Snapshot of the Probiotic Potential of Kluveromyces marxianus DMKU-1042 Using a Comparative Probiogenomics Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strain, Growth Media, and Culture Condition
2.2. Genomic DNA Isolation and Library Preparation
2.3. Data for Comparative Analysis
2.4. Genome Assembly and Annotation
2.5. Phylogenetic and Average Nucleotide Identity (ANI) Analysis
2.6. Pangenome Analysis
3. Results and Discussion
3.1. Comparative Genomic Analyses
3.2. Genome Information/Chromosomal Properties
3.3. ANI, Phylogenetic Analyses and Whole-Genome Comparison of Kluyveromyces marxianus DMKU3-1042 Strain
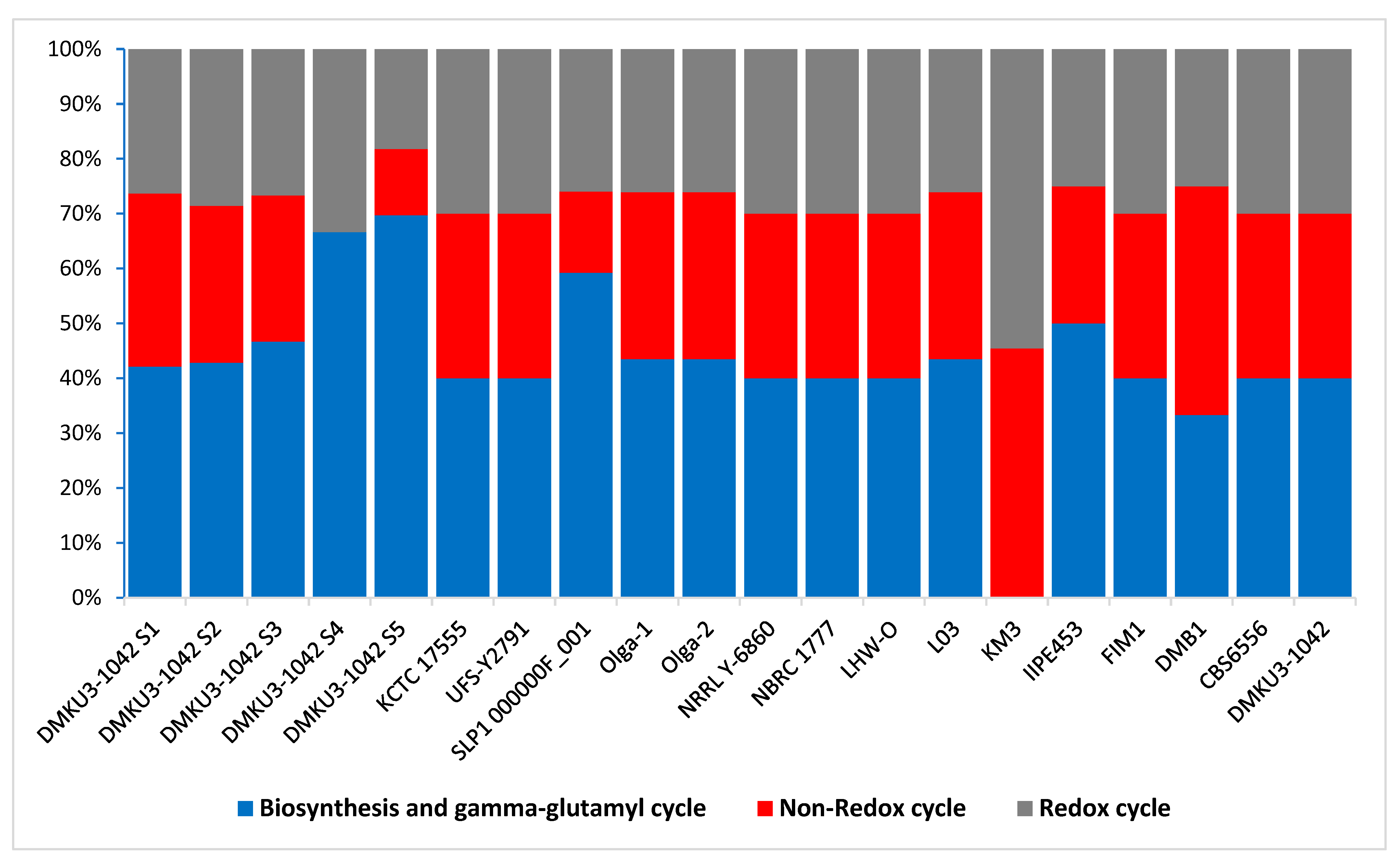
3.4. Functional Categories Related to Probiotic Capabilities
3.5. Carbohydrate Utilization and Fermentation in Kluyveromyces marxianus Strains (Genes Relevant for Sugar Utilization)
3.6. Fermentation
3.7. Vitamins, Coenzymes, and Prosthetic Groups
3.8. Stress Response and Defense Mechanisms
3.9. Amino Acids
3.10. Antioxidants, Anticoagulants, and Anti-Inflammatory Related Genes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, S.Y.; Mattanovich, D.; Villaverde, A. Systems metabolic engineering, industrial biotechnology and microbial cell factories. Microb. Cell Fact. 2012, 11, 156. [Google Scholar] [CrossRef] [PubMed]
- Abid, R.; Waseem, H.; Ali, J.; Ghazanfar, S.; Muhammad Ali, G.; Elasbali, A.M.; Alharethi, S.H.J.J.o.F. Probiotic yeast Saccharomyces: Back to nature to improve human health. J. Fungi 2022, 8, 444. [Google Scholar] [CrossRef] [PubMed]
- Delgado, S.; Sánchez, B.; Margolles, A.; Ruas-Madiedo, P.; Ruiz, L.J.N. Molecules produced by probiotics and intestinal microorganisms with immunomodulatory activity. Nutrients 2020, 12, 391. [Google Scholar] [CrossRef] [PubMed]
- Indira, M.; Venkateswarulu, T.; Abraham Peele, K.; Nazneen Bobby, M.; Krupanidhi, S.J.B. Bioactive molecules of probiotic bacteria and their mechanism of action: A review. 3 Biotech 2019, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Parapouli, M.; Vasileiadis, A.; Afendra, A.S.; Hatziloukas, E. Saccharomyces cerevisiae and its industrial applications. AIMS Microbiol. 2020, 6, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.; Jewett, M.C. Impact of systems biology on metabolic engineering of Saccharomyces cerevisiae. FEMS Yeast Res. 2008, 8, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Semumu, T.; Gamero, A. Non-Conventional Yeasts as Alternatives in Modern Baking for Improved Performance and Aroma Enhancement. Fermentation 2021, 7, 102. [Google Scholar] [CrossRef]
- Karim, A.; Gerliani, N.; Aider, M. Kluyveromyces marxianus: An emerging yeast cell factory for applications in food and biotechnology. Int. J. Food Microbiol. 2020, 333, 108818. [Google Scholar] [CrossRef]
- Leonel, L.V.; Arruda, P.V.; Chandel, A.K.; Felipe, M.G.A.; Sene, L. Kluyveromyces marxianus: A potential biocatalyst of renewable chemicals and lignocellulosic ethanol production. Crit. Rev. Biotechnol. 2021, 41, 1131–1152. [Google Scholar] [CrossRef]
- Fukuhara, H. Kluyveromyces lactis—A retrospective. FEMS Yeast Res. 2006, 6, 323–324. [Google Scholar] [CrossRef][Green Version]
- Zhang, B.L.; Yang, W.D.; Feng, W.; Omedi, J.O.; Liu, R.S.; Huang, J.X.; Zhang, L.; Zou, Q.B.; Huang, W.N.; Li, S.L. Use of Kluyveromyces marxianus prefermented wheat bran as a source of enzyme mixture to improve dough performance and bread biochemical properties. Cereal Chem. 2019, 96, 142–153. [Google Scholar] [CrossRef]
- Marcisauskas, S.; Ji, B.Y.; Nielsen, J. Reconstruction and analysis of a Kluyveromyces marxianus genome-scale metabolic model. BMC Bioinform. 2019, 20, 551. [Google Scholar] [CrossRef]
- Fonseca, G.G.; Heinzle, E.; Wittmann, C.; Gombert, A.K. The yeast Kluyveromyces marxianus and its biotechnological potential. Appl. Microbiol. Biot. 2008, 79, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Banat, B.M.; Nonklang, S.; Hoshida, H.; Akada, R. Random and targeted gene integrations through the control of non-homologous end joining in the yeast Kluyveromyces marxianus. Yeast 2010, 27, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.; Lee, D.-H.; Kim, S.H.; Kim, H.-J.; Lee, K.; Song, J.Y.; Kim, B.K.; Sung, B.H.; Park, J.C.; Sohn, J.H. Genome sequence of the thermotolerant yeast Kluyveromyces marxianus var. marxianus KCTC 17555. 2012, 11, 1584–1585. [Google Scholar] [CrossRef]
- Hong, J.; Wang, Y.; Kumagai, H.; Tamaki, H. Construction of thermotolerant yeast expressing thermostable cellulase genes. J. Biotechnol. 2007, 130, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Kerry, R.G.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.S.; Das, G. Benefaction of probiotics for human health: A review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef]
- Pace, F.; Pace, M.; Quartarone, G. Probiotics in digestive diseases: Focus on Lactobacillus GG. Minerva Gastroenterol. Dietol. 2015, 61, 273–292. [Google Scholar]
- Rabah, H.; Rosa do Carmo, F.L.; Jan, G. Dairy Propionibacteria: Versatile Probiotics. Microorganisms 2017, 5, 24. [Google Scholar] [CrossRef]
- Rad, A.H.; Mehrabany, E.V.; Alipoor, B.; Mehrabany, L.V.; Javadi, M. Do probiotics act more efficiently in foods than in supplements? Nutrition 2012, 28, 733–736. [Google Scholar] [CrossRef]
- Jin, Y.; Yu, S.; Liu, J.J.; Yun, E.J.; Lee, J.W.; Jin, Y.S.; Kim, K.H. Production of neoagarooligosaccharides by probiotic yeast Saccharomyces cerevisiae var. boulardii engineered as a microbial cell factory. Microb. Cell Fact. 2021, 20, 160. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Vergara, L.; Pereyra, C.M.; Montenegro, M.; Pena, G.A.; Aminahuel, C.A.; Cavaglieri, L.R. Encapsulated whey-native yeast Kluyveromyces marxianus as a feed additive for animal production. Food Addit. Contam. Part A 2017, 34, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Cruchet, S.; Furnes, R.; Maruy, A.; Hebel, E.; Palacios, J.; Medina, F.; Ramirez, N.; Orsi, M.; Rondon, L.; Sdepanian, V.; et al. The Use of Probiotics in Pediatric Gastroenterology: A Review of the Literature and Recommendations by Latin-American Experts. Pediatr. Drugs 2015, 17, 199–216. [Google Scholar] [CrossRef] [PubMed]
- Domig, K.J.; Kiss, H.; Petricevic, L.; Viernstein, H.; Unger, F.; Kneifel, W. Strategies for the evaluation and selection of potential vaginal probiotics from human sources: An exemplary study. Benef. Microbes 2014, 5, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Azais-Braesco, V.; Bresson, J.L.; Guarner, F.; Corthier, G. Not all lactic acid bacteria are probiotics, … but some are. Brit. J. Nutr. 2010, 103, 1079–1081. [Google Scholar] [CrossRef] [PubMed]
- McFarland, L.V. Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J. Gastroenterol. 2010, 16, 2202–2222. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Sánchez, B.; Ruiz, L.; Gueimonde, M.; Margolles, A. Omics for the study of probiotic microorganisms. Food Res. Int. 2013, 54, 1061–1071. [Google Scholar] [CrossRef]
- McFarland, L.V.; Evans, C.T.; Goldstein, E.J.C. Strain-Specificity and Disease-Specificity of Probiotic Efficacy: A Systematic Review and Meta-Analysis. Front. Med. 2018, 5, 124. [Google Scholar] [CrossRef]
- Mikheenko, A.; Prjibelski, A.; Saveliev, V.; Antipov, D.; Gurevich, A. Versatile genome assembly evaluation with QUAST-LG. Bioinformatics 2018, 34, i142–i150. [Google Scholar] [CrossRef]
- Steinbiss, S.; Silva-Franco, F.; Brunk, B.; Foth, B.; Hertz-Fowler, C.; Berriman, M.; Otto, T.D. Companion: A web server for annotation and analysis of parasite genomes. Nucleic Acids Res. 2016, 44, W29–W34. [Google Scholar] [CrossRef]
- Stamatakis, A.; Hoover, P.; Rougemont, J. A rapid bootstrap algorithm for the RAxML Web servers. Syst. Biol. 2008, 57, 758–771. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.D.; Assaf, R.; Brettin, T.; Conrad, N.; Cucinell, C.; Davis, J.J.; Dempsey, D.M.; Dickerman, A.; Dietrich, E.M.; Kenyon, R.W.; et al. Introducing the Bacterial and Viral Bioinformatics Resource Center (BV-BRC): A resource combining PATRIC, IRD and ViPR. Nucleic Acids Res. 2023, 51, D678–D689. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Richter, M.; Rossello-Mora, R.; Oliver Glockner, F.; Peplies, J. JSpeciesWS: A web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics 2016, 32, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, N.F.; Petty, N.K.; Ben Zakour, N.L.; Beatson, S.A. BLAST Ring Image Generator (BRIG): Simple prokaryote genome comparisons. BMC Genom. 2011, 12, 402. [Google Scholar] [CrossRef]
- Garrigues, C.; Johansen, E.; Crittenden, R. Pangenomics—An avenue to improved industrial starter cultures and probiotics. Curr. Opin. Biotechnol. 2013, 24, 187–191. [Google Scholar] [CrossRef]
- Valdez-Baez, J.; da Costa, F.M.R.; Pinto Gomide, A.C.; Profeta, R.; da Silva, A.L.; Sousa, T.d.J.; Viana, M.V.C.; Bentes Kato, R.; Americo, M.F.; dos Santos Freitas, A.J.B. Comparative Genomics and In Silico Evaluation of Genes Related to the Probiotic Potential of Bifidobacterium breve 1101A. Bacteria 2022, 1, 161–182. [Google Scholar] [CrossRef]
- Walter, J.; Ley, R. The human gut microbiome: Ecology and recent evolutionary changes. Annu. Rev. Microbiol. 2011, 65, 411–429. [Google Scholar] [CrossRef]
- Hildebrand, F.; Meyer, A.; Eyre-Walker, A. Evidence of selection upon genomic GC-content in bacteria. PLoS Genet. 2010, 6, e1001107. [Google Scholar] [CrossRef]
- Yang, Z.; Rannala, B. Molecular phylogenetics: Principles and practice. Nat. Rev. Genet. 2012, 13, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Jain, C.; Rodriguez-R, L.M.; Phillippy, A.M.; Konstantinidis, K.T.; Aluru, S. High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat. Commun. 2018, 9, 5114. [Google Scholar] [CrossRef] [PubMed]
- Khatri, I.; Tomar, R.; Ganesan, K.; Prasad, G.S.; Subramanian, S. Complete genome sequence and comparative genomics of the probiotic yeast Saccharomyces boulardii. Sci. Rep. 2017, 7, 371. [Google Scholar] [CrossRef] [PubMed]
- Morrissey, J.P.; Etschmann, M.M.; Schrader, J.; de Billerbeck, G.M. Cell factory applications of the yeast Kluyveromyces marxianus for the biotechnological production of natural flavour and fragrance molecules. Yeast 2015, 32, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Cadot, Y.; Caille, S.; Samson, A.; Barbeau, G.; Cheynier, V. Sensory representation of typicality of Cabernet franc wines related to phenolic composition: Impact of ripening stage and maceration time. Anal. Chim. Acta 2012, 732, 91–99. [Google Scholar] [CrossRef]
- Ng, C.Y.; Jung, M.Y.; Lee, J.; Oh, M.K. Production of 2,3-butanediol in Saccharomyces cerevisiae by in silico aided metabolic engineering. Microb. Cell Fact. 2012, 11, 68. [Google Scholar] [CrossRef]
- Farhana, A.; Lappin, S.L. Biochemistry, Lactate Dehydrogenase; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Morris, A.L.; Mohiuddin, S.S. Biochemistry, Nutrients; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Shi, L.; Tu, B.P. Acetyl-CoA and the regulation of metabolism: Mechanisms and consequences. Curr. Opin. Cell Biol. 2015, 33, 125–131. [Google Scholar] [CrossRef]
- Ziarno, M.; Zareba, D. The effect of the addition of microbial transglutaminase before the fermentation process on the quality characteristics of three types of yogurt. Food Sci. Biotechnol. 2020, 29, 109–119. [Google Scholar] [CrossRef]
- Motey, G.A.; Johansen, P.G.; Owusu-Kwarteng, J.; Ofori, L.A.; Obiri-Danso, K.; Siegumfeldt, H.; Larsen, N.; Jespersen, L. Probiotic potential of Saccharomyces cerevisiae and Kluyveromyces marxianus isolated from West African spontaneously fermented cereal and milk products. Yeast 2020, 37, 403–412. [Google Scholar] [CrossRef]
- Muhlhofer, M.; Berchtold, E.; Stratil, C.G.; Csaba, G.; Kunold, E.; Bach, N.C.; Sieber, S.A.; Haslbeck, M.; Zimmer, R.; Buchner, J. The Heat Shock Response in Yeast Maintains Protein Homeostasis by Chaperoning and Replenishing Proteins. Cell Rep. 2019, 29, 4593–4607.e4598. [Google Scholar] [CrossRef]
- Castells-Roca, L.; Garcia-Martinez, J.; Moreno, J.; Herrero, E.; Belli, G.; Perez-Ortin, J.E. Heat shock response in yeast involves changes in both transcription rates and mRNA stabilities. PLoS ONE 2011, 6, e17272. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.J.; Kim, S.; Hahn, J.S. Efficient production of acetoin in Saccharomyces cerevisiae by disruption of 2,3-butanediol dehydrogenase and expression of NADH oxidase. Sci Rep. 2016, 6, 27667. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Forman, H.J.; Choi, J. Gamma-glutamyl transpeptidase in glutathione biosynthesis. Methods Enzymol. 2005, 401, 468–483. [Google Scholar] [CrossRef] [PubMed]
- Martensson, J.; Steinherz, R.; Jain, A.; Meister, A. Glutathione ester prevents buthionine sulfoximine-induced cataracts and lens epithelial cell damage. Proc. Natl. Acad. Sci. USA 1989, 86, 8727–8731. [Google Scholar] [CrossRef] [PubMed]
- Mary, V.S.; Theumer, M.G.; Arias, S.L.; Rubinstein, H.R. Reactive oxygen species sources and biomolecular oxidative damage induced by aflatoxin B1 and fumonisin B1 in rat spleen mononuclear cells. Toxicology 2012, 302, 299–307. [Google Scholar] [CrossRef]
- Wu, G.; Bazer, F.W.; Dai, Z.; Li, D.; Wang, J.; Wu, Z. Amino acid nutrition in animals: Protein synthesis and beyond. Annu. Rev. Anim. Biosci. 2014, 2, 387–417. [Google Scholar] [CrossRef]
- Zyracka, E.; Zadrag, R.; Koziol, S.; Krzepilko, A.; Bartosz, G.; Bilinski, T. Ascorbate abolishes auxotrophy caused by the lack of superoxide dismutase in Saccharomyces cerevisiae. Yeast can be a biosensor for antioxidants. J. Biotechnol. 2005, 115, 271–278. [Google Scholar] [CrossRef]
- Paramasivan, K.; Mutturi, S. Progress in terpene synthesis strategies through engineering of Saccharomyces cerevisiae. Crit. Rev. Biotechnol. 2017, 37, 974–989. [Google Scholar] [CrossRef]
- Jeong, A.; Suazo, K.F.; Wood, W.G.; Distefano, M.D.; Li, L. Isoprenoids and protein prenylation: Implications in the pathogenesis and therapeutic intervention of Alzheimer’s disease. Crit. Rev. Biochem. Mol. Biol. 2018, 53, 279–310. [Google Scholar] [CrossRef]
- Cao, X.; Yang, S.; Cao, C.; Zhou, Y.J. Harnessing sub-organelle metabolism for biosynthesis of isoprenoids in yeast. Synth. Syst. Biotechnol. 2020, 5, 179–186. [Google Scholar] [CrossRef] [PubMed]

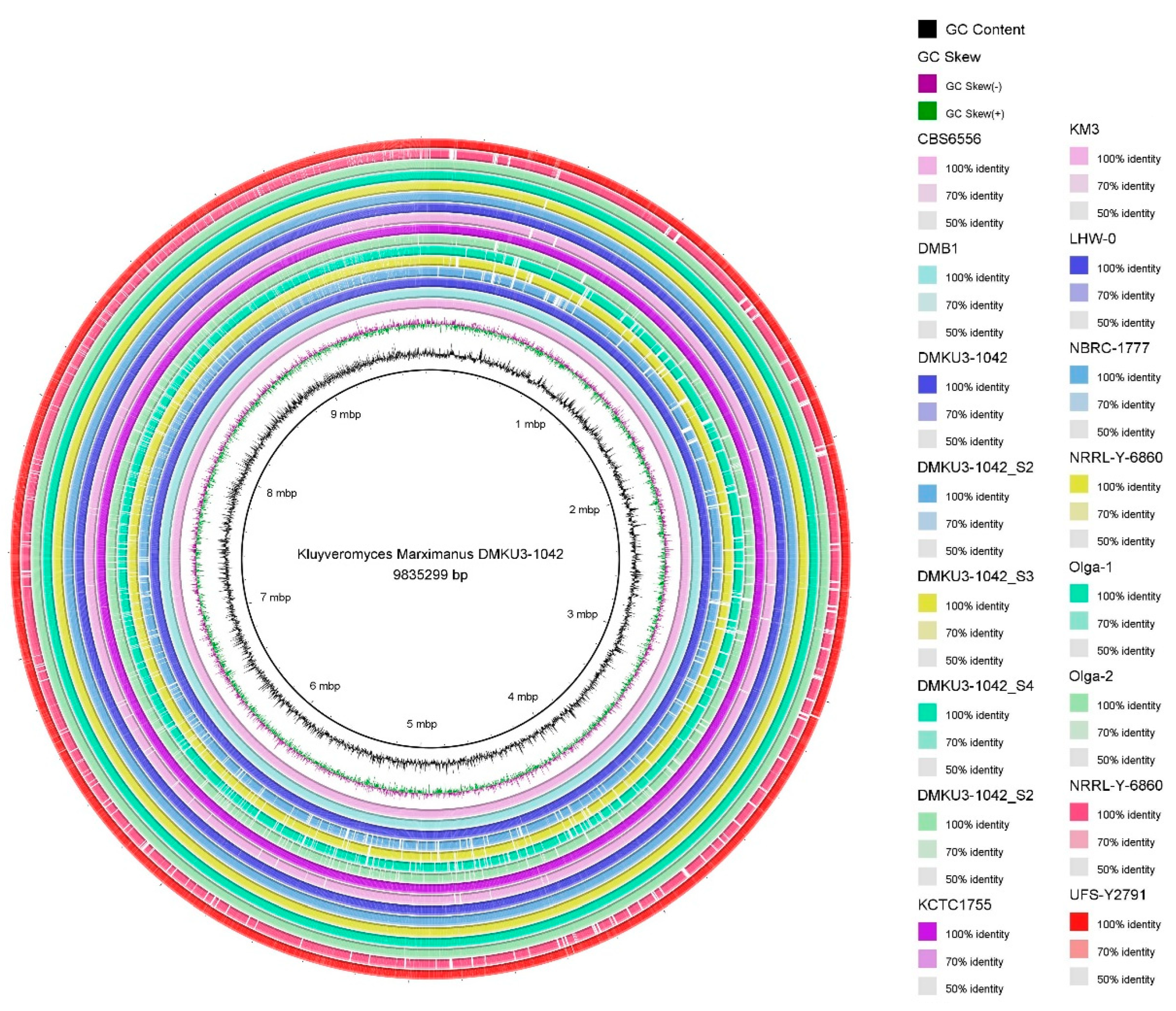
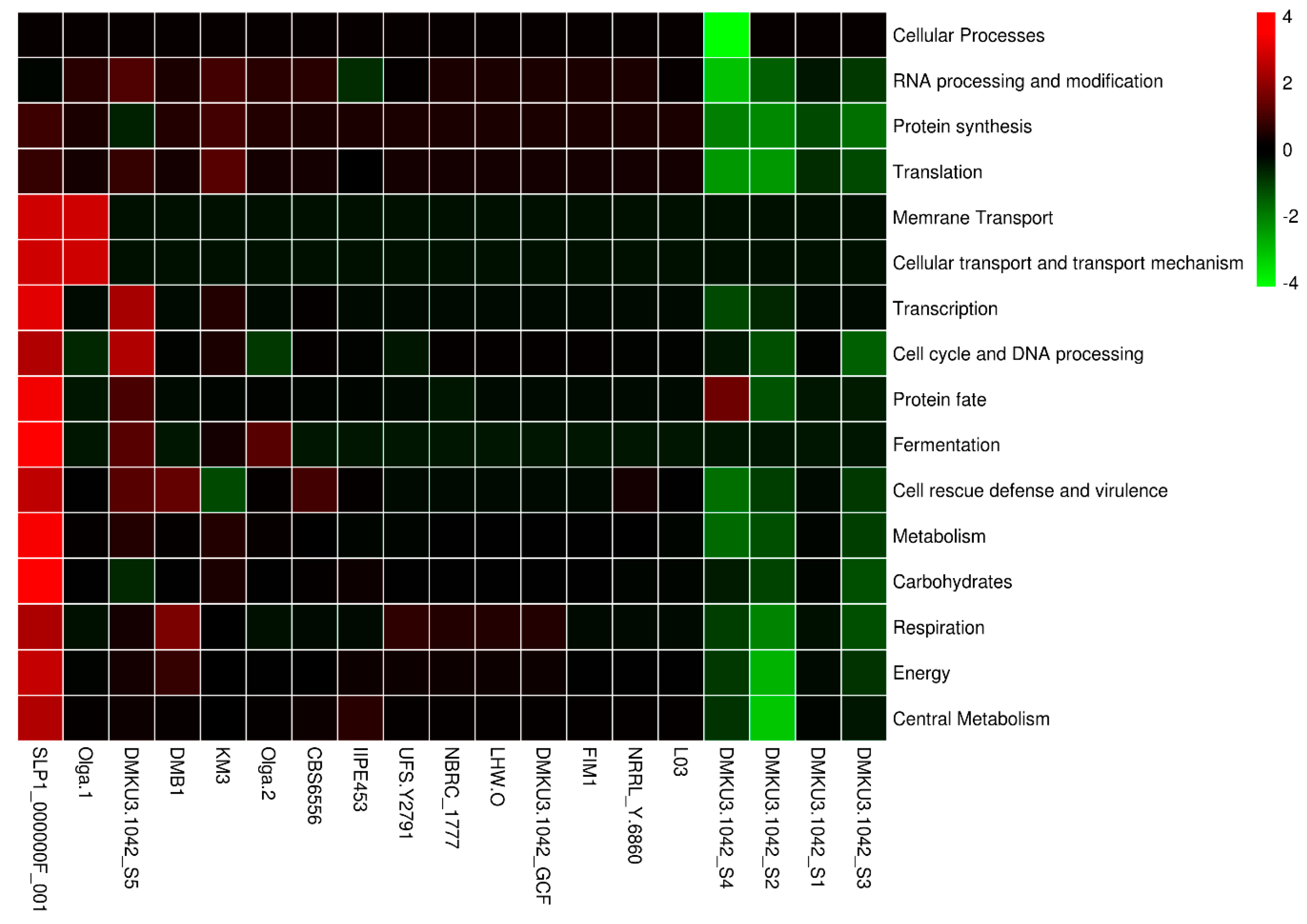
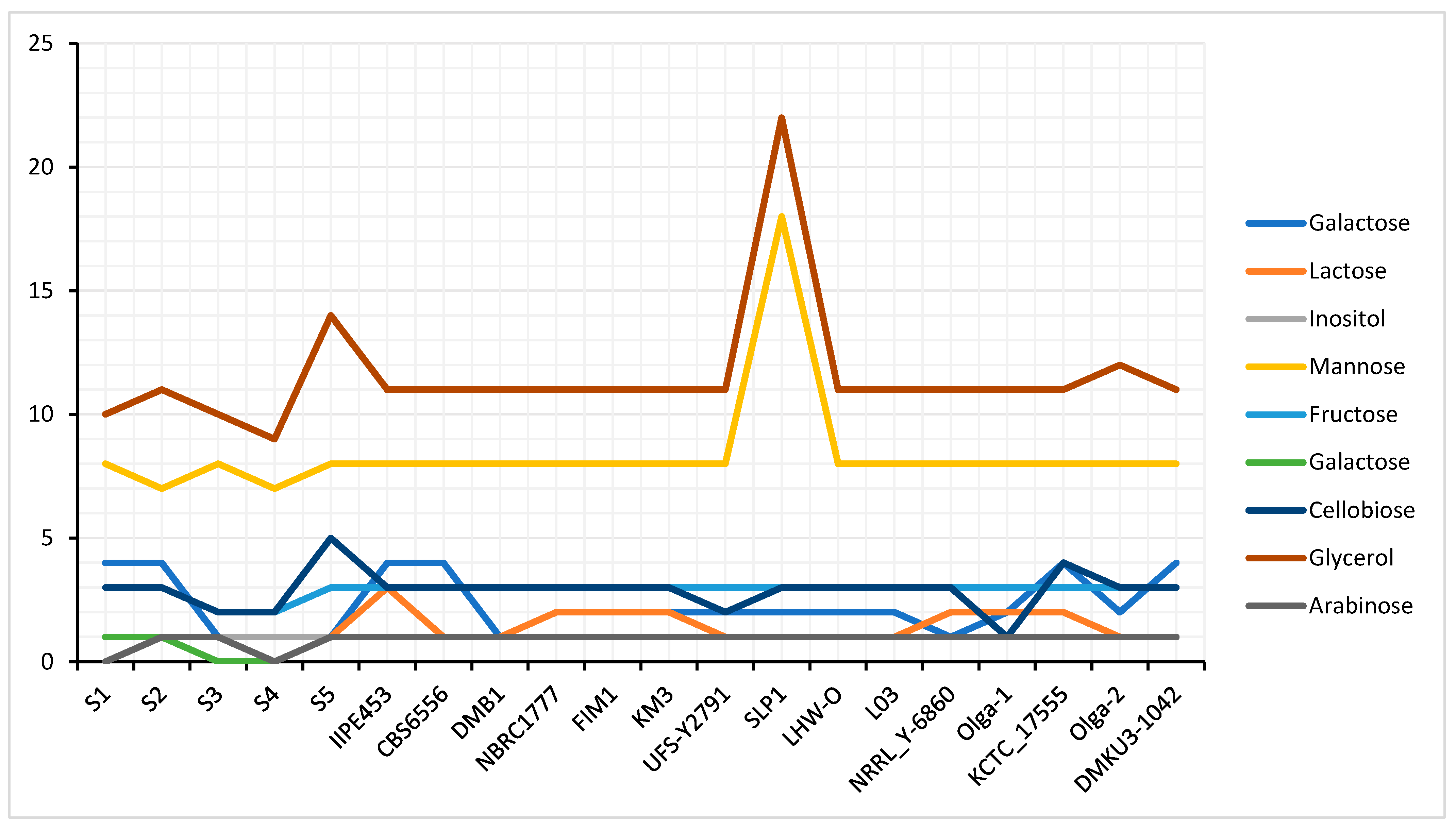
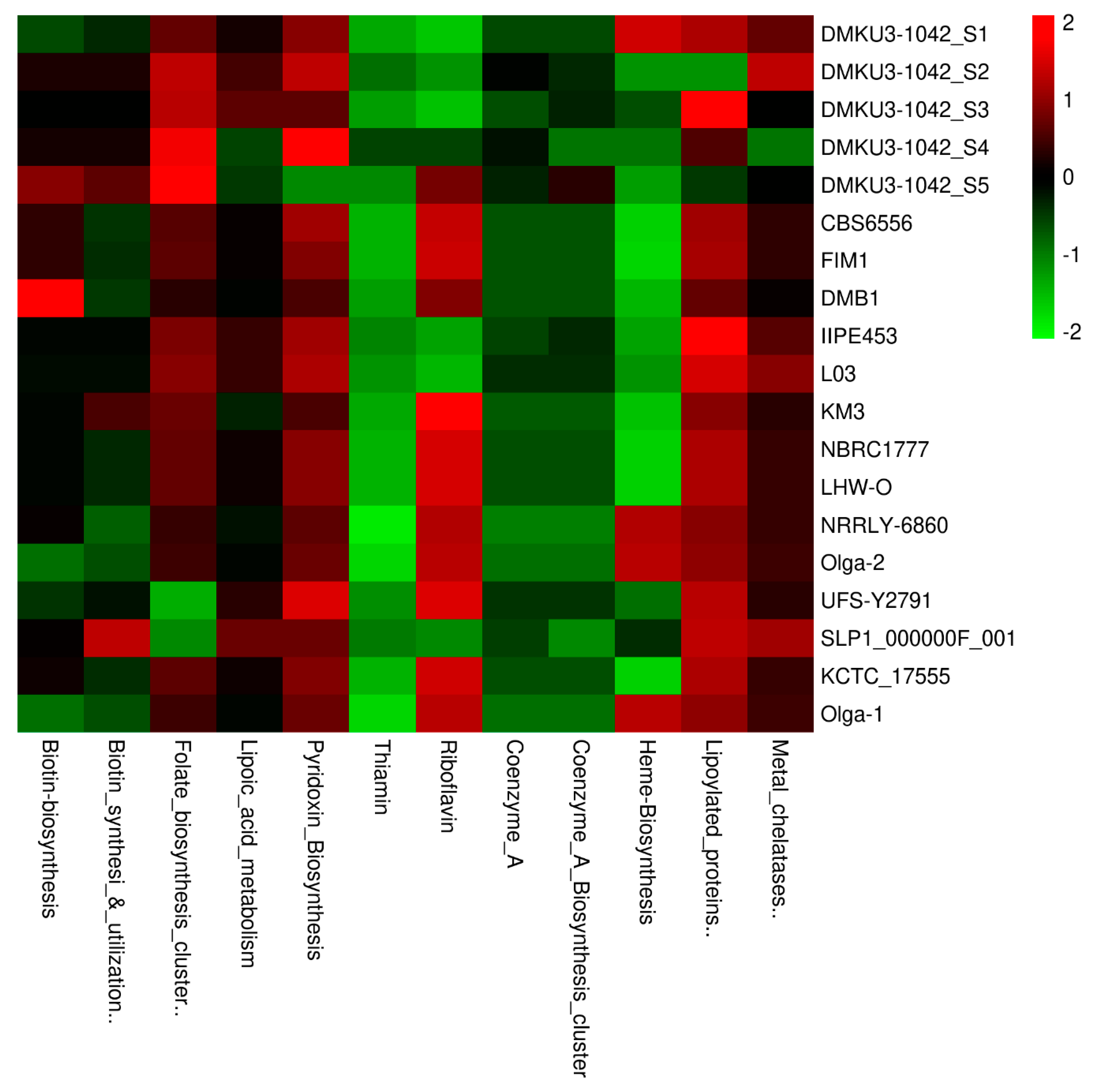
| Species | Genome Size (bp) | Average G + C Content (%) | Total CDS | Total tRNA Genes | Average Gene Density (%) | Average G + C in CDS (%) |
|---|---|---|---|---|---|---|
| DMKU3-1042_S1 | 9,835,299 | 40.59 | 9875 | 124 | 71.25 | 41.69 |
| DMKU3-1042_S2 | 9,889,048 | 40.18 | 9870 | 121 | 71.60 | 41.10 |
| DMKU3-1042_S3 | 9,806,639 | 40.20 | 9870 | 123 | 71.20 | 41.60 |
| DMKU3-1042_S4 | 9,850,219 | 40.10 | 9875 | 128 | 71.10 | 41.25 |
| DMKU3-1042_S5 | 9,843,168 | 40.2 | 9870 | 134 | 71.3 | 41.9 |
| NRRL Y-6860 | 10,837,618 | 40.2 | 8730 | 170 | - | - |
| Olga-1 | 10,595,223 | 40.13 | 10,209 | 94 | - | - |
| Olga-2 | 10,651,240 | 40.19 | 10,489 | 121 | - | - |
| SLP1000000F_001 | 9,555,365 | 39.91 | 20,917 | 157 | - | - |
| UFS-Y2791 | 10,695,463 | 40.4 | 10,446 | 26 | - | - |
| KM3 | 10,592,351 | 40.21 | 9791 | 167 | - | - |
| L03 | 10,366,177 | 40.13 | 9368 | 11 | 88.98 | 46.74 |
| LHW-O | 10,776,015 | 40.15 | 10,225 | 180 | 87.6 | 46.81 |
| NBRC 1777 | 10,895,581 | 40.12 | 8847 | 182 | - | - |
| DMKU3-1042 | 10,966,467 | 40.12 | 8821 | 177 | 87.36 | 46.79 |
| KCTC1755 | 10,920,632 | 40.14 | 8890 | 178 | 87.31 | 40.61 |
| CBS6556 | 10,894,425 | 40.25 | 8773 | 169 | 87.2 | 46.64 |
| FIM1 | 10,914,453 | 40.16 | 9167 | 172 | 87.96 | 46.78 |
| IIPE453 | 10,711,999 | 40.17 | 9228 | 145 | 93.23 | 46.68 |
| DMB1 | 11,165,408 | 40.09 | 9384 | 197 | 86.79 | 46.75 |
| DMKU3-1042 S1 | |||||||||||||||||||
| 99.66 | DMKU3-1042 S2 | ||||||||||||||||||
| 99.72 | 99.77 | DMKU3-1042 S3 | |||||||||||||||||
| 99.67 | 99.74 | 99.71 | DMKU3-1042 S4 | ||||||||||||||||
| 99.37 | 99.39 | 99.36 | 99.38 | DMKU3-1042 S5 | |||||||||||||||
| 97.16 | 97.13 | 97.05 | 97.12 | 97.46 | CBS6556 | ||||||||||||||
| 97.19 | 97.16 | 97.09 | 97.16 | 97.54 | 98.88 | DMB1 | |||||||||||||
| 97.21 | 97.18 | 97.12 | 97.16 | 97.53 | 98.99 | 99.08 | FIM1 | ||||||||||||
| 97.19 | 97.13 | 97.07 | 97.14 | 97.52 | 98.96 | 99.24 | 99.18 | IIPE453 | |||||||||||
| 97.17 | 97.14 | 97.07 | 97.13 | 97.48 | 99.86 | 98.87 | 98.98 | 99.04 | KCTC1755 | ||||||||||
| 98.48 | 98.50 | 98.45 | 98.47 | 98.28 | 97.71 | 97.71 | 97.79 | 97.82 | 97.75 | KM3 | |||||||||
| 97.23 | 97.22 | 97.13 | 97.20 | 97.54 | 97.19 | 99.04 | 99.13 | 99.12 | 99.23 | 97.88 | LHW-O | ||||||||
| 99.55 | 99.56 | 99.54 | 99.57 | 98.89 | 97.00 | 96.97 | 97.04 | 97.06 | 96.98 | 98.44 | 97.07 | LO3 | |||||||
| 97.81 | 97.16 | 97.07 | 97.15 | 97.52 | 99.03 | 99.10 | 99.07 | 99.23 | 99.09 | 97.81 | 99.26 | 97.15 | NBRC1777 | ||||||
| 97.57 | 97.56 | 97.50 | 97.57 | 97.59 | 98.15 | 98.21 | 98.22 | 98.32 | 98.19 | 97.66 | 98.36 | 97.57 | 98.19 | NRRLY-6880 | |||||
| 99.34 | 99.31 | 99.32 | 99.32 | 98.72 | 97.02 | 97.02 | 97.09 | 97.13 | 97.04 | 98.26 | 97.15 | 99.42 | 97.03 | 97.46 | Olga1 | ||||
| 99.32 | 99.31 | 99.29 | 99.31 | 98.70 | 97.03 | 97.00 | 97.07 | 97.12 | 97.04 | 98.26 | 97.13 | 99.41 | 97.03 | 97.45 | 99.82 | Olga2 | |||
| 97.16 | 97.15 | 97.07 | 97.14 | 97.52 | 98.97 | 99.25 | 99.13 | 99.37 | 99.03 | 97.79 | 99.15 | 97.15 | 99.12 | 98.29 | 97.10 | 97.10 | DMKU3-1042_GCF | ||
| 95.25 | 95.15 | 95.08 | 95.17 | 95.27 | 95.87 | 95.90 | 95.94 | 95.92 | 95.93 | 95.27 | 96.03 | 95.19 | 95.91 | 95.44 | 95.16 | 95.15 | 95.85 | SLP1 | |
| 94.63 | 94.60 | 94.49 | 94.57 | 94.33 | 94.50 | 94.40 | 94.48 | 94.62 | 94.51 | 94.46 | 94.60 | 94.57 | 94.47 | 94.48 | 94.55 | 94.56 | 94.46 | 95.85 | UFS-Y2791 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ullah, M.; Rizwan, M.; Raza, A.; Xia, Y.; Han, J.; Ma, Y.; Chen, H. Snapshot of the Probiotic Potential of Kluveromyces marxianus DMKU-1042 Using a Comparative Probiogenomics Approach. Foods 2023, 12, 4329. https://doi.org/10.3390/foods12234329
Ullah M, Rizwan M, Raza A, Xia Y, Han J, Ma Y, Chen H. Snapshot of the Probiotic Potential of Kluveromyces marxianus DMKU-1042 Using a Comparative Probiogenomics Approach. Foods. 2023; 12(23):4329. https://doi.org/10.3390/foods12234329
Chicago/Turabian StyleUllah, Mati, Muhammad Rizwan, Ali Raza, Yutong Xia, Jianda Han, Yi Ma, and Huayou Chen. 2023. "Snapshot of the Probiotic Potential of Kluveromyces marxianus DMKU-1042 Using a Comparative Probiogenomics Approach" Foods 12, no. 23: 4329. https://doi.org/10.3390/foods12234329
APA StyleUllah, M., Rizwan, M., Raza, A., Xia, Y., Han, J., Ma, Y., & Chen, H. (2023). Snapshot of the Probiotic Potential of Kluveromyces marxianus DMKU-1042 Using a Comparative Probiogenomics Approach. Foods, 12(23), 4329. https://doi.org/10.3390/foods12234329








