Fermented Vegetables: Health Benefits, Defects, and Current Technological Solutions
Abstract
1. Introduction
2. The Dynamic Process of the Fermentation of Pickled Vegetables
2.1. Microbial Diversity in Fermented Vegetables
2.2. The Nutrition Composition Changes during the Fermentation Procedure of Fermented Vegetables
2.2.1. The Dynamic Changes in the Primary Nutrients
2.2.2. Generation of Other Nutritious Substances
2.2.3. Factors Affecting the Nutrient Changes in Fermented Vegetables
3. Health Benefits of Fermented Vegetables
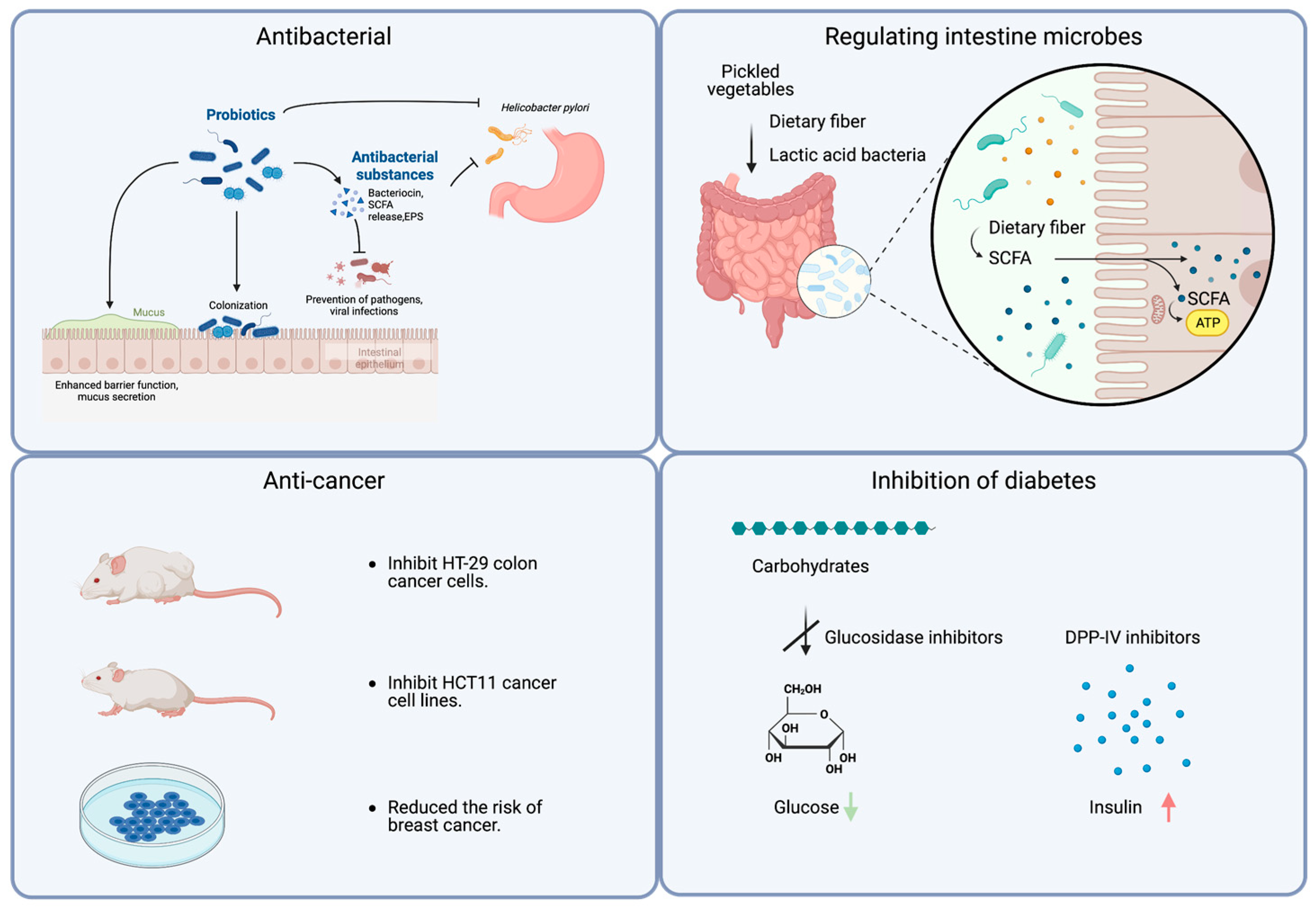
3.1. Antibacterial
3.2. Regulating the Intestine Microbes and Improving Intestine Health
3.3. Anti-Cancer
3.4. Inhibition of Diabetes
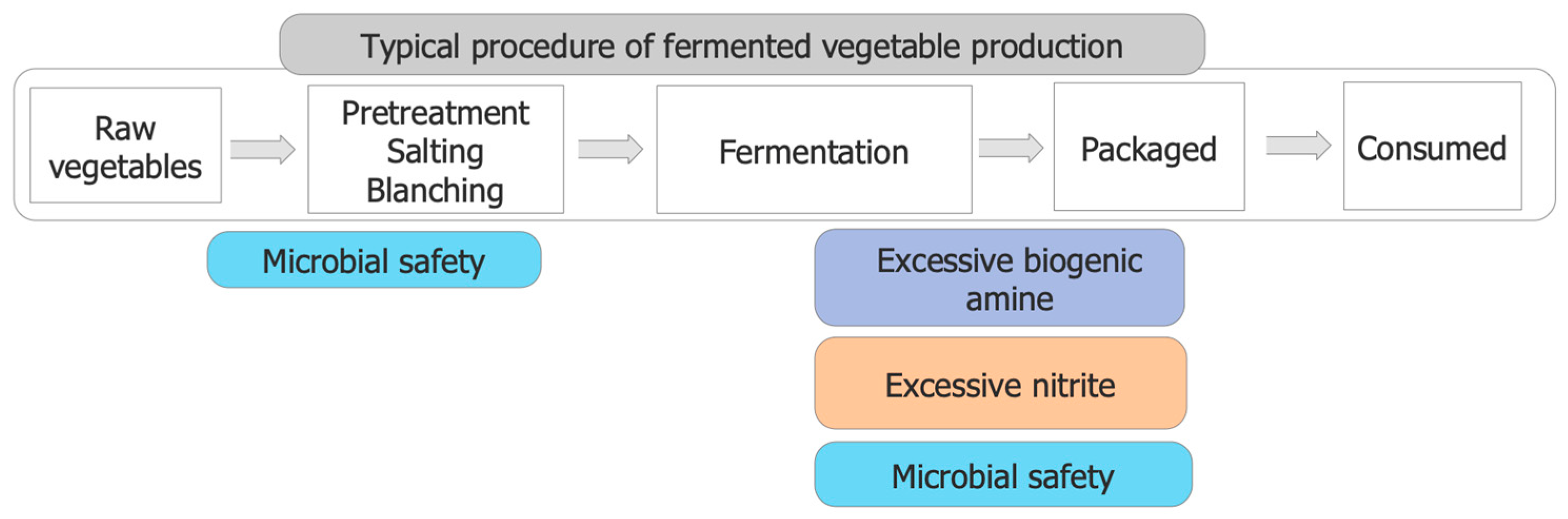
4. Safety Problems in Fermented Vegetables and Current Solutions
4.1. Biogenic Amine
4.2. Nitrite
| Potential Mechanism | Factor That Play a Main Role | Main Results | Ref. |
|---|---|---|---|
| Acid degradation | The organic acid produced by the lactic acid bacteria | A low pH caused by the metabolic products (lactic acid, acetic acid, butyric acid, tartaric acid, succinic acid, citric acid, and malic acid) of the lactic acid bacteria, which cause the degradation of nitrite. Mixed strain fermentation has a more significant degrading effects. | [131,133,134] |
| Enzyme degradation | The nitrite reductase enzyme system exist in the microorganism | The nitrite reductase system of nitrite-reducing bacteria consists of genes such as nirK, nirS, and nirBD, which could convert nitrite into NO2, NO, and N2. | [123,135] |
| Metabolic pathway degradation | The particular metabolic pathway of the microorganisms | Received electrons generated by the glycolysis/gluconeogenesis and citrate cycle, which eventually convert nitrite to L-glutamine. Decarboxylation reaction. Denitrification. | [135,141] |
4.3. Microbial Safety
4.3.1. High-Pressure Processing (HPP) Technology
4.3.2. Ultrasound Technology
4.3.3. Cold Plasma Technology
4.3.4. Photodynamic Sterilization
4.3.5. Pulsed Electric Field Technology (PEF)
4.3.6. How Far Are Non-Thermal Sterilization Technologies Being Applied in the Industry?
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| GABA | γ-aminobutyric acid |
| LAB | lactic acid bacteria |
| Thr | threonine |
| Ser | serine |
| Gly | glycine |
| Ala | alanine |
| Pro | proline |
| DPP-IV | dipeptidyl peptidase-IV |
| GLP-1 | glucagon-like peptide one |
| GIP | gastric inhibitory peptide |
| BA | biogenic amine |
| MCO | multicopper oxidase |
| PUT | putrescine |
| CAD | cadaverine |
| HIS | histamine |
| TYR | tyramine |
| SPE | spermine |
| PHE | phenethylamine |
| HPP | high-pressure processing |
| TPC | total plate count |
| IDF | insoluble dietary fiber |
| CP | cold plasma |
| PEF | pulsed electric field technology |
References
- Yongsawas, R.; Inta, A.; Kampuansai, J.; Pandith, H.; Suwannarach, N.; Lamyong, S.; Chantawannakul, P.; Chitov, T.; Disayathanoowat, T. Bacterial Communities in Lanna Phak-Gard-Dong (Pickled Mustard Green) from Three Different Ethnolinguistic Groups in Northern Thailand. Biology 2022, 11, 150. [Google Scholar] [CrossRef] [PubMed]
- Galena, A.E.; Chai, J.; Zhang, J.; Bednarzyk, M.; Perez, D.; Ochrietor, J.D.; Jahan-Mihan, A.; Arikawa, A.Y. The Effects of Fermented Vegetable Consumption on the Composition of the Intestinal Microbiota and Levels of Inflammatory Markers in Women: A Pilot and Feasibility Study. PLoS ONE 2022, 17, e0275275. [Google Scholar] [CrossRef] [PubMed]
- Liang, T.; Xie, X.; Wu, L.; Li, L.; Li, H.; Xi, Y.; Feng, Y.; Xue, L.; Chen, M.; Chen, X.; et al. Microbial Communities and Physiochemical Properties of Four Distinctive Traditionally Fermented Vegetables from North China and Their Influence on Quality and Safety. Foods 2021, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Satora, P.; Skotniczny, M.; Strnad, S.; Piechowicz, W. Chemical Composition and Sensory Quality of Sauerkraut Produced from Different Cabbage Varieties. Lwt 2021, 136, 110325. [Google Scholar] [CrossRef]
- Yang, X.; Hu, W.; Xiu, Z.; Jiang, A.; Yang, X.; Sarengaowa; Ji, Y.; Guan, Y.; Feng, K. Microbial Dynamics and Volatilome Profiles during the Fermentation of Chinese Northeast Sauerkraut by Leuconostoc Mesenteroides ORC 2 and Lactobacillus plantarum HBUAS 51041 under Different Salt Concentrations. Food Res. Int. 2020, 130, 108926. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Fan, Y.; Li, T.; Yang, Y.; Zeng, F.; Wang, H.; Suo, H.; Song, J.; Zhang, Y. Microbial Composition and Correlation between Microbiota and Quality-Related Physiochemical Characteristics in Chongqing Radish Paocai. Food Chem. 2022, 369, 130897. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, J.; Liu, D. Biochemical Changes and Microbial Community Dynamics during Spontaneous Fermentation of Zhacai, a Traditional Pickled Mustard Tuber from China. Int. J. Food Microbiol. 2021, 347, 109199. [Google Scholar] [CrossRef]
- Lee, S.H.; Whon, T.W.; Roh, S.W.; Jeon, C.O. Unraveling Microbial Fermentation Features in Kimchi: From Classical to Meta-Omics Approaches. Appl. Microbiol. Biotechnol. 2020, 104, 7731–7744. [Google Scholar] [CrossRef]
- Xiao, Z.B.; Zhu, J.C.; Feng, T.; Tian, H.X.; Yu, H.Y.; Niu, Y.W.; Zhang, X.M. Comparison of Volatile Components in Chinese Traditional Pickled Peppers Using HS–SPME–GC–MS, GC–O and Multivariate Analysis. Nat. Prod. Res. 2010, 24, 1939–1953. [Google Scholar] [CrossRef]
- Yuan, L.; Xu, F.; Xu, Y.; Wu, J.; Lao, F. Production of Marinated Chinese Lotus Root Slices Using High-Pressure Processing as an Alternative to Traditional Thermal-and-Soaking Procedure. Molecules 2022, 27, 6506. [Google Scholar] [CrossRef]
- Wang, D.; Ma, Y.; Sun, X.; Zhang, M.; Zhao, Y.; Zhao, X. Effect of Dense Phase Carbon Dioxide Treatment on Physicochemical and Textural Properties of Pickled Carrot. Cyta-J. Food 2019, 17, 988–996. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, L.; Liu, L.; Bi, X.; Liu, X. Characteristics of Microbial Communities in Fermentation of Pickled Ginger and Their Correlation with Its Volatile Flavors. Food Biosci. 2023, 53, 102736. [Google Scholar] [CrossRef]
- Moore, J.F.; DuVivier, R.; Johanningsmeier, S.D. Changes in the Free Amino Acid Profile of Pickling Cucumber during Lactic Acid Fermentation. J. Food Sci. 2022, 87, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Janiszewska-Turak, E.; Tracz, K.; Bielińska, P.; Rybak, K.; Pobiega, K.; Gniewosz, M.; Woźniak, Ł.; Gramza-Michałowska, A. The Impact of the Fermentation Method on the Pigment Content in Pickled Beetroot and Red Bell Pepper Juices and Freeze-Dried Powders. Appl. Sci. 2022, 12, 5766. [Google Scholar] [CrossRef]
- Montaño, A.; Casado, F.J.; de Castro, A.; Sánchez, A.H.; Rejano, L. Vitamin Content and Amino Acid Composition of Pickled Garlic Processed with and without Fermentation. J. Agric. Food Chem. 2004, 52, 7324–7330. [Google Scholar] [CrossRef] [PubMed]
- Fayek, N.M.; Farag, M.A.; Saber, F.R. Metabolome Classification via GC/MS and UHPLC/MS of Olive Fruit Varieties Grown in Egypt Reveal Pickling Process Impact on Their Composition. Food Chem. 2021, 339, 127861. [Google Scholar] [CrossRef]
- Shang, Z.; Li, M.; Zhang, W.; Cai, S.; Hu, X.; Yi, J. Analysis of Phenolic Compounds in Pickled Chayote and Their Effects on Antioxidant Activities and Cell Protection. Food Res. Int. 2022, 157, 111325. [Google Scholar] [CrossRef]
- Tang, Y.; Chen, G.; Wang, D.; Hu, R.; Li, H.; Liu, S.; Zhang, Q.; Ming, J.; Chi, Y. Effects of Dry-Salting and Brine-Pickling Processes on the Physicochemical Properties, Nonvolatile Flavour Profiles and Bacterial Community during the Fermentation of Chinese Salted Radishes. Lwt 2022, 157, 113084. [Google Scholar] [CrossRef]
- Rao, Y.; Tao, Y.; Chen, X.; She, X.; Qian, Y.; Li, Y.; Du, Y.; Xiang, W.; Li, H.; Liu, L. The Characteristics and Correlation of the Microbial Communities and Flavors in Traditionally Pickled Radishes. Lwt 2020, 118, 108804. [Google Scholar] [CrossRef]
- Torres, S.; Verón, H.; Contreras, L.; Isla, M.I. An Overview of Plant-Autochthonous Microorganisms and Fermented Vegetable Foods. Food Sci. Hum. Wellness 2020, 9, 112–123. [Google Scholar] [CrossRef]
- Wang, D.; Chen, G.; Tang, Y.; Li, J.; Huang, R.; Ye, M.; Ming, J.; Wu, Y.; Xu, F.; Lai, X.; et al. Correlation between Autochthonous Microbial Communities and Flavor Profiles during the Fermentation of Mustard Green Paocai (Brassica juncea Coss.), a Typical Industrial-Scaled Salted Fermented Vegetable. Lwt 2022, 172, 114212. [Google Scholar] [CrossRef]
- Singhal, P.; Satya, S.; Naik, S.N. Fermented Bamboo Shoots: A Complete Nutritional, Anti-Nutritional and Antioxidant Profile of the Sustainable and Functional Food to Food Security. Food Chem. Mol. Sci. 2021, 3, 100041. [Google Scholar] [CrossRef] [PubMed]
- Major, N.; Bažon, I.; Išić, N.; Kovačević, T.K.; Ban, D.; Radeka, S.; Ban, S.G. Bioactive Properties, Volatile Compounds, and Sensory Profile of Sauerkraut Are Dependent on Cultivar Choice and Storage Conditions. Foods 2022, 11, 1218. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.F.; DuVivier, R.; Johanningsmeier, S.D. Formation of γ-Aminobutyric Acid (GABA) during the Natural Lactic Acid Fermentation of Cucumber. J. Food Compos. Anal. 2021, 96, 103711. [Google Scholar] [CrossRef]
- Lee, N.-K.; Han, K.J.; Son, S.-H.; Eom, S.J.; Lee, S.-K.; Paik, H.-D. Multifunctional Effect of Probiotic Lactococcus Lactis KC24 Isolated from Kimchi. Lwt-Food Sci. Technol. 2015, 64, 1036–1041. [Google Scholar] [CrossRef]
- Yan, J.; Huang, Y.; Gao, Z.; Zhang, Z.; Gu, Q.; Li, P. Probiotic Potential of Lactiplantibacillus plantarum ZFM4 Isolated from Pickles and Its Effects on Human Intestinal Microecology. LWT 2023, 184, 114954. [Google Scholar] [CrossRef]
- Zhu, K.; Tan, F.; Mu, J.; Yi, R.; Zhou, X.; Zhao, X. Anti-Obesity Effects of Lactobacillus fermentum CQPC05 Isolated from Sichuan Pickle in High-Fat Diet-Induced Obese Mice through PPAR-α Signaling Pathway. Microorganisms 2019, 7, 194. [Google Scholar] [CrossRef] [PubMed]
- Freijy, T.M.; Cribb, L.; Oliver, G.; Metri, N.-J.; Opie, R.S.; Jacka, F.N.; Hawrelak, J.A.; Rucklidge, J.J.; Ng, C.H.; Sarris, J. Effects of a High-Prebiotic Diet versus Probiotic Supplements versus Synbiotics on Adult Mental Health: The “Gut Feelings” Randomised Controlled Trial. Front. Neurosci. 2023, 16, 1097278. [Google Scholar] [CrossRef]
- Ragul, K.; Kandasamy, S.; Devi, P.B.; Shetty, P.H. Evaluation of Functional Properties of Potential Probiotic Isolates from Fermented Brine Pickle. Food Chem. 2020, 311, 126057. [Google Scholar] [CrossRef]
- Yi, R.; Tan, F.; Zhou, X.; Mu, J.; Li, L.; Du, X.; Yang, Z.; Zhao, X. Effects of Lactobacillus fermentum CQPC04 on Lipid Reduction in C57BL/6J Mice. Front. Microbiol. 2020, 11, 573586. [Google Scholar] [CrossRef]
- Raak, C.; Ostermann, T.; Boehm, K.; Molsberger, F. Regular Consumption of Sauerkraut and Its Effect on Human Health: A Bibliometric Analysis. Glob. Adv. Heal. Med. 2014, 3, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J.; Anto, J.M.; Czarlewski, W.; Haahtela, T.; Fonseca, S.C.; Iaccarino, G.; Blain, H.; Vidal, A.; Sheikh, A.; Akdis, C.A.; et al. Cabbage and Fermented Vegetables: From Death Rate Heterogeneity in Countries to Candidates for Mitigation Strategies of Severe COVID-19. Allergy 2020, 76, 16. [Google Scholar] [CrossRef] [PubMed]
- Komnenov, D.; Levanovich, P.E.; Rossi, N.F. Hypertension Associated with Fructose and High Salt: Renal and Sympathetic Mechanisms. Nutrients 2019, 11, 569. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Lv, Y.; Wu, C.; Liu, B.; Shu, Z.; Lin, Y. Pickled Vegetables Intake Impacts the Metabolites for Gastric Cancer. Cancer Manag. Res. 2020, 12, 8263–8273. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Capillas, C.; Herrero, A.M. Impact of Biogenic Amines on Food Quality and Safety. Foods 2019, 8, 62. [Google Scholar] [CrossRef] [PubMed]
- Chien, H.-I.; Yen, Y.-F.; Lee, Y.-C.; Wei, P.-C.; Huang, C.-Y.; Tseng, C.-H.; Yen, F.-L.; Tsai, Y.-H. Determination of the Bacterial Community of Mustard Pickle Products and Their Microbial and Chemical Qualities. Biology 2023, 12, 258. [Google Scholar] [CrossRef] [PubMed]
- Min, M.; Bunt, C.R.; Mason, S.L.; Hussain, M.A. Non-Dairy Probiotic Food Products: An Emerging Group of Functional Foods. Crit. Rev. Food Sci. 2019, 59, 2626–2641. [Google Scholar] [CrossRef]
- Liu, D.; Tong, C. Bacterial Community Diversity of Traditional Fermented Vegetables in China. LWT-Food Sci. Technol. 2017, 86, 40–48. [Google Scholar] [CrossRef]
- Behera, S.S.; Sheikha, A.F.E.; Hammami, R.; Kumar, A. Traditionally Fermented Pickles: How the Microbial Diversity Associated with Their Nutritional and Health Benefits? J. Funct. Food 2020, 70, 103971. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. Bibliometrix: An R-Tool for Comprehensive Science Mapping Analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Chen, C.; Cheng, G.; Liu, Y.; Yi, Y.; Chen, D.; Zhang, L.; Wang, X.; Cao, J. Correlation between Microorganisms and Flavor of Chinese Fermented Sour Bamboo Shoot: Roles of Lactococcus and Lactobacillus in Flavor Formation. Food Biosci. 2022, 50, 101994. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Alagarsamy, K.; Suganthy, N.; Thangaleela, S.; Kesika, P.; Chaiyasut, C. The Role and Significance of Bacillus and Lactobacillus Species in Thai Fermented Foods. Ferment 2022, 8, 635. [Google Scholar] [CrossRef]
- Zou, Y.; Zhong, Y.; Huang, L.; Xu, W.; Wu, Y.; Gao, J.; Zhong, K.; Gao, H. Effects of Brown Sugar Addition and Fermentation Time on Metabolites and Microbial Communities of Yibin Yacai. Lwt 2022, 165, 113720. [Google Scholar] [CrossRef]
- Zhang, S.; Shang, Z.; Liu, Z.; Hu, X.; Yi, J. Flavor Production in Fermented Chayote Inoculated with Lactic Acid Bacteria Strains: Genomics and Metabolomics Based Analysis. Food Res. Int. 2022, 163, 112224. [Google Scholar] [CrossRef] [PubMed]
- Okoye, C.O.; Dong, K.; Wang, Y.; Gao, L.; Li, X.; Wu, Y.; Jiang, J. Comparative Genomics Reveals the Organic Acid Biosynthesis Metabolic Pathways among Five Lactic Acid Bacterial Species Isolated from Fermented Vegetables. New Biotechnol. 2022, 70, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Peng, Z.; Huang, T.; Xiao, Y.; Li, J.; Xie, M.; Xiong, T. Comparison of Bacterial Diversity in Traditionally Homemade Paocai and Chinese Spicy Cabbage. Food Microbiol. 2019, 83, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Shang, Z.; Zhang, S.; Li, M.; Zhang, X.; Ren, H.; Hu, X.; Yi, J. Dynamic Analysis of Flavor Properties and Microbial Communities in Chinese Pickled Chili Pepper (Capsicum frutescens L.): A Typical Industrial-Scale Natural Fermentation Process. Food Res. Int. 2022, 153, 110952. [Google Scholar] [CrossRef]
- Jin, Y.; Qi, Y.; Fan, M.; Zhang, J.; Kong, B.; Shao, B. Biotransformation of Carbendazim in Cowpea Pickling Process. Food Chem. 2023, 415, 135766. [Google Scholar] [CrossRef]
- Kanpiengjai, A.; Nuntikaew, P.; Wongsanittayarak, J.; Leangnim, N.; Khanongnuch, C. Isolation of Efficient Xylooligosaccharides-Fermenting Probiotic Lactic Acid Bacteria from Ethnic Pickled Bamboo Shoot Products. Biology 2022, 11, 638. [Google Scholar] [CrossRef]
- Dong, L.; Zhu, J.; Li, X.; Li, J. Effect of Tea Polyphenols on the Physical and Chemical Characteristics of Dried-Seasoned Squid (Dosidicus gigas) during Storage. Food Control 2013, 31, 586–592. [Google Scholar] [CrossRef]
- Li, X.; Liu, D. Effects of Wheat Bran Co-Fermentation on the Quality and Bacterial Community Succession during Radish Fermentation. Food Res. Int. 2022, 157, 111229. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.-L.; Zhao, Q.-H.; Lu, Y.; Tang, J.; Cai, T.; Rao, Y.; Liu, L.; Zhang, Q. Tetracycline Residue Alters Profile of Lactic Acid Bacterial Communities and Metabolites of Ginger Pickle during Spontaneous Fermentation. Food Res. Int. 2022, 155, 111109. [Google Scholar] [CrossRef] [PubMed]
- Šalić, A.; Šamec, D. Changes in the Content of Glucosinolates, Polyphenols and Carotenoids during Lactic-Acid Fermentation of Cruciferous Vegetables: A Mini Review. Food Chem. X 2022, 16, 100457. [Google Scholar] [CrossRef] [PubMed]
- Dallal, M.M.S.; Zamaniahari, S.; Davoodabadi, A.; Hosseini, M.; Rajabi, Z. Identification and Characterization of Probiotic Lactic Acid Bacteria Isolated from Traditional Persian Pickled Vegetables. GMS Hyg. Infect. Control 2017, 12, Doc5. [Google Scholar] [CrossRef]
- Shang, Z.; Ye, Z.; Li, M.; Ren, H.; Cai, S.; Hu, X.; Yi, J. Dynamics of Microbial Communities, Flavor, and Physicochemical Properties of Pickled Chayote during an Industrial-Scale Natural Fermentation: Correlation between Microorganisms and Metabolites. Food Chem. 2022, 377, 132004. [Google Scholar] [CrossRef] [PubMed]
- Kiczorowski, P.; Kiczorowska, B.; Samolińska, W.; Szmigielski, M.; Winiarska-Mieczan, A. Effect of Fermentation of Chosen Vegetables on the Nutrient, Mineral, and Biocomponent Profile in Human and Animal Nutrition. Sci. Rep. 2022, 12, 13422. [Google Scholar] [CrossRef] [PubMed]
- Ifesan, B.O.T.; Egbewole, O.O.; Ifesan, B.T. Effect of Fermentation on Nutritional Composition of Selected Commonly Consumed Green Leafy Vegetables in Nigeria. Int. J. Appl. Sci. Biotechnol. 2014, 2, 291–297. [Google Scholar] [CrossRef]
- Jin, Q.; Xie, F.; Luo, J.; Huang, X.; Wen, J.; Zhang, W.; Wu, J.; He, J.; Wang, Z. Investigation of Functional and Structural Properties of Insoluble Dietary Fiber From Sichuan Natural Fermented Pickles With Different Salting Treatments. Starch Stärke 2018, 70, 1800047. [Google Scholar] [CrossRef]
- Li, W.; Jin, Q.; Wu, Q.; Zhang, W.; Luo, Y.; Gu, S.; Wu, J.; Wang, Z. Effect of a Hybrid Process, High Hydrostatic Pressure Treatment Combined with Mixed-strain Fermentation, on the Quality of the Dietary Fibre in Pickled Vegetables. Int. J. Food Sci. Technol. 2020, 55, 2650–2659. [Google Scholar] [CrossRef]
- Misci, C.; Taskin, E.; Dall’Asta, M.; Fontanella, M.C.; Bandini, F.; Imathiu, S.; Sila, D.; Bertuzzi, T.; Cocconcelli, P.S.; Puglisi, E. Fermentation as a Tool for Increasing Food Security and Nutritional Quality of Indigenous African Leafy Vegetables: The Case of Cucurbita sp. Food Microbiol. 2021, 99, 103820. [Google Scholar] [CrossRef]
- Misci, C.; Taskin, E.; Vaccari, F.; Dall’Asta, M.; Vezzulli, F.; Fontanella, M.C.; Bandini, F.; Imathiu, S.; Sila, D.; Bertuzzi, T.; et al. Evolution of Microbial Communities and Nutritional Content of Fermented Amaranthus Sp. Leaves. Int. J. Food Microbiol. 2022, 362, 109445. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wu, D.; Liu, J.; Li, G.; Zhang, Z.; Chen, C.; Zhang, L.; Li, J. Characterization of Xanthine Oxidase Inhibitory Activities of Phenols from Pickled Radish with Molecular Simulation. Food Chem. X 2022, 14, 100343. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Li, R.W.; Yang, H.; Tan, Z.; Liu, F. Recent Advances in Developing Butyrogenic Functional Foods to Promote Gut Health. Crit. Rev. Food Sci. 2022, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Ciska, E.; Honke, J.; Drabińska, N. Changes in Glucosinolates and Their Breakdown Products during the Fermentation of Cabbage and Prolonged Storage of Sauerkraut: Focus on Sauerkraut Juice. Food Chem. 2021, 365, 130498. [Google Scholar] [CrossRef] [PubMed]
- Palani, K.; Harbaum-Piayda, B.; Meske, D.; Keppler, J.K.; Bockelmann, W.; Heller, K.J.; Schwarz, K. Influence of Fermentation on Glucosinolates and Glucobrassicin Degradation Products in Sauerkraut. Food Chem. 2016, 190, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, Y.; Xiao, H.; Huang, H.; Deng, G.; Chen, M.; Jiang, L. Bacterial Communities and Volatile Organic Compounds in Traditional Fermented Salt-Free Bamboo Shoots. Food Biosci. 2022, 50, 102006. [Google Scholar] [CrossRef]
- Scheers, N.; Rossander-Hulthen, L.; Torsdottir, I.; Sandberg, A.-S. Increased Iron Bioavailability from Lactic-Fermented Vegetables Is Likely an Effect of Promoting the Formation of Ferric Iron (Fe3+). Eur. J. Nutr. 2016, 55, 373–382. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, X.; Wu, X.; Zhang, Z.; Wu, D.; Chen, C.; Su, W.; Zhang, L.; Li, J.; Wang, H.-M.D. Several Natural Phytochemicals from Chinese Traditional Fermented Food-Pickled Raphanus sativus L.: Purification and Characterization. Food Chem. X 2022, 15, 100390. [Google Scholar] [CrossRef]
- Kumakura, K.; Kato, R.; Kobayashi, T.; Sekiguchi, A.; Kimura, N.; Takahashi, H.; Takahashi, A.; Matsuoka, H. Nutritional Content and Health Benefits of Sun-Dried and Salt-Aged Radish (Takuan-Zuke). Food Chem. 2017, 231, 33–41. [Google Scholar] [CrossRef]
- Di, H.; Ma, J.; Zhang, Y.; Wei, J.; Yang, J.; Ma, J.; Bian, J.; Xu, J.; Huang, Z.; Tang, Y.; et al. Correlations between Flavor and Glucosinolates and Changes in Quality-Related Physiochemical Characteristics of Guizhou Suancai during the Fermentation Process. Food Chem. 2023, 405, 134965. [Google Scholar] [CrossRef]
- Martinez-Villaluenga, C.; Peñas, E.; Frias, J.; Ciska, E.; Honke, J.; Piskula, M.K.; Kozlowska, H.; Vidal-Valverde, C. Influence of Fermentation Conditions on Glucosinolates, Ascorbigen, and Ascorbic Acid Content in White Cabbage (Brassica oleracea Var. Capitata Cv. Taler) Cultivated in Different Seasons. J. Food Sci. 2009, 74, C62–C67. [Google Scholar] [CrossRef] [PubMed]
- Hallmann, E.; Marszałek, K.; Lipowski, J.; Jasińska, U.; Kazimierczak, R.; Średnicka-Tober, D.; Rembiałkowska, E. Polyphenols and Carotenoids in Pickled Bell Pepper from Organic and Conventional Production. Food Chem. 2019, 278, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Sawada, K.; Koyano, H.; Yamamoto, N.; Yamada, T. The Relationships between Microbiota and the Amino Acids and Organic Acids in Commercial Vegetable Pickle Fermented in Rice-Bran Beds. Sci. Rep. 2021, 11, 1791. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, D. Nutritional Content Dynamics and Correlation of Bacterial Communities and Metabolites in Fermented Pickled Radishes Supplemented With Wheat Bran. Front. Nutr. 2022, 9, 840641. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.; Mahmood, K.; Kamilah, H.; Sulaiman, S.; Ibrahim, M.; Ariffin, F. Effects of Blanching and Pickling Process on the Alcohol Acyltransferase (AAT) Activity, Myristicin Content and Quality Parameters of Pickled Nutmeg (Myristica fragrans). J. Food Sci. Technol. 2022, 59, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Peñas, E.; Pihlava, J.M.; Vidal-Valverde, C.; Frias, J. Influence of Fermentation Conditions of Brassica oleracea L. Var. Capitata on the Volatile Glucosinolate Hydrolysis Compounds of Sauerkrauts. Lwt-Food Sci. Technol. 2012, 48, 16–23. [Google Scholar] [CrossRef]
- Liu, L.; She, X.; Chen, X.; Qian, Y.; Tao, Y.; Li, Y.; Guo, S.; Xiang, W.; Liu, G.; Rao, Y. Microbiota Succession and Chemical Composition Involved in the Radish Fermentation Process in Different Containers. Front. Microbiol. 2020, 11, 445. [Google Scholar] [CrossRef]
- Liu, L.; She, X.; Qian, Y.; Li, Y.; Tao, Y.; Che, Z.; Liu, G.; Rao, Y. Effect of Different Fermenting Containers on the Deterioration of Sichuan Pickle. Lwt 2019, 111, 829–836. [Google Scholar] [CrossRef]
- Li, F.; Zhou, H.; Zhou, X.; Yi, R.; Mu, J.; Zhao, X.; Liu, W. Lactobacillus plantarum CQPC05 Isolated from Pickled Vegetables Inhibits Constipation in Mice. Appl. Sci. 2019, 9, 159. [Google Scholar] [CrossRef]
- Choi, E.A.; Chang, H.C. Cholesterol-Lowering Effects of a Putative Probiotic Strain Lactobacillus plantarum EM Isolated from Kimchi. Lwt-Food Sci. Technol. 2015, 62, 210–217. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, H.; Zhang, C.; Han, M.; Du, J.; Yang, X.; Li, W. Production, Purification and Characterization of ‘Iturin A-2′ a Lipopeptide with Antitumor Activity from Chinese Sauerkraut Bacterium Bacillus velezensis T701. Int. J. Pept. Res. Ther. 2021, 27, 2135–2147. [Google Scholar] [CrossRef]
- Yang, Y.; Pei, J. Isolation and Characterization of an Enterococcus Strain from Chinese Sauerkraut with Potential for Lead Removal. Eur. Food Res. Technol. 2020, 246, 2055–2064. [Google Scholar] [CrossRef]
- Hu, J.; Tian, X.; Wei, T.; Wu, H.; Lu, J.; Lyu, M.; Wang, S. Anti-Helicobacter pylori Activity of a Lactobacillus sp. PW-7 Exopolysaccharide. Foods 2021, 10, 2453. [Google Scholar] [CrossRef] [PubMed]
- Jeong, M.; Park, J.-M.; Han, Y.-M.; Park, K.Y.; Lee, D.H.; Yoo, J.-H.; Cho, J.Y.; Hahm, K.-B. Dietary Prevention of Helicobacter Pylori-Associated Gastric Cancer with Kimchi. Oncotarget 2015, 6, 29513–29526. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jang, S.; Lee, J.; Jung, U.; Choi, H.-S.; Suh, H.J. Identification of an Anti-Listerial Domain from Pediococcus pentosaceus T1 Derived from Kimchi, a Traditional Fermented Vegetable. Food Control 2014, 43, 42–48. [Google Scholar] [CrossRef]
- Joo, N.E.; Ritchie, K.; Kamarajan, P.; Miao, D.; Kapila, Y.L. Nisin, an Apoptogenic Bacteriocin and Food Preservative, Attenuates HNSCC Tumorigenesis via CHAC1. Cancer Med. 2012, 1, 295–305. [Google Scholar] [CrossRef]
- Song, J.; Peng, S.; Yang, J.; Zhou, F.; Suo, H. Isolation and Identification of Novel Antibacterial Peptides Produced by Lactobacillus fermentum SHY10 in Chinese Pickles. Food Chem. 2021, 348, 129097. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, C.; Lei, P.; Xin, X.; Liu, D.; Yi, H. Isolation, Purification, Identification, and Discovery of the Antibacterial Mechanism of Ld-Phenyllactic Acid Produced by Lactiplantibacillus plantarum CXG9 Isolated from a Traditional Chinese Fermented Vegetable. Food Control 2022, 132, 108490. [Google Scholar] [CrossRef]
- Li, J.; Huang, S.-Y.; Deng, Q.; Li, G.; Su, G.; Liu, J.; Wang, H.-M.D. Extraction and Characterization of Phenolic Compounds with Antioxidant and Antimicrobial Activities from Pickled Radish. Food Chem. Toxicol. 2020, 136, 111050. [Google Scholar] [CrossRef]
- Thriene, K.; Hansen, S.S.; Binder, N.; Michels, K.B. Effects of Fermented Vegetable Consumption on Human Gut Microbiome Diversity—A Pilot Study. Ferment 2022, 8, 118. [Google Scholar] [CrossRef]
- Li, J.; Deng, Q.; Zhang, Y.; Wu, D.; Li, G.; Liu, J.; Zhang, L.; Wang, H.D. Three Novel Dietary Phenolic Compounds from Pickled Raphanus sativus L. Inhibit Lipid Accumulation in Obese Mice by Modulating the Gut Microbiota Composition. Mol. Nutr. Food Res. 2021, 65, 2000780. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, E.S.; Garnås, E.; Jensen, K.J.; Hansen, L.H.; Olsen, P.S.; Ritz, C.; Krych, L.; Nielsen, D.S. Lacto-Fermented Sauerkraut Improves Symptoms in IBS Patients Independent of Product Pasteurisation—A Pilot Study. Food Funct. 2018, 9, 5323–5335. [Google Scholar] [CrossRef] [PubMed]
- Šola, K.F.; Vladimir-Knežević, S.; Hrabač, P.; Mucalo, I.; Saso, L.; Verbanac, D. The Effect of Multistrain Probiotics on Functional Constipation in the Elderly: A Randomized Controlled Trial. Eur. J. Clin. Nutr. 2022, 76, 1675–1681. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Song, J.-L.; Ju, J.-H.; Kang, S.-A.; Park, K.-Y. Anticancer Effects of Kimchi Fermented for Different Times and with Added Ingredients in Human HT-29 Colon Cancer Cells. Food Sci. Biotechnol. 2015, 24, 629–633. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Pan, Y.; Kwack, K.-B.; Chung, J.H.; Park, K.-Y. Increased Anticancer Activity of Organic Kimchi with Starters Demonstrated in HT-29 Cancer Cells. Appl. Sci. 2023, 13, 6654. [Google Scholar] [CrossRef]
- Pathak, D.R.; Stein, A.D.; He, J.-P.; Noel, M.M.; Hembroff, L.; Nelson, D.A.; Vigneau, F.; Shen, T.; Scott, L.J.; Charzewska, J.; et al. Cabbage and Sauerkraut Consumption in Adolescence and Adulthood and Breast Cancer Risk among US-Resident Polish Migrant Women. Int. J. Environ. Res. Public Health 2021, 18, 10795. [Google Scholar] [CrossRef]
- Cai, Y.; Yang, X.; Chen, S.; Tian, K.; Xu, S.; Deng, R.; Chen, M.; Yang, Y.; Liu, T. Regular Consumption of Pickled Vegetables and Fermented Bean Curd Reduces the Risk of Diabetes: A Prospective Cohort Study. Front. Public Health 2023, 11, 1155989. [Google Scholar] [CrossRef]
- Li, M.; Bao, X.; Zhang, X.; Ren, H.; Cai, S.; Hu, X.; Yi, J. Exploring the Phytochemicals and Inhibitory Effects against α-Glucosidase and Dipeptidyl Peptidase-IV in Chinese Pickled Chili Pepper: Insights into Mechanisms by Molecular Docking Analysis. Lwt 2022, 162, 113467. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Dang, Y.-M.; Ha, J.-H. Effect of Various Seasoning Ingredients on the Accumulation of Biogenic Amines in Kimchi during Fermentation. Food Chem. 2022, 380, 132214. [Google Scholar] [CrossRef]
- da Silva, M.B.; Rodrigues, L.F.O.S.; Monteiro, G.C.; Monar, G.R.S.; Gomez, H.A.G.; Junior, S.S.; Minatel, I.O.; Lima, G.P.P. Evaluation of Biogenic Amines and Nitrate in Raw and Pickled Jurubeba (Solanum paniculatum L.) Fruit. J. Food Sci. Technol. 2019, 56, 2970–2978. [Google Scholar] [CrossRef]
- Liu, L.; Du, P.; Zhang, G.; Mao, X.; Zhao, Y.; Wang, J.; Duan, C.; Li, C.; Li, X. Residual Nitrite and Biogenic Amines of Traditional Northeast Sauerkraut in China. Int. J. Food Prop. 2016, 20, 2448–2455. [Google Scholar] [CrossRef]
- Zhao, N.; Lai, H.; Wang, Y.; Huang, Y.; Shi, Q.; He, W.; Zhu, S.; Li, Y.; Zhu, Y.; Li, H.; et al. Assessment of Biogenic Amine and Nitrite Production in Low-Salt Paocai during Fermentation as Affected by Reused Brine and Fresh Brine. Food Biosci. 2021, 41, 100958. [Google Scholar] [CrossRef]
- Park, Y.K.; Lee, J.H.; Mah, J.-H. Occurrence and Reduction of Biogenic Amines in Kimchi and Korean Fermented Seafood Products. Foods 2019, 8, 547. [Google Scholar] [CrossRef] [PubMed]
- Özogul, F.; Hamed, I. The Importance of Lactic Acid Bacteria for the Prevention of Bacterial Growth and Their Biogenic Amines Formation: A Review. Crit. Rev. Food Sci. 2017, 58, 1660–1670. [Google Scholar] [CrossRef]
- Barbieri, F.; Montanari, C.; Gardini, F.; Tabanelli, G. Biogenic Amine Production by Lactic Acid Bacteria: A Review. Foods 2019, 8, 17. [Google Scholar] [CrossRef]
- Alan, Y.; Topalcengiz, Z.; Dığrak, M. Biogenic Amine and Fermentation Metabolite Production Assessments of Lactobacillus plantarum Isolates for Naturally Fermented Pickles. Lwt 2018, 98, 322–328. [Google Scholar] [CrossRef]
- Yu, Y.; Li, L.; Xu, Y.; An, K.; Shi, Q.; Yu, Y.; Xu, Z. Evaluation of the Relationship among Biogenic Amines, Nitrite and Microbial Diversity in Fermented Mustard. Molecules 2021, 26, 6173. [Google Scholar] [CrossRef]
- Jin, Y.H.; Lee, J.H.; Park, Y.K.; Lee, J.-H.; Mah, J.-H. The Occurrence of Biogenic Amines and Determination of Biogenic Amine-Producing Lactic Acid Bacteria in Kkakdugi and Chonggak Kimchi. Foods 2019, 8, 73. [Google Scholar] [CrossRef]
- Ye, H.; Lang, X.; Ji, Y.; Li, S.; Xin, N.; Meng, X.; Zhang, T.; Shen, X.; Zhao, C. The Interaction between Lactobacillus plantarum SC-5 and Its Biogenic Amine Formation with Different Salt Concentrations in Chinese Dongbei Suancai. Food Res. Int. 2021, 150, 110813. [Google Scholar] [CrossRef]
- Świder, O.; Wójcicki, M.; Bujak, M.; Juszczuk-Kubiak, E.; Szczepańska, M.; Roszko, M.Ł. Time Evolution of Microbial Composition and Metabolic Profile for Biogenic Amines and Free Amino Acids in a Model Cucumber Fermentation System Brined with 0.5% to 5.0% Sodium Chloride. Molecules 2021, 26, 5796. [Google Scholar] [CrossRef]
- Majcherczyk, J.; Surówka, K. Effects of Onion or Caraway on the Formation of Biogenic Amines during Sauerkraut Fermentation and Refrigerated Storage. Food Chem. 2019, 298, 125083. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, C.; Xu, W.; Lu, Z.; Fu, R.; He, X.; Ma, Z.; Zhang, H. Evaluation of Degradation Capability of Nitrite and Biogenic Amines of Lactic Acid Bacteria Isolated from Pickles and Potential in Sausage Fermentation. J. Food Process. Pres. 2022, 46, e16141. [Google Scholar] [CrossRef]
- Priyanka, V.; Ramesha, A.; Gayathri, D.; Vasudha, M. Molecular Characterization of Non-Biogenic Amines Producing Lactobacillus plantarum GP11 Isolated from Traditional Pickles Using HRESI-MS Analysis. J. Food Sci. Technol. 2021, 58, 2216–2226. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Jin, Y.H.; Pawluk, A.M.; Mah, J.-H. Reduction in Biogenic Amine Content in Baechu (Napa Cabbage) Kimchi by Biogenic Amine-Degrading Lactic Acid Bacteria. Microorganisms 2021, 9, 2570. [Google Scholar] [CrossRef] [PubMed]
- Qi, Q.; Huang, J.; Zhou, R.; Jin, Y.; Wu, C. Characterising the Mechanism of Abating Biogenic Amines Accumulation by Cocultures of Zygosaccharomyces rouxii and Tetragenococcus halophilus. Lwt 2022, 164, 113672. [Google Scholar] [CrossRef]
- Zhao, J.; Niu, C.; Du, S.; Liu, C.; Zheng, F.; Wang, J.; Li, Q. Reduction of Biogenic Amines Formation during Soybean Paste Fermentation by Using Staphylococcus carnosus M43 and Pediococcus acidilactici M28 as Starter Culture. Lwt 2020, 133, 109917. [Google Scholar] [CrossRef]
- Callejón, S.; Sendra, R.; Ferrer, S.; Pardo, I. Identification of a Novel Enzymatic Activity from Lactic Acid Bacteria Able to Degrade Biogenic Amines in Wine. Appl. Microbiol. Biotechnol. 2014, 98, 185–198. [Google Scholar] [CrossRef]
- Li, B.; Shiling, L. The Importance of Amine Oxidases on the Biogenic Amine Degradation in Fermented Foods: A Review. Process. Biochem. 2020, 99, 331–339. [Google Scholar] [CrossRef]
- Callejón, S.; Sendra, R.; Ferrer, S.; Pardo, I. Cloning and Characterization of a New Laccase from Lactobacillus plantarum J16 CECT 8944 Catalyzing Biogenic Amines Degradation. Appl. Microbiol. Biotechnol. 2016, 100, 3113–3124. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, Y.; Xu, B.; Wang, D.; Jiang, W. Characterisation and Application of Halomonas shantousis SWA25, a Halotolerant bacterium with Multiple Biogenic Amine Degradation Activity. Food Addit. Contam. Part A 2016, 33, 674–682. [Google Scholar] [CrossRef]
- Luo, W.; Wu, W.; Du, X.; Yu, Y.; Wu, J.; Xu, Y.; Li, L. Regulation of the Nitrite, Biogenic Amine and Flavor Quality of Cantonese Pickle by Selected Lactic Acid Bacteria. Food Biosci. 2023, 53, 102554. [Google Scholar] [CrossRef]
- Yu, Y.; Yu, Y.; Xu, Z. Evaluation of Nitrite, Ethyl Carbamate, and Biogenic Amines in Four Types of Fermented Vegetables. Foods 2021, 10, 3150. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Shao, Y. Effects of Microbial Diversity on Nitrite Concentration in Pao Cai, a Naturally Fermented Cabbage Product from China. Food Microbiol. 2018, 72, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.-T.; Wu, Z.-Y.; Zhang, W.-X. Effects of Garlic Addition on Bacterial Communities and the Conversions of Nitrate and Nitrite in a Simulated Pickle Fermentation System. Food Control 2020, 113, 107215. [Google Scholar] [CrossRef]
- Niu, P.; Wang, F.; Yuan, K.; Li, X.; Yang, X.; Guo, Y. Alkaline-Extracted Thinned Young Apple Polyphenols as an Effective Scavenger against Nitrite in Pickles: A Comparative Study with Ethanol-Extracted Polyphenols. Food Control 2021, 130, 108387. [Google Scholar] [CrossRef]
- Chen, Y.; Li, Q.; Xia, C.; Yang, F.; Xu, N.; Wu, Q.; Hu, Y.; Xia, L.; Wang, C.; Zhou, M. Effect of Selenium Supplements on the Antioxidant Activity and Nitrite Degradation of Lactic Acid Bacteria. World J. Microbiol. Biotechnol. 2019, 35, 61. [Google Scholar] [CrossRef]
- Ren, D.; Chen, P.; Li, W.; Su, X.; Bao, K.; Wang, Y.; Wang, J.; Liu, H. Screening, Mutagenesis of Nitrite-Degrading Lactobacilli in Chinese Traditional Fermented Sauerkraut and Its Application in the Production of Sauerkraut. J. Food Saf. 2016, 36, 474–481. [Google Scholar] [CrossRef]
- Xia, Y.; Liu, X.; Wang, G.; Zhang, H.; Xiong, Z.; Sun, Y.; Ai, L. Characterization and Selection of Lactobacillus brevis Starter for Nitrite Degradation of Chinese Pickle. Food Control 2017, 78, 126–131. [Google Scholar] [CrossRef]
- Liu, D.; Wang, P.; Zhang, X.; Xu, X.; Wu, H.; Li, L. Characterization of Nitrite Degradation by Lactobacillus casei Subsp. Rhamnosus LCR 6013. PLoS ONE 2014, 9, e93308. [Google Scholar] [CrossRef]
- Zhang, X.; Han, J.; Zheng, X.; Yan, J.; Chen, X.; Zhou, Q.; Zhao, X.; Gu, Q.; Li, P. Use of Lactiplantibacillus plantarum ZJ316 as a Starter Culture for Nitrite Degradation, Foodborne Pathogens Inhibition and Microbial Community Modulation in Pickled Mustard Fermentation. Food Chem. X 2022, 14, 100344. [Google Scholar] [CrossRef]
- Hang, S.; Zeng, L.; Han, J.; Zhang, Z.; Zhou, Q.; Meng, X.; Gu, Q.; Li, P. Lactobacillus plantarum ZJ316 Improves the Quality of Stachys Sieboldii Miq. Pickle by Inhibiting Harmful Bacteria Growth, Degrading Nitrite and Promoting the Gut Microbiota Health in Vitro. Food Funct. 2021, 13, 1551–1562. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Feng, T.; Du, G.; Chen, J. Evaluation of the Impact on Food Safety of a Lactobacillus Coryniformis Strain from Pickled Vegetables with Degradation Activity against Nitrite and Other Undesirable Compounds. Food Addit. Contam. Part 2016, 33, 623–630. [Google Scholar] [CrossRef]
- Du, R.; Song, G.; Zhao, D.; Sun, J.; Ping, W.; Ge, J. Lactobacillus casei Starter Culture Improves Vitamin Content, Increases Acidity and Decreases Nitrite Concentration during Sauerkraut Fermentation. Int. J. Food Sci. Technol. 2018, 53, 1925–1931. [Google Scholar] [CrossRef]
- Huang, Y.; Jia, X.; Yu, J.; Chen, Y.; Liu, D.; Liang, M. Effect of Different Lactic Acid Bacteria on Nitrite Degradation, Volatile Profiles, and Sensory Quality in Chinese Traditional Paocai. Lwt 2021, 147, 111597. [Google Scholar] [CrossRef]
- Fei, Y.; Liu, D.; Luo, T.; Chen, G.; Wu, H.; Li, L.; Yu, Y. Molecular Characterization of Lactobacillus plantarum DMDL 9010, a Strain with Efficient Nitrite Degradation Capacity. PLoS ONE 2014, 9, e113792. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Liu, D.; Jia, X.; Liang, M.; Lu, Y.; Liu, J. Whole Genome Sequencing of Lactobacillus plantarum DMDL 9010 and Its Effect on Growth Phenotype under Nitrite Stress. Lwt 2021, 149, 111778. [Google Scholar] [CrossRef]
- Zhang, W.; Tian, G.; Feng, S.; Wong, J.H.; Zhao, Y.; Chen, X.; Wang, H.; Ng, T.B. Boletus Edulis Nitrite Reductase Reduces Nitrite Content of Pickles and Mitigates Intoxication in Nitrite-Intoxicated Mice. Sci. Rep. 2015, 5, 14907. [Google Scholar] [CrossRef]
- Li, Y.; Xiong, D.; Yuan, L.; Fan, P.; Xiao, Y.; Chen, J.; Feng, W. Transcriptome and Protein Networks to Elucidate the Mechanism Underlying Nitrite Degradation by Lactiplantibacillus plantarum. Food Res. Int. 2022, 156, 111319. [Google Scholar] [CrossRef]
- Yao, K.; Liu, D.; Liang, M.; Brennan, C.S.; Brennan, M. Detection of Nitrite Degradation by Lactobacillus plantarum DMDL9010 through the Anaerobic Respiration Electron Transport Chain Using Proteomic Analysis. Int. J. Food Sci. Technol. 2021, 56, 1608–1622. [Google Scholar] [CrossRef]
- Xia, C.; Tian, Q.; Kong, L.; Sun, X.; Shi, J.; Zeng, X.; Pan, D. Metabolomics Analysis for Nitrite Degradation by the Metabolites of Limosilactobacillus fermentum RC4. Foods 2022, 11, 1009. [Google Scholar] [CrossRef]
- Zeng, X.; Pan, Q.; Guo, Y.; Wu, Z.; Sun, Y.; Dang, Y.; Cao, J.; He, J.; Pan, D. Potential Mechanism of Nitrite Degradation by Lactobacillus fermentum RC4 Based on Proteomic Analysis. J. Proteom. 2019, 194, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Hu, X.; Yang, S.; Wang, K.; Zeng, C.; Hou, Z.; Cui, H.; Liu, S.; Zhu, L. Denitrifying Halophilic Archaea Derived from Salt Dominate the Degradation of Nitrite in Salted Radish during Pickling. Food Res. Int. 2022, 152, 110906. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zeng, C.; Hou, Z.; Wang, Y.; Xu, Q.; Isobe, K.; Senoo, K.; Zhu, L. The Complete Genome Sequence of the Archaeal Isolate Halomicrobium Sp. ZPS1 Reveals the Nitrogen Metabolism Characteristics under Hypersaline Conditions. Ann. Microbiol. 2020, 70, 29. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, F.; Gong, C.; Tan, X.; Ren, Y.; Yao, K.; Zhang, Q.; Chi, Y. Physicochemical, Microbial, and Aroma Characteristics of Chinese Pickled Red Peppers (Capsicum annuum) with and without Biofilm. Rsc. Adv. 2020, 10, 6609–6617. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Shang, Z.; Li, M.; Qu, Y.; Long, H.; Yi, J. Evaluation of the Physiochemical and Aromatic Qualities of Pickled Chinese Pepper (Paojiao) and Their Influence on Consumer Acceptability by Using Targeted and Untargeted Multivariate Approaches. Food Res. Int. 2020, 137, 109535. [Google Scholar] [CrossRef] [PubMed]
- Bao, R.; Fan, A.; Hu, X.; Liao, X.; Chen, F. Effects of High Pressure Processing on the Quality of Pickled Radish during Refrigerated Storage. Innov. Food Sci. Emerg. 2016, 38, 206–212. [Google Scholar] [CrossRef]
- Zhao, N.; Ge, L.; Huang, Y.; Wang, Y.; Wang, Y.; Lai, H.; Wang, Y.; Zhu, Y.; Zhang, J. Impact of Cold Plasma Processing on Quality Parameters of Packaged Fermented Vegetable (Radish Paocai) in Comparison with Pasteurization Processing: Insight into Safety and Storage Stability of Products. Innov. Food Sci. Emerg. 2020, 60, 102300. [Google Scholar] [CrossRef]
- Choi, E.J.; Park, H.W.; Kim, S.B.; Ryu, S.; Lim, J.; Hong, E.J.; Byeon, Y.S.; Chun, H.H. Sequential Application of Plasma-Activated Water and Mild Heating Improves Microbiological Quality of Ready-to-Use Shredded Salted Kimchi Cabbage (Brassica pekinensis L.). Food Control 2019, 98, 501–509. [Google Scholar] [CrossRef]
- Song, H.; Dang, Y.-M.; Ha, S.; Ha, J.-H. Effect of Ultraviolet-C Light-Emitting Diode Irradiation on Inactivation of White Colony-Forming Yeast in Kimchi Seasoning. Food Control 2022, 140, 109157. [Google Scholar] [CrossRef]
- Rendueles, E.; Omer, M.K.; Alvseike, O.; Alonso-Calleja, C.; Capita, R.; Prieto, M. Microbiological Food Safety Assessment of High Hydrostatic Pressure Processing: A Review. Lwt-Food Sci. Technol. 2011, 44, 1251–1260. [Google Scholar] [CrossRef]
- Ma, J.; Wang, H.; Yu, L.; Yuan, W.; Fu, W.; Gao, F.; Jiang, Y. Dynamic Self-Recovery of Injured Escherichia coli O157:H7 Induced by High Pressure Processing. Lwt 2019, 113, 108308. [Google Scholar] [CrossRef]
- Wu, S.-M.; Wu, C.-P.; Lin, Y.-H.; Wu, Y.-H.; Huang, B.-C.; Wang, C.-Y. Effect of High Pressure Pretreatment on Myrosinase-Glucosinolate System, Physicochemical and Bacterial Properties during Fermentation of Brine-Pickled Radishes. Food Res. Int. 2022, 162, 112018. [Google Scholar] [CrossRef] [PubMed]
- Peñas, E.; Limón, R.I.; Vidal-Valverde, C.; Frias, J. Effect of Storage on the Content of Indole-Glucosinolate Breakdown Products and Vitamin C of Sauerkrauts Treated by High Hydrostatic Pressure. Lwt-Food Sci. Technol. 2013, 53, 285–289. [Google Scholar] [CrossRef]
- Li, J.; Zhao, F.; Liu, H.; Li, R.; Wang, Y.; Liao, X. Fermented Minced Pepper by High Pressure Processing, High Pressure Processing with Mild Temperature and Thermal Pasteurization. Innov. Food Sci. Emerg. 2016, 36, 34–41. [Google Scholar] [CrossRef]
- Liu, L.; Deng, X.; Huang, L.; Li, Y.; Zhang, Y.; Chen, X.; Guo, S.; Yao, Y.; Yang, S.; Tu, M.; et al. Comparative Effects of High Hydrostatic Pressure, Pasteurization and Nisin Processing Treatments on the Quality of Pickled Radish. Lwt 2022, 167, 113833. [Google Scholar] [CrossRef]
- Chen, F.; Chen, Y.; Wang, Y.; Ding, S.; Qin, Y.; Jiang, L.; Wang, R. High Pressure Processing Improves the Texture Quality of Fermented Minced Pepper by Maintaining Pectin Characteristics during Storage. J. Food Sci. 2022, 87, 2427–2439. [Google Scholar] [CrossRef]
- Yang, Z.; Duan, X.; Yang, J.; Wang, H.; Liu, F.; Xu, X.; Pan, S. Effects of High Hydrostatic Pressure and Thermal Treatment on Texture Properties of Pickled Kohlrabi. Lwt 2022, 157, 113078. [Google Scholar] [CrossRef]
- Yu, Y.; Zhao, J.; Liu, J.; Wu, J.; Wang, Z.; Sun, Z. Improving the Function of Pickle Insoluble Dietary Fiber by Coupling Enzymatic Hydrolysis with HHP Treatment. J. Food Sci. Technol. 2022, 59, 4634–4643. [Google Scholar] [CrossRef]
- Yu, Z.; Su, Y.; Zhang, Y.; Zhu, P.; Mei, Z.; Zhou, X.; Yu, H. Potential Use of Ultrasound to Promote Fermentation, Maturation, and Properties of Fermented Foods: A Review. Food Chem. 2021, 357, 129805. [Google Scholar] [CrossRef]
- Liao, X.; Li, J.; Suo, Y.; Chen, S.; Ye, X.; Liu, D.; Ding, T. Multiple Action Sites of Ultrasound on Escherichia coli and Staphylococcus aureus. Food Sci. Hum. Wellness 2018, 7, 102–109. [Google Scholar] [CrossRef]
- Gao, S.; Hemar, Y.; Ashokkumar, M.; Paturel, S.; Lewis, G.D. Inactivation of Bacteria and Yeast Using High-Frequency Ultrasound Treatment. Water Res. 2014, 60, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Rajiuddin, S.M.; Vigre, H.; Musavian, H.S.; Kohle, S.; Krebs, N.; Hansen, T.B.; Gantzer, C.; Schultz, A.C. Inactivation of Hepatitis A Virus and Murine Norovirus on Surfaces of Plastic, Steel and Raspberries Using Steam-Ultrasound Treatment. Food Environ. Virol. 2020, 12, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Luo, Y.; Qi, B.; Luo, J.; Wan, Y. Improving the Hydrolysis Efficiency of Soy Sauce Residue Using Ultrasonic Probe-Assisted Enzymolysis Technology. Ultrason. Sonochem. 2017, 35, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-S.; Yang, C.-Y.; Fang, T.J. Strategic Ultrasound-Induced Stress Response of Lactic Acid Bacteria on Enhancement of β-Glucosidase Activity for Bioconversion of Isoflavones in Soymilk. J. Microbiol. Meth. 2018, 148, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Mai-Prochnow, A.; Clauson, M.; Hong, J.; Murphy, A.B. Gram Positive and Gram Negative Bacteria Differ in Their Sensitivity to Cold Plasma. Sci. Rep. 2016, 6, 38610. [Google Scholar] [CrossRef] [PubMed]
- Pankaj, S.K.; Wan, Z.; Keener, K.M. Effects of Cold Plasma on Food Quality: A Review. Foods 2018, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Hyun, J.-E.; Lee, S.-Y. Blue Light-Emitting Diodes as Eco-Friendly Non-Thermal Technology in Food Preservation. Trends Food Sci. Technol. 2020, 105, 284–295. [Google Scholar] [CrossRef]
- Oh, Y.-J.; Park, Y.-R.; Hong, J.; Lee, D.-Y. Metagenomic, Metabolomic, and Functional Evaluation of Kimchi Broth Treated with Light-Emitting Diodes (LEDs). Metabolites 2021, 11, 472. [Google Scholar] [CrossRef]
- Zhang, S.; Sun, L.; Ju, H.; Bao, Z.; Zeng, X.; Lin, S. Research Advances and Application of Pulsed Electric Field on Proteins and Peptides in Food. Food Res. Int. 2020, 139, 109914. [Google Scholar] [CrossRef]
- Li, J.; Shi, J.; Wang, T.; Huang, X.; Zou, X.; Li, Z.; Zhang, D.; Zhang, W.; Xu, Y. Effects of Pulsed Electric Field Pretreatment on Mass Transfer Kinetics of Pickled Lotus Root (Nelumbo nucifera Gaertn.). Lwt 2021, 151, 112205. [Google Scholar] [CrossRef]
- Coutinho, N.M.; Silveira, M.R.; Guimarães, J.T.; Fernandes, L.M.; Pimentel, T.C.; Silva, M.C.; Borges, F.O.; Fernandes, F.A.N.; Rodrigues, S.; Freitas, M.Q.; et al. Are Consumers Willing to Pay for a Product Processed by Emerging Technologies? The Case of Chocolate Milk Drink Processed by Cold Plasma. Lwt 2021, 138, 110772. [Google Scholar] [CrossRef]
- Jaeger, H.; Knorr, D.; Szabó, E.; Hámori, J.; Bánáti, D. Impact of Terminology on Consumer Acceptance of Emerging Technologies through the Example of PEF Technology. Innov. Food Sci. Emerg. 2015, 29, 87–93. [Google Scholar] [CrossRef]


| Fermented Vegetables | Probiotics | Function | Main Results | Ref. |
|---|---|---|---|---|
| Szechwan-style pickled vegetables | Lactobacillus Plantarum CQPC05 | Inhibits constipation. | Up-regulated the mRNA expression of the stem cell factor receptor (c-Kit and SCF) and glial cell-derived neurotrophic factor genes, down-regulated the transient receptor potential cation channel subfamily V member 1 and inducible nitric oxide synthase. | [79] |
| Kimchi | Lactococcus lactis KC24 | Antimicrobial, anti-inflammatory, antioxidant, anti-cancer. | Listeria monocytogenes and Staphylococcus aureus inhibition. Nitric oxide reduction. Inhibited gastric carcinoma (AGS), colon carcinoma (HT-29 and LoVo), breast carcinoma (MCF-7), and lung carcinoma (SK-MES-1) cells. | [25] |
| Kimchi | Lactobacillus plantarum EM | Lower cholesterol. | Cholesterol was removed by the cell wall fraction of the probiotics under the mechanism of enzymatic assimilation and was cell wall concentration-dependent. | [80] |
| Mango pickle | Bacillus licheniformis KT921419 | Anti-cancer. | Works against the HT-29 colon cancer cell line | [29] |
| Chinese Sauerkraut | Bacillus velezensis T701 | Antitumor. | The lipopeptide iturin A-2 produced by the strain showed good cytotoxic activities against Hela, MCF-7 and BT474 cell lines which related to cervical and breast cancer. | [81] |
| Sauerkraut | Enterococcus | Heavy metal elimination. | Eliminated heavy metals such as Cu, Pb, and Cd that are difficult to eliminate through cooking | [82] |
| Strains | Isolation Origin | Characterization of the Strain and the Main Effects | Ref. |
|---|---|---|---|
| Lactobacillus plantarum GP11 | Homemade pickled samples | Show no biogenic amine production ability. Exhibit antifungal activity against the Aspergillus sp. and Penicillium sp., which always leads to the contamination of the pickled vegetables. | [113] |
| L. brevis SC-2 | Fermented mustard | A lower capacity of biogenic amine-producing ability, 13.95 mg/kg total biogenic amine producing ability with corresponding precursors; did not produce tryptamine, putrescine, and cadaverine, and could reduce the content of the biogenic amine in the fermented mustard from 137.16 mg/kg to 39.16 mg/kg | [107] |
| L. plantarum GZ-2 | Fermented mustard | A lower capacity of biogenic amine-producing ability, 4.65 mg/kg total biogenic amine-producing ability with corresponding precursors, and could reduce the content of the biogenic amine of the fermented mustard. | [107] |
| L. brevis PK08 | Kimchi | Has a multicopper oxidase gene, and showed a high reduction in tyramine content. | [114] |
| Limosilactobacillus fermentum G9 | Cantonese pickles (containing mustard, cabbage, and bamboo shoots) | Has no biogenic amine-producing ability and could significantly reduce the biogenic amine content of Cantonese pickles to nearly 25 mg/kg compared to 150 mg/kg in the naturally fermented sample. | [121] |
| Non-Thermal Technology | Sterilization Effects | Effects on the Sensory Quality of Fermented Vegetables | Ref. |
|---|---|---|---|
| HPP | HPP treatment at 550 MPa for 5 min reduces total plate count (TPC) and substantially inactivates yeast and mold in the pickled radish, and maintained microbial safety of pickles in sixty days of storage. | Might have adverse effects on the sensory quality of the pickled radish, and the treatment parameters should be prioritized. | [146] |
| Maintained the shelf life of the marinated lotus root slices. | HPP treatment could retain the color and improve the flavor of the marinated lotus root slices. | [10] | |
| Cold plasma | Could eliminated 5.00 logCFU/g of microorganisms under the CP treatment (voltage 60 kV, frequency 50 Hz, implementing time 60 s) | Increase the firmness of the radish paocai, could alleviate the softening and browning of radish paocai. | [147] |
| Plasma activated water, generated by an AC bi-polar pulsed power supply (driving frequency 14.3 kHz, a peak-to-peak voltage 18 kV) for 120 s, could cause a reduction of 2.0, 2.2, 1.8, 0.9 log CFU/g mesophilic aerobic bacteria, lactic acid bacteria, yeast and moulds of ready-to-use shredded, salted kimchi. | Could reduce the salinity of peroxidase activity of the product. | [148] | |
| Photodynamic | Could inhibit the while colony-forming yeast in kimchi seasoning | Maintain the volatile compounds in the kimchi seasoning | [149] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, X.; Cui, F.; Wang, D.; Lv, X.; Li, X.; Li, J. Fermented Vegetables: Health Benefits, Defects, and Current Technological Solutions. Foods 2024, 13, 38. https://doi.org/10.3390/foods13010038
Tan X, Cui F, Wang D, Lv X, Li X, Li J. Fermented Vegetables: Health Benefits, Defects, and Current Technological Solutions. Foods. 2024; 13(1):38. https://doi.org/10.3390/foods13010038
Chicago/Turabian StyleTan, Xiqian, Fangchao Cui, Dangfeng Wang, Xinran Lv, Xuepeng Li, and Jianrong Li. 2024. "Fermented Vegetables: Health Benefits, Defects, and Current Technological Solutions" Foods 13, no. 1: 38. https://doi.org/10.3390/foods13010038
APA StyleTan, X., Cui, F., Wang, D., Lv, X., Li, X., & Li, J. (2024). Fermented Vegetables: Health Benefits, Defects, and Current Technological Solutions. Foods, 13(1), 38. https://doi.org/10.3390/foods13010038






