Curcumin-Mediated Photodynamic Treatment Enhances Storage Quality of Fresh Wolfberries via Antioxidant System Modulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Photosensitizer and Illumination Source Instrument
2.3. Sample Treatment
2.4. Evaluation of Fruit Decay Rate and Weight Loss Rate
2.5. Assessment of the Hardness of Fresh Wolfberry
2.6. Evaluation of the Hue of Fresh Wolfberry
2.7. Analysis of Total Soluble Solids (TSS), Titratable Acid (TA), and Solid-Acid Ratio (TSS/TA) in Fresh Wolfberry
2.8. Quantitative Analysis of Soluble Protein, Flavonoid, and Betaine Contents in Fresh Wolfberry
2.9. Assessment of MDA and LOX Levels in Fresh Wolfberry
2.10. Determination of H2O2 Content and Rate of •O2− Production in Fresh Wolfberry
2.11. Evaluation of SOD, CAT, and POD Enzymatic Activities in Fresh Wolfberry
2.12. Determination of ASA, GSH Contents and APX, GR, MDHAR, and DHAR Enzyme Activities in Fresh Wolfberry
2.13. Statistical Analysis
3. Results
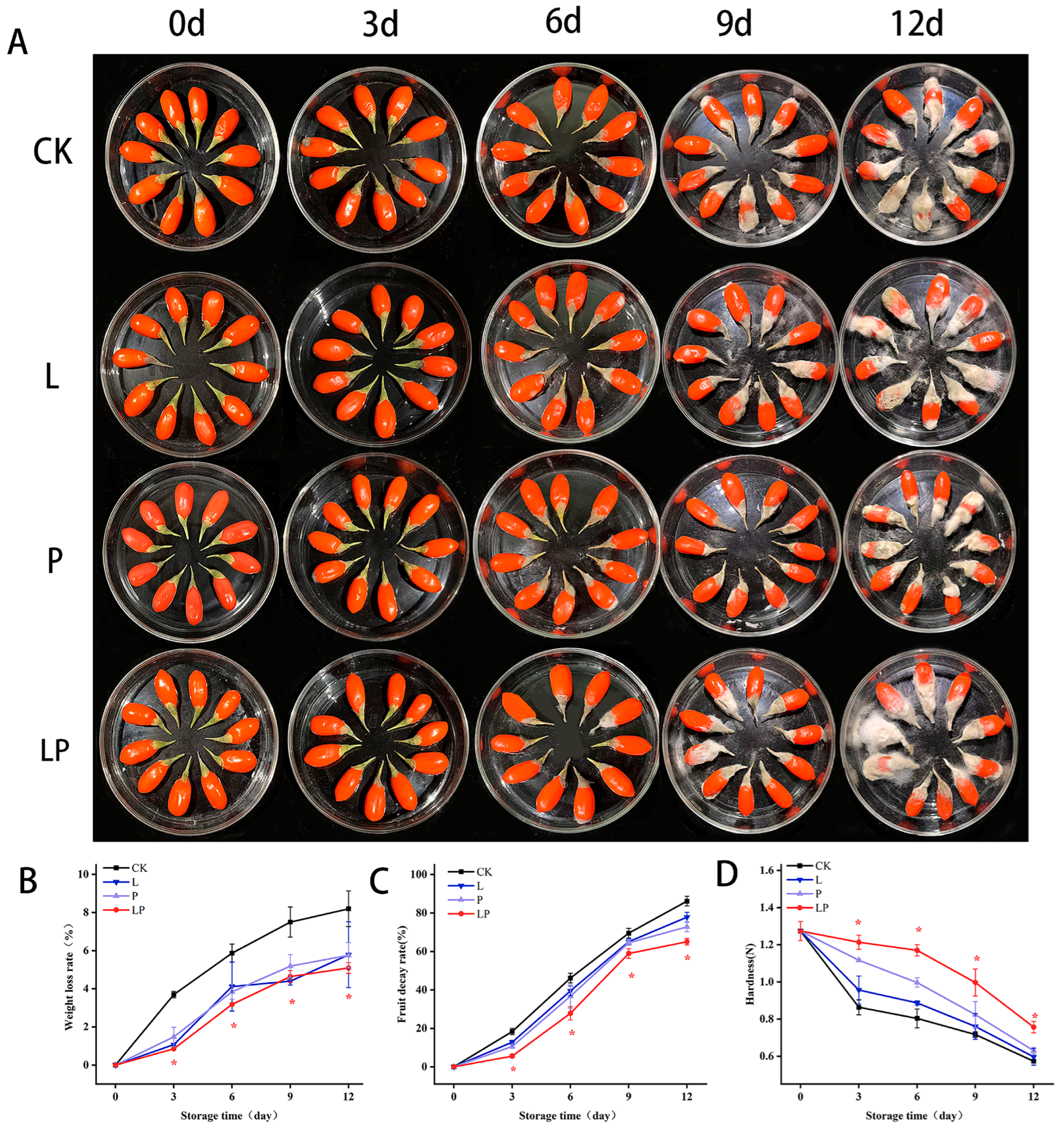
3.1. Appearance and Changes of Weight Loss, Hardness, and Decay of Fruit Wolfberry During Storage
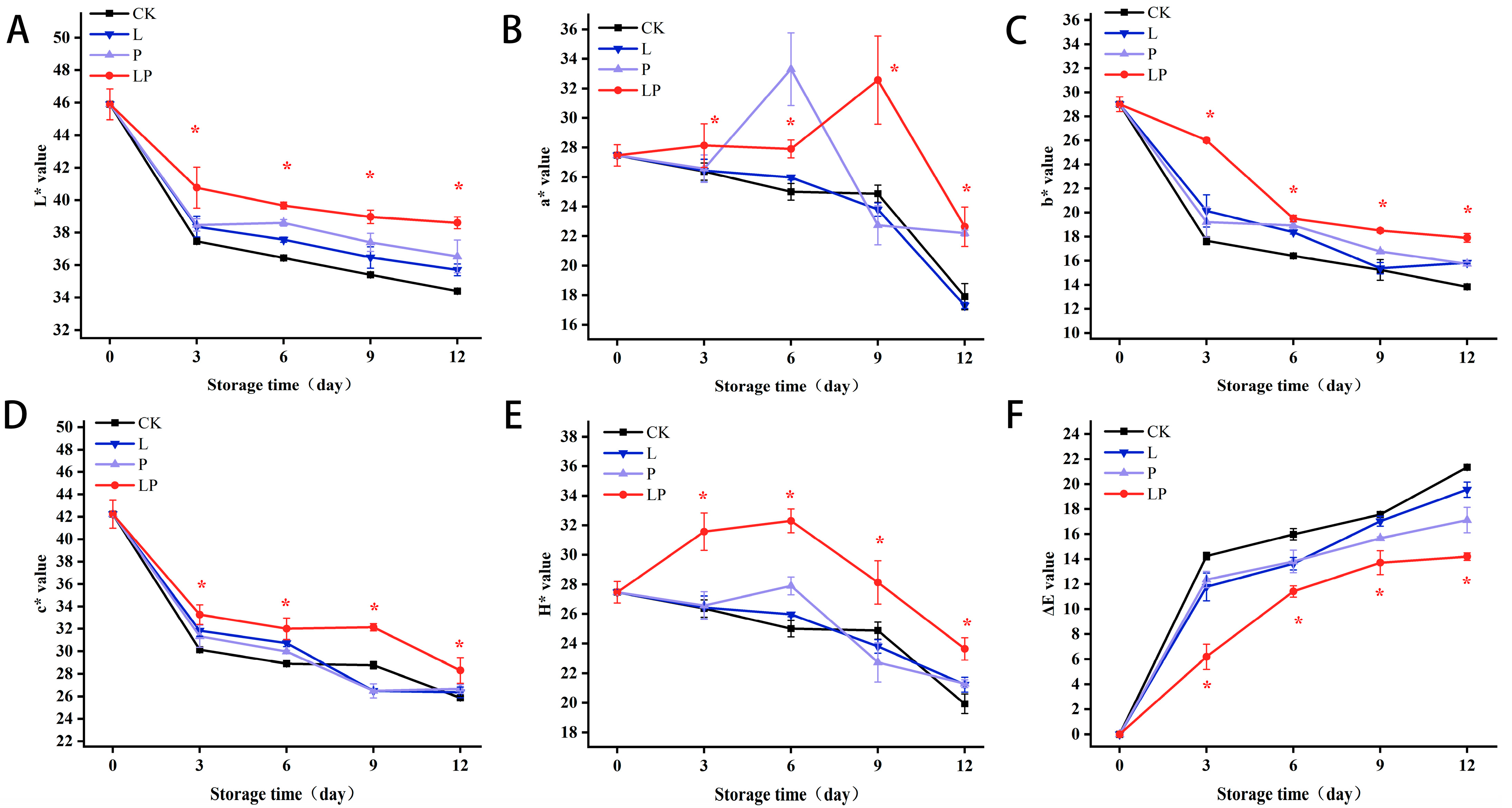
3.2. Color Changes
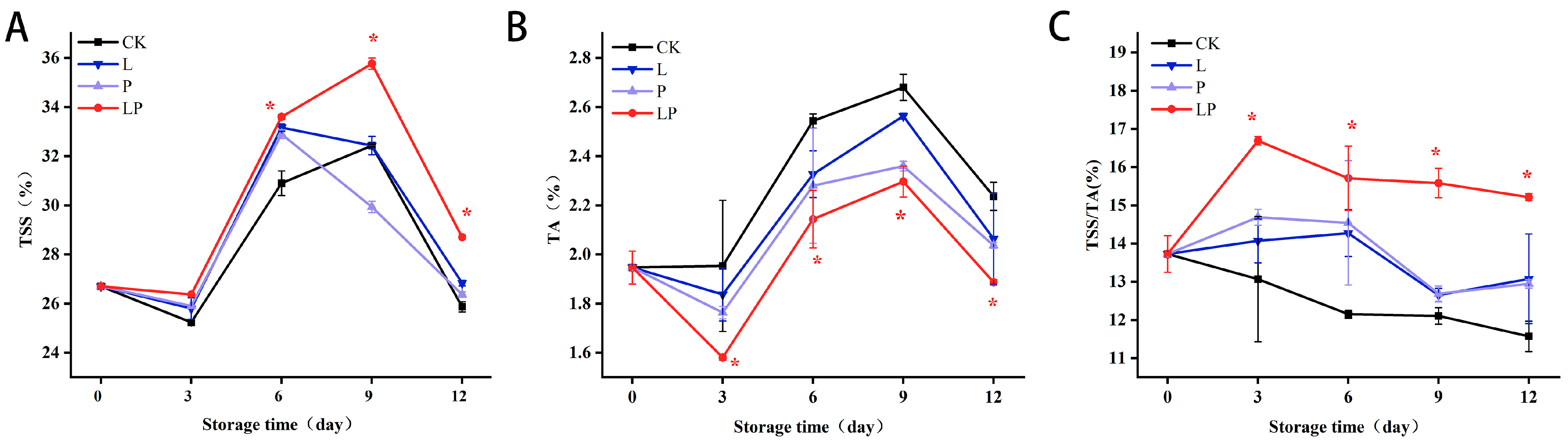
3.3. Effect on TSS, TA, and TSS/TA
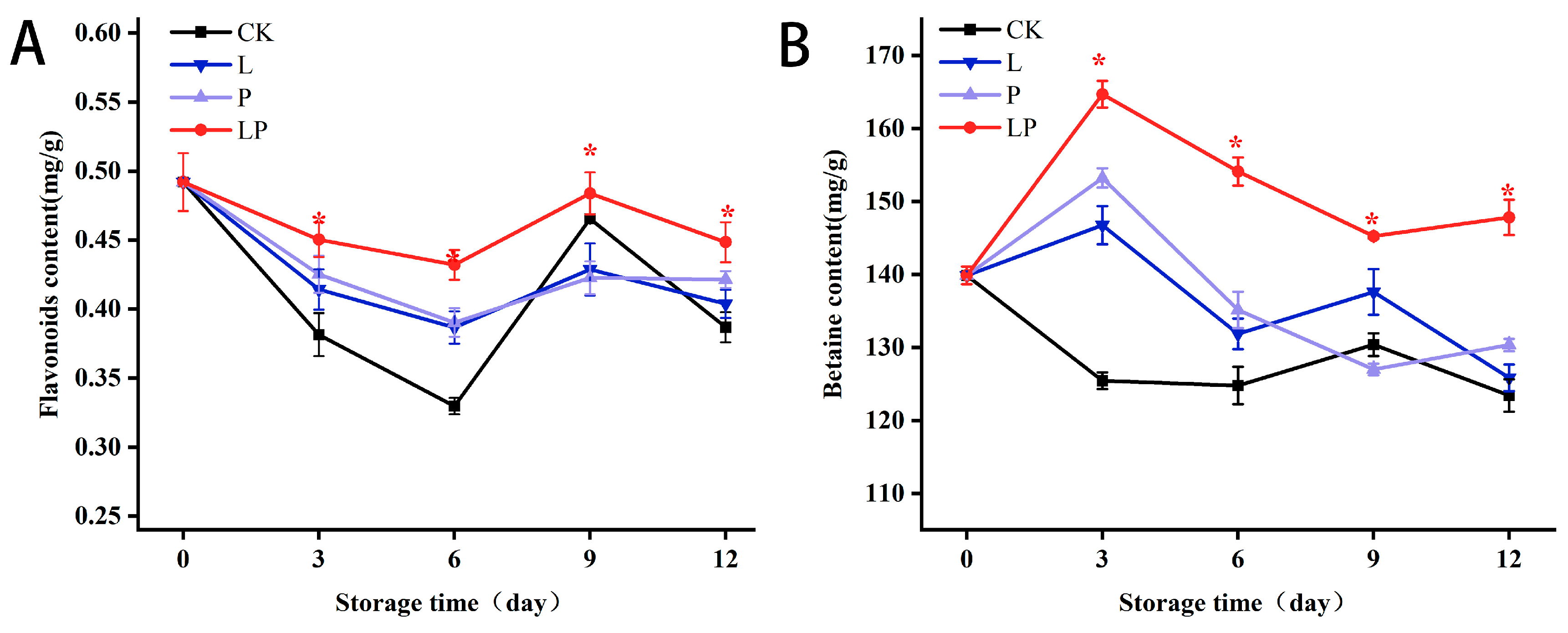
3.4. Effects on Flavonoids and Betaine Content
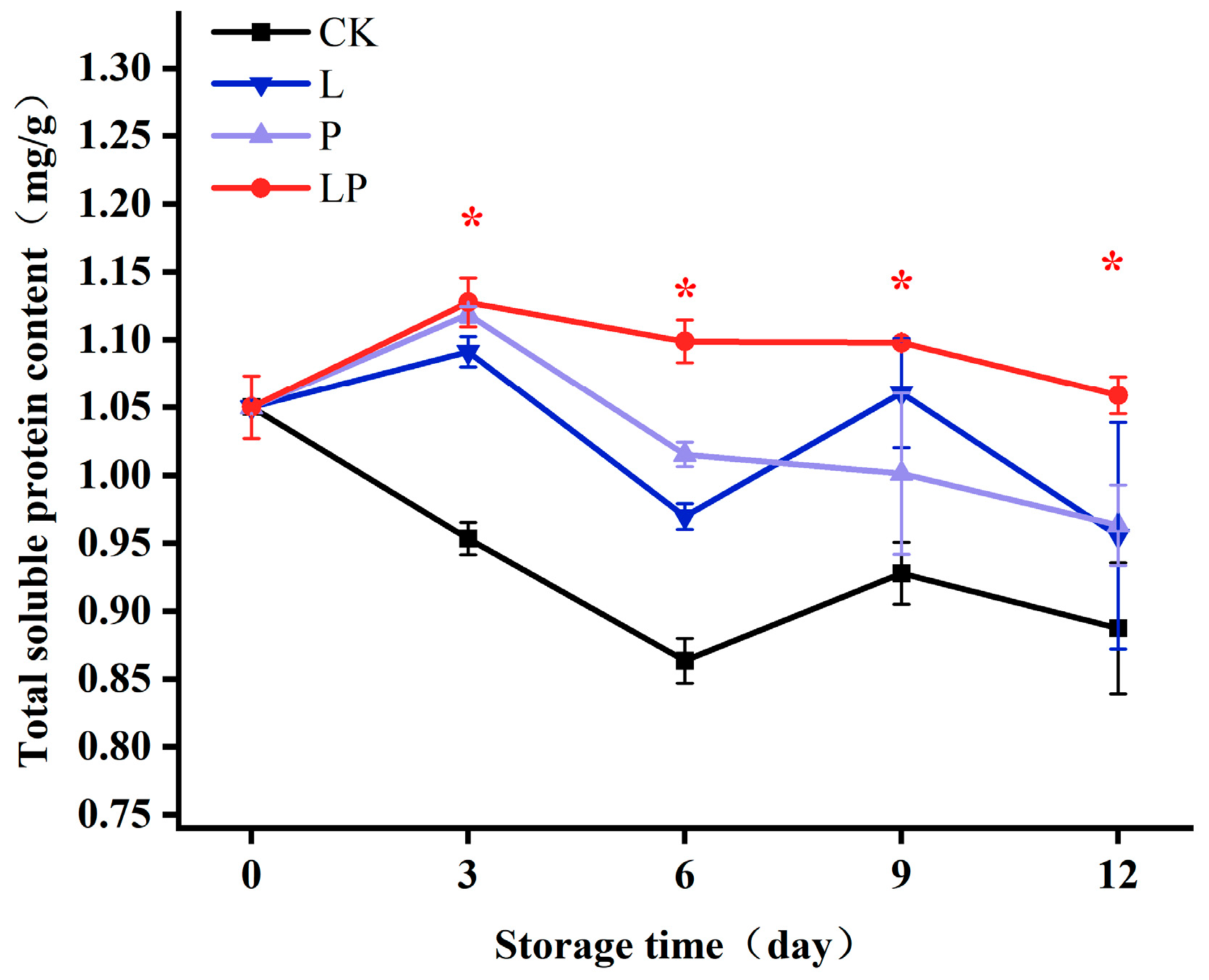
3.5. Effects on Soluble Protein
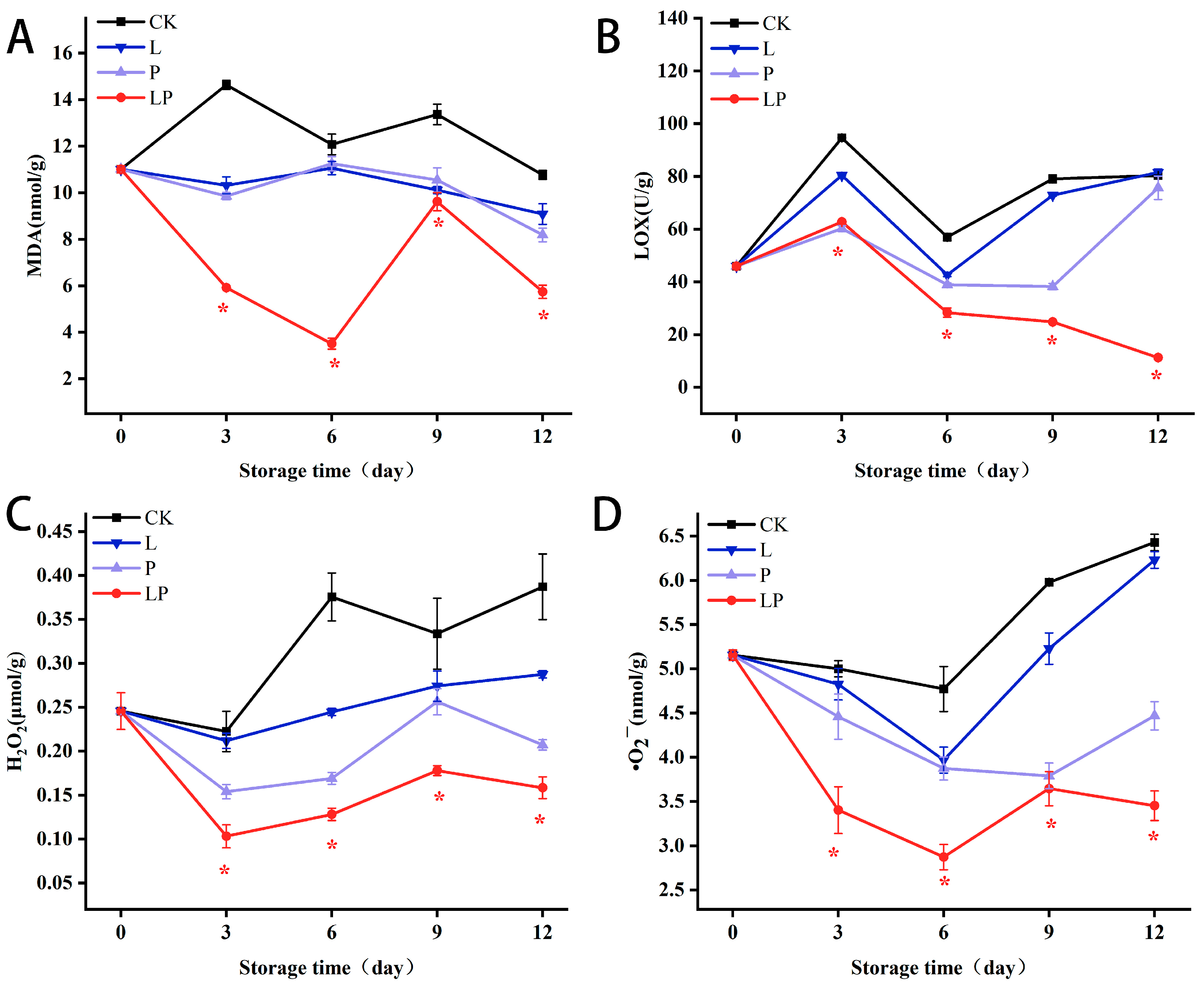
3.6. Effect on MDA, LOX, H2O2 and •O2−
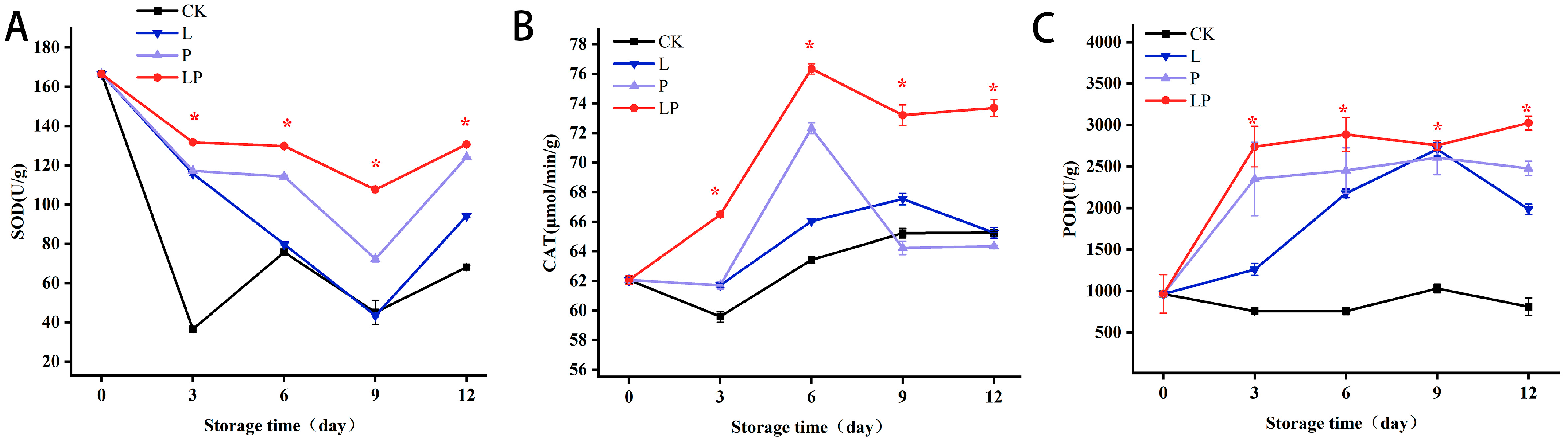
3.7. Effects on the Activities of SOD, CAT, and POD
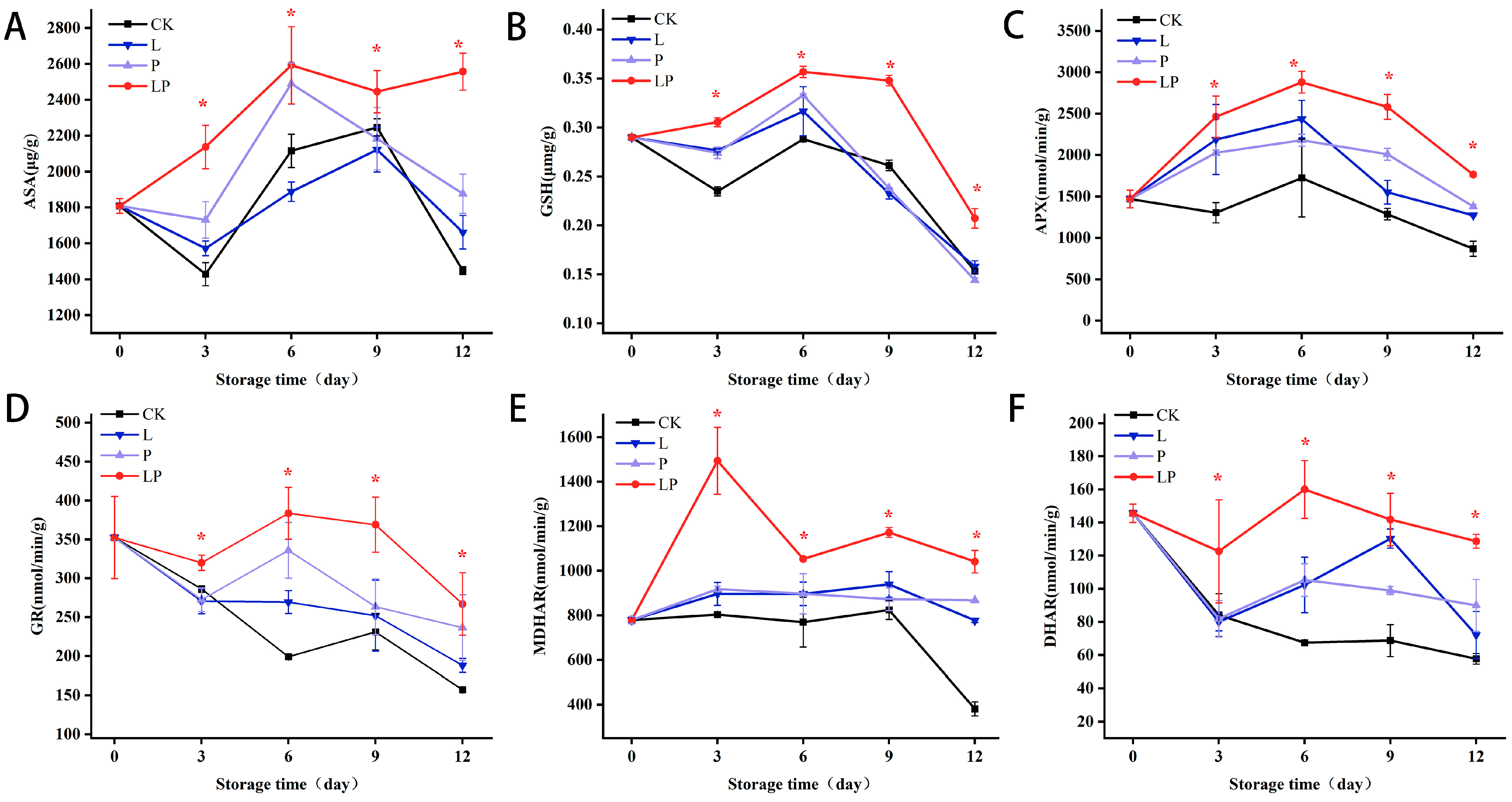
3.8. Effects on AsA and GSH Content, and Related Enzyme Activities in the AsA-GSH Cycle
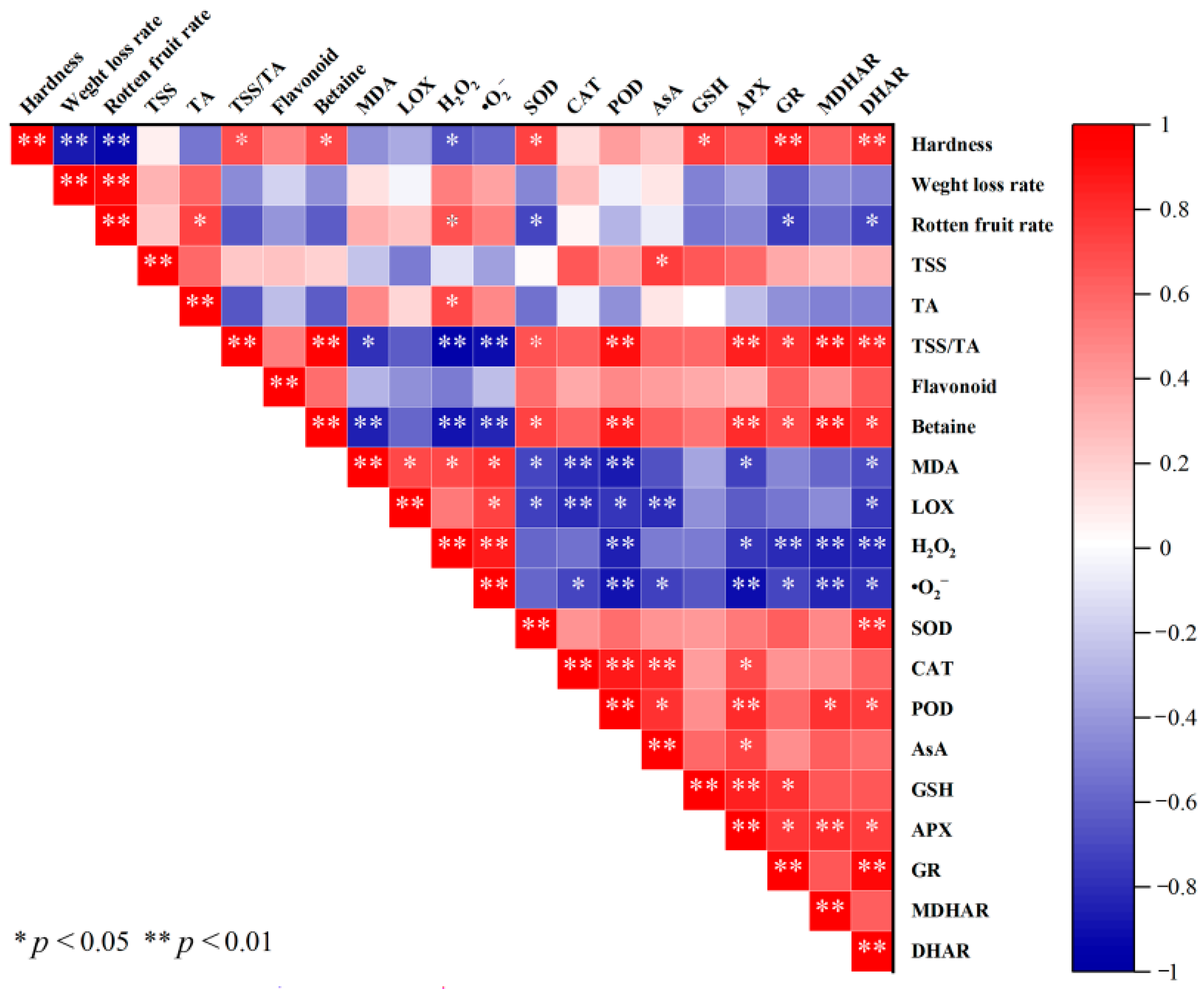
3.9. Analysis of Correlation Between Quality Indexes and Antioxidant System of Fresh Wolfberry During Storage Period
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, J.; Yan, Y.M.; Zhang, L.T.; Mi, J.; Yu, L.M.; Zhang, F.F.; Lu, L.; Luo, Q.; Li, X.Y.; Zhou, X.; et al. A comprehensive review of goji berry processing and utilization. Food Sci. Nutr. 2023, 11, 7445–7457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.Y.; Ma, Z.M.; Wang, J.J.; Wang, P.; Lu, D.Y.; Deng, S.F.; Lei, H.L.; Gao, Y.F.; Tao, Y.Y. Treatment with exogenous salicylic acid maintains quality, increases bioactive compounds, and enhances the antioxidant capacity of fresh goji (Lycium barbarum L.) fruit during storage. LWT-Food Sci. Technol. 2021, 140, 110837. [Google Scholar] [CrossRef]
- Zhou, Z.L.; Li, P.Z.; Chen, R.X.; Cai, X.Y.; Zhang, W.J.; Fan, P.H.; Su, J.Y. A Review of Curcumin-Mediated photodynamic bactericidal technology for food preservation: Limitations and improvement strategies. Food Microbiol. 2025, 131, 104802. [Google Scholar] [CrossRef]
- Li, H.R.; Ni, Y.S.; Zhao, J.S.; Li, Y.M.; Xu, B.C. Photodynamic inactivation of edible photosensitizers for fresh food preservation: Comprehensive mechanism of action and enhancement strategies. Compr. Rev. Food Sci. Food Saf. 2024, 23, e70006. [Google Scholar] [CrossRef]
- Yang, M.; Chao, H.J.; Hou, Z.H.; Wang, L.L.; Xu, W.Z.; Zhao, X. Antimicrobial activity of octyl gallate nanoemulsion combined with photodynamic technology and its effect on food preservation. Int. J. Food Microbiol. 2025, 429, 111023. [Google Scholar] [CrossRef]
- Dong, L.; Qin, J.R.; Tai, L.Y.; Mou, K.Y.; Liao, X.J.; Chen, F.; Hu, X.S. Inactivation of Bacillus subtilis by curcumin-mediated photodynamic technology through inducing oxidative stress response. Microorganisms 2022, 10, 802. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.L.; Chen, L.; Li, S.J.; Feng, K.L.; Liu, G.; Dong, H.; Xu, G.Z.; Ou, H.J.; Liu, Y.; Zhao, Y.; et al. Antifungal activity of curcumin-mediated photodynamic inactivation against Fusarium graminearum on maize. Grain Oil Sci. Technol. 2025, 8, 21–31. [Google Scholar] [CrossRef]
- Zhi, J.J.; Tang, Q.J.; Wu, S.J.; Kong, B.; Jiang, J.L.; Li, Z.J.; Wang, Y.M.; Xue, C.H. Degradation of curcumin-mediated photodynamic technology (PDT) on polycyclic aromatic hydrocarbons in oysters and toxicity evaluation of PDT-treated oysters. Int. J. Food Sci. Technol. 2022, 57, 7194–7204. [Google Scholar] [CrossRef]
- Long, Y.H.; Sun, Y.; Zhou, B.; Zhu, G.; Chen, X.L.; Qi, Y.J.; Wang, K. Photosensitization of riboflavin reduces the susceptibility to gray mold in postharvest kiwifruit. Postharvest Biol. Technol. 2024, 212, 112836. [Google Scholar] [CrossRef]
- Bhavya, M.L.; Hebbar, H.U. Sono-photodynamic inactivation of Escherichia coli and Staphylococcus aureus in orange juice. Ultrason. Sonochem. 2019, 57, 108–115. [Google Scholar] [CrossRef]
- Zou, Y.; Yu, Y.S.; Cheng, L.N.; Li, L.; Zou, B.; Wu, J.J.; Zhou, W.; Li, J.; Xu, Y.J. Effects of curcumin-based photodynamic treatment on quality attributes of fresh-cut pineapple. LWT-Food Sci. Technol. 2021, 141, 110902. [Google Scholar] [CrossRef]
- Lee, I.H.; Cho, E.R.; Kang, D.H. The effect of quercetin mediated photodynamic inactivation on apple juice properties at different temperature and its bactericidal mechanism. Food Control. 2023, 144, 109362. [Google Scholar] [CrossRef]
- Teng, X.X.; Zhang, M.; Mujumdar, A.S. Phototreatment (below 1100 nm) improving quality attributes of fresh-cut fruits and vegetables: A review. Food Res. Int. 2023, 163, 112252. [Google Scholar] [CrossRef]
- Lin, Y.L.; Lai, D.N.; Wang, D.H.; Zhou, F.; Tan, B.K.; Zhang, Z.G.; Hu, J.M.; Lin, S.L. Application of curcumin-mediated antibacterial photodynamic technology for preservation of fresh Tremella fuciformis. LWT-Food Sci. Technol. 2021, 147, 111657. [Google Scholar] [CrossRef]
- Dou, J.F.; Kou, X.H.; Wu, C.E.; Fan, G.J.; Li, T.T.; Li, X.J.; Zhou, D.D.; Yan, Z.C.; Zhu, J.P. Recent advances and development of postharvest management research for fresh jujube fruit: A review. Sci. Hortic. 2023, 310, 111769. [Google Scholar] [CrossRef]
- Seididamyeh, M.; Netzel, M.E.; Mereddy, R.; Sultanbawa, Y. Curcumin-mediated photodynamic treatment to extend the postharvest shelf-life of strawberries. J. Food Sci. 2024, 89, 6616–6627. [Google Scholar] [CrossRef]
- Prasad, A.; Wynands, E.; Roche, S.M.; Romo-Bernal, C.; Allan, N.; Olson, M.; Levengood, S.; Andersen, R.; Loebel, N.; Sabino, C.P.; et al. Photodynamic inactivation of foodborne Bacteria: Screening of 32 potential photosensitizers. Foods 2024, 13, 453. [Google Scholar] [CrossRef]
- Song, L.L.; Zhang, F.; Yu, J.S.; Wei, C.L.; Han, Q.M.; Meng, X.H. Antifungal effect and possible mechanism of curcumin mediated photodynamic technology against Penicillium expansum. Postharvest Biol. Technol. 2020, 167, 111234. [Google Scholar] [CrossRef]
- Yuan, Y.; Liu, Q.Y.; Huang, Y.J.; Qi, M.Y.; Yan, H.Y.; Li, W.L.; Zhuang, H. Antibacterial efficacy and mechanisms of curcumin-based photodynamic treatment against Staphylococcus aureus and its application in juices. Molecules. 2022, 27, 7136. [Google Scholar] [CrossRef]
- Huang, J.M.; Chen, B.; Li, H.H.; Zeng, Q.H.; Wang, J.J.; Liu, H.Q.; Pan, Y.J.; Zhao, Y. Enhanced antibacterial and antibiofilm functions of the curcumin-mediated photodynamic inactivation against Listeria monocytogenes. Food Control. 2020, 108, 106886. [Google Scholar] [CrossRef]
- Ni, Z.J.; Xue, Y.; Wang, W.; Du, J.; Thakur, K.; Ma, W.P.; Wei, Z.J. Carbon dots-mediated photodynamic treatment reduces postharvest senescence and decay of grapes by regulating the antioxidant system. Foods 2024, 13, 2717. [Google Scholar] [CrossRef] [PubMed]
- Elam, E.; Lv, Y.M.; Wang, W.; Thakur, K.; Ma, W.P.; Ni, Z.J.; Wei, Z.J. Effects of nitric oxide on postharvest storage quality of Lycium barbarum fruit. Food Sci. Technol. 2022, 42, e84122. [Google Scholar] [CrossRef]
- Liu, C.H.; Zheng, H.H.; Sheng, K.L.; Liu, W.; Zheng, L. Effects of melatonin treatment on the postharvest quality of strawberry fruit. Postharvest Biol. Technol. 2018, 139, 47–55. [Google Scholar] [CrossRef]
- Xing, Y.G.; Yang, H.; Guo, X.L.; Bi, X.F.; Liu, X.C.; Xu, Q.L.; Wang, Q.; Li, W.X.; Li, X.L.; Shui, Y.R.; et al. Effect of chitosan/Nano-TiO2 composite coatings on the postharvest quality and physicochemical characteristics of mango fruits. Sci. Hortic. 2020, 263, 109135. [Google Scholar] [CrossRef]
- Yang, Q.Z.; Hou, J.; Wang, F.; Qi, Y.J.; Zhao, Q.F. Cold shock treatment alleviates pitting in sweet cherry fruit by enhancing antioxidant enzymes activity and regulating membrane lipid metabolism. J. Sci. Food Agric. 2025, 105, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.W.; Jing, G.Q.; Zhu, S.H. Nitric oxide (NO) involved in antioxidant enzyme gene regulation to delay mitochondrial damage in peach fruit. Postharvest Biol. Technol. 2022, 192, 111993. [Google Scholar] [CrossRef]
- Lu, X.H.; Zhang, H.J.; Zhang, N.; Dong, C.H.; Ji, H.P.; Yu, J.Z.; Ban, Z.J.; Yan, R.X.; Zhang, T.; Chen, C.K.; et al. Effects of ozone treatment on gene profiling involved in ASA-GSH cycle in postharvest cantaloupe. Sci. Hortic. 2023, 312, 111843. [Google Scholar] [CrossRef]
- Zhang, Y.T.; Li, S.L.; Deng, M.Y.; Gui, R.; Liu, Y.Q.; Chen, X.P.; Lin, Y.X.; Li, M.Y.; Wang, Y.; He, W.; et al. Blue light combined with salicylic acid treatment maintained the postharvest quality of strawberry fruit during refrigerated storage. Food Chem. X 2022, 15, 100384. [Google Scholar] [CrossRef]
- Du, Y.H.; Mi, S.N.; Wang, H.H.; Yuan, S.F.; Yang, F.W.; Yu, H.; Xie, Y.F.; Guo, Y.H.; Cheng, Y.L.; Yao, W.R. Intervention mechanisms of cold plasma pretreatment on the quality, antioxidants and reactive oxygen metabolism of fresh wolfberries during storage. Food Chem. 2024, 431, 137106. [Google Scholar] [CrossRef]
- Wang, J.J.; Zhao, L.N.K.; Tian, W.; Zhang, H.Y.; Wang, P.; Zhan, Q.; Fan, H.W.; Yu, X. Carvacrol maintains antioxidant capacity in goji fruit by increasing the content of bioactive compounds. LWT-Food Sci. Technol. 2024, 212, 117035. [Google Scholar] [CrossRef]
- Patiño, L.S.; Castellanos, D.A.; Herrera, A.O. Influence of 1-MCP and modified atmosphere packaging in the quality and preservation of fresh basil. Postharvest Biol. Technol. 2018, 136, 57–65. [Google Scholar] [CrossRef]
- Ban, Z.J.; Wei, W.W.; Yang, X.Z.; Feng, J.H.; Guan, J.F.; Li, L. Combination of heat treatment and chitosan coating to improve postharvest quality of wolfberry (Lycium barbarum). Int. J. Food Sci. Technol. 2015, 50, 1019–1025. [Google Scholar] [CrossRef]
- Chang, X.J.; Liang, Y.G.; Guo, T.J.; Wang, Y.; Yang, J.L. Combined treatment of acidic electrolyzed water and high-voltage electrostatic field improves the storage quality of huping jujube (Ziziphus jujuba Mill. cv. Huping). Foods 2023, 12, 2762. [Google Scholar] [CrossRef]
- He, X.L.; Wu, C.; Lu, L.; Yan, X.X.; Yu, H.; Kang, N.B. Influence of acidic electrolyzed water combined with vacuum precooling treatment on quality and antioxidant performance of fresh Lycium barbarum L. J. Food Process Preserv. 2022, 46, e17149. [Google Scholar] [CrossRef]
- Damas-Job, M.D.; Soriano-Melgar, L.D.A.; Rodríguez-Herrera, R.; Peralta-Rodríguez, R.D.; Rivera-Cabrera, F.; Martínez-Vazquez, D.G. Effect of broccoli fresh residues-based extracts on the postharvest quality of cherry tomato (Solanum lycopersicum L.) fruits. Sci. Hortic. 2023, 317, 112076. [Google Scholar] [CrossRef]
- Yang, J.N.; Yin, J.T.; Wang, K.; Zhao, L.; Yang, Z.B.; Cai, Y.T.; Lou, J.F.; Huang, C.; Shen, Q. Advanced technology in fruit preservation: Effects of nanoscale charged water particles on storage quality and reactive oxygen species in blueberries. Food Res. Int. 2024, 198, 115331. [Google Scholar] [CrossRef]
- Du, J.; Ni, Z.J.; Wang, W.; Thakur, K.; Ma, R.H.; Ma, W.P.; Wei, Z.J. Carbon dot-mediated photodynamic treatment improves the quality attributes of post-harvest Goji Berries (Lycium barbarum L.) via regulating the antioxidant system. Foods 2024, 13, 955. [Google Scholar] [CrossRef]
- Chen, Y.H.; Hung, Y.C.; Chen, M.Y.; Lin, M.S.; Lin, H.T. Enhanced storability of blueberries by acidic electrolyzed oxidizing water application may be mediated by regulating ROS metabolism. Food Chem. 2019, 270, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Anee, T.I.; Parvin, K.; Nahar, K.; AL Mahmud, J.; Fujita, M. Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 2019, 8, 384. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, H.; Zhang, L.A.; Sun, Q.J. Horseradish peroxidase-mediated synthesis of an antioxidant gallic acid-g-chitosan derivative and its preservation application in cherry tomatoes. RSC Adv. 2018, 8, 20363–20371. [Google Scholar] [CrossRef] [PubMed]
- Shen, A.; Zhang, T.Z.; Li, S.Z.; Xiao, M.R.; Tian, Z.J.; Zhang, J.; Lu, T.T.; Yang, W.W. Innovative chitosan-onion polysaccharide composite films: A study on the preservation effects on cherry tomatoes. J. Food Sci. 2024, 89, 5712–5723. [Google Scholar] [CrossRef]
- Li, T.T.; Yan, Z.C.; Li, Y.F.; Kou, X.H.; Wu, C.; Xu, D.Y.; Zhou, D.D.; Cong, K.P.; Fan, G.J.; Li, X.J. Mechanistic insights into the enhancement of storage quality characteristics of fresh goji berry through non-thermal optical treatments (UV-C and IPL). Food Chem. 2025, 479, 143662. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, Y.-F.; Ma, W.-P.; Ma, R.-H.; Thakur, K.; Ni, Z.-J.; Wang, W.; Wei, Z.-J. Curcumin-Mediated Photodynamic Treatment Enhances Storage Quality of Fresh Wolfberries via Antioxidant System Modulation. Foods 2025, 14, 2843. https://doi.org/10.3390/foods14162843
Shen Y-F, Ma W-P, Ma R-H, Thakur K, Ni Z-J, Wang W, Wei Z-J. Curcumin-Mediated Photodynamic Treatment Enhances Storage Quality of Fresh Wolfberries via Antioxidant System Modulation. Foods. 2025; 14(16):2843. https://doi.org/10.3390/foods14162843
Chicago/Turabian StyleShen, Yan-Fei, Wen-Ping Ma, Run-Hui Ma, Kiran Thakur, Zhi-Jing Ni, Wei Wang, and Zhao-Jun Wei. 2025. "Curcumin-Mediated Photodynamic Treatment Enhances Storage Quality of Fresh Wolfberries via Antioxidant System Modulation" Foods 14, no. 16: 2843. https://doi.org/10.3390/foods14162843
APA StyleShen, Y.-F., Ma, W.-P., Ma, R.-H., Thakur, K., Ni, Z.-J., Wang, W., & Wei, Z.-J. (2025). Curcumin-Mediated Photodynamic Treatment Enhances Storage Quality of Fresh Wolfberries via Antioxidant System Modulation. Foods, 14(16), 2843. https://doi.org/10.3390/foods14162843







