Use of Oenological Tannins to Protect the Colour of Rosé Wine in a Bioprotection Strategy with Metschnikowia pulcherrima
Abstract
1. Introduction
2. Materials and Methods
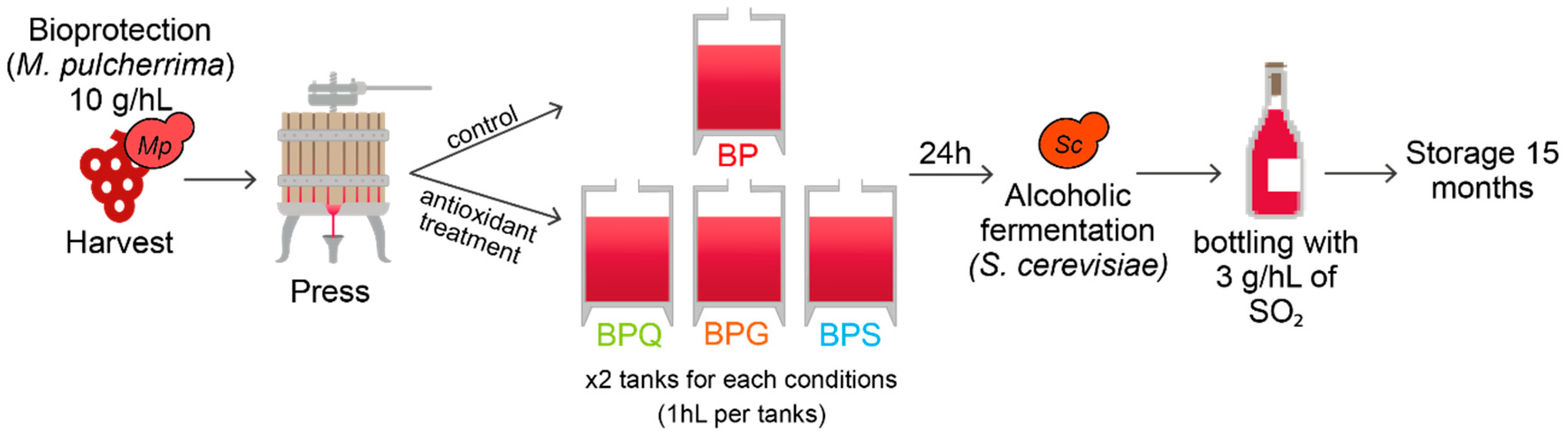
2.1. Yeast Strains and Experimental Conditions
2.2. Experimental Sampling
2.3. Microbiological Analysis
2.4. Fermentation Kinetics and Wine Composition
2.5. Colorimetric Analyses by Tristimulus Coordinates (L*a*b*)
2.6. Anthocyanins Analyses
2.7. UHPLC Analysis of Phenolic Compounds
2.8. Statistical Analysis
3. Results
3.1. Microbial Behavour
3.2. Fermentation Kinetics Analysis and Must and Wine Composition
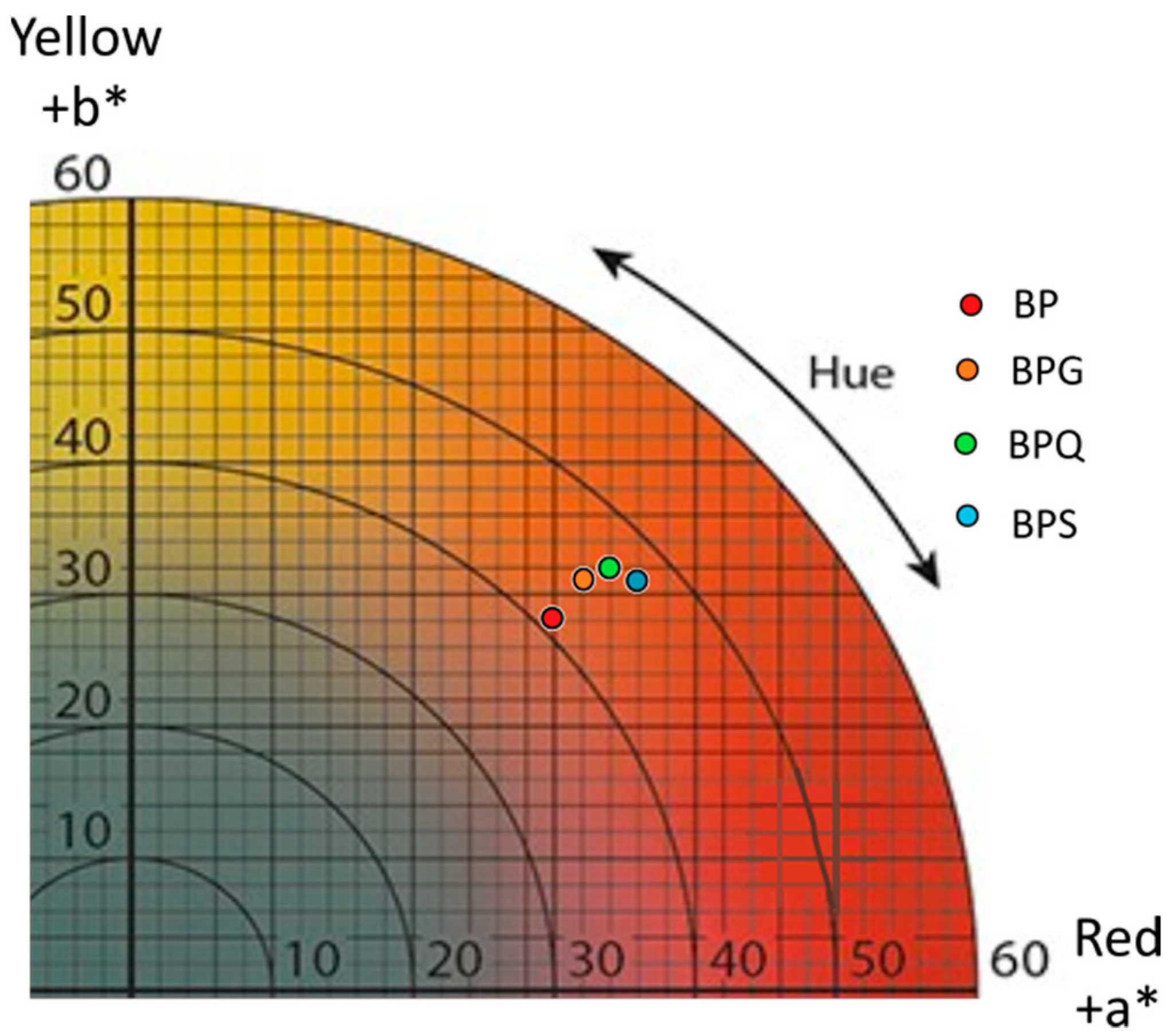
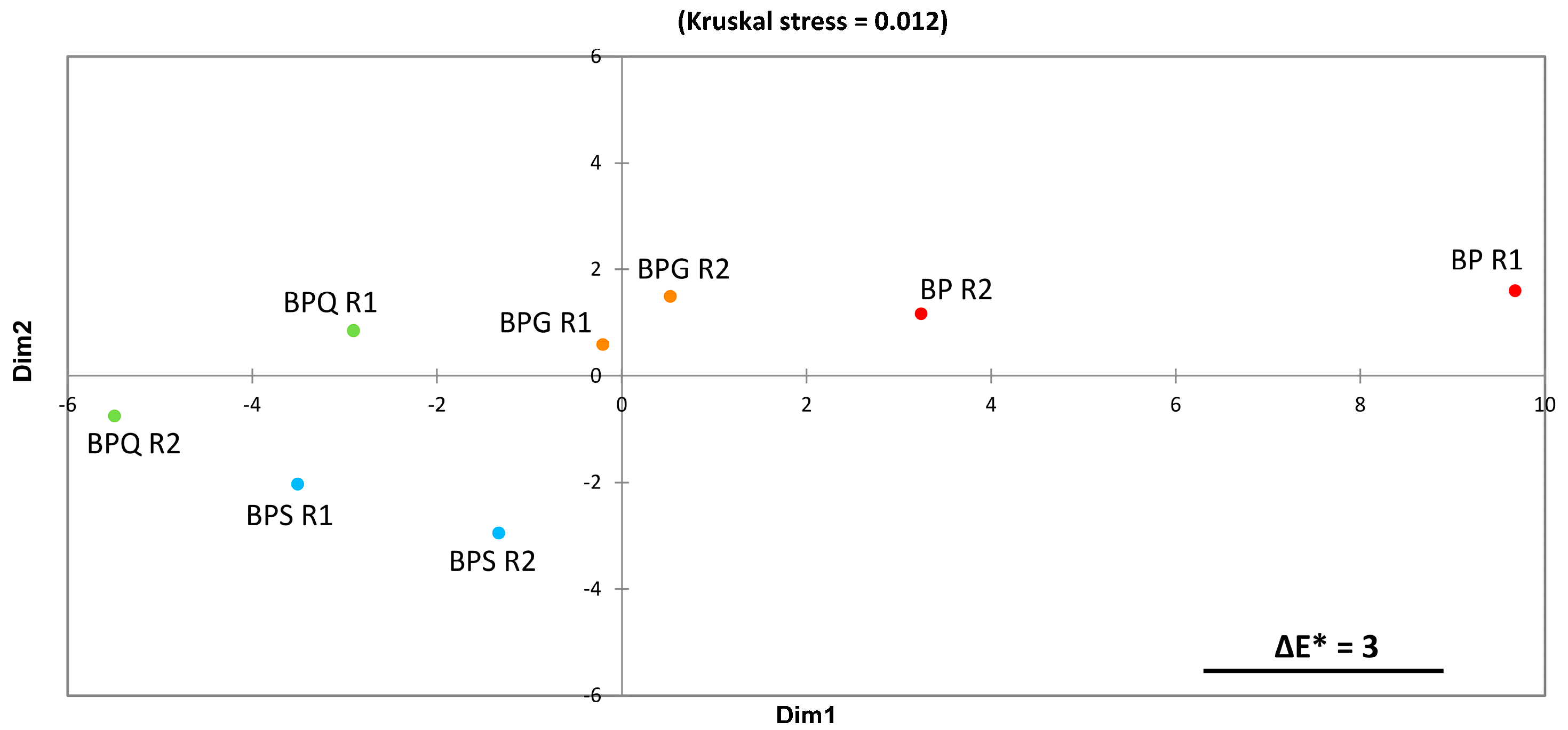
3.3. Colorimetric Analysis by Tri-Dimensional Coordinates L*a*b*
3.4. Anthocyanin Analysis and Phenolic Compound Determination
3.4.1. Anthocyanin Analysis by Spectrophotometric Analyses
3.4.2. Phenolic Compound Analyses (UHPLC Analyses)
4. Discussion
4.1. Microbial Analyses
4.2. Oenological Analysis of Wines
4.3. Wine Colour
4.4. Phenolic Composition of Wines
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roudil, L.; Russo, P.; Berbegal, C.; Albertin, W.; Spano, G.; Capozzi, V. Non-Saccharomyces Commercial Starter Cultures: Scientific Trends, Recent Patents and Innovation in the Wine Sector. Recent Pat. Food Nutr. Agric. 2020, 11, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Simonin, S.; Alexandre, H.; Nikolantonaki, M.; Coelho, C.; Tourdot-Maréchal, R. Inoculation of Torulaspora Delbrueckii as a Bio-Protection Agent in Winemaking. Food Res. Int. 2018, 107, 451–461. [Google Scholar] [CrossRef]
- Simonin, S.; Roullier-Gall, C.; Ballester, J.; Schmitt-Kopplin, P.; Quintanilla-Casas, B.; Vichi, S.; Peyron, D.; Alexandre, H.; Tourdot-Maréchal, R. Bio-Protection As an Alternative to Sulphites: Impact on Chemical and Microbial Characteristics of Red Wines. Front. Microbiol. 2020, 11, 1308. [Google Scholar] [CrossRef]
- Windholtz, S.; Dutilh, L.; Lucas, M.; Maupeu, J.; Vallet-Courbin, A.; Farris, L.; Coulon, J.; Masneuf-Pomarède, I. Population Dynamics and Yeast Diversity in Early Winemaking Stages without Sulfites Revealed by Three Complementary Approaches. Appl. Sci. 2021, 11, 2494. [Google Scholar] [CrossRef]
- Windholtz, S.; Vinsonneau, E.; Farris, L.; Thibon, C.; Masneuf-Pomarède, I. Yeast and Filamentous Fungi Microbial Communities in Organic Red Grape Juice: Effect of Vintage, Maturity Stage, SO2, and Bioprotection. Front. Microbiol. 2021, 12, 748416. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Guo, A.; Wang, H. Mechanisms of Oxidative Browning of Wine. Food Chem. 2008, 108, 1–13. [Google Scholar] [CrossRef]
- Yang, H.; Tian, T.; Gu, H.; Li, X.; Cai, G.; Sun, J.; Wu, D.; Lu, J. Analysis of Factors Related to Browning of Dangshan Pear (Pyrus Spp.) Wine. Food Chem. 2020, 308, 125665. [Google Scholar] [CrossRef]
- Oliveira, C.M.; Ferreira, A.C.S.; De Freitas, V.; Silva, A.M.S. Oxidation Mechanisms Occurring in Wines. Food Res. Int. 2011, 44, 1115–1126. [Google Scholar] [CrossRef]
- Coetzee, C. Oxidation Treatments Affecting Sauvignon Blanc Wine Sensory and Chemical Composition. PhD Thesis, Stellenbosch University, Institute for Wine Biotechnology, Faculty of AgriSciences, South Africa, 2014. [Google Scholar]
- Hoenicke, K.; Simat, T.J.; Steinhart, H.; Christoph, N.; Geßner, M.; Köhler, H.-J. ‘Untypical Aging off-Flavor’ in Wine: Formation of 2-Aminoacetophenone and Evaluation of Its Influencing Factors. Anal. Chim. Acta 2002, 458, 29–37. [Google Scholar] [CrossRef]
- Mayr, C.M.; Capone, D.L.; Pardon, K.H.; Black, C.A.; Pomeroy, D.; Francis, I.L. Quantitative Analysis by GC-MS/MS of 18 Aroma Compounds Related to Oxidative Off-Flavor in Wines. J. Agric. Food Chem. 2015, 63, 3394–3401. [Google Scholar] [CrossRef]
- Ugliano, M. Oxygen Contribution to Wine Aroma Evolution during Bottle Aging. J. Agric. Food Chem. 2013, 61, 6125–6136. [Google Scholar] [CrossRef] [PubMed]
- Simonin, S. Etude de la Bio-Protection en œnologie. Univresité de Bourgogne Franche-Compte, Dijon, France, 2019. [Google Scholar]
- Danilewicz, J.C.; Seccombe, J.T.; Whelan, J. Mechanism of Interaction of Polyphenols, Oxygen, and Sulfur Dioxide in Model Wine and Wine. Am. J. Enol. Vitic. 2008, 59, 128–136. [Google Scholar] [CrossRef]
- Carrascón, V.; Bueno, M.; Fernandez-Zurbano, P.; Ferreira, V. Oxygen and SO2 Consumption Rates in White and Rosé Wines: Relationship with and Effects on Wine Chemical Composition. J. Agric. Food Chem. 2017, 65, 9488–9495. [Google Scholar] [CrossRef]
- Carrascón, V.; Vallverdú-Queralt, A.; Meudec, E.; Sommerer, N.; Fernandez-Zurbano, P.; Ferreira, V. The Kinetics of Oxygen and SO2 Consumption by Red Wines. What Do They Tell about Oxidation Mechanisms and about Changes in Wine Composition? Food Chem. 2018, 241, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Danilewicz, J.C.; Standing, M.J. Reaction Mechanisms of Oxygen and Sulfite in Red Wine. Am. J. Enol. Vitic. 2018, 69, 189–195. [Google Scholar] [CrossRef]
- Ugliano, M.; Slaghenaufi, D.; Picariello, L.; Olivieri, G. Oxygen and SO2 Consumption of Different Enological Tannins in Relationship to Their Chemical and Electrochemical Characteristics. J. Agric. Food Chem. 2020, 68, 13418–13425. [Google Scholar] [CrossRef] [PubMed]
- OIV Code International Des Pratiques OEnologiques. Available online: https://www.oiv.int/fr/standards/code-international-des-pratiques-oenologiques (accessed on 12 December 2022).
- Amarowicz, R.; Janiak, M. Hydrolysable Tannins. Ref. Modul. Food Sci. 2018, 3, 7. [Google Scholar] [CrossRef]
- Khanbabaee, K.; van Ree, T. Tannins: Classification and Definition. Nat. Prod. Rep. 2001, 18, 641–649. [Google Scholar] [CrossRef]
- Jourdes, M.; Pouységu, L.; Deffieux, D.; Teissedre, P.-L.; Quideau, S. Hydrolyzable Tannins: Gallotannins and Ellagitannins. In Natural Products; Ramawat, K.G., Mérillon, J.-M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1975–2010. ISBN 978-3-642-22143-9. [Google Scholar]
- Versari, A.; du Toit, W.; Parpinello, G.P. Oenological Tannins: A Review: Oenological Tannins. Aust. J. Grape Wine Res. 2013, 19, 1–10. [Google Scholar] [CrossRef]
- Magalhães, L.M.; Ramos, I.I.; Reis, S.; Segundo, M.A. Antioxidant Profile of Commercial Oenological Tannins Determined by Multiple Chemical Assays. Aust. J. Grape Wine Res. 2014, 20, 72–79. [Google Scholar] [CrossRef]
- Moilanen, J.; Karonen, M.; Tähtinen, P.; Jacquet, R.; Quideau, S.; Salminen, J.-P. Biological Activity of Ellagitannins: Effects as Anti-Oxidants, pro-Oxidants and Metal Chelators. Phytochemistry 2016, 125, 65–72. [Google Scholar] [CrossRef]
- Vignault, A.; Pascual, O.; Jourdes, M.; Moine, V.; Fermaud, M.; Roudet, J.; Canals, J.M.; Teissedre, P.-L.; Zamora, F. Impact of Enological Tannins on Laccase Activity. OENO One 2019, 53, 27–38. [Google Scholar] [CrossRef]
- Gombau, J.; Vignault, A.; Pascual, O.; Gómez-Alonso, S.; Gracía-Romero, E.; Hermosín, I.; Canals, J.M.; Teissedre, P.-L.; Zamora, F. Influence of Oenological Tannins on Malvidin-3-O-Monoglucoside Copigmentation in a Model Wine Solution. OENO One 2019, 53, 531–547. [Google Scholar] [CrossRef]
- Remy, S.; Fulcrand, H.; Labarbe, B.; Cheynier, V.; Moutounet, M. First Confirmation in Red Wine of Products Resulting from Direct Anthocyanin-Tannin Reactions. J. Sci. Food Agric. 2000, 80, 745–751. [Google Scholar] [CrossRef]
- Vignault, A.; González-Centeno, M.R.; Pascual, O.; Gombau, J.; Jourdes, M.; Moine, V.; Iturmendi, N.; Canals, J.M.; Zamora, F.; Teissedre, P.-L. Chemical Characterization, Antioxidant Properties and Oxygen Consumption Rate of 36 Commercial Oenological Tannins in a Model Wine Solution. Food Chem. 2018, 268, 210–219. [Google Scholar] [CrossRef]
- Pallmann, C.L.; Brown, J.A.; Olineka, T.L.; Cocolin, L.; Mills, D.A.; Bisson, L.F. Use of WL Medium to Profile Native Flora Fermentations. Am. J. Enol. Vitic. 2001, 52, 198–203. [Google Scholar]
- Kluyver, A.J.; van der Walt, J.P.; van Triet, A.J. Pulcherrimin, The Pigment of Candida pulcherrima. Proc. Natl. Acad. Sci. USA 1953, 39, 583–593. [Google Scholar] [CrossRef]
- MacDonald, J. Biosynthesis of Pulcherriminic Acid. Biochem. J. 1965, 96, 533–538. [Google Scholar] [CrossRef]
- Gerbaux, V.; Briffox, C.; Dumont, A.; Krieger, S. Influence of Inoculation with Malolactic Bacteria on Volatile Phenols in Wines. Am. J. Enol. Vitic. 2009, 60, 233–235. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Stonestreet, E. Le Dosage Des Anthocyanes Dans Le Vin Rouge [Determination of Anthocyanins in Red Wine]. Bull Soc. Chim. Fr. 1965, 9, 2649–2652. [Google Scholar]
- Glories, Y. Recherche Sur La Matière Colorante Des Vins Rouges. Ph.D. Thesis, Université de Bordeaux II, Nouvelle-Aquitaine, France, 1978. [Google Scholar]
- Chris Somers, T.; Evans, M.E. Spectral Evaluation of Young Red Wines: Anthocyanin Equilibria, Total Phenolics, Free and Molecular SO2, “Chemical Age”. J. Sci. Food Agric. 1977, 28, 279–287. [Google Scholar] [CrossRef]
- Popîrdă, A.; Luchian, C.E.; Colibaba, L.C.; Focea, E.C.; Nicolas, S.; Noret, L.; Cioroiu, I.B.; Gougeon, R.; Cotea, V.V. Carbon-Isotope Ratio (Δ13C) and Phenolic-Compounds Analysis in Authenticity Studies of Wines from Dealu Mare and Cotnari Regions (Romania). Agronomy 2022, 12, 2286. [Google Scholar] [CrossRef]
- Martínez, J.A.; Melgosa, M.; Pérez, M.M.; Hita, E.; Negueruela, A.I. Note. Visual and Instrumental Color Evaluation in Red Wines. Food Sci. Technol. Int. 2001, 7, 439–444. [Google Scholar] [CrossRef]
- Guillamón, J.M.; Mas, A. Acetic Acid Bacteria. In Acetic Acid Bacteria; König, H., Unden, G., Fröhlich, J., Eds.; Biology of Microorganisms on Grapes, in Must and in Wine; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 10.1007/978-3-540-85463-0_2. [Google Scholar]
- Park, H.; Hwang, Y.-S. Genome-Wide Transcriptional Responses to Sulfite in Saccharomyces Cerevisiae. J. Microbiol. 2008, 46, 542–548. [Google Scholar] [CrossRef]
- Jackowetz, J.N.; Dierschke, S.; Mira de Orduña, R. Multifactorial Analysis of Acetaldehyde Kinetics during Alcoholic Fermentation by Saccharomyces Cerevisiae. Food Res. Int. 2011, 44, 310–316. [Google Scholar] [CrossRef]
- Boulton, R.B.; Singleton, V.L.; Bisson, L.F.; Kunkee, R.E. The Role of Sulfur Dioxide in Wine; Principles and Practices of Winemaking; Springer: Boston, MA, USA, 1999. [Google Scholar]
- Liu, Y.-X.; Liang, N.-N.; Wang, J.; Pan, Q.-H.; Duan, C.-Q. Effect of the Prefermentative Addition of Five Enological Tannins on Anthocyanins and Color in Red Wines. J. Food Sci. 2013, 78, C25–C30. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Escott, C.; Loira, I.; Del Fresno, J.; Morata, A.; Tesfaye, W.; Calderon, F.; Benito, S.; Suárez-Lepe, J. The Effects of Pre-Fermentative Addition of Oenological Tannins on Wine Components and Sensorial Qualities of Red Wine. Molecules 2016, 21, 1445. [Google Scholar] [CrossRef] [PubMed]
- Gombau, J.; Vignault, A.; Pascual, O.; Canals, J.M.; Teissedre, P.-L.; Zamora, F. Influence of Supplementation with Different Oenological Tannins on Malvidin-3-Monoglucoside Copigmentation. In Proceedings of the BIO Web of Conferences, 39th World Congress of Vine and Wine, online conferences, Brazil, 26 October 2016; Volume 7. [Google Scholar] [CrossRef]
- García-Estévez, I.; Alcalde-Eon, C.; Puente, V.; Escribano-Bailón, M. Enological Tannin Effect on Red Wine Color and Pigment Composition and Relevance of the Yeast Fermentation Products. Molecules 2017, 22, 2046. [Google Scholar] [CrossRef] [PubMed]
- Zaffalon, P.-L.; Dienes-Nagy, Á.; Nardone, D.; Vuichard, F.; Koestel, C.; Rösti, J. Anthocyanes libres des vins, une analyse pour différencier des cépages suisses. Rev. Suisse Vitic. Arboric. Hortic. 2014, 46, 310–316. [Google Scholar]
- Ribéreau-Gayon, P.; Dubourbieu, D.; Donèche, B.; Lonvaud, A. Handbook of Enology, Volume 1: The Microbiology of Wines and Vinification; John Wiley and Sons Ltd.: Chichester, UK, 2006. [Google Scholar]
- Waterhouse, A.L.; Sacks, G.L.; Jeffery, D.W. Understanding Wine Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2016; ISBN 978-1-118-73072-0. [Google Scholar]
| Tank * | Day | Yeats (CFU/mL) | Metschnikowia pulcherrima (%) | Saccharomyces (%) | Hanseniaspora (%) | Lactic Acid Bacteria (CFU/mL) | Acetic Acid Bacteria (CFU/mL) |
|---|---|---|---|---|---|---|---|
| BP | 1 | 3.2 ± 0.54 × 107 ** | 2.45 ± 0.47 | 95.10 ± 0.93 | 2.45 ± 0.47 | 683 ± 24 | 1600 ± 471 |
| BPG | 1 | 7.6 ± 0.50 × 107 | 7.15 ± 4.21 | 91.53 ± 4.13 | 1.33 ± 0.09 | 550 ± 24 | 1517 ± 212 |
| BPQ | 1 | 1.1 ± 1.3 × 108 | 3.34 ± 2.67 | 95.64 ± 2.05 | 1.02 ± 0.62 | 567 ± 47 | 1500 ± 189 |
| BPS | 1 | 5.4 ± 5.3 × 107 | 7.53 ± 0.23 | 91.31 ± 0.14 | 1.16 ± 0.09 | 567 ± 47 | 1267 ± 189 |
| BP | 2 | 2.8 ± 0.69 × 108 | ND *** | 100 ± 0 | ND | 50 ± 24 | 100 ± 47 |
| BPG | 2 | 2.3 ± 0.02 × 108 | 0.22 ± 0.31 | 99.78 ± 0.31 | ND | ND | ND |
| BPQ | 2 | 2.4 ± 0.02 × 108 | 0.21 ± 0.30 | 99.79 ± 0.30 | ND | 83 ± 24 | ND |
| BPS | 2 | 2.2 ± 0.18 × 108 | ND | 100 ± 0 | ND | 83 ± 24 | ND |
| BP | 4 | 2.9 ± 0.96 × 108 | ND | 100 ± 0 | ND | 167 ± 47 | 817 ± 24 |
| BPG | 4 | 2.7 ± 0.16 × 108 | ND | 100 ± 0 | ND | 600 ± 566 | 2133 ± 2923 |
| BPQ | 4 | 3.3 ± 0.15 × 108 | ND | 100 ± 0 | ND | 417 ± 354 | 4733 ± 6505 |
| BPS | 4 | 3.2 ± 0.12 × 108 | ND | 100 ± 0 | ND | 1383 ± 1815 | 300 ± 283 |
| Condition * | Total SO2 (mg/L) | Bound SO2 (mg/L) | Free SO2 (mg/L) |
|---|---|---|---|
| BP | 22.58 ± 0.86 ab ** | 15.58 ± 1.25 a | 7.00 ± 0.59 a |
| BPG | 18.64 ± 4.17 a | 11.55 ± 4.05 a | 6.20 ± 0.63 a |
| BPQ | 19.79 ± 3.12 a | 13.80 ± 3.17 a | 6.03 ± 0.46 a |
| BPS | 27.25 ± 2.42 b | 20.55 ± 2.83 b | 6.70 ± 0.97 a |
| Spectrophotometric Analyses | UHPLC Analyses | ||||
|---|---|---|---|---|---|
| Total Anthocyanins (mg/L) | Free Anthocyanins (mg/L) | Combined Anthocyanins (mg/L) | Ionisation Index (%) | Anthocyanins (mg/L of Malvidine-3-O-glucoside) | |
| BP * | 16.19 ± 3.09 ** | 4.67 ± 0.82 | 11.52 ± 2.27 | 34.32 ± 22.88 | 1.07 ± 0.04 |
| BPG | 17.50 ± 3.71 | 2.63 ± 2.47 | 14.88 ± 6.16 | 47.95 ± 0.53 | 0.91 ± 0.08 |
| BPQ | 16.63 ± 0.00 | 2.19 ± 3.09 | 14.44 ± 3.09 | 48.62 ± 0.88 | 0.96 ± 0.12 |
| BPS | 18.38 ± 2.47 | 2.19 ± 0.62 | 16.19 ± 1.86 | 24.40 ± 34.51 | 1.16 ± 0.22 |
| Compounds (mg/L) | BP * | BPG | BPQ | BPS |
|---|---|---|---|---|
| Caffeic acid | 1.70 ± 0.08 a ** | 1.66 ± 0.02 a | 1.66 ± 0.06 a | 1.64 ± 0.12 a |
| Caftaric acid | 63.54 ± 1.50 a | 61.86 ± 1.93 a | 63.11 ± 1.62 a | 63.61 ± 1.21 a |
| Coumaric acid | 0.30 ± 0.01 a | 0.32 ± 0.03 ab | 0.38 ± 0 b | 0.35 ± 0 ab |
| Coutaric acid | 11.20 ± 0.64 a | 11.19 ± 0.42 a | 11.74 ± 0.01 a | 12.58 ± 0.57 a |
| Gallic acid | 0.30 ± 0.01 b | 11.99 ± 1.22 a | 1.69 ± 0.73 b | 0.33 ± 0.02 b |
| Gentisic acid | 0.34 ± 0.01 a | 0.34 ± 0 a | 0.40 ± 0.02 ab | 0.48 ± 0.04 b |
| Hydroxybenzoic acid | 0.38 ± 0.07 a | 0.41 ± 0.02 a | 0.44 ± 0.05 a | 0.51 ± 0.01 a |
| Protocatechuic acid | 1.80 ± 0.37 a | 2.01 ± 0.02 a | 2.34 ± 0.12 a | 1.83 ± 0.11 a |
| Catechin | 1.44 ± 0 a | 1.40 ± 0.04 a | 1.36 ± 0.07 a | 1.54 ± 0.19 a |
| B1 dimer | 0.08 ± 0.01 a | 0.07 ± 0.01 a | 0.06 ± 0.01 a | 0.07 ± 0.02 a |
| B2 dimer | 0.02 ± 0.02 a | 0.02 ± 0.01 a | 0.02 ± 0 a | 0.02 ± 0.01 a |
| Epicatechin | 0.20 ± 0 a | 0.18 ± 0 a | 0.16 ± 0.01 a | 0.11 ± 0.13 a |
| Grape Reaction Product | 14.82 ± 0.81 a | 14.74 ± 0.35 a | 14.82 ± 0.1 a | 14.70 ± 0.16 a |
| Hydroxytyrosol | 0.39 ± 0.05 a | 0.37 ± 0 a | 0.39 ± 0.01 a | 0.42 ± 0 a |
| Tyrosol | 9.64 ± 0.26 a | 9.56 ± 0.03 a | 9.22 ± 0.25 a | 8.96 ± 0.5 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puyo, M.; Simonin, S.; Klein, G.; David-Vaizant, V.; Quijada-Morín, N.; Alexandre, H.; Tourdot-Maréchal, R. Use of Oenological Tannins to Protect the Colour of Rosé Wine in a Bioprotection Strategy with Metschnikowia pulcherrima. Foods 2023, 12, 735. https://doi.org/10.3390/foods12040735
Puyo M, Simonin S, Klein G, David-Vaizant V, Quijada-Morín N, Alexandre H, Tourdot-Maréchal R. Use of Oenological Tannins to Protect the Colour of Rosé Wine in a Bioprotection Strategy with Metschnikowia pulcherrima. Foods. 2023; 12(4):735. https://doi.org/10.3390/foods12040735
Chicago/Turabian StylePuyo, Maëlys, Scott Simonin, Géraldine Klein, Vanessa David-Vaizant, Natalia Quijada-Morín, Hervé Alexandre, and Raphaëlle Tourdot-Maréchal. 2023. "Use of Oenological Tannins to Protect the Colour of Rosé Wine in a Bioprotection Strategy with Metschnikowia pulcherrima" Foods 12, no. 4: 735. https://doi.org/10.3390/foods12040735
APA StylePuyo, M., Simonin, S., Klein, G., David-Vaizant, V., Quijada-Morín, N., Alexandre, H., & Tourdot-Maréchal, R. (2023). Use of Oenological Tannins to Protect the Colour of Rosé Wine in a Bioprotection Strategy with Metschnikowia pulcherrima. Foods, 12(4), 735. https://doi.org/10.3390/foods12040735






