Physiochemical Characterization and Antioxidant Potential of Sorghum and Cork Oak as Valuable Additives to Traditional Trida Pasta
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Solvents
2.2. Raw Materials
2.3. Chemical Composition of Raw Materials
2.4. Pasta Preparation
2.5. Pasta Characterization
2.5.1. Measurement of the Length, Width, and Thickness of Pasta
2.5.2. Determination of Water Absorption Index and Water Solubility Index
2.6. Pasta Cooking Quality
2.6.1. Optimum Cooking Time
2.6.2. Water Absorption Capacity
2.6.3. Cooking Loss
2.7. Texture Profile Analysis
2.8. Color Profile Evaluation
2.9. Functional Components and Antioxidant Activity Analyses of Raw Materials and Pasta Products
2.9.1. Accelerated Solvent Extraction (ASE)
2.9.2. Qualitative Analysis of Selected Polyphenolic Compounds by High-Performance Liquid Chromatography Coupled to a Mass Spectrometer (HPLC/ESI-TOF-MS)
2.9.3. Determination of the Total Content of Polyphenolic Compounds
2.9.4. Determination of Total Flavonoid Content
2.9.5. Spectrophotometric Analysis of the Obtained Extracts to Determine Their Antioxidant Properties
2.9.6. TLC-DPPH Test of the Obtained Extracts
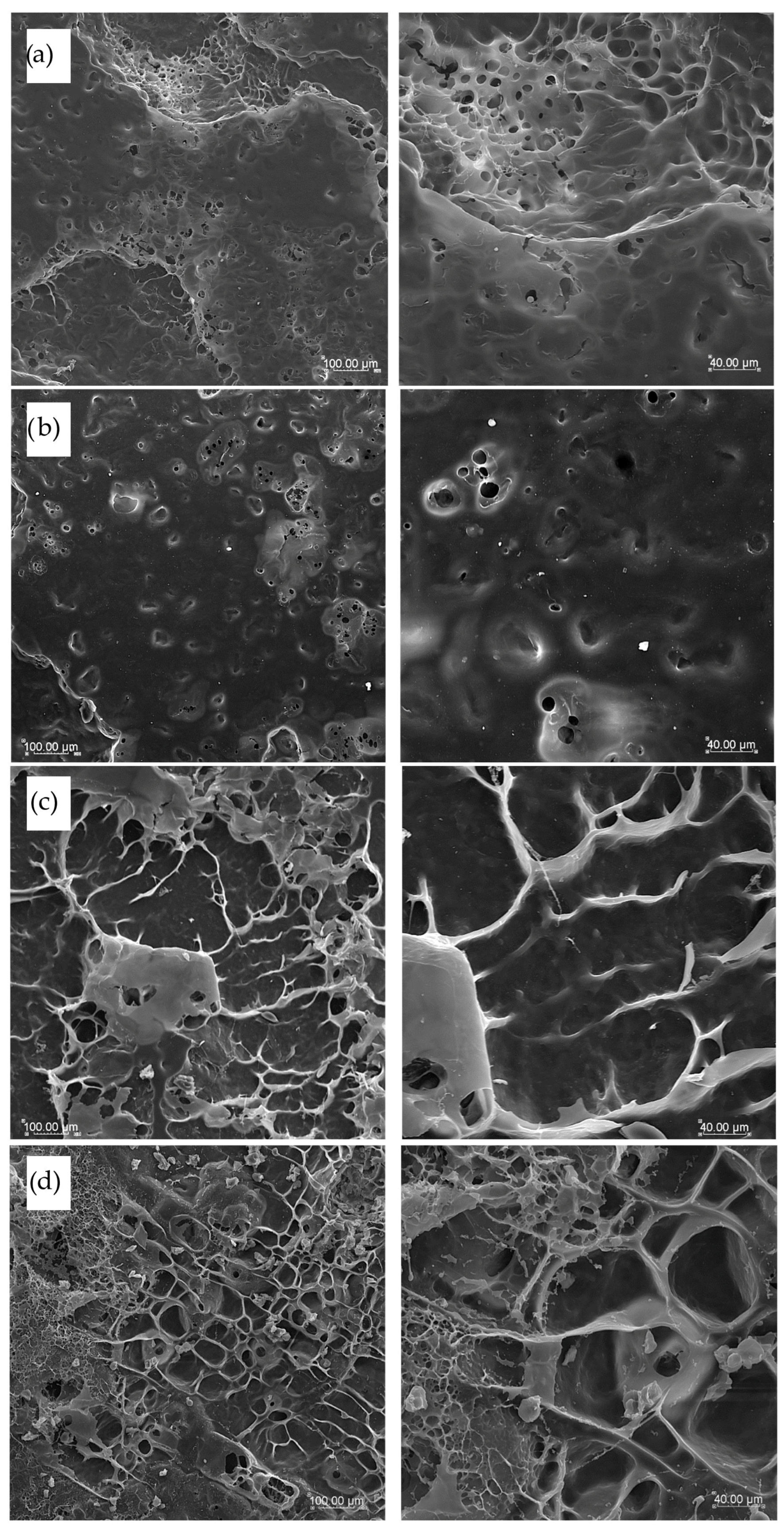
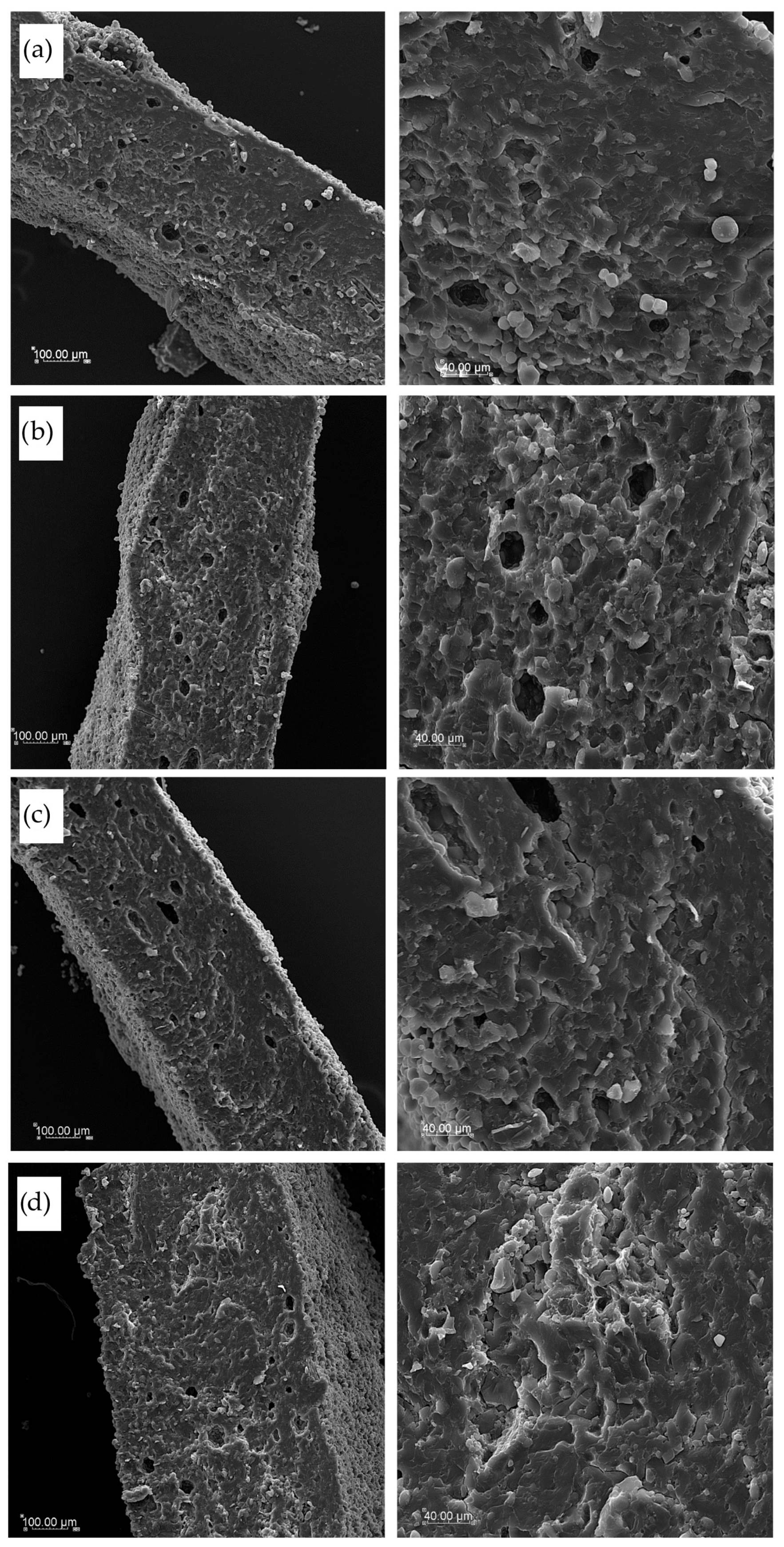
2.10. Microstructure Observations
2.11. Statistical Analysis
3. Results and Discussion
3.1. Results of Chemical Composition of Raw Materials
3.2. Pasta Characterization Results
3.2.1. Geometric Dimensions of Pasta Samples
3.2.2. Water Absorption Index (WAI) and Water Solubility Index (WSI) Results of Pasta
3.2.3. Results of Pasta Color Profile
3.3. Cooking Behavior of Pasta
3.4. Texture Profile of Cooked Pasta
3.5. Results of Functional Components and Antioxidant Activity of Raw Materials and Pasta Products
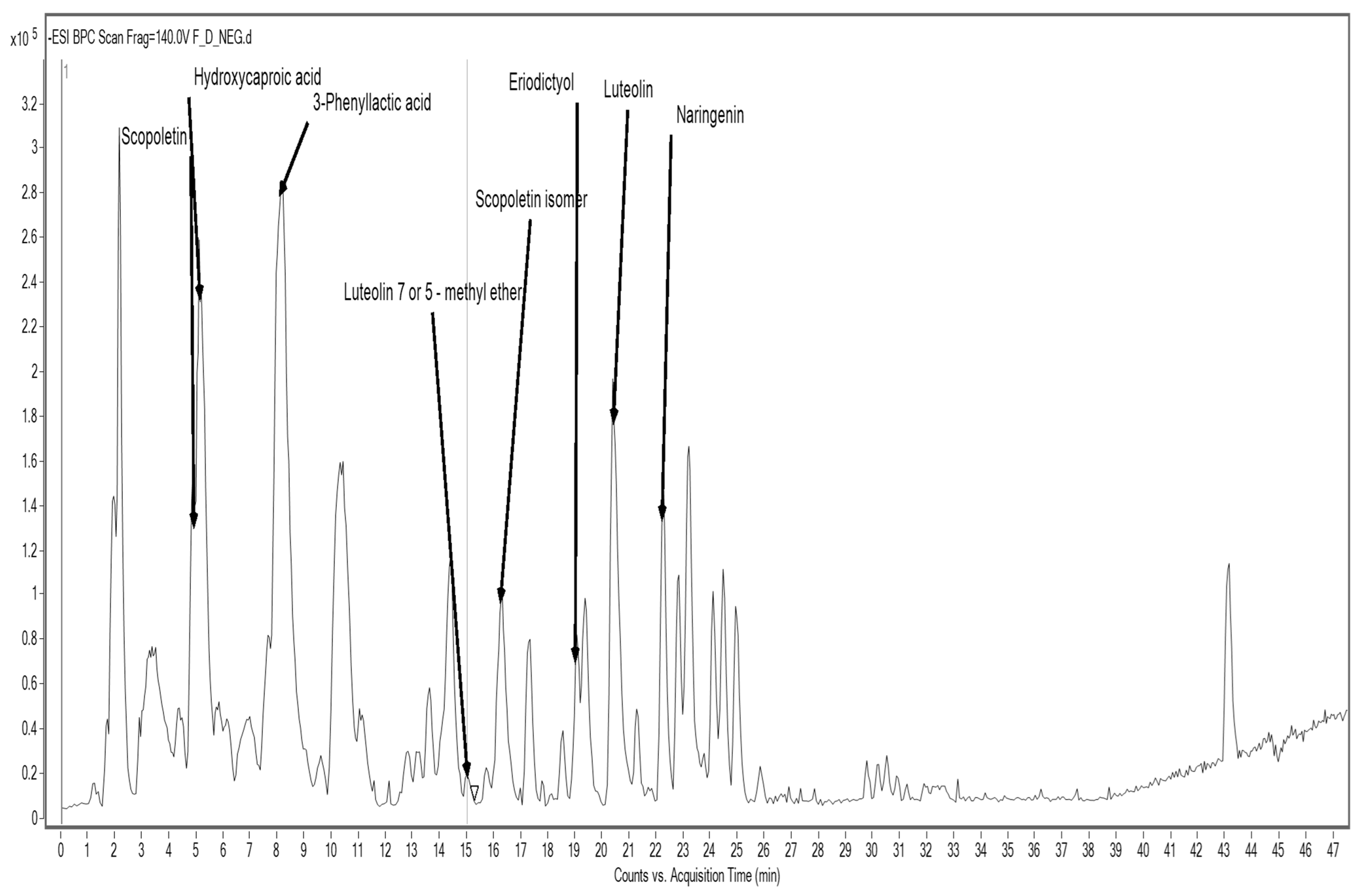
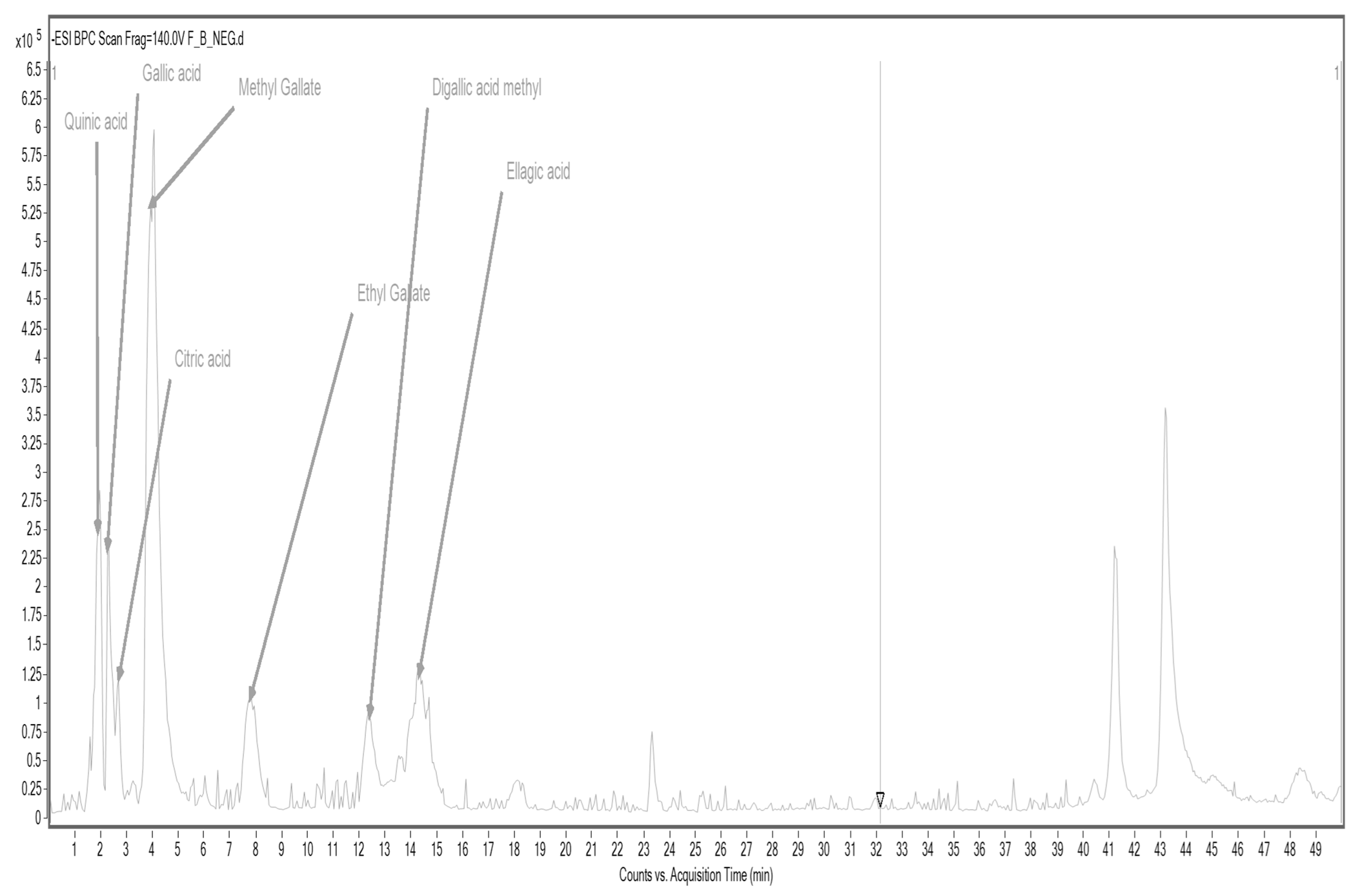
3.5.1. Results of Qualitative Analysis of Selected Polyphenolic Compounds in Raw Materials by HPLC/ESI-TOF-MS
3.5.2. Results of Total Content of Polyphenolic Compounds (TPC)
3.5.3. Results of Total Flavonoid Content (TFC)
3.5.4. Results of Antioxidant Properties of Raw Materials and Pasta by Spectrophotometric Method Using DPPH
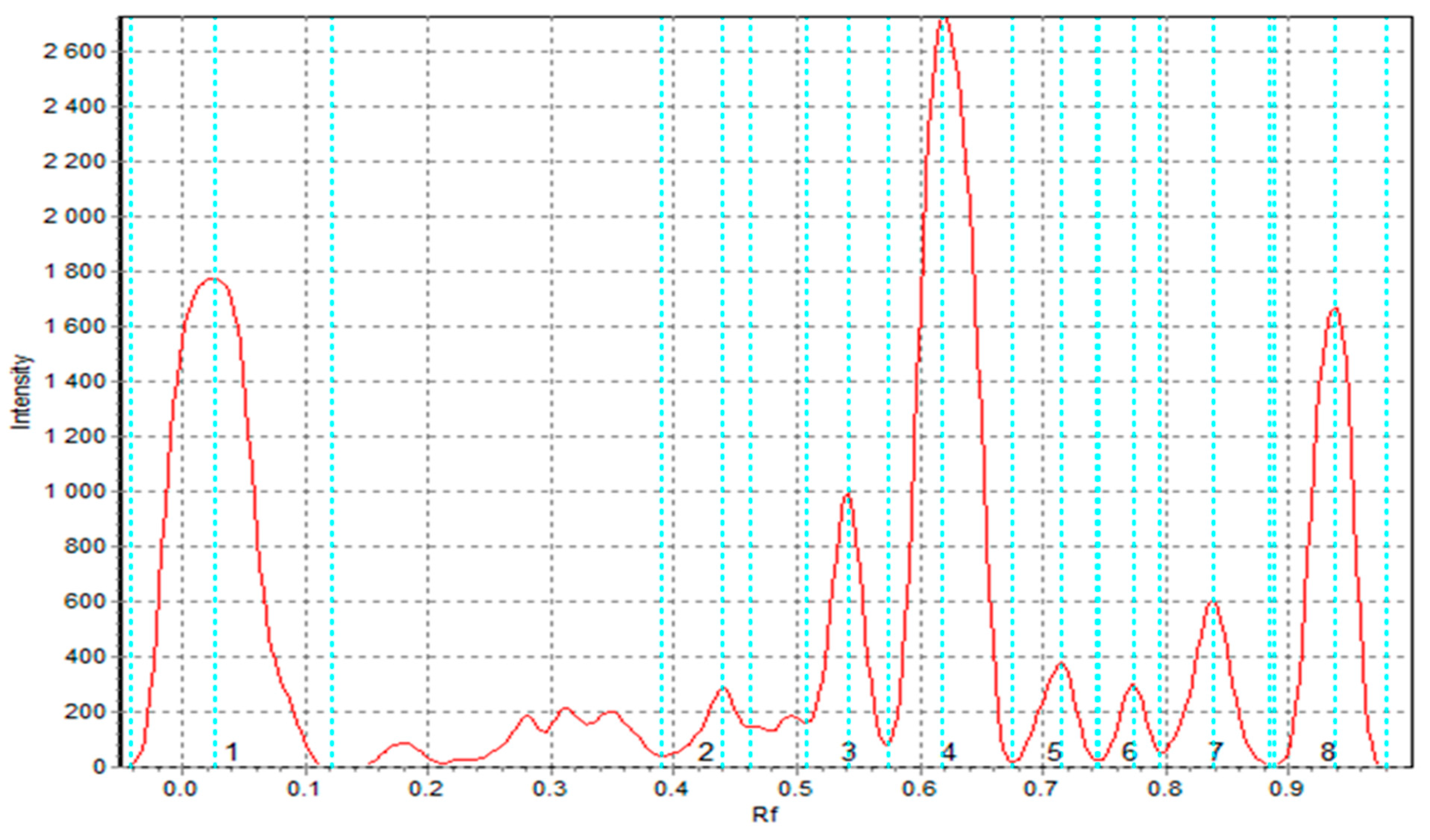
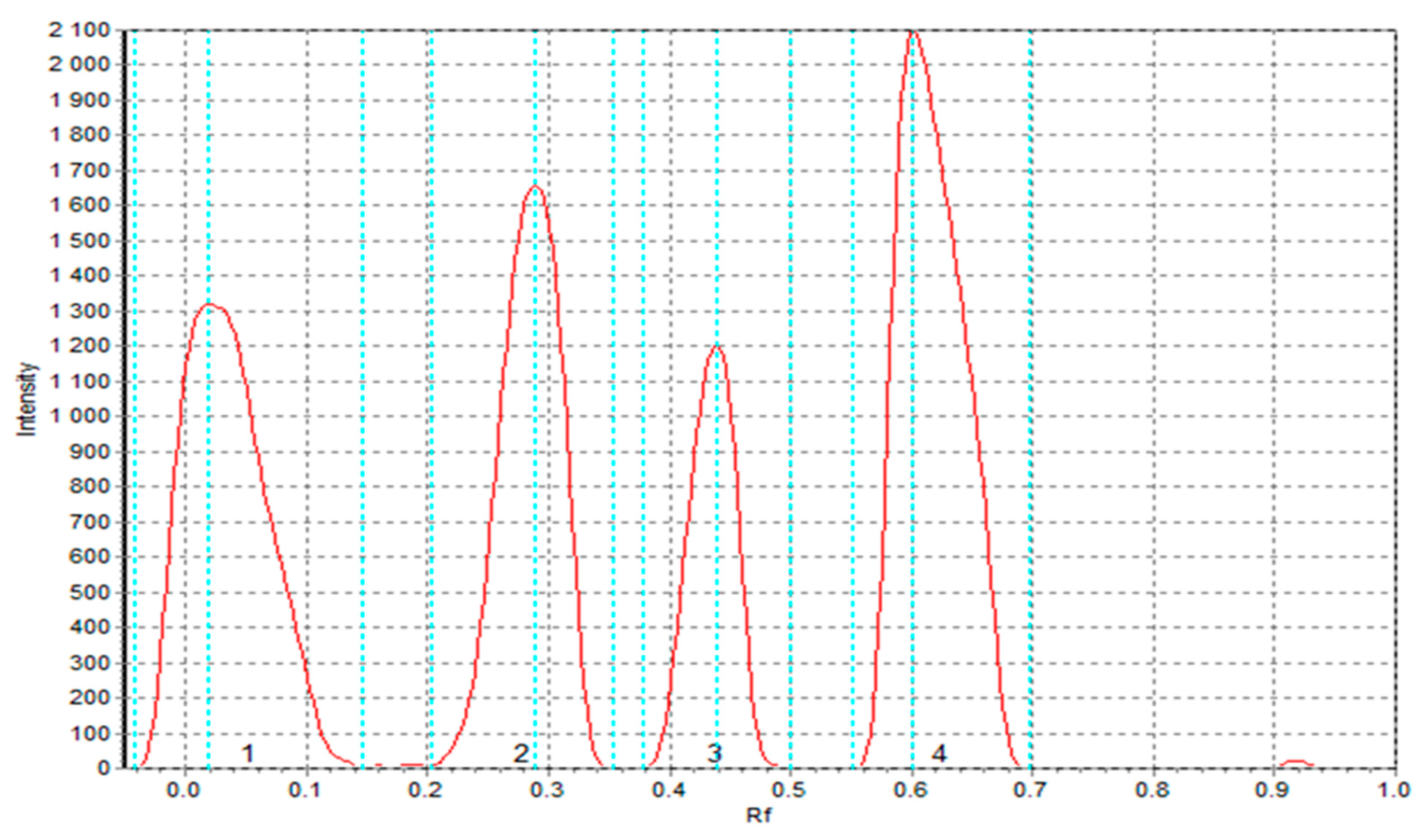
3.5.5. TLC-DPPH Results of the Extracts Obtained from Raw Materials and Pasta Products
3.6. Pasta Microstructure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Romano, A.; Ferranti, P.; Gallo, V.; Masi, P. New ingredients and alternatives to durum wheat semolina for a high quality dried pasta. Curr. Opin. Food Sci. 2021, 41, 249–259. [Google Scholar] [CrossRef]
- Bresciani, A.; Pagani, M.A.; Marti, A. Pasta-making process: A narrative review on the relation between process variables and pasta quality. Foods 2022, 11, 256. [Google Scholar] [CrossRef] [PubMed]
- Kezih, R.; Bekhouche, F.; Merazka, A. Some traditional Algerian products from durum wheat. Afr. J. Food Sci. 2014, 8, 30–34. [Google Scholar] [CrossRef]
- Carpentieri, S.; Larrea-Wachtendorff, D.; Donsì, F.; Ferrari, G. Functionalization of pasta through the incorporation of bioactive compounds from agri-food by-products: Fundamentals, opportunities, and drawbacks. Trends Food Sci. Technol. 2022, 122, 49–65. [Google Scholar] [CrossRef]
- Rawat, N.; Indrani, D. Functional ingredients of wheat-based bakery, traditional, pasta, and other food products. Food Rev. Int. 2015, 31, 125–146. [Google Scholar] [CrossRef]
- Zocchi, D.M.; Bondioli, C.; Hamzeh Hosseini, S.; Miara, M.D.; Musarella, C.M.; Mohammadi, D.; Khan Manduzai, A.; Dilawer Issa, K.; Sulaiman, N.; Khatib, C.; et al. Food Security beyond cereals: A cross-geographical comparative study on Acorn Bread Heritage in the Mediterranean and the Middle East. Foods 2022, 11, 3898. [Google Scholar] [CrossRef]
- Taib, M.; Bouyazza, L.; Lyoussi, B. Acorn oil: Chemistry and functionality. J. Food Qual. 2020, 1, 8898370. [Google Scholar] [CrossRef]
- Cantos, E.; Espín, J.C.; López-Bote, C.; de la Hoz, L.; Ordóñez, J.A.; Tomás-Barberán, F.A. Phenolic compounds and fatty acids from acorns (Quercus spp.), the main dietary constituent of free-ranged Iberian pigs. J. Agric. Food Chem. 2003, 51, 6248–6255. [Google Scholar] [CrossRef]
- Pontieri, P.; Troisi, J.; Calcagnile, M.; Aramouni, F.; Tilley, M.; Smolensky, D.; Del Giudice, L. Nutritional composition, fatty acid content, and mineral content of nine sorghum (Sorghum bicolor) inbred varieties. Foods 2024, 13, 3634. [Google Scholar] [CrossRef]
- Ronoh, A.K.; Serrem, C.A.; Tumwebaze, S.B.; Were, G.M. Effect of fortifying sorghum and wheat with Longhorn grasshopper (Ruspolia differens) powder on nutritional composition and consumer acceptability of biscuits. Food Sci. Nutr. 2024, 12, 3492–3507. [Google Scholar] [CrossRef] [PubMed]
- Charyulu, D.K.; Afari-Sefa, V.; Gumma, M.K. Trends in Global Sorghum Production: Perspectives and Limitations. In Omics and Biotechnological Approaches for Product Profile-Driven Sorghum Improvement; Springer Nature: Singapore, 2024; pp. 1–19. [Google Scholar]
- Belmouloud, R.; Bourekoua, H.; Wójtowicz, A.; Mahroug, H.; Ayad, R.; Krajewska, M.; Różyło, R. Physicochemical, functional, antioxidant, pasting and FT-IR spectroscopic properties of fermented acorns and sorghum using traditional Algerian processes. Processes 2024, 12, 1647. [Google Scholar] [CrossRef]
- Maaroufi, C.; Melcion, J.P.; De Monredon, F.; Giboulot, B.; Guibert, D.; Le Guen, M.P. Fractionation of pea flour with pilot scale sieving. I. Physical and chemical characteristics of pea seed fractions. Anim. Feed Sci. Technol. 2020, 85, 61–78. [Google Scholar] [CrossRef]
- Boucheham, N.; Galet, L.; Patry, S.; Zidoune, M.N. Physicochemical and hydration properties of different cereal and legume gluten-free powders. Food Sci. Nutr. 2019, 7, 3081–3092. [Google Scholar] [CrossRef] [PubMed]
- N’Goran, C.A.; Petit, J.; N’Guessan, A.A.; Gonnety, J.T.; Scher, J. Improvement of the production process and the sensory and nutritional quality of m’bahou, a traditional plantain semolina, enriched with soy or cowpea. Int. J. Food Sci. Technol. 2023, 58, 4171–4185. [Google Scholar] [CrossRef]
- ISO 20483:2006; Cereals and Pulses—Determination of the Nitrogen Content and Calculation of the Crude Protein Content—Kjeldahl Method. International Organization for Standardization: Geneva, Switzerland, 2006.
- AOAC International. Official Method 920.39—Fat (Crude) or Ether Extract in Foods. In Official Methods of Analysis, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- AOAC International. Official Method 985.29—Total Dietary Fiber in Foods. In Official Methods of Analysis, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Callister, W.D.; Rethwisch, D.G. Materials Science and Engineering: An Introduction, 10th ed.; Wiley: Hoboken, NJ, USA, 2018. [Google Scholar]
- Wójtowicz, A.; Mościcki, L. Influence of legume type and addition level on quality characteristics, texture and microstructure of enriched precooked pasta. LWT Food Sci. Technol. 2014, 59, 1175–1185. [Google Scholar] [CrossRef]
- AACC. Approved Methods of the American Association of Cereal Chemists; AACC: St. Paul, MN, USA, 2000. [Google Scholar]
- Chillo, S.; Laverse, J.; Falcone, P.; Del Nobile, M. Quality of spaghetti in base amaranthus wholemeal flour added with quinoa, broad bean and chick pea. J. Food Eng. 2008, 84, 101–107. [Google Scholar] [CrossRef]
- Cavazza, A.; Corradini, C.; Rinaldi, M.; Salvadeo, P.; Borromei, C.; Massini, R. Evaluation of pasta thermal treatment by determination of carbohydrates, furosine, and color indices. Food Bioprocess Technol. 2013, 6, 2721–2731. [Google Scholar] [CrossRef]
- Romankiewicz, D.; Hassoon, W.H.; Cacak-Pietrzak, G.; Sobczyk, M.; Wirkowska-Wojdyła, M.; Ceglińska, A.; Dziki, D. The effect of chia seeds (Salvia hispanica L.) addition on quality and nutritional value of wheat bread. J. Food Qual. 2017, 2017, 7352631. [Google Scholar] [CrossRef]
- Oniszczuk, A.; Podgórski, R. Influence of different extraction methods on the quantification of selected flavonoids and phenolic acids from Tilia cordata inflorescence. Ind. Crops Prod. 2015, 76, 509–514. [Google Scholar] [CrossRef]
- Lee, M.J.; Chung, I.-M.; Kim, H.; Jung, M.Y. High resolution LC–ESI-TOF-mass spectrometry method for fast separation, identification, and quantification of 12 isoflavones in soybeans and soybean products. Food Chem. 2015, 176, 254–262. [Google Scholar] [CrossRef]
- Kasprzak-Drozd, K.; Oniszczuk, T.; Kowalska, I.; Mołdoch, J.; Combrzyński, M.; Gancarz, M.; Dobrzański, B.; Kondracka, A.; Oniszczuk, A. Effect of the production parameters and in vitro digestion on the content of polyphenolic compounds, phenolic acids, and antiradical properties of innovative snacks enriched with wild garlic (Allium ursinum L.) leaves. Int. J. Mol. Sci. 2022, 23, 14458. [Google Scholar] [CrossRef] [PubMed]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef]
- Cieśla, Ł.; Kryszeń, J.; Stochmal, A.; Oleszek, W.; Waksmundzka-Hajnos, M. Approach to develop a standardized TLC-DPPH test for assessing free radical scavenging properties of selected phenolic compounds. J. Pharm. Biomed. Anal. 2012, 70, 126–135. [Google Scholar] [CrossRef]
- Gopalan, C.; Ramasastri, B.V.; Balasubramanian, S.C. Nutritive Value of Indian Foods; National Institute of Nutrition (NIN), Indian Council of Medical Research: Hyderabad, India, 2004. [Google Scholar]
- Zhang, Y.; Zhao, X.; Chen, L.; Liu, Y. Influence of water activity on shelf life and quality of cereal products: A review. Food Control 2021, 121, 107567. [Google Scholar] [CrossRef]
- Ranhotra, G.S.; Gelroth, J.A.; Lorenz, K. Nutritional composition of wheat and wheat products: The effect of bran, germ and fiber. Cereal Chem. 2020, 97, 327–335. [Google Scholar] [CrossRef]
- Călinoiu, L.F.; Gheldiu, A.M.; Vodnar, D.C.; Socaci, S.A. Wholegrain and bran fraction in cereal-based foods: Health impacts and recent advances. Nutrients 2022, 14, 298. [Google Scholar] [CrossRef]
- Bhatta, M.; Arendt, E.K.; Gallagher, E. The impact of milling on nutritional and functional properties of wheat and rye flours: A review. Trends Food Sci. Technol. 2020, 100, 120–134. [Google Scholar] [CrossRef]
- Cui, J.; Lian, Y.; Zhao, C.; Du, H.; Han, Y.; Gao, W.; Xiao, H.; Zheng, J. Dietary fibers from fruits and vegetables and their health benefits via modulation of gut microbiota. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1514–1532. [Google Scholar] [CrossRef]
- Silva, F.A.; Lopes, C.M.; Santos, L.M. Development of low-calorie functional foods: Trends and challenges. Food Res. Int. 2019, 123, 310–321. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. Codex Standard for Pasta (CODEX STAN 212-1999); Food and Agriculture Organization: Rome, Italy; World Health Organization: Geneva, Switzerland, 2019; Available online: https://www.fao.org/fao-who-codexalimentarius/codex-texts/list-standards/en/ (accessed on 11 August 2025).
- Giuberti, G.; Cerioli, C.; Galli, V.; Gallo, V. Influence of raw material and processing parameters on pasta quality: A review. Food Res. Int. 2020, 137, 109706. [Google Scholar] [CrossRef]
- Kim, S.K.; Kim, H.; Lee, M. Moisture content and water activity effects on microbial stability and shelf life of dried pasta products. Food Microbiol. 2021, 94, 103641. [Google Scholar] [CrossRef]
- Allai, F.M.; Junaid, P.M.; Azad, Z.R.A.A.; Gul, K.; Dar, B.N.; Siddiqui, S.A.; Loenzo, J.M. Impact of moisture content on microstructural, thermal, and techno-functional characteristics of extruded whole-grain-based breakfast cereal enriched with Indian horse chestnut flour. Food Chem. X 2023, 20, 100959. [Google Scholar] [CrossRef] [PubMed]
- Cappa, C.; Cattivelli, L.; Borrelli, G.M. Role of starch damage and protein content on water absorption indices of pasta made from durum wheat. LWT Food Sci. Technol. 2020, 117, 108608. [Google Scholar] [CrossRef]
- Rashid, M.A.; Khan, M.I.; Husain, S.Z. Influence of damaged starch on cooking quality and nutrient retention in pasta products. J. Food Process. Preserv. 2019, 43, e14069. [Google Scholar] [CrossRef]
- Sujka, K.; Cacak-Pietrzak, G.; Sułek, A.; Murgrabia, K.; Dziki, D. Buckwheat hull-enriched pasta: Physicochemical and sensory properties. Molecules 2022, 27, 4065. [Google Scholar] [CrossRef]
- Sissons, M.; Abecassis, J.; Marchylo, B.; Cubadda, R. Methods used to assess and predict quality of durum wheat, semolina, and pasta. In Durum Wheat: Chemistry and Technology, 2nd ed.; Sissons, M., Abecassis, J., Marchylo, B., Carcea, M., Eds.; AACC International, Inc.: St. Paul, MN, USA, 2012; pp. 213–234. [Google Scholar] [CrossRef]
- Oladunmoye, O. Physico-Chemical, Nutritional and Sensory Qualities of Cassava-Wheat Semolina Macaroni Noodles. Doctoral Dissertation, University of Ibadan, Ibadan, Nigeria, 2012. Available online: http://ir.library.ui.edu.ng/handle/123456789/9462 (accessed on 11 August 2025).
- Feillet, P.; Autran, J.C.; Icard-Vernière, C. Pasta brownness: An assessment. J. Cereal Sci. 2000, 32, 215–233. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W.; Waniska, R.D.; Rooney, W.L. Phenolic compounds and antioxidant activity of sorghum grains of varying genotypes. J. Agric. Food Chem. 2005, 53, 6813–6818. [Google Scholar] [CrossRef]
- Rather, M.A.; Thakur, R.; Hoque, M.; Das, R.S.; Miki, K.S.L.; Teixeira-Costa, B.E.; Gupta, A.K. Sorghum (Sorghum bicolor): Phytochemical composition, bio-functional, and technological characteristics. In Nutri-Cereals: Nutraceutical and Techno-Functional Potential; Sharma, R., Nanda, V., Sharma, S., Eds.; CRC Press: Boca Raton, FL, USA, 2023; pp. 45–77. [Google Scholar]
- Biernacka, B.; Dziki, D.; Gawlik-Dziki, U.; Różyło, R. Common wheat pasta enriched with cereal coffee: Quality and physical and functional properties. LWT Food Sci. Technol. 2020, 139, 110516. [Google Scholar] [CrossRef]
- Martín-Esparza, M.E.; Raigón, M.D.; Raga, A.; Albors, A. High fibre tiger nut pasta and xanthan gum: Cooking quality, microstructure, physico-chemical properties and consumer acceptance. Food Sci. Biotechnol. 2018, 27, 1075–1084. [Google Scholar] [CrossRef]
- Bustos, M.C.; Perez, G.T.; Leon, A.E. Structure and quality of pasta enriched with functional ingredients. RSC Adv. 2015, 5, 30780–30792. [Google Scholar] [CrossRef]
- Martínez, M.M.; Li, C.; Dhital, S.; Gidley, M.J. Structural basis and kinetics of starch digestion. Trends Food Sci. Technol. 2021, 108, 524–535. [Google Scholar] [CrossRef]
- Guerra, A.; Bresciani, A.; Marti, A.; Pagani, M.A.; Iametti, S. Understanding the protein-starch matrix in pasta: Implications for quality and digestibility. LWT Food Sci. Technol. 2023, 177, 114547. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Zee, S.Y.; Corke, H. Physicochemical properties related to quality of rice noodles. Cereal Chem. 1999, 76, 861–867. [Google Scholar] [CrossRef]
- Bolarinwa, I.F.; Oyesiji, O.O. Gluten free rice-soy pasta: Proximate composition, textural properties and sensory attributes. Heliyon 2021, 7, e06052. [Google Scholar] [CrossRef]
- Rao, S.; Santhakumar, A.B.; Chinkwo, K.A.; Wu, G.; Johnson, S.K.; Blanchard, C.L. Characterization of phenolic compounds and antioxidant activity in sorghum grains. J. Cereal Sci. 2018, 84, 103–111. [Google Scholar] [CrossRef]
- De Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant’Ana, H.M. Sorghum (Sorghum bicolor L.): Nutrients, bioactive compounds, and potential impact on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Ma, Q.; Zheng, N.; Wang, R.; Visentin, S.; He, L.; Liu, S. Plant Polyphenol-Based Injectable Hydrogels: Advances and Biomedical Applications. Adv. Healthc. Mater. 2025, 14, 2500445. [Google Scholar] [CrossRef]
- Sejbuk, M.; Mirończuk-Chodakowska, I.; Karav, S.; Witkowska, A.M. Dietary polyphenols, food processing and gut microbiome: Recent findings on bioavailability, bioactivity, and gut microbiome interplay. Antioxidants 2024, 13, 1220. [Google Scholar] [CrossRef]
- Durazzo, A.; Azzini, E.; Turfani, V.; Polito, A.; Maiani, G.; Carcea, M. Effect of cooking on lignans content in whole-grain pasta made with different cereals and other seeds. Cereal Chem. 2013, 90, 193–198. [Google Scholar] [CrossRef]
- Punia, H.; Tokas, J.; Malik, A.; Sangwan, S. Characterization of phenolic compounds and antioxidant activity in sorghum (Sorghum bicolor L. Moench) grains. Cereal Res. Commun. 2021, 49, 343–353. [Google Scholar] [CrossRef]
- Przybylska-Balcerek, A.; Frankowski, J.; Stuper-Szablewska, K. Bioactive compounds in sorghum. Eur. Food Res. Technol. 2019, 245, 1075–1080. [Google Scholar] [CrossRef]
- Altunkaya, A.; Gökmen, V.; Skibsted, L.H. pH dependent antioxidant activity of lettuce (L. sativa) and synergism with added phenolic antioxidants. Food Chem. 2016, 190, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Yousif, A.M.; Johnson, S.K.; Gamlath, S. Acute effect of sorghum flour containing pasta on plasma total polyphenols, antioxidant capacity and oxidative stress markers in healthy subjects: A randomised controlled trial. Clin. Nutr. 2015, 34, 415–421. [Google Scholar] [CrossRef]
- Girard, A.L.; Awika, J.M. Effects of edible plant polyphenols on gluten protein functionality and potential applications of polyphenol–gluten interactions. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2164–2199. [Google Scholar] [CrossRef]
- Ribeiro, M.; Sousa, T.d.; Poeta, P.; Bagulho, A.S.; Igrejas, G. Review of Structural Features and Binding Capacity of Polyphenols to Gluten Proteins and Peptides In Vitro: Relevance to Celiac Disease. Antioxidants 2020, 9, 463. [Google Scholar] [CrossRef] [PubMed]
- Seraglio, S.K.T.; Valese, A.C.; Daguer, H.; Bergamo, G.; Azevedo, M.S.; Nehring, P.; Gonzaga, L.V.; Fett, R.; Costa, A.C.O. Effect of in vitro gastrointestinal digestion on the bioaccessibility of phenolic compounds, minerals, and antioxidant capacity of Mimosa scabrella Bentham honeydew honeys. Food Res. Int. 2017, 99, 670–678. [Google Scholar] [CrossRef]
- Romano, A.; di Luccia, A.; Romano, R.; Sarghini, F.; Masi, P. Microscopic and thermal characteristics of experimental models of starch, gliadins, glutenins and gluten from semolina. Chem. Eng. Trans. 2015, 43, 163–168. [Google Scholar] [CrossRef]
- Lu, H.; Wang, C.; Guo, T.; Xie, Y.; Feng, W.; Li, S. Starch composition and its granules distribution in wheat grains in relation to post-anthesis high temperature and drought stress treatments. Starch/Starke 2014, 66, 419–428. [Google Scholar] [CrossRef]
- Geera, B.P.; Nelson, J.E.; Souza, E.; Huber, K.C. Composition properties of A- B-type starch granules of wild-type, partial waxy, waxy soft wheat. Cereal Chem. 2006, 83, 551–557. [Google Scholar] [CrossRef]
- Haziman, M.L.; Ishaq, M.I.; Qonit, M.A.H.; Lestari, E.G.; Susilawati, P.N.; Widarsih, W.; Syukur, C.; Herawati, H.; Arief, R.; Santosa, B.; et al. Sorghum starch review: Structural properties, interactions with proteins and polyphenols, and modification of physicochemical properties. Food Chem. 2025, 463, 139810. [Google Scholar] [CrossRef]
- Gallo, V.; Romano, A.; Masi, P. Does the presence of fibres affect the microstructure and in vitro starch digestibility of commercial Italian pasta? Food Struct. 2020, 24, 100139. [Google Scholar] [CrossRef]



| Sample Type | Semolina (g) | Enrichment Flour (g) | Salt (g) | Water (mL) |
|---|---|---|---|---|
| Control T1–T4 | 1000 (S1–S4) | 0 | 10 | 400 |
| Sorghum-Enriched TSF | 990 (S2 Kenza) | 10 (1%) | 10 | 450 |
| Cork Oak-Enriched TCF | 960 (S2 Kenza) | 40 (4%) | 10 | 600 |
| Sample | Moisture (%) | Protein (%) | Fat (%) | Ash (%) | Fiber (%) | Digestible Carbohydrates (%) | Energy (kJ) | Energy (kcal) |
|---|---|---|---|---|---|---|---|---|
| S1 | 11.56 b | 12.02 d | 1.44 d | 0.77 d | 5.70 c | 68.51 a | 1468 | 346 |
| S2 | 11.95 a | 13.07 a | 1.52 d | 0.82 c | 5.97 c | 66.67 b | 1460 | 345 |
| S3 | 11.07 c | 12.83 b | 1.48 d | 0.74 e | 5.96 c | 67.92 a | 1475 | 348 |
| S4 | 11.09 c | 12.25 c | 1.83 c | 0.83 c | 6.19 c | 67.81 a | 1478 | 349 |
| SF | 8.96 e | 8.42 e | 3.17 b | 2.69 a | 28.27 b | 48.49 c | 1311 | 313 |
| CF | 9.60 d | 4.33 f | 8.69 a | 1.90 b | 30.07 a | 45.41 d | 1408 | 337 |
| Sample | Length (mm) | Width (mm) | Thickness (mm) |
|---|---|---|---|
| T1 | 13.31 ± 1.60 a | 13.25 ± 1.75 a | 0.81 ± 0.14 a |
| T2 | 13.50 ± 2.00 a | 12.81 ± 1.96 a | 0.75 ± 0.07 a |
| T3 | 13.45 ± 1.45 a | 12.95 ± 1.95 a | 0.66 ± 0.14 a |
| T4 | 14.00 ± 1.00 a | 13.25 ± 1.25 a | 0.67 ± 0.13 a |
| TSF | 13.90 ± 1.40 a | 13.36 ± 1.83 a | 0.63 ± 0.12 a |
| TCF | 14.35 ± 1.15 a | 13.85 ± 1.35 a | 0.75 ± 0.03 a |
| Sample | Moisture Content (%) | WAI (%) | WSI (%) |
|---|---|---|---|
| T1 | 10.887 ± 0.014 d | 1.60 ± 0.02 a | 6.23 ± 0.16 c |
| T2 | 11.468 ± 0.004 a | 1.57 ± 0.02 ab | 6.38 ± 0.45 c |
| T3 | 11.203 ± 0.015 b | 1.60 ± 0.01 a | 6.62 ± 0.38 c |
| T4 | 11.239 ± 0.021 ab | 1.61 ± 0.03 a | 5.81 ± 0.33 c |
| TSF | 10.905 ± 0.039 d | 1.54 ± 0.01 b | 10.19 ± 1.13 a |
| TCF | 11.179 ± 0.002 c | 1.57 ± 0.01 ab | 7.33 ± 0.22 b |
| Sample | L* | a* | b* | ΔE |
|---|---|---|---|---|
| T1 | 59.95 ± 0.8 bc | 1.17 ± 0.3 b | 10.45 ± 0.7 a | Ref |
| T2 | 61.14 ± 0.7 ab | 1.02 ± 0.4 b | 9.65 ± 0.6 a | 1.44 |
| T3 | 60.33 ± 0.6 abc | 0.82 ± 0.3 b | 9.20 ± 0.5 a | 1.35 |
| T4 | 61.42 ± 0.7 a | 0.85 ± 0.4 b | 10.64 ± 0.6 a | 1.52 |
| TSF | 57.74 ± 0.9 c | 1.25 ± 0.5 b | 7.48 ± 0.7 b | 3.70 |
| TCF | 46.20 ± 1.0 d | 4.71 ± 0.6 a | 10.59 ± 0.8 a | 14.20 |
| Sample | OCT (min) | WAC (%) | CL (%) |
|---|---|---|---|
| T1 | 2.00 ± 0.01 b | 241.99 ± 0.86 c | 7.85 ± 0.03 b |
| T2 | 2.00 ± 0.01 b | 253.69 ± 1.08 b | 7.90 ± 0.02 b |
| T3 | 2.00 ± 0.02 b | 233.04 ± 0.57 d | 7.58 ± 0.03 c |
| T4 | 1.30 ± 0.01 c | 299.82 ± 0.12 a | 8.08 ± 0.04 a |
| TSF | 2.15 ± 0.17 ab | 236.88 ± 1.05 a | 7.70 ± 0.02 bc |
| TCF | 2.30 ± 0.05 a | 214.33 ± 1.23 e | 7.58 ± 0.03 c |
| Sample | Hardness (N) | Adhesiveness (mJ) | Cohesiveness (-) | Gumminess (N) | Springiness (-) | Chewiness (N·mm) |
|---|---|---|---|---|---|---|
| T1 | 208.50 ± 5.50 bc | 20.11 ± 2.67 ab | 11.24 ± 1.27 ab | 30.66 ± 1.20 cd | 0.27 ± 0.01 b | 8.02 ± 0.21 cd |
| T2 | 243.50 ± 6.50 ab | 19.39 ± 3.04 ab | 9.49 ± 3.31 bc | 46.04 ± 2.13 b | 0.33 ± 0.01 ab | 15.21 ± 0.94 b |
| T3 | 219.50 ± 25.5 bc | 18.35 ± 4.78 ab | 8.75 ± 2.35 bc | 35.35 ± 5.94 c | 0.36 ± 0.02 a | 12.61 ± 1.61 bc |
| T4 | 166.50 ± 4.50 d | 14.77 ± 2.51 b | 9.44 ± 1.17 bc | 20.71 ± 2.21 e | 0.27 ± 0.01 b | 5.64 ± 0.41 e |
| TSF | 191.50 ± 2.50 cd | 14.17 ± 0.95 b | 7.88 ± 1.02 c | 33.24 ± 1.33 cd | 0.29 ± 0.02 b | 9.52 ± 0.97 c |
| TCF | 276.00 ± 29.00 a | 22.17 ± 4.21 a | 13.59 ± 3.61 a | 54.84 ± 4.63 a | 0.35 ± 0.02 a | 18.85 ± 0.69 a |
| Formula | [M + H]+ | [M + Na]+ | [M − H]− | Fragments | Compounds | Rf (min) |
|---|---|---|---|---|---|---|
| C10H8O4 | - | - | 191.0379 | 176.0186; 148.0234 124.0461 | Scopoletin | 4.971 |
| C6H12O3 | 131.0718 | 129.0567; 113.0564 | Hydroxycaproic acid | 5.209 | ||
| C9H10O3 | - | - | 165.0559 | 147.0444; 119.0498; 103.0561 | 2-Hydroxy-3-phenylpropanoic acid (3-Phenyllactic acid) | 8.225 |
| C16H12O6 | - | - | 299.0597 | 165.0226; 133.0273 | Luteolin methyl ether (7or5) | 15.184 |
| C10H8O4 | - | - | 191.0361 | 176.0124; 148.0210 | Scopoletin isomer | 16.255 |
| C15H12O6 | - | - | 287.0606 | 151.0058; 135.0462 107.0156 | Eriodictyol | 18.969 |
| C15H10O6 | - | - | 285.0415 | 151.0013; 133.0292; 107.0148 | Luteolin | 20.345 |
| C15H12O5 | - | - | 271.0644 | 177.0259; 151.0031; 119.0519 | Naringenin | 22.301 |
| Formula | [M + H]+ | [M + Na]+ | [M − H]− | Fragments | Compound | Rf [min] |
|---|---|---|---|---|---|---|
| C7H12O6 | 191.0563 | 173.0521; 127.0490 | Quinic acid | 1.905 | ||
| C7H6O5 | 169.0145 | 125.0237 | Gallic acid | 2.334 | ||
| C6H8O7 | 191.0207 | 111.0125 | Citric acid | 2.714 | ||
| C9H10O5 | - | - | 197.0470 | 169.0125; 125.0228 | Ethyl gallate | 7.457 |
| C15H20O10 | 335.0431 | 183.0329 | Digallic acid methyl | 12.530 | ||
| C8H8O5 | 183.0228 | 168.0004; 124.0130 | Methyl gallate | 4.000 | ||
| C14H6O8 | 301.0008 | 145.0276 | Ellagic acid | 14.502 |
| Sample | TPC (mg GAE/mL Extract) | TFC (mg QE/mL Extract) |
|---|---|---|
| S1 | 0.099 ± 0.004 g | 0.299 ± 0.013 f |
| S2 | 0.123 ± 0.001 e | 0.328 ± 0.012 e |
| S3 | 0.105 ± 0.002 g | 0.417 ± 0.027 d |
| S4 | 0.118 ± 0.002 f | 0.581 ± 0.052 c |
| SF | 1.048 ± 0.056 b | 0.947 ± 0.031 b |
| CF | 1.715 ± 0.050 a | 1.615 ± 0.003 a |
| T2 | 0.116 ± 0.002 f | 0.310 ± 0.010 f |
| TSF | 0.299 ± 0.015 d | 0.254 ± 0.011 g |
| TCF | 0.314 ± 0.015 c | 0.299 ± 0.000 f |
| Sample | RSA After 10 Min (%) | RSA After 20 Min (%) | RSA After 30 Min (%) |
|---|---|---|---|
| S1 | 24.87 ± 1.12 g | 28.27 ± 0.53 g | 30.19 ± 0.08 g |
| S2 | 27.53 ± 0.83 f | 31.68 ± 0.68 f | 34.40 ± 0.15 f |
| S3 | 22.11 ± 1.00 h | 25.66 ± 1.08 h | 27.89 ± 1.16 h |
| S4 | 31.44 ± 1.08 e | 36.90 ± 1.08 e | 40.17 ± 1.23 e |
| SF | 77.84 ± 0.50 b | 78.45 ± 0.50 b | 78.65 ± 0.50 b |
| CF | 91.10 ± 0.39 a | 91.32 ± 0.54 a | 91.43 ± 0.54 a |
| T2 | 25.80 ± 0.75 g | 30.20 ± 0.60 f | 33.00 ± 0.20 f |
| TSF | 45.60 ± 0.29 d | 51.19 ± 0.44 d | 53.10 ± 0.49 d |
| TCF | 54.41 ± 0.64 c | 61.71 ± 0.93 c | 64.15 ± 0.93 c |
| Sample | After 0 Min | After 10 Min | After 30 Min | Reference | Description |
|---|---|---|---|---|---|
| SF | 1.860 b | 1.867 b | 1.857 b | 1.000 | Sorghum flour |
| TSF | 1.362 c | 1.371 c | 1.372 c | 1.000 | Pasta with sorghum flour |
| CF | 2.789 a | 2.883 a | 2.887 a | 1.000 | Cork oak flour |
| TCF | 1.564 b | 1.566 b | 1.569 b | 1.000 | Pasta with cork oak flour |
| S2 | 1.000 d | 1.000 d | 1.000 d | 1.000 | Semolina |
| T2 | 1.090 cd | 1.115 cd | 1.112 cd | 1.000 | Control pasta with semolina S2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabouni, R.; Himed, L.; Akachat, B.; Wójtowicz, A.; Kasprzak-Drozd, K.; Namoune, H.; Merniz, S.; D’Elia, M.; Rastrelli, L.; Oniszczuk, A. Physiochemical Characterization and Antioxidant Potential of Sorghum and Cork Oak as Valuable Additives to Traditional Trida Pasta. Foods 2025, 14, 2832. https://doi.org/10.3390/foods14162832
Sabouni R, Himed L, Akachat B, Wójtowicz A, Kasprzak-Drozd K, Namoune H, Merniz S, D’Elia M, Rastrelli L, Oniszczuk A. Physiochemical Characterization and Antioxidant Potential of Sorghum and Cork Oak as Valuable Additives to Traditional Trida Pasta. Foods. 2025; 14(16):2832. https://doi.org/10.3390/foods14162832
Chicago/Turabian StyleSabouni, Rima, Louiza Himed, Belkis Akachat, Agnieszka Wójtowicz, Kamila Kasprzak-Drozd, Hacène Namoune, Salah Merniz, Maria D’Elia, Luca Rastrelli, and Anna Oniszczuk. 2025. "Physiochemical Characterization and Antioxidant Potential of Sorghum and Cork Oak as Valuable Additives to Traditional Trida Pasta" Foods 14, no. 16: 2832. https://doi.org/10.3390/foods14162832
APA StyleSabouni, R., Himed, L., Akachat, B., Wójtowicz, A., Kasprzak-Drozd, K., Namoune, H., Merniz, S., D’Elia, M., Rastrelli, L., & Oniszczuk, A. (2025). Physiochemical Characterization and Antioxidant Potential of Sorghum and Cork Oak as Valuable Additives to Traditional Trida Pasta. Foods, 14(16), 2832. https://doi.org/10.3390/foods14162832











