Leaching of Phytochemicals from Beans during Hydration, Kinetics, and Modeling
Abstract
1. Introduction
2. Bean Hydration Process
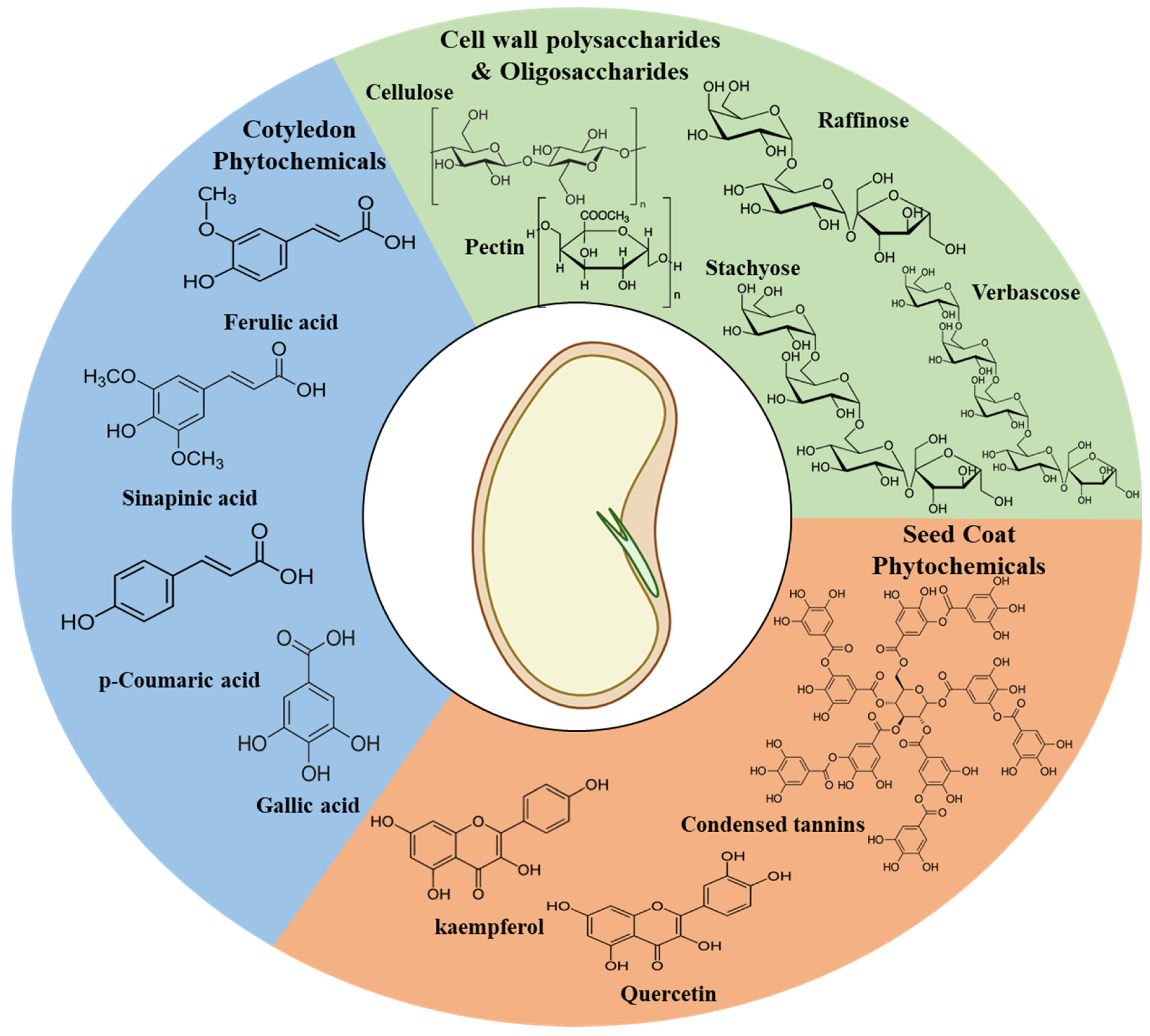
3. Hypothesized Mechanism of Phytochemical Leach out during Hydration
| Common Phytochemicals | References |
|---|---|
| Syringic acid | [33] |
| p-Coumaric acid | |
| Ferulic acid | |
| Rutin | |
| Quercetin- derivatives | |
| Luteolin | |
| - (-) catechin | |
| kaempferol derivatives | [35] |
| Gallic acid | |
| delphinidin | |
| cyanidin | |
| petunidin | |
| pelargonidin | |
| malvidin aglycones | |
| Raffinose | [9,10,11] |
| Stachyose | |
| Verbascose | |
| Cellulose | |
| Hemi cellulose | |
| Pectin | |
| Lignin | |
| Tannins |
4. Modeling Approaches in Phytochemical Leach Out
4.1. Mechanistic Modeling
4.2. Major Assumptions in Existing Studies
4.3. Governing Equations
4.3.1. Washing Stage
4.3.2. Diffusion Stage
5. Phenomenological Models
5.1. Power Law Extraction Model
5.2. Peleg’s Extraction Model
5.3. Weibull Extraction Model
5.4. Two-Site Kinetic Model
6. Future Scope
7. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chigwedere, C.M.; Olaoye, T.F.; Kyomugasho, C.; Jamsazzadeh Kermani, Z.; Pallares Pallares, A.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Mechanistic insight into softening of Canadian wonder common beans (Phaseolus vulgaris) during cooking. Food Res. Int. 2018, 106, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Giusti, F.; Capuano, E.; Sagratini, G.; Pellegrini, N. A comprehensive investigation of the behaviour of phenolic compounds in legumes during domestic cooking and in vitro digestion. Food Chem. 2019, 285, 458–467. [Google Scholar] [CrossRef]
- Aiking, H.; de Boer, J. The next protein transition. Trends Food Sci. Technol. 2020, 105, 515–522. [Google Scholar] [CrossRef]
- Augustin, M.A.; Cole, M.B. Towards a sustainable food system by design using faba bean protein as an example. Trends Food Sci. Technol. 2022, 125, 1–11. [Google Scholar] [CrossRef]
- Köpke, U.; Nemecek, T. Ecological services of faba bean. Field Crop Res. 2010, 115, 217–233. [Google Scholar] [CrossRef]
- Mesfin, S.; Gebresamuel, G.; Haile, M.; Zenebe, A.; Desta, G. Mineral Fertilizer Demand for Optimum Biological Nitrogen Fixation and Yield Potentials of Legumes in Northern Ethiopia. Sustainability 2020, 12, 6449. [Google Scholar] [CrossRef]
- Borges, C.W.C.; Jorge, L.M.d.M.; Jorge, R.M.M. Kinetic modeling and thermodynamic properties of soybean cultivar (BRS257) during hydration process. J. Food Process Eng. 2017, 40, e12579. [Google Scholar] [CrossRef]
- Ghafoor, M.; Misra, N.N.; Mahadevan, K.; Tiwari, B.K. Ultrasound assisted hydration of navy beans (Phaseolus vulgaris). Ultrason. Sonochem 2014, 21, 409–414. [Google Scholar] [CrossRef]
- Kumar, G.; Le, D.T.; Durco, J.; Cianciosi, S.; Devkota, L.; Dhital, S. Innovations in legume processing: Ultrasound-based strategies for enhanced legume hydration and processing. Trends Food Sci. Technol. 2023, 139, 104122. [Google Scholar] [CrossRef]
- Devkota, L.; He, L.; Bittencourt, C.; Midgley, J.; Haritos, V.S. Thermal and pulsed electric field (PEF) assisted hydration of common beans. LWT—Food Sci. Technol. 2022, 158, 113163. [Google Scholar] [CrossRef]
- Perera, D.; Kumar, G.; Devkota, L.; Dhital, S. Bioactive Nutrient Retention during Thermal-Assisted Hydration of Lupins. Foods 2023, 12, 709. [Google Scholar] [CrossRef] [PubMed]
- Buhl, T.F.; Christensen, C.H.; Hammershoj, M. Aquafaba as an egg white substitute in food foams and emulsions: Protein composition and functional behavior. Food Hydrocoll. 2019, 96, 354–364. [Google Scholar] [CrossRef]
- Matella, N.J.; Dolan, K.D.; Stoeckle, A.W.; Bennink, M.R.; Lee, Y.S.; Uebersax, M.A. Use of Hydration, Germination, and α-Galactosidase Treatments to Reduce Oligosaccharides in Dry Beans. J. Food Sci. 2006, 70, C203–C207. [Google Scholar] [CrossRef]
- Frisullo, P.; Laverse, J.; Barnabà, M.; Navarini, L.; Del Nobile, M.A. Coffee beans microstructural changes induced by cultivation processing: An X-ray microtomographic investigation. J. Food Eng. 2012, 109, 175–181. [Google Scholar] [CrossRef]
- Gargiulo, L.; Sorrentino, G.; Mele, G. 3D imaging of bean seeds: Correlations between hilum region structures and hydration kinetics. Food Res. Int. 2020, 134, 109211. [Google Scholar] [CrossRef]
- Kikuchi, K.; Koizumi, M.; Ishida, N.; Kano, H. Water uptake by dry beans observed by micro-magnetic resonance imaging. Ann. Bot. 2006, 98, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Miano, A.C.; García, J.A.; Augusto, P.E.D. Correlation between morphology, hydration kinetics and mathematical models on Andean lupin (Lupinus mutabilis Sweet) grains. LWT—Food Sci. Technol. 2015, 61, 290–298. [Google Scholar] [CrossRef]
- Smykal, P.; Vernoud, V.; Blair, M.W.; Soukup, A.; Thompson, R.D. The role of the testa during development and in establishment of dormancy of the legume seed. Front. Plant Sci. 2014, 5, 351. [Google Scholar] [CrossRef]
- Mikac, U.; Sepe, A.; Sersa, I. MR microscopy for noninvasive detection of water distribution during soaking and cooking in the common bean. Magn. Reson. Imaging 2015, 33, 336–345. [Google Scholar] [CrossRef]
- Garnczarska, M.; Zalewski, T.; Kempka, M. Changes in water status and water distribution in maturing lupin seeds studied by MR imaging and NMR spectroscopy. J. Expimental Bot. 2007, 58, 3961–3969. [Google Scholar] [CrossRef]
- Nasar-Abbas, S.M.; Plummer, J.A.; Siddique, K.H.M.; White, P.; Harris, D.; Dods, K. Cooking quality of faba bean after storage at high temperature and the role of lignins and other phenolics in bean hardening. LWT—Food Sci. Technol. 2008, 41, 1260–1267. [Google Scholar] [CrossRef]
- Plumb, G.W.; De Pascual-Teresa, S.; Santos-Buelga, C.; Cheynier, V.; Williamson, G. Antioxidant properties of catechins and proanthocyanidins: Effect of polymerisation, galloylation and glycosylation. Free Radic. Res. 1998, 29, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Li, X.; Qi, X.; Ren, Y. Pathway analysis of the biodegradation of lignin by Brevibacillus thermoruber. Bioresour. Technol. 2021, 341, 125875. [Google Scholar] [CrossRef] [PubMed]
- Siah, S.; Wood, J.A.; Agboola, S.; Konczak, I.; Blanchard, C.L. Effects of soaking, boiling and autoclaving on the phenolic contents and antioxidant activities of faba beans (Vicia faba L.) differing in seed coat colours. Food Chem. 2014, 142, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Zaupa, M.; Calani, L.; Del Rio, D.; Brighenti, F.; Pellegrini, N. Characterization of total antioxidant capacity and (poly)phenolic compounds of differently pigmented rice varieties and their changes during domestic cooking. Food Chem. 2015, 187, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Villaluenga, C.; Frías, J.; Vidal-Valverde, C. Raffinose family oligosaccharides and sucrose contents in 13 Spanish lupin cultivars. Food Chem. 2005, 91, 645–649. [Google Scholar] [CrossRef]
- Chen, Y.; Chang, S.K.C.; Zhang, Y.; Hsu, C.Y.; Nannapaneni, R. Gut microbiota and short chain fatty acid composition as affected by legume type and processing methods as assessed by simulated in vitro digestion assays. Food Chem. 2020, 312, 126040. [Google Scholar] [CrossRef] [PubMed]
- Ashaolu, T.J.; Ashaolu, J.O.; Adeyeye, S.A.O. Fermentation of prebiotics by human colonic microbiota in vitro and short-chain fatty acids production: A critical review. J. Appl. Microbiol. 2021, 130, 677–687. [Google Scholar] [CrossRef]
- Matella, N.J.; Mishra, D.K.; Dolan, K.D. Hydration, Blanching and Thermal Processing of Dry Beans. In Dry Beans and Pulses Production, Processing and Nutrition; Wiley: Hoboken, NJ, USA, 2012; pp. 129–154. [Google Scholar]
- Wei, X.; Wanasundara, J.P.D.; Shand, P. Short-term germination of faba bean (Vicia faba L.) and the effect on selected chemical constituents. Appl. Food Res. 2022, 2, 100030. [Google Scholar] [CrossRef]
- Díaz, A.M.; Caldas, G.V.; Blair, M.W. Concentrations of condensed tannins and anthocyanins in common bean seed coats. Food Res. Int. 2010, 43, 595–601. [Google Scholar] [CrossRef]
- Ranilla, L.G.; Genovese, M.I.; Lajolo, F.M. Polyphenols and antioxidant capacity of seed coat and cotyledon from Brazilian and Peruvian bean cultivars (Phaseolus vulgaris L.). J. Agric. Food Chem. 2007, 55, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.B.; Neupane, P.; Bhattarai, S.P.; Trotter, T.; Naiker, M. Phenolic profiles and acetylcholinesterase inhibitory activity in Australian adzuki bean. Food Chem. Adv. 2023, 3, 100361. [Google Scholar] [CrossRef]
- Troszyńska, A.; Ciska, E. Phenolic compounds of seed coats of white and coloured varieties of pea (Pisum sativum L.) and their total antioxidant activity. Czech J. Food Sci. 2002, 20, 15–22. [Google Scholar] [CrossRef]
- Rodriguez Madrera, R.; Campa Negrillo, A.; Suarez Valles, B.; Ferreira Fernandez, J.J. Characterization of extractable phenolic profile of common bean seeds (Phaseolus vulgaris L.) in a Spanish diversity panel. Food Res. Int. 2020, 138, 109713. [Google Scholar] [CrossRef] [PubMed]
- Sedlakova, V.; Zeljkovic, S.C.; Stefelova, N.; Smykal, P.; Hanacek, P. Phenylpropanoid Content of Chickpea Seed Coats in Relation to Seed Dormancy. Plants 2023, 12, 2687. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.H.; Joardder, M.U.H.; Kumar, C.; Karim, M.A. Multiphase porous media modelling: A novel approach to predicting food processing performance. Crit. Rev. Food Sci. Nutr. 2018, 58, 528–546. [Google Scholar] [CrossRef] [PubMed]
- Purlis, E. Modelling convective drying of foods: A multiphase porous media model considering heat of sorption. J. Food Eng. 2019, 263, 132–146. [Google Scholar] [CrossRef]
- Mkaouar, S.; Gelicus, A.; Bahloul, N.; Allaf, K.; Kechaou, N. Kinetic study of polyphenols extraction from olive (Olea europaea L.) leaves using instant controlled pressure drop texturing. Sep. Purif. Technol. 2016, 161, 165–171. [Google Scholar] [CrossRef]
- Datta, A.K. Porous media approaches to studying simultaneous heat and mass transfer in food processes. I: Problem formulations. J. Food Eng. 2007, 80, 80–95. [Google Scholar] [CrossRef]
- Datta, A.K. Porous media approaches to studying simultaneous heat and mass transfer in food processes. II: Property data and representative results. J. Food Eng. 2007, 80, 96–110. [Google Scholar] [CrossRef]
- Bello, I.; Adeniyi, A.; Mukaila, T.; Hammed, A. Developing and modelling of sustainable protein extraction using ammonium hydroxide-A recoverable and reusable solvent. Food Bioprod. Process. 2023, 140, 16–28. [Google Scholar] [CrossRef]
- Simeonov, E.; Yaneva, Z.; Chilev, C. Kinetics of green solid-liquid extraction of useful compounds from plant materials: Kinetics coefficients and modeling. Green. Process Synth. 2018, 7, 68–73. [Google Scholar] [CrossRef]
- Crank, J. The Mathematics of Diffusion; Oxford University Press: Oxford, UK, 1979. [Google Scholar]
- Alonso-Riano, P.; Sanz Diez, M.T.; Blanco, B.; Beltran, S.; Trigueros, E.; Benito-Roman, O. Water Ultrasound-Assisted Extraction of Polyphenol Compounds from Brewer’s Spent Grain: Kinetic Study, Extract Characterization, and Concentration. Antioxidants 2020, 9, 265. [Google Scholar] [CrossRef] [PubMed]
- Jokić, S.; Velić, D.; Bilić, M.; Bucić-Kojić, A.; Planinić, M.; Tomas, S. Modelling of solid-liquid extraction process of total polyphenols from soybeans. Czech J. Food Sci. 2010, 28, 206–212. [Google Scholar] [CrossRef]
- Patil, S.S.; Deshannavar, U.B.; Ramasamy, M.; Emani, S.; Khalilpoor, N.; Issakhov, A.; Baghban, A. Study of Extraction Kinetics of Total Polyphenols from Curry Leaves. Int. J. Chem. Eng. 2021, 2021, 9988684. [Google Scholar] [CrossRef]
- Fazil, F.N.; Azzimi, N.S.; Yahaya, B.H.; Kamalaldin, N.A.; Zubairi, S.I. Kinetics Extraction Modelling and Antiproliferative Activity of Clinacanthus nutans Water Extract. Sci. World J. 2016, 2016, 7370536. [Google Scholar] [CrossRef] [PubMed]
- Sukor, N.F.; Selvam, V.P.; Jusoh, R.; Kamarudin, N.S.; Rahim, S.A. Intensified DES mediated ultrasound extraction of tannic acid from onion peel. J. Food Eng. 2021, 296, 110437. [Google Scholar] [CrossRef]
- Anbalagan, K.; Kumar, M.M.; Ilango, K.; Mohankumar, R.; Priya, R.L. Prelusive scale extraction of mangiferin from leaves: Assessing solvent competency, process optimization, kinetic study and diffusion modelling. Ind. Crop Prod. 2019, 140, 111703. [Google Scholar] [CrossRef]
- Lin, C.F.; Xia, G.B.; Liu, S.B. Modeling and comparison of extraction kinetics of 8 catechins, gallic acid and caffeine from representative white teas. LWT—Food Sci. Technol. 2017, 83, 1–9. [Google Scholar] [CrossRef]
- Kashaninejad, M.; Sanz, M.T.; Blanco, B.; Beltrán, S.; Niknam, S.M. Freeze dried extract from olive leaves: Valorisation, extraction kinetics and extract characterization. Food Bioprod. Process. 2020, 124, 196–207. [Google Scholar] [CrossRef]
- Barrientos-Lezcano, J.C.; Gallo-Machado, J.; Marin-Palacio, L.D.; Builes, S. Extraction kinetics and physicochemical characteristics of Colombian propolis. J. Food Process Eng. 2023, 46, 14272. [Google Scholar] [CrossRef]
- Benito-Roman, O.; Blanco, B.; Sanz, M.T.; Beltran, S. Subcritical Water Extraction of Phenolic Compounds from Onion Skin Wastes (Allium cepa cv. Horcal): Effect of Temperature and Solvent Properties. Antioxidants 2020, 9, 1233. [Google Scholar] [CrossRef] [PubMed]
- Duba, K.S.; Casazza, A.A.; Mohamed, H.B.; Perego, P.; Fiori, L. Extraction of polyphenols from grape skins and defatted grape seeds using subcritical water: Experiments and modeling. Food Bioprod. Process. 2015, 94, 29–38. [Google Scholar] [CrossRef]
- Natolino, A.; Da Porto, C. Kinetic models for conventional and ultrasound assistant extraction of polyphenols from defatted fresh and distilled grape marc and its main components skins and seeds. Chem. Eng. Res. Des. 2020, 156, 1–12. [Google Scholar] [CrossRef]
- Islam, M.N.; Jo, Y.-T.; Jung, S.-K.; Park, J.-H. Thermodynamic and kinetic study for subcritical water extraction of PAHs. J. Ind. Eng. Chem. 2013, 19, 129–136. [Google Scholar] [CrossRef]
- Sovová, H. Rate of the vegetable oil extraction with supercritical CO2—I. Modelling of extraction curves. Chem. Eng. Sci. 1994, 49, 409–414. [Google Scholar] [CrossRef]
- Peng, X.J.; Liu, N.; Wang, M.X.; Liang, B.; Feng, C.T.; Zhang, R.S.; Wang, X.F.; Hu, X.K.; Gu, H.Y.; Xing, D.M. Recent advances of kinetic model in the separation of essential oils by microwave-assisted hydrodistillation. Ind. Crop Prod. 2022, 187, 115418. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, G.; Perera, D.; Sudheer, K.P.; Zhang, P.; Dhital, S. Leaching of Phytochemicals from Beans during Hydration, Kinetics, and Modeling. Foods 2024, 13, 354. https://doi.org/10.3390/foods13020354
Kumar G, Perera D, Sudheer KP, Zhang P, Dhital S. Leaching of Phytochemicals from Beans during Hydration, Kinetics, and Modeling. Foods. 2024; 13(2):354. https://doi.org/10.3390/foods13020354
Chicago/Turabian StyleKumar, Gaurav, Dilini Perera, Kundukulangara Pulissery Sudheer, Pangzhen Zhang, and Sushil Dhital. 2024. "Leaching of Phytochemicals from Beans during Hydration, Kinetics, and Modeling" Foods 13, no. 2: 354. https://doi.org/10.3390/foods13020354
APA StyleKumar, G., Perera, D., Sudheer, K. P., Zhang, P., & Dhital, S. (2024). Leaching of Phytochemicals from Beans during Hydration, Kinetics, and Modeling. Foods, 13(2), 354. https://doi.org/10.3390/foods13020354








