The Cardioprotective Role of Nitrate-Rich Vegetables
Abstract
1. Introduction
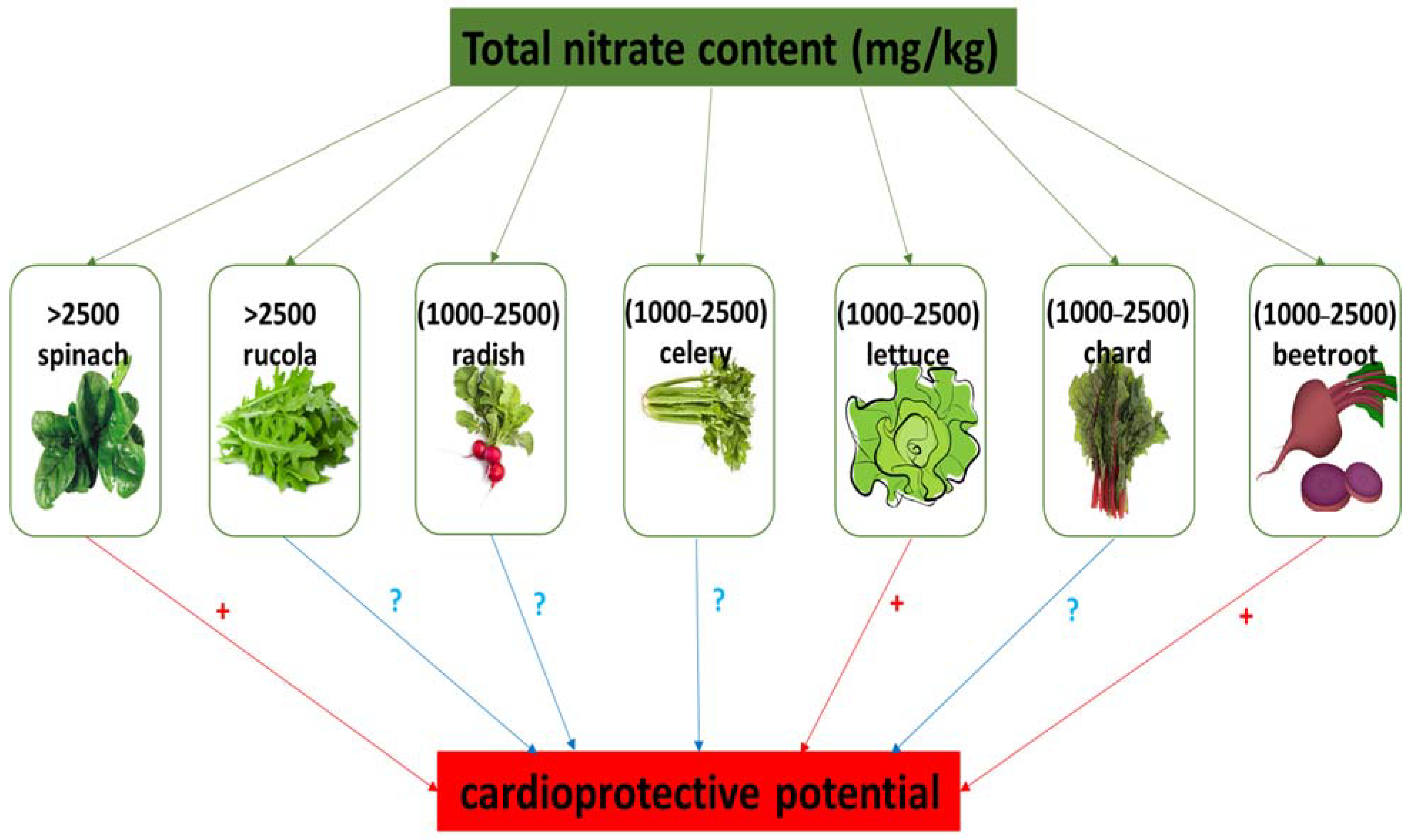
2. Dietary Sources of Nitrate
| Nitrate Content | Dietary Sources | References |
|---|---|---|
| Vegetables | ||
| Very high (>2500 mg/kg fw) | Celery, cress, chervil, lettuce, beetroot, spinach, rucola | [18] |
| High (1000 to 2500 mg/kg fw) | Celeriac, Chinese cabbage, endive, fennel, leek, parsley | [18] |
| Medium (500 to 1000 mg/kg fw) | Cabbage, dill, turnip | [18] |
| Low (200 to <500 mg/kg fw) | Broccoli, carrot, cauliflower, cucumber, pumpkin, chicory | [18] |
| Very Low (<200 mg/kg fw) | Artichoke, asparagus, garlic, onion, green bean, mushroom, pepper, potato, sweet potato, tomato, watermelon, apple, banana, grape, pear, orange, strawberry | [18,35] |
| Very low (<20 mg/kg fw) | Meat | [10] |
| Very low (5 mg per 100 g) | Water | [10] |
3. Regulatory Limits of Dietary Nitrate and Nitrite
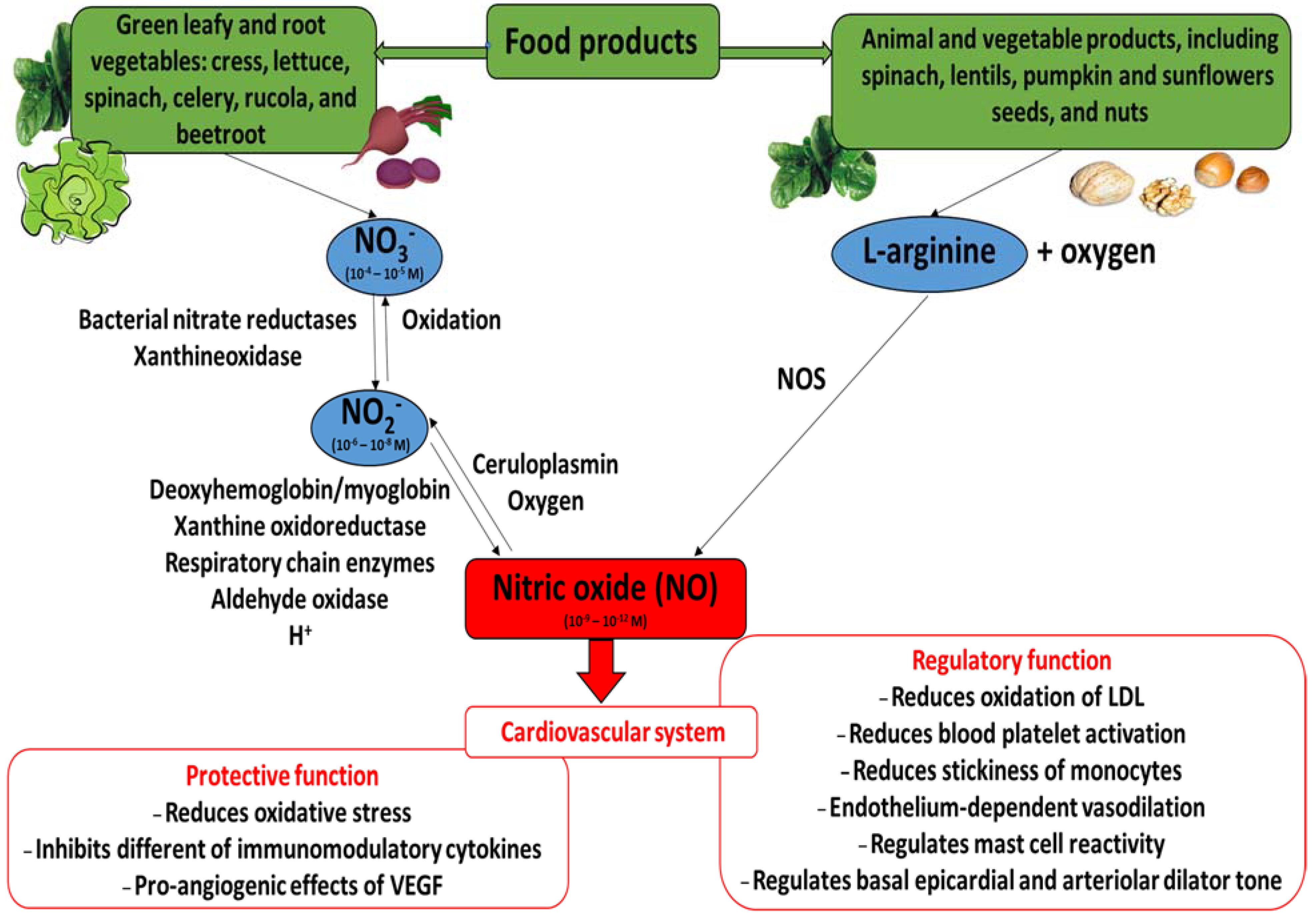
4. Nitric Oxide and Cardiovascular System
5. Role of Vegetable Nitrate in CVDs
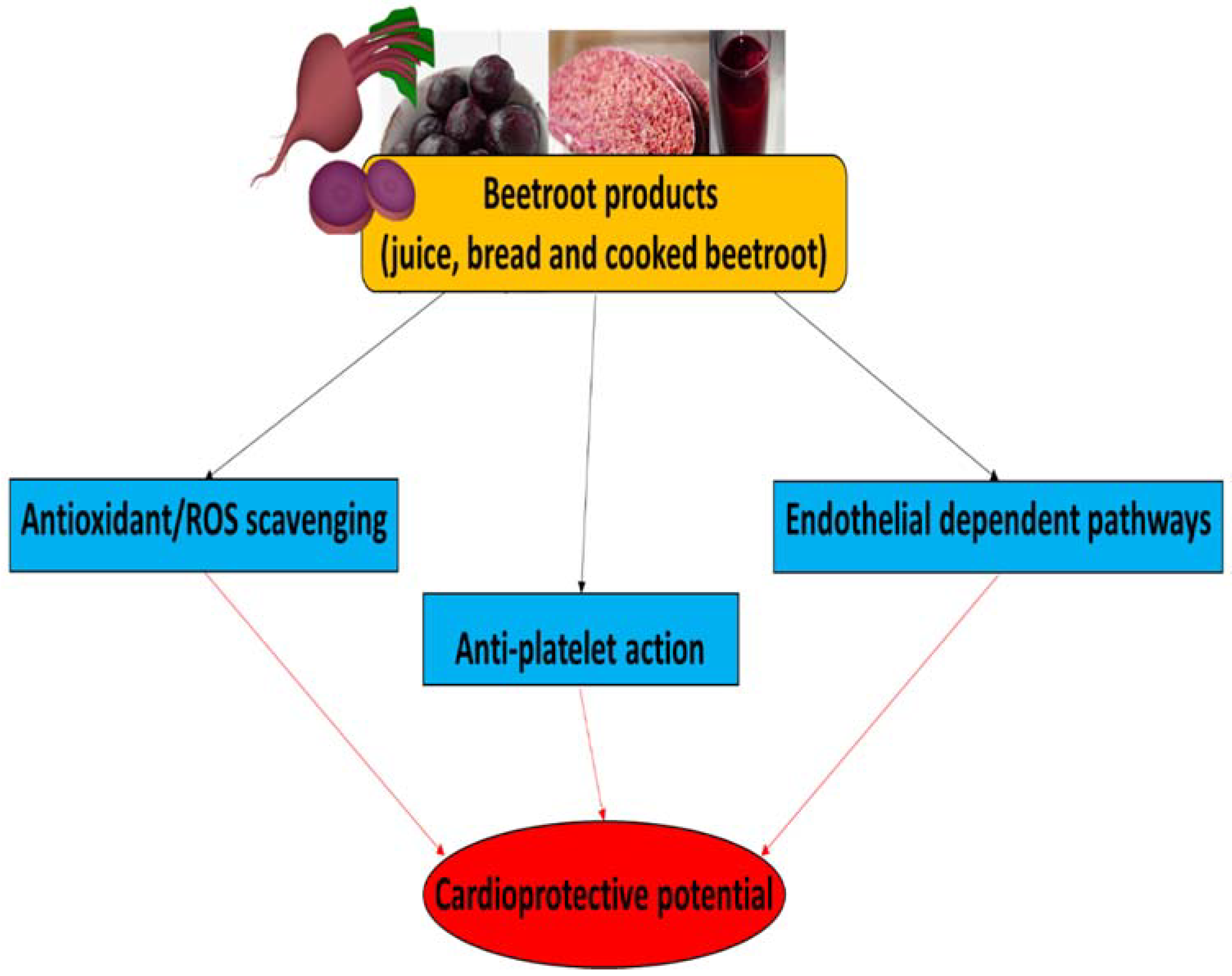
6. Beetroot Products
7. Spinach Products
8. Lettuce and Other Nitrate-Rich Vegetable Products
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Govoni, M.; Jansson, E.A.; Weitzberg, E.; Lundberg, J.O. The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide 2008, 19, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; et al. Acute blood pressure lowering, vsoprotective, and antiplatelet properties of dietry nitrate via bioconversion to nitrite. Hypertension 2008, 51, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Olas, B. Gasomediators (NO, CO, and H2S) and their role in hemostasis and thrombosis. Clin. Chim. Acta 2015, 445, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Khatri, J.; Mills, C.E.; Maskell, P.; Odongerel, C.; Webb, A.J. It is rocket science—Why dietary nitrate is hard to “beet”! Part I—twists and turns in the realization of the nitrate-nitrite-NO pathway. Br. J. Clin. Pharmacol. 2017, 83, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.E.; Khatri, J.; Maskell, P.; Odongerel, C.; Webb, A.J. It is rocket science—Why dietary nitrate is hard to “beet”! Part II: Further mechanisms and therapeutic potential of the nitrate-nitrite-NO pathway. Br. J. Clin. Pharmacol. 2017, 83, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Malone-Povolny, M.; Maloney, S.E.; Schoenfisch, M.H. Nitric oxide therapy for diabetic wound healing. Adv. Healthc. Mater. 2019, 8, e1801210. [Google Scholar] [CrossRef]
- Jones, A.M.; Vanhatalo, A.; Seals, D.R.; Rossman, M.J.; Piknova, B.; Jonvik, K.L. Dietary nitrate and nitric oxide metabolism: Mouth, circulation, skeletal muscle, and exercise performance. Med. Sci. Sports Exerc. 2021, 1, 280–294. [Google Scholar] [CrossRef]
- Dudhe, R.; Dudhe, A.C.; Raut, S.D. Significance of inorganic nitrate supplement in cardiovascular health. Cardiovasc. Hematol. Agents Med. Chem. 2022, 20, 83–89. [Google Scholar] [CrossRef]
- Kiani, A.K.; Bonetti, G.; Medori, M.C.; Caruso, P.; Manganotti, P.; Fioretti, F.; Nodari, S.; Connelly, S.T.; Bertelli, M. Dietary supplements for improving nitric-oxide synthesis. J. Prev. Med. Hyg. 2022, 63, E239–E245. [Google Scholar]
- Rajendra, A.; Bondonno, N.P.; Rainey-Smith, S.R.; Gardener, S.L.; Hodgson, J.M.; Bondonno, C.P. Potential role of dietary nitrate in relation to cardiovascular and cerebrovascular health, cognition, cognitive decline and dementia: A review. Food Funct. 2022, 13, 12572–12589. [Google Scholar] [CrossRef]
- Capper, T.E.; Siervo, M.; Clifford, T.; Taylor, G.; Iqbal, W.; West, D.; Stevenson, E.J. Pharmacokinetic profile of incremental oral doses of dietary nitrate in young and older adults: A crossover randomized trial. J. Nutr. 2022, 152, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Capper, T.; Clifford, T.; Taylor, G.; Iqbal, W.; West, D.; Stevenson, E.; Siervo, M. Ageing modifies acute resting blood pressure responses to incremental consumption of dietary nitrate: A randomized, croos-over clinical trial. Br. J. Nutr. 2023, 129, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.M.; Townsend, J.R.; Pinzone, A.G.; Hoffman, J.R. Supplementation with nitric oxide precursors for strength performance: A review of the current literature. Nutrients 2023, 15, 660. [Google Scholar] [CrossRef] [PubMed]
- Tejada-Jimenez, M.; Chamizo-Ampudia, A.; Calatrava, V.; Galvan, A.; Fernandez, E.; Liamas, A. From the eukaryotic molybdenum cofactor biosynthesis to the moonlighting enzyme mARC. Molecules 2018, 23, 3287. [Google Scholar] [CrossRef] [PubMed]
- Sparacino-Watkins, C.E.; Tejero, J.; Sun, B.; Gauthier, M.C.; Thomas, J.; Ragireddy, V.; Merchant, B.A.; Wang, J.; Azarov, I.; Basu, P.; et al. Nitrite reductase and nitric-oxide synthase activity of the mitochondrial molybdopterin enzymes mARC1 and mARC2. J. Biol. Chem. 2014, 289, 10345–10358. [Google Scholar] [CrossRef] [PubMed]
- Bryan, N.S. Cardioproetctive actions of nitrite therapy and dietary considerations. Front. Biosci. 2009, 14, 4793–4808. [Google Scholar] [CrossRef] [PubMed]
- Hord, N.G.; Ghannam, J.S.; Garg, H.K.; Berens, P.D.; Bryan, N.S. Nitrate and nitrite content of human, formula, bovine, and soy milks: Implications for dietary nitrite and nitrate recommendations. Breastfeed Med. 2011, 6, 393–399. [Google Scholar] [CrossRef]
- Hord, N.G. Dietary nitrates, nitrites, and cardiovascular disease. Curr. Atheroscler. Rep. 2011, 13, 484–492. [Google Scholar] [CrossRef]
- Hord, N.G.; Tang, Y.; Bryan, N.S. Food sources of nitrates and nitrites: The physiologic context for potential health benefits. Am. J. Clin. Nutr. 2009, 90, 2. [Google Scholar] [CrossRef]
- Verma, T.; Sinha, M.; Bansal, N.; Raj Yadav, S.; Shah, K.; Chauhan, N.G. Plants used as antihypertensive. Nat. Prod. Bioprosp. 2021, 11, 155–184. [Google Scholar] [CrossRef]
- Rolnik, A.; Olas, B. Vegetables from Cucurbitaceae family anf their products: Positive effect on human health. Nutrition 2020, 78, 110788. [Google Scholar] [CrossRef] [PubMed]
- Rolnik, A.; Olas, B. The plants of the Asteraceae family as agents in the protection of human health. Intern. J. Molec. Sci. 2021, 22, 3009. [Google Scholar] [CrossRef]
- Benjamin, C.J.R.; Sousa, Y.B.A.; Porto, A.A.; de Moraes Pontes, Y.M.; Tavares, S.S.; da Silva Rodrigues, G.; da Silva, L.S.L.; da Silva Goncalves, L.; Uimaraes, C.S.; Rebelo, M.A.; et al. Nitrate-rich beet juice intake on cardiovascular performance in response to exercise in postmenopausal women with arterial hypertension: Study protocol for randomized controlled trial. Trials 2023, 24, 94. [Google Scholar] [CrossRef]
- Ahluwalia, A.; Gladwin, M.; Coleman, G.D.; Hord, N.; Howard, G.; Kim-Shapiro, D.B.; Lajous, M.; Larsen, F.J.; Lefer, D.J.; McClure, L.A.; et al. Dietary nitrate and the epidemiology of cardiovascular disease: Report from a National Heart, lung, and blood institute workshop. J. Am. Heart Assoc. 2016, 5, e003402. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Nitrate in vegetables. Scientific opinion of the panel on contaminants in the food chain. EFSA J. 2008, 689, 1–79. [Google Scholar]
- Rahimi, P.; Mesbah-Namin, A.S.; Ostadrahimi, A.; Abedimanesh, S.; Separham, A.; Jafarabadi, M.A. Effects of betalains on atherogenic risk factors in patients with atheroserotic caradiovascular disease. Food Funct. 2019, 10, 8286–8297. [Google Scholar] [CrossRef]
- Anjana, A.; Umar, S.; Iqbal, M.; Abrol, Y.P. Are nitrate concentrations in leafy vegetables within safe limits? Curr. Sci. 2007, 92, 355–360. [Google Scholar]
- Ding, Z.; Johanningsmeir, S.D.; Price, R.; Reynolds, R.; Troung, V.D.; Payton, S.C.; Breidt, F. Evaluation of nitrate and nitrite content in pickled fruit and vegetable products. Food Control. 2018, 90, 304–311. [Google Scholar] [CrossRef]
- Jones, A.M. Dietary nitrate supplementation and exercise performance. Sports Med. 2014, 44, S35–S45. [Google Scholar] [CrossRef]
- Tan, R.; Cano, L.; Lago-Rodriguez, A.; Dominguez, R. The effects of dietary nitrate supplementation on explosive exercise performance: A systemiatic review. Int. J. Environ. Res. Public Health 2022, 19, 762. [Google Scholar] [CrossRef]
- Van Velzen, A.G.; Sips, A.J.; Schothorst, R.C.; Lambers, A.C.; Meulenbelt, J. The oral bioavailability of nitrate from nitrate-rich vegetables in human. Toxicol. Lett. 2008, 181, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Wootton-Beard, P.C.; Moran, A.; Ryan, L. Stability of the total antioxidant capacity and total polyphenol content of 23 commercially available vegetable juices before and after in vitro digestion measured by FRAP, DPPH, ABTS and Folin-Ciocalteu methods. Food Res. Int. 2011, 44, 217–224. [Google Scholar] [CrossRef]
- Wootton-Beard, P.C.; Ryan, L. Combined use of multiple methodologies for the measurement of total antioxidant capacity in UK commercially available vegetable juices. Plant Foods Hum. Nutr. 2012, 67, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Cocksedge, S.P.; Causer, A.J.; Winyard, P.G.; Jones, A.M.; Bailey, S.J. Oral temperature and pH influence dietary nitrate metabolism in healthy adults. Nutrients 2023, 15, 784. [Google Scholar] [CrossRef]
- Uddin, R.; Thakur, M.U.; Uddin, M.Z.; Islam, G.M.R. Study of nitrate levels in fruits and vegetables to assess the potential health risks in Bangladesh. Sci. Rep. 2021, 1, 4704. [Google Scholar] [CrossRef] [PubMed]
- Ocampo, D.A.B.; Paipilla, A.F.; Marin, E.; Vargas-Molina, S.; Petro, J.L.; Perez-Idarraga, A. Dietary nitrate from beetroot juice for hypertension: A systematic review. Biomolecules 2018, 2, 1–11. [Google Scholar]
- Zamani, P.; Rawat, D.; Shiva-Kumar, P.; Geraci, S.; Bhuva, R.; Konda, P.; Doulias, P.T.; Ischiropoulos, H.; Townsend, R.R.; Margulies, K.B.; et al. Effect of inorganicnitrate on exercise capacity in heart failure with preserved ejection fraction. Circulation 2015, 131, 371–380. [Google Scholar] [CrossRef]
- Zamani, H.; de Joode, M.E.J.R.; Hossein, I.J.; Henckens, N.F.T.; Guggeis, M.A.; Berends, J.E.; de Kok, T.M.C.M.; van Breda, S.G.J. The benefits and risks of beetrrot juice consumption: A systematic review. Crit. Rev. Food Sci. Nutr. 2021, 61, 788–804. [Google Scholar] [CrossRef]
- Clifford, T.; Howatson, G.; West, D.J.; Stevenson, E.J. The potential benefits of red beetroot supplementation in health and disease. Nutrients 2015, 7, 2801–2822. [Google Scholar] [CrossRef] [PubMed]
- dos Baiao, D.S.; da Silva, D.V.T.; Paschoalin, V.M.F. Beetroot, a remarkable vegetable: Its nitrate and phytochemical contents can be adjusted in novel formulations to benefit health and support cardiovascular disease therapies. Antioxidants 2020, 9, 960. [Google Scholar] [CrossRef] [PubMed]
- Corleto, K.A.; Singh, J.; Jayaprakasha, G.K.; Patil, B.S. Storage stability of dietary nitrate and phenolic compounds in beetrrot (Beta vulgaris) and arugula (Eruca sativa) juices. J. Food Sci. 2018, 83, 1237–1248. [Google Scholar] [CrossRef]
- WHO. Nitrate and Nitrite. In WHO Food Additive Series; WHO: Geneva, Switzerland, 2014; Volume 50, pp. 1–14. [Google Scholar]
- EFSA. EFSA Confirms Safe Levels for Nitrites and Nitrtes to Food; EFSA: Parma, Italy, 2017. [Google Scholar]
- Karwowska, M.; Kononiuk, A. Nitrates/nitrites in food—Risk for nitrative stress and benefits. Antioxidant 2020, 9, 241. [Google Scholar] [CrossRef]
- Shi, M.; Gu, J.; Wu, H.; Rauf, A.; Emran, T.B.; Khan, Z.; Mitra, S.; Aljohani, A.S.M.; Alhumaydhi, F.A.; Al-Awthan, Y.S.; et al. Phytochemicals, nutrition, metabolism, bioavailability, and health benefits in lettuce—A comprehensive review. Antioxidants 2022, 11, 1158. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Carlstrom, M.; Weitzberg, E. Metabolic effects of dietary nitrate in health and disease. Cell Metab. 2018, 28, 9–22. [Google Scholar] [CrossRef]
- Tricker, A.R.; Preussmann, R. Carcinogenic N-nitrosamines in the diet: Occurrence, formation, mechanism and carcinogenesis potential. Mut. Res. 1991, 259, 277–289. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, Y.; Hu, Z.; Wu, S.; Jin, C. Beetroot as a functional food with huge health benefits: Antioxidant, antitumor, physical function, and chronic metabolomics activity. Food Sci. Nutr. 2021, 9, 6406–6420. [Google Scholar] [CrossRef] [PubMed]
- Bescos, R.; Sureda, A.; Tur, J.A.; Pons, A. The effect of nitric-oxide-related supplements on human performance. Sports Med. 2012, 42, 99–117. [Google Scholar] [CrossRef] [PubMed]
- Grimble, G.K. Adverse gastrointestinal effects of arginine and related amino acids. J. Nutr. 2007, 137, 1693S–1701S. [Google Scholar] [CrossRef] [PubMed]
- Joseph, P.; Leong, D.; McKee, M.; Anand, S.S.; Schwalm, J.-D.; Teo, K.; Mente, A.; Yusuf, S. Reducing the global burden of cardiovascular disease. Part 1: The epidemiology and risk factors. Circ. Res. 2017, 121, 677–694. [Google Scholar] [CrossRef] [PubMed]
- Lakatta, E.G. Arterial and cardiac aging major shereholders in cardiovascular disease enterpriss. Part III: Cellular and molecular clues to heart and arterial aging. Circulation 2003, 107, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.; Berglund, L.; Larsson, A.; Sundstrom, J. Endothelial function in resistance and conduit arteries and 5-year risk of cardiovascular disease. Circulation 2011, 123, 1545–1551. [Google Scholar] [CrossRef]
- Seals, D.R.; Jablonski, K.L.; Donato, A.J. Aging and vascular endothelial function in humans. Clin. Sci. 2011, 120, 357–375. [Google Scholar] [CrossRef]
- Sindler, A.L.; Fleenor, B.S.; Calvert, J.W.; Marshall, K.D.; Zigler, M.L.; Lefer, D.J.; Seals, D.R. Nitrite supplementation reverses vascular endothelial dysfunction and large elastic artery stiffness with aging. Aging Cell 2011, 10, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Sindler, A.L.; Devan, A.E.; Fleenor, B.S.; Seals, D.R. Inorganic nitrite supplementation for healthy arterial aging. J. Appl. Physiol. 2014, 116, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Fleenor, B.S.; Sindler, A.L.; Eng, J.S.; Nair, D.P.; Dodson, R.B.; Seals, D.R. Sodium nitrite de-stiffening of large elastic arteries with aging: Role of normalization of advanced glycation end-products. Exp. Gerontol. 2012, 47, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, A.; Mitechell, K.; Blackwell, J.R.; Vanhatalo, A.; Jones, A.M. High-nitrate vegetable diet increases plasma nitrate and nitrite concentrations and reduces blood pressure in healthy women. Public Health Nutr. 2015, 18, 2669–2678. [Google Scholar] [CrossRef] [PubMed]
- Berry, M.J.; Justus, N.W.; Hauser, J.I.; Case, A.H.; Helms, C.C.; Basu, S.; Miller, G.D. Dietary nitrate supplementation improves exercise performance and decreases blood pressure in COPD patients. Nitric Oxide 2015, 48, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Khambata, R.S.; Robertson, A.; Caulfield, M.J.; Ahluwalia, A. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients. A randomized, phase 2, double-blind, placebo-controlled study. Hypertension 2015, 65, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Woodward, K.A.; Santos-Parker, J.R.; Lubieniecki, K.L.; Nagy, E.; Bryan, N.S.; Chonchol, M.; Justice, J.N.; Seals, D.R.; Rossman, M.J. Sodium nitrite supplementation improves vascular endothelial function but not motor or cognitive function in middle-aged and older adults. FASEB J. 2019, 33, 833.13. [Google Scholar] [CrossRef]
- Bailey, S.J.; Winyard, P.; Vanhatalo, A.; Blockwell, J.R.; Di Menna, F.J.; Wilkerson, D.P.; Tarr, J.; Benjmanin, N.; Jones, A.M. Dietary nitrate supplementation reduces the O2 cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J. Appl. Physiol. 2009, 107, 1144–1155. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Pavey, T.G.; Wilkerson, D.P.; Benjamin, N.; Winyard, P.G.; Jones, A.M. Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R1121–R1131. [Google Scholar] [CrossRef]
- Milton-Laskibar, I.; Martinez, J.A.; Portillo, M.P. Current knowledge on beetrrot bioactive compounds: Role of nitrate and betalains in health and disease. Foods 2021, 10, 1314. [Google Scholar] [CrossRef]
- Ninfali, P.; Angelino, D. Nutritional and functional potential of Beta vulgaris cicla and rubra. Fitoterapia 2013, 89, 188–199. [Google Scholar] [CrossRef] [PubMed]
- dos Baiao, D.S.; da Silva, D.V.T.; Del Aguila, E.M.; Paschoalin, V.M.F. Nutritional, bioactive and physicochemical characteristics of different beetroot formulations. In Food Additivies; Karunaratne, D.N., Pamunuowa, G., Eds.; Intech Open: London, UK, 2017; Chapter 2; pp. 21–44. [Google Scholar]
- da Silva, D.V.T.; dos Santo Baiao, D.; Ferreira, V.F.; Paschoalin, V.M.F. Betanin as amultipath oxidative stress and inflammation modulator: A beetroot pigment with protective effects on cardiovascular disease pathogenesis. Crit. Rev. Food Sci. Nutr. 2022, 62, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Drinks, J.W. Nitrate 3000 Concentrate. 2019. Available online: https://www.beet-it.com/buy-now/beet-it-sport-nitrate-3000 (accessed on 4 June 2019).
- Brzezinska-Rojek, J.; Sagatovych, S.; Malinowska, P.; Gadaj, P.; Prokopowicz, M.; Grembecka, M. Antioxidant capacity, nitrate and nitrite content in beetroot-based dietary supplements. Foods 2023, 12, 1017. [Google Scholar] [CrossRef]
- Hobbs, D.A.; Goulding, M.G.; Nguyen, A.; Malaver, T.; Walker, C.F.; George, T.W.; Lovegrove, J.A. Acute ingestion of beetroot bread increases endothelium-independent vasodilation and lowers diastolic blood pressure in healthy men: A randomized controlled trial. J. Nutr. 2013, 143, 1399–1405. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, D.A.; Kaffa, N.; George, T.W.; Methven, L.; Lovegrove, J.B. Blood pressure-lowering effects of beetroot juice and novel beetroot-enriched bread products in normotensive male subjects. Br. J. Nutr. 2012, 108, 2066–2074. [Google Scholar] [CrossRef] [PubMed]
- Czyżewska, A.; Klewicka, E.; Libudzisz, Z. The influence of lactic acid fermentation proces of red beet juice on the stability of biologically colorants. Eur. Food Res. Technol. 2006, 223, 110–116. [Google Scholar] [CrossRef]
- Stanaway, L.; Rutherfurd-Markwick, K.; Page, R.; Ali, A. Performance and health benefits of dietary nitrate supplementation in older adults: A systematic review. Nutrients 2017, 9, 1171. [Google Scholar] [CrossRef] [PubMed]
- Remington, J.; Winters, K. Effectiveness of dietary inorganic nitrate for lowering blood pressure in hypertensive adults: A systematic review. JBI Database Syst. Rev. Imploment. Rep. 2019, 17, 365–389. [Google Scholar] [CrossRef]
- Siervo, M.; Lara, J.; Ogbonmwan, I.; Mathers, J.C. Inorganic nitrate and beetroot juice supplementation reduces blood pressure in dults: A systematic review and meta-analysis. J. Nutr. 2013, 143, 818–826. [Google Scholar] [CrossRef]
- Kim, D.J.; Roe, C.A.; Somani, Y.B.; Moore, D.J.; Barrett, M.A.; Flanagan, M.; Kim-Shapiro, D.B.; Basu, S.; Muller, M.D.; Proctor, D.N. Effects of acute dietary nitrate supplementation on aortic blood pressures and pulse wave characteristics in post-menopausal women. Nitric Oxide 2019, 85, 10–16. [Google Scholar] [CrossRef]
- Velmurugan, S.; Kapil, V.; Ghosh, S.M.; Davies, S.; McKnight, A.; Aboud, Z.; Khambata, R.S.; Webb, A.J.; Poole, A.; Ahluwalia, A. Antiplatelet effects of dietary nitrate in healthy volunteers: Involvement of cGMP and influence of sex. Free Radic. Biol. Med. 2013, 65, 1521–1532. [Google Scholar] [CrossRef] [PubMed]
- Craig, J.C.; Broxterman, R.M.; Smith, J.R.; Allen, J.D.; Barstow, T.J. Effect of dietry nitrate supplementation on conduit artery blood flow, muscle oxygenation, and metabolic rate during handgrip exercise. J. Appl. Physiol. 2018, 125, 254–262. [Google Scholar] [CrossRef] [PubMed]
- de Vries, C.J.; DeLorey, D.S. Effect of acute dietary nitrate supplementation on sympathetic vasoconstriction at rest and during exercise. J. Appl. Physiol. 2019, 127, 81–88. [Google Scholar] [CrossRef]
- Notay, K.; Incognito, A.V.; Millar, P.J. Acute beetroot juice supplementation on systemaitic nerve activity: A randomized, double-blind, placebo-controlled proof-of-concept study. Am. J. Physiol. 2017, 313, H59–H65. [Google Scholar]
- Perez, J.M.; Dobson, J.L.; Ryan, G.A.; Riggs, A.J. The effects of beetroot juice on VO2max and blood pressure during submaximal exercise. Intern. J. Exerc. Sci. 2019, 12, 332–342. [Google Scholar]
- Ryan, L.; Prescott, S.L. Stability of the antioxidant capacity of twenty-five commercially available fruit juices subjected to an in vitro digestion. Int. J. Food Sci. Technol. 2010, 45, 1191–1197. [Google Scholar] [CrossRef]
- Joris, P.J.; Mensink, R.P. Beetroot juice improves in overweight and slightly obese men postprandial endothelial function after consumption of a mixed meal. Aterosclerosis 2013, 231, 78–83. [Google Scholar] [CrossRef]
- Lidder, S.; Webb, A.J. Vascular effects of dietary nitrate (as found in green leafy vegetables and beetroot) via the nitrate-nitrite-nitric oxide pathway. Br. J. Clin. Pharmacol. 2013, 75, 677–696. [Google Scholar] [CrossRef]
- Bakker, E.; Engan, H.; Patrician, A.; Schagatay, E.; Karlsen, T.; Wisloff, U.; Gaustad, S.E. Acute dietary nitrate supplementation improves arterial endothelial function at high altitude: A double –blinded randomized controlled cross over study. Nitric Oxide 2015, 50, 58–64. [Google Scholar] [CrossRef]
- Keen, J.T.; Levitt, E.L.; Hodges, G.J.; Wong, B.J. Short-term dietary nitrate supplementation augments cutaneous vasodilatation and reduces mean arterial pressure in healthy humans. Microvasc. Res. 2015, 98, 48–53. [Google Scholar] [CrossRef]
- Lee, J.S.; Stebbins, C.L.; Jung, E.; Nho, H.; Kim, J.K.; Chang, M.J.; Choi, H.M. Effects of chronic dietary nitrate supplementation on the hemodynamic response to dynamic exercise. Am. J. Physiol. 2015, 309, R459–R466. [Google Scholar] [CrossRef]
- Levitt, E.L.; Keen, J.T.; Wong, B.J. Augmented reflex cutaneous vasodilatation following short-term dietary nitrate supplementation in humans. Exp. Physiol. 2015, 100, 708–718. [Google Scholar] [CrossRef]
- Lara, J.; Ashor, A.W.; Oggioni, C.; Ahluwalia, A.; Mathers, J.C. Effects of inorganic nitrate and beetroot supplementation on endothelial function: A systemiatic review and meta-analysis. Eur. J. Nutr. 2016, 55, 451–459. [Google Scholar] [CrossRef]
- Kenjale, A.A.; Ham, K.L.; Stabler, T.; Robbins, J.L.; Johnson, J.L.; Vanbruggen, M.; Privette, G.; Yim, E.; Kraus, W.E.; Allen, J.D. Dietary nitrate supplementation enhances exercise performance in peripheral arterial disease. J. Appl. Physiol. 2011, 110, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- McKnight, G.M.; Duncan, C.W.; Leifert, C.; Golden, M.H. Dietary nitrate in man: Frein or foe? Br. J. Nutr. 1999, 81, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Richardson, G.; Hicks, S.L.; O’Byrne, S.; Frost, M.T.; Moore, K.; Benjamin, N.; McKnight, G.M. The ingestion of inorganic nitrate increases gastric S-nitrosothiol levels and inhibits platelet function in humans. Nitric Oxide 2002, 7, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Morgado, M.; de Oliveira, G.V.; Vasconcellos, J.; Monteiro, M.L.; Conte-Junior, C.; Pierucci, A.P.T.R.; Alvares, T.S. Development of a beetroot-based nutritional gel containing high content of bioaccessible dietary nitrate and antioxidants. Int. Food Sci. Nutr. 2016, 67, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Houshialsadat, Z.; Gaeini, Z.; Bahadoran, Z.; Azizi, F. Functional properties of beetroot (Beta vulgaris) in management of cardio-metabolic diseases. Nutr. Metab. 2020, 17, 3. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.L.; Moreau, R. Functionl properties of spinach (Spinacia oleracea L.) phytochemicals and bioactives. Food Funct. 2016, 7, 3337–3353. [Google Scholar] [CrossRef]
- Panda, V.; Bhandare, N.; Mistry, K.; Sudhamani, S.; Dande, P. Cardioprotective potential of Spinacia oleracea (Spinach) against isoproterenol-induced myocardial infraction in rats. Arch. Physiol. Biochem. 2022, 128, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Arru, L.; Mussi, F.; Forti, L.; Buschini, A. Biological effect of different spinach extracts in comparison with the individual components of the phytocomplex. Foods 2021, 10, 382. [Google Scholar] [CrossRef] [PubMed]
- Jovanovski, E.; Bosco, L.; Khan, K.; Au-Yeung, F.; Ho, H.; Zurbau, A.; Jenkins, A.L.; Vuksan, V. Effect of spinach, a high dietary nitrate source, on arterial stiffness and related homodynamic measures: A randomized, controlled trial in helthy adults. Clin. Nutr. Res. 2015, 4, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.; Morrison, S.; Hedges, L.; Kerkhofs, N.; Lister, C. Phenolics contribute significantly to higher antioxidant activity of red lettuce compared to green lettuce. In Proceedings of the XXII International Conference on Polyphenols, Helsinki, Finland, 25–28 August 2004; pp. 273–274. [Google Scholar]
- Rolnik, A.; Soluch, A.; Kowalska, I.; Olas, B. Antioxidant and hemostatic properties of preparations from Asteraceae family and their chemical composition—Comparative studies. Biomed. Pharmacol. 2021, 142, 111982. [Google Scholar] [CrossRef] [PubMed]
- Rolnik, A.; Stochmal, A.; Olas, B. The in vitro anti-platelet activities of plant extracts from the Asterceae family. Biomed. Pharmacol. 2022, 149, 112809. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Gil, M.I.; Yang, Q.; Tomas-Barberan, F.A. Bioactive compounds inlettuce: Highlighting the benefits to human health and impacts of preharvest and postharvest practices. Comp. Rev. Food Sci. Food Saf. 2022, 21, 4–45. [Google Scholar] [CrossRef] [PubMed]
- Nicolle, C.; Cardinault, N.; Gueux, E.; Jaffrelo, L.; Rock, E.; Mazur, A.; Amoroux, P.; Remesy, C. Health effect of vegetable-based diet: Lettuce consumption improves cholesterol metabolism and antioxidant status in the rat. Clin. Nutr. 2004, 23, 605–614. [Google Scholar] [CrossRef]
- Abdalla, M.A.; Wenzel-Storjohann, A.; Sulieman, S.; Tasdemir, D.; Muhling, K.H. Comparative metabolite profile, biological activity and overall quality of three lettuce (Lactuca sativa L.; Asteracea) cultivars in response to sulfur nutrition. Pharmaceutics 2021, 13, 713. [Google Scholar] [CrossRef]
- Kammoun, I.; Salah, H.B.; Saad, H.B.; Cherif, B.; Droguet, M.; Magne, C.; Kallel, C.; Boudawara, O.; Hakim, A.; Gharsallah, N.; et al. Hypolipidemic and cardioprotective effects of Ulva lactuca ethanolic extract in hypochoelsterolemic mice. Arch. Physiol. Biochem. 2018, 124, 313–325. [Google Scholar] [CrossRef]
- Kooti, W.; Daraei, N. A review of the antioxidant activity of celery (Apium graveolens L). Top. Rev. Artic. 2017, 22, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Bagar, S.P.; Sharma, N.; Sanwal, N.; Lorenzo, J.M.; Sahu, J.K. Bioactive potential of beetroot (Beta vulgaris). Food Res. Int. 2022, 158, 111556. [Google Scholar]
| Beetroot Product | NO3− (mmol) | NO2− (mmol) |
|---|---|---|
| Cereal bar | 14.0 ± 0.05 | 0.2 ± 0.01 |
| Gel | 6.3 ± 0.01 | 0.11 ± 0.02 |
| Chips | 6.9 ± 0.02 | 0.13 ± 0.02 |
| Fresh juice | 4.1 ± 0.01 | 0.1 ± 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olas, B. The Cardioprotective Role of Nitrate-Rich Vegetables. Foods 2024, 13, 691. https://doi.org/10.3390/foods13050691
Olas B. The Cardioprotective Role of Nitrate-Rich Vegetables. Foods. 2024; 13(5):691. https://doi.org/10.3390/foods13050691
Chicago/Turabian StyleOlas, Beata. 2024. "The Cardioprotective Role of Nitrate-Rich Vegetables" Foods 13, no. 5: 691. https://doi.org/10.3390/foods13050691
APA StyleOlas, B. (2024). The Cardioprotective Role of Nitrate-Rich Vegetables. Foods, 13(5), 691. https://doi.org/10.3390/foods13050691






