Study of Physico-Chemical Properties of Dough and Wood Oven-Baked Pizza Base: The Effect of Leavening Time
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Pizza Dough Ingredients
2.2. Dough and Pizza Making
2.3. Image Analysis of Leavening Kinetics
2.4. Stress-Relaxation Test
2.5. Elastic Modulus Determination
2.6. Thermal Analysis
2.7. Reducing Sugars, Free Amino Groups, and Acrylamide Analysis
2.8. In Vitro Digestible and Resistant Starch
2.9. SEM of Dough and Pizza Samples
2.10. Statistical Analysis
3. Results and Discussion
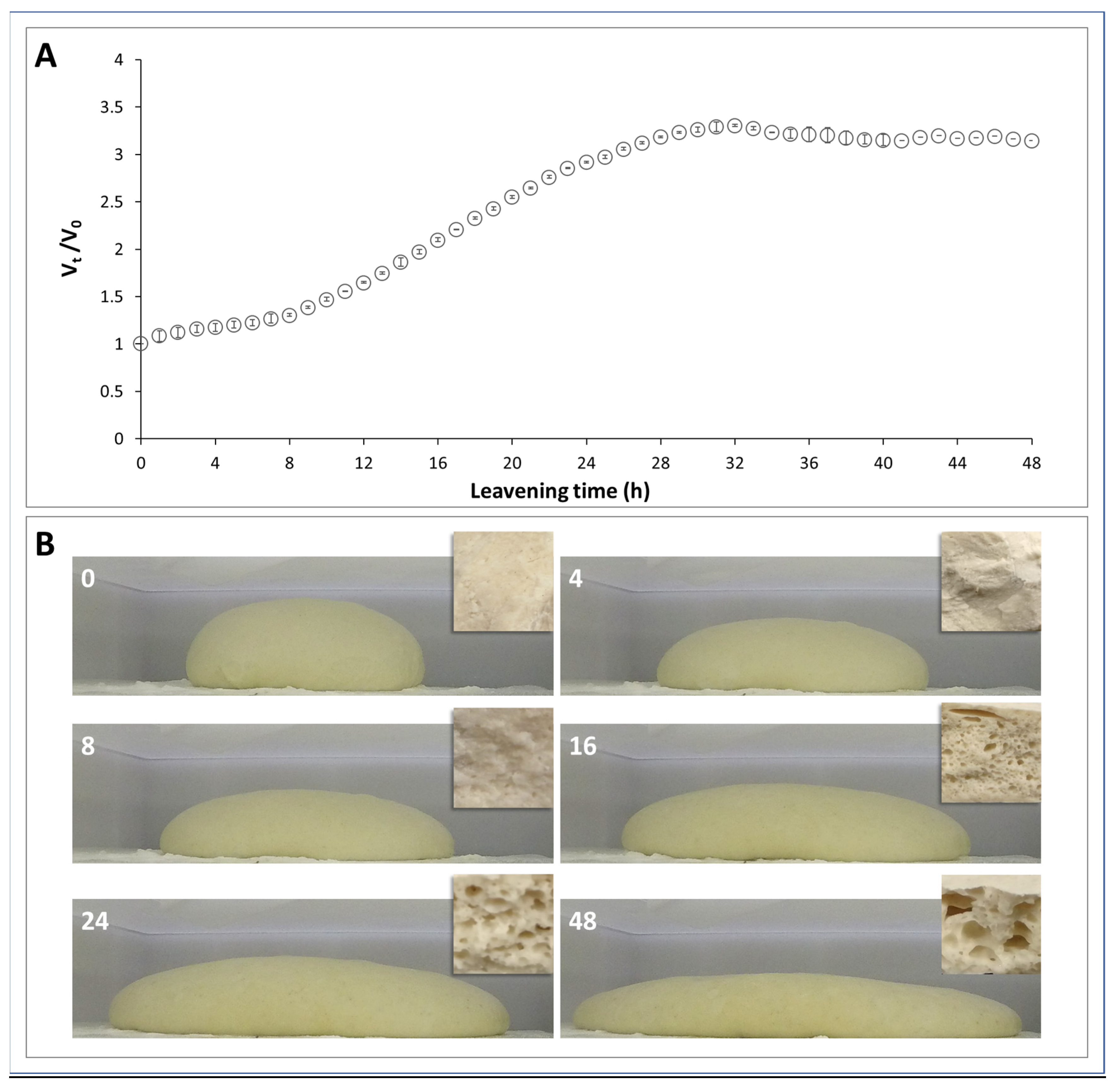
3.1. Dough Volume during Leavening
3.2. Rheological Properties of Pizza Dough
3.3. Thermal Properties
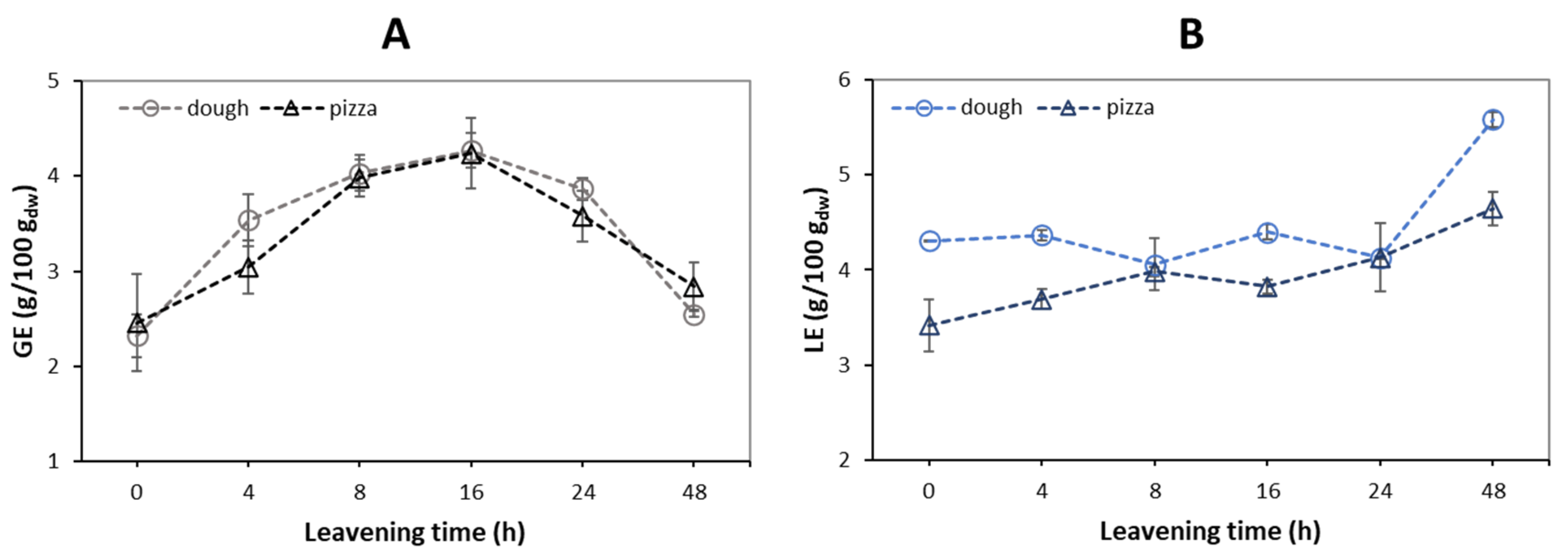
3.4. Reducing Sugars, Free Amino Groups, and Digestible Starch in Dough and Pizza
3.5. Acrylamide Levels in Pizza Base Leavened for Different Times
3.6. Morphology of Dough and Pizza Base during Leavening
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Commission Regulation (EU) 97/2010. Entering a name in the register of traditional specialities guaranteed (Pizza Napoletana (TSG). Off. J. Eur. Union 2010, 34, 7–13. [Google Scholar]
- Ganguly, M. Naples’ Pizza Spinning Given UNESCO ‘Intangible Heritage’ Status. 2017. Available online: http://edition.cnn.com/2017/12/07/europe/naples-unesco-pizza-intl/index.html (accessed on 25 March 2023).
- Dini, I.; Izzo, L.; Graziani, G.; Ritieni, A. The Nutraceutical Properties of “Pizza Napoletana Marinara TSG” a traditional food rich in bioaccessible antioxidants. Antioxidants 2021, 10, 495. [Google Scholar] [CrossRef]
- Caporaso, N.; Panariello, V.; Sacchi, R. The “True” Neapolitan Pizza: Assessing the influence of extra virgin olive oil on pizza volatile compounds and lipid oxidation. J. Culin. Sci. Technol. 2015, 13, 29–48. [Google Scholar] [CrossRef]
- Piscopo, A.; Zappia, A.; Mincione, A.; Silletti, R.; Summo, C.; Pasqualone, A. Effect of oil type used in Neapolitan pizza TSG topping on its physical, chemical, and sensory properties. Foods 2023, 12, 41. [Google Scholar] [CrossRef]
- Iaccarino, I.O.; Russo, O.; Iacone, R.; D’Elia, L.; Giacco, R.; Volpe, M.G.; Strazzullo, P. A lower sodium Neapolitan pizza prepared with seawater in place of salt: Nutritional properties, sensory characteristics, and metabolic effects. Nutrients 2020, 12, 3533. [Google Scholar] [CrossRef] [PubMed]
- Falciano, A.; Sorrentino, A.; Masi, P.; Di Pierro, P. Development of Functional Pizza Base Enriched with Jujube (Ziziphus jujuba) Powder. Foods 2022, 11, 1458. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Barros, L.; Calhelha, R.C.; Carocho, M.; Ćirić, A.; Sokovic, M.; Dias, M.M.; Santos-Buelga, C.; Barreiro, M.F.; Ferreira, I.C. Tarragon phenolic extract as a functional ingredient for pizza dough: Comparative performance with ascorbic acid (E300). J. Funct. Foods 2016, 26, 268–278. [Google Scholar] [CrossRef]
- Covino, C.; Sorrentino, A.; Di Pierro, P.; Aiello, A.; Romano, R.; Masi, P. Asparaginase enzyme reduces acrylamide levels in fried and wood oven baked pizza base. Food Chem. Adv. 2023, 2, 100206. [Google Scholar] [CrossRef]
- Falciano, A.; Moresi, M.; Masi, P. Phenomenology of Neapolitan Pizza Baking in a Traditional Wood-Fired Oven. Foods 2023, 12, 890. [Google Scholar] [CrossRef]
- Ciarmiello, M.; Morrone, B. Why not using electric ovens for Neapolitan pizzas? A thermal analysis of a high temperature electric pizza oven. Energy Procedia 2016, 101, 1010–1017. [Google Scholar] [CrossRef]
- Dey, S.; Maurya, C.; Hettiarachchy, N.; Seo, H.-S.; Zhou, W. Textural characteristics and color analyses of 3D printed gluten-free pizza dough and crust. J. Food Sci. Technol. 2023, 60, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Janssen, A.M.; van Vliet, T.; Vereijken, J.M. Fundamental and empirical rheological behaviour of wheat flour doughs and comparison with bread making performance. J. Cereal Sci. 1996, 23, 43–54. [Google Scholar] [CrossRef]
- Masi, P.; Romano, A.; Coccia, E. The Artisanal Production Process of Neapolitan Pizza, in The Neapolitan Pizza: A Scientific Guide Bout the Artisanal Process; Doppia Voce: Naples, Italy, 2015; pp. 67–96. [Google Scholar]
- Marshall, W.E.; Chrastil, J. Interaction of food proteins with starch. Biochem. Food Prot. 1992, 3, 75–97. [Google Scholar] [CrossRef]
- Song, Y.; Zheng, Q. Dynamic rheological properties of wheat flour dough and proteins. Trends Food Sci. Technol. 2007, 18, 132–138. [Google Scholar] [CrossRef]
- Srikaeo, K.; Furst, J.E.; Ashton, J.F.; Hosken, R.W. Microstructural changes of starch in cooked wheat grains as affected by cooking temperatures and times. LWT-Food Sci. Technol. 2006, 39, 528–533. [Google Scholar] [CrossRef]
- Peleg, M. Characterisation of the stress relaxation curves of solid foods. J. Food Sci. 1979, 44, 277–281. [Google Scholar] [CrossRef]
- Masi, P. Study of the influence of temperature on the rheological behaviour of gluten by means of dynamic mechanical analysis. In Food Properties and Computer-Aided Engineering of Food Processing Systems; Singh, R.P., Medina, A.G., Eds.; NATO Science Series E (NSSE); Springer: Dordrecht, The Netherlands, 1989; Volume 168, pp. 357–363. [Google Scholar] [CrossRef]
- Zhang, Y.; Hong, T.; Yu, W.; Yang, N.; Jin, Z.; Xu, X. Structural, thermal and rheological properties of gluten dough: Comparative changes by dextran, weak acidification and their combination. Food Chem. 2020, 330, 127154. [Google Scholar] [CrossRef]
- Lapčíková, B.; Burešová, I.; Lapčík, L.; Dabash, V.; Valenta, T. Impact of particle size on wheat dough and bread characteristics. Food Chem. 2019, 297, 124938. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Lee, K.; Drescher, D.G. Fluorometric amino-acid analysis with o-phthaldialdehyde (OPA). Int. J. Biochem. 1978, 9, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Englyst, H.N.; Kingman, S.M.; Hudson, G.J.; Cummings, J.H. Measurement of resistant starch in vitro and in vivo. Br. J. Nutr. 1996, 75, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Sluková, M.; Levková, J.; Michalcová, A.; Horáčková, Š.; Skřivan, P. Effect of the dough mixing process on the quality of wheat and buckwheat proteins. Czech J. Food Sci. 2017, 35, 522–531. [Google Scholar] [CrossRef]
- Struyf, N.; Verspreet, J.; Verstrepen, K.J.; Courtin, C.M. Investigating the impact of α-amylase, α-glucosidase and glucoamylase action on yeast-mediated bread dough fermentation and bread sugar levels. J. Cereal Sci. 2017, 75, 35–44. [Google Scholar] [CrossRef]
- Scanlon, M.G.; Zghal, M.C. Bread properties and crumb structure. Food Res. Int. 2001, 34, 841–864. [Google Scholar] [CrossRef]
- Romano, A.; Toraldo, G.; Cavella, S.; Masi, P. Description of leavening of bread dough with mathematical modelling. J. Food Eng. 2007, 83, 142–148. [Google Scholar] [CrossRef]
- Turbin-Orger, A.; Boller, E.; Chaunier, L.; Chiron, H.; Della Valle, G.; Réguerre, A.L. Kinetics of bubble growth in wheat flour dough during proofing studied by computed X-ray micro-tomography. J. Cereal Sci. 2012, 56, 676–683. [Google Scholar] [CrossRef]
- Launay, B.; Michon, C. Biaxial extension of wheat flour doughs: Lubricated squeezing flow and stress relaxation properties. J. Texture Stud. 2008, 39, 496–529. [Google Scholar] [CrossRef]
- Yang, Y.; Guana, E.; Zhang, T.; Li, M.; Bian, K. Influence of water addition methods on water mobility characterisation and rheological properties of wheat flour dough. J. Cereal Sci. 2019, 89, 102791. [Google Scholar] [CrossRef]
- Fan, J.L.; Han, N.; Chen, H.Q. Physicochemical and structural properties of wheat gluten/rice starch dough-like model. J. Cereal Sci. 2021, 98, 103181. [Google Scholar] [CrossRef]
- Li, W.; Dobraszcsyk, B.J.; Schofield, J.D. Stress relaxation behavior of wheat dough, gluten, and gluten protein fractions. Cereal Chem. 2003, 80, 333–338. [Google Scholar] [CrossRef]
- Goesaert, H.; Brijs, K.; Veraverbeke, W.S.; Courtin, C.M.; Gebruers, K.; Delcour, J.A. Wheat flour constituents: How they impact bread quality, and how to impact their functionality. Food Sci. Technol. 2005, 16, 12–30. [Google Scholar] [CrossRef]
- Rosell, C.M.; Foegeding, A. Interaction of hydroxypropylmethylcellulose with gluten proteins: Small deformation properties during thermal treatment. Food Hydrocoll. 2007, 21, 1092–1100. [Google Scholar] [CrossRef]
- Connelly, R.K.; McIntier, R.L. Rheological properties of yeasted and non yeasted wheat doughs developed under different mixing conditions. J. Sci. Food Agric. 2008, 88, 2309–2323. [Google Scholar] [CrossRef]
- Marchetti, L.; Cardós, M.; Campaña, L.; Ferrero, C. Effect of glutens of different quality on dough characteristics and breadmaking performance. LWT-Food Sci. Technol. 2012, 46, 224–231. [Google Scholar] [CrossRef]
- Miller, K.A.; Hoseney, R.C. Dynamic rheological properties of wheat starch-gluten doughs. Cereal Chem. 1999, 76, 105–109. [Google Scholar] [CrossRef]
- Salvador, A.; Sanz, T.; Fiszman, S.M. Dynamic rheological characteristics of wheat flour–water doughs. Effect of adding NaCl, sucrose and yeast. Food Hydrocoll. 2006, 20, 780–786. [Google Scholar] [CrossRef]
- Jekle, M.; Mühlberger, K.; Becker, T. Starch–gluten interactions during gelatinization and its functionality in dough like model systems. Food Hydrocoll. 2016, 54, 196–201. [Google Scholar] [CrossRef]
- Dreese, P.C.; Faubion, J.M.; Hoseney, R.C. Dynamic rheological properties of flour, gluten and gluten-starch doughs. I. Temperature-dependent changes during heating. Cereal Chem. 1988, 65, 348–353. [Google Scholar]
- Ooms, N.; Delcour, J.A. How to impact gluten protein network formation during wheat flour dough making. Curr. Opin. Food Sci. 2015, 25, 88–97. [Google Scholar] [CrossRef]
- Bernklau, I.; Neußer, C.; Moroni, A.V.; Gysler, C.; Spagnolello, A.; Chung, W.; Becker, T. Structural, textural and sensory impact of sodium reduction on long fermented pizza. Food Chem. 2017, 234, 398–407. [Google Scholar] [CrossRef]
- Fessas, D.; Schiraldi, A. Water properties in wheat flour dough I: Classical thermogravimetry approach. Food Chem. 2001, 72, 237–244. [Google Scholar] [CrossRef]
- Wehrli, M.C.; Kratky, T.; Schopf, M.; Scherf, K.A.; Becker, T.; Jekle, M. Thermally induced gluten modification observed with rheology and spectroscopies. Int. J. Biol. Macromol. 2021, 173, 26–33. [Google Scholar] [CrossRef]
- Khatkar, B.S.; Barak, S.; Mudgil, D. Effects of gliadin addition on the rheological, microscopic and thermal characteristics of wheat gluten. Int. J. Biol. Macromol. 2013, 53, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Addo, K.; Xiong, Y.L.; Blanchard, S.P. Thermal and dynamic rheological properties of wheat flour fractions. Food Res. Int. 2000, 34, 329–335. [Google Scholar] [CrossRef]
- Yiltirak, S.; Kocadağlı, T.; Çelik, E.E.; Kanmaz, E.O.; Gökmen, V. Effects of Sprouting and Fermentation on Free Asparagine and Reducing Sugars in Wheat, Einkorn, Oat, Rye, Barley, and Buckwheat and on Acrylamide and 5-Hydroxymethylfurfural Formation during Heating. J. Agric. Food Chem. 2021, 69, 9419–9433. [Google Scholar] [CrossRef] [PubMed]
- Meerts, M.; Cervera, A.R.; Struyf, N.; Cardinaels, R.; Courtin, C.M.; Moldenaers, P. The effects of yeast metabolites on the rheological behaviour of the dough matrix in fermented wheat flour dough. J. Cereal Sci. 2018, 82, 183–189. [Google Scholar] [CrossRef]
- Nielsen, P.M.; Petersen, D.; Dambmann, C. Improved Method for Determining Food Protein Degree of Hydrolysis. J. Food Sci. 2001, 66, 642–646. [Google Scholar] [CrossRef]
- Di Cagno, R.; De Angelis, M.; Lavermicocca, P.; De Vincenzi, M.; Giovannini, C.; Faccia, M.; Gobbetti, M. Proteolysis by sourdough lactic acid bacteria: Effects on wheat flour protein fractions and gliadin peptides involved in human cereal intolerance. Appl. Environ. Microbiol. 2002, 68, 623–633. [Google Scholar] [CrossRef]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar]
- Romano, A.; Mackie, A.; Farina, F.; Aponte, M.; Sarghini, F.; Masi, P. Characterisation, in vitro digestibility and expected glycemic index of commercial starches as uncooked ingredients. J. Food Sci. Technol. 2016, 53, 4126–4134. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Barker, H.; Fielden, H.; Baldwin, J.M.; Goff, D.V. Glycemic index of foods: A physiological basis for carbohydrate exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Rosin, P.M.; Lajolo, F.M.; Menezes, E.W. Measurement and characterisation of dietary starches. J. Food Compos. Anal. 2002, 15, 367–377. [Google Scholar] [CrossRef]
- Ronda, F.; Rivero, P.; Caballero, P.A.; Quilez, J. High insoluble fibre content increases in vitro starch digestibility in partially baked breads. Int. J. Food Sci. Nutr. 2012, 63, 971–977. [Google Scholar] [CrossRef]
- Ells, L.J.; Seal, C.J.; Kettlitz, B.; Bal, W.; Mathers, J.C. Postprandial glycaemic, lipaemic and haemostatic responses to ingestion of rapidly and slowly digested starches in healthy young women. Br. J. Nutr. 2005, 94, 948–955. [Google Scholar] [CrossRef]
- Friedman, M. Chemistry, biochemistry, and safety of acrylamide. A review. J. Agric. Food Chem. 2003, 51, 4504–4526. [Google Scholar] [CrossRef]
- Wang, S.; Yu, J.; Xin, Q.; Wang, S.; Copeland, L. Effects of starch damage and yeast fermentation on acrylamide formation in bread. Food Control 2017, 73, 230–236. [Google Scholar] [CrossRef]
- Gökmen, V.; Palazoğlu, T.K. Acrylamide Formation in Foods during Thermal Processing with a Focus on Frying. Food Bioproc. Technol. 2008, 1, 35–42. [Google Scholar] [CrossRef]
- Dessev, T.; Lalanne, V.; Keramat, J.; Jury, V.; Prost, C.; Le-Bail, A. Influence of Baking Conditions on Bread Characteristics and Acrylamide Concentration. J. Food Sci. Nutr. Res. 2020, 3, 291–310. [Google Scholar] [CrossRef]
- Ee, K.Y.; Eng, M.K.; Lee, M.L. Physicochemical, thermal and rheological properties of commercial wheat flours and corresponding starches. Food Sci. Technol. 2020, 40, 51–59. [Google Scholar] [CrossRef]
- Yan, B.; Yang, H.; Wu, Y.; Lian, H.; Zhang, H.; Chen, W.; Fan, D.; Zhao, X. Quality Enhancement Mechanism of Alkali-Free Chinese Northern Steamed Bread by Sourdough Acidification. Molecules 2020, 25, 726. [Google Scholar] [CrossRef]
- Marti, A.; Torri, L.; Casiraghi, M.C.; Franzetti, L.; Limbo, S.; Morandin, F.; Pagani, M.A. Wheat germ stabilization by heat-treatment or sourdough fermentation: Effects on dough rheology and bread properties. LWT-Food Sci. Technol. 2014, 59, 1100–1106. [Google Scholar] [CrossRef]
- Peng, B.; Li, Y.; Ding, S.; Yang, J. Characterisation of textural, rheological, thermal, microstructural, and water mobility in wheat flour dough and bread affected by trehalose. Food Chem. 2017, 233, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Di Cagno, R.; Rizzello, C.G.; Gobbetti, M. Adverse reactions to gluten: Exploitation of sourdough fermentation. In Wheat and Rice in Disease Prevention and Health; Watson, R.R., Preedy, V.R., Zibadi, S., Eds.; Academic Press: London, UK, 2014; Chapter 14; pp. 171–177. [Google Scholar] [CrossRef]
- Lindsay, M.P.; Skerritt, J.H. The glutenin macropolymer of wheat flour doughs: Structure–function perspectives. Trends Food Sci. Technol. 1999, 10, 247–253. [Google Scholar] [CrossRef]




| Leavening Time (h) | Compression W (J) | Relaxation a (%) | Decay Rate b (N s−1) | Diameter (cm) |
|---|---|---|---|---|
| 0 | 6.59 ± 0.41 a | 82.5 ± 0.7 a | 0.387 ± 0.014 a | 21.50 ± 2.07 a |
| 4 | 29.96 ± 2.52 b | 79.7 ± 0.3 b | 0.409 ± 0.007 b | 29.25 ± 0.53 b |
| 8 | 16.16 ± 0.69 c | 78.9 ± 0.4 b | 0.342 ± 0.008 c | 26.06 ± 1.05 c |
| 16 | 19.72 ± 1.10 d | 80.8 ± 0.3 c | 0.359 ± 0.015 c | 27.50 ± 0.46 cd |
| 24 | 15.97 ± 1.43 c | 80.9 ± 1.0 c | 0.347 ± 0.010 c | 28.37 ± 0.44 bd |
| 48 | 7.65 ± 0.40 a | 85.2 ± 0.3 d | 0.283 ± 0.013 d | 28.06 ± 0.42 bd |
| Leavening Time (h) | Ti (°C) | Tg (°C) | Te (°C) | ∆H (J/g) |
|---|---|---|---|---|
| 0 | 68.17 ± 0.01 a | 75.40 ± 0.01 a | 85.15 ± 0.33 a | 0.613 ± 0.015 a |
| 4 | 68.09 ± 0.47 a | 75.12 ± 0.30 a | 86.40 ± 0.42 a | 0.709 ± 0.056 b |
| 8 | 68.08 ± 0.65 a | 75.53 ± 0.03 a | 86.70 ± 1.12 a | 0.805 ± 0.045 b |
| 16 | 66.54 ± 0.29 b | 74.14 ± 0.27 b | 87.46 ± 0.42 a | 0.975 ± 0.013 c |
| 24 | 68.29 ± 0.27 a | 75.39 ± 0.22 a | 88.00 ± 0.67 a | 0.917 ± 0.036 d |
| 48 | 67.65 ± 0.63 ab | 74.36 ± 0.08 b | 86.94 ± 1.68 a | 0.730 ± 0.090 b |
| Leavening Time (h) | RDS | SDS | RS | TS | SDRI | RAG | |
|---|---|---|---|---|---|---|---|
| (%) | |||||||
| A | |||||||
| dough | 0 | 12.15 ± 0.96 a | 12.48 ± 0.70 a | 44.07 ± 1.75 a | 68.71 ± 3.42 a | - | - |
| 4 | 11.81 ± 0.07 a | 12.06 ± 0.54 a | 36.89 ± 0.33 b | 60.76 ± 0.29 a | - | - | |
| 8 | 13.29 ± 0.54 a | 11.43 ± 1.44 a | 38.59 ± 1.05 b | 63.32 ± 3.04 a | - | - | |
| 16 | 9.76 ± 0.36 b | 12.22 ± 2.02 a | 38.83 ± 0.62 b | 60.81 ± 2.28 a | - | - | |
| 24 | 7.25 ± 0.12 c | 12.12 ± 1.27 a | 46.78 ± 2.93 a | 66.16 ± 4.33 a | - | - | |
| 48 | 9.25 ± 0.39 b | 8.72 ± 0.59 a | 47.99 ± 0.43 a | 65.96 ± 0.23 a | - | - | |
| B | |||||||
| pizza base | 0 | 46.06 ± 3.01 a | 26.08 ± 5.87 a | 0.58 ± 0.05 a | 72.72 ± 2.82 a | 63.46 ± 6.60 a | 51.18 ± 3.44 a |
| 4 | 53.25 ± 1.07 ab | 11.53 ± 5.79 b | 0.50 ± 0.01 a | 65.28 ± 4.73 a | 81.84 ± 7.57 b | 59.17 ± 1.19 ab | |
| 8 | 65.65 ± 0.75 c | 1.62 ± 0.76 c | 0.48 ± 0.03 a | 67.75 ± 1.55 a | 96.91 ± 1.11 c | 72.95 ± 0.83 c | |
| 16 | 67.15 ± 0.87 c | 1.63 ± 0.87 c | 0.44 ± 0.02 a | 69.23 ± 0.02 a | 97.00 ± 1.28 c | 74.61 ± 0.97 c | |
| 24 | 64.58 ± 3.93 c | 1.67 ± 1.13 c | 0.45 ± 0.07 a | 66.71 ± 2.87 a | 96.78 ± 1.73 c | 71.76 ± 4.37 c | |
| 48 | 59.02 ± 2.19 bc | 3.22 ± 0.76 c | 0.49 ± 0.02 a | 62.73 ± 1.45 a | 94.07 ± 1.32 c | 65.58 ± 2.44 bc | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Covino, C.; Sorrentino, A.; Di Pierro, P.; Masi, P. Study of Physico-Chemical Properties of Dough and Wood Oven-Baked Pizza Base: The Effect of Leavening Time. Foods 2023, 12, 1407. https://doi.org/10.3390/foods12071407
Covino C, Sorrentino A, Di Pierro P, Masi P. Study of Physico-Chemical Properties of Dough and Wood Oven-Baked Pizza Base: The Effect of Leavening Time. Foods. 2023; 12(7):1407. https://doi.org/10.3390/foods12071407
Chicago/Turabian StyleCovino, Clelia, Angela Sorrentino, Prospero Di Pierro, and Paolo Masi. 2023. "Study of Physico-Chemical Properties of Dough and Wood Oven-Baked Pizza Base: The Effect of Leavening Time" Foods 12, no. 7: 1407. https://doi.org/10.3390/foods12071407
APA StyleCovino, C., Sorrentino, A., Di Pierro, P., & Masi, P. (2023). Study of Physico-Chemical Properties of Dough and Wood Oven-Baked Pizza Base: The Effect of Leavening Time. Foods, 12(7), 1407. https://doi.org/10.3390/foods12071407






