Residues of Priority Organic Micropollutants in Eruca vesicaria (Rocket) Irrigated by Reclaimed Wastewater: Optimization of a QuEChERS SPME-GC/MS Protocol and Risk Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Standards
2.2. Instrumentation and Chromatographic Conditions
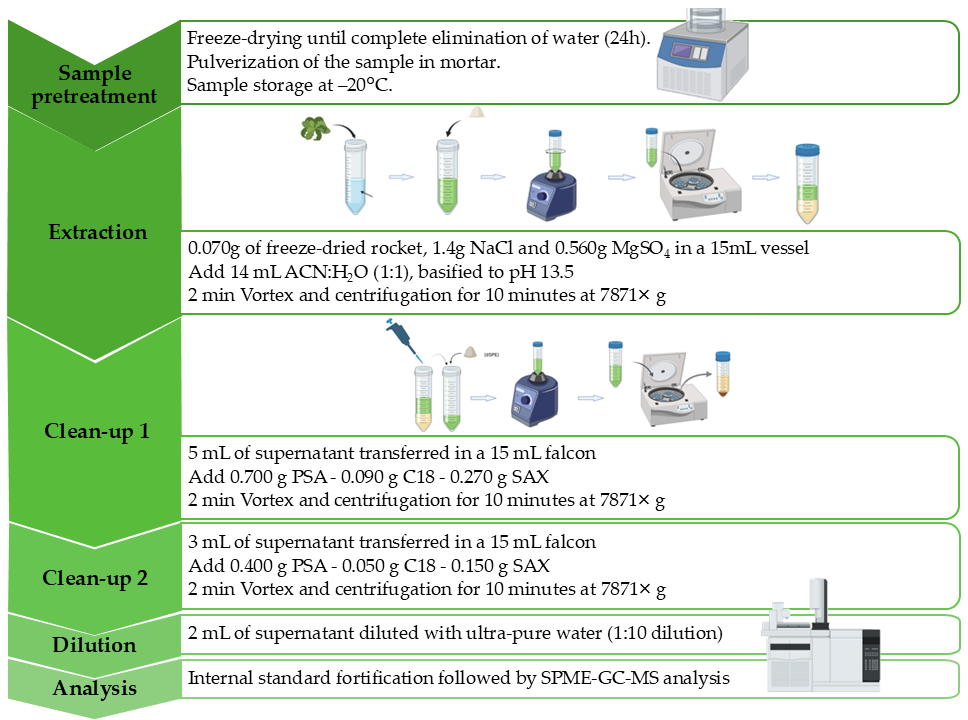
2.3. Sample Collection and Preparation
2.4. QuEChERS Extraction Protocol
2.5. SPME–GC–MS Optimization
2.6. Protocol Validation
2.6.1. Extraction Recoveries
2.6.2. Linearity
2.6.3. MDL and MQL
2.6.4. Precision
2.7. Risk Analysis
3. Results and Discussions
3.1. Optimization of SPME Conditions
3.1.1. Fiber Selection and Extract Dilution
3.1.2. Extraction Temperature
3.2. Optimization of QuEChERS Extraction and Cleanup
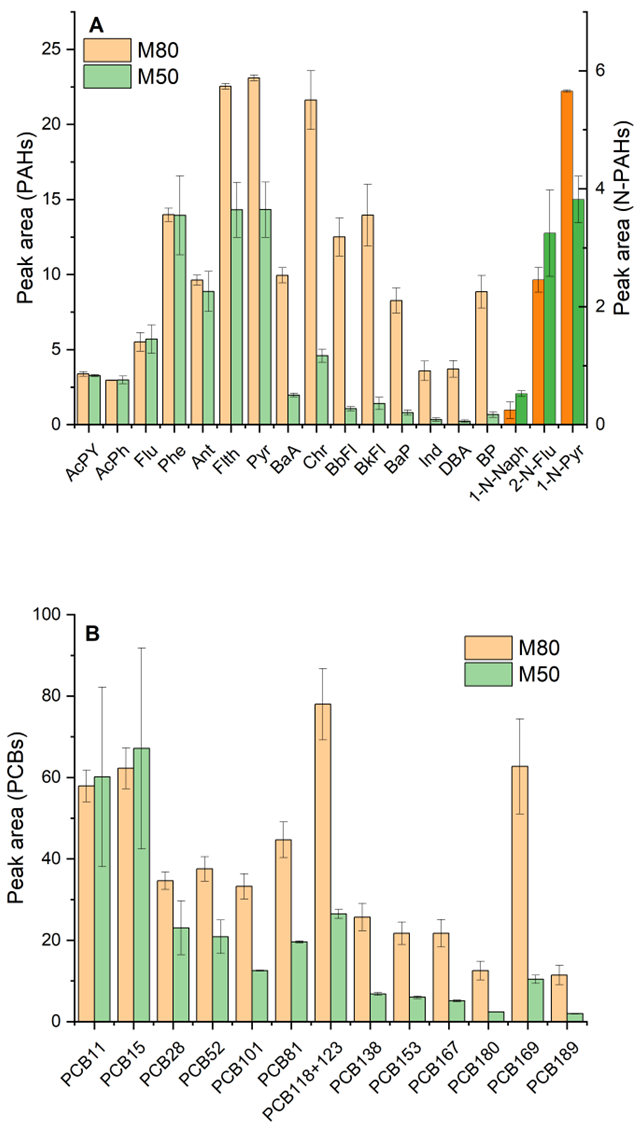
3.2.1. Extraction pH
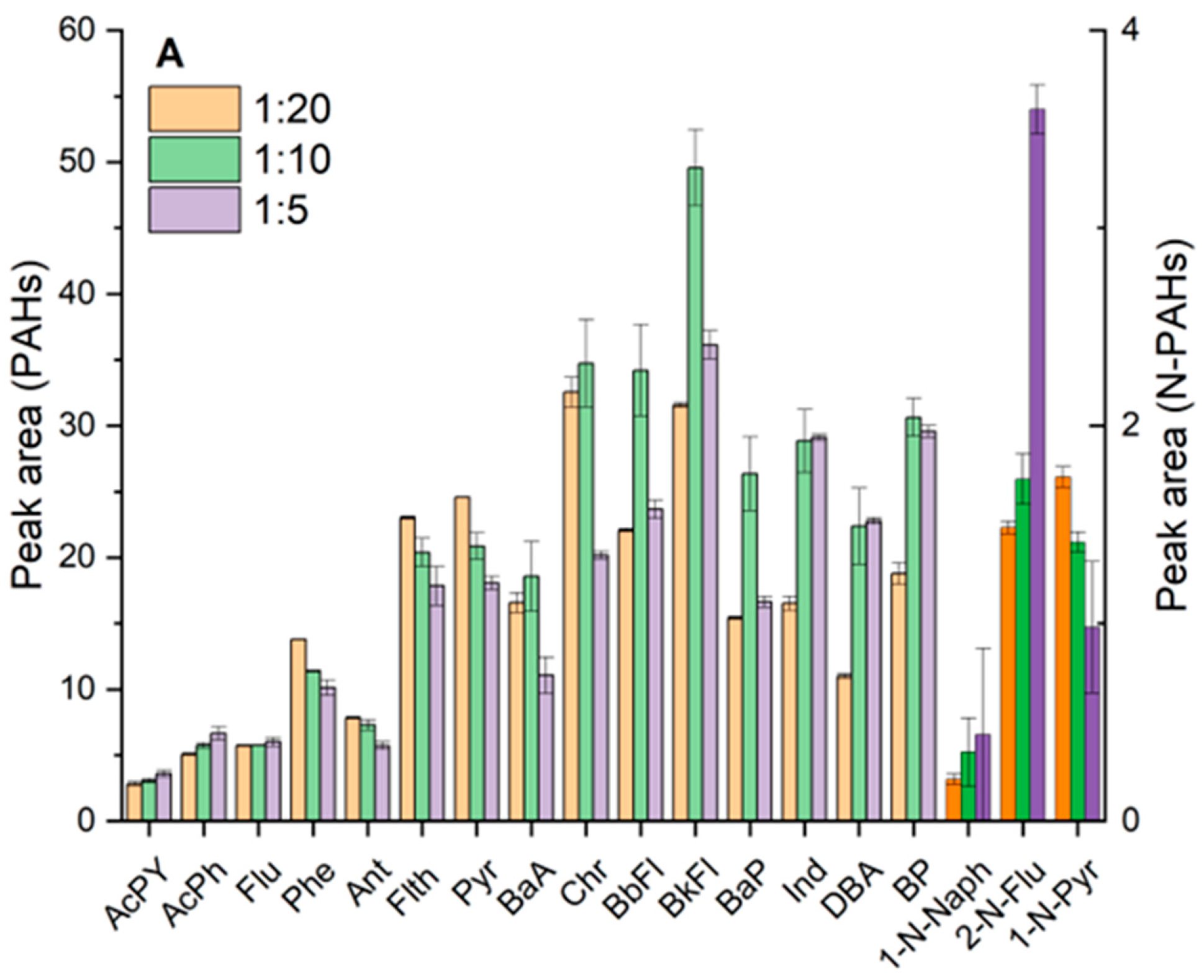
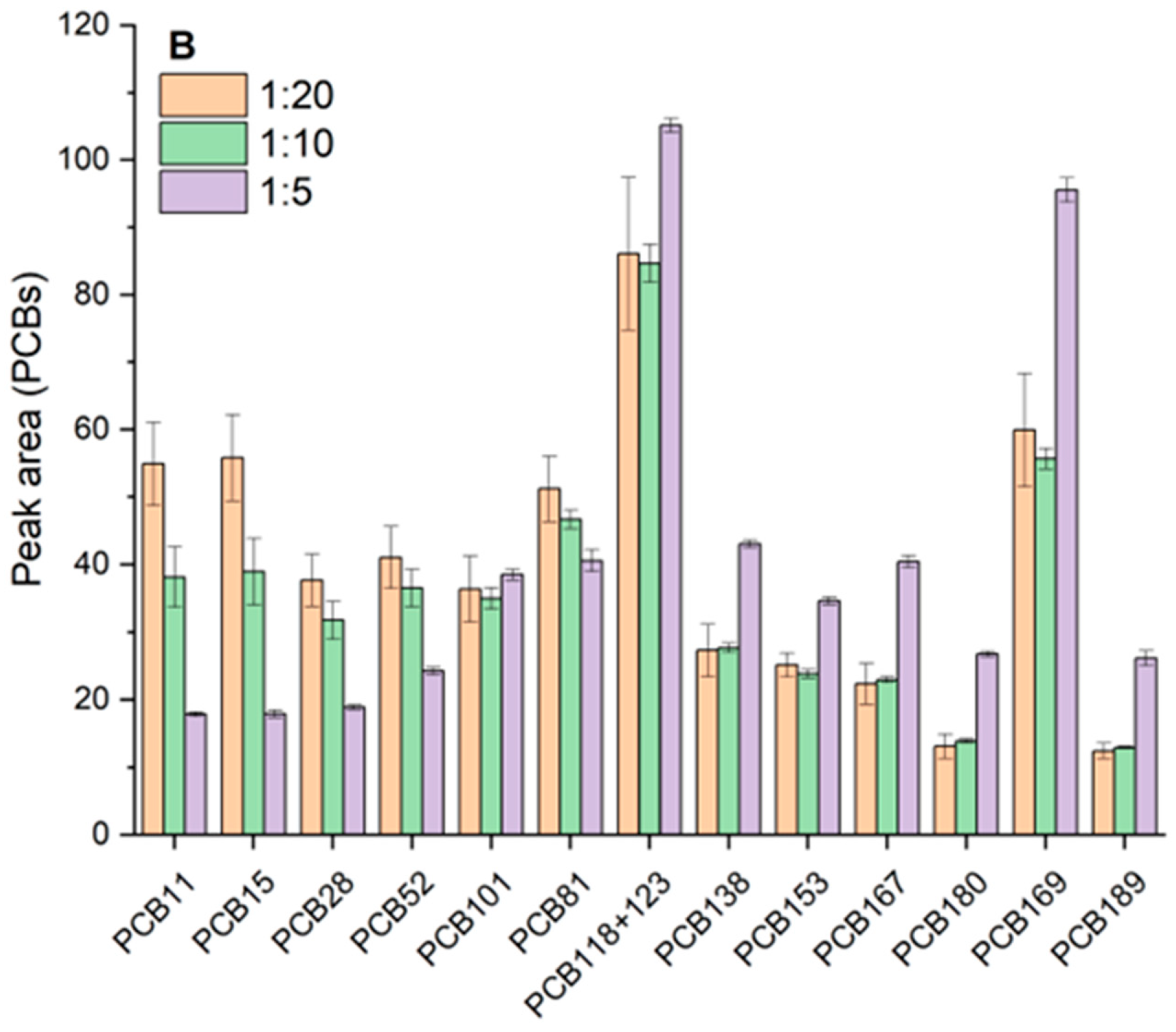
3.2.2. Dispersive-SPE Sorbents and Cleanup Strategy
3.3. Protocol Validation
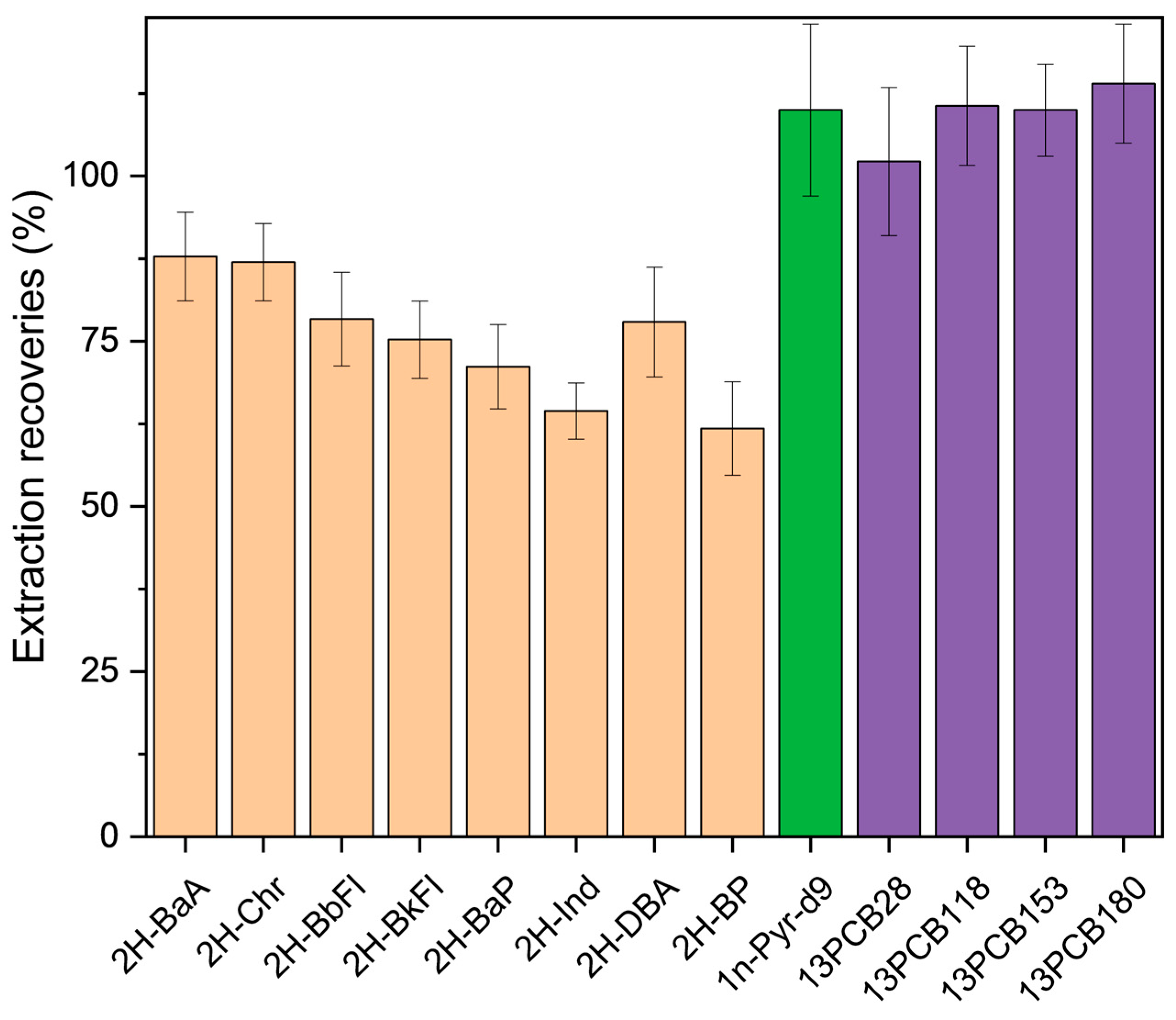
3.3.1. Extraction Recoveries
3.3.2. Linearity and Sensitivity
3.3.3. Precision
3.4. Greenness Evaluation and Economical Aspects
3.5. Real Samples
3.5.1. Analysis
3.5.2. Risk Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heyde, B.J.; Braun, M.; Soufi, L.; Lüneberg, K.; Gallego, S.; Amelung, W.; Axtmann, K.; Bierbaum, G.; Glaeser, S.P.; Grohmann, E. Transition from irrigation with untreated wastewater to treated wastewater and associated benefits and risks. npj Clean Water 2025, 8, 6. [Google Scholar] [CrossRef]
- Helmecke, M.; Fries, E.; Schulte, C. Regulating water reuse for agricultural irrigation: Risks related to organic micro-contaminants. Environ. Sci. Eur. 2020, 32, 4. [Google Scholar] [CrossRef]
- Zhang, J.; Fan, S.; Du, X.; Yang, J.; Wang, W.; Hou, H. Accumulation, allocation, and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in soil-Brassica chinensis system. PLoS ONE 2015, 10, e0115863. [Google Scholar] [CrossRef]
- Möller, L.; Lax, I.; Eriksson, L.C. Nitrated polycyclic aromatic hydrocarbons: A risk assessment for the urban citizen. Environ. Health Perspect. 1993, 101, 309–315. [Google Scholar][Green Version]
- Dybing, E.; Schwarze, P.E.; Nafstad, P.; Victorin, K.; Penning, T.M. Polycyclic aromatic hydrocarbons in ambient air and cancer. Air Pollut. Cancer 2013, 161. [Google Scholar]
- Olatunji, O.S. Evaluation of selected polychlorinated biphenyls (PCBs) congeners and dichlorodiphenyltrichloroethane (DDT) in fresh root and leafy vegetables using GC-MS. Sci. Rep. 2019, 9, 538. [Google Scholar] [CrossRef]
- Khillare, P.; Jyethi, D.S.; Sarkar, S. Health risk assessment of polycyclic aromatic hydrocarbons and heavy metals via dietary intake of vegetables grown in the vicinity of thermal power plants. Food Chem. Toxicol. 2012, 50, 1642–1652. [Google Scholar] [CrossRef]
- Lee, H.; Cho, Y.; Jung, G.; Kim, H.; Jeong, W. Comparison of recovery efficiency and matrix effect reduction in pesticide residue analysis: QuEChERS with d-SPE, SPE, and FaPEx in apples and Korean cabbage. Anal. Methods 2023, 15, 3709–3716. [Google Scholar] [CrossRef]
- Aguas, Y.; Hincapie, M.; Martínez-Piernas, A.B.; Agüera, A.; Fernández-Ibáñez, P.; Nahim-Granados, S.; Polo-López, M.I. Reclamation of real urban wastewater using solar advanced oxidation processes: An assessment of microbial pathogens and 74 organic microcontaminants uptake in lettuce and radish. Environ. Sci. Technol. 2019, 53, 9705–9714. [Google Scholar] [CrossRef]
- Rivoira, L.; Del Bubba, M.; Cecconi, G.; Castiglioni, M.; Testa, V.; Isola, M.; Bruzzoniti, M.C. Experimental design and multiple response optimization for the extraction and quantitation of thirty-four priority organic micropollutants in tomatoes through the QuEChERS approach. Separations 2023, 10, 174. [Google Scholar] [CrossRef]
- Calderón-Preciado, D.; Matamoros, V.; Savé, R.; Muñoz, P.; Biel, C.; Bayona, J. Uptake of microcontaminants by crops irrigated with reclaimed water and groundwater under real field greenhouse conditions. Environ. Sci. Pollut. Res. 2013, 20, 3629–3638. [Google Scholar] [CrossRef]
- Bruzzoniti, M.C.; Rivoira, L.; Castiglioni, M.; Cagno, E.; Kettab, A.; Fibbi, D.; Del Bubba, M. Optimization and validation of a method based on QuEChERS extraction and gas chromatographic-mass spectrometric analysis for the determination of polycyclic aromatic hydrocarbons and polychlorinated biphenyls in olive fruits irrigated with treated wastewaters. Separations 2022, 9, 82. [Google Scholar]
- Li, Y.; Zhou, W.; Jiang, R.W.; Pawliszyn, J. Solid-phase microextraction with recessed matrix compatible coating for in situ sampling of per-and polyfluoroalkyl substances in meat. Food Chem. 2025, 480, 143891. [Google Scholar] [CrossRef]
- Bruzzoniti, M.C.; Checchini, L.; De Carlo, R.M.; Orlandini, S.; Rivoira, L.; Del Bubba, M. QuEChERS sample preparation for the determination of pesticides and other organic residues in environmental matrices: A critical review. Anal. Bioanal. Chem. 2014, 406, 4089–4116. [Google Scholar] [CrossRef]
- Zhao, L.; Meng, C.-K. Optimized Method Development of Large Volume Injection for GC/MS/MS of Food Pesticides; Agilent Technologies, 2012. Available online: https://www.agilent.com/cs/library/applications/5991-1196EN.pdf (accessed on 20 June 2025).
- Al-Alam, J.; Fajloun, Z.; Chbani, A.; Millet, M. A multiresidue method for the analysis of 90 pesticides, 16 PAHs, and 22 PCBs in honey using QuEChERS–SPME. Anal. Bioanal. Chem. 2017, 409, 5157–5169. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency (EPA). Example Exposure Scenarios; EPA/600/R-03/036; National Center for Environmental Assessment: Washington, DC, USA, 2003. [Google Scholar]
- Agilent Technologies Inc. Solid Phase Microextraction Fundamentals. 2023. Available online: https://www.agilent.com/cs/library/technicaloverviews/public/te-solid-phase-microextraction-fundamentals-spme-arrow-5994-5775en-agilent.pdf (accessed on 22 May 2025).
- Merck Life Science. SPME for GC Analysis. 2020. Available online: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/product/documents/386/930/spme-gc-analysis-br1410en-ms.pdf (accessed on 22 May 2025).
- Pócsová, T.; Okanovič, S.; Hrouzková, S. Green Analytical Method Using Single-Drop Microextraction Followed by Gas Chromatography for Nitro Compound Detection in Environmental Water and Forensic Rinse Water. Molecules 2025, 30, 1894. [Google Scholar] [CrossRef]
- Galamba, N.; Paiva, A.; Barreiros, S.; Simões, P. Solubility of polar and nonpolar aromatic molecules in subcritical water: The role of the dielectric constant. J. Chem. Theory Comput. 2019, 15, 6277–6293. [Google Scholar] [CrossRef]
- Yabalak, E.; Akay, S.; Kayan, B.; Gizir, A.M.; Yang, Y. Solubility and decomposition of organic compounds in subcritical water. Molecules 2023, 28, 1000. [Google Scholar] [CrossRef]
- Terrigno, V.; Della Posta, S.; Pietrangeli, G.; Tonto, T.C.; Locato, V.; De Gara, L.; Fanali, C. Extraction and Analysis of Phenolic Compounds from Rocket: Development of a Green and Innovative DES-Based Extraction Method. Molecules 2025, 30, 1177. [Google Scholar] [CrossRef]
- Li, T.; Xu, J.; Wu, H.; Wang, G.; Dai, S.; Fan, J.; He, H.; Xiang, W. A saponification method for chlorophyll removal from microalgae biomass as oil feedstock. Mar. Drugs 2016, 14, 162. [Google Scholar] [CrossRef]
- Mol, H.G.; Rooseboom, A.; Van Dam, R.; Roding, M.; Arondeus, K.; Sunarto, S. Modification and re-validation of the ethyl acetate-based multi-residue method for pesticides in produce. Anal. Bioanal. Chem. 2007, 389, 1715–1754. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EU) 2023/915 of 25 April 2023 on Maximum Levels for Certain Contaminants in Food and Repealing Regulation (EC) No 1881/2006; 2023. Available online: https://eur-lex.europa.eu/eli/reg/2023/915/oj/eng (accessed on 20 June 2025).
- Guidance SANTE 11312/2021—Analytical Quality Control and Method Validation Procedures for Pesticide Residues Analysis in Food and Feed. 2022. Available online: https://www.accredia.it/wp-content/uploads/2021/02/SANTE_11312_2021.pdf (accessed on 20 June 2025).
- Jeong, J.; Koo, M.; Lee, J.-G. QuEChERS method development for the GC–MS analysis of polycyclic aromatic hydrocarbons in food. Food Sci. Biotechnol. 2025, 34, 2749–2761. [Google Scholar] [CrossRef]
- Paris, A.; Ledauphin, J.; Poinot, P.; Gaillard, J.-L. Polycyclic aromatic hydrocarbons in fruits and vegetables: Origin, analysis, and occurrence. Environ. Pollut. 2018, 234, 96–106. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Wojnowski, W.; Tobiszewski, M. AGREE—Analytical GREEnness metric approach and software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar] [CrossRef] [PubMed]
- European Commission. SCF/CS/CNTM/PAH/29 ADD1; 2002. Available online: https://www.google.com/url?sa=t&source=web&rct=j&opi=89978449&url=https://ec.europa.eu/food/fs/sc/scf/out153_en.pdf (accessed on 20 May 2025).
- U.S. Environmental Protection Agency, National Center for Environmental Assessment, Office of Research and Development. Update for Chapter 9 of the Exposure Factors Handbook. Intake of Fruits and Vegetables; EPA/600/R–18/098F; EPA: Washington, DC, USA, 2018. [Google Scholar]
- Rivoira, L.; Castiglioni, M.; Kettab, A.; Ouazzani, N.; Al-Karablieh, E.; Boujelben, N.; Fibbi, D.; Coppini, E.; Giordani, E.; Bubba, M.D. Impact of effluents from wastewater treatments reused for irrigation: Strawberry as case study. Environ. Eng. Manag. J. 2019, 18, 2133–2143. [Google Scholar]

| Analites | R2 | MDL (μg/kg) | MQL (μg/kg) |
|---|---|---|---|
| AcPY | 0.999 | 0.3 | 1.0 |
| AcPh | 0.997 | 0.2 | 0.7 |
| Flu | 0.998 | 0.5 | 1.3 |
| Phe | 0.998 | 0.2 | 0.6 |
| Ant | 0.999 | 0.1 | 0.4 |
| Flth | 0.997 | 0.3 | 1.3 |
| Pyr | 0.999 | 0.2 | 0.7 |
| BaA | 0.998 | 0.3 | 0.9 |
| Chr | 0.999 | 0.3 | 1.0 |
| BbFl | 0.989 | 0.2 | 0.7 |
| BkFl | 0.997 | 0.4 | 1.3 |
| BaP | 0.996 | 0.4 | 1.1 |
| Ind | 0.985 | 0.3 | 0.7 |
| DBA | 0.994 | 0.3 | 1.9 |
| BP | 0.988 | 0.3 | 0.8 |
| 1-N-Naph | 0.997 | 2.4 | 7.2 |
| 2-N-Flu | 0.996 | 6.7 | 20.3 |
| 1-N-Pyr | 0.995 | 3.4 | 10.2 |
| PCB11 | 0.994 | 0.8 | 2.5 |
| PCB15 | 0.999 | 0.8 | 2.5 |
| PCB28 | 0.998 | 0.8 | 2.4 |
| PCB52 | 0.997 | 0.9 | 2.6 |
| PCB101 | 0.995 | 0.7 | 2.2 |
| PCB81 | 0.998 | 0.7 | 2.3 |
| PCB118 + 123 | 0.996 | 0.5 | 1.6 |
| PCB138 | 0.997 | 0.4 | 1.2 |
| PCB153 | 0.996 | 0.4 | 1.3 |
| PCB167 | 0.999 | 0.2 | 0.7 |
| PCB180 | 0.999 | 0.4 | 1.2 |
| PCB169 | 0.997 | 0.5 | 1.7 |
| PCB189 | 0.997 | 0.5 | 1.5 |
| Intraday Precision [RSD%] | Interday Precision [RSD%] | |
|---|---|---|
| BaA-d12 | 5 | 10 |
| Chr-d12 | 12 | 12 |
| BbFl-d12 | 8 | 10 |
| BkFl-d12 | 9 | 12 |
| BaP-d12 | 12 | 13 |
| Ind-d12 | 18 | 16 |
| DBA-d14 | 18 | 15 |
| BP-d12 | 18 | 17 |
| 13 C12-PCB28 | 8 | 9 |
| 13 C12-PCB52 | 9 | 9 |
| 13 C12-PCB118 | 8 | 9 |
| 13 C12-PCB153 | 20 | 7 |
| RW1 | RW2 | RW3 | RW4 | RW5 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AcPy | 9.0 | ± | 1.1 | 7.9 | ± | 0.5 | 8.0 | ± | 0.2 | 5.9 | ± | 0.2 | 5.9 | ± | 2.0 |
| AcPh | 6.3 | ± | 2.1 | 8.4 | ± | 0.9 | 6.8 | ± | 0.3 | 4.9 | ± | 0.1 | 3.0 | ± | 0.9 |
| Flu | 18.1 | ± | 0.4 | 16.2 | ± | 0.8 | 18.2 | ± | 2.6 | 12.9 | ± | 0.3 | 10.4 | ± | 2.6 |
| Phe | 60.8 | ± | 4.6 | 45.5 | ± | 1.6 | 50.8 | ± | 5.5 | 37.2 | ± | 0.5 | 30.9 | ± | 11.6 |
| Ant | 5.1 | ± | 0.1 | 4.9 | ± | 0.6 | 5.8 | ± | 0.4 | 4.4 | ± | 0.2 | 3.2 | ± | 0.1 |
| Flth | 6.1 | ± | 0.1 | 3.8 | ± | 0.3 | 5.0 | ± | 0.7 | 3.0 | ± | 0.5 | 3.4 | ± | 1.0 |
| Pyr | 5.5 | ± | 0.1 | 3.3 | ± | 0.2 | 4.9 | ± | 0.6 | 2.8 | ± | 0.3 | 3.1 | ± | 0.8 |
| BaA | <MQL | <MQL | <MQL | <MQL | <MQL | ||||||||||
| RW6 | RW7 | RW8 | C1 | ||||||||||||
| AcPy | 6.1 | ± | 0.6 | 11.6 | ± | 0.2 | 10.2 | ± | 0.8 | 11.7 | ± | 0.1 | |||
| AcPh | 3.8 | ± | 0.0 | 7.9 | ± | 0.3 | 7.5 | ± | 1.6 | 13.3 | ± | 0.1 | |||
| Flu | 11.2 | ± | 0.5 | 27.7 | ± | 0.8 | 26.4 | ± | 3.1 | 13.4 | ± | 0.2 | |||
| Phe | 26.0 | ± | 2.4 | 77.8 | ± | 4.9 | 69.0 | ± | 5.0 | 14.3 | ± | 1.3 | |||
| Ant | 3.3 | ± | 0.4 | 7.6 | ± | 0.2 | 8.3 | ± | 0.1 | 5.5 | ± | 0.1 | |||
| Flth | 1.6 | ± | 0.4 | 7.6 | ± | 0.6 | 6.3 | ± | 0.1 | 10.4 | ± | 0.1 | |||
| Pyr | 2.1 | ± | 0.2 | 6.8 | ± | 0.4 | 5.5 | ± | 0.0 | 18.0 | ± | 0.1 | |||
| BaA | <MQL | 1.1 | ± | 0.0 | 1.1 | ± | 0.0 | <MQL | |||||||
| RW1 | RW2 | RW3 | RW4 | RW5 | |
|---|---|---|---|---|---|
| Cancer Risk | 1.3 × 10−8 | 1.3 × 10−8 | 2.0 × 10−8 | 9.4 × 10−9 | 7.0 × 10−9 |
| RW6 | RW7 | RW8 | C1 | ||
| Cancer Risk | 7.2 × 10−9 | 2.4 × 10−8 | 2.3 × 10−8 | 1.6 × 10−8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivoira, L.; Di Bonito, S.; Libonati, V.; Del Bubba, M.; Beldean-Galea, M.S.; Bruzzoniti, M.C. Residues of Priority Organic Micropollutants in Eruca vesicaria (Rocket) Irrigated by Reclaimed Wastewater: Optimization of a QuEChERS SPME-GC/MS Protocol and Risk Assessment. Foods 2025, 14, 2963. https://doi.org/10.3390/foods14172963
Rivoira L, Di Bonito S, Libonati V, Del Bubba M, Beldean-Galea MS, Bruzzoniti MC. Residues of Priority Organic Micropollutants in Eruca vesicaria (Rocket) Irrigated by Reclaimed Wastewater: Optimization of a QuEChERS SPME-GC/MS Protocol and Risk Assessment. Foods. 2025; 14(17):2963. https://doi.org/10.3390/foods14172963
Chicago/Turabian StyleRivoira, Luca, Simona Di Bonito, Veronica Libonati, Massimo Del Bubba, Mihail Simion Beldean-Galea, and Maria Concetta Bruzzoniti. 2025. "Residues of Priority Organic Micropollutants in Eruca vesicaria (Rocket) Irrigated by Reclaimed Wastewater: Optimization of a QuEChERS SPME-GC/MS Protocol and Risk Assessment" Foods 14, no. 17: 2963. https://doi.org/10.3390/foods14172963
APA StyleRivoira, L., Di Bonito, S., Libonati, V., Del Bubba, M., Beldean-Galea, M. S., & Bruzzoniti, M. C. (2025). Residues of Priority Organic Micropollutants in Eruca vesicaria (Rocket) Irrigated by Reclaimed Wastewater: Optimization of a QuEChERS SPME-GC/MS Protocol and Risk Assessment. Foods, 14(17), 2963. https://doi.org/10.3390/foods14172963









