Antibacterial Effects of Theaflavins against Staphylococcus aureus and Salmonella paratyphi B: Role of Environmental Factors and Food Matrices
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Bacterial Culture Suspensions
2.3. Evaluation of Antibacterial Activity
2.4. Effect of Environmental Factors on the Antibacterial Activity of TFs
2.5. Effect of Food Matrices on the Antibacterial Activity of TFs
2.6. Statistical Analysis
3. Results
3.1. Antibacterial Effects of TFs Concentrations
3.2. Effect of Environmental Factors
3.2.1. Role of Temperature
3.2.2. Role of pH
3.2.3. Role of NaCl
3.3. Effect of Food Matrices
3.3.1. Role of Sucrose
3.3.2. Role of Skimmed Milk Powder
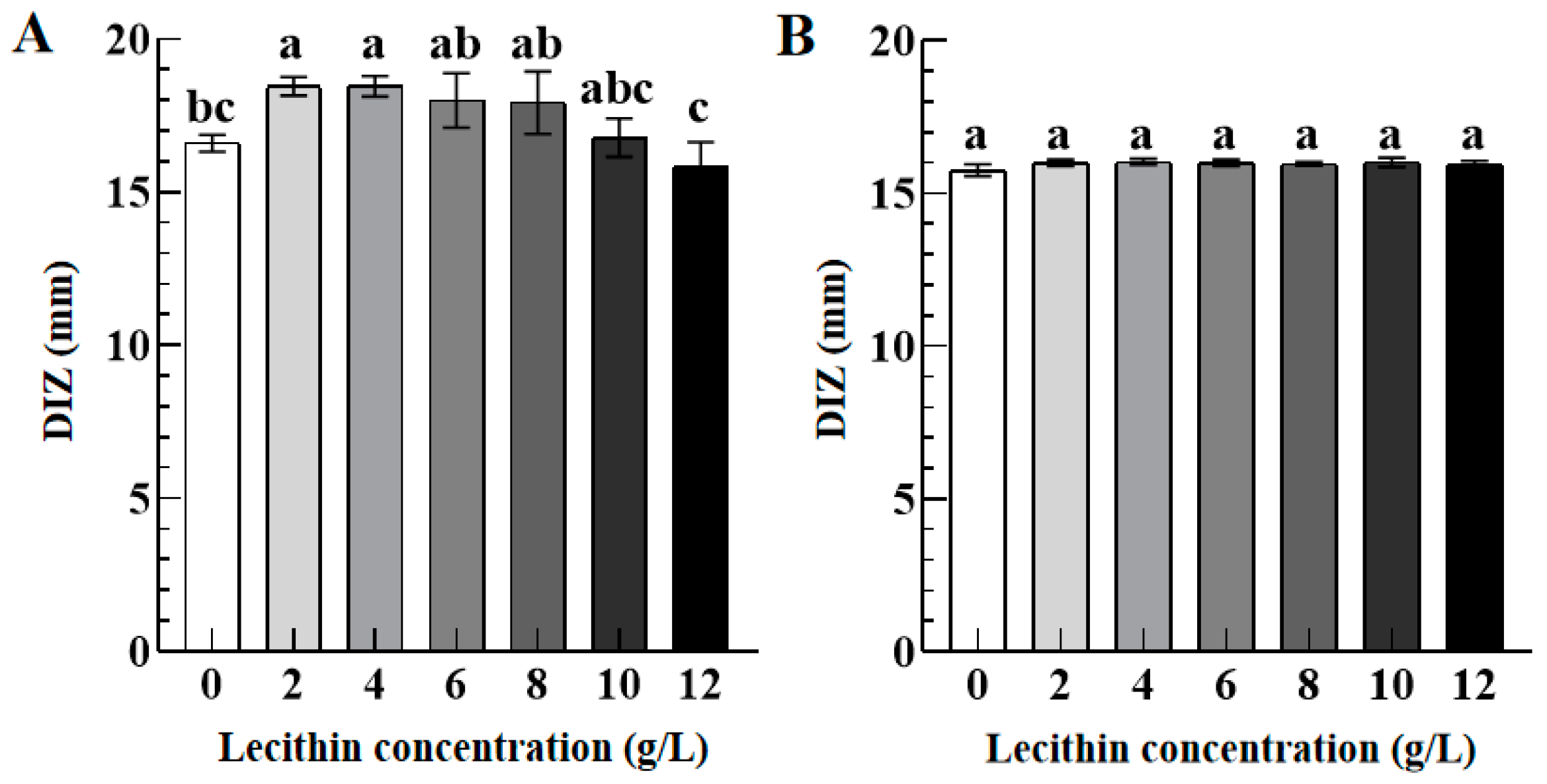
3.3.3. Role of Lecithin
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Bintsis, T. Microbial pollution and food safety. AIMS Microbiol. 2018, 4, 377–396. [Google Scholar] [CrossRef] [PubMed]
- Moretro, T.; Langsrud, S. Residential Bacteria on Surfaces in the Food Industry and Their Implications for Food Safety and Quality. Compr. Rev. Food Sci. Food Safe 2017, 16, 1022–1041. [Google Scholar] [CrossRef] [PubMed]
- Wangprasertkul, J.; Siriwattanapong, R.; Harnkarnsujarit, N. Antifungal packaging of sorbate and benzoate incorporated biodegradable films for fresh noodles. Food Control 2021, 123, 107763. [Google Scholar] [CrossRef]
- Piper, J.D.; Piper, P.W. Benzoate and Sorbate Salts: A Systematic Review of the Potential Hazards of These Invaluable Preservatives and the Expanding Spectrum of Clinical Uses for Sodium Benzoate. Compr. Rev. Food Sci. Food Safe 2017, 16, 868–880. [Google Scholar] [CrossRef] [PubMed]
- Martiny, T.R.; Raghavan, V.; de Moraes, C.C.; da Rosa, G.S.; Dotto, G.L. Optimization of green extraction for the recovery of bioactive compounds from Brazilian olive crops and evaluation of its potential as a natural preservative. J. Environ. Chem. Eng. 2021, 9, 105130. [Google Scholar] [CrossRef]
- Bae, J.Y.; Seo, Y.H.; Oh, S.W. Antibacterial activities of polyphenols against foodborne pathogens and their application as antibacterial agents. Food Sci. Biotechnol. 2022, 31, 985–997. [Google Scholar] [CrossRef]
- Shan, Z.; Nisar, M.F.; Li, M.; Zhang, C.; Wan, C.C. Theaflavin Chemistry and Its Health Benefits. Oxid. Med. Cell. Longev. 2021, 2021, 6256618. [Google Scholar] [CrossRef]
- Takemoto, M.; Takemoto, H. Synthesis of Theaflavins and Their Functions. Molecules 2018, 23, 918. [Google Scholar] [CrossRef]
- Xu, Y.; Jin, Y.X.; Wu, Y.Y.; Tu, Y.Y. Isolation and Purification of Four Individual Theaflavins Using Semi-Preparative High Performance Liquid Chromatography. J. Liq. Chromatogr. R T 2010, 33, 1791–1801. [Google Scholar] [CrossRef]
- Sato, J.; Tomita, A.; Sonoda, T.; Miyamoto, T. Theaflavin and its derivatives exert antibacterial action against Bacillus coagulans through adsorption to cell surface phospholipids. J. Appl. Microbiol. 2022, 133, 1781–1790. [Google Scholar] [CrossRef]
- Adhikary, B.; Yadav, S.K.; Roy, K.; Bandyopadhyay, S.K.; Chattopadhyay, S. Black tea and theaflavins assist healing of indomethacin-induced gastric ulceration in mice by antioxidative action. Evid. Based Complement. Alternat. Med. 2011, 2011, 546560. [Google Scholar] [CrossRef]
- He, H.F. Research progress on theaflavins: Efficacy, formation, and preparation. Food Nutr. Res. 2017, 61, 1344521. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Jie, G.; Dong, F.; Xu, Y.; Watanabe, N.; Tu, Y. Radical-scavenging abilities and antioxidant properties of theaflavins and their gallate esters in H2O2-mediated oxidative damage system in the HPF-1 cells. Toxicol. Vitro 2008, 22, 1250–1256. [Google Scholar] [CrossRef]
- Gao, X.; Xia, L.; Fan, Y.; Jin, C.; Xiong, G.; Hao, X.; Fu, L.; Lian, W. Evaluation of coloration, nitrite residue and antioxidant capacity of theaflavins, tea polyphenols in cured sausage. Meat Sci. 2022, 192, 108877. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Tu, C.H.; Mao, J.L.; Benjakul, S.; Zhang, B. Impact of theaflavin soaking pretreatment on oxidative stabilities and physicochemical properties of semi-dried large yellow croaker (Pseudosciaena crocea) fillets during storage. Food Packaging Shelf 2022, 32, 100852. [Google Scholar] [CrossRef]
- Wang, T.H.; Hsia, S.M.; Wu, C.H.; Ko, S.Y.; Chen, M.Y.; Shih, Y.H.; Shieh, T.M.; Chuang, L.C.; Wu, C.Y. Evaluation of the Antibacterial Potential of Liquid and Vapor Phase Phenolic Essential Oil Compounds against Oral Microorganisms. PLoS ONE 2016, 11, e0163147. [Google Scholar] [CrossRef]
- Liu, W.; Hansen, J.N. Some chemical and physical properties of nisin, a small-protein antibiotic produced by Lactococcus lactis. Appl. Environ. Microbiol. 1990, 56, 2551–2558. [Google Scholar] [CrossRef] [PubMed]
- Zangeneh, M.; Khorrami, S.; Khaleghi, M. Bacteriostatic activity and partial characterization of the bacteriocin produced by L. plantarum sp. isolated from traditional sourdough. Food Sci. Nutr. 2020, 8, 6023–6030. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.C.; Guo, S.Y.; Li, S.Y.; Liu, R.; Li, S.N.; Ge, Q.F. Effects of different environmental factors and food matrices on the inhibitory activity of nisin against listeria monocytogenes. Food Ferment Ind. 2022, 48, 209–221. [Google Scholar]
- Grimalt-Alemany, A.; Lezyk, M.; Asimakopoulos, K.; Skiadas, I.V.; Gavala, H.N. Cryopreservation and fast recovery of enriched syngas-converting microbial communities. Water Res. 2020, 177, 115747. [Google Scholar] [CrossRef] [PubMed]
- Mota-Meira, M.; LaPointe, G.; Lacroix, C.; Lavoie, M.C. MICs of mutacin B-Ny266, nisin A, vancomycin, and oxacillin against bacterial pathogens. Antimicrob. Agents Chemother. 2000, 44, 24–29. [Google Scholar] [CrossRef]
- de Azevedo, P.O.S.; Converti, A.; Gierus, M.; Oliveira, R.P.S. Antimicrobial activity of bacteriocin-like inhibitory substance produced by Pediococcus pentosaceus: From shake flasks to bioreactor. Mol. Biol. Rep. 2019, 46, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.H.; Marshall, D.L. Effect of pH on the Minimum Inhibitory Concentration of Monolaurin Against Listeria monocytogenes (1). J. Food Prot. 1992, 55, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Servent, A.; Cazals, G.; Perfetto, C.; Achir, N. Kinetic modeling of four folates in a model solution at different temperatures and pH to mimic their behavior in foods during processing. J. Food Process. Eng. 2023, 46, e14288. [Google Scholar] [CrossRef]
- Lee, H.J.; Tokle, I.F.; Lunestad, B.T.; Lerfall, J.; Hoel, S.; Jakobsen, A.N. The effect of food processing factors on the growth kinetics of Aeromonas strains isolated from ready-to-eat seafood. Int. J. Food Microbiol. 2023, 384, 109985. [Google Scholar] [CrossRef]
- Pei, J.; Yu, H.; Qiu, W.; Mei, J.; Xie, J. Antimicrobial Effect of Epigallocatechin Gallate Against Shewanella putrefaciens ATCC 8071: A Study Based on Cell Membrane and Biofilm. Curr. Microbiol. 2022, 79, 297. [Google Scholar] [CrossRef]
- Kim, N.H.; Kim, H.W.; Moon, H.; Rhee, M.S. Sodium chloride significantly enhances the bactericidal actions of carvacrol and thymol against the halotolerant species Escherichia coli O157:H7, Listeria monocytogenes, and Staphylococcus aureus. LWT 2020, 122, 109015. [Google Scholar] [CrossRef]
- Xiao, H.Q.; Li, Y.Z.; Lin, Q.L.; Zhao, M.J.; Liu, J.; Zhou, Q. Antimicrobial stability of metal antimicrobial peptide SIF4 in ar-tificial simulated food systems. Food Ferment Ind. 2021, 47, 121–125. [Google Scholar]
- Ferreira, S.; Domingues, F. The antimicrobial action of resveratrol against Listeria monocytogenes in food-based models and its antibiofilm properties. J. Sci. Food Agric. 2016, 96, 4531–4535. [Google Scholar] [CrossRef] [PubMed]
- Ramadan, M.F.; Asker, M.M.S. Antimicrobical and Antivirial Impact of Novel Quercetin-Enriched Lecithin. J. Food Biochem. 2009, 33, 557–571. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, Q.; Li, H.; Qiu, Z.; Yu, Y. Comparative Assessment of the Antibacterial Efficacies and Mechanisms of Different Tea Extracts. Foods 2022, 11, 620. [Google Scholar] [CrossRef]
- Borges, A.; Saavedra, M.J.; Simões, M. The activity of ferulic and gallic acids in biofilm prevention and control of pathogenic bacteria. Biofouling 2012, 28, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Bouarab-Chibane, L.; Forquet, V.; Lantéri, P.; Clément, Y.; Léonard-Akkari, L.; Oulahal, N.; Degraeve, P.; Bordes, C. Antibacterial Properties of Polyphenols: Characterization and QSAR (Quantitative Structure–Activity Relationship) Models. Front. Microbiol. 2019, 10, 829. [Google Scholar] [CrossRef]
- Oulahal, N.; Degraeve, P. Phenolic-Rich Plant Extracts with Antimicrobial Activity: An Alternative to Food Preservatives and Biocides? Front. Microbiol. 2021, 12, 753518. [Google Scholar] [CrossRef]
- Hosseiniyan Khatibi, S.M.; Zununi Vahed, F.; Sharifi, S.; Ardalan, M.; Mohajel Shoja, M.; Zununi Vahed, S. Osmolytes resist against harsh osmolarity: Something old something new. Biochimie 2019, 158, 156–164. [Google Scholar] [CrossRef]
- Ignasimuthu, K.; Prakash, R.; Murthy, P.S.; Subban, N. Enhanced bioaccessibility of green tea polyphenols and lipophilic activity of EGCG octaacetate on gram-negative bacteria. LWT Food Sci. Technol. 2019, 105, 103–109. [Google Scholar] [CrossRef]
- Fathima, A.; Rao, J.R. Selective toxicity of Catechin-a natural flavonoid towards bacteria. Appl. Microbiol. Biotechnol. 2016, 100, 6395–6402. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Oh, Y.J.; Lim, J.; Youn, M.; Lee, I.; Pak, H.K.; Park, W.; Jo, W.; Park, S. AFM study of the differential inhibitory effects of the green tea polyphenol (-)-epigallocatechin-3-gallate (EGCG) against Gram-positive and Gram-negative bacteria. Food Microbiol. 2012, 29, 80–87. [Google Scholar] [CrossRef]
- Jiang, J.; Shi, B.; Zhu, D.Q.; Cai, Q.X.; Chen, Y.R.; Li, J.N.; Qi, K.Z.; Zhang, M. Characterization of a novel bacteriocin produced by Lactobacillus sakei LSJ618 isolated from traditional Chinese fermented radish. Food Control 2012, 23, 338–344. [Google Scholar] [CrossRef]
- Lun Su, Y.; Leung, L.K.; Huang, Y.; Chen, Z.-Y. Stability of tea theaflavins and catechins. Food Chem. 2003, 83, 189–195. [Google Scholar] [CrossRef]
- Başyiğit, B.; Sağlam, H.; Köroğlu, K.; Karaaslan, M. Compositional analysis, biological activity, and food protecting ability of ethanolic extract of Quercus infectoria gall. J. Food Process. Preserv. 2020, 44, e14692. [Google Scholar] [CrossRef]
- Roila, R.; Valiani, A.; Ranucci, D.; Ortenzi, R.; Servili, M.; Veneziani, G.; Branciari, R. Antimicrobial efficacy of a polyphenolic extract from olive oil by-product against “Fior di latte” cheese spoilage bacteria. Int. J. Food Microbiol. 2019, 295, 49–53. [Google Scholar] [CrossRef]
- Stojković, D.; Petrović, J.; Soković, M.; Glamočlija, J.; Kukić-Marković, J.; Petrović, S. In situ antioxidant and antimicrobial activities of naturally occurring caffeic acid, p-coumaric acid and rutin, using food systems. J. Sci. Food Agric. 2013, 93, 3205–3208. [Google Scholar] [CrossRef] [PubMed]
- Algharib, S.A.; Dawood, A.; Xie, S. Nanoparticles for treatment of bovine Staphylococcus aureus mastitis. Drug Deliv. 2020, 27, 292–308. [Google Scholar] [CrossRef] [PubMed]
- Rda, A.; Cvab, C.; Coa, B. The synergistic effect of thurincin H and power ultrasound: An alternative for the inactivation of Listeria innocua ATCC 33090 and Escherichia coli K-12 in liquid food matrices. Food Control 2022, 135, 108778. [Google Scholar]
- Buldain, D.; Castillo, L.G.; Marchetti, M.L.; Lozano, K.J.; Bandoni, A.; Mestorino, N. Modeling the Growth and Death of Staphylococcus aureus against Melaleuca armillaris Essential Oil at Different pH Conditions. Antibiotics 2021, 10, 222. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Lambert, J.D.; Prabhu, S.; Meng, X.; Lu, H.; Maliakal, P.; Ho, C.T.; Yang, C.S. Delivery of tea polyphenols to the oral cavity by green tea leaves and black tea extract. Cancer Epidemiol. Biomark. Prev. 2004, 13, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Zou, Y.; Lee, H.Y.; Ahn, J. Effect of NaCl on the biofilm formation by foodborne pathogens. J. Food Sci. 2010, 75, 580–585. [Google Scholar] [CrossRef]
- Bremer, E.; Kramer, R. Responses of Microorganisms to Osmotic Stress. Annu. Rev. Microbiol. 2019, 73, 313–334. [Google Scholar] [CrossRef]
- Wenjiao, F.; Yunchuan, C.; Junxiu, S.; Yongkui, Z. Effects of tea polyphenol on quality and shelf life of pork sausages. J. Food Sci. Technol. 2014, 51, 191–195. [Google Scholar] [CrossRef]
- Ikigai, H.; Nakae, T.; Hara, Y.; Shimamura, T. Bactericidal catechins damage the lipid bilayer. Biochim. Biophys. Acta 1993, 1147, 132–136. [Google Scholar] [CrossRef]
- Esbelin, J.; Santos, T.; Hebraud, M. Desiccation: An environmental and food industry stress that bacteria commonly face. Food Microbiol. 2018, 69, 82–88. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. The potential application of plant essential oils as natural food preservatives in soft cheese. Food Microbiol. 2001, 18, 463–470. [Google Scholar] [CrossRef]
- Rowan-Nash, A.D.; Korry, B.J.; Mylonakis, E.; Belenky, P. Cross-Domain and Viral Interactions in the Microbiome. Microbiol. Mol. Biol. Rev. 2019, 83, e00044-18. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.X.; Yu, J.; Chen, N.; Zeng, W.C. Effects and mechanism of tea polyphenols on the quality of oil during frying process. J. Food Sci. 2020, 85, 3786–3796. [Google Scholar] [CrossRef]
- Gramza, A.; Korczak, J. Tea constituents (Camellia sinensis L.) as antioxidants in lipid systems. Trends Food Sci. Technol. 2005, 16, 351–358. [Google Scholar] [CrossRef]









Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Shan, H.; Li, P.; Liu, Y.; Zhang, X.; Xu, J.; Li, S. Antibacterial Effects of Theaflavins against Staphylococcus aureus and Salmonella paratyphi B: Role of Environmental Factors and Food Matrices. Foods 2023, 12, 2615. https://doi.org/10.3390/foods12132615
Wang J, Shan H, Li P, Liu Y, Zhang X, Xu J, Li S. Antibacterial Effects of Theaflavins against Staphylococcus aureus and Salmonella paratyphi B: Role of Environmental Factors and Food Matrices. Foods. 2023; 12(13):2615. https://doi.org/10.3390/foods12132615
Chicago/Turabian StyleWang, Jun, Hongyan Shan, Ping Li, Yanan Liu, Xun Zhang, Jingguo Xu, and Songnan Li. 2023. "Antibacterial Effects of Theaflavins against Staphylococcus aureus and Salmonella paratyphi B: Role of Environmental Factors and Food Matrices" Foods 12, no. 13: 2615. https://doi.org/10.3390/foods12132615
APA StyleWang, J., Shan, H., Li, P., Liu, Y., Zhang, X., Xu, J., & Li, S. (2023). Antibacterial Effects of Theaflavins against Staphylococcus aureus and Salmonella paratyphi B: Role of Environmental Factors and Food Matrices. Foods, 12(13), 2615. https://doi.org/10.3390/foods12132615







