Production of Polyhydroxyalkanoates for Biodegradable Food Packaging Applications Using Haloferax mediterranei and Agrifood Wastes
Abstract
1. Introduction
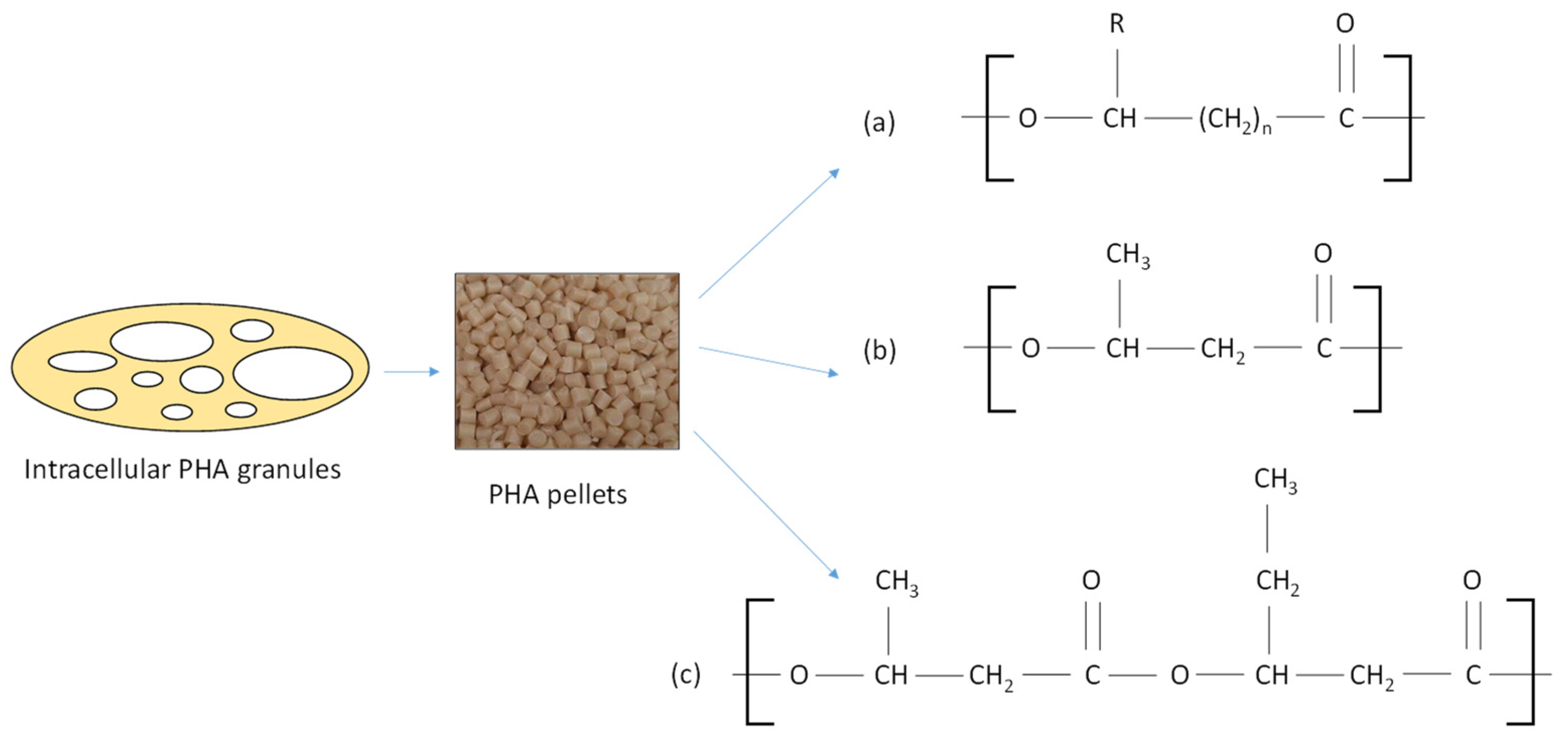
2. Structure, Properties, and Degradation Mechanism of PHAs
3. Production of PHAs
4. Downstream Processing of PHAs
5. Production of PHAs from Inexpensive Carbon Sources
6. PHA Producing Microorganisms: The Case of Haloferax mediterranei
| Reference | Substrate | gCDM/L | gPHA/100 gCDM (1) | g PHA/L | Product Yield Coefficient (2) (YP/S) | Volumetric Productivity | Other Findings |
|---|---|---|---|---|---|---|---|
| [4] | Simulants of green macroalgae hydrolysates | 3.8 ± 0.2 | 58% | 2.2 ± 0.12 g/L | --- | 0.035 g/L h | Conditions for fermentation are to be optimized to increase the yield and productivity of PHA production. |
| [6] | Sesame seed waste water hydrolysates (acid hydrolysis) | 50 | 75% | 0.53 g/L to 20.9 g/L with glucose supplementation | --- | --- | Less than 1 g sugar/L hydrolysate. Glucose supplementation needed. |
| [80] | Biodiesel industry-derived by-products | --- | 75.4% | 16.2 g/L from crude glycerol phase | 0.19 g/g | 0.12 g/L h | 10% mol HV, 253 kDa polydispersity 2.7 |
| [86] | Olive mill wastewater | --- | 43% | Max 0.2 g/L | --- | --- | 6.5% mol HV |
| [87] | Synthetic molasses wastewater | 6 | --- | 5 g/L | --- | 390–620 mg/L h | Increased T (35 °C) improves production |
| [52] | Chlorella biomass hydrolysates (acid hydrolysis) | 6 | 55.5% | 4 g/L | --- | --- | PHBV with 10.5% mol HV |
| [1] | Food Waste-Derived Nutrients | 3 | --- | 2 g/L | 0.41–0.54 gPHBV/g acetate | --- | Up to 23% wt. HV |
| [88] | Fermented food waste | 7.0 ± 0.7 | --- | 4.5 ± 0.2 g/L PHA | 0.3 g PHBV/g COD | --- | 10–30% wt. HV |
| [89] | Ricotta cheese exhausted whey enzymatically treated | 6 (lab), 18 (pilot) | 13.5% lab 9% pilot | 1.0 g/L lab 1.3 g/L pilot | 0.05–0.14 | 0.05–0.21 g/L h | Approx 16% HV |
| [16] | Vinasse from etanol industry | --- | 70% | 19.7 g/L | 0.87 (based on total carbohydrates) | 0.21 g/L h (based on total carbohydrates) | PHBV 12–14 mol% |
| [83] | Rice-based ethanol stillage | 71% | 16.42 g/L | 0.35 | 0.17 g/L h | PHBV 15.4% HV | |
| [39] | Stillage from rice-based ethanol manufacture | 63% | 13.12 g/L | 0.27 | 0.14 g/L h | PHBV 17.9% HV | |
| [78] | Chemically hydrolyzed cheese whey | 7.54 | 53% | 7.92 g/L | --- | 4.04 g/L day | 1.5 mol% HV. The polymer presented a molecular mass of 4.4 × 105, with a polydispersity index of 1.5. |
| [90] | Extruded rice bran and extruded cornstarch | 140 | 55.6% | 77.8 g/L | --- | --- | Repeated fed-batch fermentation |
| [72] | Whey lactose | --- | 73% PHA 50% PHBV | --- | 0.29 | 0.09 g/L h | H. mediterranei was a more effective producer than Pseudomonas hydrogenovora and Hydrogenophaga pseudoflava |
| [76] | Whey lactose | --- | 50% | 5.5 g/L | --- | 0.05 g/L h | --- |
| [77] | Waste bread | 2–3 | --- | 1.53 g/L | 20–25 mg/g, depending on the extraction procedure | --- | 10–13% (w/w) HV depending on the extraction procedure. Solvent-free extraction could be used |
| [13] | Silkworm excrement | --- | --- | 1.73 g/L | --- | --- | 16% mol HV |
| [91] | Hydrolyzed rapeseed meal | 5–10 | --- | 0.512 g/L | --- | --- | --- |
7. Strategies for Improving the Economic Viability of PHAs Production and Profitability: Packaging and Other Applications
8. Conclusions and Future Challenges
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, K.; Zhang, R. Production of polyhydroxyalkanoates (PHA) by Haloferax mediterranei from food waste derived nutrients for biodegradable plastic applications. J. Microbiol. Biotechnol. 2021, 31, 338. [Google Scholar] [CrossRef] [PubMed]
- Joyline, M.; Aruna, K. Production and characterization of polyhydroxyalkanoates (PHA) by Bacillus megaterium strain JHA using inexpensive agro-industrial wastes. Int. J. Rec. Sci. Res. 2019, 10, 33359–33374. [Google Scholar]
- Angra, V.; Sehgal, R.; Gupta, R. Trends in PHA production by microbially diverse and functionally distinct communities. Microb. Ecol. 2023, 85, 572–585. [Google Scholar] [CrossRef]
- Ghosh, S.; Gnaim, R.; Greiserman, S.; Fadeev, L.; Gozin, M.; Golberg, A. Macroalgal biomass subcritical hydrolysates for the production of polyhydroxyalkanoate (PHA) by Haloferax mediterranei. Bioresour. Technol. 2019, 271, 166–173. [Google Scholar] [CrossRef]
- Plastics Europe AISBL (2022). Plastic the Facts 2022. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2022/ (accessed on 10 February 2024).
- Alsafadi, D.; Alhesan, J.S.A.; Mansoura, A.; Oqdeha, S. Production of polyhydroxyalkanoate from sesame seed wastewater by sequencing batch reactor cultivation process of Haloferax mediterranei. Arab. J. Chem. 2023, 16, 104584. [Google Scholar] [CrossRef]
- Ibrahim, M.I.; Alsafadi, D.; Alamry, K.A.; Oves, M.; Alosaimi, A.M.; Hussein, M.A. A promising antimicrobial bionanocomposite based poly(3-hydroxybutyrate-co-3-hydroxyvalerate) reinforced silver doped zinc oxide nanoparticles. Sci. Rep. 2022, 12, 14299. [Google Scholar] [CrossRef]
- Romero Castelán, E.; Rodríguez-Hernández, A.I.; Chavarría-Hernández, N.; López-Ortega, M.A.; del Rocio López-Cuellar, M. Natural antimicrobial systems protected by complex polyhydroxyalkanoate matrices for food biopackaging applications—A review. Int. J. Biol. Macromol. 2023, 233, 123418. [Google Scholar] [CrossRef]
- Vigneswari, S.; Noor, M.S.M.; Amelia, T.S.M.; Balakrishnan, K.; Adnan, A.; Bhubalan, K.; Ramakrishna, S. Recent advances in the biosynthesis of polyhydroxyalkanoates from lignocellulosic feedstocks. Life 2021, 11, 807. [Google Scholar] [CrossRef]
- Khosravi-Darani, K.; Bucci, D.Z. Application of poly (hydroxyalkanoate) in food packaging: Improvements by nanotechnology. CABEQ 2015, 29, 275–285. [Google Scholar]
- Ferre-Guell, A.; Winterburn, J. Biosynthesis and Characterization of Polyhydroxyalkanoates with Controlled Composition and Microstructure. Biomacromolecules 2018, 19, 996–1005. [Google Scholar] [CrossRef]
- Narayanan, A.; Sajeev Kumar, V.A.; Ramana, K.V. Production and characterization of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) from Bacillus mycoides DFC1 using rice husk hydrolyzate. Waste Biomass Valorization 2014, 5, 109–118. [Google Scholar] [CrossRef]
- Cai, S.; Wu, Y.; Liu, R.; Jia, H.; Qiu, Y.; Jiang, M.; Cai, L. Study on the production of high 3HV content PHBV via an open fermentation with waste silkworm excrement as the carbon source by the haloarchaeon Haloferax mediterranei. Front. Microbial. 2022, 13, 981605. [Google Scholar] [CrossRef] [PubMed]
- Policastro, G.; Panico, A.; Fabbricino, M. Improving biological production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) co-polymer: A critical review. Rev. Environ. Sci. Biotechnol. 2021, 20, 479–513. [Google Scholar] [CrossRef]
- Priya, A.; Hathi, Z.; Haque, M.A.; Kumar, S.; Kumar, A.; Singh, E.; Lin, C.S. Effect of levulinic acid on production of polyhydroxyalkanoates from food waste by Haloferax mediterranei. Environ. Res. 2022, 214, 114001. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Pramanik, A.; Maji, S.K.; Haldar, S.; Mukhopadhyay, U.K.; Mukherjee, J. Utilization of vinasse for production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei. AMB Express 2012, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, P.B.; Malafaia, C.B. Perspectives on the production, structural characteristics and potential applications of bioplastics derived from polyhydroxyalkanoates. Int. J. Biol. Macromol. 2017, 107, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V.A. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing and potential applications in packaging. EXPRESS Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Petersen, K.; Nielsen, P.V.; Bertelsen, G.; Lawther, M.; Olsen, M.B.; Nilsson, N.H.; Mortensen, G. Potential of biobased materials for food packaging. Trends Food Sci. Technol. 1999, 10, 52–68. [Google Scholar] [CrossRef]
- Dhar, P.; Bhardwaj, U.; Kumar, A.; Katiyar, V. Poly (3-hydroxybutyrate)/cellulose nanocrystal films for food packaging applications: Barrier and migration studies. Polym. Eng. Sci. 2015, 55, 2388–2395. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Poly (3-hydroxybutyrate)/ZnO bionanocomposites with improved mechanical, barrier and antibacterial properties. Int. J. Mol. Sci. 2014, 15, 10950–10973. [Google Scholar] [CrossRef]
- Zhijiang, C.; Guang, Y. Optical nanocomposites prepared by incorporating bacterial cellulose nanofibrils into poly (3-hydroxybutyrate). Mater. Lett. 2011, 65, 182–184. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Castro-Lopez, M.D.M.; Rayón, E.; Barral-Losada, L.F.; López-Vilariño, J.M.; López, J.; González-Rodríguez, M.V. Plasticized poly (lactic acid)–poly (hydroxybutyrate)(PLA–PHB) blends incorporated with catechin intended for active food-packaging applications. J. Agric. Food Chem. 2014, 62, 10170–10180. [Google Scholar] [CrossRef] [PubMed]
- Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. Nanostructured interlayers of zein to improve the barrier properties of high barrier polyhydroxyalkanoates and other polyesters. J. Food Eng. 2014, 127, 1–9. [Google Scholar] [CrossRef]
- Laycock, B.; Halley, P.; Pratt, S.; Werker, A.; Lant, P. The chemomechanical properties of microbial polyhydroxyalkanoates. Prog. Polym. Sci. 2013, 38, 536–583. [Google Scholar] [CrossRef]
- Requena, R.; Jiménez, A.; Vargas, M.; Chiralt, A. Effect of plasticizers on thermal and physical properties of compression-moulded poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] films. Polym. Test. 2016, 56, 45–53. [Google Scholar] [CrossRef]
- Requena, R.; Jiménez, A.; Vargas, M.; Chiralt, A. Poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)] active bilayer films obtained by compression moulding and applying essential oils at the interface. Polym. Int. 2016, 65, 883–891. [Google Scholar] [CrossRef]
- Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. High barrier polyhydroxyalcanoate food packaging film by means of nanostructured electrospun interlayers of zein. Food Hydrocoll. 2013, 32, 106–114. [Google Scholar] [CrossRef]
- Laycock, B.; Arcos-Hernandez, M.V.; Langford, A.; Buchanan, J.; Halley, P.J.; Werker, A.; Pratt, S. Thermal properties and crystallization behavior of fractionated blocky and random polyhydroxyalkanoate copolymers from mixed microbial cultures. J. Appl. Polym. Sci. 2014, 131, 40836. [Google Scholar] [CrossRef]
- Modi, S.; Koelling, K.; Vodovotz, Y. Assessment of PHB with varying hydroxyvalerate content for potential packaging applications. Eur. Polym. J. 2011, 47, 179–186. [Google Scholar] [CrossRef]
- Sanchez-Garcia, M.D.; Lagaron, J.M.; Hoa, S.V. Effect of addition of carbon nanofibers and carbon nanotubes on properties of thermoplastic biopolymers. Compos. Sci. Technol. 2010, 70, 1095–1105. [Google Scholar] [CrossRef]
- Savenkova, L.; Gercberga, Z.; Bibers, I.; Kalnin, M. Effect of 3-hydroxy valerate content on some physical and mechanical properties of polyhydroxyalkanoates produced by Azotobacter chroococcum. Process Biochem. 2000, 36, 445–450. [Google Scholar] [CrossRef]
- Orts, W.J.; Marchessault, R.H.; Bluhm, T.L.; Hamer, G.K. Observation of strain-induced β form in poly (β-hydroxyalkanoates). Macromol. 1990, 23, 5368–5370. [Google Scholar] [CrossRef]
- Holmes, P.A. Biologically produced (R)-3-hydroxy-alkanoate polymers and copolymers. In Developments in Crystalline Polymers; Springer: Dordrecht, The Netherlands, 1988; pp. 1–35. [Google Scholar]
- Guérin, P.; Renard, E.; Langlois, V. Degradation of Natural and Artificial Poly[(R)-3-hydroxyalkanoate]s: From Biodegradation to Hydrolysis. In Plastics from Bacteria—Microbiology Monographs; Chen, G.Q., Ed.; Springer: Berlin/Heidelberg, Germany, 2010; Volume 14. [Google Scholar]
- Laycock, B.; Nikolić, M.; Colwell, J.M.; Gauthier, E.; Halley, P.; Bottle, S.; George, G. Lifetime prediction of biodegradable polymers. Prog. Polym. Sci. 2017, 71, 144–189. [Google Scholar] [CrossRef]
- Raza, Z.A.; Tariq, M.R.; Majeed, M.I.; Banat, I.M. Recent developments in bioreactor scale production of bacterial polyhydroxyalkanoates. Bioprocess Biosyst. Eng. 2019, 42, 901–919. [Google Scholar] [CrossRef]
- Koller, M. A review on established and emerging fermentation schemes for microbial production of polyhydroxyalkanoate (PHA) biopolyesters. Ferment. 2018, 4, 30. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Jana, K.; Haldar, S.; Bhowmic, A.; Mukhopadhyay, U.K.; De, S.; Mukherjee, J. Integration of poly-3-(hydroxybutyrate-co-hydroxyvalerate) production by Haloferax mediterranei through utilization of stillage from rice-based ethanol manufacture in India and its techno-economic analysis. World J. Microbiol. Biotechnol. 2015, 31, 717–727. [Google Scholar] [CrossRef]
- Patel, S.K.; Das, D.; Kim, S.C.; Cho, B.K.; Kalia, V.C.; Lee, J.K. Integrating strategies for sustainable conversion of waste biomass into dark fermentative hydrogen and value-added products Renew. Sustain. Energy Rev. 2021, 150, 111491. [Google Scholar] [CrossRef]
- Sohn, Y.J.; Son, J.; Lim, H.J.; Lim, S.H.; Park, S.J. Valorization of lignocellulosic biomass for polyhydroxyalkanoate production: Status and perspectives. Bioresour. Technol. 2022, 360, 127575. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atlić, A.; Braunegg, G. Biosynthesis of high quality polyhydroxyalkanoate co- and terpolyesters for potential medical application by the archaeon Haloferax mediterranei. Macromol. Symp. 2007, 253, 33–39. [Google Scholar] [CrossRef]
- Garcia, I.L.; Dorado Perez, M.P.; Lopez, J.A.; Villar, M.A.; Yanniotis, S.; Koutinas, A. Design and techno-economic evaluation of microbial biopolymer production from food industry wastes and agricultural crops. In Proceedings of the ICEF11 International Congress on Engineering and Food, Food Process Engineering in a Changing World, Atenas, Greece, 22–26 May 2011. [Google Scholar]
- Liu, H.; Kumar, V.; Jia, L.; Sarsaiya, S.; Kumar, D.; Juneja, A.; Awasthi, M.K. Biopolymer poly-hydroxyalkanoates (PHA) production from apple industrial waste residues: A review. Chemosphere 2021, 284, 131427. [Google Scholar] [CrossRef] [PubMed]
- Jacquel, N.; Lo, C.W.; Wei, Y.H.; Wu, H.S.; Wang, S.S. Isolation and purification of bacterial poly (3-hydroxyalkanoates). Biochem. Eng. J. 2008, 39, 15–27. [Google Scholar] [CrossRef]
- Jung, I.L.; Phyo, K.H.; Kim, K.C.; Park, H.K.; Kim, I.G. Spontaneous liberation of intracellular polyhydroxybutyrate granules in Escherichia coli. Res. Microbiol. 2005, 156, 865–873. [Google Scholar] [CrossRef]
- Saratale, G.D.; Oh, M.K. Characterization of poly-3-hydroxybutyrate (PHB) produced from Ralstonia eutropha using an alkali-pretreated biomass feedstock. Int. J. Biol. Macromol. 2015, 80, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. Established and advanced approaches for recovery of microbial polyhydroxyalkanoate (PHA) biopolyesters from surrounding microbial biomass. EuroBiotech. J. 2020, 4, 113–126. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Otari, S.V.; Jeon, J.M.; Gurav, R.; Choi, Y.K.; Bhatia, R.K.; Yang, Y.H. Biowaste-to-bioplastic (polyhydroxyalkanoates): Conversion technologies, strategies, challenges, and perspective. Bioresour. Technol. 2021, 326, 124733. [Google Scholar] [CrossRef]
- Koller, M.; Niebelschütz, H.; Braunegg, G. Strategies for recovery and purification of poly [(R)-3-hydroxyalkanoates](PHA) biopolyesters from surrounding biomass. Eng. Life Sci. 2013, 13, 549–562. [Google Scholar] [CrossRef]
- Neves, A.; Müller, J. Use of enzymes in extraction of polyhydroxyalkanoates produced by Cupriavidus necator. Biotechnol. Prog. 2012, 28, 1575–1580. [Google Scholar] [CrossRef] [PubMed]
- Khomlaem, C.; Aloui, H.; Kim, B.S. Biosynthesis of polyhydroxyalkanoates from defatted Chlorella biomass as an inexpensive substrate. Appl. Sci. 2021, 11, 1094. [Google Scholar] [CrossRef]
- Leong, Y.K.; Show, P.L.; Lan, J.C.W.; Loh, H.S.; Lam, H.L.; Ling, T.C. Economic and environmental analysis of PHAs production process. Clean. Technol. Environ. Policy 2017, 19, 1941–1953. [Google Scholar] [CrossRef]
- Koller, M.; Maršálek, L.; de Sousa Dias, M.M.; Braunegg, G. Producing microbial polyhydroxyalkanoate (PHA) biopolyesters in a sustainable manner. New Biotechnol. 2017, 37, 24–38. [Google Scholar] [CrossRef]
- Obruca, S.; Benesova, P.; Marsalek, L.; Marova, I. Use of lignocellulosic materials for PHA production. Chem. Biochem. Eng. Q. 2015, 29, 135–144. [Google Scholar] [CrossRef]
- Van Dyk, J.S.; Pletschke, B. A review of lignocellulose bioconversion using enzymatic hydrolysis and synergistic cooperation between enzymes—Factors affecting enzymes, conversion and synergy. Biotechnol. Adv. 2012, 30, 1458–1480. [Google Scholar] [CrossRef] [PubMed]
- Khare, S.K.; Pandey, A.; Larroche, C. Current perspectives in enzymatic saccharification of lignocellulosic biomass. Biochem. Eng. J. 2015, 102, 38–44. [Google Scholar] [CrossRef]
- Chundawat, S.P.; Venkatesh, B.; Dale, B.E. Effect of particle size based separation of milled corn stover on AFEX pretreatment and enzymatic digestibility. Biotechnol. Bioeng. 2007, 96, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Capolupo, L.; Faraco, V. Green methods of lignocellulose pretreatment for biorefinery development. Appl. Microbiol. Biotechnol. 2016, 100, 9451–9467. [Google Scholar] [CrossRef]
- Gullón, P.; Romani, A.; Vila, C.; Garrote, G.; Parajo, J.C. Potential of hydrothermal treatments in lignocellulose biorefineries. Biofpr 2012, 6, 219–232. [Google Scholar] [CrossRef]
- Ahn, J.; Jho, E.H.; Kim, M.; Nam, K. Increased 3HV concentration in the bacterial production of 3-hydroxybutyrate (3HB) and 3-hydroxyvalerate (3HV) copolymer with acid-digested rice straw waste. J. Environ. Polym. Degrad. 2016, 24, 98–103. [Google Scholar] [CrossRef]
- Lenihan, P.; Orozco, A.; O’Neill, E.; Ahmad, M.N.M.; Rooney, D.W.; Walker, G.M. Dilute acid hydrolysis of lignocellulosic biomass. Chem. Eng. J. 2010, 156, 395. [Google Scholar] [CrossRef]
- Dos Santos, A.C.; Ximenes, E.; Kim, Y.; Ladisch, M.R. Lignin–enzyme interactions in the hydrolysis of lignocellulosic biomass. Trends Biotechnol. 2019, 37, 518–531. [Google Scholar] [CrossRef]
- Kucera, D.; Benesova, P.; Ladicky, P.; Pekar, M.; Sedlacek, P.; Obruca, S. Production of polyhydroxyalkanoates using hydrolyzates of spruce sawdust: Comparison of hydrolyzates detoxification by application of overliming, active carbon, and lignite. Bioenergy 2017, 4, 53. [Google Scholar] [CrossRef]
- Singhania, R.R.; Patel, A.K.; Singh, A.; Haldar, D.; Soam, S.; Chen, C.W.; Dong, C.D. Consolidated bioprocessing of lignocellulosic biomass: Technological advances and challenges. Bioresour. Technol. 2022, 354, 127153. [Google Scholar] [CrossRef]
- Van-Thuoc, D.; Quillaguamán, J.; Mamo, G.; Mattiasson, B. Utilization of agricultural residues for poly(3-hydroxybutyrate) production by Halomonas boliviensis LC1. J. Appl. Microbiol. 2008, 104, 420–428. [Google Scholar] [CrossRef]
- Kanekar, P.P.; Kanekar, S.P.; Kelkar, A.S.; Dhakephalkar, P.K. Halophiles–taxonomy, diversity, physiology and applications. In Microorganisms in Environmental Management; Springer: Dordrecht, The Netherlands, 2012; pp. 1–34. [Google Scholar]
- Wang, K.; Hobby, A.M.; Chen, Y.; Chio, A.; Zhang, R. Techno-economic Analysis on An Industrial-scale Production System of Biodegradable Plastics from Cheese By-products by Haloferax mediterranei. In Proceedings of the ASABE Annual International Virtual Meeting American Society of Agricultural and Biological Engineers, Virtual, 12–16 July 2021; p. 1. [Google Scholar]
- Rodriguez-Valera, F.; Lillo, J.G. Halobacteria as producers of polyhydroxyalkanoates. FEMS Microbiol. Rev. 1992, 103, 181–186. [Google Scholar] [CrossRef]
- Tan, G.Y.A.; Chen, C.L.; Li, L.; Ge, L.; Wang, L.; Razaad, I.M.N.; Wang, J.Y. Start a research on biopolymer polyhydroxyalkanoate (PHA): A review. Polymers 2014, 6, 706–754. [Google Scholar] [CrossRef]
- Oren, A. Diversity of halophilic microorganisms: Environments, phylogeny, physiology, and applications. J. Ind. Microbiol. Biotechnol. 2002, 28, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Atlić, A.; Gonzalez-Garcia, Y.; Kutschera, C.; Braunegg, G. Polyhydroxyalkanoate (PHA) biosynthesis from whey lactose. In Macromolecular Symposia; WILEY-VCH Verlag: Weinheim, Germany, 2008; Volume 272, pp. 87–92. [Google Scholar]
- Lillo, J.G.; Rodriguez-Valera, F. Effects of culture conditions on poly (β-hydroxybutyric acid) production by Haloferax mediterranei. Appl. Environ. Microbiol. 1990, 56, 2517–2521. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.-Q.; Zhang, J. Microbial polyhydroxyalkanoates as medical implant biomaterials. Artif. Cells Nanomed. Biotechnol. 2017, 46, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.; Winterburn, J. Enhanced biosynthesis of polyhydroxyalkanoates by continuous feeding of volatile fatty acids in Haloferax mediterranei. Biochem. Eng. J. 2022, 179, 108307. [Google Scholar]
- Koller, M.; Hesse, P.; Bona, R.; Kutschera, C.; Atli, A.; Braunegg, G. Potential of various archae and eubacterial strains as industrial polyhydroxyalkanoate producers from whey. Macromol. Biosci. 2007, 7, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, M.; Salvatori, G.; Alfano, S.; Martinelli, A.; Verni, M.; Pontonio, E.; Rizzello, C.G. Exploitation of wasted bread as substrate for polyhydroxyalkanoates production through the use of Haloferax mediterranei and seawater. Front. Microbial. 2022, 13, 1000962. [Google Scholar] [CrossRef] [PubMed]
- Pais, J.; Serafim, L.S.; Freitas, F.; Reis, M.A.M. Conversion of cheese whey into poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. New Biotechnol. 2016, 33, 224–230. [Google Scholar] [CrossRef]
- Ferre-Guell, A.; Winterburn, J. Production of the copolymer poly (3-hydroxybutyrate-co-3-hydroxyvalerate) with varied composition using different nitrogen sources with Haloferax mediterranei. Extremophiles 2017, 21, 1037–1047. [Google Scholar] [CrossRef] [PubMed]
- Hermann-Krauss, C.; Koller, M.; Muhr, A.; Fasl, H.; Stelzer, F.; Braunegg, G. Archaeal production of polyhydroxyalkanoate (PHA) Co- and terpolyesters from biodiesel industry-derived by-products. Archaea 2013, 129267. [Google Scholar] [CrossRef] [PubMed]
- Moorkoth, D.; Nampoothiri, K.M. Production and characterization of poly(3-hydroxy butyrate-co-3 hydroxyvalerate) (PHBV) by a novel halotolerant mangrove isolate. Bioresour. Technol. 2016, 201, 253–260. [Google Scholar] [CrossRef]
- Fernandez-Castillo, R.; Rodriguez-Valera, F.; Gonzalez-Ramos, J.; Ruiz-Berraquero, F. Accumulation of Poly (β-Hydroxybutyrate) by Halobacteria. Appl. Environ. Microbiol. 1986, 51, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Saha, J.; Haldar, S.; Bhowmic, A.; Mukhopadhyay, U.K.; Mukherjee, J. Production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei using rice-based ethanol stillage with simultaneous recovery and re-use of medium salts. Extremophiles 2014, 18, 463–470. [Google Scholar] [CrossRef]
- Koller, M. Recycling of waste streams of the biotechnological poly (hydroxyalkanoate) production by Haloferax mediterranei on whey. Int. J. Polym. Sci. 2015, 2015, 370164. [Google Scholar] [CrossRef]
- Zhao, D.; Cai, L.; Wu, J.; Li, M.; Liu, H.; Han, J.; Xiang, H. Improving polyhydroxyalkanoate production by knocking out the genes involved in exopolysaccharide biosynthesis in Haloferax mediterranei. Appl. Microbiol. Biotechnol. 2013, 97, 3027–3036. [Google Scholar] [CrossRef] [PubMed]
- Alsafadi, D.; Al-Mashaqbeh, O. A one-stage cultivation process for the production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) from olive mill wastewater by Haloferax mediterranei. New Biotechnol. 2017, 34, 47–53. [Google Scholar] [CrossRef]
- Cui, Y.W.; Zhang, H.Y.; Ji, S.Y.; Wang, Z.W. Kinetic analysis of the temperature effect on polyhydroxyalkanoate production by Haloferax mediterranei in synthetic molasses wastewater. J. Polym. Environ. 2017, 25, 277–285. [Google Scholar] [CrossRef]
- Wang, K.; Chen, C.; Zhang, R. Process Development of Polyhydroxyalkanoates Production by Halophiles Valorising Food Waste. Bioenergy 2022, 9, 630. [Google Scholar] [CrossRef]
- Raho, S.; Carofiglio, V.E.; Montemurro, M. Production of the polyhydroxyalkanoate PHBV from ricotta cheese exhausted whey by Haloferax mediterranei fermentation. Foods 2020, 9, 1459. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.Y.; Duan, K.J.; Huang, S.Y.; Chen, C.W. Production of polyhydroxyalkanoates from inexpensive extruded rice bran and starch by Haloferax mediterranei. J. Ind. Microbiol. Biotechnol. 2006, 33, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Khamplod, T.; Wongsirichot, P.; Winterburn, J. Production of polyhydroxyalkanoates from hydrolysed rapeseed meal by Haloferax mediterranei. Bioresour. Technol. 2023, 386, 129541. [Google Scholar] [CrossRef] [PubMed]
- Stanley, A.; Mutturi, S.; Vijayendra, S.V.N. Halophilic Microorganisms as Potential Producers of Polyhydroxyalkanoates Kuddus, M. (Ed.). In Bioplastics for Sustainable Development; Springer: Singapore, 2021. [Google Scholar]
- Rodriguez-Perez, S.; Serrano, A.; Pantión, A.A.; Alonso-Fariñas, B. Challenges of scaling-up PHA production from waste streams. A review. J. Environ. Manag. 2018, 205, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K.; Battistuzzi, F. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Yadav, B.; Talan, A.; Tyagi, R.D.; Drogui, P. Concomitant production of value-added products with polyhydroxyalkanoate (PHA) synthesis: A review. Bioresour. Technol. 2021, 337, 125419. [Google Scholar] [CrossRef] [PubMed]
- Lorantfy, B.; Ruschitzka, P.; Herwig, C. Investigation of physiological limits and conditions for robust bioprocessing of an extreme halophilic archaeon using external cell retention system. Biochem. Eng. J. 2014, 90, 140–148. [Google Scholar] [CrossRef]
- Antón, J.; Meseguer, I.; Rodriguez-Valera, F. Production of an extracellular polysaccharide by Haloferax mediterranei. Appl.Environ. Microbiol. 1988, 54, 2381–2386. [Google Scholar] [CrossRef]
- Fang, C.J.; Ku, K.L.; Lee, M.H.; Su, N.W. Influence of nutritive factors on C50 carotenoids production by Haloferax mediterranei ATCC 33500 with two-stage cultivation. Bioresour. Technol. 2010, 101, 6487–6493. [Google Scholar] [CrossRef]
- Giani, M.; Garbayo, I.; Vílchez, C.; Martínez-Espinosa, R.M. Haloarchaeal carotenoids: Healthy novel compounds from extreme environments. Mar. Drugs 2019, 17, 524. [Google Scholar] [CrossRef]
- Platas, G.; Meseguer, I.; Amils, R. Purification and biological characterization of halocin H1 from Haloferax mediterranei M2a. Int. Microbiol. 2002, 5, 15–19. [Google Scholar] [CrossRef]
- Kumar, V.; Singh, B.; van Belkum, M.J.; Diep, D.B.; Chikindas, M.L.; Ermakov, A.M.; Tiwari, S.K. Halocins, natural antimicrobials of Archaea: Exotic or special or both? Biotechnol. Adv. 2021, 53, 107834. [Google Scholar] [CrossRef]
- Ladhari, S.; Vu, N.N.; Boisvert, C.; Saidi, A.; Nguyen-Tri, P. Recent Development of Polyhydroxyalkanoates (PHA)-based Materials for Antibacterial applications: A review. ACS Appl. Bio Mater. 2023, 6, 1398–1430. [Google Scholar] [CrossRef]
- Keshavarz, T.; Roy, I. Polyhydroxyalkanoates: Bioplastics with a green agenda. Curr. Opin. Microbiol. 2010, 13, 321–326. [Google Scholar] [CrossRef]
- Madison, L.L.; Huisman, G.W. Metabolic engineering of poly (3-hydroxyalkanoates): From DNA to plastic. Microbiol. Mol. Biol. Rev. 1999, 63, 21–53. [Google Scholar] [CrossRef]
- Gumel, A.M.; Annuar, M.S.M.; Chisti, Y. Recent advances in the production, recovery and applications of polyhydroxyalkanoates. J. Environ. Polym. Degrad. 2013, 21, 580–605. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Dalla Rosa, M. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Peelman, N.; Ragaert, P.; De Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; Devlieghere, F. Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef]
- Choi, J.S.; Park, W.H. Effect of biodegradable plasticizers on thermal and mechanical properties of poly (3-hydroxybutyrate). Polym. Test. 2004, 23, 455–460. [Google Scholar] [CrossRef]
- Meereboer, K.W.; Misra, M.; Mohanty, A.K. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. [Google Scholar] [CrossRef]
- Requena, R.; Vargas, M.; Chiralt, A. Obtaining antimicrobial bilayer starch and polyester-blend films with carvacrol. Food Hydrocoll. 2018, 83, 118–133. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Fortunati, E.; Dominici, F.; Rayón, E.; López, J.; Kenny, J.M. PLA-PHB/cellulose based films: Mechanical, barrier and disintegration properties. Polym. Degrad. Stab. 2014, 107, 139–149. [Google Scholar] [CrossRef]
- Soares da Silva, F.; Matos, M.; Dourado, F.; Reis, M.; Branco, P.; Poças, F.; Gama, M. Development of a layered bacterial nanocellulose-PHBV composite for food packaging. J. Sci. Food Agric. 2023, 103, 1077–1087. [Google Scholar] [CrossRef]
- Melendez-Rodriguez, B.; Castro-Mayorga, J.L.; Reis, M.A.; Sammon, C.; Cabedo, L.; Torres-Giner, S.; Lagaron, J.M. Preparation and characterization of electrospun food biopackaging films of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) derived from fruit pulp biowaste. Front. Sustain. Food Syst. 2018, 2, 38. [Google Scholar] [CrossRef]
- Râpă, M.; Stefan, M.; Popa, P.A.; Toloman, D.; Leostean, C.; Borodi, G.; Matei, E. Electrospun nanosystems based on PHBV and ZnO for ecological food packaging. Polymers 2021, 13, 2123. [Google Scholar] [CrossRef]
- Melendez-Rodriguez, B.; Torres-Giner, S.; Lorini, L.; Valentino, F.; Sammon, C.; Cabedo, L.; Lagaron, J.M. Valorization of municipal biowaste into electrospun poly (3-hydroxybutyrate-co-3-hydroxyvalerate) biopapers for food packaging applications. ACS Appl. Bio Mater. 2020, 3, 6110–6123. [Google Scholar] [CrossRef]
- Chen, C.; Xu, Z.; Ma, Y.; Liu, J.; Zhang, Q.; Tang, Z.; Fu, K.; Yang, F.; Xie, J. Properties, vapour-phase antimicrobial and antioxidant activities of active PVA packaging films incorporated with clove oil. Food Control 2018, 88, 105–112. [Google Scholar] [CrossRef]
- Vilela, C.; Kurek, M.; Hayouka, Z.; Röcker, B.; Yildirim, S.; Antunes, M.D.C.; Nilsen-Nygaard, J.; Pettersen, M.K.; Freire, C.S.R. A concise guide to active agents for active food packaging. Trends Food Sci. Technol. 2018, 80, 212–222. [Google Scholar] [CrossRef]
- Yildirim, S.; Röcker, B.; Pettersen, M.K.; Nilsen-Nygaard, J.; Ayhan, Z.; Rutkaite, R.; Radusin, T.; Suminska, P.; Marcos, B.; Coma, V. Active Packaging Applications for Food. CRFSFS 2017, 17, 165–199. [Google Scholar] [CrossRef]
- Xavier, J.R.; Babusha, S.T.; George, J.; Ramana, K.V. Material properties and antimicrobial activity of polyhydroxybutyrate (PHB) films incorporated with vanillin. Appl. Biochem. Biotechnol. 2015, 176, 1498–1510. [Google Scholar] [CrossRef]
- Melendez-Rodriguez, B.; Figueroa-Lopez, K.J.; Bernardos, A.; Martínez-Máñez, R.; Cabedo, L.; Torres-Giner, S.; Lagaron, J.M. Electrospun antimicrobial films of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) containing eugenol essential oil encapsulated in mesoporous silica nanoparticles. Nanomater. 2019, 9, 227. [Google Scholar] [CrossRef]
- Requena, R.; Vargas, M.; Chiralt, A. Eugenol and carvacrol migration from PHBV films and antibacterial action in different food matrices. Food Chem. 2019, 277, 38–45. [Google Scholar] [CrossRef]
- Moll, E.; Chiralt, A. Polyhydroxybutyrate-co-hydroxyvalerate (PHBV) with Phenolic Acids for Active Food Packaging. Polymers 2023, 15, 4222. [Google Scholar] [CrossRef]
- Castro-Mayorga, J.L.; Fabra, M.J.; Lagaron, J.M. Stabilized nanosilver based antimicrobial poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanocomposites of interest in active food packaging. IFSET 2016, 33, 524–533. [Google Scholar] [CrossRef]
- Ojha, N.; Das, N. Fabrication and characterization of biodegradable PHBV/SiO2 nanocomposite for thermo-mechanical and antibacterial applications in food packaging. IET Nanobiotech. 2020, 14, 785–795. [Google Scholar] [CrossRef]
- Bonnenfant, C.; Gontard, N.; Aouf, C. PHBV-based polymers as food packaging: Physical-chemical and structural stability under reuse conditions. Polymer 2023, 270, 125784. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Iiyas, S.; Hezam, A.; Mathur, S.; Byrappa, K. Smart Fortified PHBV-CS Biopolymer with ZnO–Ag Nanocomposites for Enhanced Shelf Life of Food Packaging. ACS Appl. Mater. Interfaces 2019, 11, 48309–48320. [Google Scholar] [CrossRef]
- Hernández-García, E.; Vargas, M.; González-Martínez, C.; Chiralt, A. Biodegradable antimicrobial films for food packaging: Effect of antimicrobials on degradation. Foods 2021, 10, 1256. [Google Scholar] [CrossRef]

| Type of PHAs | Production Technique | Tg (°C) | Tm (°C) | EM (GPa) | TS (MPa) | E (%) | Xc (%) | WVP·1015 | OP·1019 | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| PHB | Solvent casting | −2–9 | 160–180 | 1.2–3.6 | 19–44 | 2–5 | 57–63 | 2–10 | 8 | [20,21,22] |
| Melt blending | 10–18 | 170–180 | 1.5–3.5 | 8–40 | 0.8–2.1 | 45–50 | 8 | 5–8 | [23,24,25] | |
| P(3HB-3HV8 *) | Melt blending | 7 | 170.6–171.2 | 1.7 | 37 | 3.4 | 73.8–99.06 | 5.7 | - | [26,27] |
| P(3HB-3HV12 *) | Solvent casting | - | 140–155 | 0.6 | 6.4 | 1.4 | 33 | 50 | 15 | [28] |
| Melt blending | −3–2 | 140–155 | 1.7 | 14.6 | 1.2 | 37 | 4 | 15–18 | [29,30,31] | |
| P(3HB-3HV50–55 *) | Solvent casting | −10 | 77 | - | 16 | 1200 | - | - | - | [29] |
| Melt blending | 0 | 162 | 0.4 | 13.4 | 230 | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atarés, L.; Chiralt, A.; González-Martínez, C.; Vargas, M. Production of Polyhydroxyalkanoates for Biodegradable Food Packaging Applications Using Haloferax mediterranei and Agrifood Wastes. Foods 2024, 13, 950. https://doi.org/10.3390/foods13060950
Atarés L, Chiralt A, González-Martínez C, Vargas M. Production of Polyhydroxyalkanoates for Biodegradable Food Packaging Applications Using Haloferax mediterranei and Agrifood Wastes. Foods. 2024; 13(6):950. https://doi.org/10.3390/foods13060950
Chicago/Turabian StyleAtarés, Lorena, Amparo Chiralt, Chelo González-Martínez, and Maria Vargas. 2024. "Production of Polyhydroxyalkanoates for Biodegradable Food Packaging Applications Using Haloferax mediterranei and Agrifood Wastes" Foods 13, no. 6: 950. https://doi.org/10.3390/foods13060950
APA StyleAtarés, L., Chiralt, A., González-Martínez, C., & Vargas, M. (2024). Production of Polyhydroxyalkanoates for Biodegradable Food Packaging Applications Using Haloferax mediterranei and Agrifood Wastes. Foods, 13(6), 950. https://doi.org/10.3390/foods13060950






