Color Characterization of Bordeaux Red Wines Produced without Added Sulfites
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Red Wine Colorimetric Analysis
2.2.1. CIELab Analysis
2.2.2. Color Intensity Analysis
2.3. Total Phenolics Index
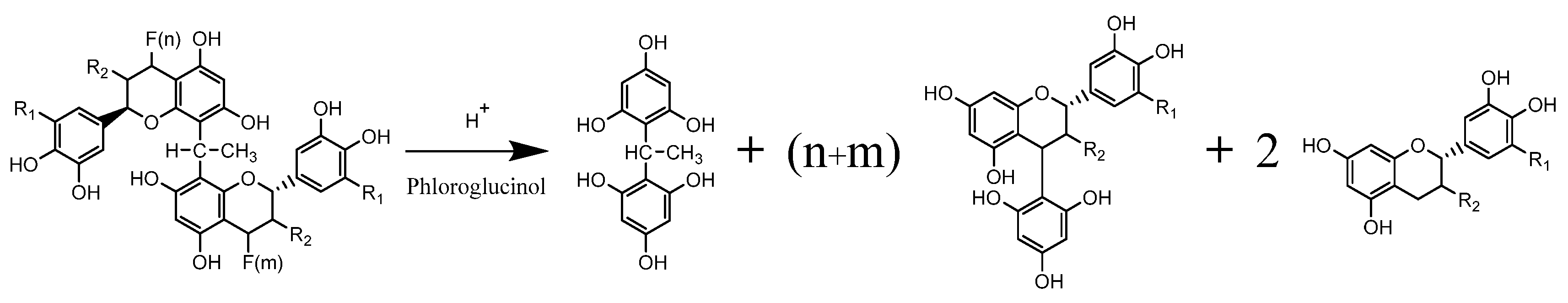
2.4. Polymeric Pigments Analyses by UPLC-DAD/ESI-QTof
2.4.1. Fractionation
2.4.2. Acidic Depolymerization
2.4.3. UPLC-DAD/ESI-QTof
2.5. Statistical Analysis
3. Results and Discussion
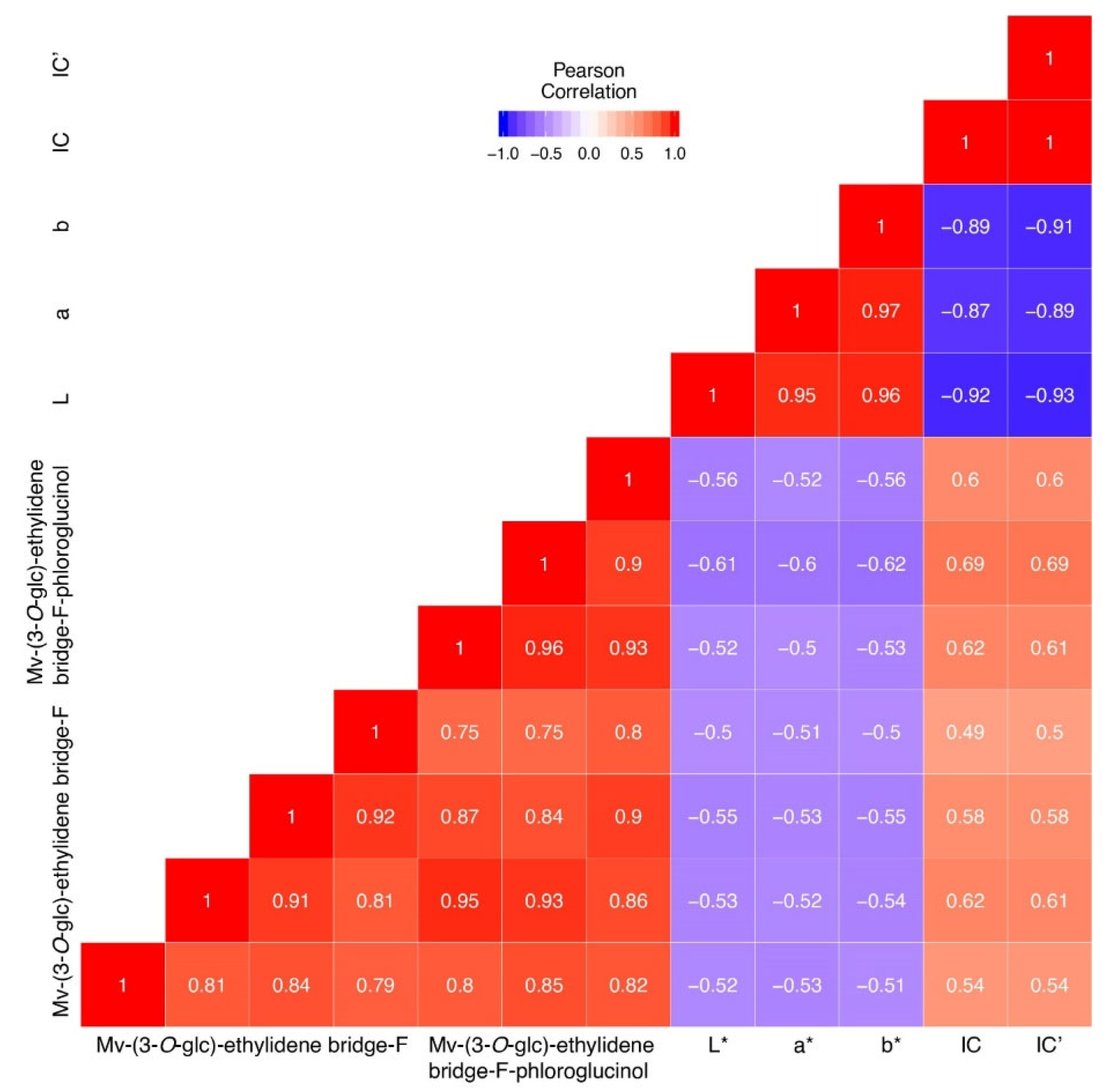
3.1. Color Parameters
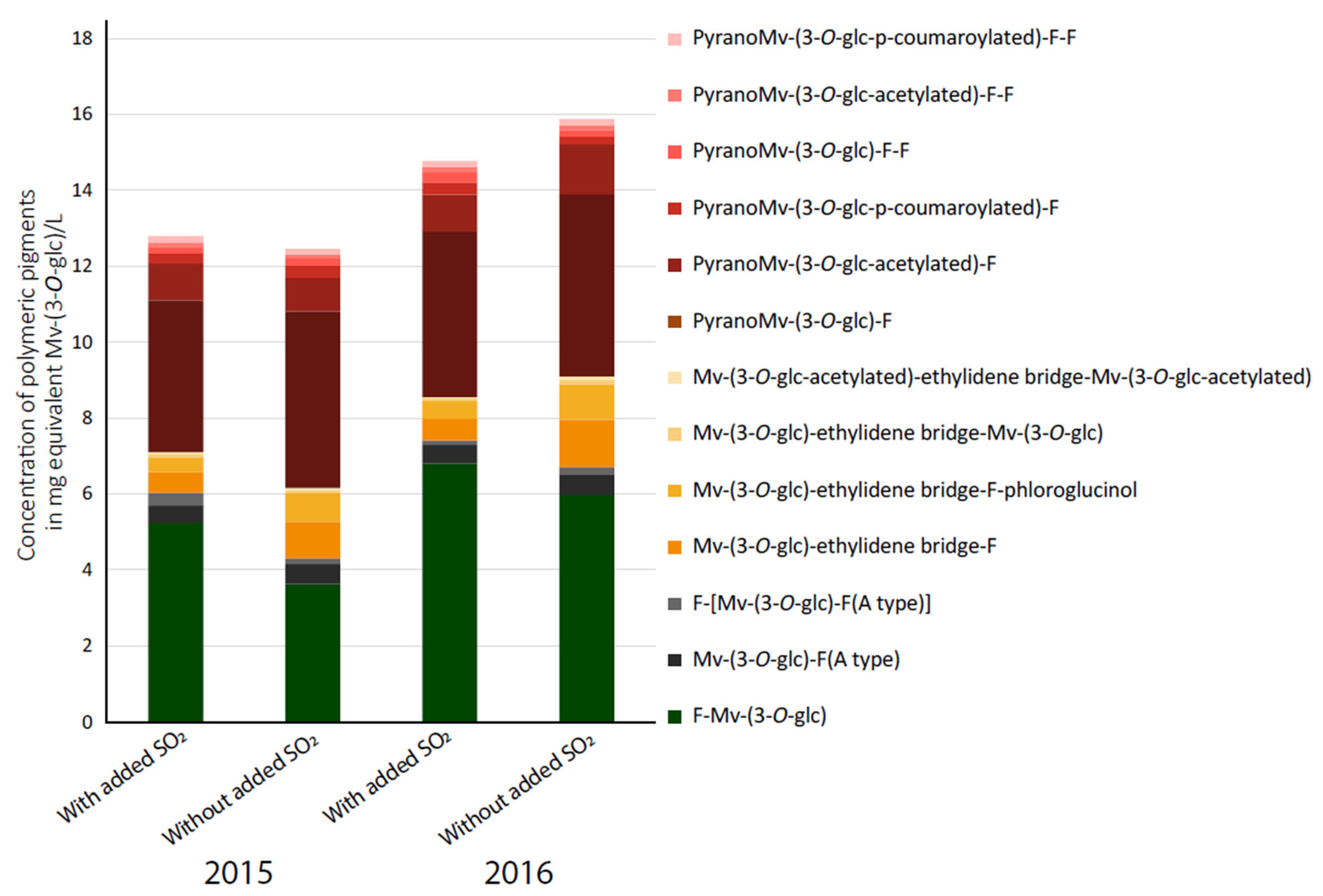
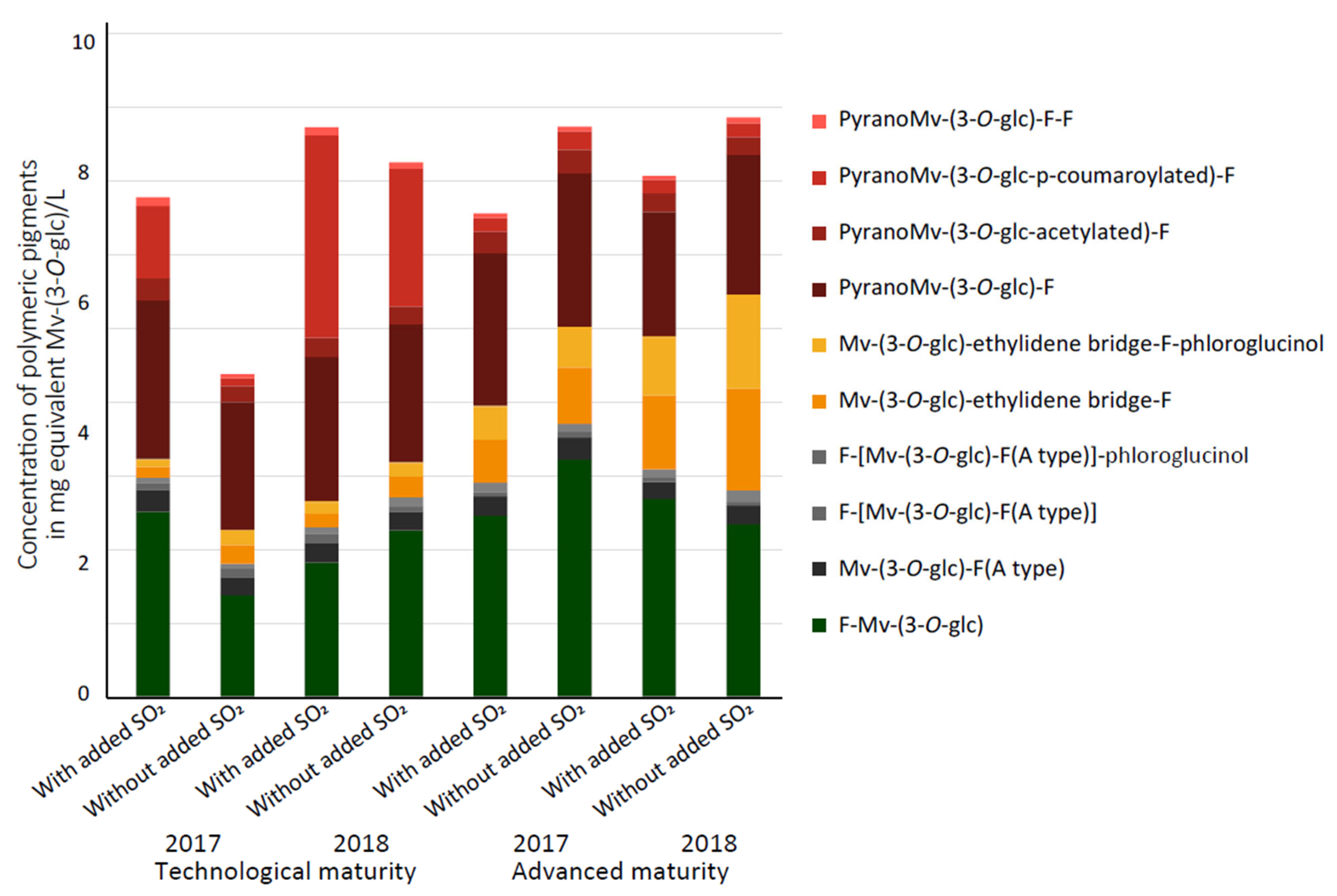
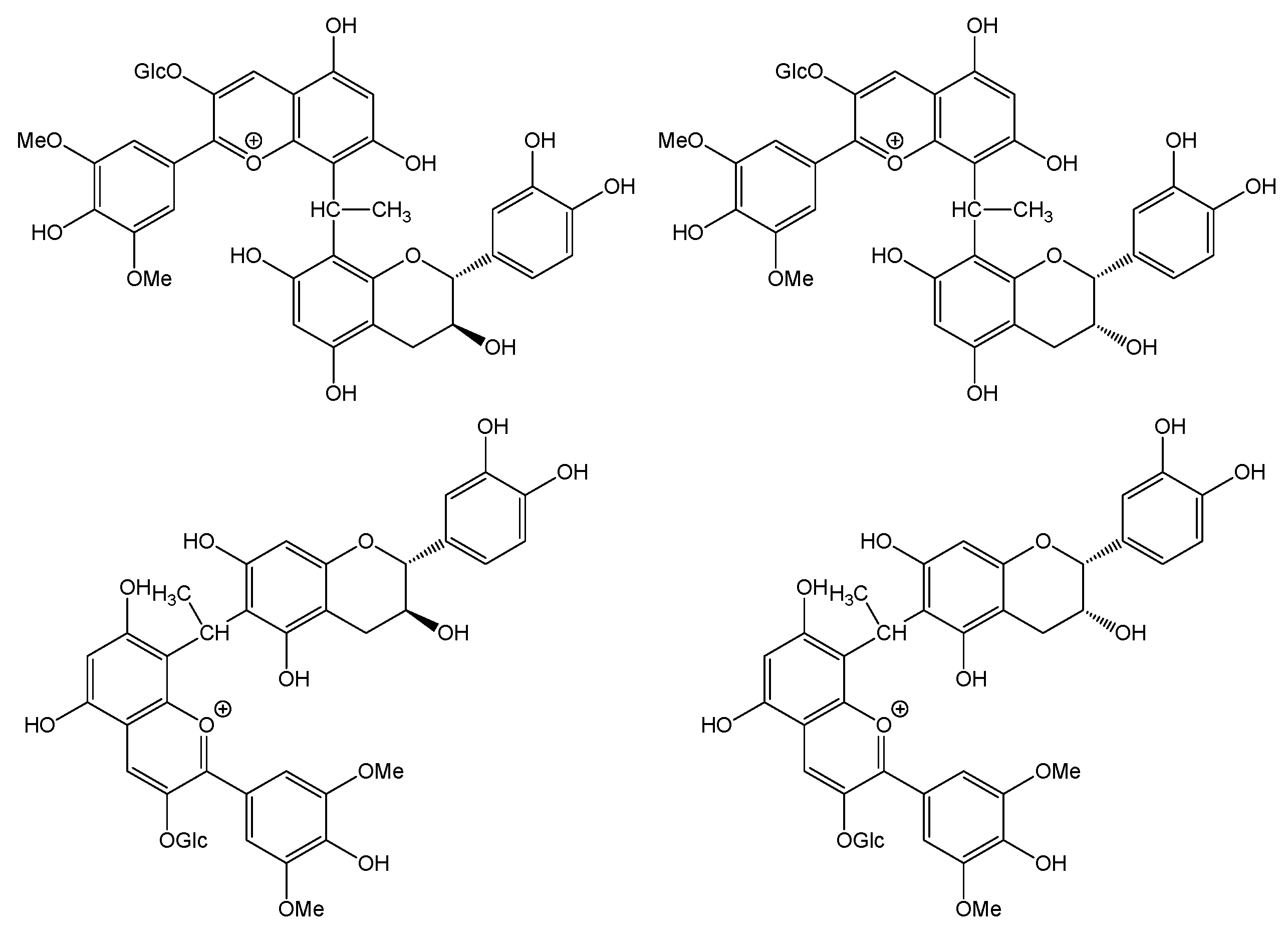
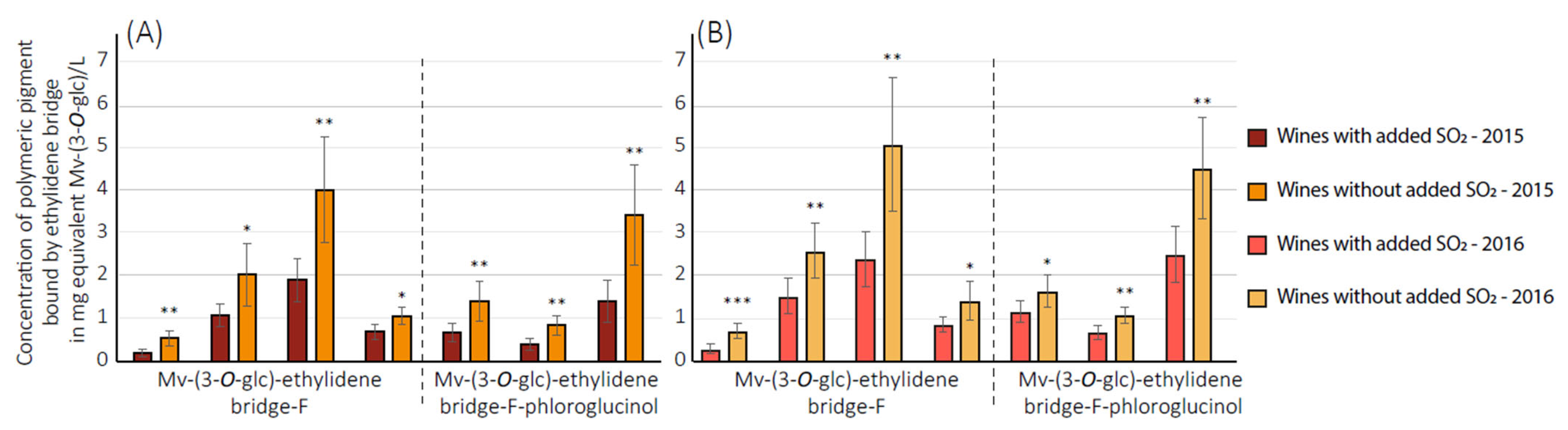
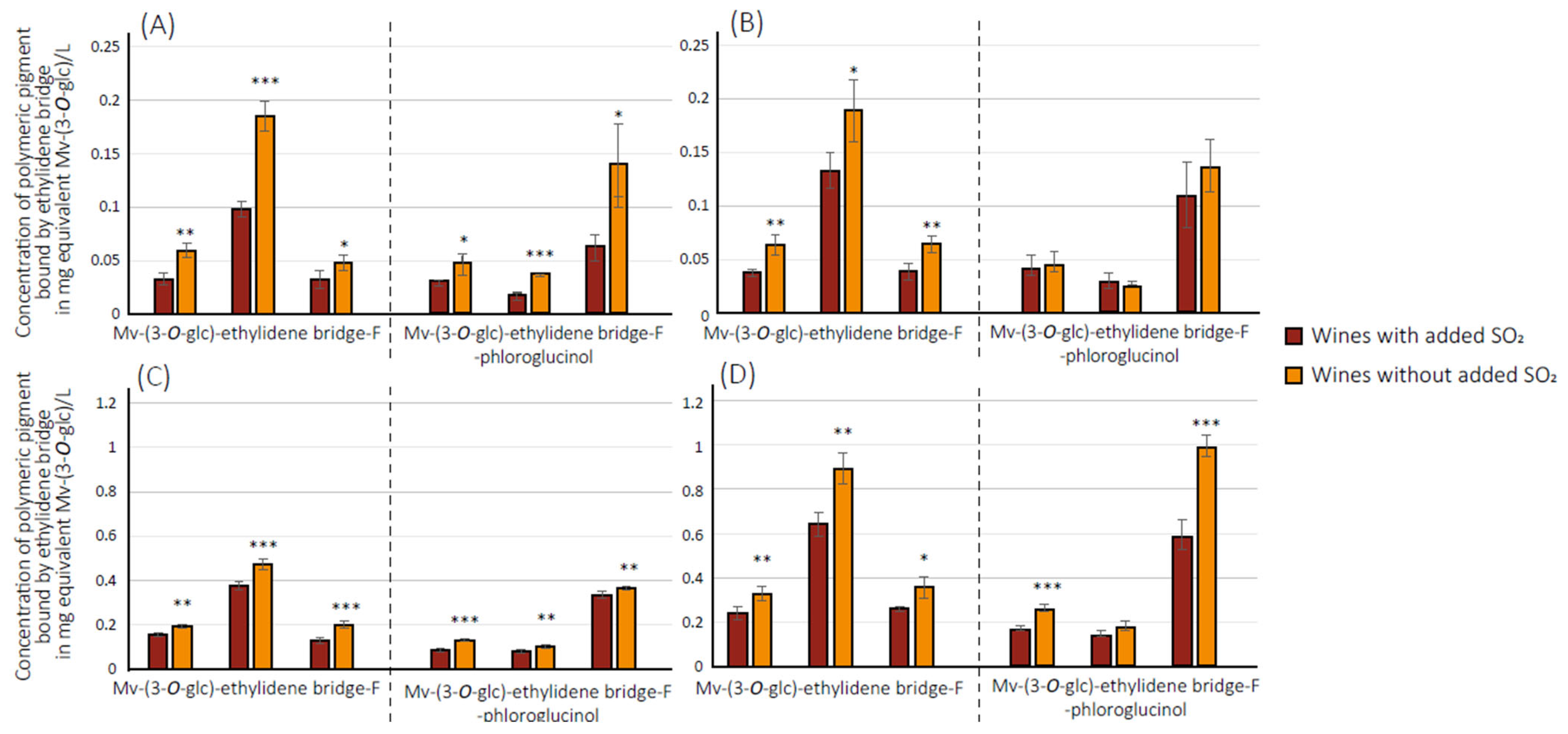
3.2. Quantification of Polymeric Pigments in Wines
3.3. Quantification of 2,2′-Ethylidenediphloroglucinol (EDP) in Wines
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Appendix A
Appendix B
| Sugar (g/L) | Total Acidity (g/L H2SO4) | pH | |
|---|---|---|---|
| Without SO2 Technological maturity 2017 | 222 | 3.10 | 3.45 |
| With SO2 Technological maturity 2017 | 222 | 3.10 | 3.45 |
| Without SO2 Advanced maturity 2017 | 234 | 2.70 | 3.58 |
| With SO2 Advanced maturity 2017 | 234 | 2.70 | 3.58 |
| Without SO2 Technological maturity 2018 | 216 | 2.80 | 3.57 |
| With SO2 Technological maturity 2018 | 216 | 2.80 | 3.57 |
| Without SO2 Advanced maturity 2018 | 219 | 2.65 | 3.61 |
| With SO2 Advanced maturity 2018 | 219 | 2.65 | 3.61 |
| No. | Name | Molecular Formula [M]+ | m/z | Retention Time (min) |
|---|---|---|---|---|
| P1 | F-Mv-(3-O-glc) | [C38H37O18]+ | 781.1974 | 4.720 |
| P2 | 5.404 | |||
| P3 | 6.679 | |||
| P4 | 7.172 | |||
| P5 | Mv-(3-O-glc)-F(A type) | [C38H37O18]+ | 783.2131 | 6.690 |
| P6 | 7.514 | |||
| P11 | F-(Mv-(3-O-glc)-F(Atype))-phloroglucinol | [C44H41O18]+ | 907.2291 | 5.490 |
| P12 | 5.648 | |||
| P13 | Mv-(3-O-glc)-ethylidene bridge-F | [C40H41O18]+ | 809.2287 | 7.839 |
| P14 | 7.980 | |||
| P15 | 8.263 | |||
| P16 | 8.396 | |||
| P17 | Mv-(3-O-glc)-ethylidene bridge-F-phloroglucinol | [C46H45O21]+ | 933.2448 | 7.562 |
| P18 | 7.845 | |||
| P19 | 8.053 | |||
| P21 | Mv-(3-O-glc)-ethylidene bridge- Mv-(3-O-glc) | [C48H51O24]+ | 1011.2765 | 9.536 |
| P22 | 11.209 | |||
| P25 | Mv-(3-O-glc-acetylated)-ethylidene bridge- Mv-(3-O-glc-acetylated) | [C52H55O26]+ | 1095.2976 | 11.028 |
| P26 | 12.886 | |||
| P42 | PyranoMv-(3-O-glc)-F | [C40H37O18]+ | 805.1974 | 9.336 |
| P43 | 9.886 | |||
| P44 | PyranoMv-(3-O-glc-acetylated)-F | [C42H39O19]+ | 847.2080 | 9.611 |
| P45 | 9.819 | |||
| P46 | PyranoMv-(3-O-glc-p-coumaroylated)-F | [C49H43O20]+ | 951.2342 | 9.769 |
| P47 | PyranoMv-(3-O-glc-)-F-F | [C45H49O24]+ | 1093.2610 | 7.353 |
| P48 | 7.436 | |||
| P49 | PyranoMv-(3-O-glc-acetylated)-F-F | [C57H51O25]+ | 1135.2714 | 7.561 |
| P50 | 7.644 | |||
| P51 | PyranoMv-(3-O-glc-p-coumaroylated)-F-F | [C64H55O26]+ | 1239.2976 | 8.468 |
| P52 | 8.468 | |||
| P53 | 8.468 | |||
| P54 | 8.468 |
| Vintage | Wine | L* | a* | b* | C* | h* | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | ||
| 2015 | With added SO2 | 21.38 | 4.66 | 50.88 | 2.99 | 34.71 | 6.26 | 61.69 | 5.84 | 0.59 | 0.06 |
| Without added SO2 | 15.91 | 4.80 | 46.72 | 4.56 | 26.56 | 7.25 | 53.87 | 7.53 | 0.50 | 0.03 | |
| 2016 | With added SO2 | 20.13 | 3.91 | 50.82 | 2.17 | 32.33 | 4.21 | 60.26 | 3.92 | 0.56 | 0.07 |
| Without added SO2 | 12.16 | 2.52 | 43.37 | 3.11 | 20.71 | 3.9 | 48.12 | 4.61 | 0.44 | 0.05 | |
References
- Dubernet, M. Les “polyphenoloxydases” du raisin sain et du raisin parasite par Botrytis cinerea. CR Acad. Sci. 1973, 277, 975–978. [Google Scholar]
- Carr, J.G.; Davies, P.A.; Sparks, A.H. The Toxicity of Sulphur Dioxide towards Certain Lactic Acid Bacteria from Fermented Apple Juice. J. Appl. Bacteriol. 1976, 40, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Albertin, W.; Miot-Sertier, C.; Bely, M.; Marullo, P.; Coulon, J.; Moine, V.; Colonna-Ceccaldi, B.; Masneuf-Pomarede, I. Oenological prefermentation practices strongly impact yeast population dynamics and alcoholic fermentation kinetics in Chardonnay grape must. Int. J. Food Microbiol. 2014, 178, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Usseglio-Tomasset, L. Properties and use of sulphur dioxide. Food Addit. Contam. 1992, 9, 399–404. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Traité D’oenologie, Tome 1—Microbiologie du Vin—Vinification, 6th ed.; Dunot: Paris, France, 2012; Volume 1. [Google Scholar]
- Vally, H.; Misso, N.L.A.; Madan, V. Clinical effects of sulphite additives. Clin. Exp. Allergy 2009, 39, 1643–1651. [Google Scholar] [CrossRef]
- Amato, M.; Ballco, P.; López-Galán, B.; De Magistris, T.; Verneau, F. Exploring consumers’ perception and willingness to pay for “Non-Added Sulphite” wines through experimental auctions: A case study in Italy and Spain. Wine Econ. Policy 2017, 6, 146–154. [Google Scholar] [CrossRef]
- Li, L.; Sun, B. Grape and wine polymeric polyphenols: Their importance in enology. Crit. Rev. Food Sci. Nutr. 2019, 59, 563–579. [Google Scholar] [CrossRef]
- García-Estévez, I.; Cruz, L.; Oliveira, J.; Mateus, N.; de Freitas, V.; Soares, S. First evidences of interaction between pyranoanthocyanins and salivary proline-rich proteins. Food Chem. 2017, 228, 574–581. [Google Scholar] [CrossRef]
- Soares, S.; Kohl, S.; Thalmann, S.; Mateus, N.; Meyerhof, W.; De Freitas, V. Different phenolic compounds activate distinct human bitter taste receptors. J. Agric. Food Chem. 2013, 61, 1525–1533. [Google Scholar] [CrossRef]
- Hornedo-Ortega, R.; González-Centeno, M.R.; Chira, K.; Jourdes, M.; Teissedre, P.-L. Phenolic Compounds of Grapes and Wines: Key Compounds and Implications in Sensory Perception. In Winemaking-Stabilization, Aging Chemistry and Biochemistry; IntechOpen: London, UK, 2020. [Google Scholar]
- Pinasseau, L.; Vallverdú-Queralt, A.; Verbaere, A.; Roques, M.; Meudec, E.; Le Cunff, L.; Péros, J.-P.; Ageorges, A.; Sommerer, N.; Boulet, J.-C. Cultivar diversity of grape skin polyphenol composition and changes in response to drought investigated by LC-MS based metabolomics. Front. Plant Sci. 2017, 8, 1826. [Google Scholar] [CrossRef]
- Smeriglio, A.; Barreca, D.; Bellocco, E.; Trombetta, D. Proanthocyanidins and hydrolysable tannins: Occurrence, dietary intake and pharmacological effects. Br. J. Pharmacol. 2017, 174, 1244–1262. [Google Scholar] [CrossRef] [PubMed]
- Somers, T. The polymeric nature of wine pigments. Phytochemistry 1971, 10, 2175–2186. [Google Scholar] [CrossRef]
- Hayasaka, Y.; Kennedy, J.A. Mass spectrometric evidence for the formation of pigmented polymers in red wine. Aust. J. Grape Wine Res. 2003, 9, 210–220. [Google Scholar] [CrossRef]
- Es-Safi, N.-E.; Fulcrand, H.; Cheynier, V.; Moutounet, M. Competition between (+)-catechin and (−)-epicatechin in acetaldehyde-induced polymerization of flavanols. J. Agric. Food Chem. 1999, 47, 2088–2095. [Google Scholar] [CrossRef]
- de Freitas, V.; Mateus, N. Formation of pyranoanthocyanins in red wines: A new and diverse class of anthocyanin derivatives. Anal. Bioanal. Chem. 2011, 401, 1463–1473. [Google Scholar] [CrossRef]
- Blouin, J. Le SO2 en Œnologie; Dunod: Paris, France, 2014; ISBN 9782100710492. [Google Scholar]
- Gambuti, A.; Strollo, D.; Erbaggio, A.; Lecce, L.; Moio, L. Effect of winemaking practices on color indexes and selected bioactive phenolics of Aglianico wine. J. Food Sci. 2007, 72, S623–S628. [Google Scholar] [CrossRef]
- Bakker, J.; Bridle, P.; Bellworthy, S.J.; Garcia-Viguera, C.; Reader, H.P.; Watkins, S.J. Effect of sulphur dioxide and must extraction on colour, phenolic composition and sensory quality of red table wine. J. Sci. Food Agric. 1998, 78, 297–307. [Google Scholar] [CrossRef]
- Pelonnier-Magimel, E.; Windholtz, S.; Pomarède, I.M.; Barbe, J.-C. Sensory characterisation of wines without added sulfites via specific and adapted sensory profile. OENO One 2020, 54, 671–685. [Google Scholar] [CrossRef]
- Pelonnier-Magimel, E.; Mangiorou, P.; Darriet, P.; de Revel, G.; Jourdes, M.; Marchal, A.; Marchand, S.; Pons, A.; Riquier, L.; Teissedre, P.-L.; et al. Sensory characterisation of Bordeaux red wines produced without added sulfites. OENO One 2020, 54, 733–743. [Google Scholar] [CrossRef]
- Zeng, L.; Teissèdre, P.; Jourdes, M. Structures of polymeric pigments in red wine and their derived quantification markers revealed by high-resolution quadrupole time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 2016, 30, 81–88. [Google Scholar] [CrossRef]
- Paul, F. Die alkalimetrische Bestimmung der freien, gebundenen und gesamten schwefligen Säure mittels des Apparates von Lieb und Zacherl. Mitt. Klosterneuburg 1958, 8, 21–27. [Google Scholar]
- Commission Internationale de l’Eclairage. Colorimetry, 2nd ed.; Publication CIE no. 15.2 (TC-1.3); Bureau Central de la CIE: Paris, France, 1986.
- Ayala, F.; Echávarri, J.; Negueruela, A. A new simplified method for measuring the color of wines. I. Red and rose wines. Am. J. Enol. Vitic. 1997, 48, 357–363. [Google Scholar] [CrossRef]
- Sudraud, P. Interpretation des courbes d’absorption des vins rouges. Ann. Technol. Agric 1958, 7, 203–208. [Google Scholar]
- Glories, Y. La couleur des vins rouges: 2e. Partie: Mesure, origine et interprétation. Connaiss. Vigne Vin 1984, 18. [Google Scholar] [CrossRef]
- Organisation Internationale de la Vigne et du Vin. Recueil des Méthodes Internationales D’analyse des Vins et des Moûts; OIV: Dijon, France, 1990. [Google Scholar]
- Kennedy, J.A.; Jones, G.P. Analysis of Proanthocyanidin Cleavage Products Following Acid-Catalysis in the Presence of Excess Phloroglucinol. J. Agric. Food Chem. 2001, 49, 1740–1746. [Google Scholar] [CrossRef] [PubMed]
- Drinkine, J.; Lopes, P.; Kennedy, J.A.; Teissedre, P.-L.; Saucier, C. Ethylidene-Bridged Flavan-3-ols in Red Wine and Correlation with Wine Age. J. Agric. Food Chem. 2007, 55, 6292–6299. [Google Scholar] [CrossRef]
- Martínez, J.; Melgosa, M.; Pérez, M.; Hita, E.; Negueruela, A. Note. Visual and instrumental color evaluation in red wines. Food Sci. Technol. Int. 2001, 7, 439–444. [Google Scholar] [CrossRef]
- Pérez-Magariño, S.; González-San José, M.L. Polyphenols and colour variability of red wines made from grapes harvested at different ripeness grade. Food Chem. 2006, 96, 197–208. [Google Scholar] [CrossRef]
- Picinelli, A.; Bakker, J.; Bridle, P. Model wine solutions: Effect of sulphur dioxide on colour and composition during ageing. Vitis 1994, 33, 31–35. [Google Scholar]
- Santos-Buelga, C.; Bravo-Haro, S.; Rivas-Gonzalo, J.C. Interactions between catechin and malvidin-3-monoglucoside in model solutions. Z. Lebensm.-Unters. Forsch. 1995, 201, 269–274. [Google Scholar] [CrossRef]
- Lee, D.F.; Swinny, E.E.; Jones, G.P. NMR identification of ethyl-linked anthocyanin–flavanol pigments formed in model wine ferments. Tetrahedron Lett. 2004, 45, 1671–1674. [Google Scholar] [CrossRef]
- Drinkine, J.; Lopes, P.; Kennedy, J.A.; Teissedre, P.-L.; Saucier, C. Analysis of ethylidene-bridged flavan-3-ols in wine. J. Agric. Food Chem. 2007, 55, 1109–1116. [Google Scholar] [CrossRef]
- Morata, A.; Calderón, F.; González, M.; Gómez-Cordovés, M.; Suárez, J. Formation of the highly stable pyranoanthocyanins (vitisins A and B) in red wines by the addition of pyruvic acid and acetaldehyde. Food Chem. 2007, 100, 1144–1152. [Google Scholar] [CrossRef]
- Avizcuri, J.-M.; Sáenz-Navajas, M.-P.; Echávarri, J.-F.; Ferreira, V.; Fernández-Zurbano, P. Evaluation of the impact of initial red wine composition on changes in color and anthocyanin content during bottle storage. Food Chem. 2016, 213, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Dallas, C.; Ricardo-da-Silva, J.M.; Laureano, O. Products formed in model wine solutions involving anthocyanins, procyanidin B2, and acetaldehyde. J. Agric. Food Chem. 1996, 44, 2402–2407. [Google Scholar] [CrossRef]
- Atanasova, V.; Fulcrand, H.; Le Guernevé, C.; Cheynier, V.; Moutounet, M. Structure of a new dimeric acetaldehyde malvidin 3-glucoside condensation product. Tetrahedron Lett. 2002, 43, 6151–6153. [Google Scholar] [CrossRef]
- Cheynier, V.; Duenas-Paton, M.; Salas, E.; Maury, C.; Souquet, J.-M.; Sarni-Manchado, P.; Fulcrand, H. Structure and properties of wine pigments and tannins. Am. J. Enol. Vitic. 2006, 57, 298–305. [Google Scholar] [CrossRef]
- Sims, C.A.; Morris, J.R. Effects of pH, Sulfur Dioxide, Storage Time, and Temperature on the Color and Stability of Red Muscadine Grape Wine. Am. J. Enol Vitic. 1984, 35, 35–39. [Google Scholar] [CrossRef]
- He, F.; Liang, N.-N.; Mu, L.; Pan, Q.-H.; Wang, J.; Reeves, M.J.; Duan, C.-Q. Anthocyanins and Their Variation in Red Wines II. Anthocyanin Derived Pigments and Their Color Evolution. Molecules 2012, 17, 1483–1519. [Google Scholar] [CrossRef]
- Es-Safi, N.-E.; Fulcrand, H.; Cheynier, V.; Moutounet, M. Studies on the acetaldehyde-induced condensation of (−)-epicatechin and malvidin 3-O-glucoside in a model solution system. J. Agric. Food Chem. 1999, 47, 2096–2102. [Google Scholar] [CrossRef]
- Weber, F.; Winterhalter, P. Synthesis and structure elucidation of ethyliden-linked anthocyanin—Flavan-3-ol oligomers. Food Res. Int. 2014, 65, 69–76. [Google Scholar] [CrossRef]
- Zeng, L. Étude de la Composition Macromoléculaire du Raisin et des Vins: Impact sur la Qualité Sensorielle. Ph.D. Thesis, Université de Bordeaux, Bordeaux, France, 2015. [Google Scholar]

| L* | a* | b* | CI | CD | ΔE*ab | |||
|---|---|---|---|---|---|---|---|---|
| 2017 | Technological maturity | With SO2 | 16.75 | 46.45 | 27.88 | 0.643 | 0.729 | 17.62 |
| Without SO2 | 8.64 | 38.05 | 14.68 | 0.937 | 1.087 | |||
| Advanced maturity | With SO2 | 8.66 | 38.35 | 14.73 | 0.999 | 1.085 | 4.22 | |
| Without SO2 | 7.06 | 35.60 | 11.95 | 1.071 | 1.238 | |||
| 2018 | Technological maturity | With SO2 | 17.51 | 48.39 | 28.88 | 0.715 | 0.817 | 4.81 |
| Without SO2 | 15.50 | 45.65 | 25.48 | 0.713 | 0.823 | |||
| Advanced maturity | With SO2 | 12.46 | 43.30 | 21.22 | 0.887 | 1.023 | 2.44 | |
| Without SO2 | 13.71 | 44.49 | 22.99 | 0.676 | 0.805 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelonnier-Magimel, E.; Chira, K.; Teissèdre, P.-L.; Jourdes, M.; Barbe, J.-C. Color Characterization of Bordeaux Red Wines Produced without Added Sulfites. Foods 2023, 12, 2358. https://doi.org/10.3390/foods12122358
Pelonnier-Magimel E, Chira K, Teissèdre P-L, Jourdes M, Barbe J-C. Color Characterization of Bordeaux Red Wines Produced without Added Sulfites. Foods. 2023; 12(12):2358. https://doi.org/10.3390/foods12122358
Chicago/Turabian StylePelonnier-Magimel, Edouard, Kléopatra Chira, Pierre-Louis Teissèdre, Michaël Jourdes, and Jean-Christophe Barbe. 2023. "Color Characterization of Bordeaux Red Wines Produced without Added Sulfites" Foods 12, no. 12: 2358. https://doi.org/10.3390/foods12122358
APA StylePelonnier-Magimel, E., Chira, K., Teissèdre, P.-L., Jourdes, M., & Barbe, J.-C. (2023). Color Characterization of Bordeaux Red Wines Produced without Added Sulfites. Foods, 12(12), 2358. https://doi.org/10.3390/foods12122358






