductive TCA cycle metabolism fuels glutamine- and glucose-stimulated insulin secretion. Cell Metab. 2021, 33, 804–817. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Xie, J.; Pan, Y. System sensory analysis of yogurt based on texture analyzer. J. Dairy Sci. 2024, in press. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, S.; Shen, Q.; Zhu, M.-J. A metabolomic explanation on beneficial effects of dietary Goji on intestine inflammation. J. Funct. Foods 2019, 53, 109–114. [Google Scholar] [CrossRef]
- Zhang, P.; Liu, J.; Xiong, B.; Zhang, C.; Kang, B.; Gao, Y.; Li, Z.; Ge, W.; Cheng, S.; Hao, Y.; et al. Microbiota from alginate oligosaccharide-dosed mice successfully mitigated small intestinal mucositis. Microbiome 2020, 8, 112. [Google Scholar] [CrossRef]
- Zhang, P.; Tang, F.; Cai, W.; Zhao, X.; Shan, C. Evaluating the effect of lactic acid bacteria fermentation on quality, aroma, and metabolites of chickpea milk. Front. Nutr. 2022, 9, 1069714. [Google Scholar] [CrossRef] [PubMed]
- Settachaimongkon, S.; Nout, M.J.R.; Fernandes, E.C.A.; Hettinga, K.A.; Vervoort, J.M.; van Hooijdonk, T.C.M.; Zwietering, M.H.; Smid, E.J.; van Valenberg, H.J.F. Influence of different proteolytic strains of Streptococcus thermophilus in co-culture with Lactobacillus delbrueckii subsp. bulgaricus on the metabolite profile of set-yoghurt. Int. J. Food Microbiol. 2014, 177, 29–36. [Google Scholar] [CrossRef]
- Kang, C.; Zhang, Y.; Zhang, M.; Qi, J.; Zhao, W.; Gu, J.; Guo, W.; Li, Y. Screening of specific quantitative peptides of beef by LC–MS/MS coupled with OPLS-DA. Food Chem. 2022, 387, 132932. [Google Scholar] [CrossRef]
- Zhang, L.; Mi, S.; Liu, R.-B.; Sang, Y.-X.; Wang, X.-H. Evaluation of Volatile Compounds during the Fermentation Process of Yogurts by Streptococcus thermophilus Based on Odor Activity Value and Heat Map Analysis. Int. J. Anal. Chem. 2020, 1, 3242854. [Google Scholar] [CrossRef]
- Coppen, A.; Swade, C.; Jones, S.A.; Armstrong, R.A.; Blair, J.A.; Leeming, R.J. Depression and tetrahydrobiopterin: The folate connection. J. Affect. Disord. 1989, 16, 103–107. [Google Scholar] [CrossRef]
- De Schutter, D.P.; Saison, D.; Delvaux, F.; Derdelinckx, G.; Rock, J.-M.; Neven, H.; Delvaux, F.R. Characterization of volatiles in unhopped wort. J. Agric. Food Chem. 2008, 56, 246–254. [Google Scholar] [CrossRef]
- Song, H.; Lee, S.Y. Production of succinic acid by bacterial fermentation. Enzym. Microb. Technol. 2006, 39, 352–361. [Google Scholar] [CrossRef]
- Presley, B.; Bianchi, B.; Coleman, J.; Diamond, F.; McNally, G. Efficiency of extraction and conversion of pseudoephedrine to methamphetamine from tamper-resistant and non-tamper-resistant formulations. J. Pharm. Biomed. Anal. 2018, 156, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.C.; Duus Johansen, J. Public consultation on perfumery materials in the framework of Council Directive 76/768/EEC relative to cosmetic products. Contact Dermat. 2008, 5, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Potter, L.R. Regulation and therapeutic targeting of peptide-activated receptor guanylyl cyclases. Pharmacol. Ther. 2011, 130, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Gass, M.L.S.; Khan, S. Chapter 77—Estrogen and Estrogen Analogs for Prevention and Treatment of Osteoporosis. In Osteoporosis, 4th ed.; Academic Press: Cambridge, MA, USA, 2013; pp. 1805–1825. [Google Scholar] [CrossRef]
- Cai, W.; Tang, F.; Wang, Y.; Zhang, Z.; Xue, Y.; Zhao, X.; Guo, Z.; Shan, C. Bacterial diversity and flavor profile of Zha-Chili, a traditional fermented food in China. Food Res. Int. 2021, 141, 110112. [Google Scholar] [CrossRef] [PubMed]
- Newstead, S. Insights into L-type heteromeric amino acid transporters. Nat. Struct. Mol. Biol. 2019, 26, 395–396. [Google Scholar] [CrossRef]
- Paradkar, A. Clavulanic acid production by Streptomyces clavuligerus: Biogenesis, regulation and strain improvement. J. Antibiot. 2013, 66, 411–420. [Google Scholar] [CrossRef]
- Liras, P.; Rodroguez-Garcia, A. Clavulanic acid, a beta-lactamase inhibitor: Biosynthesis and molecular genetics. Appl. Microbiol. Biotechnol. 2000, 54, 467–475. [Google Scholar] [CrossRef]
- Ku, J.W.K.; Gan, Y.-H. New roles for glutathione: Modulators of bacterial virulence and pathogenesis. Redox Biol. 2021, 44, 102012. [Google Scholar] [CrossRef]
- Qiu, M.; Jiang, J.; Jiang, W.; Zhang, W.; Jiang, Y.; Xin, F.; Jiang, M. The biosynthesis of L-phenylalanine-derived compounds by engineered microbes. Biotechnol. Adv. 2024, 77, 108448. [Google Scholar] [CrossRef]
- Li, J.; Ilangovan, U.; Daubner, S.C.; Hinck, A.P.; Fitzpatrick, P.F. Direct evidence for a phenylalanine site in the regulatory domain of phenylalanine hydroxylase. Arch. Biochem. Biophys. 2011, 505, 250–255. [Google Scholar] [CrossRef] [PubMed]
| Groups | Total Number Of Viable Bacteria (LgCFU/mL) | Acidity (°T) | Water-Holding Capacity (%) |
|---|---|---|---|
| SC | 9.13 ± 0.02 f | 88.31 ± 1.22 e | 28.97 ± 0.52 e |
| SS | 9.83 ± 0.02 a | 104.60 ± 0.70 b | 48.99 ± 1.14 a |
| FCS | 9.56 ± 0.03 c | 109.70 ± 0.87 a | 38.03 ± 1.20 c |
| SCS | 9.46 ± 0.01 d | 90.01 ± 0.81 de | 31.35 ± 0.79 de |
| TCS | 9.29 ± 0.01 e | 91.81 ± 0.16 d | 33.22 ± 0.07 d |
| OCS | 9.25 ± 0.02 e | 86.49 ± 0.17 f | 39.12 ± 0.47 bc |
| RCS | 9.73 ± 0.03 b | 98.10 ± 1.42 c | 42.23 ± 0.08 b |
| Groups | Hardness/N | Maximum Adhesion/N | Adhesion/mJ | Adhesive/N |
|---|---|---|---|---|
| SC | 0.322 ± 0.021 c | 0.062 ± 0.015 b | 0.217 ± 0.025 d | 0.105 ± 0.014 c |
| SS | 0.473 ± 0.014 a | 0.133 ± 0.011 a | 0.536 ± 0.017 a | 0.197 ± 0.008 a |
| FCS | 0.413 ± 0.016 b | 0.118 ± 0.011 a | 0.487 ± 0.010 ab | 0.178 ± 0.010 ab |
| SCS | 0.370 ± 0.025 bc | 0.110 ± 0.012 a | 0.480 ± 0.021 ab | 0.158 ± 0.011 b |
| TCS | 0.412 ± 0.010 b | 0.102 ± 0.006 a | 0.357 ± 0.019 c | 0.145 ± 0.012 b |
| OCS | 0.368 ± 0.021 bc | 0.062 ± 0.018 b | 0.297 ± 0.036 c | 0.111 ± 0.008 c |
| RCS | 0.458 ± 0.012 a | 0.113 ± 0.006 a | 0.434 ± 0.028 b | 0.179 ± 0.011 ab |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Ma, R.; Zhu, L.; Wang, J.; Wang, R.; Wu, X.; Liu, X.; Huang, X.; Zhang, L.; Liu, B. Improvement of Quality of Sour Camel Milk by Extract of Sparassis crispa: Physicochemical Properties, Sensory Quality and Metabolic Changes. Foods 2025, 14, 3042. https://doi.org/10.3390/foods14173042
Zhao L, Ma R, Zhu L, Wang J, Wang R, Wu X, Liu X, Huang X, Zhang L, Liu B. Improvement of Quality of Sour Camel Milk by Extract of Sparassis crispa: Physicochemical Properties, Sensory Quality and Metabolic Changes. Foods. 2025; 14(17):3042. https://doi.org/10.3390/foods14173042
Chicago/Turabian StyleZhao, Lina, Ruping Ma, Linyan Zhu, Jinzhi Wang, Rui Wang, Xiaojun Wu, Xiaoyan Liu, Xinhong Huang, Lianchao Zhang, and Bin Liu. 2025. "Improvement of Quality of Sour Camel Milk by Extract of Sparassis crispa: Physicochemical Properties, Sensory Quality and Metabolic Changes" Foods 14, no. 17: 3042. https://doi.org/10.3390/foods14173042
APA StyleZhao, L., Ma, R., Zhu, L., Wang, J., Wang, R., Wu, X., Liu, X., Huang, X., Zhang, L., & Liu, B. (2025). Improvement of Quality of Sour Camel Milk by Extract of Sparassis crispa: Physicochemical Properties, Sensory Quality and Metabolic Changes. Foods, 14(17), 3042. https://doi.org/10.3390/foods14173042







