Hydroxypropyl Methylcellulose and Gum Arabic Composite Edible Coatings Amended with Geraniol to Control Postharvest Brown Rot and Maintain Quality of Cold-Stored Plums
Abstract
1. Introduction
2. Materials and Methods
2.1. Coating Materials and Ingredients
2.2. Preparation of Antifungal Edible Coatings
2.3. Fruit Samples
2.4. Effect of Coatings on Brown Rot Control
2.4.1. Fungal Pathogen and Inoculum Preparation
2.4.2. Curative Activity of Antifungal Coatings
2.5. Effect of Coatings on Plum Fruit Quality
2.5.1. Plum Weight Loss
2.5.2. Plum Flesh Firmness
2.5.3. Plum Peel Color
2.5.4. Plum Juice Quality
2.5.5. Ethanol and Acetaldehyde Content
2.5.6. Plum Physiological Disorders
2.5.7. Plum Sensory Analysis
2.6. Statistical Analysis
3. Result and Discussion
3.1. Effect of Coatings on Brown Rot Development
3.2. Effect of Coatings on Plum Fruit Quality
3.2.1. Plum Weight Loss
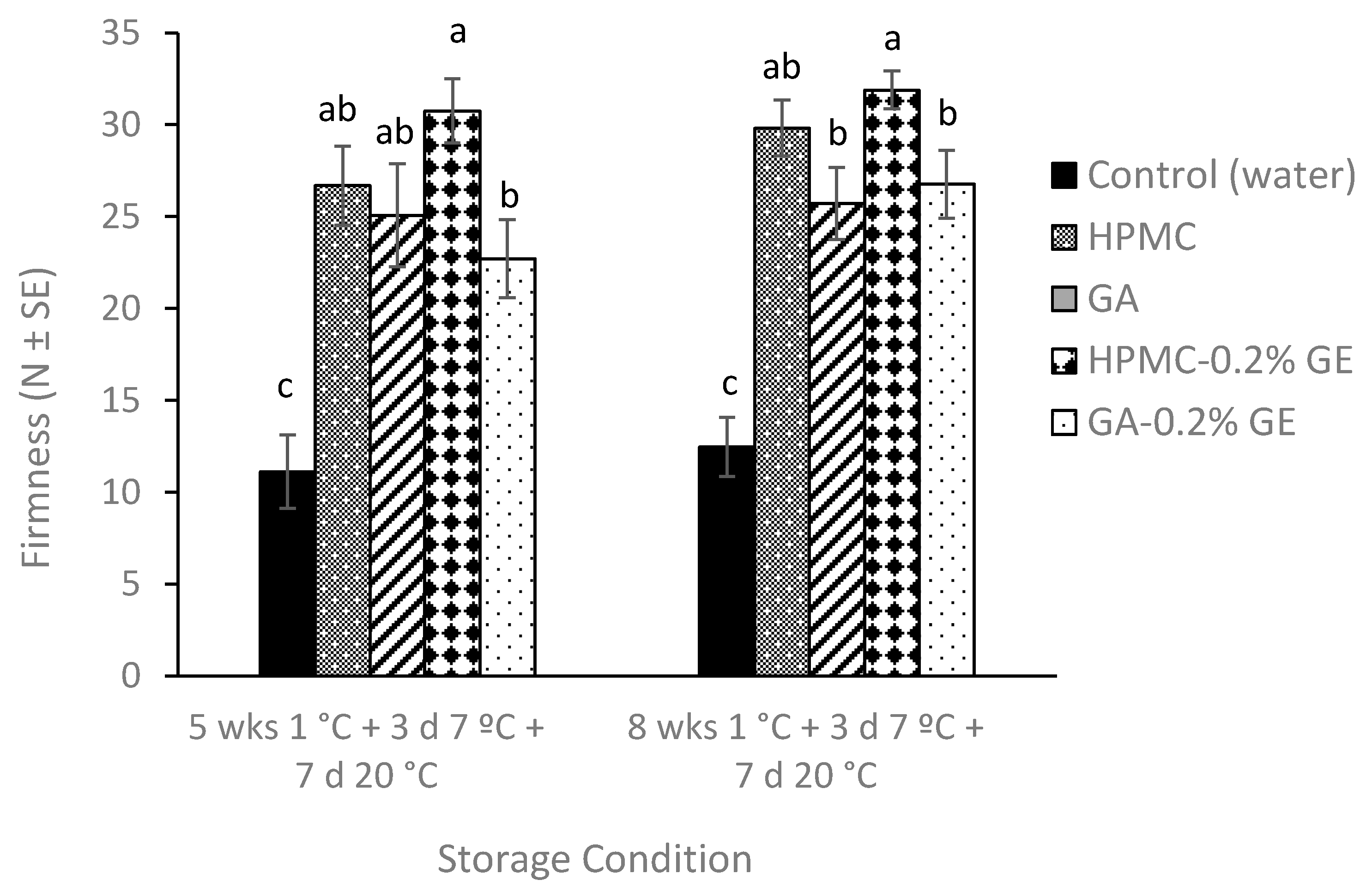
3.2.2. Plum Flesh Firmness
3.2.3. Plum Peel Color
3.2.4. Plum Internal Quality
3.2.5. Ethanol and Acetaldehyde Contents
3.2.6. Plum Physiological Disorders
3.2.7. Plum Sensory Quality
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pérez-Gago, M.B.; Rojas, C.; Del Río, M.A. Effect of hydroxypropyl methylcellulose-lipid edible composite coatings on plum (cv. Autumn giant) quality during storage. J. Food Sci. 2003, 68, 879–883. [Google Scholar]
- Wang, C.Y. Approaches to reduce chilling injury of fruits and vegetables. Hortic. Rev. 1993, 15, 63–95. [Google Scholar]
- Lurie, S.; Crisosto, C.H. Chilling injury in peach and nectarine. Postharvest Biol. Technol. 2005, 37, 195–208. [Google Scholar] [CrossRef]
- Karabulut, O.A.; Smilanick, J.L.; Crisosto, C.H.; Palou, L. Control of brown rot of stone fruits by brief heated water immersion treatments. Crop. Prot. 2010, 29, 903–906. [Google Scholar] [CrossRef]
- Mari, M.; Spadaro, D.; Casals, C.; Collina, M.; De Cal, A.; Usall, J. Stone fruits. In Postharvest Pathology of Fresh Horticultural Produce; Palou, L., Smilanick, J.L., Eds.; CRC Press; Taylor and Francis Group: Boca Raton, FL, USA, 2020; pp. 111–140. ISBN 9781138630833. [Google Scholar]
- Karaca, H.; Pérez-Gago, M.B.; Taberner, V.; Palou, L. Evaluating food additives as antifungal agents against Monilinia fructicola in vitro and in hydroxypropyl methylcellulose–lipid composite edible coatings for plums. Int. J. Food Microbiol. 2014, 179, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Usall, J.; Casals, C.; Sisquella, M.; Palou, L.; De Cal, A. Alternative technologies to control postharvest diseases of stone fruits. Stewart Postharvest Rev. 2015, 11, 1–6. [Google Scholar]
- Choi, W.S.; Singh, S.; Lee, Y.S. Characterization of edible film containing essential oils in hydroxypropyl methylcellulose and its effect on quality attributes of ‘Formosa’ plum (Prunus salicina L.). LWT—Food Sci. Technol. 2016, 70, 213–222. [Google Scholar] [CrossRef]
- Shiekh, K.A.; Ngiwngam, K.; Tongdeesoontorn, W. Polysaccharide-based active coatings incorporated with bioactive compounds for reducing postharvest losses of fresh fruits. Coatings 2022, 12, 8. [Google Scholar] [CrossRef]
- Patil, V.A.; Baswal, A.K.; Parab, A.; Patil, V.; Jakhar, V. Effect of hydroxypropyl methylcellulose and methylcellulose-based edible coatings on storage life and quality of horticultural crops: Review. Pharma Innov. J. 2022, 11, 2746–2752. [Google Scholar]
- Baswal, A.K.; Dhaliwal, H.S.; Singh, Z.; Mahajan, B.V.C.; Kalia, A.; Gill, K.S. Influence of carboxy methylcellulose, chitosan and beeswax coatings on cold storage life and quality of Kinnow mandarin fruit. Sci. Hortic. 2020, 260, 108887. [Google Scholar] [CrossRef]
- De Souza, E.L.; Ramos Berger, L.R.; Marín, A.; Pérez-Gago, M.B.; Palou, L. Chitosan and other edible coatings for postharvest disease control. In Postharvest Pathology of Fresh Horticultural Produce; Palou, L., Smilanick, J.L., Eds.; CRC Press; Taylor and Francis Group: Boca Raton, FL, USA, 2020; pp. 677–711. ISBN 9781138630833. [Google Scholar]
- Basiak, E.; Linke, M.; Debeaufort, F.; Lenart, A.; Geyer, M. Impact of Biodegradable Materials on the Quality of Plums. Coatings 2022, 12, 226. [Google Scholar] [CrossRef]
- Kaur, P.; Sandhu, K.S.; Bangar, S.P.; Whiteside, W.S. Effect of edible coating from octenyl succinic anhydride modified rye starch to extend shelf life of plums. Int. J. Food Sci. Technol. 2023, 58, 987–994. [Google Scholar] [CrossRef]
- More, P.R.; Pegu, K.; Arya, S.S. Post-harvest application of micellar pomegranate peel extract (MPPE) enriched starch-casein composite coating to preserve the plum (Prunus salicina L.) fruit during cold and ambient storage. J. Food Process. Preserv. 2022, 46, e17226. [Google Scholar] [CrossRef]
- Thakur, R.; Pristijono, P.; Golding, J.B.; Stathopoulos, C.E.; Scarlett, C.J.; Bowyer, M.; Vuong, Q.V. Development and application of rice starch based edible coating to improve the postharvest storage potential and quality of plum fruit (Prunus salicina). Sci. Hortic. 2018, 237, 59–66. [Google Scholar] [CrossRef]
- Martínez-Romero, D.; Zapata, P.J.; Guillén, F.; Paladines, D.; Castillo, S.; Valero, D.; Serrano, M. The addition of rosehip oil to Aloe gels improves their properties as postharvest coatings for maintaining quality in plum. Food Chem. 2017, 217, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Adiletta, G.; Gliottone, G.; Di Matteo, M.; Petriccione, M. Response of qualitative traits and antioxidant systems to chitosan postharvest treatment in ‘black Golden’ Japanese plum. Foods 2022, 11, 853. [Google Scholar] [CrossRef]
- Mahmoudi, R.; Razavi, F.; Rabiei, V.; Gohari, G.; Palou, L. Application of Glycine betaine coated chitosan nanoparticles alleviate chilling injury and maintain quality of plum (Prunus domestica L.) fruit. Int. J. Biol. Macromol. 2022, 207, 965–977. [Google Scholar] [CrossRef]
- Kumar, P.; Sethi, S.; Sharma, R.R.; Srivastav, M.; Varghese, E. Effect of chitosan coating on postharvest life and quality of plum during storage at low temperature. Sci. Hortic. 2017, 226, 104–109. [Google Scholar] [CrossRef]
- Panahirad, S.; Naghshiband-Hassani, R.; Bergin, S.; Katam, R.; Mahna, N. Improvement of postharvest quality of plum (Prunus domestica L.) using polysaccharide-based edible coatings. Plants 2020, 9, 1148. [Google Scholar] [CrossRef]
- Fawole, O.A.; Riva, S.C.; Opara, U.L. Efficacy of edible coatings in alleviating shrivel and maintaining quality of Japanese plum (Prunus salicina Lindl.) during export and shelf life conditions. Agronomy 2020, 10, 1023. [Google Scholar] [CrossRef]
- Andrade, S.C.; Baretto, T.A.; Arcanjo, N.M.; Madruga, M.S.; Meireles, B.; Cordeiro, Â.M.; Magnani, M. Control of Rhizopus soft rot and quality responses in plums (Prunus domestica L.) coated with gum arabic, oregano and rosemary essential oils. J. Food Process. Preserv. 2017, 41, e13251. [Google Scholar] [CrossRef]
- Gunaydin, S.; Karaca, H.; Palou, L.; de la Fuente, B.; Pérez-Gago, M.B. Effect of hydroxypropyl methylcellulose-beeswax composite edible coatings formulated with or without antifungal agents on physicochemical properties of plums during cold storage. J. Food Qual. 2017, 2017, 8573549. [Google Scholar] [CrossRef]
- Rajeswara Rao, B.R.; Sastry, K.P.; Saleem, S.M.; Prakasa Rao, E.V.S.; Syamasundar, K.V.; Ramesh, S. Volatile flower oils of three genotypes of rose-scented geranium (Pelargonium sp.). Flavour Fragr. J. 2000, 15, 105–107. [Google Scholar] [CrossRef]
- Iijima, Y.; Gang, D.R.; Fridman, E.; Lewinsohn, E.; Pichersky, E. Characterization of geraniol synthase from the peltate glands of sweet basil. Plant Physiol. 2004, 134, 370–379. [Google Scholar] [CrossRef]
- Mączka, W.; Wińska, K.; Grabarczyk, M. One hundred faces of geraniol. Molecules 2020, 25, 3303. [Google Scholar] [CrossRef] [PubMed]
- Parikh, L.; Agindotan, B.O.; Burrows, M.E. Antifungal activity of plant-derived essential oils on pathogens of pulse crops. Plant Dis. 2021, 105, 1692–1701. [Google Scholar] [CrossRef] [PubMed]
- Tsao, R.; Zhou, T. Antifungal activity of monoterpenoids against postharvest pathogens Botrytis cinerea and Monilinia fructicola. J. Essent. Oil Res. 2000, 12, 113–121. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Palou, L.; Taberner, V.; Fernández-Catalán, A.; Argente-Sanchis, M.; Pitta, E.; Pérez-Gago, M.B. Natural Pectin-Based Edible Composite Coatings with Antifungal Properties to Control Green Mold and Reduce Losses of ‘Valencia’Oranges. Foods 2022, 11, 1083. [Google Scholar] [CrossRef]
- Crisosto, C.H.; Mitchell, F.G.; Ju, Z. Susceptibility to chilling injury of peach, nectarine, and plum cultivars grown in California. HortScience 1999, 34, 1116–1118. [Google Scholar] [CrossRef]
- ISO 8586:2012; Sensory Analysis—General Guidelines for the Selection, Training and Monitoring of Selected Assessors and Expert Sensory Assessors. International Organization for Standardization: Geneva, Switzerland, 2012.
- Kim, I.H.; Lee, H.; Kim, J.E.; Song, K.B.; Lee, Y.S.; Chung, D.S.; Min, S.C. Plum coatings of lemongrass oil-incorporating carnauba wax-based nanoemulsion. J. Food Sci. 2013, 78, E1551–E1559. [Google Scholar] [CrossRef]
- Chutia, M.; Bhuyan, P.D.; Pathak, M.G.; Sarma, T.C.; Boruah, P. Antifungal activity and chemical composition of Citrus reticulata Blanco essential oil against phytopathogens from North East India. LWT-Food Sci. Technol. 2009, 42, 777–780. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, Z.; Wei, M.; Xie, Y.; He, S.; Shi, H.; Lin, Z. Evaluation of the antifungal activity of individual and combine monoterpenes against Rhizopus stolonifer and Absidia coerulea. Environ. Sci. Pollut. Res. 2019, 26, 7804–7809. [Google Scholar] [CrossRef] [PubMed]
- Scariot, F.J.; Foresti, L.; Delamare, A.P.L.; Echeverrigaray, A.S. Activity of monoterpenoids on the in vitro growth of two Colletotrichum species and the mode of action on C. acutatum. Pestic. Biochem. Physiol. 2020, 170, 104698. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Catalan, A.; Palou, L.; Taberner, V.; Grimal, A.; Argente-Sanchis, M.; Perez-Gago, M.B. Hydroxypropyl Methylcellulose-Based Edible Coatings Formulated with Antifungal Food Additives to Reduce Alternaria Black Spot and Maintain Postharvest Quality of Cold-Stored ‘Rojo Brillante’Persimmons. Agronomy 2021, 11, 757. [Google Scholar] [CrossRef]
- Palou, L.; Valencia-Chamorro, S.A.; Pérez-Gago, M.B. Antifungal edible coatings for fresh citrus fruit: A review. Coatings 2015, 5, 962–986. [Google Scholar] [CrossRef]
- Navarro-Tarazaga, M.L.; Massa, A.; Pérez-Gago, M.B. Effect of beeswax content on hydroxypropyl methylcellulose-based edible film properties and postharvest quality of coated plums (cv. Angeleno). LWT-Food Sci. Technol. 2011, 44, 2328–2334. [Google Scholar] [CrossRef]
- Bai, J.; Baldwin, E.A.; Hagenmaier, R.H. Alternatives to shellac coatings provide comparable gloss, internal gas modification, and quality for ‘Delicious’ apple fruit. HortScience 2002, 37, 559–563. [Google Scholar] [CrossRef]
- Fagundes, C.; Palou, L.; Monteiro, A.R.; Perez-Gago, M.B. Effect of antifungal hydroxypropyl methylcellulose-beeswax edible coatings on gray mold development and quality attributes of cold-stored cherry tomato fruit. Postharvest Biol. Technol. 2014, 92, 1–8. [Google Scholar] [CrossRef]
- Pastor, C.; Sánchez-González, L.; Marcilla, A.; Chiralt, A.; Cháfer, M.; González-Martínez, C. Quality and safety of table grapes coated with hydroxypropylmethylcellulose edible coatings containing propolis extract. Postharvest Biol. Technol. 2011, 60, 64–70. [Google Scholar] [CrossRef]
- Kingwascharapong, P.; Arisa, K.; Karnjanapratum, S.; Tanaka, F.; Tanaka, F. Effect of gelatin-based coating containing frog skin oil on the quality of persimmon and its characteristics. Sci. Hortic. 2020, 260, 108864. [Google Scholar] [CrossRef]
- Guimarães, J.E.; de la Fuente, B.; Pérez-Gago, M.B.; Andradas, C.; Carbó, R.; Mattiuz, B.H.; Palou, L. Antifungal activity of GRAS salts against Lasiodiplodia theobromae in vitro and as ingredients of hydroxypropyl methylcellulose-lipid composite edible coatings to control Diplodia stem-end rot and maintain postharvest quality of citrus fruit. Int. J. Food Microbiol. 2019, 301, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Blay, V.; Pérez-Gago, M.B.; de la Fuente, B.; Carbó, R.; Palou, L. Edible coatings formulated with antifungal GRAS salts to control citrus anthracnose caused by Colletotrichum gloeosporioides and preserve postharvest fruit quality. Coatings 2020, 10, 730. [Google Scholar] [CrossRef]
- Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M. Effects of alginate edible coating on preserving fruit quality in four plum cultivars during postharvest storage. Postharvest Biol. Technol. 2013, 77, 1–6. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, Q.; Ge, L.; Zhang, G.; Zhang, X.; Zhang, X. Chitin enhances biocontrol of Rhodotorula mucilaginosa to postharvest decay of peaches. Int. J. Biol. Macromol. 2016, 88, 465–475. [Google Scholar] [CrossRef]
- Kasim, R.; Kasim, M.U. The effect of ultraviolet irradiation (UV-C) on chilling injury of cucumbers during cold storage. J. Food Agric. Environ. 2008, 6, 50. [Google Scholar]
- Fernandez-Cancelo, P.; Munoz, P.; Echeverria, G.; Larrigaudiere, C.; Teixido, N.; Munne-Bosch, S.; Gine-Bordonaba, J. Ethylene and abscisic acid play a key role in modulating apple ripening after harvest and after cold-storage. Postharvest Biol. Technol. 2022, 188, 111902. [Google Scholar] [CrossRef]
- Kumar, P.; Sethi, S.; Sharma, R.R.; Srivastav, M.; Singh, D.; Varghese, E. Edible coatings influence the cold-storage life and quality of ‘Santa Rosa’ plum (Prunus salicina Lindell). J. Food Sci. Technol. 2018, 55, 2344–2350. [Google Scholar] [CrossRef]
- Paladines, D.; Valero, D.; Valverde, J.M.; Díaz-Mula, H.; Serrano, M.; Martínez-Romero, D. The addition of rosehip oil improves the beneficial effect of Aloe vera gel on delaying ripening and maintaining postharvest quality of several stone fruit. Postharvest Biol. Technol. 2014, 92, 23–28. [Google Scholar] [CrossRef]
- Riva, S.C.; Opara, U.O.; Fawole, O.A. Recent developments on postharvest application of edible coatings on stone fruit: A review. Sci. Hortic. 2020, 262, 109074. [Google Scholar] [CrossRef]
- Mahfoudhi, N.; Hamdi, S. Use of Almond Gum and Gum Arabic as Novel Edible Coating to Delay Postharvest Ripening and to Maintain Sweet Cherry (Prunus avium) Quality during Storage. J. Food Process. Preserv. 2015, 39, 1499–1508. [Google Scholar] [CrossRef]
- Li, H.; Huang, Z.; Addo, K.A.; Yu, Y. Evaluation of postharvest quality of plum (Prunus salicina L. cv. ‘French’) treated with layer-by-layer edible coating during storage. Sci. Hortic. 2022, 304, 111310. [Google Scholar] [CrossRef]
- Johnston, J.W.; Gunaseelan, K.; Pidakala, P.; Wang, M.; Schaffer, R.J. Co-ordination of early and late ripening events in apples is regulated through differential sensitivities to ethylene. J. Exp. Bot. 2009, 60, 2689–2699. [Google Scholar] [CrossRef] [PubMed]
- Pech, J.C.; Bouzayen, M.; Latché, A.J.P.S. Climacteric fruit ripening: Ethylene-dependent and independent regulation of ripening pathways in melon fruit. Plant Sci. 2008, 175, 114–120. [Google Scholar] [CrossRef]
- Salehi, F. Edible coating of fruits and vegetables using natural gums: A review. Int. J. Fruit Sci. 2020, 20, S570–S589. [Google Scholar] [CrossRef]
- Tahir, H.E.; Xiaobo, Z.; Mahunu, G.K.; Arslan, M.; Abdalhai, M.; Zhihua, L. Recent developments in gum edible coating applications for fruits and vegetables preservation: A review. Carbohydr. Polym. 2019, 224, 115141. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, L.; Fan, K. Recent advances in polysaccharide-based edible coatings for preservation of fruits and vegetables: A review. Crit. Rev. Food Sci. Nutr. 2022, 1–16. [Google Scholar] [CrossRef]
- Ghadermazi, R.; Hamdipour, S.; Sadeghi, K.; Ghadermazi, R.; Khosrowshahi Asl, A. Effect of various additives on the properties of the films and coatings derived from hydroxypropyl methylcellulose—A review. Food Sci. Nutr. 2019, 7, 3363–3377. [Google Scholar] [CrossRef]
- Navarro-Tarazaga, M.L.; Sothornvit, R.; Pérez-Gago, M.B. Effect of plasticizer type and amount on hydroxypropyl methylcellulose-beeswax edible film properties and postharvest quality of coated plums (cv. Angeleno). J. Agric. Food Chem. 2008, 56, 9502–9509. [Google Scholar] [CrossRef]
- Ghadermazi, R.; Keramat, J.; Goli, S.A.H. Antioxidant activity and physical properties of hydroxypropylmethylcellulose films enriched with essential oils. J. Food Nutr. Res. 2016, 55, 22–32. [Google Scholar]
- Klangmuang, P.; Sothornvit, R. Barrier properties, mechanical properties and antimicrobial activity of hydroxypropyl methylcellulose-based nanocomposite films incorporated with Thai essential oils. Food Hydrocoll. 2016, 61, 609–616. [Google Scholar] [CrossRef]
- Crisosto, C.H.; Garner, D.; Crisosto, G.M.; Bowerman, E. Increasing ‘Blackamber’ plum (Prunus salicina Lindell) consumer acceptance. Postharvest Biol. Technol. 2004, 34, 237–244. [Google Scholar] [CrossRef]
- Candan, A.P.; Graell, J.; Larrigaudiere, C. Roles of climacteric ethylene in the development of chilling injury in plums. Postharvest Biol. Technol. 2008, 47, 107–112. [Google Scholar] [CrossRef]
- Lurie, S. Controlled atmosphere storage to decrease physiological disorders in nectarines. Int. J. Food Sci. Technol. 1992, 27, 507–514. [Google Scholar] [CrossRef]
- Panahirad, S.; Nasser, M.; Hassani, B.; Ghanbarzadeh, B.; Nahandi, F.Z. Plum shelf life enhancement by edible coating based on pectin and carboxymethyl cellulose. J. Biodivers. Environ. Sci. 2015, 7, 423–430. [Google Scholar]


| Storage Period at 1 °C | Treatment | L* | a* | b* | C* | Hue |
|---|---|---|---|---|---|---|
| 5 weeks | Control | 27.5 ± 0.4 b,c | 11.0 ± 1.0 d | 1.4 ± 0.5 d | 11.2± 1.1 d | 2.7 ± 2.5 d |
| HPMC | 26.7 ± 0.4 c | 18.1 ± 1.0 b | 6.3 ± 0.7 b | 19.3± 1.2 b | 17.3 ± 1.3 a,b | |
| GA | 27.7 ± 0.4 a,b,c | 14.7 ± 0.9 c | 3.3 ± 0.5 c | 15.1± 1.0 c | 11.0 ± 1.3 c | |
| HPMC-0.2% GE | 28.7 ± 0.5 a | 22.6 ± 0.8 a | 9.0 ± 0.6 a | 24.4± 1.0 a | 21.3 ± 0.7 a | |
| GA-0.2% GE | 27.9 ± 0.4 a,b | 16.8 ± 1.1 b,c | 5.1 ± 0.6 b | 17.6± 1.2 b,c | 15.0 ± 1.3 b,c | |
| 8 weeks | Control | 28.5 ± 0.4 a | 11.9 ± 0.8 c | 0.7 ± 0.4 c | 12.1 ± 0.8 c | 1.0 ± 2.2 d |
| HPMC | 24.9 ± 0.4 c | 15.6 ± 1.0 b | 4.3 ± 0.5 b | 16.2 ± 1.1 b | 14.4 ± 0.8 a,b | |
| GA | 27.3 ± 0.4 b | 14.6 ±1.1 b | 3.2 ± 0.7 b | 15.0 ± 1.3 b | 9.8 ± 1.5 c | |
| HPMC-0.2% GE | 26.9 ± 0.3 b | 19.5 ± 0.7 a | 6.5 ± 0.5 a | 20.6 ± 0.9 a | 17.8 ± 0.9 a | |
| GA-0.2% GE | 26.3 ± 0.3 b | 14.1 ±0.7 b,c | 3.0 ± 0.3 b | 14.5 ± 0.7 b,c | 11.5 ± 0.8 b,c |
| Storage Period at 1 °C | Treatment | TA (g/L Malic Acid) | SSC (°Brix) | MI |
|---|---|---|---|---|
| 5 weeks | Control | 6.6 ± 0.4 a | 16.9 ± 0.1 b | 25.6 ± 0.7 a |
| HPMC | 7.0 ± 0.5 a | 17.4 ± 0.0 a | 25.1 ± 0.7 a | |
| GA | 6.6 ± 0.2 a | 16.7 ± 0.3 b | 25.2 ± 0.3 a | |
| HPMC-0.2% GE | 6.9 ± 0.6 a | 17.1 ± 0.1 a,b | 24.9 ± 0.8 a | |
| GA-0.2% GE | 7.0 ± 0.6 a | 16.2 ± 0.2 c | 23.2 ± 0.6 b | |
| 8 weeks | Control | 6.6 ± 0.2 a | 16.5 ± 0.2 b | 25.1 ± 0.5 b |
| HPMC | 7.1 ± 0.4 a | 17.1 ± 0.1 a | 24.0 ± 0.5 b | |
| GA | 6.8 ± 0.4 a | 17.4 ± 0.3 a | 25.5 ± 0.4 b | |
| HPMC-0.2% GE | 6.5 ± 0.6 a | 16.3 ± 0.2 b | 25.0 ± 0.9 b | |
| GA-0.2% GE | 6.1 ± 0.2 a | 17.3 ± 0.1 a | 28.2 ± 0.3 a |
| Storage Period at 1 °C | Treatment | Ethanol (mg/L) | Acetaldehyde (mg/L) |
|---|---|---|---|
| 5 weeks | Control | 13.5 ± 7.3 c | 5.7 ± 1.2 a |
| HPMC | 51.3 ± 3.7 b | 4.0 ± 0.3 b,c | |
| GA | 3.1 ± 0.2 c | 3.0 ± 0.6 c | |
| HPMC-0.2% GE | 94.0 ± 19.5 a | 5.2 ± 0.9 a,b | |
| GA-0.2% GE | 16.6 ± 5.9 c | 3.8 ± 0.7 b,c | |
| 8 weeks | Control | 21.3 ± 6.9 c | 15.8 ± 1.2 b |
| HPMC | 212.7 ± 91.8 b | 12.5 ± 2.2 c | |
| GA | 15.4 ± 1.2 c | 8.9 ± 2.0 d | |
| HPMC-0.2% GE | 587.7± 44.5 a | 20.7 ± 1.4 a | |
| GA-0.2% GE | 46.2 ± 14.1 c | 6.7± 0.8 d |
| Treatment | Storage Conditions | |
|---|---|---|
| 5 Weeks 1 °C + 3 d 7 °C + 7 d 20 °C | 8 Weeks 1 °C + 3 d 7 °C + 7 d 20 °C | |
| Control | 2.4 ± 0.1 a | 3.0 ± 0.0 a |
| HPMC | 1.9 ± 0.2 b | 1.9 ± 0.2 c |
| GA | 2.1 ± 0.2 a,b | 2.7 ± 0.1 a,b |
| HPMC-0.2% GE | 1.5 ± 0.1 c | 1.9 ± 0.2 c |
| GA-0.2% GE | 2.0 ± 0.2 b | 2.6 ± 0.1 b |
| Storage Period at 1 °C | Treatment | Overall Flavor | Off-Flavor | Firmness |
|---|---|---|---|---|
| 5 weeks | Control | 6.3 ± 0.4 a | 1.1 ± 0.1 a | 2.5 ± 0.2 b |
| HPMC | 5.6 ± 0.3 a | 1.4 ± 0.1 a | 3.3 ± 0.3 a | |
| GA | 5.8 ± 0.4 a | 1.3 ± 0.2 a | 3.7 ± 0.2 a | |
| HPMC-0.2% GE | 5.8 ± 0.4 a | 1.3 ± 0.2 a | 3.8 ± 0.2 a | |
| GA-0.2% GE | 6.0 ± 0.5 a | 1.1 ± 0.1 a | 3.6 ± 0.2 a | |
| 8 weeks | Control | 4.2 ± 0.5 a | 1.0 ± 0.1 a | 2.8 ± 0.2 b |
| HPMC | 4.3 ± 0.3 a | 1.0 ± 0.1 a | 3.3 ± 0.2 a | |
| GA | 4.3 ± 0.4 a | 1.0 ± 0.2 a | 3.3 ± 0.2 a | |
| HPMC-0.2% GE | 4.5 ± 0.4 a | 1.0 ± 0.2 a | 3.5 ± 0.2 a | |
| GA-0.2% GE | 5.0 ± 0.4 a | 1.0 ± 0.0 a | 4.0 ± 0.2 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asgarian, Z.S.; Palou, L.; Souza, R.F.L.d.; Quintanilla, P.G.; Taberner, V.; Karimi, R.; Pérez-Gago, M.B. Hydroxypropyl Methylcellulose and Gum Arabic Composite Edible Coatings Amended with Geraniol to Control Postharvest Brown Rot and Maintain Quality of Cold-Stored Plums. Foods 2023, 12, 2978. https://doi.org/10.3390/foods12152978
Asgarian ZS, Palou L, Souza RFLd, Quintanilla PG, Taberner V, Karimi R, Pérez-Gago MB. Hydroxypropyl Methylcellulose and Gum Arabic Composite Edible Coatings Amended with Geraniol to Control Postharvest Brown Rot and Maintain Quality of Cold-Stored Plums. Foods. 2023; 12(15):2978. https://doi.org/10.3390/foods12152978
Chicago/Turabian StyleAsgarian, Zahra Sadat, Lluís Palou, Ricardo Felipe Lima de Souza, Paloma G. Quintanilla, Verònica Taberner, Rouhollah Karimi, and María Bernardita Pérez-Gago. 2023. "Hydroxypropyl Methylcellulose and Gum Arabic Composite Edible Coatings Amended with Geraniol to Control Postharvest Brown Rot and Maintain Quality of Cold-Stored Plums" Foods 12, no. 15: 2978. https://doi.org/10.3390/foods12152978
APA StyleAsgarian, Z. S., Palou, L., Souza, R. F. L. d., Quintanilla, P. G., Taberner, V., Karimi, R., & Pérez-Gago, M. B. (2023). Hydroxypropyl Methylcellulose and Gum Arabic Composite Edible Coatings Amended with Geraniol to Control Postharvest Brown Rot and Maintain Quality of Cold-Stored Plums. Foods, 12(15), 2978. https://doi.org/10.3390/foods12152978








