Glyphosate and Glufosinate Residues in Honey and Other Hive Products
Abstract
1. Introduction
2. Methods
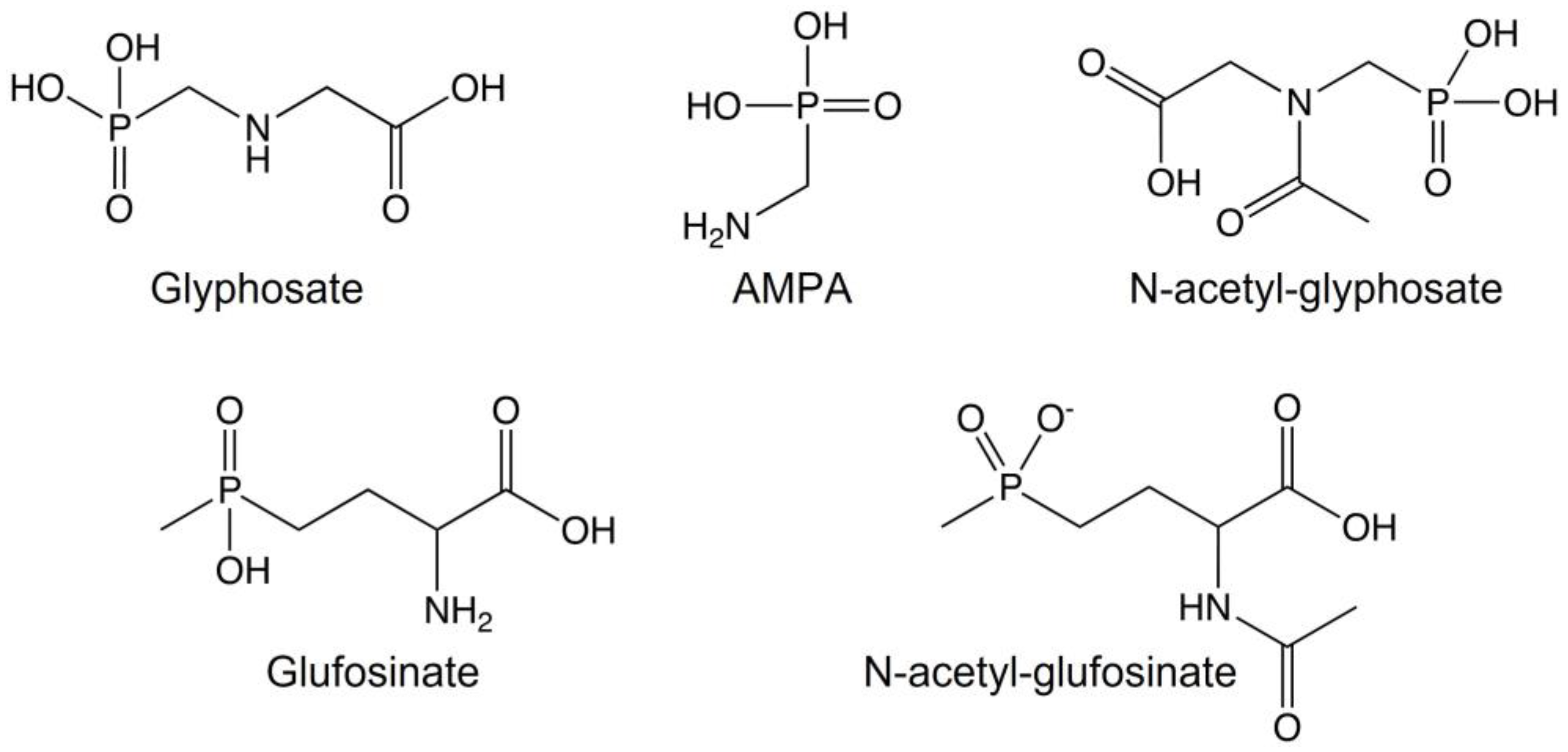
3. Chemical Properties and Mechanism of Action of Glyphosate and Glufosinate
4. Degradation of Glyphosate and Glufosinate
5. Glyphosate and Glufosinate Effects on Honeybees’ Health
6. Regulatory Agencies’ Assessment on Health Effects in Humans and Regulatory Developments
7. Analytical Methods for the Determination of Glyphosate, Glufosinate, and Their Metabolites in Honey and Other Hive Products
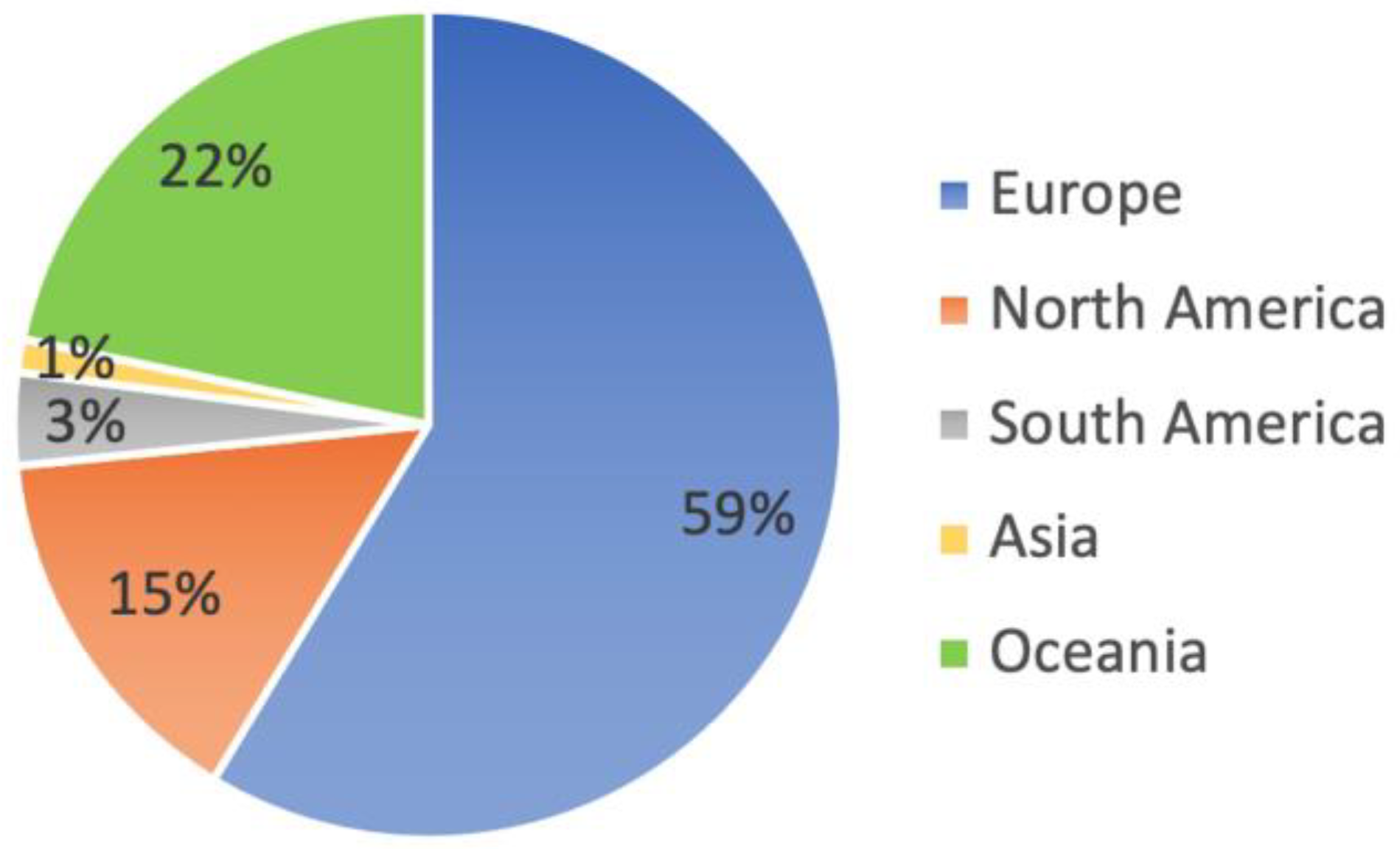
8. Reported Levels of Herbicide Contamination in Hive Products
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- de Oliveira, R.C.; do Nascimento Queiroz, S.C.; da Luz CF, P.; Porto, R.S.; Rath, S. Bee Pollen as a Bioindicator of Environmental Pesticide Contamination. Chemosphere 2016, 163, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Battisti, L.; Potrich, M.; Sampaio, A.R.; de Castilhos Ghisi, N.; Costa-Maia, F.M.; Abati, R.; dos Reis Martinez, C.B.; Sofia, S.H. Is Glyphosate Toxic to Bees? A Meta-Analytical Review. Sci. Total Environ. 2021, 767, 145397. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Pant, K.; Brar, D.S.; Thakur, A.; Nanda, V. A Review on Api-Products: Current Scenario of Potential Contaminants and Their Food Safety Concerns. Food Control 2023, 145, 109499. [Google Scholar] [CrossRef]
- Lajmanovich, R.C.; Attademo, A.M.; Lener, G.; Cuzziol Boccioni, A.P.; Peltzer, P.M.; Martinuzzi, C.S.; Demonte, L.D.; Repetti, M.R. Glyphosate and Glufosinate Ammonium, Herbicides Commonly Used on Genetically Modified Crops, and Their Interaction with Microplastics: Ecotoxicity in Anuran Tadpoles. Sci. Total Environ. 2022, 804, 150177. [Google Scholar] [CrossRef]
- Dill, G.M. Glyphosate-Resistant Crops: History, Status and Future. Pest Manag. Sci. 2005, 61, 219–224. [Google Scholar] [CrossRef]
- Marketresearch. Available online: https://www.marketresearch.com/Global-Industry-Analysts-v1039/Glyphosate-32281218/ (accessed on 18 January 2023).
- More, S.J.; Auteri, D.; Rortais, A.; Pagani, S. EFSA Is Working to Protect Bees and Shape the Future of Environmental Risk Assessment. EFSA J. 2021, 19, e190101. [Google Scholar] [CrossRef] [PubMed]
- Bosco, L.; Bergero, M.; Panella, F.; Oldani, S.; Concari, C. Dossier | 2017–2019 Environmental Bio-Monitoring with Bees. Available online: https://www.lapisonline.it/wps/wp-content/uploads/2021/09/ENGL_dossier_02_2019_biomonitorag.pdf (accessed on 30 January 2023).
- Verdini, E.; Pecorelli, I. The Current Status of Analytical Methods Applied to the Determination of Polar Pesticides in Food of Animal Origin: A Brief Review. Foods 2022, 11, 1527. [Google Scholar] [CrossRef]
- Soares, D.; Silva, L.; Duarte, S.; Pena, A.; Pereira, A. Glyphosate Use, Toxicity and Occurrence in Food. Foods 2021, 10, 2785. [Google Scholar] [CrossRef]
- Dill, G.M.; Sammons, R.D.; Feng, P.C.; Kohn, F.; Kretzmer, K.; Mehrsheikh, A.; Bleeke, M.; Honegger, J.L.; Farmer, D.; Wright, D. Glyphosate: Discovery, Development, Applications, and Properties. In Glyphosate Resistance in Crops and Weeds: History, Development, and Management; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 1–33. [Google Scholar] [CrossRef]
- Takano, H.K.; Dayan, F.E. Glufosinate-ammonium: A Review of the Current State of Knowledge. Pest Manag. Sci. 2020, 76, 3911–3925. [Google Scholar] [CrossRef]
- European Commission Review Report for the Active Substance Glyphosate. 29 June 2001. Available online: https://ec.europa.eu/food/fs/ph_ps/pro/eva/existing/list1_glyphosate_en.pdf (accessed on 18 January 2023).
- Singh, S.; Kumar, V.; Gill, J.P.K.; Datta, S.; Singh, S.; Dhaka, V.; Kapoor, D.; Wani, A.B.; Dhanjal, D.S.; Kumar, M. Herbicide Glyphosate: Toxicity and Microbial Degradation. Int. J. Environ. Res. Public Health 2020, 17, 7519. [Google Scholar] [CrossRef]
- Ojelade, B.S.; Durowoju, O.S.; Adesoye, P.O.; Gibb, S.W.; Ekosse, G.-I. Review of Glyphosate-Based Herbicide and Aminomethylphosphonic Acid (AMPA): Environmental and Health Impacts. Appl. Sci. 2022, 12, 8789. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Conclusion Regarding the Peer Review of the Pesticide Risk Assessment of the Active Substance Glufosinate. EFSA J. 2005, 27, 1–81. [Google Scholar] [CrossRef]
- Johnson, R.M. Honeybee Toxicology. Annu. Rev. Entomol. 2015, 60, 415–434. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Advancing Environmental Risk Assessment for Bees and Other Insect Pollinators. Available online: https://www.efsa.europa.eu/en/events/advancing-environmental-risk-assessment-bees-and-other-insect-pollinators (accessed on 18 January 2023).
- U.S. Environmental Protection Agency. U.S. Environmental Protection Agency’s Policy to Mitigate the Acute Risk to Bees from Pesticide Products. Available online: https://www.regulations.gov/document/EPA-HQ-OPP-2014-0818-0477 (accessed on 30 January 2023).
- European Food Safety Authority. EFSA Explains Risk Assessment: Glyphosate. Available online: https://www.efsa.europa.eu/en/corporate/pub/glyphosate151112 (accessed on 30 January 2023).
- IUPAC Pesticides Properties DataBase. Available online: http://sitem.herts.ac.uk/aeru/iupac/index.htm (accessed on 18 January 2023).
- FAO/WHO. Pesticide Residues in Food 2016; FAO/WHO: Rome, Italy, 2016. Available online: https://www.fao.org/3/I6926EN/bookmarks-i6926en.pdf (accessed on 30 January 2023).
- International Agency for Research on Cancer, World Health Organization. Some Organophosphate Insecticides and Herbicides. Lyon, France, 2017. Available online: https://www.iarc.who.int/news-events/iarc-monographs-volume-112-some-organophosphate-insecticides-and-herbicides-3/ (accessed on 30 January 2023).
- Benbrook, C.M. How Did the US EPA and IARC Reach Diametrically Opposed Conclusions on the Genotoxicity of Glyphosate-Based Herbicides? Environ. Sci. Eur. 2019, 31, 2. [Google Scholar] [CrossRef]
- European Parliament. Renewing the Approval of the Active Substance Glyphosate. Available online: https://www.europarl.europa.eu/doceo/document/TA-8-2017-0395_EN.pdf (accessed on 30 January 2023).
- European Commission. Extension of the Approval Period of Glyphosate. Available online: https://food.ec.europa.eu/plants/pesticides/approval-active-substances/renewal-approval/glyphosate_en (accessed on 12 January 2023).
- Donthi, D.N.R.; Kumar, A.D.D. Glufosinate Ammonium An Overview, Pesticide Action Network, India. 2022. Available online: https://pan-india.org/wp-content/uploads/2022/12/Glufosinate-Ammonium-An-overview_PAN-India_Dec2022.pdf (accessed on 30 January 2023).
- Commission Regulation (EU) 2020/1068 of 15 May 2020 Amending Annexes I and V to Regulation (EU) No 649/2012 of the European Parliament and of the Council Concerning the Export and Import of Hazardous Chemicals. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32020R1068&from=EN (accessed on 23 February 2023).
- Commission Regulation (EU) 2018/62 of 17 January 2018 Replacing Annex I to Regulation (EC) No 396/2005 of the European Parliament and of the Council. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32018R0062&from=EN (accessed on 24 February 2023).
- Commission Regulation (EU) No 293/2013 of 20 March 2013 Amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as Regards Maximum Residue Levels for Emamectin Benzoate, Etofenprox, Etoxazole, Flutriafol, Glyphosate, Phosmet, Pyraclostrobin, Spinosad and Spirotetramat in or on Certain Products. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32013R0293&from=EN (accessed on 23 February 2023).
- Commission Regulation (EU) 2016/1002 of 17 June 2016 Amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as Regards Maximum Residue Levels for AMTT, Diquat, Dodine, Glufosinate and Tritosulfuron in or on Certain Products. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32016R1002&from=EN (accessed on 23 February 2023).
- Japan Food Chemical Research Foundation. Search Engine for MRLs. Available online: http://db.ffcr.or.jp/front/food_group_detail?id=20600 (accessed on 18 January 2023).
- Australia New Zealand Food Standards Code–Schedule 20–Maximum Residue Limits. Available online: https://www.legislation.gov.au/Series/F2015L00468 (accessed on 18 January 2023).
- Thompson, T.S.; van den Heever, J.P.; Limanowka, R.E. Determination of Glyphosate, AMPA, and Glufosinate in Honey by Online Solid-Phase Extraction-Liquid Chromatography-Tandem Mass Spectrometry. Food Addit. Contam. Part A 2019, 36, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Emily Guo, F.R. Survey of Glyphosate Residues in Honey, Corn and Soy Products. J. Environ. Anal. Toxicol. 2014, 5, 1–8. [Google Scholar] [CrossRef]
- Medici, S.K.; Maggi, M.D.; Galetto, L.; del Rosario Iglesias, M.; Sarlo, E.G.; Recavarren, M.I.; Salar, P.E.; Eguaras, M.J. Influence of the Agricultural Landscape Surrounding Apis Mellifera Colonies on the Presence of Pesticides in Honey. Apidologie 2022, 53, 21. [Google Scholar] [CrossRef]
- Bergero, M.; Bosco, L.; Giacomelli, A.; Angelozzi, G.; Perugini, M.; Merola, C. Agrochemical Contamination of Honey and Bee Bread Collected in the Piedmont Region, Italy. Environments 2021, 8, 62. [Google Scholar] [CrossRef]
- Jansons, M.; Pugajeva, I.; Bartkevics, V.; Karkee, H.B. LC-MS/MS Characterisation and Determination of Dansyl Chloride Derivatised Glyphosate, Aminomethylphosphonic Acid (AMPA), and Glufosinate in Foods of Plant and Animal Origin. J. Chromatogr. B 2021, 1177, 122779. [Google Scholar] [CrossRef]
- El Agrebi, N.; Tosi, S.; Wilmart, O.; Scippo, M.-L.; de Graaf, D.C.; Saegerman, C. Honeybee and Consumer’s Exposure and Risk Characterisation to Glyphosate-Based Herbicide (GBH) and Its Degradation Product (AMPA): Residues in Beebread, Wax, and Honey. Sci. Total Environ. 2020, 704, 135312. [Google Scholar] [CrossRef]
- Raimets, R.; Bontšutšnaja, A.; Bartkevics, V.; Pugajeva, I.; Kaart, T.; Puusepp, L.; Pihlik, P.; Keres, I.; Viinalass, H.; Mänd, M.; et al. Pesticide Residues in Beehive Matrices Are Dependent on Collection Time and Matrix Type but Independent of Proportion of Foraged Oilseed Rape and Agricultural Land in Foraging Territory. Chemosphere 2020, 238, 124555. [Google Scholar] [CrossRef] [PubMed]
- de Souza, A.P.F.; Rodrigues, N.R.; Reyes, F.G.R. Glyphosate and Aminomethylphosphonic Acid (AMPA) Residues in Brazilian Honey. Food Addit. Contam. Part B 2021, 14, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, L.M.; Nobile, M.; Panseri, S.; Arioli, F. Detection of Glyphosate and Its Metabolites in Food of Animal Origin Based on Ion-Chromatography-High Resolution Mass Spectrometry (IC-HRMS). Food Addit. Contam. Part A 2019, 36, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Pareja, L.; Jesús, F.; Heinzen, H.; Hernando, M.D.; Rajski, Ł.; Fernández-Alba, A.R. Evaluation of Glyphosate and AMPA in Honey by Water Extraction Followed by Ion Chromatography Mass Spectrometry. A Pilot Monitoring Study. Anal. Methods 2019, 11, 2123–2128. [Google Scholar] [CrossRef]
- Berg, C.J.; King, H.P.; Delenstarr, G.; Kumar, R.; Rubio, F.; Glaze, T. Glyphosate Residue Concentrations in Honey Attributed through Geospatial Analysis to Proximity of Large-Scale Agriculture and Transfer off-Site by Bees. PLoS ONE 2018, 13, e0198876. [Google Scholar] [CrossRef]
- John, J.; Liu, H. Glyphosate Monitoring in Water, Foods, and Urine Reveals an Association between Urinary Glyphosate and Tea Drinking: A Pilot Study. Int. J. Environ. Health Eng. 2018, 7, 2. [Google Scholar] [CrossRef]
- Zoller, O.; Rhyn, P.; Rupp, H.; Zarn, J.A.; Geiser, C. Glyphosate Residues in Swiss Market Foods: Monitoring and Risk Evaluation. Food Addit. Contam. Part B 2018, 11, 83–91. [Google Scholar] [CrossRef]
- Chamkasem, N.; Vargo, J. Development and Independent Laboratory Validation of an Analytical Method for the Direct Determination of Glyphosate, Glufosinate, and Aminomethylphosphonic Acid in Honey by Liquid Chromatography/Tandem Mass Spectrometry. JRS 2017, 5, 1–9. [Google Scholar] [CrossRef]
- Karise, R.; Raimets, R.; Bartkevics, V.; Pugajeva, I.; Pihlik, P.; Keres, I.; Williams, I.H.; Viinalass, H.; Mänd, M. Are Pesticide Residues in Honey Related to Oilseed Rape Treatments? Chemosphere 2017, 188, 389–396. [Google Scholar] [CrossRef]
- Anastassiades, M.; Mack, D.; Wildgrube, C.; Barth, A.; Sigalov, I.; Görlich, S.; Dörk, D.; Cerchia, G. Quick Method for the Analysis of Highly Polar Pesticides in Food Involving Extraction with Acidified Methanol and LC- or IC-MS/MS Measurement in Food of Plant Origin (QuPPe-PO-Method). Available online: https://www.eurl-pesticides.eu/docs/public/tmplt_article.asp?LabID=200&CntID=1115&Theme_ID=1&Pdf=False&Lang=EN (accessed on 18 January 2023).
- European Food Safety Authority. Management of Left-censored Data in Dietary Exposure Assessment of Chemical Substances. EFSA J. 2010, 8, 1557. [Google Scholar] [CrossRef]
- New Zealand National Chemical Residues Programme Report Results for Agricultural Compound Residues in Honey. Available online: https://www.mpi.govt.nz/dmsdocument/44002-Results-for-1-July-2019-30-June-2020 (accessed on 30 January 2023).
- EU Pesticides Database. Available online: https://food.ec.europa.eu/plants/pesticides/eu-pesticides-database_en (accessed on 30 January 2023).
- Yaqub, G.; Khalid, M.; Ikram, A.; Sohail, A. Monitoring and Risk Assessment Due to Presence of Metals and Pesticides Residues in Honey Samples from the Major Honey Producing Forest Belts and Different Brands. Food Sci. Technol. 2020, 40, 331–335. [Google Scholar] [CrossRef]
- Panseri, S.; Bonerba, E.; Nobile, M.; Di Cesare, F.; Mosconi, G.; Cecati, F.; Arioli, F.; Tantillo, G.; Chiesa, L. Pesticides and Environmental Contaminants in Organic Honeys According to Their Different Productive Areas toward Food Safety Protection. Foods 2020, 9, 1863. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA); Carrasco Cabrera, L.; Medina Pastor, P. The 2019 European Union Report on Pesticide Residues in Food. EFSA J. 2021, 19, e06491. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA); Medina-Pastor, P.; Triacchini, G. The 2018 European Union Report on Pesticide Residues in Food. EFSA J. 2020, 18, e06057. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). The 2017 European Union Report on Pesticide Residues in Food. EFSA J. 2019, 17, e05743. [Google Scholar] [CrossRef]
- Ruiz-Toledo, J.; Sánchez, D. Risk Characterization to Glyphosate in Honeybee and Consumer’s Exposed to Honeybee-Collected Pollen in Southeastern Mexico. 2022. Available online: https://europepmc.org/article/PPR/PPR523428 (accessed on 18 January 2023). [CrossRef]

| Country | Matrix | Analyte | MRL (µg/kg) |
|---|---|---|---|
| EU | Honey and other apiculture products | Glyphosate | 50 |
| EU | Honey and other apiculture products | Glufosinate (Sum of glufosinate isomers, its salts, MPP, and N-acetyl-glufosinate) | 50 |
| Japan | Honey (Including royal jelly) | Glyphosate (Sum of glyphosate and N-acetyl-glyphosate) | 50 |
| Australia | Honey | Glyphosate (Sum of glyphosate, N-acetyl-glyphosate, and AMPA) | 200 |
| New Zealand | Honey | Glyphosate (Sum of glyphosate, N-acetyl-glyphosate, and AMPA) | 200 |
| Ref. | Matrix | Analyte | LOD (µg/kg) | LOQ (µg/kg) | Derivatization | Instrumentation |
|---|---|---|---|---|---|---|
| [36] | Honey | Glyphosate | 1 | n.r. | FMOC | LC-MS/MS |
| AMPA | ||||||
| [37] | Honey | Glyphosate | 3 | 10 | - | LC-MS/MS |
| Beebread | ||||||
| [38] | Honey | Glyphosate | n.r. | 25 | Dansyl cloride | LC-MS/MS |
| AMPA | 10 | |||||
| Glufosinate | 10 | |||||
| [39] | Honey | Glyphosate AMPA | 1 | 10 | - | LC-MS/MS |
| Beebread | ||||||
| Wax | ||||||
| [40] | Honey | Glyphosate | n.r. | 50 | - | LC-MS/MS |
| Beebread | ||||||
| [41] | Honey | Glyphosate | 20 | 40 | OPA | HPLC-FLD |
| AMPA | ||||||
| [42] | Honey | Glyphosate | n.r. | 9.26 | - | IC-HRMS |
| AMPA | 4.3 | |||||
| Glufosinate | 5.05 | |||||
| [43] | Honey | Glyphosate | n.r. | 5 | - | IC-HRMS |
| AMPA | 20 | |||||
| [34] | Honey | Glyphosate | n.r. | 1 | FMOC | LC-MS/MS |
| AMPA | ||||||
| Glufosinate | ||||||
| [44] | Honey | Glyphosate | n.r. | 15 | - | ELISA |
| [45] | Honey | Glyphosate | 0.075 | 0.4 | - | ELISA |
| [46] | Honey | Glyphosate | n.r. | 1 | - | LC-MS/MS |
| AMPA | 2.5 | |||||
| [47] | Honey | Glyphosate | n.r. | 16 | - | LC-MS/MS |
| [48] | Honey | Glyphosate | n.r. | 50 | - | LC-MS/MS |
| [35] | Honey | Glyphosate | n.r. | 15 | - | ELISA |
| Matrix | Ref. | Origin | Analyte | Total Sample (Number) | LC % | Quantifiable Samples | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Number | % | Range | Mean | Median | ||||||
| Honey | [36] | Argentina | Gly | 30 | 50 | 15 | 50 | 2.0–27.5 | n.r. | n.r. |
| AMPA | 70 | 9 | 30 | 1.9–18.1 | n.r. | n.r. | ||||
| Honey | [51] | New Zealand | Gly | 360 | 79 | 78 | 21 | n.r. | n.r. | n.r. |
| Honey | [37] | Italy | Gly | 84 | 50 | 42 | 50 | 10–34 | 17.1 | n.r. |
| Honey | [52] | Various EU countries | Gly | 115 | 94 | 7 | 6 | 29–5500 | 1153 | 270 |
| AMPA | 35 | 100 | 0 | - | - | - | - | |||
| N-Acetyl Gly | 1 | 100 | 0 | - | - | - | - | |||
| Glu | 14 | 100 | 0 | - | - | - | - | |||
| N-Acetyl Glu | 1 | 100 | 0 | - | - | - | - | |||
| Honey | [53] | Pakistan | Gly | 25 | 80 | 5 | 20 | 440–3500 | 2004 | n.r. |
| Honey | [39] | Belgium | Gly | 10 | 90 | 1 | 10 | 11 | 11 | 11 |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
| Honey | [54] | Italy | Gly | 98 | 100 | 0 | 0 | - | - | - |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
| Glu | 100 | 0 | 0 | - | - | - | ||||
| Honey | [41] | Brazil | Gly | 40 | 63 | 15 | 37 | 40–220 | n.r. | 70 |
| AMPA | 98 | 1 | 2 | 20–100 | - | - | ||||
| Honey | [55] | EU | Gly | 249 | 93 | 17 | 7 | n.r. | n.r. | n.r. |
| Honey | [8] | Italy | Gly | 176 | 50 | 88 | 50 | 10–790 | n.r. | n.r. |
| Honey | [42] | Italy | Gly | 10 | 100 | 0 | 0 | - | - | - |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
| Glu | 100 | 0 | 0 | - | - | - | ||||
| Honey | [43] | Uruguay & EU countries | Gly | 32 | 19 | 26 | 81 | n.r. | n.r. | n.r. |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
| Honey | [34] | Canada | Gly | 200 | 2 | 197 | 98 | 1–49.8 | n.r. | 4.9 |
| AMPA | 1 | 198 | 99 | 1–50 | n.r. | 10.3 | ||||
| Glu | 38 | 125 | 62 | 1–33 | n.r. | 1.4 | ||||
| Honey | [56] | EU | Gly | 157 | 94 | 9 | 6 | n.r. | n.r. | n.r. |
| Honey | [44] | USA (Hawaii) | Gly | 59 | 73 | 16 | 27 | 15–342 | 33.5 LB 118.3 UB | n.r. |
| Honey | [46] | Switzerland | Gly | 16 | 6 | 15 | 94 | 1–15.9 | 4.6 | 3 |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
| Honey | [57] | EU | Gly | 186 | 87 | 24 | 13 | n.r. | n.r. | n.r. |
| Honey | [48] | Estonia | Gly | 33 | 36 | 21 | 64 | 14–62 | 44 | n.r. |
| Honey | [47] | USA | Gly | 16 | 44 | 9 | 56 | n.r. | n.r. | n.r. |
| Honey | [35] | USA | Gly | 69 | 41 | 41 | 59 | 17–63 | n.r. | n.r. |
| Beebread | [37] | Italy | Gly | 84 | 46 | 45 | 54 | 10–542 | n.r. | n.r. |
| Beebread | [39] | Belgium | Gly | 81 | 9 | 74 | 91 | 10–700 | 55.5 | 26 |
| AMPA | 82 | 21 | 18 | 10–250 | 67.1 | 44 | ||||
| Wax | [39] | Belgium | Gly | 100 | 68 | 32 | 32 | 10–320 | 62 | 36 |
| AMPA | 100 | 0 | 0 | - | - | - | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rampazzo, G.; Gazzotti, T.; Zironi, E.; Pagliuca, G. Glyphosate and Glufosinate Residues in Honey and Other Hive Products. Foods 2023, 12, 1155. https://doi.org/10.3390/foods12061155
Rampazzo G, Gazzotti T, Zironi E, Pagliuca G. Glyphosate and Glufosinate Residues in Honey and Other Hive Products. Foods. 2023; 12(6):1155. https://doi.org/10.3390/foods12061155
Chicago/Turabian StyleRampazzo, Giulia, Teresa Gazzotti, Elisa Zironi, and Giampiero Pagliuca. 2023. "Glyphosate and Glufosinate Residues in Honey and Other Hive Products" Foods 12, no. 6: 1155. https://doi.org/10.3390/foods12061155
APA StyleRampazzo, G., Gazzotti, T., Zironi, E., & Pagliuca, G. (2023). Glyphosate and Glufosinate Residues in Honey and Other Hive Products. Foods, 12(6), 1155. https://doi.org/10.3390/foods12061155







