Evaluation of Recovery Methods for Fragaria vesca L. Oil: Characteristics, Stability and Bioactive Potential
Abstract
1. Introduction
2. Materials and Methods
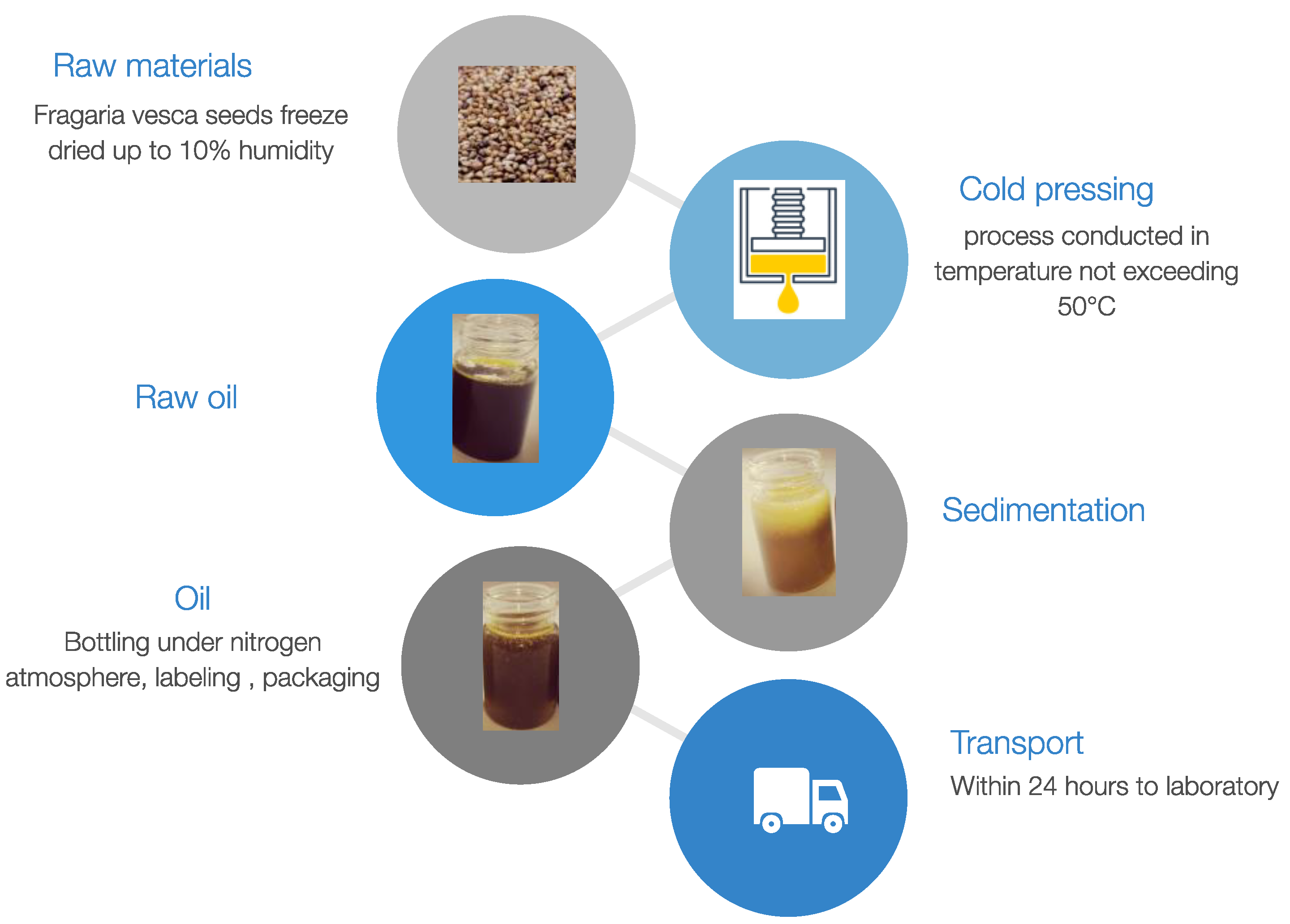
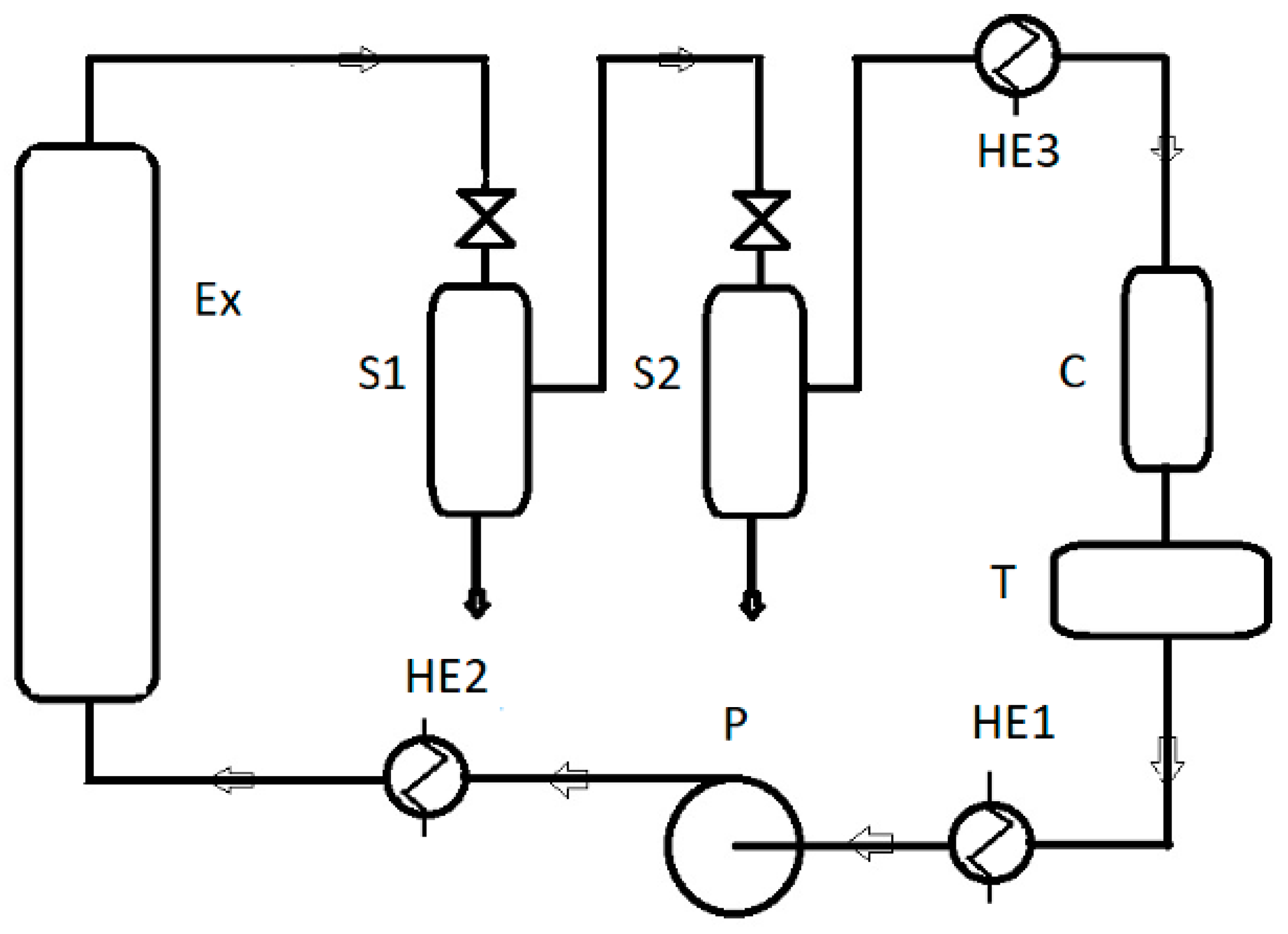
2.1. Samples
2.2. Oils’ Characteristics
2.2.1. Fatty Acid Composition
2.2.2. Antiradical Capacity
2.2.3. Quality Parameters
2.2.4. Phytosterol and Squalene Contents
2.2.5. Tocopherol and Carotenoid Contents
2.2.6. Polyphenolic Extraction and Analysis
2.2.7. Oxidative Stability Studies
2.3. Cytotoxicity Assays
2.3.1. Cell Culture
2.3.2. Viability Measurements with the MTT Method
2.3.3. Cytotoxicity Measurements with the LDH Leakage Assay
2.3.4. Viability/Proliferative Ability Measurements with the SRB Assay (Total Protein Content)
2.4. Statistical Analysis
3. Results
3.1. Oils’ Chemical Characteristics
3.1.1. Fatty Acid Composition
3.1.2. DPPH Radical Scavenging Activity of WSOs
3.1.3. Quality Parameters
3.1.4. Phytosterol and Squalene Content
3.1.5. Tocopherol and Carotenoid Contents
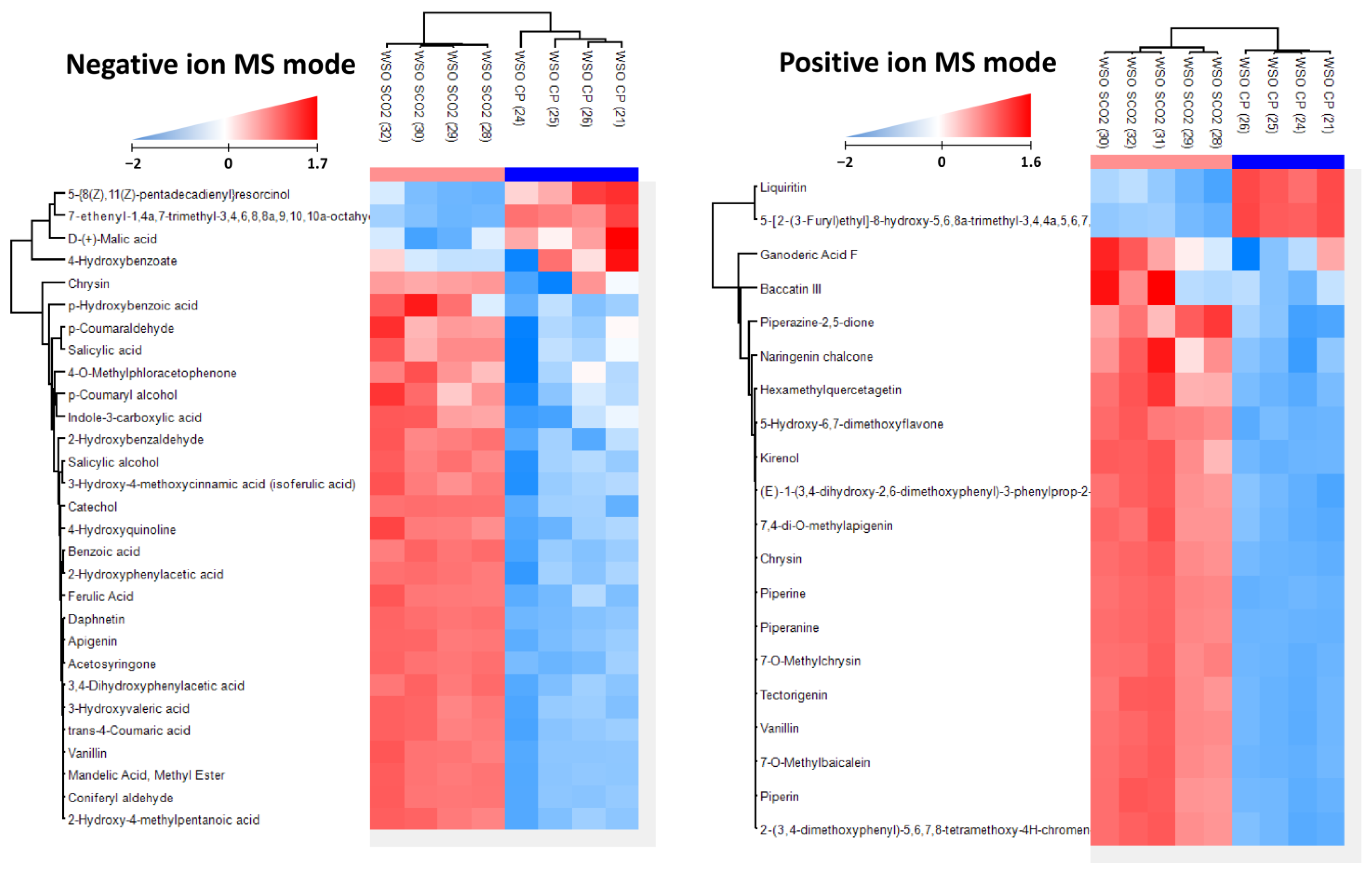
3.1.6. Polyphenol Content and Composition
3.1.7. Oxidative Stability Studies
3.2. In Vitro Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ulrich, D.; Olbricht, K. Diversity of volatile patterns in sixteen Fragaria vesca L. accessions in comparison to cultivars of Fragaria× ananassa. J. Appl. Bot. Food Qual. 2013, 86, 37–46. [Google Scholar] [CrossRef]
- Gasperotti, M.; Masuero, D.; Mattivi, F.; Vrhovsek, U. Overall dietary polyphenol intake in a bowl of strawberries: The influence of Fragaria spp. in nutritional studies. J. Funct. Foods 2015, 18, 1057–1069. [Google Scholar] [CrossRef]
- Dyduch-Siemińska, M.; Najda, A.; Dyduch, J.; Gantner, M.; Klimek, K. The content of secondary metabolites and antioxidant activity of wild strawberry fruit (Fragaria vesca L.). J. Anal. Methods Chem. 2015, 2015, 831238. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.; Kallio, H. Characterization of seed oils of wild, edible Finnish berries. Z. Für Lebensm. Und-Forsch. A 1997, 204, 300–307. [Google Scholar] [CrossRef]
- Grajzer, M.; Wiatrak, B.; Gębarowski, T.; Boba, A.; Rój, E.; Gorczyca, D.; Prescha, A. Bioactive Compounds of Raspberry Oil Emulsions Induced Oxidative Stress via Stimulating the Accumulation of Reactive Oxygen Species and NO in Cancer Cells. Oxid. Med. Cell. Longev. 2021, 2021, 5561672. [Google Scholar] [CrossRef]
- Grajzer, M.; Wiatrak, B.; Gębarowski, T.; Matkowski, A.; Grajeta, H.; Rój, E.; Kulma, A.; Prescha, A. Chemistry, oxidative stability and bioactivity of oil extracted from Rosa rugosa (Thunb.) seeds by supercritical carbon dioxide. Food Chem. 2021, 335, 127649. [Google Scholar] [CrossRef]
- Tereshchuk, L.; Starovoytova, K.; Babich, O.; Dyshlyuk, L.; Sergeeva, I.; Pavsky, V.; Ivanova, S.; Prosekov, A. Sea buckthorn and rosehip oils with chokeberry extract to prevent hypercholesterolemia in mice caused by a high-fat diet in vivo. Nutrients 2020, 12, 2941. [Google Scholar] [CrossRef]
- Guici El Kouacheur, K.; Cherif, H.S.; Saidi, F.; Bensouici, C.; Fauconnier, M.L. P runus amygdalus var. amara (bitter almond) seed oil: Fatty acid composition, physicochemical parameters, enzyme inhibitory activity, antioxidant and anti-inflammatory potential. J. Food Meas. Charact. 2022, 17, 371–384. [Google Scholar] [CrossRef]
- Rasor, A.S.; Duncan, S.E. Fats and Oils—Plant Based. In Food Processing; John Wiley & Sons, Ltd.: Oxford, UK, 2014; pp. 457–480. [Google Scholar] [CrossRef]
- Vavpot, V.J.; Williams, R.J.; Williams, M.A. Extrusion/Expeller® pressing as a means of processing green oils and meals. In Green Vegetable Oil Processing; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar] [CrossRef]
- Prescha, A.; Świędrych, A.; Biernat, J.; Szopa, J. Increase in Lipid Content in Potato Tubers Modified by 14-3-3 Gene Overexpression. J. Agric. Food Chem. 2001, 49, 3638–3643. [Google Scholar] [CrossRef] [PubMed]
- Tuberoso, C.I.G.; Kowalczyk, A.; Sarritzu, E.; Cabras, P. Determination of antioxidant compounds and antioxidant activity in commercial oilseeds for food use. Food Chem. 2007, 103, 1494–1501. [Google Scholar] [CrossRef]
- Espín, J.C.; Soler-Rivas, C.; Wichers, H.J. Characterization of the total free radical scavenger capacity of vegetable oils and oil fractions using 2, 2-diphenyl-1-picrylhydrazyl radical. J. Agric. Food Chem. 2000, 48, 648–656. [Google Scholar] [CrossRef]
- Shukla, V.; Dutta, P.; Artz, W. Camelina oil and its unusual cholesterol content. J. Am. Oil Chem. Soc. 2002, 79, 965–969. [Google Scholar] [CrossRef]
- Fromm, M.; Bayha, S.; Kammerer, D.R.; Carle, R. Identification and quantitation of carotenoids and tocopherols in seed oils recovered from different Rosaceae species. J. Agric. Food Chem. 2012, 60, 10733–10742. [Google Scholar] [CrossRef] [PubMed]
- Pirisi, F.M.; Angioni, A.; Cabras, P.; Garau, V.L.; di Teulada, M.T.S.; dos Santos, M.K.; Bandino, G. Phenolic compounds in virgin olive oils I. Low-wavelength quantitative determination of complex phenols by high-performance liquid chromatography under isocratic elution. J. Chromatogr. A 1997, 768, 207–213. [Google Scholar] [CrossRef]
- Balon, K.; Wiatrak, B. PC12 and THP-1 cell lines as neuronal and microglia model in neurobiological research. Appl. Sci. 2021, 11, 3729. [Google Scholar] [CrossRef]
- Bopp, S.K.; Lettieri, T. Comparison of four different colorimetric and fluorometric cytotoxicity assays in a zebrafish liver cell line. BMC Pharmacol. 2008, 8, 1–11. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Lipid oxidation and improving the oxidative stability. Chem. Soc. Rev. 2010, 39, 4067–4079. [Google Scholar] [CrossRef]
- Milala, J.; Grzelak-Błaszczyk, K.; Sójka, M.; Kosmala, M.; Dobrzyńska-Inger, A.; Rój, E. Changes of bioactive components in berry seed oils during supercritical CO2 extraction. J. Food Process. Preserv. 2018, 42, e13368. [Google Scholar] [CrossRef]
- Yang, B.; Ahotupa, M.; Määttä, P.; Kallio, H. Composition and antioxidative activities of supercritical CO2-extracted oils from seeds and soft parts of northern berries. Food Res. Int. 2011, 44, 2009–2017. [Google Scholar] [CrossRef]
- Pieszka, M.; Tombarkiewicz, B.; Roman, A.; Migdał, W.; Niedziółka, J. Effect of bioactive substances found in rapeseed, raspberry and strawberry seed oils on blood lipid profile and selected parameters of oxidative status in rats. Environ. Toxicol. Pharmacol. 2013, 36, 1055–1062. [Google Scholar] [CrossRef]
- Rombaut, N.; Savoire, R.; Thomasset, B.; Bélliard, T.; Castello, J.; Van Hecke, É.; Lanoisellé, J.-L. Grape seed oil extraction: Interest of supercritical fluid extraction and gas-assisted mechanical extraction for enhancing polyphenol co-extraction in oil. Comptes Rendus Chim. 2014, 17, 284–292. [Google Scholar] [CrossRef]
- Krstić, Đ.D.; Ristivojević, P.M.; Gašić, U.M.; Lazović, M.; Akšić, M.M.F.; Milivojević, J.; Morlock, G.E.; Milojković-Opsenica, D.M.; Trifković, J.Đ. Authenticity assessment of cultivated berries via phenolic profiles of seeds. Food Chem. 2023, 402, 134184. [Google Scholar] [CrossRef] [PubMed]
- Nyam, K.L.; Tan, C.P.; Lai, O.M.; Long, K.; Che Man, Y.B. Optimization of supercritical CO 2 extraction of phytosterol-enriched oil from Kalahari melon seeds. Food Bioproc. Technol. 2011, 4, 1432–1441. [Google Scholar] [CrossRef]
- Abbasi, H.; Rezaei, K.; Emamdjomeh, Z.; Mousavi, S.M.E. Effect of various extraction conditions on the phenolic contents of pomegranate seed oil. Eur. J. Lipid Sci. Technol. 2008, 110, 435–440. [Google Scholar] [CrossRef]
- Bai, G.; Ma, C.; Chen, X. Phytosterols in edible oil: Distribution, analysis and variation during processing. Grain Oil Sci. Technol. 2021, 4, 33–44. [Google Scholar] [CrossRef]
- Wejnerowska, G.; Heinrich, P.; Gaca, J. Separation of squalene and oil from Amaranthus seeds by supercritical carbon dioxide. Sep. Purif. Technol. 2013, 110, 39–43. [Google Scholar] [CrossRef]
- Van Hoed, V.; De Clercq, N.; Echim, C.; Andjelkovic, M.; Leber, E.; Dewettinck, K.; Verhé, R. Berry seeds: A source of specialty oils with high content of bioactives and nutritional value. J. Food Lipids 2009, 16, 33–49. [Google Scholar] [CrossRef]
- Farhoosh, R. Reliable determination of the induction period and critical reverse micelle concentration of lipid hydroperoxides exploiting a model composed of pseudo-first and-second order reaction kinetics. Lwt 2018, 98, 406–410. [Google Scholar] [CrossRef]
- Grajzer, M.; Szmalcel, K.; Kuźmiński, Ł.; Witkowski, M.; Kulma, A.; Prescha, A. Characteristics and Antioxidant Potential of Cold-Pressed Oils—Possible Strategies to Improve Oil Stability. Foods 2020, 9, 1630. [Google Scholar] [CrossRef]
- Farhoosh, R. Initiation and propagation kinetics of inhibited lipid peroxidation. Sci. Rep. 2021, 11, 6864. [Google Scholar] [CrossRef]
- Scheers, E.; Ekwall, B.; Dierickx, P. In vitro long-term cytotoxicity testing of 27 MEIC chemicals on Hep G2 cells and comparison with acute human toxicity data. Toxicol. In Vitro 2001, 15, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, J. Modulating effect of fatty acids and sterols on skin aging. J. Funct. Foods 2019, 57, 135–140. [Google Scholar] [CrossRef]
- Skorkowska-Telichowska, K.; Hasiewicz-Derkacz, K.; Gębarowski, T.; Kulma, A.; Moreira, H.; Kostyn, K.; Gębczak, K.; Szyjka, A.; Wojtasik, W.; Gąsiorowski, K. Emulsions made of oils from seeds of GM flax protect V79 cells against oxidative stress. Oxid. Med. Cell. Longev. 2016, 2016, 7510759. [Google Scholar] [CrossRef]
- McIntyre, B.S.; Briski, K.P.; Tirmenstein, M.A.; Fariss, M.W.; Gapor, A.; Sylvester, P.W. Antiproliferative and apoptotic effects of tocopherols and tocotrienols on normal mouse mammary epithelial cells. Lipids 2000, 35, 171. [Google Scholar] [CrossRef] [PubMed]
- Bruggisser, R.; von Daeniken, K.; Jundt, G.; Schaffner, W.; Tullberg-Reinert, H. Interference of plant extracts, phytoestrogens and antioxidants with the MTT tetrazolium assay. Planta Med. 2002, 68, 445–448. [Google Scholar] [CrossRef]
- Dinse, G.E. An em algorithm for fitting a four-parameter logistic model to binary dose-response data. J. Agric. Biol. Environ. Stat. 2011, 16, 221–232. [Google Scholar] [CrossRef]
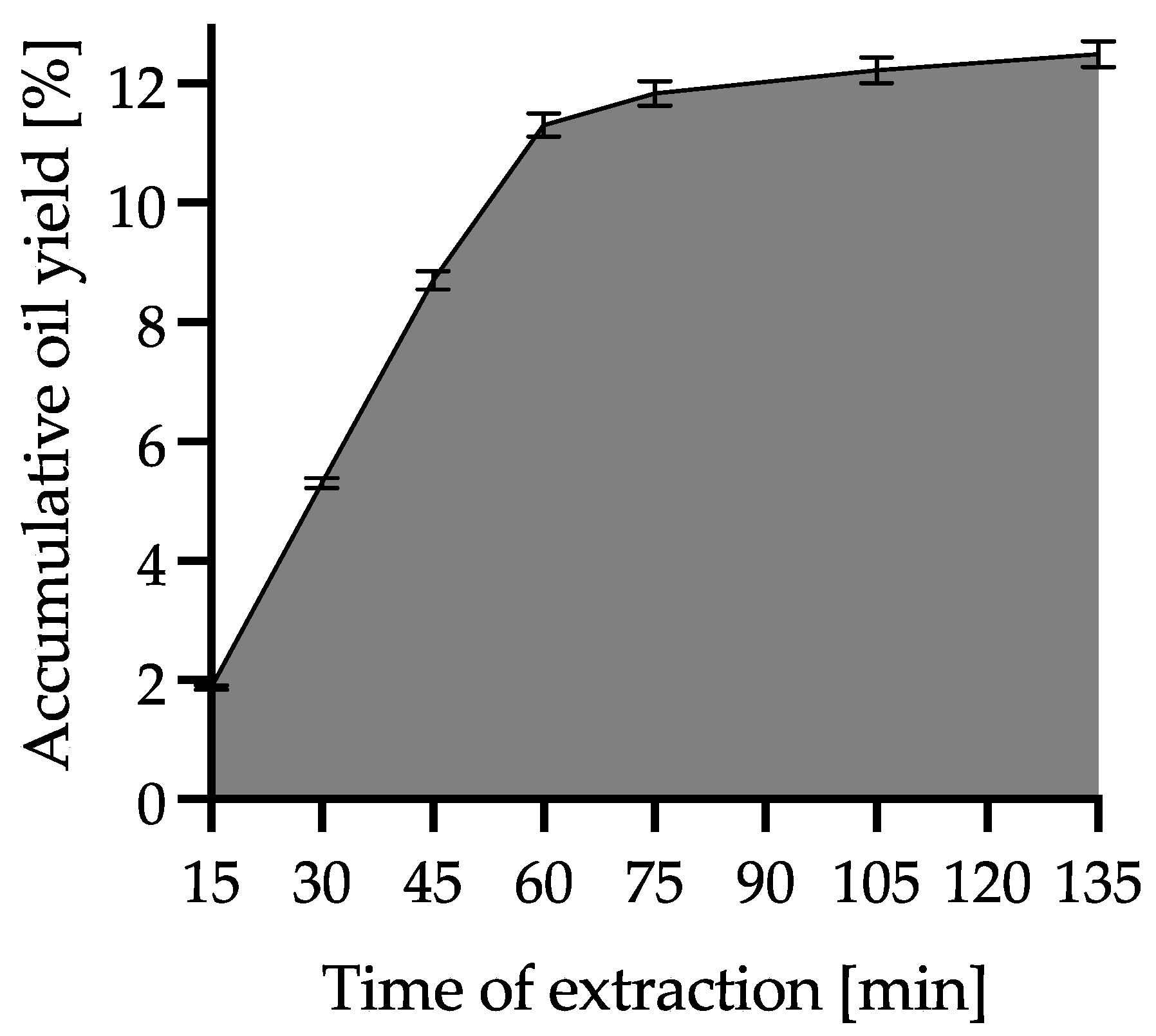
| SCO2E Parameters | |
|---|---|
| Time of extraction [min] | 135 |
| Pressure of extraction [bar] | 300 |
| Temperature of extraction [°C] | 40 |
| Batch weight [g] | 9850 |
| CO2 pump flow [kg/h] | 130 |
| WSO | ||||||||
|---|---|---|---|---|---|---|---|---|
| Basic Characteristics | CP | SCO215 | SCO230 | SCO245 | SCO260 | SCO275 | SCO2105 | SCO2135 |
| Fatty acids [%] | ||||||||
| C18:0 | 2.756 ± 0.016 a | 2.040 ± 0.051 b | 1.91 ± 0.004 c | 1.972 ± 0.118 b | 2.606 ± 0.022 d | 2.746 ± 0.041 a | 2.622 ± 0.012 d | 2.571 ± 0.045 d |
| C18:1 n-9 | 17.517 ± 0.014 a | 14.502 ± 0.422 bd | 12.714 ± 0.017 c | 12.784 ± 0.167 c | 14.125 ± 0.105 b | 14. 783 ± 0.202 d | 14.386 ± 0.199 bd | 14.143 ± 0.082 b |
| C18:2 n-6 | 47.769 ± 0.025 a | 41.060 ± 0.145 b | 40.973 ± 0.73 b | 40.776 ± 0.138 c | 40.195 ± 0.023 de | 40.066 ± 0.023 d | 40.205 ± 0.075 de | 40.327 ± 0.061 e |
| C18:3 n-3 | 30.840 ± 0.108 a | 41.296 ± 0.390 b | 43.304 ± 0.105 c | 43.124 ± 0.152 c | 41.218 ± 0.112 b | 40.04 ± 0.23 d | 40.748 ± 0.158 e | 41.080 ± 0.121 be |
| PUFA | 78.615 ± 0.194 a | 82.358 ± 0.527 b | 84.277 ± 0.172 c | 84.151 ± 0.278 c | 81.414 ± 0.162 d | 80.024± 0.298 e | 80.954 ± 0.17 d | 81.325± 0.084 d |
| n-6/n-3 | 1.548 ± 0.007 a | 0.994 ± 0.006 be | 0.946 ± 0.001 c | 0.946 ± 0.001 c | 0.975 ± 0.003 d | 1.003 ± 0.006 b | 0.987 ± 0.002 e | 0.984 ± 0.001 de |
| Antiradical capacity [mM TEAC/kg] | ||||||||
| Oil | 1.031 ± 0.059 a | 1.243 ± 0.036 b | 1.255 ± 0.041 b | 0.919 ± 0.024 c | 0.767 ± 0.013 d | 1.210 ±0.029 b | 1.247 ± 0.020 b | 1.227 ± 0.010 b |
| LP | 0.775 ± 0.075 ad | 1.007 ± 0.060 b | 1.276 ± 0.050 c | 0.960 ± 0.031 b | 0.620 ± 0.027 a | 0.918 ± 0.150 bd | 0.891 ± 0.094 bd | 0.861 ± 0.085 bd |
| HP | 0.369 ± 0.030 a | 0.442 ± 0.027 be | 0.563 ± 0.027 c | 0.195 ± 0.008 d | 0.251 ± 0.054 d | 0.476 ± 0.025 b | 0.420 ± 0.031 abe | 0.404 ± 0.056 ae |
| Quality parameters | ||||||||
| Acid Value (AV) | 1.010 ± 0.014 ab | 1.288 ± 0.018 a | 0.838 ± 0.004 ab | 0.682 ± 0.004 b | 2.227 ± 0.003 c | 6.485 ± 0.238 d | 8.846 ± 0.445 e | 9.404 ± 0.506 f |
| [mg KOH/g] | ||||||||
| Peoxide Value (PV) | 1.678 ± 0.027 a | 1.322 ± 0.027 a | 2.960 ± 0.089 b | 1.352 ± 0.004 a | 8.768 ± 0.001 c | 7.963 ± 0.573 d | 12.910 ± 0.368 e | 11.655 ± 0.493 f |
| [mEq O2/Kg] |
| WSO | ||||
|---|---|---|---|---|
| Minor Compounds [mg/kg] | CP | SCO215 | SCO230 | SCO245 |
| Phytosterols | ||||
| Campesterol | 117.38 ± 24.01 ac | 86.50 ± 14.21 a | 175.29 ± 11.72 c | 206.64 ± 72.47 b |
| Stigmasterol | 25.46 ± 3.55 a | 20.24 ± 2.53 b | ND c | 18.20 ± 1.01 b |
| β-Sitosterol | 3511.26± 179.39 a | 3382.36 ± 143.39 a | 5106.712 ± 478.57 b | 4252.24 ± 56.20 c |
| Δ5-Avenasterol | 133.2 ± 15.97 a | 86.40 ± 8.21 b | 114.00 ± 9.78 ba | 135.36 ± 5.48 ba |
| Total | 3787.31 ± 214.57 a | 3575.50 ± 143.56 a | 5396.100 ± 457.08 b | 4574.74 ± 177.50 c |
| Squalene | 31.4 ± 3.33 a | 23.13 ± 4.60 b | 11.12 ± 0.36 c | 12.71 ± 2.10 c |
| Tocopherols | ||||
| α-Tocopherol | 451.99 ± 179.57 a | 174.78 ± 7.26 b | 168.45 ± 33.55 b | 34.45 ± 4.46 b |
| γ-Tocopherol | 466.68 ± 169.7 a | 859.48 ± 107.07 b | 985.25 ± 210.36 b | 205.21 ± 43.03 a |
| δ-Tocopherol | 299.78 ± 60.79 a | 792.74 ± 68.69 b | 771.23 ± 93.96 b | 142.81 ± 26.43 a |
| β-Tocopherol | ND | 73.97 ± 12.56 b | 77.28 ± 19.82 b | 19.33 ± 2.09 c |
| Total | 1218.45 ± 408.14 abc | 1900.98 ± 79.10 c | 1795.08 ± 357.69 c | 401.80 ± 76.01 ab |
| Carotenoids | ||||
| all-trans-β-Carotene | 55.15 ± 12.67 a | 53.20 ± 14.78 a | 27.24 ± 7.55 b | 24.17 ± 2.04 b |
| all-trans-Lutein | 14.4 ± 2.73 a | 10.13 ± 1.53 b | 5.413 ± 1.41 c | 3.23 ± 0.29 c |
| all-trans-Zeaxanthin | 39.88 ± 0.37 a | 39.52 ± 0.14 a | 39.43 ± 0.23 b | 39.07 ± 0.01 b |
| all-trans-α-Cryptoxanthin | 14.36 ± 0.07 | ND | ND c | ND |
| Total | 123.78 ± 15.09 a | 102.85 ± 16.18 a | 77.96 ± 9.19 b | 66.47 ± 2.34 b |
| Total Polyphenols | 15.12 ± 1.40 a | 58.45 ± 2.58 a | 40.48 ± 1.93 b | 45.32 ±0.06 b |
| Temperature Applied | Oxidation Parameter | WSO | |||
|---|---|---|---|---|---|
| CP | SCO215 | SCO230 | SCO245 | ||
| Isothermal conditions, time [min] | |||||
| 100 °C | OIT | 386.07 ± 29.34 a | 225.66 ± 23.03 b | 443.92 ± 44.95 c | 6.85 ± 0.44 d |
| Propagation | 96.78 ± 5.58 a | 86.97 ± 5.18 b | 72.01 ± 5.07 c | 58.89 ± 3.11 c | |
| 110 °C | OIT | 134.21± 4.36 a | 128.66 ± 9.02 b | 184.76 ± 7.11 c | 6.86 ± 0.44 d |
| Propagation | 59.68 ± 3.31 a | 55.67 ± 15.43 b | 70.16 ± 9.96 c | 58.90 ± 3.10 a | |
| 120 °C | OIT | 84.23 ± 2.83 a | 61.12 ± 3.61 b | 84.05 ± 7.40 a | 0.09 ± 0.01 c |
| Propagation | 15.17 ± 3.56 a | 12.07 ± 0.97 a | 22.94 ± 2.05 b | 19.90 ± 2.18 b | |
| Ea [kJ/mol] | 100.41 ± 5.85 a | 85.23 ± 12.70 b | 105.00 ± 5.91 a | ND | |
| Non-isothermal conditions, temperature [°C] | |||||
| Rate 5 °C/min | Ton | 164.42 ± 1.32 a | 157.57 ± 3.96 b | 167.33 ± 0.68 a | 144.28 ± 1.54 c |
| Propagation | 157.69 ± 2.58 ab | 161.53 ± 3.26 a | 153.21 ± 1.01 b | 177.84 ± 1.99 c | |
| Rate 10 °C/min | Ton | 174.03 ± 0.58 a | 167.92 ± 3.19 a | 175.46 ± 5.08 a | 145.86 ± 8.97 b |
| Propagation | 157.15 ± 1.46 ab | 159.95 ± 3.42 ab | 151.79 ± 1.16 a | 170.50 ± 15.11 b | |
| Rate 15 °C/min | Ton | 184.58 ± 1.77 a | 181.99 ± 3.81 a | 185.85 ± 2.01 a | 164.56 ± 5.72 b |
| Propagation | 153.43 ± 2.11 a | 88.04 ± 7.26 b | 151.32 ± 2.43 a | 169.40 ± 2.70 c | |
| WSO | MTT Assay | LDH Assay | SRB Assay |
|---|---|---|---|
| CC50 ± SD (%) | |||
| CP | Non-toxic | Non-toxic | Non-toxic |
| SCO215 | 3.35 ± 0.67% | Non-toxic | Non-toxic |
| SCO230 | 5.49 ± 0.91% | Non-toxic | Non-toxic |
| SCO245 | Non-toxic | Non-toxic | Non-toxic |
| WSO | Description | Grade |
|---|---|---|
| CP (5%) | Discrete intracytoplasmic granules, no cell lysis | 0 |
| SCO215 (5%) | Around 15% of the cells are round and loosely attached, with culture density lower than the blank sample | 1 |
| SCO230 (5%) | Around 5–10% of the cells are round and loosely attached, with culture density lower than the blank sample | 1 |
| SCO245 (5%) | Discrete intracytoplasmic granules, no cell lysis | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grajzer, M.; Wiatrak, B.; Jawień, P.; Marczak, Ł.; Wojakowska, A.; Wiejak, R.; Rój, E.; Grzebieluch, W.; Prescha, A. Evaluation of Recovery Methods for Fragaria vesca L. Oil: Characteristics, Stability and Bioactive Potential. Foods 2023, 12, 1852. https://doi.org/10.3390/foods12091852
Grajzer M, Wiatrak B, Jawień P, Marczak Ł, Wojakowska A, Wiejak R, Rój E, Grzebieluch W, Prescha A. Evaluation of Recovery Methods for Fragaria vesca L. Oil: Characteristics, Stability and Bioactive Potential. Foods. 2023; 12(9):1852. https://doi.org/10.3390/foods12091852
Chicago/Turabian StyleGrajzer, Magdalena, Benita Wiatrak, Paulina Jawień, Łukasz Marczak, Anna Wojakowska, Rafał Wiejak, Edward Rój, Wojciech Grzebieluch, and Anna Prescha. 2023. "Evaluation of Recovery Methods for Fragaria vesca L. Oil: Characteristics, Stability and Bioactive Potential" Foods 12, no. 9: 1852. https://doi.org/10.3390/foods12091852
APA StyleGrajzer, M., Wiatrak, B., Jawień, P., Marczak, Ł., Wojakowska, A., Wiejak, R., Rój, E., Grzebieluch, W., & Prescha, A. (2023). Evaluation of Recovery Methods for Fragaria vesca L. Oil: Characteristics, Stability and Bioactive Potential. Foods, 12(9), 1852. https://doi.org/10.3390/foods12091852







