Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Soybean Whey, Desalted Soybean Whey and Concentrated Soybean Whey
2.3. Preparation of Soybean Whey Yogurt
2.4. pH, Titratable Acidity (TA), Buffering Capacity and Viable Cell Counts of LAB
2.5. Composition Analysis
2.5.1. Total Solids, Protein and Sugar
2.5.2. SDS-PAGE
2.6. Texture Properties
2.7. Microstructures
2.8. Activity of Urease and Trypsin Inhibitors
2.8.1. Urease Activity
2.8.2. Trypsin Inhibitory Activity (TIA)
2.9. Volatile Compounds
2.10. Rheological Analysis
2.11. Sensory Evaluation
2.12. Statistical Analysis
3. Results and Discussion
3.1. Basic Composition of Soybean Whey after Desalination and Concentration
3.2. Fermentation Characteristics of Soybean Whey after Desalination and Concentration
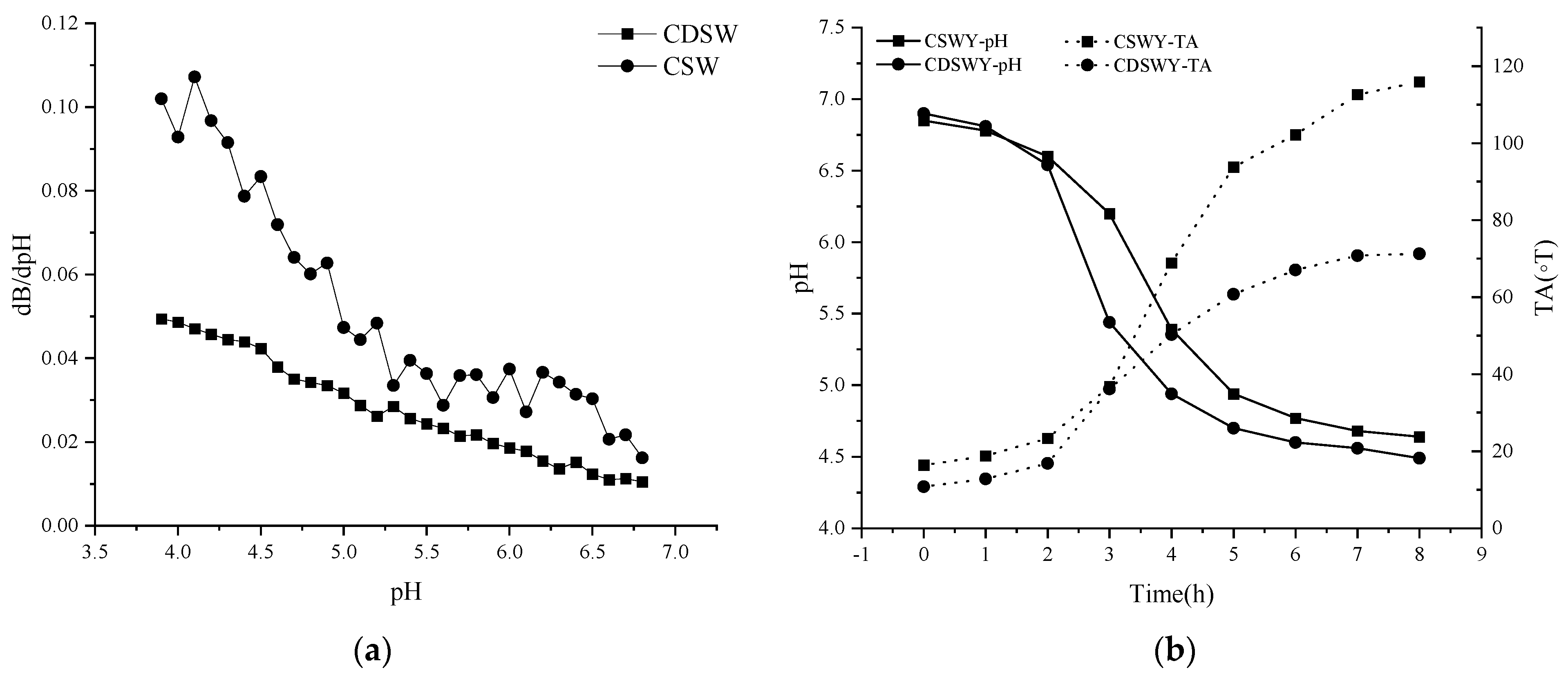
3.2.1. Buffering Capacity
3.2.2. Changes in pH and TA during Fermentation
3.3. Protein Compositions of Soybean Whey Proteins after Desalination, Concentration and and Fermentation
3.4. Textural Property of Soybean Whey Yogurt
3.5. Microstructure of Soybean Whey Yogurt
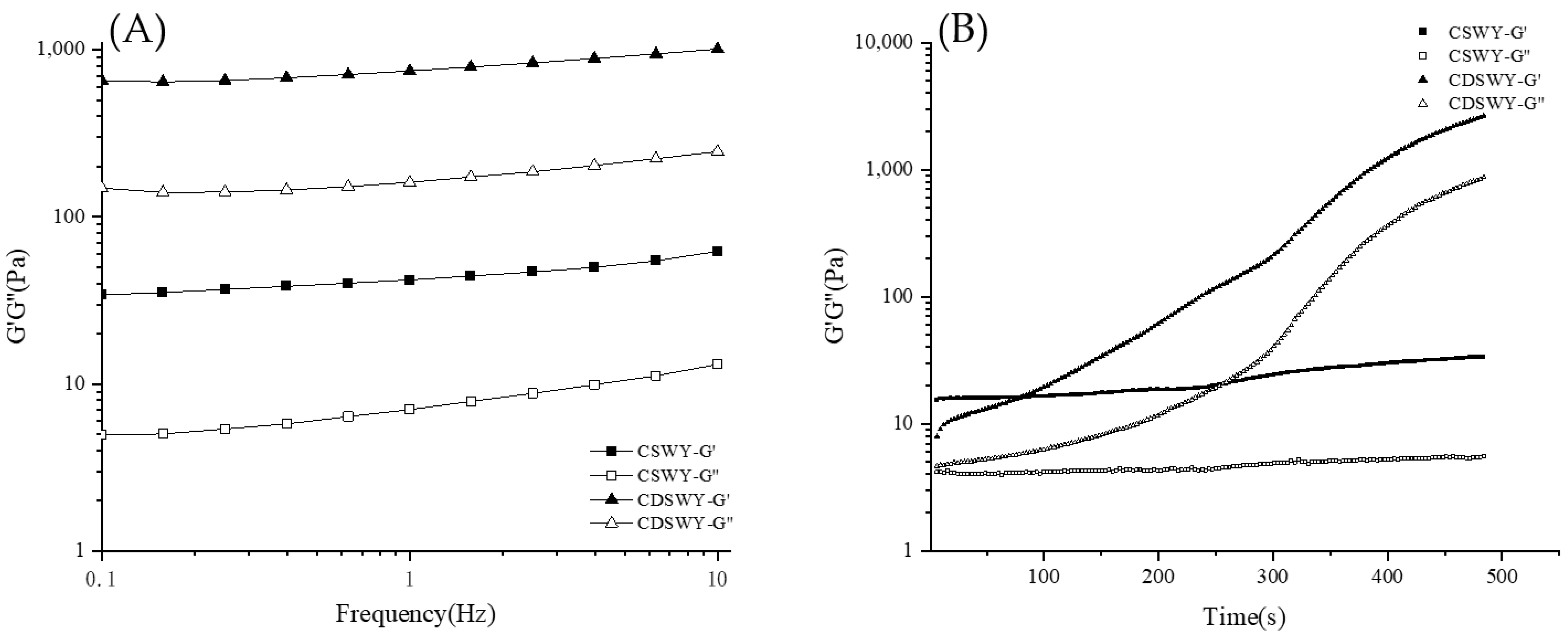
3.6. Gelation Kinetics and Viscoelastic Property of Soybean Whey Yogurt
3.7. Activities of Urease and Trypsin Inhibitors
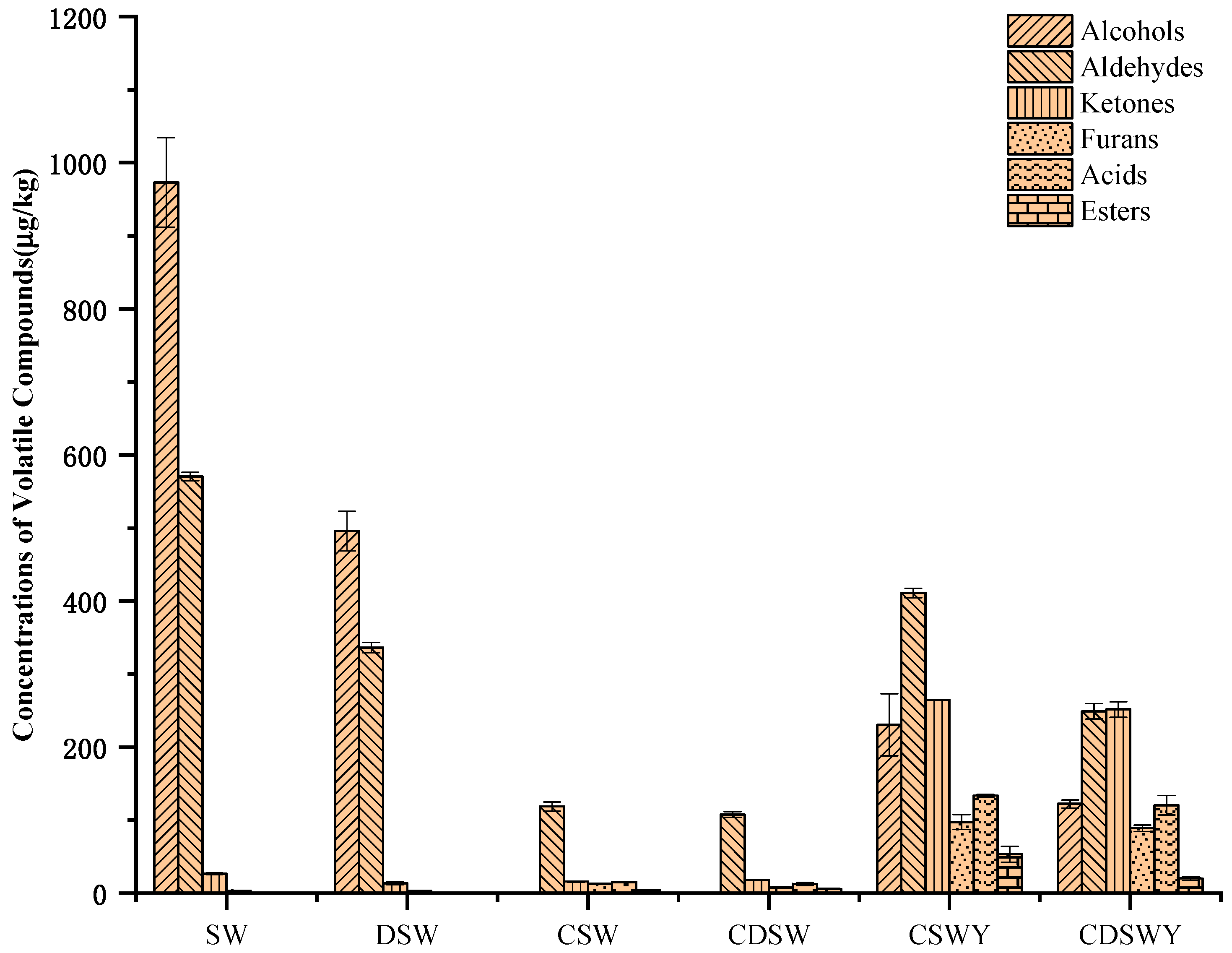
3.8. Volatile Flavor Compounds of Soybean Whey after Desalination, Concentration and Fermentation
| (a) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Compounds | Odor Description | Threshold (μg/kg in Water) | SW | OAVs | Relative Contribution Rate (%) | DSW | OAVs | Relative Contribution Rate (%) |
| (μg/kg | (μg/kg | |||||||
| Sample) | Sample) | |||||||
| 1-Octen-3-one | Mushroom, earthy, fruity | 0.007 | 5.92 ± 0.43 | 845.40 ± 62.02 | 60.41 ± 3.29 | 6.87 ± 0.00 | 981.89 ± 0.46 | 77.79 ± 0.74 |
| 1-Octen-3-ol | Mushroom | 1.5 | 570.29 ± 66.16 | 380.19 ± 44.10 | 27.22 ± 3.67 | 301.80 ± 20.17 | 201.20 ± 13.45 | 15.94 ± 0.92 |
| Hexanal | Green, grass | 4.5 | 474.82 ± 5.77 | 105.52 ± 1.28 | 7.55 ± 0.23 | 284.87 ± 14.15 | 63.31 ± 3.15 | 5.02 ± 0.29 |
| (E,E)-2,4-Nonadienal | Fat, fried | 0.1 | 2.99 ± 1.17 | 29.92 ± 11.74 | 2.13 ± 0.80 | - | - | - |
| (E)-2-Nonenal | Grease, tallow, grass | 0.19 | 4.28 ± 0.68 | 22.52 ± 3.58 | 1.61 ± 0.29 | 1.73 ± 0.25 | 9.08 ± 1.30 | 0.72 ± 0.10 |
| Nonanal | Wax, citrus, fat, flowers | 1 | 5.25 ± 0.54 | 5.25 ± 0.54 | 0.38 ± 0.03 | 2.96 ± 0.08 | 2.96 ± 0.08 | 0.23 ± 0.01 |
| Heptanal | Grease, grass | 2.8 | 13.61 ± 1.94 | 4.86 ± 0.69 | 0.35 ± 0.04 | 7.60 ± 0.59 | 2.71 ± 0.21 | 0.21 ± 0.01 |
| (E)-2-Octenal | Cucumber, vegetable | 3 | 5.99 ± 1.07 | 2.00 ± 0.36 | 0.14 ± 0.02 | 3.39 ± 0.54 | 1.13 ± 0.18 | 0.09 ± 0.01 |
| Octanal | Lemon and fruit | 0.8 | 1.33 ± 0.07 | 1.66 ± 0.09 | 0.12 ± 0.00 | - | - | - |
| Pentanal | Fermentation, yogurt | 12 | 16.72 ± 0.15 | 1.39 ± 0.01 | 0.10 ± 0.00 | 9.04 ± 1.62 | <1 | - |
| (b) | ||||||||
| Compounds | Odor Description | Threshold (μg/kg in Water) | CSW (μg/kg Sample) | OAVs | Relative Contribution Rate (%) | CDSW (μg/kg Sample) | OAVs | Relative Contribution Rate (%) |
| (E)-2-Nonenal | Grease, tallow, grass | 0.19 | 5.15 ± 0.23 | 27.12 ± 0.59 | 27.31 ± 1.76 | 2.83 ± 0.24 | 14.89 ± 1.27 | 20.23 ± 0.71 |
| β-damascenone | Apples, roses, honey | 0.056 | 1.19 ± 0.11 | 21.32 ± 1.01 | 21.44 ± 1.59 | 0.44 ± 0.09 | 7.81 ± 1.62 | 11.62 ± 1.79 |
| Nonanal | Wax, citrus, fat, flowers | 1 | 18.95 ± 0.99 | 18.95 ± 0.49 | 19.06 ± 0.60 | 25.09 ± 0.64 | 25.09 ± 0.64 | 37.56 ± 2.98 |
| (E,E)-2,4-Nonadienal | Fat, fried | 0.1 | 0.77 ± 0.03 | 7.66 ± 0.15 | 7.71 ± 0.47 | 0.64 ± 0.09 | 6.41 ± 0.90 | 9.55 ± 0.82 |
| Hexanal | Green, grass | 4.5 | 27.28 ± 0.44 | 6.06 ± 0.05 | 6.10 ± 0.23 | 10.94 ± 0.80 | 2.43 ± 0.18 | 3.64 ± 0.46 |
| (E,E)-2,4-Decadienal | Fried, wax, fat | 0.07 | 0.34 ± 0.04 | 4.80 ± 0.32 | 4.83 ± 0.54 | - | - | - |
| 2,4-Decadienal | Fat | 0.3 | 1.02 ± 0.02 | 3.39 ± 0.04 | 3.41 ± 0.15 | 0.81 ± 0.01 | 2.69 ± 0.04 | 4.02 ± 0.12 |
| (E)-2-Octenal | Cucumber, vegetable | 3 | 7.88 ± 0.34 | 2.63 ± 0.06 | 2.64 ± 0.17 | 4.91 ± 0.28 | 1.64 ± 0.09 | 2.45 ± 0.27 |
| Octanal | Lemon and fruit | 0.8 | 1.91 ± 0.11 | 2.38 ± 0.07 | 2.40 ± 0.19 | 2.10 ± 0.24 | 2.62 ± 0.30 | 3.91 ± 0.24 |
| (E)-2-Heptenal | Soap, fat, almond | 13 | 25.62 ± 5.32 | 1.97 ± 0.20 | 1.98 ± 0.37 | 20.42 ± 4.83 | 1.57 ± 0.37 | 2.33 ± 0.43 |
| 2-Pentylfuran | Grass, beany, butter | 6 | 10.29 ± 0.08 | 1.72 ± 0.01 | 1.73 ± 0.05 | 5.50 ± 0.56 | <1 | - |
| Decanal | Earthy, mushroom | 3 | 4.15 ± 0.19 | 1.38 ± 0.03 | 1.39 ± 0.09 | 5.38 ± 0.06 | 1.79 ± 0.02 | 2.68 ± 0.11 |
| (c) | ||||||||
| Compounds | Odor Description | a Threshold (μg/kg in Water) | CSWY (μg/kg Samples) | OAVs | Relative Contribution Rate (%) | CDSWY (μg/kg Samples) | OAVs | Relative Contribution Rate (%) |
| 2,3-Butanedione | Butter | 0.059 | 70.05 ± 0.86 | 1187.34 ± 14.62 | 58.38 ± 0.68 | 78.92 ± 6.06 | 1337.64 ± 102.63 | 65.47 ± 1.11 |
| 2,4-Decadienal | Fat | 0.3 | 122.11 ± 19.50 | 407.04 ± 64.99 | 19.83 ± 2.92 | 112.93 ± 2.87 | 376.43 ± 9.58 | 18.45 ± 0.63 |
| (E,E)-2,4-Decadienal | Fried, wax, fat | 0.07 | 16.00 ± 0.49 | 228.55 ± 6.94 | 11.15 ± 0.48 | 10.47 ± 0.26 | 149.59 ± 3.67 | 7.34 ± 0.62 |
| β-damascenone | Apples, roses, honey | 0.056 | 3.40 ± 0.10 | 60.78 ± 1.84 | 2.96 ± 0.05 | 2.22 ± 0.16 | 39.73 ± 2.86 | 1.94 ± 0.02 |
| (E,E)-2,4-Nonadienal | Fat, fried | 0.1 | 4.04 ± 1.03 | 40.36 ± 10.32 | 1.97 ± 0.53 | 3.07 ± 0.21 | 30.73 ± 2.15 | 1.50 ± 0.02 |
| (E)-2-Decenaldehyde | Chicken oil, oranges | 0.3 | 7.14 ± 0.76 | 23.81 ± 2.54 | 1.16 ± 0.11 | 8.07 ± 0.26 | 26.89 ± 0.87 | 1.32 ± 0.04 |
| (E)-2-Nonenal | Grease, tallow, grass | 0.19 | 6.85 ± 1.12 | 36.04 ± 5.89 | 1.75 ± 0.22 | 5.56 ± 1.48 | 29.25 ± 7.78 | 1.42 ± 0.30 |
| 2-Pentylfuran | Grass, beany, butter | 6 | 91.05 ± 9.78 | 15.18 ± 1.63 | 0.74 ± 0.09 | 80.77 ± 2.72 | 13.46 ± 0.45 | 0.66 ± 0.02 |
| (E)-2-Heptenal | Soap, fat, almond | 13 | 175.26 ± 14.92 | 13.48 ± 1.15 | 0.66 ± 0.06 | 34.93 ± 0.48 | 2.69 ± 0.04 | 0.13 ± 0.01 |
| Nonanal | Wax, citrus, fat, flowers | 1 | 12.85 ± 7.28 | 12.85 ± 7.28 | 0.62 ± 0.35 | 15.91 ± 0.71 | 15.91 ± 0.71 | 0.78 ± 0.08 |
| (E)-2-Octenal | Cucumber, vegetable | 3 | 14.81 ± 1.37 | 4.94 ± 0.46 | 0.24 ± 0.03 | 14.81 ± 1.37 | 4.94 ± 0.46 | 0.20 ± 0.01 |
| Hexanal | Green, grass | 4.5 | 19.90 ± 0.26 | 4.42 ± 0.06 | 0.22 ± 0.01 | 18.08 ± 2.16 | 4.02 ± 0.48 | 0.20 ± 0.01 |
| Octanal | Lemon and fruit | 0.8 | 3.34 ± 0.71 | 4.18 ± 0.89 | 0.20 ± 0.04 | 2.78 ± 0.00 | 3.47 ± 0.00 | 0.17 ± 0.01 |
| 2,3-Pentanedione | Cream, butter | 20 | 76.48 ± 6.34 | 3.82 ± 0.32 | 0.19 ± 0.02 | 45.28 ± 1.56 | 2.26 ± 0.08 | 0.11 ± 0.00 |
| Ethyl hexanoate | Fruit | 5 | 16.92 ± 5.19 | 3.38 ± 1.04 | 0.17 ± 0.05 | 2.40 ± 0.80 | <1 | - |
| 3-Hydroxy-2-butanone | Cream, fat | 14 | 26.04 ± 1.60 | 1.86 ± 0.11 | 0.09 ± 0.01 | 55.99 ± 0.73 | 4.00 ± 0.05 | 0.20 ± 0.01 |
| 1-Octen-3-ol | Mushroom | 1.5 | 1.83 ± 0.12 | 1.22 ± 0.08 | 0.06 ± 0.00 | 1.17 ± 0.02 | <1 | - |
| Heptanal | Grease, grass | 2.8 | - | - | - | 3.02 ± 0.16 | 1.08 ± 0.06 | 0.05 ± 0.01 |
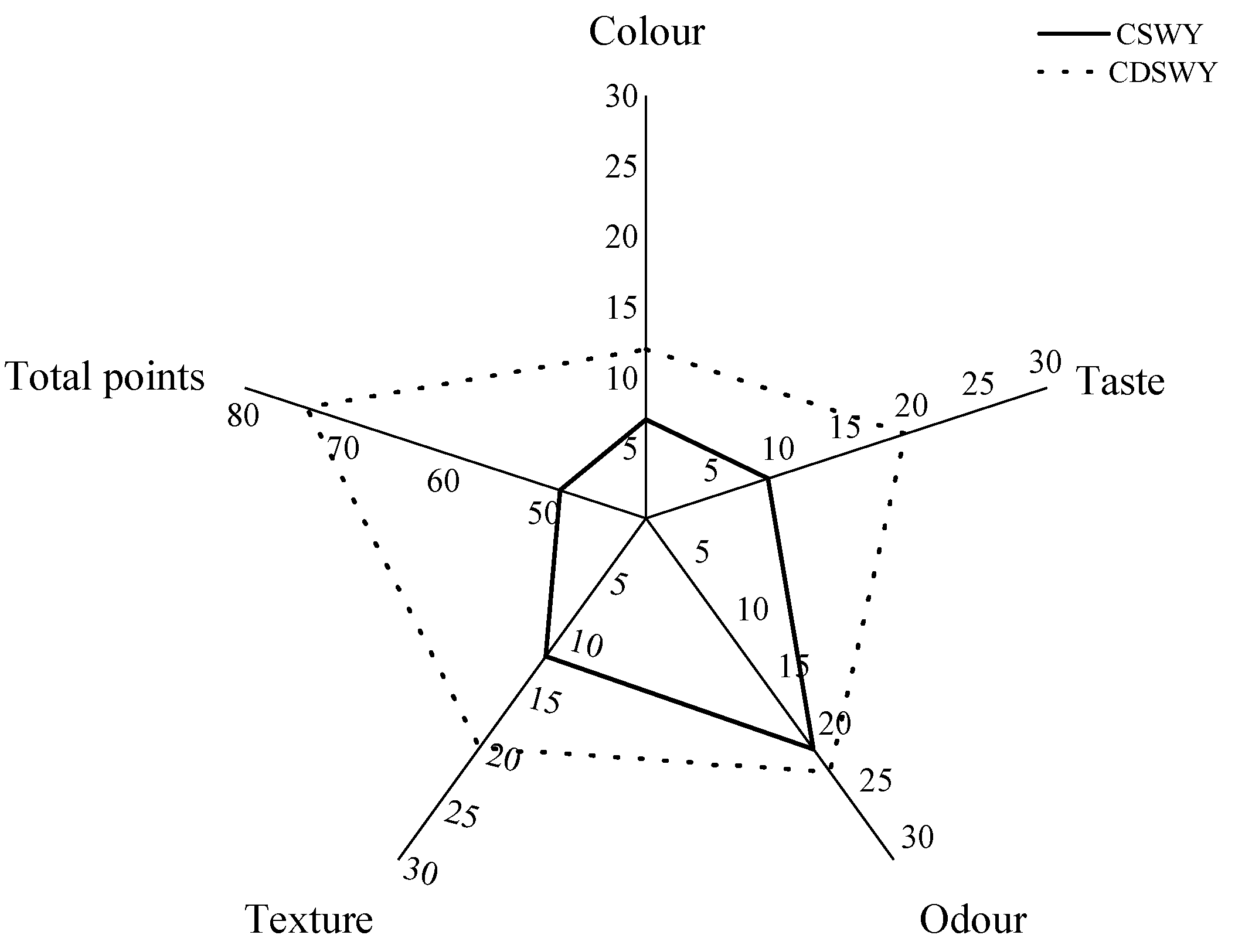
3.9. Sensory Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, W.; Zhang, H.X.; Wu, Z.L.; Wang, Y.J.; Wang, L.J. Recovery of Isoflavone Aglycones from Soy Whey Wastewater Using Foam Fractionation and Acidic Hydrolysis. J. Agric. Food Chem. 2013, 61, 7366–7372. [Google Scholar] [CrossRef] [PubMed]
- Sanjukta, S.; Rai, A.K. Production of Bioactive Peptides During Soybean Fermentation and Their Potential Health Benefits. Trends Food Sci. Technol. 2016, 50, 1–10. [Google Scholar] [CrossRef]
- Chua, J.Y.; Lu, Y.; Liu, S.Q. Evaluation of Five Commercial Non-Saccharomyces Yeasts in Fermentation of Soy (Tofu) Whey into an Alcoholic Beverage. Food Microbiol. 2018, 76, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.H.; Tang, S.J.; Azi, F.; Hu, W.X.; Dong, M.S. Use of Kombucha Consortium to Transform Soy Whey into a Novel Functional Beverage. J. Funct. Food. 2019, 52, 81–89. [Google Scholar] [CrossRef]
- Tu, C.H.; Azi, F.; Huang, J.; Xu, X.; Xing, G.L.; Dong, M.S. Quality and Metagenomic Evaluation of a Novel Functional Beverage Produced from Soy Whey Using Water Kefir Grains. LWT-Food Sci. Technol. 2019, 113, 108258. [Google Scholar] [CrossRef]
- Xu, X.Y.; Cui, H.T.; Xu, J.X.; Yuan, Z.H.; Liu, X.Q.; Fan, X.R.; Li, J.; Zhu, D.S.; Liu, H. Effects of Different Probiotic Fermentations on the Quality, Soy Isoflavone and Equol Content of Soy Protein Yogurt Made from Soy Whey and Soy Embryo Powder. LWT-Food Sci. Technol. 2022, 157, 113096. [Google Scholar] [CrossRef]
- Li, X.F.; Liu, X.; Hua, Y.F.; Chen, Y.M.; Kong, X.Z.; Zhang, C.M. Effects of Water Absorption of Soybean Seed on the Quality of Soymilk and the Release of Flavor Compounds. RSC Adv. 2019, 9, 2906–2918. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guo, S.T.; Liu, Z.H.; Chang, S.K.C. Off-Flavor Related Volatiles in Soymilk as Affected by Soybean Variety, Grinding, and Heat-Processing Methods. J. Agric. Food Chem. 2012, 60, 7457–7462. [Google Scholar] [CrossRef]
- Yuan, S.H.; Chang, S.K.C. Selected Odor Compounds in Cooked Soymilk as Affected by Soybean Materials and Direct Steam Injection. J. Food Sci. 2007, 72, 481–486. [Google Scholar] [CrossRef]
- Yi, C.P.; Li, Y.S.; Zhu, H.; Liu, Y.L.; Quan, K. Effect of Lactobacillus Plantarum Fermentation on the Volatile Flavors of Mung Beans. LWT-Food Sci. Technol. 2021, 146, 111434. [Google Scholar] [CrossRef]
- Ray, M.; Rousseau, D. Stabilization of Oil-in-Water Emulsions Using Mixtures of Denatured Soy Whey Proteins and Soluble Soybean Polysaccharides. Food Res. Int. 2013, 52, 298–307. [Google Scholar] [CrossRef]
- Holm, H.; Reseland, J.E.; Thorsen, L.I.; Flatmark, A.; Hanssen, L.E. Raw Soybeans Stimulate Human Pancreatic Proteinase Secretion. J. Nutr. 1992, 122, 1407–1416. [Google Scholar] [CrossRef]
- Rouhana, A.; AdlerNissen, J.; Cogan, U.; Frokiaer, H. Heat Inactivation Kinetics of Trypsin Inhibitors During High Temperature Short Time Processing of Soymilk. J. Food Sci. 1996, 61, 265–269. [Google Scholar] [CrossRef]
- Bueno, R.D.; Borges, L.L.; God, P.I.V.G.; Piovesan, N.D.P.; Teixeira, A.I.; Cruz, C.D.; Barros, E.G. Quantification of Anti-Nutritional Factors and Their Correlations with Protein and Oil in Soybeans. An. Acad. Bras. Cienc. 2018, 90, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Prasad, J.; McJarrow, P.; Gopal, P. Heat and Osmotic Stress Responses of Probiotic Lactobacillus Rhamnosus Hn001 (Dr20) in Relation to Viability after Drying. Appl. Environ. Microbiol. 2003, 69, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Zhang, Y.P.; Zhu, Y.; Mao, S.M.; Li, Y. Proteomic Analyses to Reveal the Protective Role of Glutathione in Resistance of Lactococcus Lactis to Osmotic Stress. Appl. Environ. Microbiol. 2010, 76, 3177–3186. [Google Scholar] [CrossRef] [PubMed]
- Li, X.F.; Hua, Y.F.; Chen, Y.M.; Kong, X.Z.; Zhang, C.M. The Selective Complex Behavior between Soybean Whey Proteins and Ι-Carrageenan and Isolation of the Major Proteins of the Soybean Whey. Food Hydrocoll. 2016, 56, 207–217. [Google Scholar] [CrossRef]
- Dashper, S.G.; Saion, B.N.; Stacey, M.A.; Manton, D.J.; Cochrane, N.J.; Stanton, D.P.; Yuan, Y.; Reynolds, E.C. Acidogenic Potential of Soy and Bovine Milk Beverages. J. Dent. 2012, 40, 736–741. [Google Scholar] [CrossRef]
- Rogatsky, E. Pandora Box of Bca Assay. Investigation of the Accuracy and Linearity of the Microplate Bicinchoninic Protein Assay: Analytical Challenges and Method Modifications to Minimize Systematic Errors. Anal. Biochem. 2021, 631, 114321. [Google Scholar] [CrossRef]
- Wang, X.L.; Kong, X.Z.; Zhang, C.M.; Hua, Y.F.; Chen, Y.M.; Li, X.F. Comparison of Physicochemical Properties and Volatile Flavor Compounds of Plant-Based Yoghurt and Dairy Yoghurt. Food Res. Int. 2023, 164, 112375. [Google Scholar] [CrossRef]
- Wu, C.; Hua, Y.F.; Chen, Y.M.; Kong, X.Z.; Zhang, C.M. Effect of Temperature, Ionic Strength and 11s Ratio on the Rheological Properties of Heat-Induced Soy Protein Gels in Relation to Network Proteins Content and Aggregates Size. Food Hydrocoll. 2017, 66, 389–395. [Google Scholar] [CrossRef]
- Kaur, H.; Mishra, H.N.; Kumar, P. Textural Properties of Mango Soy Fortified Probiotic Yoghurt: Optimisation of Inoculum Level of Yoghurt and Probiotic Culture. Int. J. Food Sci. Technol. 2009, 44, 415–424. [Google Scholar] [CrossRef]
- Leonard, R.H. Quantitative Range of Nessler’s Reaction with Ammonia. Clin. Chem. 1963, 12, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zhao, L.P.; Zhang, C.M.; Kong, X.Z.; Hua, Y.F.; Chen, Y.M. Comparative Effects of Ohmic, Induction Cooker, and Electric Stove Heating on Soymilk Trypsin Inhibitor Inactivation. J. Food Sci. 2015, 80, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Achouri, A.; Boye, J.I.; Zamani, Y. Identification of Volatile Compounds in Soymilk Using Solid-Phase Microextraction-Gas Chromatography. Food Chem. 2006, 99, 759–766. [Google Scholar] [CrossRef]
- Sedighi, M.; Behvand Usefi, M.M.; Ismail, A.F.; Ghasemi, M. Environmental Sustainability and Ions Removal through Electrodialysis Desalination: Operating Conditions and Process Parameters. Desalination 2023, 549, 116319. [Google Scholar] [CrossRef]
- Salaün, F.; Mietton, B.; Gaucheron, F. Buffering Capacity of Dairy Products. Int. Dairy J. 2005, 15, 95–109. [Google Scholar] [CrossRef]
- Parvarei, M.M.; Fazeli, M.R.; Mortazavian, A.M.; Nezhad, S.S.; Mortazavi, S.A.; Golabchifar, A.A.; Khorshidian, N. Comparative Effects of Probiotic and Paraprobiotic Addition on Microbiological, Biochemical and Physical Properties of Yogurt. Food Res. Int. 2021, 140, 110030. [Google Scholar] [CrossRef] [PubMed]
- Zotta, T.; Parente, E.; Ricciardi, A. Viability Staining and Detection of Metabolic Activity of Sourdough Lactic Acid Bacteria under Stress Conditions. World J. Microbiol. Biotechnol. 2009, 25, 1119–1124. [Google Scholar] [CrossRef]
- Fonseca, F.; Pénicaud, C.; Tymczyszyn, E.E.; Gómez-Zavaglia, A.; Passot, S. Factors Influencing the Membrane Fluidity and the Impact on Production of Lactic Acid Bacteria Starters. Appl. Microbiol. Biotechnol. 2019, 103, 6867–6883. [Google Scholar] [CrossRef]
- Nsogning, S.D.; Fischer, S.; Becker, T. Investigating on the Fermentation Behavior of Six Lactic Acid Bacteria Strains in Barley Malt Wort Reveals Limitation in Key Amino Acids and Buffer Capacity. Food Microbiol. 2018, 73, 245–253. [Google Scholar] [CrossRef]
- Utsumi, S.; Matsumura, Y.; Mori, T. Structure-Function Relationships of Soy Proteins. In Food Proteins and Their Applications; CRC Press: Boca Raton, FL, USA, 2017; pp. 257–292. [Google Scholar]
- Wang, C.H.; Damodaran, S. Thermal Gelation of Globular Proteins: Influence of Protein Conformation on Gel Strength. J. Agric. Food Chem. 1991, 39, 433–438. [Google Scholar] [CrossRef]
- Kella, N.K.; Kinsella, J.E. Structural Stability of Β-Lactoglobulin in the Presence of Kosmotropic Salts a Kinetic and Thermodynamic Study. Int. J. Pept. Protein Res. 1988, 32, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.L.; Li, Y.; Guo, S.T. Characteristics of Soy Protein Isolate Gel Induced by Glucono-Δ-Lactone: Effects of the Protein Concentration During Preheating. Food Hydrocoll. 2021, 113, 106525. [Google Scholar] [CrossRef]
- Nagano, T.; Hirotsuka, M.; Mori, H.; Kohyama, K.; Nishinari, K. Dynamic Viscoelastic Study on the Gelation of 7s Globulin from Soybeans. J. Agric. Food Chem. 1992, 40, 941–944. [Google Scholar] [CrossRef]
- Li, X.H.; Li, Y.; Hua, Y.F.; Qiu, A.Y.; Yang, C.; Cui, S. Effect of Concentration, Ionic Strength and Freeze-Drying on the Heat-Induced Aggregation of Soy Proteins. Food Chem. 2007, 104, 1410–1417. [Google Scholar] [CrossRef]
- Schuldt, S.; Raak, N.; Jaros, D.; Rohm, H. Acid-Induced Formation of Soy Protein Gels in the Presence of Nacl. LWT-Food Sci. Technol. 2014, 57, 634–639. [Google Scholar] [CrossRef]
- Zhou, F.F.; Pan, M.K.; Liu, Y.; Guo, N.; Zhang, Q.; Wang, J.H. Effects of Na+ on the Cold Gelation between a Low-Methoxyl Pectin Extracted from Premna Microphylla Turcz and Soy Protein Isolate. Food Hydrocoll. 2020, 104, 105762. [Google Scholar] [CrossRef]
- Wang, X.Y.; Lee, J.Y.; Wang, Y.W.; Huang, Q.R. Composition and Rheological Properties of Β-Lactoglobulin/ Pectin Coacervates: Effects of Salt Concentration and Initial Protein/Polysaccharide Ratio. Biomacromolecules 2007, 8, 992–997. [Google Scholar] [CrossRef]
- Lam, E.; Otter, D.; Huppertz, T.; Zhou, P.; Hemar, Y. Effect of Transglutaminase and Acidification Temperature on the Gelation of Reconstituted Skim Milk. Int. Dairy J. 2019, 92, 59–68. [Google Scholar] [CrossRef]
- Van Eys, J.E. Manual of Quality Analyses for Soybean Products in the Feed Industry; American Soyabean Association: St. Louis, MO, USA, 2004; Available online: http://ussec.org/wp-content/uploads/2012/09/Manual-of-Quality-Analyses-2nd-edition.pdf (accessed on 15 January 2024).
- Osman, M.A.; Reid, P.M.; Weber, C.W. Thermal Inactivation of Tepary Bean (Phaseolus acutifolius), Soybean and Lima Bean Protease Inhibitors: Effect of Acidic and Basic Ph. Food Chem. 2002, 78, 419–423. [Google Scholar] [CrossRef]
- Chen, Y.M.; Xu, Z.C.; Zhang, C.M.; Kong, X.Z.; Hua, Y.F. Heat-Induced Inactivation Mechanisms of Kunitz Trypsin Inhibitor and Bowman-Birk Inhibitor in Soymilk Processing. Food Chem. 2014, 154, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Boye, J.I.; Hu, X.Z. Nutritional Quality and Techno-Functional Changes in Raw, Germinated and Fermented Yellow Field Pea (Pisum sativum L.) Upon Pasteurization. LWT-Food Sci. Technol. 2018, 92, 147–154. [Google Scholar] [CrossRef]
- Cros, S.; Lignot, B.; Bourseau, P.; Jaouen, P.; Prost, C. Desalination of Mussel Cooking Juices by Electrodialysis: Effect on the Aroma Profile. J. Food Eng. 2005, 69, 425–436. [Google Scholar] [CrossRef]
- Cissé, M.; Vaillant, F.; Bouquet, S.; Pallet, D.; Lutin, F.; Reynes, M.; Dornier, M. Athermal Concentration by Osmotic Evaporation of Roselle Extract, Apple and Grape Juices and Impact on Quality. Innov. Food Sci. Emerg. Technol. 2011, 12, 352–360. [Google Scholar] [CrossRef]
- Aguiar, I.B.; Miranda, N.G.M.; Gomes, F.S.; Santos, M.C.S.; Freitas, D.d.G.C.; Tonon, R.V.; Cabral, L.M.C. Physicochemical and Sensory Properties of Apple Juice Concentrated by Reverse Osmosis and Osmotic Evaporation. Innov. Food Sci. Emerg. Technol. 2012, 16, 137–142. [Google Scholar] [CrossRef]
- Ali, F.; Dornier, M.; Duquenoy, A.; Reynes, M. Evaluating Transfers of Aroma Compounds During the Concentration of Sucrose Solutions by Osmotic Distillation in a Batch-Type Pilot Plant. J. Food Eng. 2003, 60, 1–8. [Google Scholar] [CrossRef]
- Morath, S.U.; Hung, R.; Bennett, J.W. Fungal Volatile Organic Compounds: A Review with Emphasis on Their Biotechnological Potential. Fungal Biol. Rev. 2012, 26, 73–83. [Google Scholar] [CrossRef]
- Feng, X.X.; Hua, Y.F. The Precursors of C8 Alcohols from Soybean: Purification, Distribution and Hydrolysis Properties of Glycosidically Bound Volatiles. J. Food Sci. 2022, 87, 178–192. [Google Scholar] [CrossRef]
- Azarnia, S.; Boye, J.I.; Warkentin, T.; Malcolmson, L.; Sabik, H.; Bellido, A.S. Volatile Flavour Profile Changes in Selected Field Pea Cultivars as Affected by Crop Year and Processing. Food Chem. 2011, 124, 326–335. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Z.Y.; Zhang, D.Q.; Shen, Q.W.; Pan, T.; Hui, T.; Ma, J.R. Characterization of Key Aroma Compounds in Beijing Roasted Duck by Gas Chromatography-Olfactometry-Mass Spectrometry, Odor-Activity Values, and Aroma-Recombination Experiments. J. Agric. Food Chem. 2019, 67, 5847–5856. [Google Scholar] [CrossRef]
- Mosaferi, S.; Jelley, R.E.; Fedrizzi, B.; Barker, D. Synthesis of D6-Deuterated Analogues of Aroma Molecules-Β-Damascenone, Β-Damascone and Safranal. Results Chem. 2022, 4, 100264. [Google Scholar] [CrossRef]
- Wang, H.; Zielinska, M.; An, K.J.; Fang, X.M.; Raghavan, G.S.V.; Zhang, Y.; Orsat, V.; Xiao, H.M.; Xiao, H.W. Effects of Vacuum-Steam Pulsed Blanching (Vspb) and Storage on Β-Carotene, Color, Total Phenolics, Antioxidant Capacity and Volatile Profiles of Dried Carrots. LWT-Food Sci. Technol. 2023, 183, 114894. [Google Scholar] [CrossRef]
- Dobrev, I.; Denkova, R.; Denkova, Z.; Filipov, E.; Yanakieva, V. Non-Traditional Yogurt from Cow’s Milk Enriched with Pea Protein. J. Food Packag. Sci. Tech. Technol. 2014, 5, 38–44. [Google Scholar]
- Nieto-Arribas, P.; Seseña, S.; Poveda, J.M.; Chicón, R.; Cabezas, L.; Palop, L. Enterococcus Populations in Artisanal Manchego Cheese: Biodiversity, Technological and Safety Aspects. Food Microbiol. 2011, 28, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Y.; Cui, H.T.; Xu, J.X.; Yuan, Z.H.; Li, J.; Liu, J.; He, L.; Zhu, D.S. Effect of Fermentation of Danisco Mixed Lactic Acid Bacteria and Single Bifidobacterium Lactis Hcs04-001 on Metabolites of Soy Yogurt. Int. J. Food Eng. 2023, 19, 509–519. [Google Scholar]
- Zhu, J.C.; Chen, F.; Wang, L.Y.; Niu, Y.W.; Yu, D.; Shu, C.; Chen, H.X.; Wang, H.L.; Xiao, Z.B. Comparison of Aroma-Active Volatiles in Oolong Tea Infusions Using Gc-Olfactometry, Gc-Fpd, and Gc-Ms. J. Agric. Food Chem. 2015, 63, 7499–7510. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.J.; Guo, G.Y.; Liu, P.P.; Chen, L.; Wang, W.W.; Zhang, J.Y.; Yin, J.F.; Ni, D.J.; Engelhardt, U.H.; Jiang, H.Y. Identification of Aroma-Active Compounds Responsible for the Floral and Sweet Odors of Congou Black Teas Using Gas Chromatography-Mass Spectrometry/Olfactometry, Odor Activity Value, and Chemometrics. J. Sci. Food Agric. 2022, 102, 5399–5410. [Google Scholar] [CrossRef] [PubMed]
- Mudgil, P.; Jumah, B.; Ahmad, M.; Hamed, F.; Maqsood, S. Rheological, Micro-Structural and Sensorial Properties of Camel Milk Yogurt as Influenced by Gelatin. LWT-Food Sci. Technol. 2018, 98, 646–653. [Google Scholar] [CrossRef]


| Samples | SW | DSW | CSW | CDSW |
|---|---|---|---|---|
| Solid (%) | 3.27 ± 0.02 c | 2.22 ± 0.01 d | 18.98 ± 0.01 a | 17.09 ± 0.02 b |
| Protein (%) | 0.47 ± 0.02 b | 0.36 ± 0.02 c | 2.50 ± 0.00 a | 2.50 ± 0.00 a |
| Carbohydrate (%) | 1.65 ± 0.03 c | 1.41 ± 0.03 d | 8.94 ± 0.01 b | 9.75 ± 0.12 a |
| Ash (%) | 0.40 ± 0.02 c | 0.09 ± 0.00 d | 2.37 ± 0.25 a | 0.76 ± 0.06 b |
| Sugars (mg/mL sample) | ||||
| Fructose | 0.91 ± 0.00 c | 0.82 ± 0.02 c | 4.21 ± 0.06 b | 4.68 ± 0.15 a |
| Sucrose | 11.31 ± 0.19 c | 8.53 ± 0.05 c | 62.51 ± 4.47 b | 69.82 ± 0.99 a |
| Raffinose | 1.30 ± 0.04 c | 0.91 ± 0.00 d | 5.33 ± 0.32 b | 6.54 ± 0.29 a |
| Stachyose | 6.79 ± 0.02 b | 5.92 ± 0.03 c | 10.16 ± 0.46 a | 10.60 ± 0.19 a |
| Yogurt Samples | Firmness/ (g) | Consistency/ (g·s) | Cohesiveness/ (g) | Work of Cohesion/ (g·s) |
|---|---|---|---|---|
| CDSWY | 432.42 ± 37.70 a | 2289.40 ± 139.81 a | −14.51 ± 1.22 b | −24.77 ± 1.16 b |
| CSWY | 33.49 ± 1.96 b | 187.2 ± 15.78 b | −7.12 ± 0.17 a | −16.78 ± 0.39 a |
| Samples | Urease Activity | Trypsin Inhibitory Activity (U/mg Protein) |
|---|---|---|
| SW | + | 615.42 ± 54.34 |
| DSW | + | 699.67 ± 6.73 |
| CSW | − | 469.70 ± 11.08 |
| CDSW | − | 538.54 ± 1.74 |
| PCSW | − | 178.29 ± 1.97 |
| PCDSW | − | 201.95 ± 21.46 |
| CSWY | − | 56.29 ± 0.28 |
| CDSWY | − | 66.50 ± 2.74 |
| Volatile Compounds | SW | DSW | CSW | CDSW | CSWY | CDSWY |
|---|---|---|---|---|---|---|
| Alcohols | ||||||
| 1-Hexanol | 233.51 ± 0.22 | 104.77 ± 1.94 | - | - | 121.60 ± 39.25 | 45.48 ± 1.07 |
| 1-Octanol | 2.13 ± 0.59 | - | 28.36 ± 3.47 | 8.12 ± 0.99 | ||
| 1-Pentanol | 48.13 ± 4.01 | 26.90 ± 0.02 | - | - | - | - |
| 2-Hexanol | 45.00 ± 0.07 | 24.83 ± 0.93 | - | - | - | - |
| 2-Octanol | 2.39 ± 0.68 | - | - | - | - | - |
| 3-Hexanol | 43.99 ± 0.17 | 23.00 ± 3.25 | - | - | - | - |
| Benzyl alcohol | 2.26 ± 0.12 | 1.37 ± 0.09 | - | - | - | - |
| 1-Dodecanol | 10.26 ± 0.92 | 4.66 ± 0.48 | - | - | - | - |
| 2-Ethyl-1-hexanol | 1.96 ± 0.16 | 2.42 ± 0.17 | - | - | - | - |
| 1-Octen-3-ol | 570.29 ± 66.16 | 301.80 ± 20.17 | - | - | 1.83 ± 0.12 | 1.17 ± 0.02 |
| (E)-2-Hepten-1-ol | 2.74 ± 0.17 | 1.70 ± 0.01 | - | - | - | - |
| (E)-2-Penten-1-ol | 6.58 ± 0.12 | 2.91 ± 0.15 | - | - | - | - |
| (E)-3-Hexen-1-ol | 1.49 ± 0.61 | 1.22 ± 0.02 | - | - | - | - |
| 1- Nonanol | - | - | - | - | 47.80 ± 1.59 | 61.02 ± 3.97 |
| Phenethyl alcohol | - | - | - | - | 11.30 ± 0.08 | 6.21 ± 0.43 |
| Aldehydes | ||||||
| Hexanal | 474.82 ± 5.77 | 284.87 ± 14.15 | 27.28 ± 0.44 | 10.94 ± 0.80 | 19.90 ± 0.26 | 18.08 ± 2.16 |
| Nonanal | 5.25 ± 0.54 | 2.96 ± 0.08 | 18.95 ± 0.99 | 25.09 ± 0.64 | 12.85 ± 7.28 | 15.91 ± 0.71 |
| Octanal | 1.33 ± 0.07 | - | 1.91 ± 0.11 | 2.10 ± 0.24 | 3.34 ± 0.71 | 2.78 ± 0.00 |
| Pentanal | 16.72 ± 0.15 | 9.04 ± 1.62 | - | - | - | - |
| Heptanal | 13.61 ± 1.94 | 7.60 ± 0.59 | 2.19 ± 0.08 | 1.47 ± 0.17 | - | 3.02 ± 0.16 |
| Decanal | - | 1.40 ± 0.06 | 4.15 ± 0.19 | 5.38 ± 0.06 | - | - |
| Benzaldehyde | 8.41 ± 1.03 | 4.17 ± 0.77 | 7.79 ± 0.15 | 6.86 ± 0.13 | 18.32 ± 0.26 | 17.03 ± 0.04 |
| (E)-2-Heptenal | 11.60 ± 0.57 | 6.22 ± 0.28 | 25.62 ± 5.32 | 20.42 ± 4.83 | 175.26 ± 14.92 | 34.93 ± 0.48 |
| (E)-2-Hexenal | 19.10 ± 5.56 | 11.37 ± 1.72 | 9.62 ± 0.23 | 5.03 ± 0.42 | 8.22 ± 0.54 | 7.46 ± 0.03 |
| (E)-2-Nonenal | 4.28 ± 0.68 | 1.73 ± 0.25 | 5.15 ± 0.23 | 2.83 ± 0.24 | 6.85 ± 1.12 | 5.56 ± 1.48 |
| (E)-2-Octenal | 5.99 ± 1.07 | 3.39 ± 0.54 | 7.88 ± 0.34 | 4.91 ± 0.28 | 14.81 ± 1.37 | 14.81 ± 1.37 |
| (E)-2-Pentenal | 6.44 ± 1.27 | 3.24 ± 1.44 | 2.76 ± 1.17 | 2.06 ± 0.09 | - | - |
| (E,E)-2,4-Nonadienal | 2.99 ± 1.17 | - | 0.77 ± 0.03 | 0.64 ± 0.09 | 4.04 ± 1.03 | 3.07 ± 0.21 |
| Phenylacetaldehyde | - | - | 1.52 ± 0.10 | 1.47 ± 0.19 | 1.63 ± 0.11 | 2.16 ± 0.32 |
| (E,E)-2,4-Decadienal | - | - | 0.34 ± 0.04 | - | 16.00 ± 0.49 | 10.47 ± 0.26 |
| (E,E)-2,4-Heptadienal | - | - | 1.34 ± 0.17 | 1.98 ± 0.15 | 3.38 ± 0.19 | 4.84 ± 0.11 |
| (E)-2-Decenaldehyde | - | - | - | - | 7.14 ± 0.76 | 8.07 ± 0.26 |
| 2,4-Decadienal | - | - | 1.02 ± 0.02 | 0.81 ± 0.01 | 122.11 ± 19.50 | 112.93 ± 2.87 |
| Ketones | ||||||
| 3-Octanone | 4.91 ± 0.35 | - | - | - | 1.72 ± 0.93 | 2.29 ± 0.10 |
| Acetophenone | 0.61 ± 0.07 | 1.14 ± 0.07 | 1.66 ± 0.58 | 2.29 ± 0.03 | 3.35 ± 0.03 | 5.97 ± 0.79 |
| 1-Octen-3-one | 5.92 ± 0.43 | 6.87 ± 0.00 | - | - | - | - |
| (E)-3-Octen-2-one | 11.58 ± 1.90 | 3.62 ± 1.67 | 5.60 ± 0.14 | 6.07 ± 0.32 | 31.95 ± 2.11 | 33.03 ± 0.14 |
| (E,E)-3,5-Octadien-2-one | 3.42 ± 0.15 | 1.57 ± 0.42 | 0.71 ± 0.16 | 2.10 ± 0.17 | 4.66 ± 0.07 | 4.23 ± 0.20 |
| 2-Nonanone | - | - | 0.24 ± 0.02 | 0.47 ± 0.08 | 7.54 ± 0.06 | 8.99 ± 0.61 |
| 4- Octanone | - | - | 2.00 ± 0.06 | 2.19 ± 0.39 | 1.04 ± 0.02 | 7.23 ± 0.90 |
| 2- Undecanone | - | - | 0.23 ± 0.01 | 0.67 ± 0.02 | 3.24 ± 0.01 | 2.75± 0.21 |
| β-damascenone | - | - | 1.19 ± 0.11 | 0.44 ± 0.09 | 3.40 ± 0.10 | 2.22 ± 0.16 |
| 6-Methyl -5-heptene -2- one | - | - | 3.81 ± 0.05 | 3.51 ± 0.02 | 5.17 ± 0.57 | 4.40 ± 0.06 |
| 2,3-Butanedione | - | - | - | - | 70.05 ± 0.86 | 78.92 ± 6.06 |
| 2,3-Pentanedione | - | - | - | - | 76.48 ± 6.34 | 44.87 ± 1.31 |
| 3-Hydroxy -2- butanone | - | - | - | - | 26.04 ± 1.60 | 55.99 ± 0.73 |
| 2- Heptanone | - | - | - | - | 29.68 ± 3.50 | 18.32 ± 0.08 |
| Furans | ||||||
| 2-Pentylfuran | 2.92 ± 0.09 | 2.57 ± 0.58 | 10.29 ± 0.08 | 5.50 ± 0.56 | 91.05 ± 9.78 | 80.77 ± 2.72 |
| 2- Butylfuran | 6.01 ± 0.45 | 6.96 ± 1.81 | ||||
| 2- Ethylfuran | 2.16 ± 0.18 | 1.94 ± 0.46 | 19.61 ± 1.53 | 12.97 ± 1.30 | ||
| Acids | ||||||
| Acetic acid | - | - | 10.03 ± 0.48 | 6.92 ± 0.76 | 52.99 ± 1.98 | 53.46 ± 0.66 |
| Butanoic acid | - | - | 0.16 ± 0.02 | 0.66 ± 0.01 | 2.67 ± 0.48 | 1.22 ± 0.34 |
| Hexanoic acid | - | - | 1.62 ± 0.37 | 3.19 ± 0.31 | 16.16 ± 0.32 | 30.94 ± 10.43 |
| Decylic acid | - | 0.92 ± 0.26 | 1.26 ± 0.43 | 2.89 ± 0.91 | 1.55 ± 0.49 | |
| Nonanoic acid | - | - | 2.69 ± 0.25 | 25.50 ± 6.83 | 34.11 ± 2.58 | 11.61 ± 3.46 |
| Benzoic acid | - | - | 0.88 ± 0.06 | 0.91 ± 0.10 | 2.97 ± 0.01 | 1.48 ± 0.03 |
| Pentanoic acid | - | - | 0.61 ± 0.06 | 0.60 ± 0.01 | 5.65 ± 0.23 | 2.84 ± 0.57 |
| Octanoic acid | 0.98 ± 0.08 | - | - | - | 8.35 ± 1.60 | 8.59 ± 0.52 |
| Heptanoic acid | 0.96 ± 0.10 | - | - | - | 7.47 ± 1.94 | 8.40 ± 0.54 |
| Esters | ||||||
| Ethyl hexanoate | - | - | 1.52 ± 0.66 | 0.26 ± 0.07 | 16.92 ± 5.19 | 2.40 ± 0.80 |
| Hexyl hexanoate | - | - | 0.32 ± 0.04 | 0.13 ± 0.01 | 4.32 ± 0.26 | 1.63 ± 0.27 |
| Ethyl nonanoate | - | - | 0.26 ± 0.04 | 0.22 ± 0.03 | 2.39 ± 0.90 | 0.83 ± 0.15 |
| Etheyl octanoat | - | - | 1.74 ± 0.52 | 4.81 ± 0.42 | 29.33 ± 4.88 | 14.81 ± 1.37 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Long, J.; Liu, J.; Hua, Y.; Zhang, C.; Li, X. Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt. Foods 2024, 13, 330. https://doi.org/10.3390/foods13020330
Zhang X, Long J, Liu J, Hua Y, Zhang C, Li X. Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt. Foods. 2024; 13(2):330. https://doi.org/10.3390/foods13020330
Chicago/Turabian StyleZhang, Xinyu, Jie Long, Jun Liu, Yufei Hua, Caimeng Zhang, and Xingfei Li. 2024. "Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt" Foods 13, no. 2: 330. https://doi.org/10.3390/foods13020330
APA StyleZhang, X., Long, J., Liu, J., Hua, Y., Zhang, C., & Li, X. (2024). Fermentation Characteristics, Antinutritional Factor Level and Flavor Compounds of Soybean Whey Yogurt. Foods, 13(2), 330. https://doi.org/10.3390/foods13020330





