pH-Sensitive Cassava Starch/Onion Peel Powder Films as Colorimetric Indicators for Minced Beef Freshness Monitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
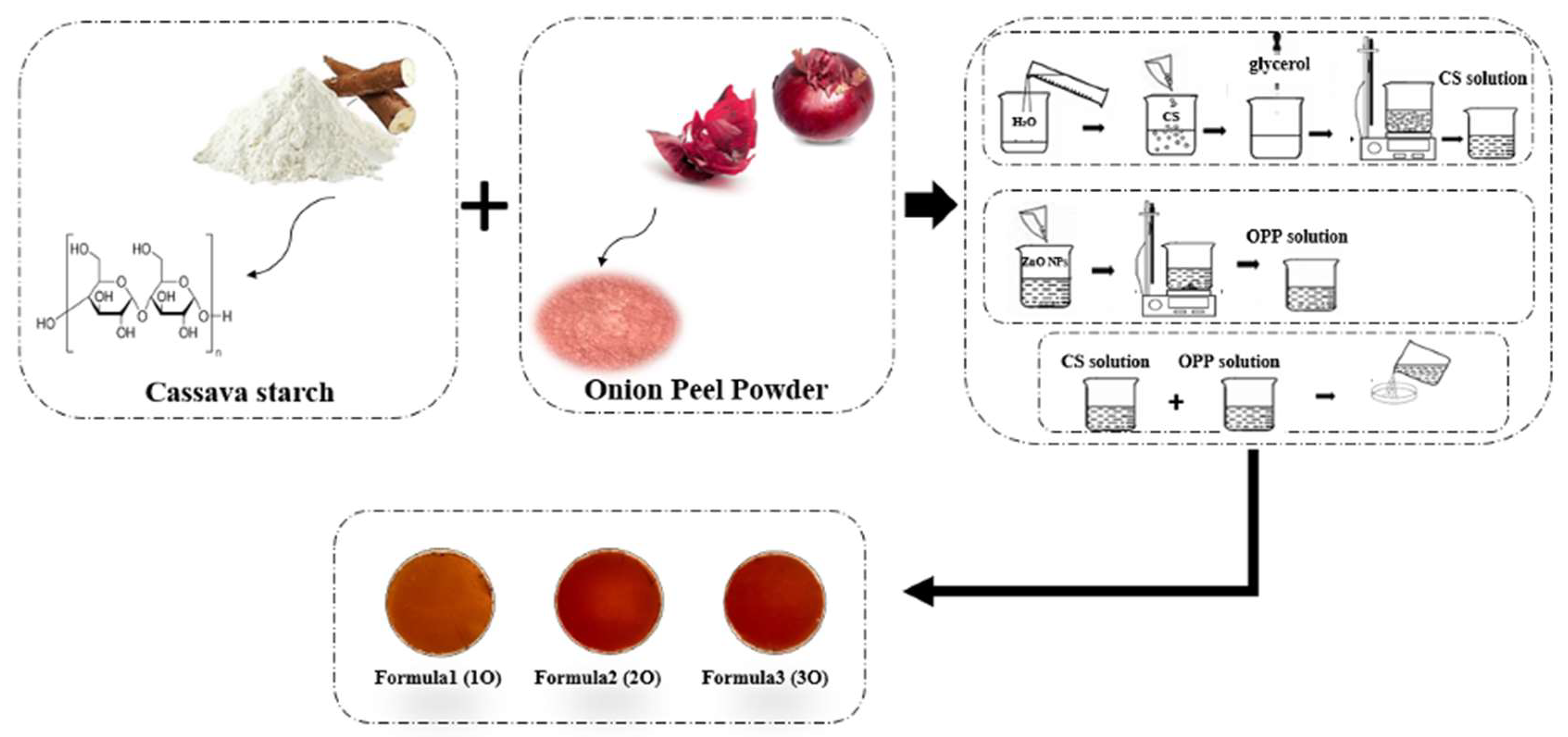
2.2. Preparation of Onion Peel Powder (OPP)
2.3. Preparation of Indicator Films
2.4. Total Phenolic Content (TPC)
2.5. Total Flavonoid Content (TFC)
2.6. Antioxidant Activity (DPPH and ABTS Assays)
2.7. Release Rate of Antioxidants
2.8. Antibacterial Activity
2.9. pH-Sensing Ability of OPP and Starch–OPP Films
2.10. Performance of the Real-Time Monitoring of Meat Freshness
2.10.1. Packaging of Minced Beef
2.10.2. Microbiological Analysis
- N = number of colonies per mL or g of product;
- ΣC\Sigma ΣC = total number of colonies counted on both dishes;
- V = volume of inoculum used (mL);
- n1 = number of plates at the first dilution;
- n2 = number of plates at the second dilution;
- D = dilution factor corresponding to the first dilution.
2.10.3. pH Measurement
2.10.4. Colorimetric Analysis of Indicator Films
2.11. Statistical Analysis
3. Results
3.1. Total Phenolic and Flavonoid Content
3.2. Antioxidant Activity
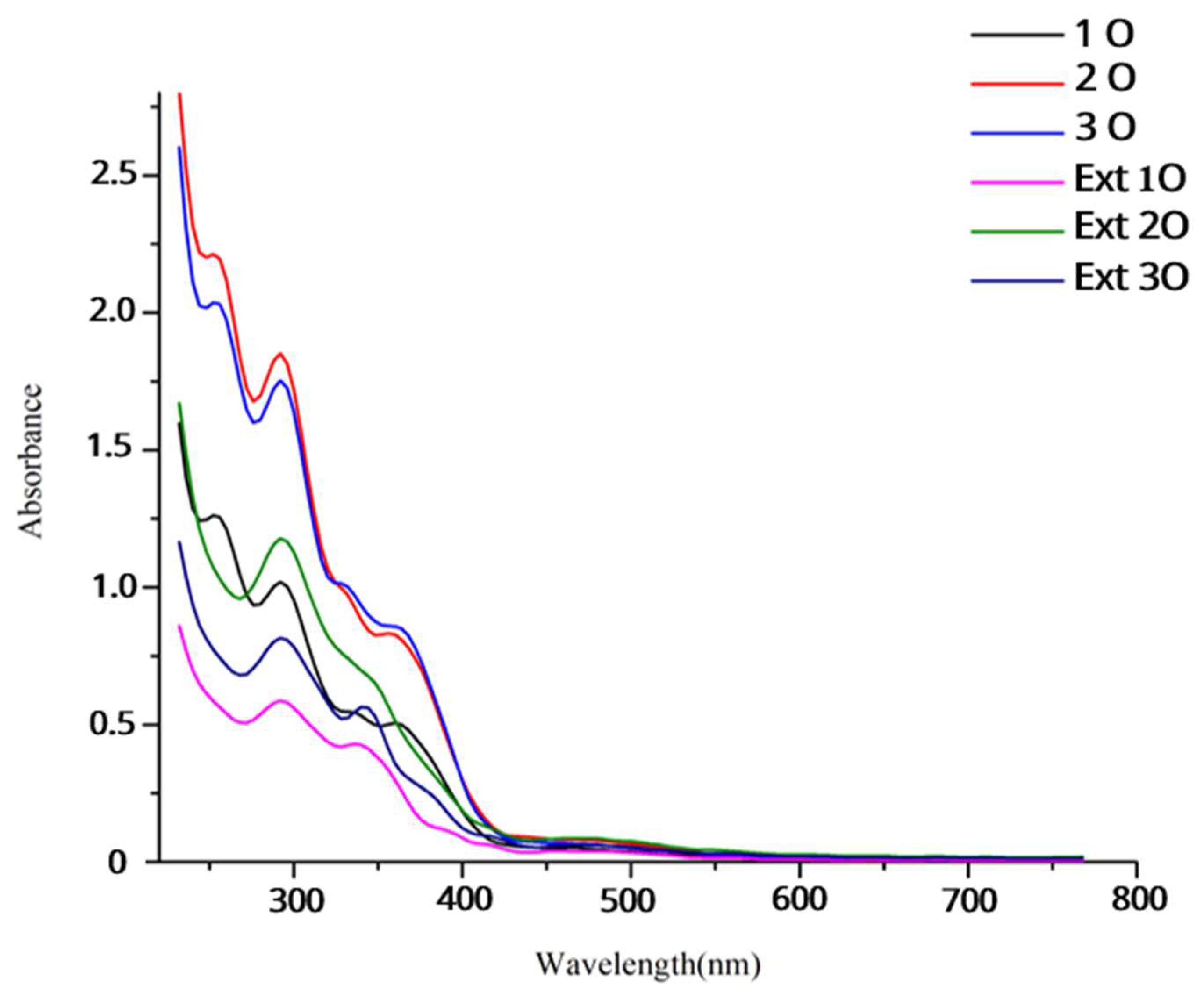
3.3. Release of Antioxidant Compounds
3.4. Antimicrobial Properties
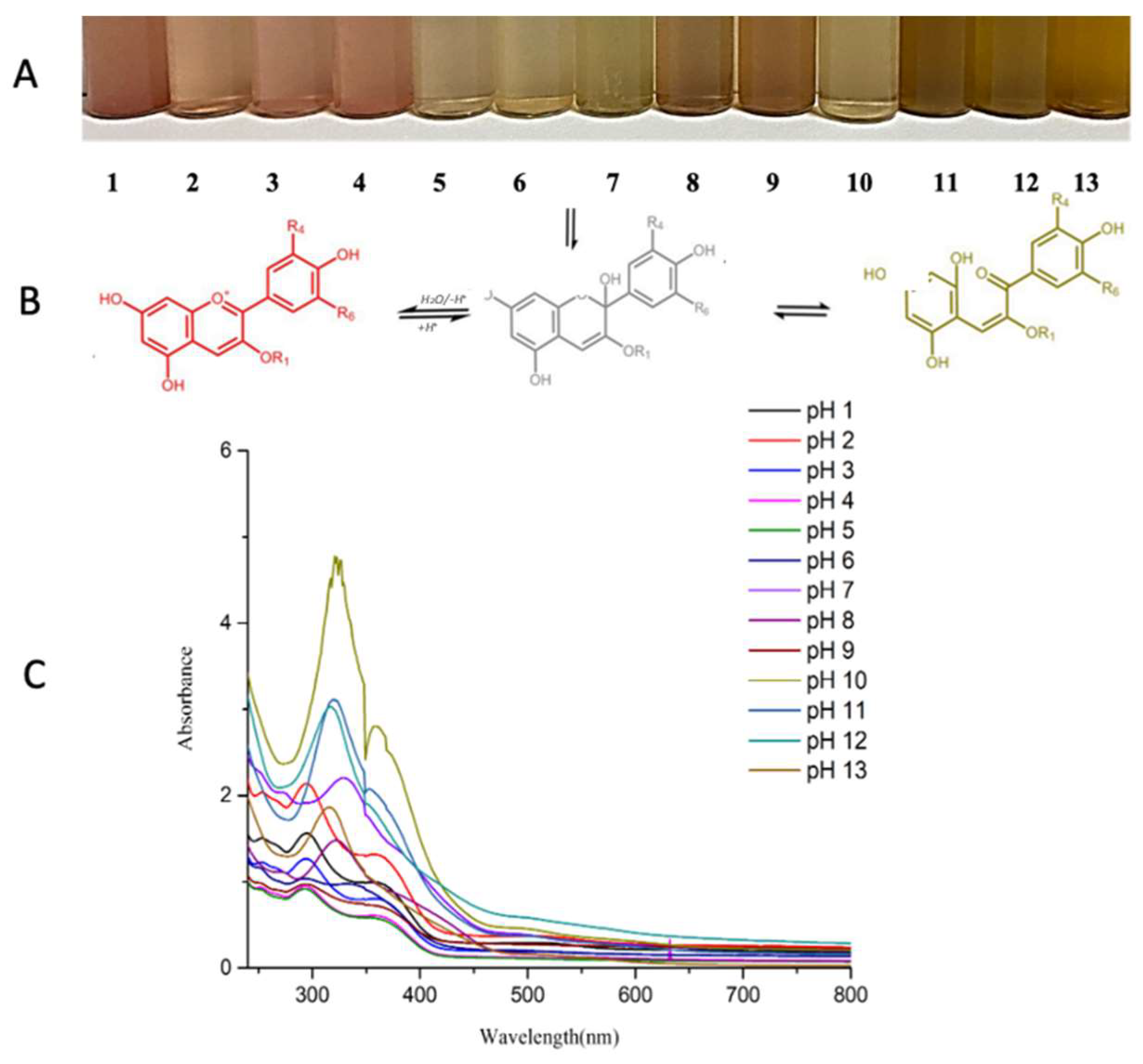
3.5. pH-Sensitive Color Change
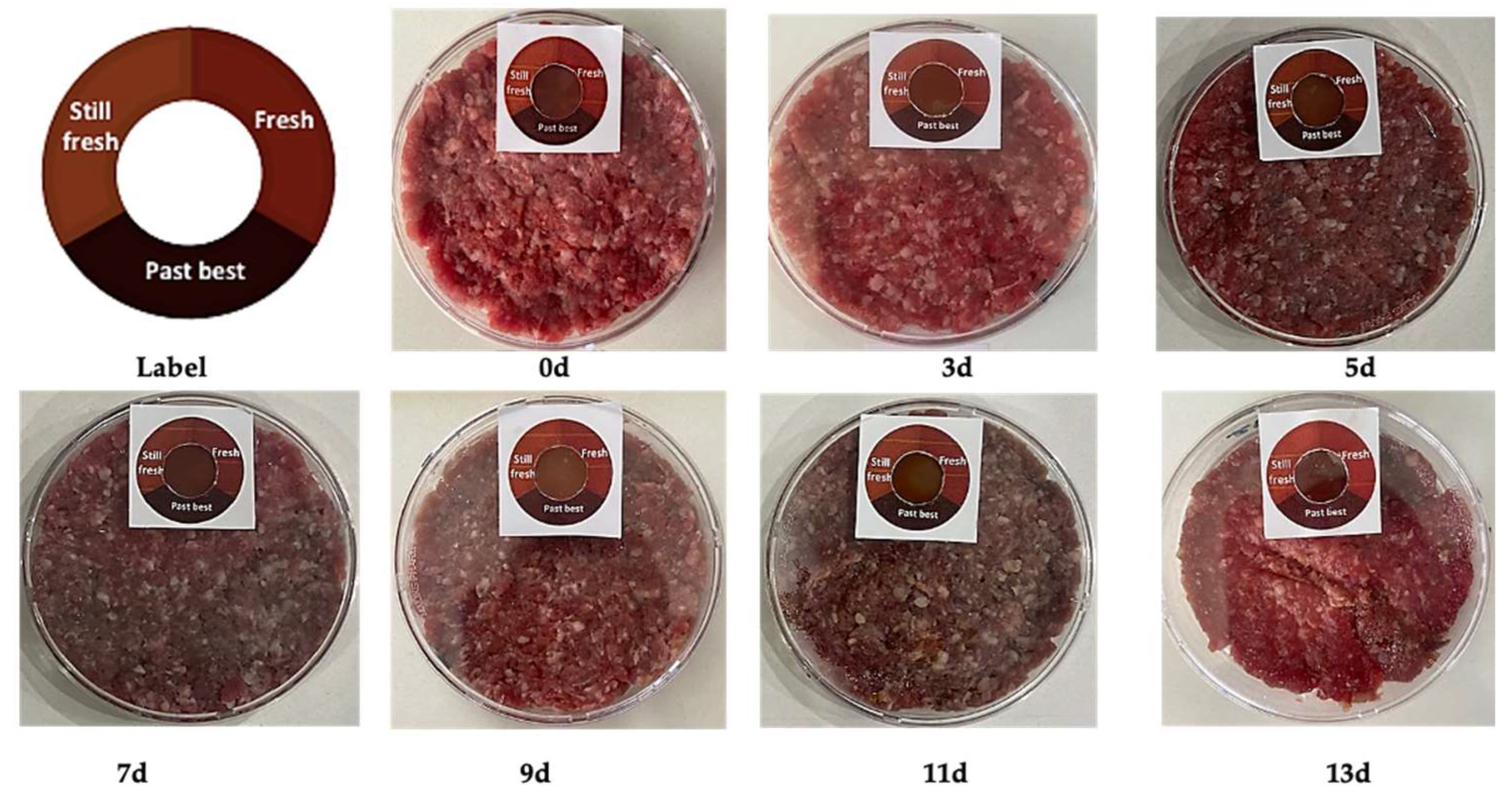
3.6. Real-Time Monitoring of Meat Freshness
4. Discussion
4.1. Food Safety and Shelf-Life Extension
4.2. Advantages of OPP-Based Colorimetric Films
4.3. Circular Bioeconomy and Valorization of Agro-Waste
4.4. Industrial Relevance and Innovation
4.5. Future Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Koshy, R.R.; Koshy, J.T.; Mary, S.K.; Sadanandan, S.; Jisha, S.; Pothan, L.A. Preparation of pH sensitive film based on starch/carbon nano dots incorporating anthocyanin for monitoring spoilage of pork. Food Control 2021, 126, 108039. [Google Scholar] [CrossRef]
- Lin, T.; Wu, Y.; Li, Z.; Song, Z.; Guo, L.; Fu, F. Visual monitoring of food spoilage based on hydrolysis-induced silver metallization of au nanorods. Anal. Chem. 2016, 88, 11022–11027. [Google Scholar] [CrossRef] [PubMed]
- Torche, A.; Chouna, T.; Djeghim, F.; Sanah, I.; Arhab, R.; D’Elia, M.; Rastrelli, L. Optimization of Cassava Starch/Onion Peel Powder-Based Bioplastics: Influence of Composition on Mechanical Properties and Biodegradability Using Central Composite Design. Foods 2025, 14, 2414. [Google Scholar] [CrossRef]
- Wu, C.; Sun, J.; Zheng, P.; Kang, X.; Chen, M.; Li, Y.; Ge, Y.; Hu, Y.; Pang, J. Preparation of an intelligent film based on chitosan/oxidized chitin nanocrystals incorporating black rice bran anthocyanins for seafood spoilage monitoring. Carbohydr. Polym. 2019, 222, 115006. [Google Scholar] [CrossRef]
- Chen, J.; Long, Z.; Wang, S.; Meng, Y.; Zhang, G.; Nie, S. Biodegradable blends of graphene quantum dots and thermoplastic starch with solid-state photoluminescent and conductive properties. Int. J. Biol. Macromol. 2019, 139, 367–376. [Google Scholar] [CrossRef]
- Santos, L.G.; Silva, G.F.A.; Gomes, B.M.; Martins, V.G. A novel sodium alginate active films functionalized with purple onion peel extract (Allium cepa). Biocatal. Agric. Biotechnol. 2021, 35, 102096. [Google Scholar] [CrossRef]
- Ju, A.; Song, K.B. Incorporation of yellow onion peel extract into the funoran-based biodegradable films as an antioxidant packaging material. Int. J. Food Sci. Technol. 2020, 55, 1671–1678. [Google Scholar] [CrossRef]
- Ndwandwe, B.K.; Malinga, S.P.; Kayitesi, E.; Dlamini, B.C. Recent developments in the application of natural pigments as pH-sensitive food freshness indicators in biopolymer-based smart packaging: Challenges and opportunities. Int. J. Food Sci. Technol. 2024, 59, 2148–2161. [Google Scholar] [CrossRef]
- Kumar, Y.; Bist, Y.; Thakur, D.; Nagar, M.; Saxena, D.C. A review on the role of pH-sensitive natural pigments in biopolymers based intelligent food packaging films. Int. J. Biol. Macromol. 2024, 276, 133869. [Google Scholar] [CrossRef]
- Karnwal, A.; Rauf, A.; Jassim, A.Y.; Selvaraj, M.; Al-Tawaha, A.R.M.S.; Kashyap, P.; Kumar, D.; Malik, T. Advanced starch-based films for food packaging: Innovations in sustainability and functional properties. Food Chem. X 2025, 29, 102662. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, P.; Chen, J.; Yan, Y.; Wu, Z. Recent advances and applications in starch for intelligent active food packaging: A review. Foods 2022, 11, 2879. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Yan, X.; Zhang, J.; Wu, X.; Luan, M.; Zhang, Q. Preparation and characterization of pH-sensitive intelligent packaging films based on cassava starch/polyvinyl alcohol matrices containing Aronia melanocarpa anthocyanins. LWT 2024, 194, 115818. [Google Scholar] [CrossRef]
- Mahović Poljaček, S.; Tomašegović, T.; Strižić Jakovljević, M.; Jamnicki Hanzer, S.; Murković Steinberg, I.; Žuvić, I.; Leskovac, M.; Lavrič, G.; Kavčič, U.; Karlovits, I. Starch-Based Functional Films Enhanced with Bacterial Nanocellulose for Smart Packaging: Physicochemical Properties, pH Sensitivity and Colorimetric Response. Polymers 2024, 16, 2259. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, S.; Xie, C.; Wang, C.; Zhong, Y.; Fan, K. Recent advances in pH-sensitive indicator films based on natural colorants for smart monitoring of food freshness: A review. Crit. Rev. Food Sci. Nutr. 2024, 64, 12800–12819. [Google Scholar] [CrossRef]
- Song, W.; Wu, N.; He, Y.; Zhao, H.; Xu, J.; Ren, L. Intelligent color changing packaging film based on esterified starch and black rice anthocyanins. Food Chem. X 2024, 24, 101930. [Google Scholar] [CrossRef] [PubMed]
- Müller, L.; Gnoyke, S.; Popken, A.M.; Böhm, V. Antioxidant capacity and related parameters of different fruit formulations. LWT 2010, 43, 992–999. [Google Scholar] [CrossRef]
- Kumar, N.; Neeraj; Ojha, A.; Singh, R. Preparation and characterization of chitosan—Pullulan blended edible films enrich with pomegranate peel extract. React. Funct. Polym. 2019, 144, 104350. [Google Scholar] [CrossRef]
- Dou, L.; Li, B.; Zhang, K.; Chu, X.; Hou, H. Physical properties and antioxidant activity of gelatin-sodium alginate edible films with tea polyphenols. Int. J. Biol. Macromol. 2018, 118, 1377–1383. [Google Scholar] [CrossRef]
- Pirsa, S.; Bener, M.; Şen, F.B. Biodegradable film of carboxymethyl cellulose modified with red onion peel powder waste and boron nitride nanoparticles: Investigation of physicochemical properties and release of active substances. Food Chem. 2024, 445, 138721. [Google Scholar] [CrossRef]
- Elhadef, K.; Chaari, M.; Akermi, S.; Ennouri, K.; Ben Hlima, H.; Fourati, M.; Mtibaa, A.C.; Ennouri, M.; Sarkar, T.; Shariati, M.A.; et al. Gelatin-sodium alginate packaging film with date pits extract: An eco-friendly packaging for extending raw minced beef shelf life. Meat Sci. 2024, 207, 109371. [Google Scholar] [CrossRef]
- Luchese, C.L.; Sperotto, N.; Spada, J.C.; Tessaro, I.C. Effect of blueberry agro-industrial waste addition to corn starch-based films for the production of a pH-indicator film. Int. J. Biol. Macromol. 2017, 104, 11–18. [Google Scholar] [CrossRef]
- ISO 4833-1: 2013; Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 C by the Pour Plate Technique. International Organization for Standardization: Geneva, Switzerland, 2020.
- Smaoui, S.; Hsouna, A.B.; Lahmar, A.; Ennouri, K.; Mtibaa-Chakchouk, A.; Sellem, I.; Najah, S.; Bouaziz, M.; Mellouli, L. Bio-preservative effect of the essential oil of the endemic Mentha piperita used alone and in combination with BacTN635 in stored minced beef meat. Meat Sci. 2016, 117, 196–204. [Google Scholar] [CrossRef]
- Medeiros Silva, V.D.; Coutinho Macedo, M.C.; Rodrigues, C.G.; Neris dos Santos, A.; de Freitas e Loyola, A.C.; Fante, C.A. Biodegradable edible films of ripe banana peel and starch enriched with extract of Eriobotrya japonica leaves. Food Biosci. 2020, 38, 100750. [Google Scholar] [CrossRef]
- Pérez-Gregorio, R.M.; García-Falcón, M.S.; Simal-Gándara, J.; Rodrigues, A.S.; Almeida, D.P.F. Identification and quantification of flavonoids in traditional cultivars of red and white onions at harvest. J. Food Compos. Anal. 2010, 23, 592–598. [Google Scholar] [CrossRef]
- Akcan, T.; Estévez, M.; Serdaroğlu, M. Antioxidant protection of cooked meatballs during frozen storage by whey protein edible films with phytochemicals from Laurus nobilis L. and Salvia officinalis. LWT 2017, 77, 323–331. [Google Scholar] [CrossRef]
- de Oliveira Filho, J.G.; Rodrigues, J.M.; Valadares, A.C.F.; de Almeida, A.B.; de Lima, T.M.; Takeuchi, K.P.; Alves, C.C.F.; de Figueiredo Sousa, H.A.; da Silva, E.R.; Dyszy, F.H.; et al. Active food packaging: Alginate films with cottonseed protein hydrolysates. Food Hydrocoll. 2019, 92, 267–275. [Google Scholar] [CrossRef]
- Thi Nguyen, T.; Pham, B.T.T.; Nhien Le, H.; Bach, L.G.; Thuc, C.N.H. Comparative characterization and release study of edible films of chitosan and natural extracts. Food Packag. Shelf Life 2022, 32, 100830. [Google Scholar] [CrossRef]
- Osojnik Črnivec, I.G.; Skrt, M.; Šeremet, D.; Sterniša, M.; Farčnik, D.; Štrumbelj, E.; Poljanšek, A.; Cebin, N.; Pogačnik, L.; Možina, S.S.; et al. Waste streams in onion production: Bioactive compounds, quercetin and use of antimicrobial and antioxidative properties. Waste Manag. 2021, 126, 476–486. [Google Scholar] [CrossRef]
- Bolumar, T.; LaPeña, D.; Skibsted, L.H.; Orlien, V. Rosemary and oxygen scavenger in active packaging for prevention of high-pressure induced lipid oxidation in pork patties. Food Packag. Shelf Life 2016, 7, 26–33. [Google Scholar] [CrossRef]
- Manso, T.; Lores, M.; de Miguel, T. Antimicrobial Activity of Polyphenols and Natural Polyphenolic Extracts on Clinical Isolates. Antibiotics 2022, 11, 46. [Google Scholar] [CrossRef]
- Makarewicz, M.; Drożdż, I.; Tarko, T.; Duda-Chodak, A. The interactions between polyphenols and microorganisms, especially gut microbiota. Antioxidants 2021, 10, 188. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Yong, H.; Qin, Y.; Liu, J.; Liu, J. Development of multifunctional food packaging films based on chitosan, TiO2 nanoparticles and anthocyanin-rich black plum peel extract. Food Hydrocoll. 2019, 94, 80–92. [Google Scholar] [CrossRef]
- Duan, Y. Ultraviolet-visible spectrum characterizations of quercetin in aqueous ethanol solution with different pH values. J. Chem. Pharm. Res. 2014, 6, 236–240. [Google Scholar]
- Ezati, P.; Rhim, J.W. pH-responsive chitosan-based film incorporated with alizarin for intelligent packaging applications. Food Hydrocoll. 2020, 102, 105629. [Google Scholar] [CrossRef]
- Jamróz, E.; Kulawik, P.; Guzik, P.; Duda, I. The verification of intelligent properties of furcellaran films with plant extracts on the stored fresh Atlantic mackerel during storage at 2 °C. Food Hydrocoll. 2019, 97, 105211. [Google Scholar] [CrossRef]
- Bouaziz, F.; Helbert, C.B.; Romdhane, M.B.; Koubaa, M.; Bhiri, F.; Kallel, F.; Chaari, F.; Driss, D.; Buon, L.; Chaabouni, S.E. Structural data and biological properties of almond gum oligosaccharide: Application to beef meat preservation. Int. J. Biol. Macromol. 2015, 72, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Docimo, T.; Celano, R.; Lambiase, A.; Di Sanzo, R.; Serio, S.; Santoro, V.; Coccetti, P.; Russo, M.; Rastrelli, L.; Piccinelli, A.L. Exploring Influence of Production Area and Harvest Time on Specialized Metabolite Content of Glycyrrhiza glabra Leaves and Evaluation of Antioxidant and Anti-Aging Properties. Antioxidants 2024, 13, 93. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, Y.; Zhao, L.; Wang, Y. Anthocyanin-based pH-sensitive smart packaging films for monitoring food freshness. J. Agric. Food Res. 2022, 9, 100340. [Google Scholar] [CrossRef]

| Films | DPPH (%) | ABTS (%) | TPC (µg/mL) | TFC (µg/mL) |
|---|---|---|---|---|
| Control | 12.95 ± 0.03 b | 42.94 ± 0.15 c | 63.71 ± 1.93 b | 15.97 ± 3.24 c |
| Formula 1 (1O) | 23.28 ± 3.31 b | 88.38 ± 0.51 b | 97.82 ± 2.22 a | 29.65 ± 3.83 b c |
| Formula 2 (2O) | 87.50 ± 8.55 a | 90.60 ± 0.60 a | 109.39 ± 2.07 a | 88.54 ± 3.09 a |
| Formula 3 (3O) | 66.62 ± 13.76 a | 89.03 ± 0.80 b | 102.33 ± 9.85 a | 43.82 ± 1.62 b |
| p-Value | p < 0.0001 | p < 0.0001 | p < 0.05 | p < 0.0001 |
| Films | Antioxidant Release (%) |
|---|---|
| Formula 1 (1O) | 70.10 ± 0.005 c |
| Formula 2 (2O) | 95.03 ± 0.025 a |
| Formula 3 (3O) | 83.31 ± 0.015 b |
| p-value | p < 0.0001 |
| Films | Bacillus cereus | Pseudomonas aeruginosa | Escherichia coli | Staphylococcus aureus |
|---|---|---|---|---|
| Control | 0.0 ± 0.0 c | 0.0 ± 0.0 c | 0.0 ± 0.0 b | 0.0 ± 0.0 b |
| Formula 1 (1O) | 0.0 ± 0.0 c | 8.06 ± 0.04 c | 7.22 ± 0.02 a | 0.0 ± 0.0 b |
| Formula 2 (2O) | 0.0 ± 0.0 c | 11.04 ± 0.03 b | 0.0 ± 0.0 b | 0.0 ± 0.0 b |
| Formula 3 (3O) | 0.0 ± 0.0 c | 0.0 ± 0.0 a | 0.0 ± 0.0 b | 0.0 ± 0.0 b |
| p-Value | p > 0.05 | p < 0.0001 | p < 0.0001 | p > 0.05 |
| pH | L* | a* | b* | ΔE |
|---|---|---|---|---|
| 1 | 38.293 ± 0.120 | 43.293 ± 0.050 | 41.540 ± 0.295 | 20.051 ± 0.073 |
| 2 | 39.433 ± 0.170 | 36.906 ± 0.070 | 36.463 ± 0.209 | 17.493 ± 0.161 |
| 3 | 39.326 ± 0.092 | 33.463 ± 0.032 | 36.310 ± 0.026 | 16.935 ± 0.080 |
| 4 | 50.300 ± 0.020 | 27.020 ± 0.026 | 35.530 ± 0.026 | 10.264 ± 0.023 |
| 5 | 47.110 ± 0.010 | 31.850 ± 0.043 | 36.460 ± 0.295 | 10.324 ± 0.142 |
| 6 | 43.703 ± 0.182 | 34.743 ± 0.081 | 40.796 ± 0.251 | 11.560 ± 0.098 |
| 7 | 48.316 ± 0.021 | 31.820 ± 0.052 | 38.683 ± 0.015 | 7.935 ± 0.032 |
| 8 | 45.110 ± 0.010 | 35.856 ± 0.066 | 35.903 ± 0.015 | 12.897 ± 0.038 |
| 9 | 42.270 ± 0.026 | 37.033 ± 0.041 | 41.153 ± 0.055 | 13.569 ± 0.055 |
| 10 | 45.350 ± 0.020 | 34.273 ± 0.025 | 38.203 ± 0.025 | 10.965 ± 0.012 |
| 11 | 40.696 ± 0.025 | 36.040 ± 0.017 | 41.190 ± 0.010 | 14.659 ± 0.008 |
| 12 | 26.856 ± 0.066 | 34.353 ± 0.041 | 33.486 ± 0.011 | 29.548 ± 0.051 |
| 13 | 14.226 ± 0.057 | 28.170 ± 0.026 | 17.783 ± 0.020 | 47.977 ± 0.008 |
| Storage Time (Day) | APC (log CFU/g) | pH | ΔE |
|---|---|---|---|
| 0 | 5.400 ± 0.010 c | 6.0430 ± 0.005 d | 42.226 ± 0.127 a |
| 3 | 5.319 ± 0.013 d | 6.151 ± 0.007 d | 40.845 ± 0.058 a |
| 5 | 5.348 ± 0.005 d | 6.051 ± 0.004 d | 44.766 ± 0.029 b |
| 7 | 5.315 ± 0.015 d | 6.083 ± 0.112 d | 47.361 ± 0.020 c |
| 9 | 6.183 ± 0.018 b | 6.843 ± 0.035 c | 49.655 ± 0.003 d |
| 11 | 6.213 ± 0.008 b | 8.800 ± 0.118 b | 58.676 ± 0.021 e |
| 13 | 6.260 ± 0.010 a | 9.570 ± 0.075 a | 70.146 ± 0.020 f |
| p-Value | p < 0.0001 | p < 0.0001 | p < 0.0001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torche, A.; Chouana, T.; Sanah, I.; Djeghim, F.; Khodja, E.A.T.; Mezreb, K.; Elboutachfaiti, R.; Delattre, C.; D’Elia, M.; Rastrelli, L. pH-Sensitive Cassava Starch/Onion Peel Powder Films as Colorimetric Indicators for Minced Beef Freshness Monitoring. Foods 2025, 14, 2974. https://doi.org/10.3390/foods14172974
Torche A, Chouana T, Sanah I, Djeghim F, Khodja EAT, Mezreb K, Elboutachfaiti R, Delattre C, D’Elia M, Rastrelli L. pH-Sensitive Cassava Starch/Onion Peel Powder Films as Colorimetric Indicators for Minced Beef Freshness Monitoring. Foods. 2025; 14(17):2974. https://doi.org/10.3390/foods14172974
Chicago/Turabian StyleTorche, Assala, Toufik Chouana, Ibtissem Sanah, Fairouz Djeghim, Esma Anissa Trad Khodja, Katiba Mezreb, Redouan Elboutachfaiti, Cedric Delattre, Maria D’Elia, and Luca Rastrelli. 2025. "pH-Sensitive Cassava Starch/Onion Peel Powder Films as Colorimetric Indicators for Minced Beef Freshness Monitoring" Foods 14, no. 17: 2974. https://doi.org/10.3390/foods14172974
APA StyleTorche, A., Chouana, T., Sanah, I., Djeghim, F., Khodja, E. A. T., Mezreb, K., Elboutachfaiti, R., Delattre, C., D’Elia, M., & Rastrelli, L. (2025). pH-Sensitive Cassava Starch/Onion Peel Powder Films as Colorimetric Indicators for Minced Beef Freshness Monitoring. Foods, 14(17), 2974. https://doi.org/10.3390/foods14172974









