Biophysical, Nutraceutical, and Technofunctional Features of Specialty Cereals: Pigmented Popcorn and Sorghum
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biological Materials
2.2. Biophysical Properties
2.3. Proximal Analysis
2.4. Nutraceutical Properties
2.4.1. Quantification of Total Phenolic Compounds
2.4.2. Quantification of Total Anthocyanins
2.5. Rheological Assessment
2.6. Structural Properties (X-ray Diffraction)
2.7. Morphological Assessments
2.8. Design of Experiments and Statistical Analysis
3. Results and Discussion
3.1. Biophysical Properties
3.2. Proximal Analysis
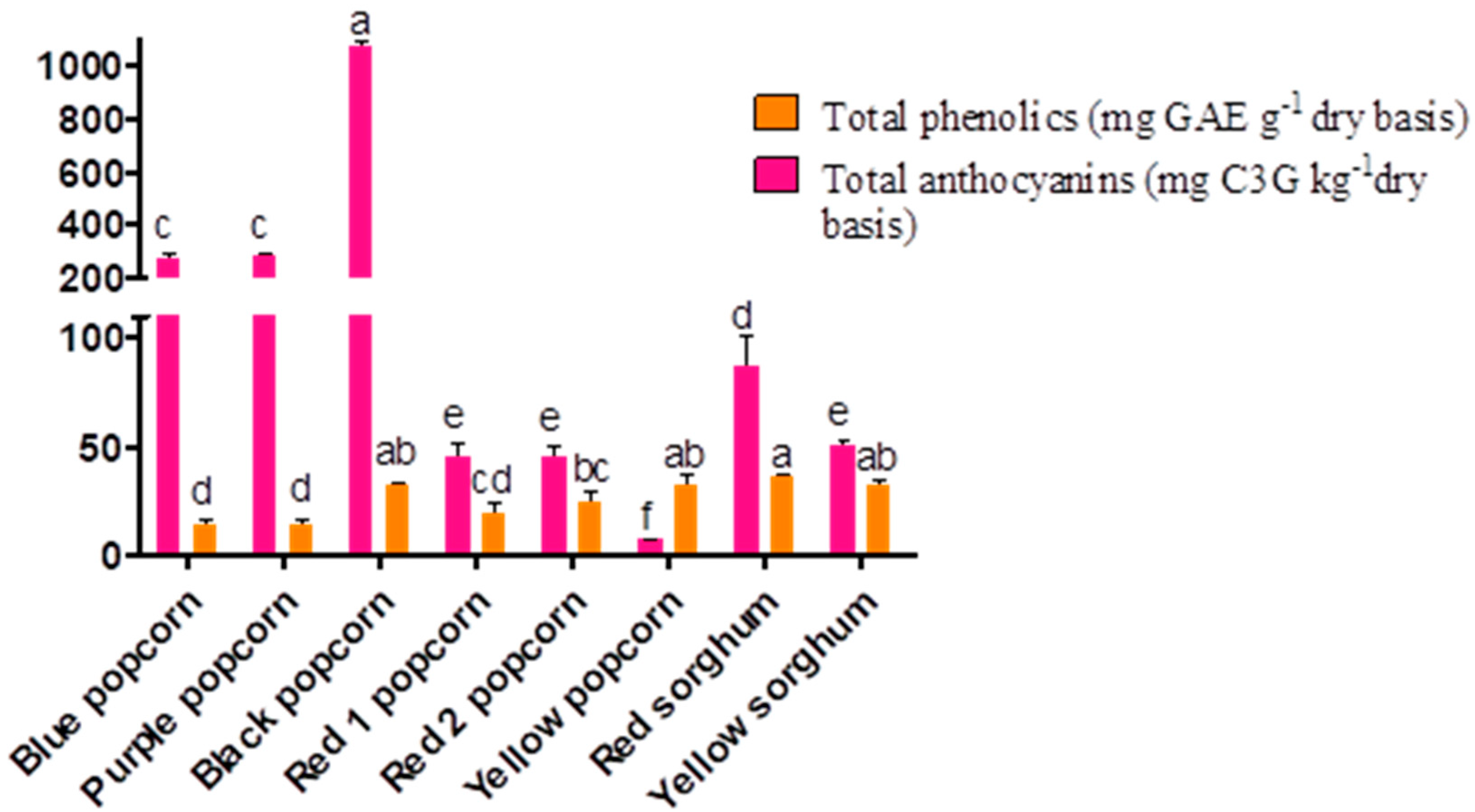
3.3. Nutraceutical Characteristics
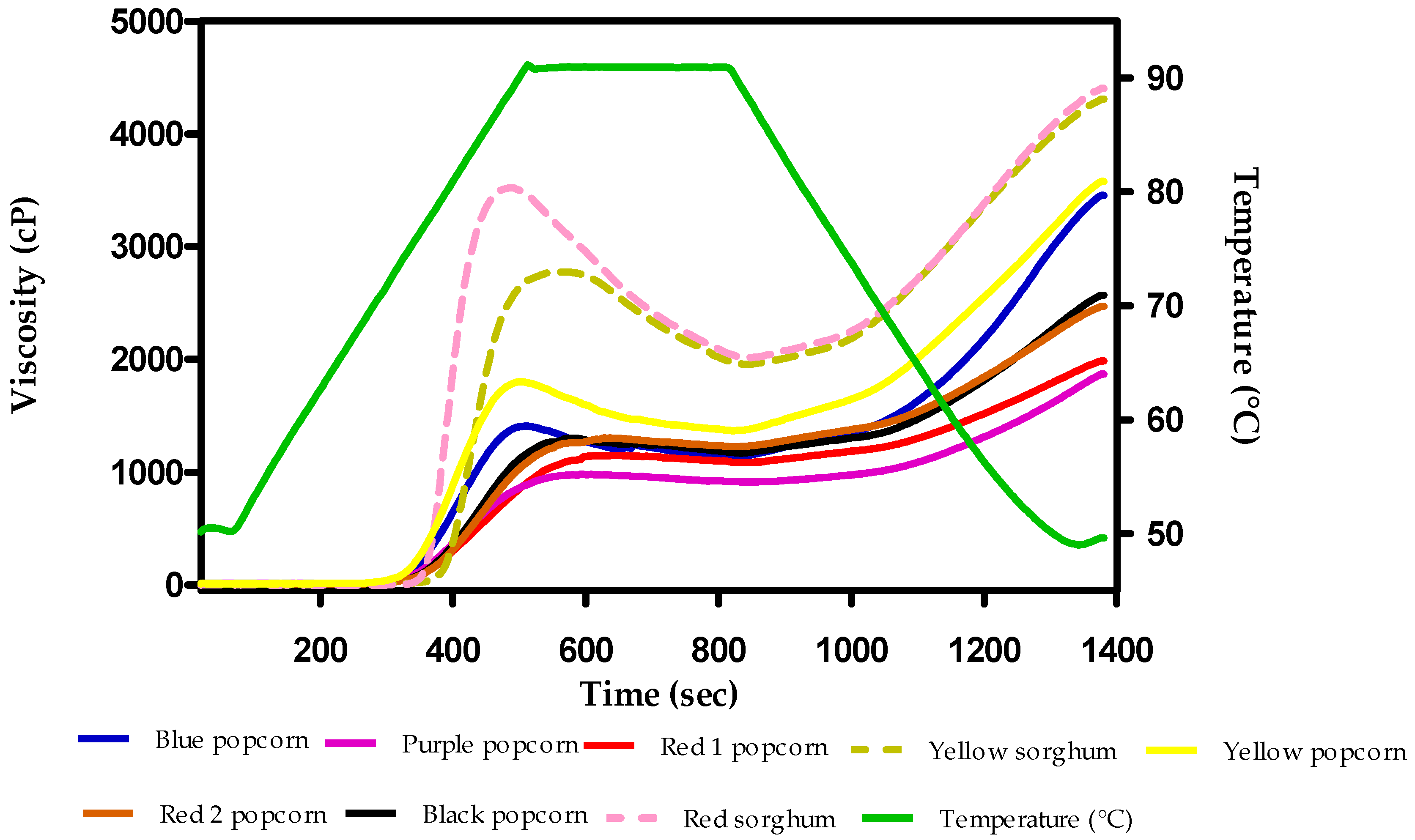
3.4. Rheological Behavior
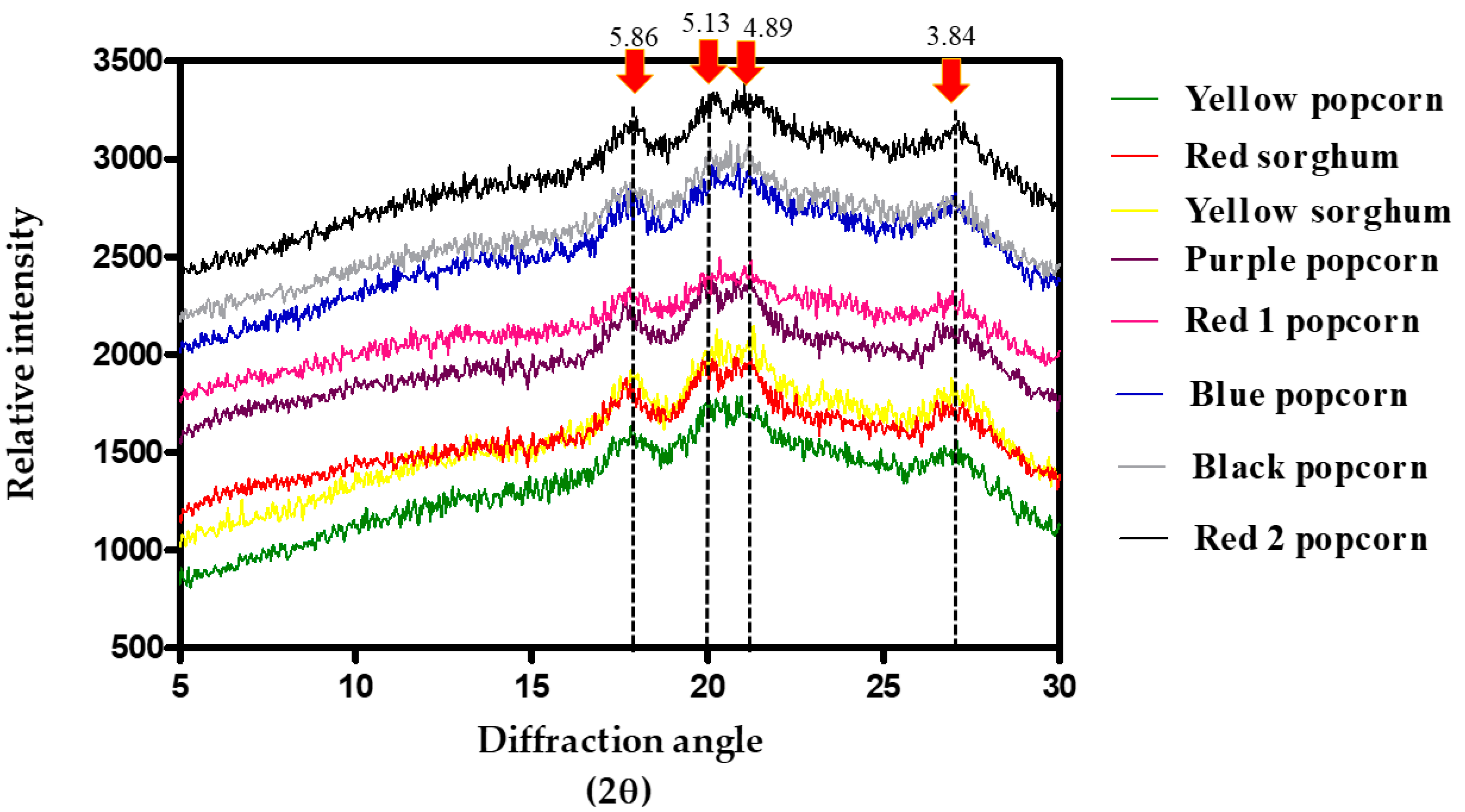
3.5. Structural Properties (X-ray Diffraction)
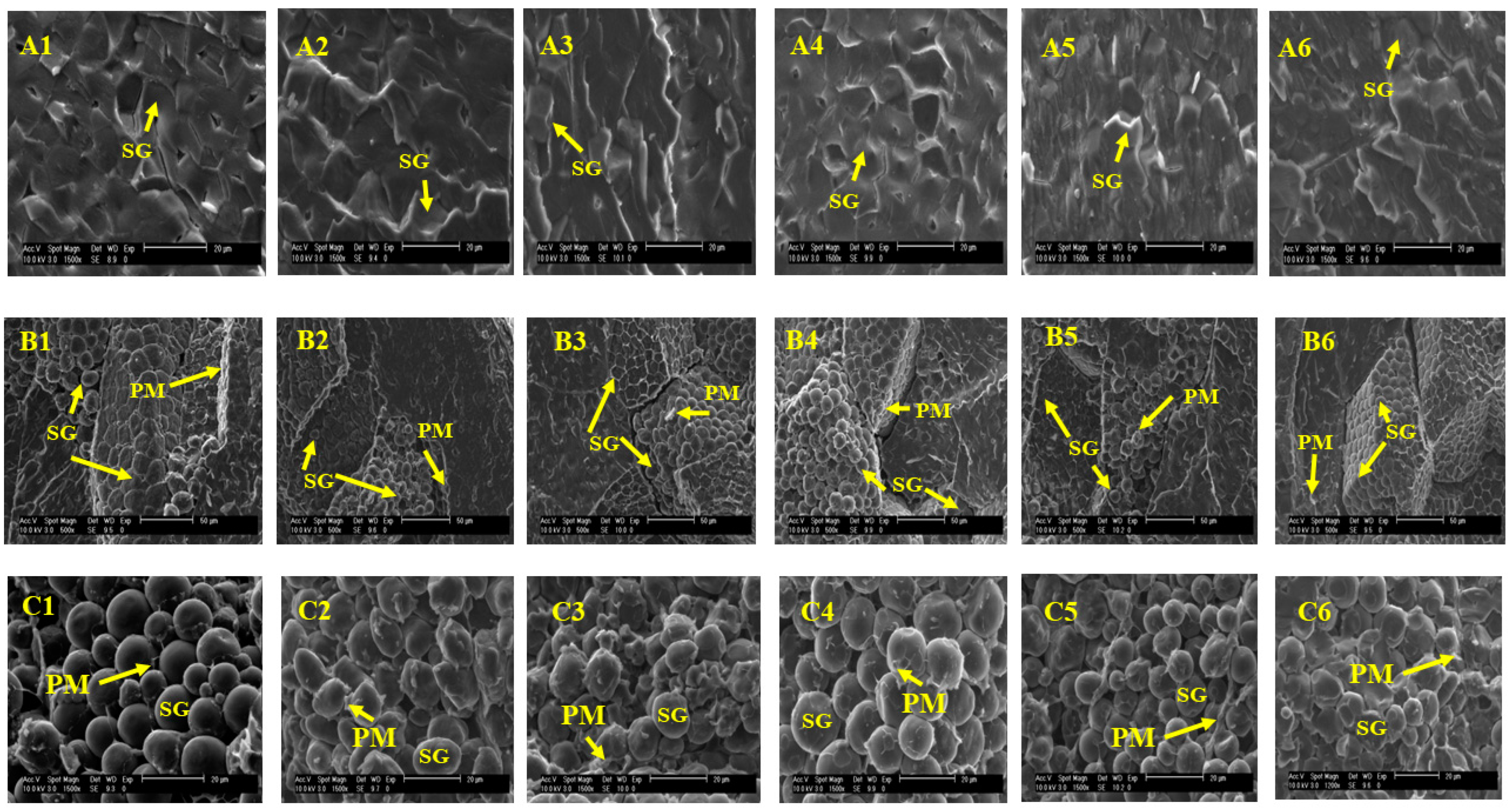
3.6. Morphological Assessments
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Torbica, A.; Belović, M.; Popović, L.; Čakarević, J.; Jovičić, M.; Pavličević, J. Comparative Study of Nutritional and Technological Quality Aspects of Minor Cereals. J. Food Sci. Technol. 2021, 58, 311–322. [Google Scholar] [CrossRef]
- Preciado-Ortiz, R.E.; Ochoa-Centeno, N.J.; Vázquez-Carrillo, M.G.; Santiago-Ramos, D.; Terrón-Ibarra, A.D. Grain Yield, Physical and Pasting Properties, and Anthocyanins of Non-Conventional Pigmented Corn Hybrids for Pozole End-Use Adapted to Subtropical Regions. Appl. Food Res. 2022, 2, 100180. [Google Scholar] [CrossRef]
- Peralta-Veran, L.; Espinosa-Leal, C.; Escalante-Aburto, A.; Preciado-Ortiz, R.E.; Puente-Garza, C.A.; Serna-Saldivar, S.O.; García-Lara, S. Effects of Pozole Broth Production on Phenolic Acids and Antioxidant Activity of Specialty Maize Landraces. J. Cereal Sci. 2022, 107, 103543. [Google Scholar] [CrossRef]
- Dudeja, I.; Gupta, M.; Mankoo, R.K.; Singh, A. Pigmented Maize: Nutritional Properties and Bioactive Profile. In Pigmented Cereals and Millets; Bangar, S.P., Maqsood, S., Siroha, A.K., Eds.; The Royal Society of Chemistry: London, UK, 2023; Volume 38, pp. 50–55. [Google Scholar]
- Espitia-Hernández, P.; Chávez González, M.L.; Ascacio-Valdés, J.A.; Dávila-Medina, D.; Flores-Naveda, A.; Silva, T.; Ruelas Chacón, X.; Sepúlveda, L. Sorghum (Sorghum bicolor L.) as a Potential Source of Bioactive Substances and Their Biological Properties. Crit. Rev. Food Sci. Nutr. 2020, 62, 2269–2280. [Google Scholar] [CrossRef] [PubMed]
- Ben Mrid, R.; Bouargalne, Y.; El Omari, R.; Nhiri, M. New Insights into the Therapeutic Effects of Phenolic Acids from Sorghum Seeds. J. Rep. Pharma Sci. 2019, 8, 91–101. [Google Scholar]
- Khalid, W.; Ali, A.; Arshad, M.S.; Afzal, F.; Akram, R.; Siddeeg, A.; Kousar, S.; Rahim, M.A.; Aziz, A.; Maqbool, Z.; et al. Nutrients and Bioactive Compounds of Sorghum bicolor L. Used to Prepare Functional Foods: A Review on the Efficacy against Different Chronic Disorders. Int. J. Food Prop. 2022, 25, 1045–1062. [Google Scholar] [CrossRef]
- Riaz, M.N. Snack Foods, Processing. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Sergio, O. Serna-Saldivar Popcorn and Other Puffed Grains. In Snack Foods, 1st ed.; CRC Press: Boca Raton, FL, USA, 2022; p. 20. [Google Scholar]
- Mohammed, R.; Are, A.K.; Bhavanasi, R.; Munghate, R.S.; Kavi Kishor, P.B.; Sharma, H.C. Quantitative Genetic Analysis of Agronomic and Morphological Traits in Sorghum, Sorghum bicolor. Front. Plant Sci. 2015, 6, 945. [Google Scholar] [CrossRef] [Green Version]
- Serna-Saldivar, S.O. Cereal Grains: Properties, Processing and Nutritional Attributes; CRC Press: Boca Ratón, FL, USA, 2010. [Google Scholar]
- American Organization of Analytical Chemists International (AOAC). Official Methods 962.09, 978.02, 925.10, 923.03. Determination of Crude Fiber, Total Nitrogen, Moisture, and Ash; AOAC: Washington, DC, USA, 2010. [Google Scholar]
- AACC. International AACC Approved Methods of Analysis, 11th Edition—AACC Method 30-20.01. Crude Fat in Grain and Stock Feeds. Available online: https://www.cerealsgrains.org/resources/Methods/Pages/30CrudeFat.aspx (accessed on 3 June 2023).
- FAO. Appendix XVI: Methods of Feed Analysis. Available online: https://www.fao.org/3/s4314e/s4314e0v.htm (accessed on 16 May 2023).
- Ainsworth, E.A.; Gillespie, K.M. Estimation of Total Phenolic Content and Other Oxidation Substrates in Plant Tissues Using Folin-Ciocalteu Reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef]
- Escalante-Aburto, A.; Ramírez-Wong, B.; Torres-Chávez, P.I.; López-Cervantes, J.; De Figueroa-Cárdenas, J.D.; Barrón-Hoyos, J.M.; Morales-Rosas, I.; García, N.P.; Gutiérrez-Dorado, R. Obtaining Ready-to-Eat Blue Corn Expanded Snacks with Anthocyanins Using an Extrusion Process and Response Surface Methodology. Molecules 2014, 19, 21066–21084. [Google Scholar] [CrossRef] [Green Version]
- Escalante-Aburto, A.; Ponce-García, N.; Ramírez-Wong, B.; Santiago-Ramos, D.; Véles-Medina, J.J.; de Dios Figueroa Cárdenas, J. Effect of Extrusion Factors and Particle Size on Starch Properties of Nixtamalized Whole Blue Corn Snacks. Starch/Staerke 2016, 68, 1111–1120. [Google Scholar] [CrossRef]
- Tian, Y.; Buriak, P.; Eckhoff, S.R. Effect of Hybrid and Physical Properties of Individual Popcorn Kernels on Expansion Volume. Cereal Chem. 2001, 78, 578–582. [Google Scholar] [CrossRef]
- Surpam, T.; Pardeshi, I.; Rokade, H. Engineering Properties of Sorghum. Int. J. Chem. Stud. 2019, 7, 108–110. [Google Scholar]
- Erkmen, O.; Bozoglu, T.F. (Eds.) Spoilage of Cereals and Cereal Products. In Food Microbiology: Principles into Practice; John Wiley & Sons: Hoboken, NJ, USA, 2016; pp. 364–375. [Google Scholar] [CrossRef]
- Farahnaky, A.; Alipour, M.; Majzoobi, M. Popping Properties of Corn Grains of Two Different Varieties at Different Moistures. J. Agric. Sci. Technol. 2013, 15, 771–780. [Google Scholar]
- Mawouma, S.; Condurache, N.N.; Turturică, M.; Constantin, O.E.; Croitoru, C.; Rapeanu, G. Chemical Composition and Antioxidant Profile of Sorghum (Sorghum bicolor (L.) Moench) and Pearl Millet (Pennisetum glaucum (L.) R.Br.) Grains Cultivated in the Far-North Region of Cameroon. Foods 2022, 11, 2026. [Google Scholar] [CrossRef] [PubMed]
- Palavecino, P.M.; Penci, M.C.; Calderón-Domínguez, G.; Ribotta, P.D. Chemical Composition and Physical Properties of Sorghum Flour Prepared from Different Sorghum Hybrids Grown in Argentina. Starch Stärke 2016, 68, 1055–1064. [Google Scholar] [CrossRef] [Green Version]
- Hernández-Becerra, E.; Gutierrez-Oñate, M.P.; Martinez-Soto, G.; Vega-Rojas, L.J.; Acosta-Osorio, A.A.; Contreras-Padilla, M.; Rodríguez-García, M.E. Physicochemical Characterization of Corn–Sorghum Nixtamalized Flours as a Function of the Steeping Time. J. Food Meas. Charact. 2016, 10, 434–443. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.; Zhao, Y. Phenolic Compounds in Whole Grain Sorghum and Their Health Benefits. Foods 2021, 10, 1921. [Google Scholar] [CrossRef]
- Shavandi, M.; Javanmard, M.; Basiri, A. Novel Popping through Infrared: Effect on Some Physicochemical Properties of Popcorn (Zea mays L. Var. Everta). LWT 2022, 155, 112955. [Google Scholar] [CrossRef]
- Coco, M.G.; Vinson, J.A. Analysis of Popcorn (Zea mays L. Var. Everta) for Antioxidant Capacity and Total Phenolic Content. Antioxidants 2019, 8, 22. [Google Scholar] [CrossRef] [Green Version]
- Bhukya, R.; Chamarthy, R.V.; Ediga, S.; Reddy, A.C.; Babu Marriboina, S.; Tonapi, V.A. Variability of Polyphenols, Antioxidant Activity and UFLC Phenolic Acid Profiles of Different Sorghum Genotypes. Sch. Int. J. Biochem. 2020, 3, 104–113. [Google Scholar] [CrossRef]
- Rao, S.; Santhakumar, A.B.; Chinkwo, K.A.; Wu, G.; Johnson, S.K.; Blanchard, C.L. Characterization of Phenolic Compounds and Antioxidant Activity in Sorghum Grains. J. Cereal Sci. 2018, 84, 103–111. [Google Scholar] [CrossRef]
- Rumler, R.; Bender, D.; Speranza, S.; Frauenlob, J.; Gamper, L.; Hoek, J.; Jäger, H.; Schönlechner, R. Chemical and Physical Characterization of Sorghum Milling Fractions and Sorghum Whole Meal Flours Obtained via Stone or Roller Milling. Foods 2021, 10, 870. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Bean, S.; Seib, P.A.; Pedersen, J.; Shi, Y.C. Structure and Functional Properties of Sorghum Starches Differing in Amylose Content. J. Agric. Food Chem. 2008, 56, 6680–6685. [Google Scholar] [CrossRef]
- Balet, S.; Guelpa, A.; Fox, G.; Manley, M. Rapid Visco Analyser (RVA) as a Tool for Measuring Starch-Related Physiochemical Properties in Cereals: A Review. Food Anal. Methods 2019, 12, 2344–2360. [Google Scholar] [CrossRef]
- Paraginski, R.T.; Ziegler, V.; Peter, M.; Rockenbach, B.A.; Vanier, N.L.; Elias, M.C. Physicochemical and Sensory Properties of Popcorn Grain Stored at Different Temperatures. Curr. Agric. Sci. Technol. 2016, 22, 14–23. [Google Scholar] [CrossRef] [Green Version]
- Truong, H.H.; Khoddami, A.; Moss, A.F.; Liu, S.Y.; Selle, P.H. The Potential of Rapid Visco-Analysis Starch Pasting Profiles to Gauge the Quality of Sorghum as a Feed Grain for Chicken-Meat Production. Anim. Nutr. 2017, 3, 11–18. [Google Scholar] [CrossRef]
- Sandhu, K.S.; Singh, N.; Kaur, M. Characteristics of the Different Corn Types and Their Grain Fractions: Physicochemical, Thermal, Morphological, and Rheological Properties of Starches. J. Food Eng. 2004, 64, 119–127. [Google Scholar] [CrossRef]
- Dome, K.; Podgorbunskikh, E.; Bychkov, A.; Lomovsky, O. Changes in the Crystallinity Degree of Starch Having Different Types of Crystal Structure after Mechanical Pretreatment. Polymers 2020, 12, 641. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.J.; Na, J.H.; Park, J.Y.; Chang, P.S. Structural Characteristics and In Vitro Digestibility of Malic Acid-Treated Corn Starch with Different PH Conditions. Molecules 2019, 24, 1900. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, F.; Ali, T.M.; Mustafa, G.; Hasnain, A. Structural, Functional and Digestibility Characteristics of Sorghum and Corn Starch Extrudates (RS3) as Affected by Cold Storage Time. Int. J. Biol. Macromol. 2020, 164, 3048–3054. [Google Scholar] [CrossRef]
- Torovo Marues de Souza Cesar, A.L.; Gomes Cheim, L.M.; Afonso Rossignoli, P.; Rodrigues, L.J.; FOnseca da Silva, F.; Pereira Takeuchi, K.; de Morais Carvalho, D.; de Melo Faria, A.M. Physical Chemicals and Rheologicals Characterization of Corn Starch (Zea mays L.) Landraces Popcorn. Res. Soc. Dev. 2021, 10, e402101321394. [Google Scholar] [CrossRef]
- Ziegler, V.; Timm, N.d.S.; Ferreira, C.D.; Goebel, J.T.; Pohndorf, R.S.; de Oliveira, M. Effects of Drying Temperature of Red Popcorn Grains on the Morphology, Technological, and Digestibility Properties of Starch. Int. J. Biol. Macromol. 2020, 145, 568–574. [Google Scholar] [CrossRef] [PubMed]
- Freire, A.I.; De, E.; Castro, M.; Pereira, A.M.; Ranielly, R.; Cruz, P.; Machado De Souza, F.B.; Filipe, W.; Chagas, T.; Cândido De Souza, J. Amylose Content and Micromorphology of Popcorn Progenies with Different Popping Expansion Volumes. Ciência Rural 2020, 50, e20180962. [Google Scholar] [CrossRef] [Green Version]
- Cui, L.; Dong, S.; Zhang, J.; Liu, P. Starch Granule Size Distribution and Morphogenesis in Maize (Zea mays L.) Grains with Different Endosperm Types. AJCS 2014, 8, 1560–1565. [Google Scholar]
- Bean, S.R.; Wilson, J.D.; Moreau, R.A.; Galant, A.; Awika, J.M.; Kaufman, R.C.; Adrianos, S.L.; Ioerger, B.P. Structure and Composition of the Sorghum Grain. Sorghum State Art Future Perspetives 2019, 58, 173–214. [Google Scholar] [CrossRef]

| Sample | 1000 Grains Weight (g) | Length (mm) | Width (mm) | Thickness (mm) |
|---|---|---|---|---|
| Blue | 163.757 ±1.43 a | 8.66 ± 0.41 ab | 5.46 ± 0.36 b | 4.34 ± 0.46 a |
| Purple | 136.16 ± 2.32 c | 8.79 ± 0.61 a | 5.41 ± 0.57 b | 4.21 ± 0.43 ab |
| Black | 95.77 ± 1.82 e | 8.07 ± 0.61 c | 4.86 ± 0.42 d | 3.96 ± 0.53 bc |
| Red 1 | 124.33± 3.14 d | 8.40 ± 0.58 bc | 5.20 ± 0.44 bc | 3.65 ± 0.36 c |
| Red 2 | 123.84 ± 0.71 d | 8.25 ± 0.40 c | 5.00 ± 0.41 cd | 3.67 ± 0.37 c |
| Yellow | 158.16 ± 1.06 b | 8.71 ± 0.49 ab | 5.89 ± 0.49 a | 3.95 ± 0.44 bc |
| Sample | 1000 Grains Weight (g) | Length (mm) | Width (mm) | Thickness (mm) |
|---|---|---|---|---|
| Red | 25.18 ± 0.31 a | 4.12 ± 0.37 a | 3.53 ± 0.22 a | 2.55 ± 0.21 a |
| Yellow | 25.52 ± 0.21 a | 4.11 ± 0.22 a | 3.40 ± 0.24 b | 2.53 ± 0.21 a |
| Sample | Moisture | Protein | Crude fat | Ash | Crude Fiber | NFE |
|---|---|---|---|---|---|---|
| % (Dry Basis) | ||||||
| Blue | 12.69 ± 0.30 abc | 11.08 ± 0.28 a | 3.00 ± 0.59 bc | 1.60 ± 0.26 a | 4.51 ± 0.21 ab | 67.08 ± 0.79 bc |
| Purple | 13.06 ± 0.12 ab | 11.25 ± 0.15 a | 3.79 ± 0.12 ab | 1.46 ± 0.23 a | 3.98 ± 0.55 abc | 66.44 ± 0.15 bc |
| Black | 11.67 ± 0.21 cd | 11.91 ± 0.34 a | 2.96 ± 0.65 bc | 1.53 ± 0.23 a | 3.88 ± 0.24 abc | 68.02 ± 1.17 b |
| Red 1 | 13.76 ± 0.31 a | 13.12 ± 0.33 a | 3.79 ± 0.13 ab | 1.83 ± 0.25 a | 2.16 ± 0.11 e | 65.29 ± 0.71 bc |
| Red 2 | 13.16 ± 0.92 ab | 12.94 ± 0.20 a | 4.11 ± 0.21 a | 1.66 ± 0.30 a | 3.48 ± 0.35 cd | 64.68 ± 1.23 c |
| Yellow | 12.55 ± 0.01 bc | 11.15 ± 2.24 a | 3.87 ± 0.48 ab | 1.73 ± 0.05 a | 3.59 ± 0.23 bc | 67.08 ± 1.83 bc |
| Sample | Moisture | Protein | Crude Fat | Ash | Crude Fiber | NFE |
|---|---|---|---|---|---|---|
| % (Dry Basis) | ||||||
| Red | 11.11 ± 0.38 a | 7.93 ± 0.24 b | 2.57 ± 0.11 b | 1.53 ± 0.05 a | 2.61 ± 0.48 b | 74.21 ± 0.85 a |
| Yellow | 11.22 ±0.03 a | 10.95 ± 0.11 a | 3.34 ± 0.22 a | 1.53 ± 0.05 a | 4.80 ± 0.22 a | 68.12 ± 0.19 b |
| Sample | Peak Viscosity | Breakdown | Holding Strength | Setback Region | Total Setback | Final Viscosity |
|---|---|---|---|---|---|---|
| cP | ||||||
| Blue | 1445 ± 7 b | 184 ± 6 b | 1261 ± 13 b | 2929 ± 87 a | 3113 ± 93 a | 4374 ± 82 a |
| Purple | 965 ± 12 f | 55 ± 12 c | 909 ± 6 e | 969 ± 15 e | 1024 ± 9 e | 1933 ± 4 d |
| Black | 1261 ± 10 d | 58 ± 8 c | 1203 ± 3 c | 2287 ± 96 b | 2345 ± 91 b | 3548 ± 88 b |
| Red 1 | 1135 ± 8 e | 47 ± 8 c | 1088 ± 1 d | 867 ± 16 e | 914 ± 92 e | 2002 ± 9 d |
| Red 2 | 1304 ± 1 c | 68 ± 7 c | 1236 ± 6 b | 1551 ± 15 d | 1618 ± 14 d | 2855 ± 14 c |
| Yellow | 1834 ± 12 a | 431 ± 31 a | 1404 ± 23 a | 1738 ± 54 c | 2168 ± 22 c | 3572 ± 45 b |
| Sample | Peak Viscosity | Breakdown | Holding Strength | Setback Region | Total Setback | Final Viscosity |
|---|---|---|---|---|---|---|
| cP | ||||||
| Red sorghum | 3564 ± 12 a | 1502 ± 3 a | 2061 ± 12 a | 833 ± 25 b | 2335 ± 23 a | 4397 ± 30 a |
| Yellow sorghum | 2750 ± 9 b | 778 ± 11 b | 1972 ± 4 b | 1551 ± 6 a | 2329 ± 50 a | 4301 ± 4 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robles-Plata, V.T.; Serna Saldivar, S.; de Dios Figueroa-Cárdenas, J.; Rooney, W.L.; Dávila-Vega, J.P.; Chuck-Hernández, C.; Escalante-Aburto, A. Biophysical, Nutraceutical, and Technofunctional Features of Specialty Cereals: Pigmented Popcorn and Sorghum. Foods 2023, 12, 2301. https://doi.org/10.3390/foods12122301
Robles-Plata VT, Serna Saldivar S, de Dios Figueroa-Cárdenas J, Rooney WL, Dávila-Vega JP, Chuck-Hernández C, Escalante-Aburto A. Biophysical, Nutraceutical, and Technofunctional Features of Specialty Cereals: Pigmented Popcorn and Sorghum. Foods. 2023; 12(12):2301. https://doi.org/10.3390/foods12122301
Chicago/Turabian StyleRobles-Plata, Valery Tixian, Sergio Serna Saldivar, Juan de Dios Figueroa-Cárdenas, William L. Rooney, Juan Pablo Dávila-Vega, Cristina Chuck-Hernández, and Anayansi Escalante-Aburto. 2023. "Biophysical, Nutraceutical, and Technofunctional Features of Specialty Cereals: Pigmented Popcorn and Sorghum" Foods 12, no. 12: 2301. https://doi.org/10.3390/foods12122301
APA StyleRobles-Plata, V. T., Serna Saldivar, S., de Dios Figueroa-Cárdenas, J., Rooney, W. L., Dávila-Vega, J. P., Chuck-Hernández, C., & Escalante-Aburto, A. (2023). Biophysical, Nutraceutical, and Technofunctional Features of Specialty Cereals: Pigmented Popcorn and Sorghum. Foods, 12(12), 2301. https://doi.org/10.3390/foods12122301








