Potential Correlation between Microbial Diversity and Volatile Flavor Substances in a Novel Chinese-Style Sausage during Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of the Novel Chinese-Style Sausage
2.2. GC-MS Analysis
2.3. DNA Extraction and PCR Amplification and Sequencing
2.4. Data Processing
3. Results and Discussion
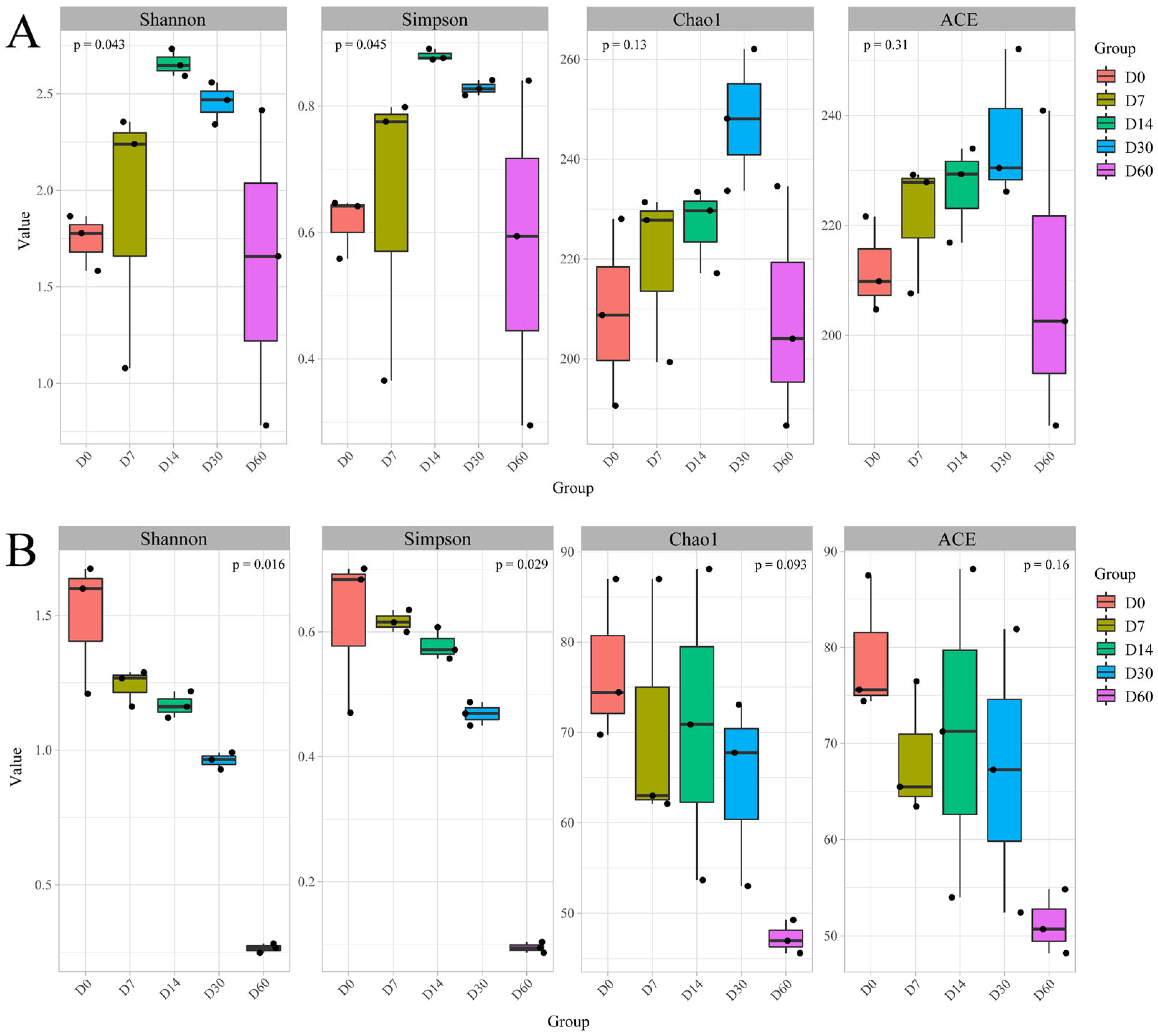
3.1. α-Diversity Analysis
3.2. Analysis of Microbial Community Structure
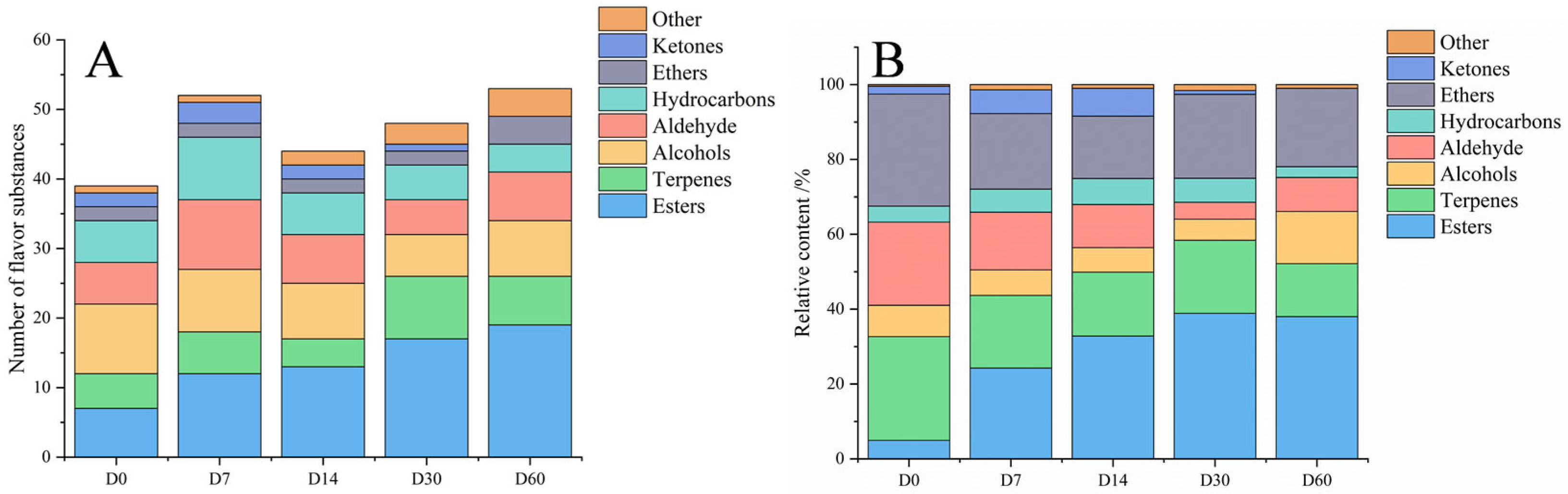
3.3. Volatile Flavor Substances in the Fermented Sausages
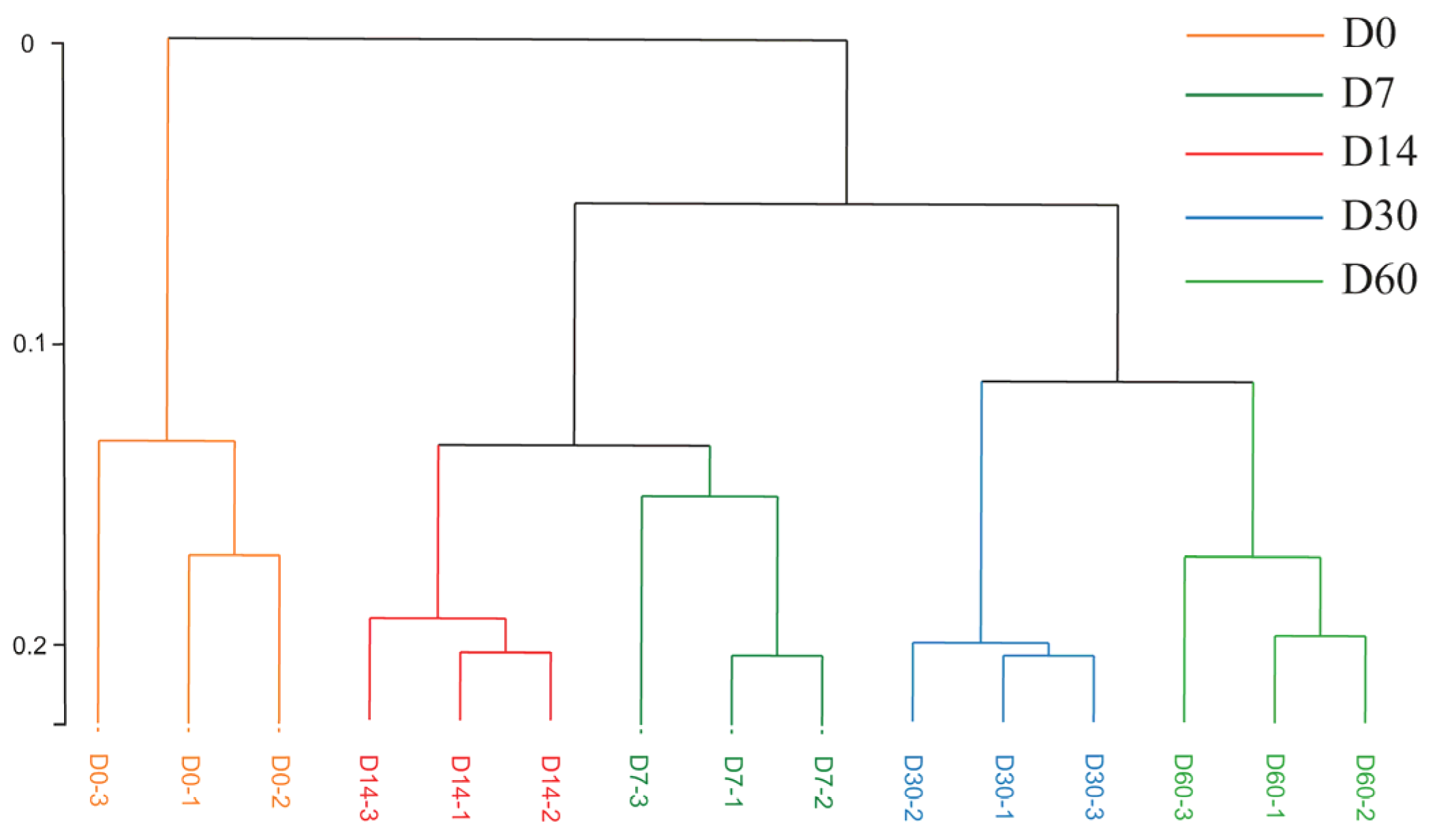
3.4. Hierarchical Clustering Analysis for Volatile Flavor Substances
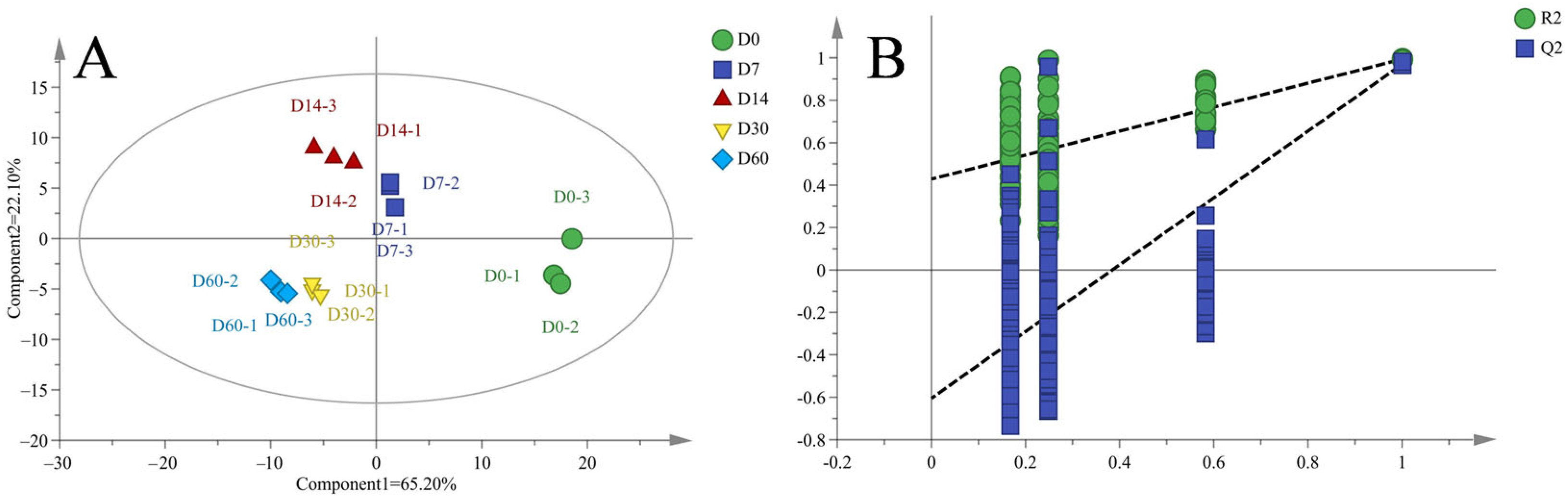
3.5. PCA Analysis for Volatile Flavor Substances
3.6. PLS-DA Analysis
3.7. Key Volatile Flavor Substances in the Sausages Stored for Different Periods
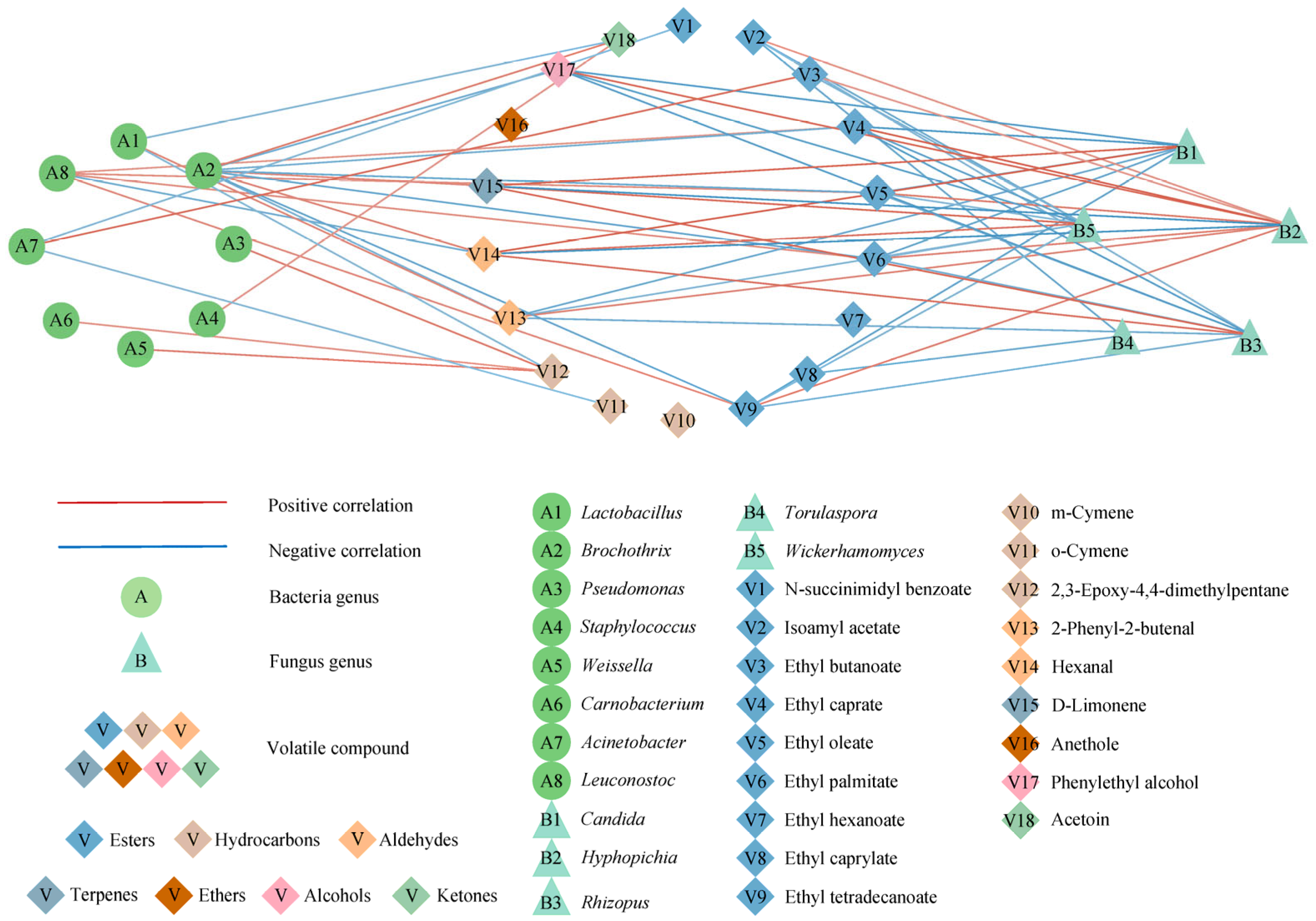
3.8. Potential Correlation between Microorganisms and Differential Volatile Flavor Substances
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Flores, M.; Corral, S.; Cano-García, L.; Salvador, A.; Belloch, C. Yeast strains as potential aroma enhancers in dry fermented sausages. Int. J. Food Microbiol. 2015, 212, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Benito, M.J.; Serradilla, M.J.; Ruiz-Moyano, S.; Martín, A.; Pérez-Nevado, F.; Córdoba, M.G. Rapid differentiation of lactic acid bacteria from autochthonous fermentation of Iberian dry-fermented sausages. Meat Sci. 2008, 80, 656–661. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Choi, Y.J.; Lee, M.J.; Park, S.J.; Oh, S.J.; Yun, Y.R.; Min, S.G.; Seo, H.Y.; Park, S.H.; Lee, M.A. Effects of combining two lactic acid bacteria as a starter culture on model kimchi fermentation. Food Res. Int. 2020, 136, 109591. [Google Scholar] [CrossRef]
- Liu, Y.; Wan, Z.; Yang, Z.; Yu, Q.; Li, L.; Wang, J. Recent Progress in Understanding the Contribution of Lactic Acid Bacteria to Flavor Formation in Salami: A Review. Food Sci. 2020, 41, 273–282. [Google Scholar] [CrossRef]
- Demeyer, D.; Raemaekers, M.; Rizzo, A.; Holck, A.; De Smedt, A.; ten Brink, B.; Hagen, B.; Montel, C.; Zanardi, E.; Murbrekk, E.; et al. Control of bioflavour and safety in fermented sausages: First results of a European project. Food Res. Int. 2000, 33, 171–180. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, Q.; Wen, R.; Wang, Y.; Qin, L.; Kong, B. Quality characteristics and flavor profile of Harbin dry sausages inoculated with lactic acid bacteria and Staphylococcus xylosus. LWT 2019, 114, 108392. [Google Scholar] [CrossRef]
- Casaburi, A.; Di Monaco, R.; Cavella, S.; Toldrá, F.; Ercolini, D.; Villani, F. Proteolytic and lipolytic starter cultures and their effect on traditional fermented sausages ripening and sensory traits. Food Microbiol. 2008, 25, 335–347. [Google Scholar] [CrossRef]
- Thonning Olesen, P.; Stahnke, L.H. The influence of environmental parameters on the catabolism of branched-chain amino acids by Staphylococcus xylosus and Staphylococcus carnosus. Food Microbiol. 2004, 21, 43–50. [Google Scholar] [CrossRef]
- Talon, R.; Walter, D.; Chartier, S.; Barrière, C.; Montel, M.C. Effect of nitrate and incubation conditions on the production of catalase and nitrate reductase by staphylococci. Int. J. Food Microbiol. 1999, 52, 47–56. [Google Scholar] [CrossRef]
- Patrignani, F.; Vannini, L.; Gardini, F.; Guerzoni, M.E.; Lanciotti, R. Variability of the lipolytic activity and volatile molecules production by a strain of Yarrowia lipolytica in pork fat and its dependence on environmental conditions. Meat Sci. 2011, 89, 21–26. [Google Scholar] [CrossRef]
- Durá, M.A.; Flores, M.; Toldrá, F. Effect of Debaryomyces spp. on the proteolysis of dry-fermented sausages. Meat Sci. 2004, 68, 319–328. [Google Scholar] [CrossRef]
- Chen, Y.H.; Liu, X.W.; Huang, J.L.; Baloch, S.; Xu, X.; Pei, X.F. Microbial diversity and chemical analysis of Shuidouchi, traditional Chinese fermented soybean. Food Res. Int. 2019, 116, 1289–1297. [Google Scholar] [CrossRef]
- Lu, Y.; Tan, X.; Lv, Y.; Yang, G.; Chi, Y.; He, Q. Physicochemical properties and microbial community dynamics during Chinese horse bean-chili-paste fermentation, revealed by culture-dependent and culture-independent approaches. Food Microbiol. 2020, 85, 103309. [Google Scholar] [CrossRef]
- Li, W.; Mei, S.; Zhou, H.; Salman Farid, M.; Hu, T.; Wu, T. Metabolite fingerprinting of the ripening process in Pixian douban using a feature-based molecular network and metabolomics analysis. Food Chem. 2023, 418, 135940. [Google Scholar] [CrossRef]
- Hussin, M.; Anzian, A.; Liew, C.X.; Muhialdin, B.J.; Mohsin, A.Z.; Fang, C.-M.; Saad, M.Z.; Ahmad, N.H.; Hassan, M.; Adnan, H.; et al. Potentially Probiotic Fermented Glutinous Rice (Oryza sativa L.) with Lactiplantibacillus plantarum Improved Immune System Response in a Small Sample of BALB/cByJ Mice. Fermentation 2022, 8, 612. [Google Scholar] [CrossRef]
- Xie, C.; Zeng, H.; Li, J.; Qin, L. Comprehensive explorations of nutritional, functional and potential tasty components of various types of Sufu, a Chinese fermented soybean appetizer. Food Sci. Technol. 2019, 39, 105–114. [Google Scholar] [CrossRef]
- Heo, S.; Park, J.; Lee, K.-G.; Lee, J.-H.; Jeong, D.-W. Quality characteristics of soybean fermented by Mucor, Rhizopus, and Aspergillus from meju. Heliyon 2023, 9, e14092. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhou, S.; Julian McClements, D.; Huang, L.; Meng, L.; Xia, X.; Dong, M. Multistarter fermentation of glutinous rice with Fu brick tea: Effects on microbial, chemical, and volatile compositions. Food Chem. 2020, 309, 125790. [Google Scholar] [CrossRef] [PubMed]
- Kamilari, E.; Efthymiou, M.; Anagnostopoulos, D.A.; Tsaltas, D. Cyprus Sausages’ Bacterial Community Identification through Metataxonomic Sequencing: Evaluation of the Impact of Different DNA Extraction Protocols on the Sausages’ Microbial Diversity Representation. Front. Microbiol. 2021, 12, 662957. [Google Scholar] [CrossRef] [PubMed]
- Unno, R.; Suzuki, T.; Osaki, Y.; Matsutani, M.; Ishikawa, M. Causality Verification for the Correlation between the Presence of Nonstarter Bacteria and Flavor Characteristics in Soft-Type Ripened Cheeses. Microbiol. Spectr. 2022, 10, e0289422. [Google Scholar] [CrossRef]
- Hu, X.; Du, H.; Ren, C.; Xu, Y. Illuminating Anaerobic Microbial Community and Cooccurrence Patterns across a Quality Gradient in Chinese Liquor Fermentation Pit Muds. Appl. Environ. Microbiol. 2016, 82, 2506–2515. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Xiang, X.; Tan, H.; Zhang, Q.; Shan, Y.; Yang, H. Potential correlation between volatiles and microbiome of Xiang xi sausages from four different regions. Food Res. Int. 2021, 139, 109943. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Li, X.; Xiao, R.; Dudu, O.E.; Yang, L.; Ma, Y. Impact of Lactobacillus paracasei IMC502 in coculture with traditional starters on volatile and non-volatile metabolite profiles in yogurt. Process Biochem. 2020, 99, 61–69. [Google Scholar] [CrossRef]
- Flores, M.; Durá, M.A.; Marco, A.; Toldrá, F. Effect of Debaryomyces spp. on aroma formation and sensory quality of dry-fermented sausages. Meat Sci. 2004, 68, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Yadav, J.S.; Bezawada, J.; Yan, S.; Tyagi, R.D.; Surampalli, R.Y. Candida krusei: Biotechnological potentials and concerns about its safety. Can. J. Microbiol. 2012, 58, 937–952. [Google Scholar] [CrossRef] [PubMed]
- Solomando, J.C.; Antequera, T.; Martín, A.; Perez-Palacios, T. Fish Oil Microcapsules as Omega-3 Enrichment Strategy: Changes in Volatile Compounds of Meat Products during Storage and Cooking. Foods 2021, 10, 745. [Google Scholar] [CrossRef]
- Sun, W.; Zhao, Q.; Zhao, H.; Zhao, M.; Yang, B. Volatile compounds of Cantonese sausage released at different stages of processing and storage. Food Chem. 2010, 121, 319–325. [Google Scholar] [CrossRef]
- Liu, Y.; Wan, Z.; Yohannes, K.W.; Yu, Q.; Yang, Z.; Li, H.; Liu, J.; Wang, J. Functional Characteristics of Lactobacillus and Yeast Single Starter Cultures in the Ripening Process of Dry Fermented Sausage. Front. Microbiol. 2020, 11, 611260. [Google Scholar] [CrossRef]
- Bel-Rhlid, R.; Berger, R.G.; Blank, I. Bio-mediated generation of food flavors—Towards sustainable flavor production inspired by nature. Trends. Food Sci. Technol. 2018, 78, 134–143. [Google Scholar] [CrossRef]
- Sulejmani, E.; Demiri, M. Volatile compounds of Macedonian fermented sausage as affected by ripening process using SPME/GC-MS. Chem. Ind. Chem. Eng. Q. 2019, 26, 71–78. [Google Scholar] [CrossRef]
- Molina, G.; Pimentel, M.R.; Pastore, G.M. Pseudomonas: A promising biocatalyst for the bioconversion of terpenes. Appl. Microbiol. Biotechnol. 2013, 97, 1851–1864. [Google Scholar] [CrossRef] [PubMed]
- Vespermann, K.A.; Paulino, B.N.; Barcelos, M.C.; Pessôa, M.G.; Pastore, G.M.; Molina, G. Biotransformation of α- and β-pinene into flavor compounds. Appl. Microbiol. Biotechnol. 2017, 101, 1805–1817. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, X.; Wang, Y.; Pan, D.; Sun, Y.; Cao, J. Study on the volatile compounds generated from lipid oxidation of Chinese bacon (unsmoked) during processing. Eur. J. Lipid Sci. Technol. 2017, 119, 1600512. [Google Scholar] [CrossRef]
- Stahnke, L.H. Aroma components from dried sausages fermented with Staphylococcus xylosus. Meat Sci. 1994, 38, 39–53. [Google Scholar] [CrossRef]
- Cheah, P.B.; Abu Hasim, N.H. Natural antioxidant extract from galangal (Alpinia galanga) for minced beef. J. Sci. Food Agric. 2000, 80, 1565–1571. [Google Scholar] [CrossRef]
- Al-Dalali, S.; Li, C.; Xu, B. Effect of frozen storage on the lipid oxidation, protein oxidation, and flavor profile of marinated raw beef meat. Food Chem. 2021, 376, 131881. [Google Scholar] [CrossRef]
- Sidira, M.; Kandylis, P.; Kanellaki, M.; Kourkoutas, Y. Effect of immobilized Lactobacillus casei on the evolution of flavor compounds in probiotic dry-fermented sausages during ripening. Meat Sci. 2015, 100, 41–51. [Google Scholar] [CrossRef]
- Bianchi, F.; Cantoni, C.; Careri, M.; Chiesa, L.; Musci, M.; Pinna, A. Characterization of the aromatic profile for the authentication and differentiation of typical Italian dry-sausages. Talanta 2007, 72, 1552–1563. [Google Scholar] [CrossRef]
- Maikhunthod, B.; Marriott, P.J. Aroma-impact compounds in dried spice as a quality index using solid phase microextraction with olfactometry and comprehensive two-dimensional gas chromatography. Food Chem. 2013, 141, 4324–4332. [Google Scholar] [CrossRef]
- Huang, L.; Sun, Z.; Zeng, X.; Pan, D.; He, J.; Shen, J. Effects of Multi-Ingredients for Nitrite on the Volatile Flavor Compounds in Cured Meat. J. Chin. Inst. Food Sci. Technol. 2021, 21, 324–333. [Google Scholar] [CrossRef]
- Ryu, J.; Seo, J.; Lee, Y.; Lim, Y.; Ahn, J.-H.; Hur, H.-G. Identification of syn- and anti-anethole-2,3-epoxides in the metabolism of trans-anethole by the newly isolated bacterium Pseudomonas putida JYR-1. J. Agric. Food Chem. 2005, 53, 5954–5958. [Google Scholar] [CrossRef]
- Worley, B.; Powers, R. Multivariate Analysis in Metabolomics. Curr. Metab. 2013, 1, 92–107. [Google Scholar] [CrossRef]
- Xu, Y.; Bai, X.; Feng, Y.; Zhao, H.; Li, X.; Li, J.; Yi, S.; Xie, J.; Guo, X. Changes of Flavor Compounds in Sea Bass during Steaming Process as Analyzed by Gas Chromatography-Ion Mobility Spectroscopy and Chemometrics. Food Sci. 2021, 42, 270–275. [Google Scholar] [CrossRef]
- Liu, D.; Qi, Y.; Zhao, N.; Cao, Y.; Xu, J.; Fan, M. Multivariate analysis reveals effect of glutathione-enriched inactive dry yeast on amino acids and volatile components of kiwi wine. Food Chem. 2020, 329, 127086. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.Y.; Aheto, J.H.; Huang, X.; Zheng, K.; Dai, C.; Wang, C.; Bai, J.W. An evaluation of biochemical, structural and volatile changes of dry-cured pork using a combined ion mobility spectrometry, hyperspectral and confocal imaging approach. J. Sci. Food Agric. 2021, 101, 5972–5983. [Google Scholar] [CrossRef]
- Rao, Y.; Qian, Y.; Tao, Y.; She, X.; Li, Y.; Chen, X.; Guo, S.; Xiang, W.; Liu, L.; Du, H.; et al. Characterization of the microbial communities and their correlations with chemical profiles in assorted vegetable Sichuan pickles. Food Control 2020, 113, 107174. [Google Scholar] [CrossRef]
- Markkinen, N.; Laaksonen, O.; Yang, B. Impact of malolactic fermentation with Lactobacillus plantarum on volatile compounds of sea buckthorn juice. Eur. Food Res. Technol. 2021, 247, 719–736. [Google Scholar] [CrossRef]
- Li, C.; Zhao, Y.; Wang, Y.; Li, L.; Huang, J.; Yang, X.; Chen, S.; Zhao, Y. Contribution of microbial community to flavor formation in tilapia sausage during fermentation with Pediococcus pentosaceus. LWT 2022, 154, 112628. [Google Scholar] [CrossRef]
- Xu, X.; Wu, B.; Zhao, W.; Lao, F.; Chen, F.; Liao, X.; Wu, J. Shifts in autochthonous microbial diversity and volatile metabolites during the fermentation of chili pepper (Capsicum frutescens L.). Food Chem. 2021, 335, 127512. [Google Scholar] [CrossRef]
- Jiang, L.; Mu, Y.; Su, W.; Tian, H.; Zhao, M.; Su, G.; Zhao, C. Effects of Pediococcus acidilactici and Rhizopus Oryzae on microbiota and metabolomic profiling in fermented dry-cure mutton sausages. Food Chem. 2023, 403, 134431. [Google Scholar] [CrossRef]



| Materials | Douban | Laozao | Douchi | Furu | Salt |
|---|---|---|---|---|---|
| Proportion of lean meat and fat | 4% | 3% | 2% | 1.5% | 1% |
| Volatile Flavor Components | RT | CAS | VIP |
|---|---|---|---|
| Hexanal | 6.071 | 66–25–1 | 2.80774 |
| Ethyl butanoate | 6.228 | 105–54–4 | 2.67423 |
| Anethole | 29.252 | 104–46–1 | 2.40669 |
| Ethyl hexanoate | 14.902 | 123–66–0 | 2.37757 |
| Ethyl caprylate | 25.189 | 106–32–1 | 2.19615 |
| Acetoin | 3.710 | 513–86–0 | 2.15824 |
| D-limonene | 16.003 | 5989–27–5 | 2.11602 |
| Phenylethyl alcohol | 20.281 | 60–12–8 | 1.93827 |
| Ethyl caprate | 32.685 | 110–38–3 | 1.89676 |
| 2,3-Epoxy–4,4-dimethylpentane | 4.328 | 53897–30–6 | 1.67419 |
| Isoamyl acetate | 9.219 | 123–92–2 | 1.65980 |
| N-succinimidyl benzoate | 12.768 | 23405–15–4 | 1.37629 |
| Ethyl tetradecanoate | 38.560 | 124–06–1 | 1.33031 |
| Ethyl palmitate | 40.757 | 628–97–7 | 1.30464 |
| m-Cymene | 15.817 | 535–77–3 | 1.30454 |
| o-Cymene | 15.822 | 527–84–4 | 1.13554 |
| 2-Phenyl–2-butenal | 28.605 | 4411–89–6 | 1.06599 |
| Ethyl oleate | 42.483 | 111–62–6 | 1.02648 |
| Volatile Flavor Components | CAS | Sensory Threshold mg/kg | ROAV | ||||
|---|---|---|---|---|---|---|---|
| D0 | D7 | D14 | D30 | D60 | |||
| Ethyl caprylate | 106–32–1 | 0.000100 | 100 | 100 | 100 | 100 | 100 |
| Ethyl hexanoate | 123–66–0 | 0.000500 | 14.94 | 22.59 | 29.11 | 19.99 | 17.35 |
| Eucalyptol | 470–82–6 | 0.000260 | 48.68 | 3.99 | 3.28 | 2.65 | 2.58 |
| Methional | 3268–49–3 | 0.000040 | - | - | - | 9.26 | 16.17 |
| Ethyl isovalerate | 108–64–5 | 0.000200 | - | 4.88 | 5.79 | 3.45 | 3.29 |
| Linalool | 78–70–6 | 0.001500 | 11.99 | 1.34 | 1.35 | 1.36 | 1.10 |
| ethyl butanoate | 105–54–4 | 0.001000 | - | - | 4.34 | 1.91 | 2.29 |
| Nonanal | 124–19–6 | 0.003500 | 5.54 | 0.87 | 0.72 | 0.65 | 0.35 |
| D-limonene | 5989–27–5 | 0.034000 | 5.95 | 0.63 | 0.53 | 0.59 | 0.40 |
| (E)–2-nonenal | 18829–56–6 | 0.000065 | - | 4.75 | 3.37 | - | - |
| Estragole | 140–67–0 | 0.007500 | 5.22 | 0.50 | 0.43 | 0.65 | 0.51 |
| Ethyl 2-methylbutanoate | 7452–79–1 | 0.000150 | - | 1.90 | 2.55 | - | 0.96 |
| Anethole | 104–46–1 | 0.100000 | 2.11 | 0.22 | 0.17 | 0.30 | 0.20 |
| Phenylacetaldehyde | 122–78–1 | 0.009000 | 1.60 | 0.17 | 0.17 | 0.21 | 0.30 |
| 1-Octen–3-ol | 3391–86–4 | 0.010000 | 1.09 | 0.14 | 0.09 | 0.06 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Kang, X.; Wang, X.; Chen, X.; Nie, X.; Xiang, L.; Liu, D.; Zhao, Z. Potential Correlation between Microbial Diversity and Volatile Flavor Substances in a Novel Chinese-Style Sausage during Storage. Foods 2023, 12, 3190. https://doi.org/10.3390/foods12173190
Chen H, Kang X, Wang X, Chen X, Nie X, Xiang L, Liu D, Zhao Z. Potential Correlation between Microbial Diversity and Volatile Flavor Substances in a Novel Chinese-Style Sausage during Storage. Foods. 2023; 12(17):3190. https://doi.org/10.3390/foods12173190
Chicago/Turabian StyleChen, Hongfan, Xinyue Kang, Xinyi Wang, Xinya Chen, Xin Nie, Lu Xiang, Dayu Liu, and Zhiping Zhao. 2023. "Potential Correlation between Microbial Diversity and Volatile Flavor Substances in a Novel Chinese-Style Sausage during Storage" Foods 12, no. 17: 3190. https://doi.org/10.3390/foods12173190
APA StyleChen, H., Kang, X., Wang, X., Chen, X., Nie, X., Xiang, L., Liu, D., & Zhao, Z. (2023). Potential Correlation between Microbial Diversity and Volatile Flavor Substances in a Novel Chinese-Style Sausage during Storage. Foods, 12(17), 3190. https://doi.org/10.3390/foods12173190






