Production of Fish Analogues from Plant Proteins: Potential Strategies, Challenges, and Outlook
Abstract
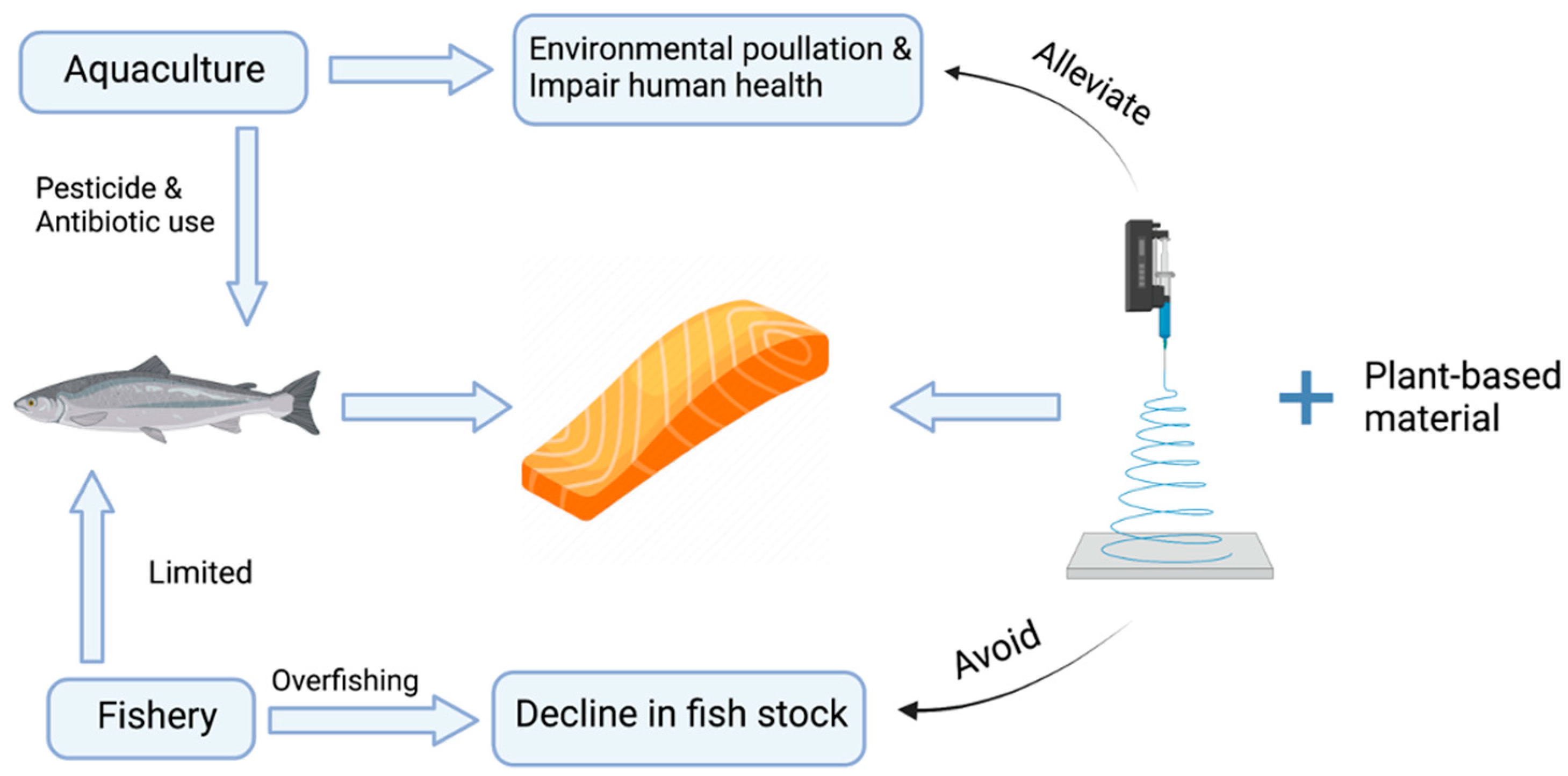
1. Present Global Seafood Production
2. Major Composition of Fish
2.1. Protein Composition
2.2. Protein Quality
| Product Type | PDCAAS (after Truncation) | Reference |
|---|---|---|
| Cod | 0.96 | [27] |
| Tuna | 0.97 | [28] |
| Salmon | 1 | [20] |
| Herring | 1 | [20] |
| Mackerel | 1 | [20] |
| Trout | 0.998 | [19] |
| Beef | 1 | [22] |
| Casein | 0.99 | [28] |
| Soy protein | 1 | [23] |
| Chickpea protein | 0.66 | [28] |
| Lentils | 0.52 | [28] |
| Kidney beans | 0.68 | [28] |
3. Fish Muscle Structure
3.1. Fish Muscle Structure
3.2. Fish Muscle Alignment
4. Processing Techniques to Produce Fish Analogue
4.1. Electrospinning
4.2. Wet Spinning
4.3. 3D-Printing
4.4. Extrusion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- The World Bank Population. Available online: https://data.worldbank.org/indicator/SP.POP.TOTL (accessed on 20 April 2022).
- Ritchie, H.; Roser, M. Fish and Overfishing. Available online: https://ourworldindata.org/fish-and-overfishing#how-much-fish-do-people-eat (accessed on 20 April 2022).
- World Wild Life Overfishing. Available online: https://www.worldwildlife.org/threats/overfishing (accessed on 20 April 2022).
- FAO. The State of World Fisheries and Aquaculture 2020. Available online: https://www.fao.org/state-of-fisheries-aquaculture (accessed on 20 April 2022).
- Naylor, R.L.; Goldburg, R.J.; Primavera, J.H.; Kautsky, N.; Beveridge, M.C.M.; Clay, J.; Folke, C.; Lubchenco, J.; Mooney, H.; Troell, M. Effect of Aquaculture on World Fish Supplies. Nature 2000, 405, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Cole, D.W.; Cole, R.; Gaydos, S.J.; Gray, J.; Hyland, G.; Jacques, M.L.; Powell-Dunford, N.; Sawhney, C.; Au, W.W. Aquaculture: Environmental, Toxicological, and Health Issues. Int. J. Hyg. Environ. Health 2009, 212, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Sheikh Abdullah, S.R.; Hasan, H.A.; Othman, A.R.; Ismail, N. Izzati Aquaculture Industry: Supply and Demand, Best Practices, Effluent and Its Current Issues and Treatment Technology. J. Environ. Manag. 2021, 287, 112271. [Google Scholar] [CrossRef] [PubMed]
- Vollmann, J.; Fritz, C.N.; Wagentristl, H.; Ruckenbauer, P. Environmental and Genetic Variation of Soybean Seed Protein Content under Central European Growing Conditions. J. Sci. Food Agric. 2000, 80, 1300–1306. [Google Scholar] [CrossRef]
- Serkissian, Madeleine Seven of the Biggest Problems Facing Fish in Our Oceans. Available online: https://marine-conservation.org/on-the-tide/seven-of-the-biggest-problems-facing-fish-in-our-oceans (accessed on 16 January 2023).
- Heal, G.; Schlenker, W. Sustainable Fisheries. Nature 2008, 455, 1044–1045. [Google Scholar] [CrossRef]
- Wanjala, Caiaphas Top 10 Most Consumed Fish in the World in 2021. Available online: https://www.tuko.co.ke/406008-top-10-consumed-fish-world-2021-photos.html (accessed on 31 December 2022).
- Ghaedian, R.; Decker, E.A.; McClements, D.J. Use of Ultrasound to Determine Cod Fillet Composition. J. Food Sci. 1997, 62, 500–504. [Google Scholar] [CrossRef]
- Isaksson, T.; Tøgersen, G.; Iversen, A.; Hildrum, K.I. Non-destructive Determination of Fat, Moisture and Protein in Salmon Fillets by Use of Near-infrared Diffuse Spectroscopy. J. Sci. Food Agric. 1995, 69, 95–100. [Google Scholar] [CrossRef]
- Khodabux, K.; L’Omelette, M.S.S.; Jhaumeer-Laulloo, S.; Ramasami, P.; Rondeau, P. Chemical and Near-Infrared Determination of Moisture, Fat and Protein in Tuna Fishes. Food Chem. 2007, 102, 669–675. [Google Scholar] [CrossRef]
- Petricorena, Z.C. Chemical composition of fish and fishery products. In Handbook of Food Chemistry; Springer: Berlin/Heidelberg, Germany, 2015; pp. 403–435. ISBN 9783642366055. [Google Scholar]
- Venugopal, V.; Shahidi, F. Structure and Composition of Fish Muscle. Food Rev. Int. 1996, 12, 175–197. [Google Scholar] [CrossRef]
- Tahergorabi, R.; Hosseini, S.V.; Jaczynski, J. Seafood Proteins; Woodhead Publishing Limited: Sawston, UK, 2011. [Google Scholar]
- Schaafsma, G. The Protein Digestibility-Corrected Amino Acid Score (PDCAAS)—A Concept for Describing Protein Quality in Foods and Food Ingredients: A Critical Review. J. AOAC Int. 2005, 88, 988–994. [Google Scholar] [CrossRef]
- El, S.N.; Kavas, A. Determination of Protein Quality of Rainbow Trout (Salmo Irideus) by in Vitro Protein Digestibility-Corrected Amino Acid Score (PDCAAS). Food Chem. 1996, 55, 221–223. [Google Scholar] [CrossRef]
- Usydus, Z.; Szlinder-Richert, J.; Adamczyk, M. Protein Quality and Amino Acid Profiles of Fish Products Available in Poland. Food Chem. 2009, 112, 139–145. [Google Scholar] [CrossRef]
- Pyz-Łukasik, R.; Paszkiewicz, W. Species Variations in the Proximate Composition, Amino Acid Profile, and Protein Quality of the Muscle Tissue of Grass Carp, Bighead Carp, Siberian Sturgeon, and Wels Catfish. J. Food Qual. 2018, 2018, 2625401. [Google Scholar] [CrossRef]
- Ertl, P.; Knaus, W.; Zollitsch, W. An Approach to Including Protein Quality When Assessing the Net Contribution of Livestock to Human Food Supply. Animal 2016, 10, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.J.; Ryan, D.J.; Mukherjea, R.; Schasteen, C.S. Protein Digestibility-Corrected Amino Acid Scores (PDCAAS) for Soy Protein Isolates and Concentrate: Criteria for Evaluation. J. Agric. Food Chem. 2011, 59, 12707–12712. [Google Scholar] [CrossRef]
- Mariotti, F. Plant Protein, Animal Protein, and Protein Quality; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780128039694. [Google Scholar]
- Berry, T.H.; Becker, D.E.; Rasmussen, O.G.; Jensen, A.H.; Norton, H.W. The Limiting Amino Acids in Soybean Protein. J. Anim. Sci. 1962, 21, 558–561. [Google Scholar] [CrossRef]
- Nikmaram, N.; Leong, S.Y.; Koubaa, M.; Zhu, Z.; Barba, F.J.; Greiner, R.; Oey, I.; Roohinejad, S. Effect of Extrusion on the Anti-Nutritional Factors of Food Products: An Overview. Food Control 2017, 79, 62–73. [Google Scholar] [CrossRef]
- Nigam, P.S.; Singh, A. Single Cell Protein: Mycelial Fungi, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 3, ISBN 9780123847331. [Google Scholar]
- Sarwar, G.; Peace, R.W.; Botting, H.G.; Brulé, D. Digestibility of Protein and Amino Acids in Selected Foods as Determined by a Rat Balance Method. Plant Foods Hum. Nutr. 1989, 39, 23–32. [Google Scholar] [CrossRef]
- Kazir, M.; Livney, Y.D. Plant-Based Seafood Analogs. Molecules 2021, 26, 1559. [Google Scholar] [CrossRef]
- Torres, J.A.; Rodrigo-García, J.; Jaczynski, J.; Chen, Y.C. Recovery of By-Products from Seafood Processing Streams. In Maximising Value Marine By-Products; Woodhead Publishing Limited: Sawston, UK, 2006; pp. 65–90. [Google Scholar] [CrossRef]
- Kiessling, A.; Ruohonen, K.; Bjørnevik, M. Muscle Fibre Growth and Quality in Fish. Arch. Tierz. Dummerstorf 2016, 49, 137–146. [Google Scholar]
- Coughlin, D.J.; Rome, L.C. The Roles of Pink and Red Muscle in Powering Steady Swimming in Scup, Stenotomus Chrysops. Am. Zool. 1996, 36, 666–677. [Google Scholar] [CrossRef]
- Nowsad, A. Structure of Fish Muscles and Composition of Fish. In Participatory Training of Trainers- A New Approach Applied in Fish Processing; Bangladesh Fisheries Research Forum: Dhaka, Bangladesh, 2007; p. 329. [Google Scholar]
- Johnston, I.A.; Davison, W.; Goldspink, G. Energy Metabolism of Carp Swimming Muscles. J. Comp. Physiol. B 1977, 114, 203–216. [Google Scholar] [CrossRef]
- Listrat, A.; Lebret, B.; Louveau, I.; Astruc, T.; Bonnet, M.; Lefaucheur, L.; Bugeon, J. How Muscle Structure and Composition Determine Meat Quality. Prod. Anim. 2015, 28, 125–136. [Google Scholar]
- Mascarello, E.; Romanello, M.G.; Scapolo, P.A. Histochemical and Immunohistochemical Profile of Pink Muscle Fibres in Some Teleosts. Histochemistry 1986, 84, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Tahergorabi, R.; Jaczynski, J. Physicochemical Changes in Surimi with Salt Substitute. Food Chem. 2012, 132, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Hultin, H.O. Postmortem Biochemistry of Meat and Fish. J. Chem. Educ. 1984, 61, 289–298. [Google Scholar] [CrossRef]
- Sato, K.; Yoshinaka, R.; Sato, M.; Shimizu, Y. Collagen Content in the Muscle of Fishes in Association with Their Swimming Movement and Meat Texture. Nippon SUISAN GAKKAISHI 1986, 52, 1595–1600. [Google Scholar] [CrossRef]
- FAO Quality and Quality Changes in Fresh Fish. Available online: https://www.fao.org/3/V7180E/V7180E04.htm (accessed on 20 April 2022).
- LAMPILA, L.E. Comparative Microstructure of Red Meat, Poultry and Fish Muscle. J. Muscle Foods 1990, 1, 247–267. [Google Scholar] [CrossRef]
- Dekkers, B.L.; Boom, R.M.; van der Goot, A.J. Structuring Processes for Meat Analogues. Trends Food Sci. Technol. 2018, 81, 25–36. [Google Scholar] [CrossRef]
- Austin, Mark Vegan Fish Flavors That Deliver More, Naturally. Available online: https://www.dsm.com/food-beverage/en_US/insights/insights/savory/plant-based-seafood.html (accessed on 31 December 2022).
- Wongkanya, R.; Chuysinuan, P.; Pengsuk, C.; Techasakul, S.; Lirdprapamongkol, K.; Svasti, J.; Nooeaid, P. Electrospinning of Alginate/Soy Protein Isolated Nanofibers and Their Release Characteristics for Biomedical Applications. J. Sci. Adv. Mater. Devices 2017, 2, 309–316. [Google Scholar] [CrossRef]
- Rampon, V.; Robert, P.; Nicolas, N.; Dufour, E. Protein Structure and Network Orientation in Edible Films Prepared by Spinning Process. J. Food Sci. 1999, 64, 313–316. [Google Scholar] [CrossRef]
- Wittek, P.; Ellwanger, F.; Karbstein, H.P.; Emin, M.A. Morphology Development and Flow Characteristics during High Moisture Extrusion of a Plant-Based Meat Analogue. Foods 2021, 10, 1753. [Google Scholar] [CrossRef] [PubMed]
- Grahl, S.; Palanisamy, M.; Strack, M.; Meier-Dinkel, L.; Toepfl, S.; Mörlein, D. Towards More Sustainable Meat Alternatives: How Technical Parameters Affect the Sensory Properties of Extrusion Products Derived from Soy and Algae. J. Clean. Prod. 2018, 198, 962–971. [Google Scholar] [CrossRef]
- Phuhongsung, P.; Zhang, M.; Devahastin, S. Investigation on 3D Printing Ability of Soybean Protein Isolate Gels and Correlations with Their Rheological and Textural Properties via LF-NMR Spectroscopic Characteristics. LWT 2020, 122, 109019. [Google Scholar] [CrossRef]
- Ramos Diaz, J.M.; Kantanen, K.; Edelmann, J.M.; Suhonen, H.; Sontag-Strohm, T.; Jouppila, K.; Piironen, V. Fibrous Meat Analogues Containing Oat Fiber Concentrate and Pea Protein Isolate: Mechanical and Physicochemical Characterization. Innov. Food Sci. Emerg. Technol. 2022, 77, 102954. [Google Scholar] [CrossRef]
- Avebe; Kyriakopoulou, K.; Keppler, J.K.; Van Der Goot, A.J. Vegan Meat Analogues. Foods 2020, 10, 9147. [Google Scholar]
- Karefyllakis, D.; van der Goot, A.J.; Nikiforidis, C.V. Multicomponent Emulsifiers from Sunflower Seeds. Curr. Opin. Food Sci. 2019, 29, 35–41. [Google Scholar] [CrossRef]
- Ramachandraiah, K. Potential Development of Sustainable 3d-Printed Meat Analogues: A Review. Sustainablity 2021, 13, 938. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A Fascinating Fiber Fabrication Technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef]
- Vega-Lugo, A.-C.; Lim, L.-T. Electrospinning of Soy Protein Isolate Nanofibers. J. Biobased Mater. Bioenergy 2008, 2, 223–230. [Google Scholar] [CrossRef]
- Cui, B.; Liang, H.; Li, J.; Zhou, B.; Chen, W.; Liu, J.; Li, B. Development and Characterization of Edible Plant-Based Fibers Using a Wet-Spinning Technique. Food Hydrocoll. 2022, 133, 107965. [Google Scholar] [CrossRef]
- Sun, J.; Zhou, W.; Yan, L.; Huang, D.; Lin, L.-y. Extrusion-Based Food Printing for Digitalized Food Design and Nutrition Control. J. Food Eng. 2018, 220, 1–11. [Google Scholar] [CrossRef]
- Lavielle, N.; Hébraud, A.; Schlatter, G.; Thöny-Meyer, L.; Rossi, R.M.; Popa, A.M. Simultaneous Electrospinning and Electrospraying: A Straightforward Approach for Fabricating Hierarchically Structured Composite Membranes. ACS Appl. Mater. Interfaces 2013, 5, 10090–10097. [Google Scholar] [CrossRef] [PubMed]
- Ozipek, B.; Karakas, H. Wet Spinning of Synthetic Polymer Fibers. In Advances in Filament Yarn Spinning of Textiles and Polymers; Elsevier: Amsterdam, The Netherlands, 2014; pp. 174–186. ISBN 9780857094995. [Google Scholar]
- Feneuil, B.; Strøm Lillebø, E.; Honstad, C.L.; Jensen, A.; Carlson, A. Elastic Modulus Measurements of Cooked Lutefisk. Phys. Fluids 2022, 34, 047122. [Google Scholar] [CrossRef]
- James, B.; Yang, S.W. Testing Meat Tenderness Using an in Situ Straining Stage with Variable Pressure Scanning Electron Microscopy. Procedia Food Sci. 2011, 1, 258–266. [Google Scholar] [CrossRef]
- Boyer, R.A. High Protein Food Product and Process for Its Preparation. US Patent 2,682,466, 1954. [Google Scholar]
- TOLSTOGUZOV, V.B. CREATION OF FIBROUS STRUCTURES BY SPINNERETLESS SPINNING. In Food Structure; Elsevier: Amsterdam, The Netherlands, 1988; pp. 181–196. [Google Scholar]
- Dick, A.; Bhandari, B.; Prakash, S. 3D Printing of Meat. Meat Sci. 2019, 153, 35–44. [Google Scholar] [CrossRef]
- Chen, J.; Mu, T.; Goffin, D.; Blecker, C.; Richard, G.; Richel, A.; Haubruge, E. Application of Soy Protein Isolate and Hydrocolloids Based Mixtures as Promising Food Material in 3D Food Printing. J. Food Eng. 2019, 261, 76–86. [Google Scholar] [CrossRef]
- Shahbazi, M.; Jäger, H.; Ettelaie, R.; Chen, J. Construction of 3D Printed Reduced-Fat Meat Analogue by Emulsion Gels. Part I: Flow Behavior, Thixotropic Feature, and Network Structure of Soy Protein-Based Inks. Food Hydrocoll. 2021, 120, 106967. [Google Scholar] [CrossRef]
- Godoi, F.C.; Prakash, S.; Bhandari, B.R. 3d Printing Technologies Applied for Food Design: Status and Prospects. J. Food Eng. 2016, 179, 44–54. [Google Scholar] [CrossRef]
- Shand, P.J.; Ya, H.; Pietrasik, Z.; Wanasundara, P.K.J.P.D. Transglutaminase Treatment of Pea Proteins: Effect on Physicochemical and Rheological Properties of Heat-Induced Protein Gels. Food Chem. 2008, 107, 692–699. [Google Scholar] [CrossRef]
- Dong, X.; Pan, Y.; Zhao, W.; Huang, Y.; Qu, W.; Pan, J.; Qi, H.; Prakash, S. Impact of Microbial Transglutaminase on 3D Printing Quality of Scomberomorus Niphonius Surimi. LWT 2020, 124, 109123. [Google Scholar] [CrossRef]
- Wang, T.; Kaur, L.; Furuhata, Y.; Aoyama, H.; Singh, J. 3D Printing of Textured Soft Hybrid Meat Analogues. Foods 2022, 11, 478. [Google Scholar] [CrossRef] [PubMed]
- Wittek, P.; Zeiler, N.; Karbstein, H.P.; Emin, M.A. High Moisture Extrusion of Soy Protein: Investigations on the Formation of Anisotropic Product Structure. Foods 2021, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, M.; Bhandari, B. 3D Printing of Steak-Like Foods Based on Textured Soybean Protein. Foods 2021, 10, 2011. [Google Scholar] [CrossRef] [PubMed]
- Cheftel, J.C.; Kitagawa, M.; Queguiner, C. New Protein Texturization Processes by Extrusion Cooking at High Moisture Levels. Food Rev. Int. 1992, 8, 235–275. [Google Scholar] [CrossRef]
- Feng, Y.; Lee, Y. Effect of Specific Mechanical Energy on In-Vitro Digestion and Physical Properties of Extruded Rice-Based Snacks. Food Nutr. Sci. 2014, 05, 1818–1827. [Google Scholar] [CrossRef]
- Zahari, I.; Ferawati, F.; Helstad, A.; Ahlström, C.; Östbring, K.; Rayner, M.; Purhagen, J.K. Development of High-Moisture Meat Analogues with Hemp and Soy Protein Using Extrusion Cooking. Foods 2020, 9, 772. [Google Scholar] [CrossRef]
- Lin, S.; Huff, H.E.; Hsieh, F. Extrusion Process Parameters, Sensory Characteristics, and Structural Properties of a High Moisture Soy Protein Meat Analog. J. Food Sci. 2002, 67, 1066–1072. [Google Scholar] [CrossRef]
- Chen, F.L.; Wei, Y.M.; Zhang, B. Chemical Cross-Linking and Molecular Aggregation of Soybean Protein during Extrusion Cooking at Low and High Moisture Content. LWT 2011, 44, 957–962. [Google Scholar] [CrossRef]
- Samard, S.; Ryu, G.H. Physicochemical and Functional Characteristics of Plant Protein-Based Meat Analogs. J. Food Process. Preserv. 2019, 43, e14123. [Google Scholar] [CrossRef]
- McClements, D.J.; Grossmann, L. The Science of Plant-Based Foods: Constructing next-Generation Meat, Fish, Milk, and Egg Analogs. Compr. Rev. Food Sci. Food Saf. 2021, 20, 4049–4100. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, V.J.; Garcia, C.E.R. Annatto Carotenoids as Additives Replacers in Meat Products. In Alternative and Replacement Foods; Elsevier: Amsterdam, The Netherlands, 2018; pp. 355–384. [Google Scholar]
- Mottram, D.S.; Madruga, M.S.; Whitfield, F.B. Some Novel Meatlike Aroma Compounds from the Reactions of Alkanediones with Hydrogen Sulfide and Furanthiols. J. Agric. Food Chem. 1995, 43, 189–193. [Google Scholar] [CrossRef]
- Beniwal, A.S.; Singh, J.; Kaur, L.; Hardacre, A.; Singh, H. Meat Analogs: Protein Restructuring during Thermomechanical Processing. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1221–1249. [Google Scholar] [CrossRef] [PubMed]
- Preece, K.E.; Hooshyar, N.; Zuidam, N.J. Whole Soybean Protein Extraction Processes: A Review. Innov. Food Sci. Emerg. Technol. 2017, 43, 163–172. [Google Scholar] [CrossRef]


| Fish | Moisture (%) | Protein (%) | Fat (%) | References |
|---|---|---|---|---|
| Cod, Atlantic (Gadus morhua) | 80.8 | 18.2 | 0.11 | [12] |
| Atlantic salmon (Salmo salar) | 65.6 | 19.6 | 14.4 | [13] |
| Tuna, yellowfin (Thunnus albacares) | 70.3–72.7 | 21.8–25.1 | 3.1–5.7 | [14] |
| Tuna, skipjack (Katsuwonus pelamis) | 66.5–72.3 | 20.72–27.98 | 3.47–5.73 | [14] |
| Processing Techniques | Raw Material | Products | References |
|---|---|---|---|
| Electrospinning | Soy protein isolate and alginate | Nanofibers | [44] |
| Wet spinning | Soy protein isolate | Edible films | [45] |
| Extrusion | Hemp and soy protein | Meat analogue | [46] |
| Extrusion | Soy and algae | Meat analogue | [47] |
| 3D printing | Soy protein isolate and xanthan gum | Gels | [48] |
| Extrusion | Pea protein and out fiber | Fibrous Meat analogue | [49] |
| Ingredients | Functionality | Example of Food | Reference |
|---|---|---|---|
| Polysaccharide (Carrageenan; Methylcellulose) | Texture enhancement; thickening agent | Meat analogues | [50] |
| Oleosomes | Emulsification/Stabilization | Meat analogues | [51] |
| Plant fat (Coconut oil; Canola oil; Sunflower oil) | Texture (juiciness) enhancement | Plant-based burger | [50] |
| Leghemoglobin | Meat flavor | Plant-based meat | [52] |
| Maxavor® Fish YE | Fish flavor | Plant-based seafood | [43] |
| Techniques | Plant Protein Sources | Processing Parameters | Images | Ref. |
|---|---|---|---|---|
| High moisture extrusion | Soy protein isolate | Extrusion temperature = 95 °C, Screw speed = 600 rpm, Moisture content = 57% |  | [46] |
| 3D printing | Pea protein isolate | Nozzle diameter = 1.54 mm, Extrusion speed = 15 mm/s |  | [69] |
| High moisture extrusion | Soy protein isolate | Extrusion temperature = 124 °C, Screw speed = 250 rpm, Water content = 50% |  | [70] |
| 3D printing | Textured soybean protein | Nozzle diameter = 0.8 mm, Extrusion speed = 20 mm/s |  | [71] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhong, C.; Feng, Y.; Xu, Y. Production of Fish Analogues from Plant Proteins: Potential Strategies, Challenges, and Outlook. Foods 2023, 12, 614. https://doi.org/10.3390/foods12030614
Zhong C, Feng Y, Xu Y. Production of Fish Analogues from Plant Proteins: Potential Strategies, Challenges, and Outlook. Foods. 2023; 12(3):614. https://doi.org/10.3390/foods12030614
Chicago/Turabian StyleZhong, Chengxuan, Yiming Feng, and Yixiang Xu. 2023. "Production of Fish Analogues from Plant Proteins: Potential Strategies, Challenges, and Outlook" Foods 12, no. 3: 614. https://doi.org/10.3390/foods12030614
APA StyleZhong, C., Feng, Y., & Xu, Y. (2023). Production of Fish Analogues from Plant Proteins: Potential Strategies, Challenges, and Outlook. Foods, 12(3), 614. https://doi.org/10.3390/foods12030614






