Insoluble Dietary Fiber from Soybean Residue (Okara) Exerts Anti-Obesity Effects by Promoting Hepatic Mitochondrial Fatty Acid Oxidation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Animal Experimental Design
2.3. Biochemical Analysis
2.4. Analysis of Enzyme Contents Related to Fatty Acid Oxidation
2.5. Histological Investigation
2.6. Analysis of Hepatic Fatty Acid Composition and Content
2.7. Western Blot Analysis
2.8. Statistical Analysis
3. Results
3.1. Effects of HPSIDF on Body Weight, Food Intake and Fat Accumulation in HFD-Fed Mice
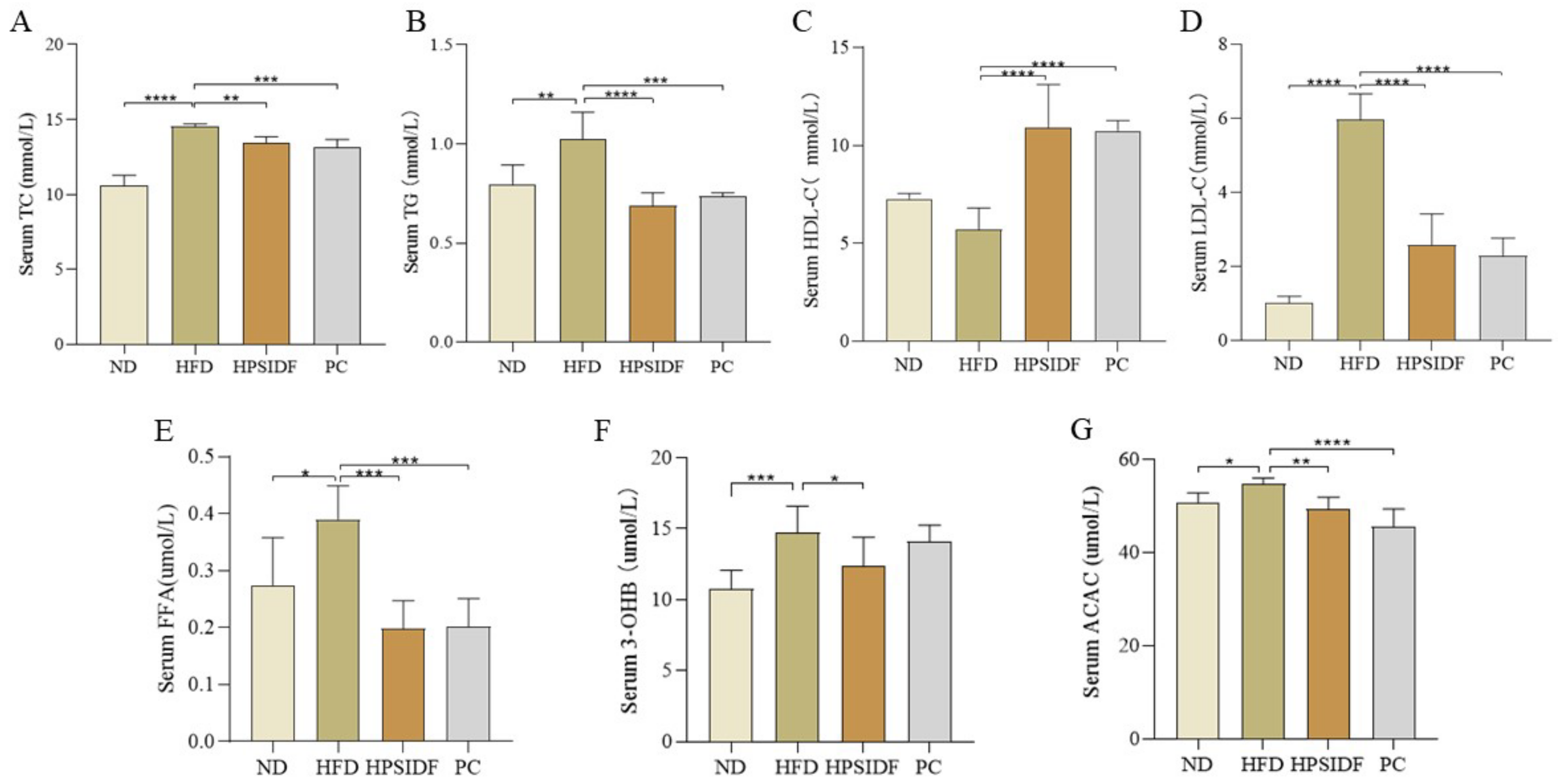
3.2. Effects of HPSIDF on Serum Biochemical Indicators in HFD-Fed Mice
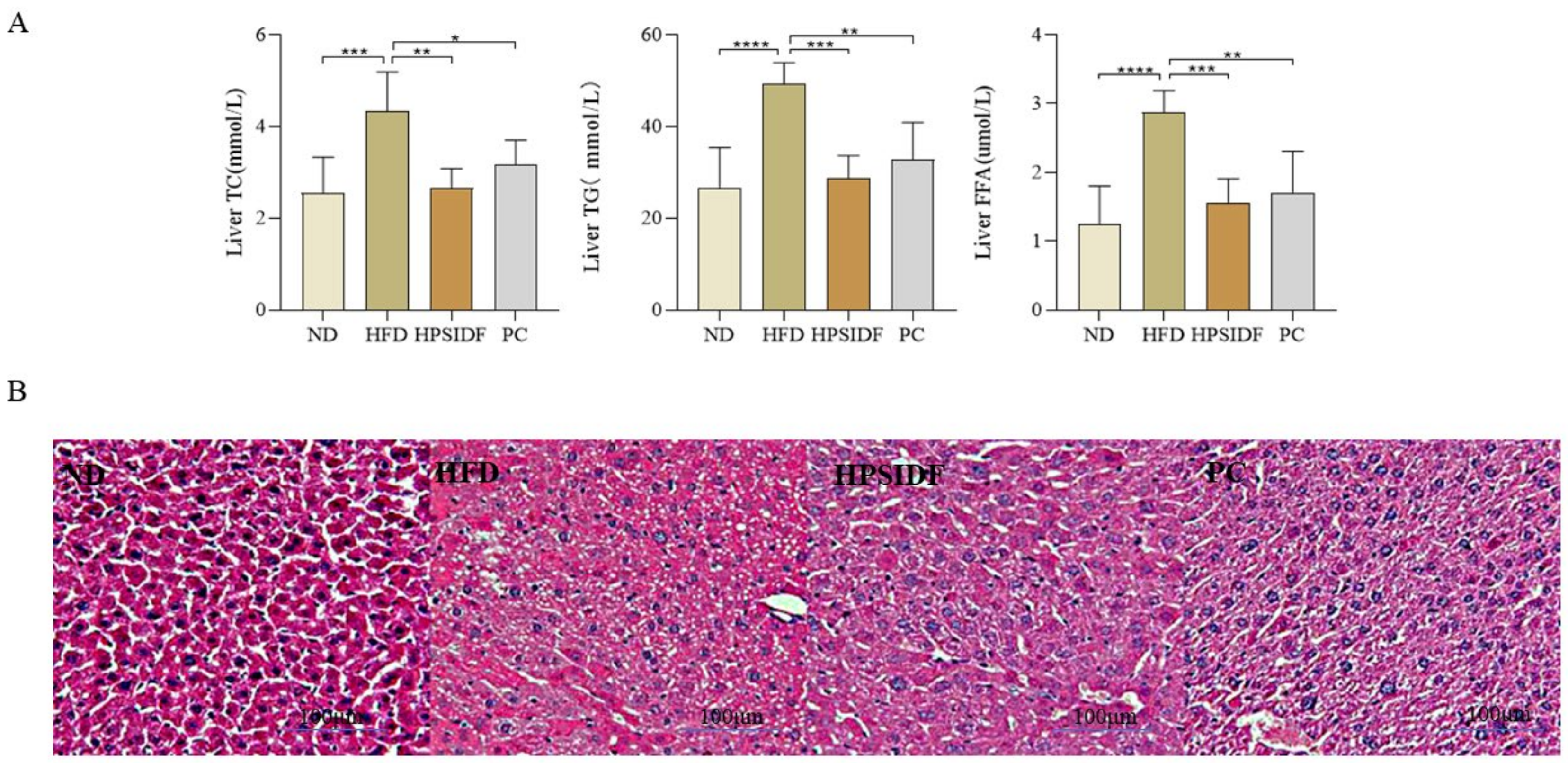
3.3. Effects of HPSIDF on Hepatic Steatosis in HFD-Fed Mice
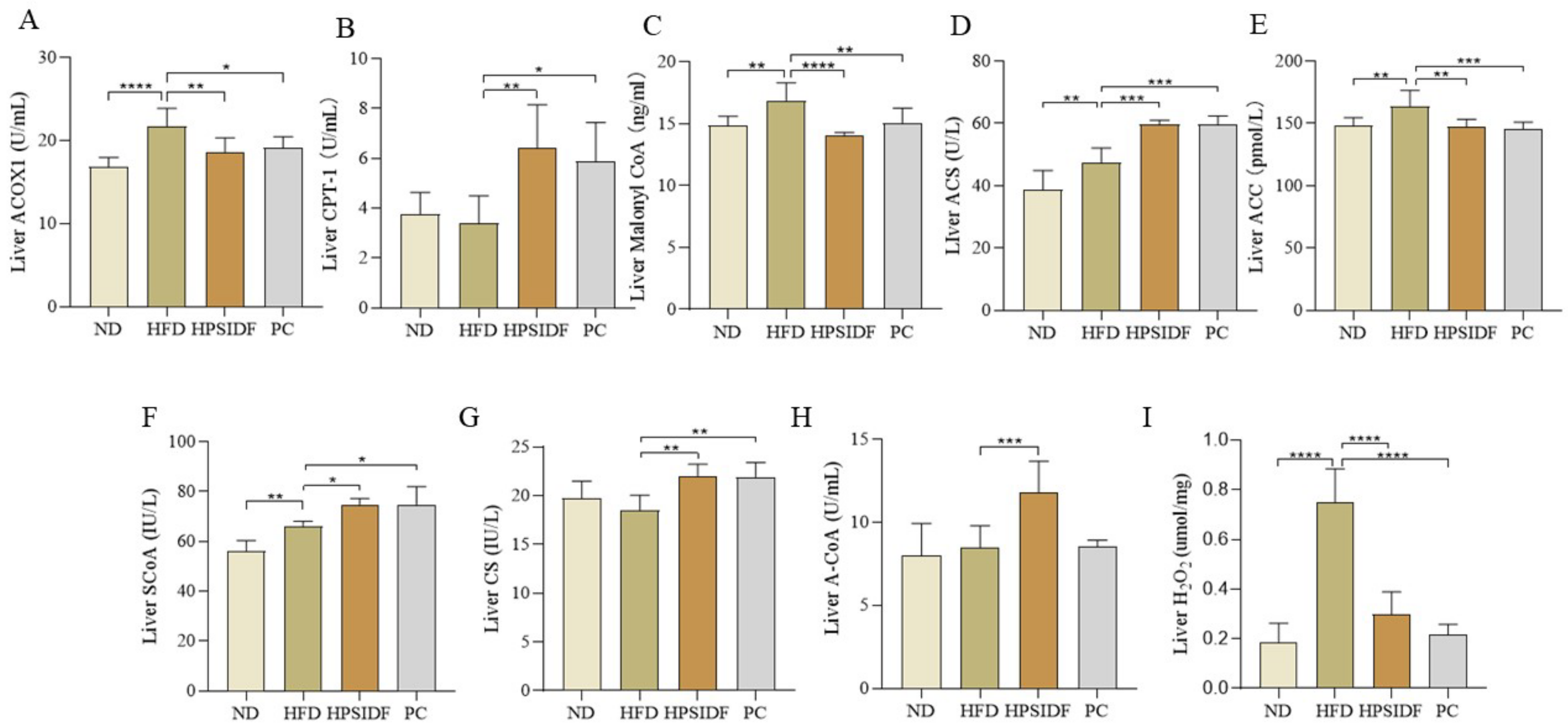
3.4. Effects of HPSIDF on the Activity of Enzymes Related to Hepatic Fatty Acid Oxidation and the Production of Intermediate and Final Products in HFD-Fed Mice
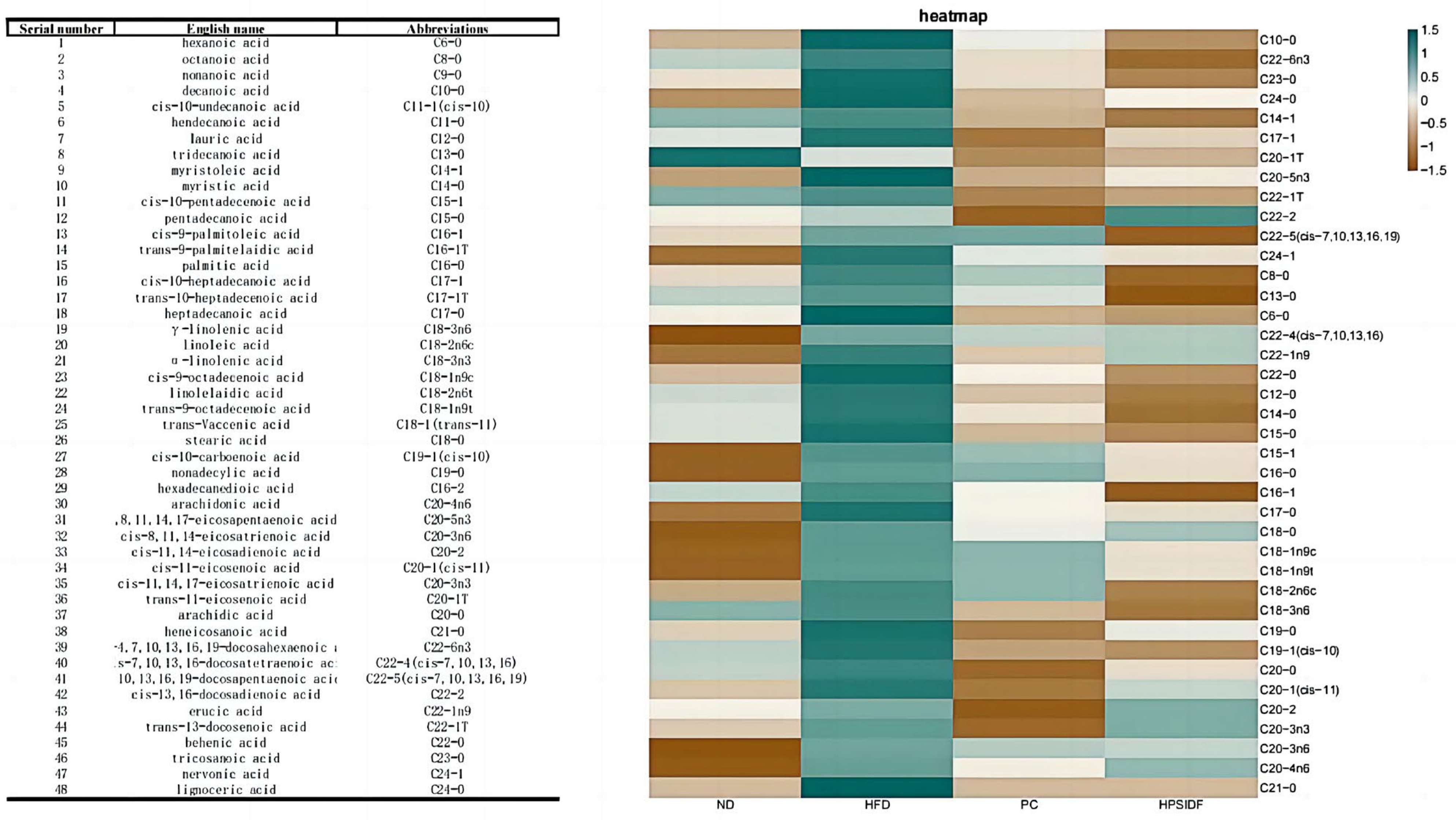
3.5. Effects of HPSIDF on Hepatic Fatty Acid Content and Composition in HFD-Fed Mice
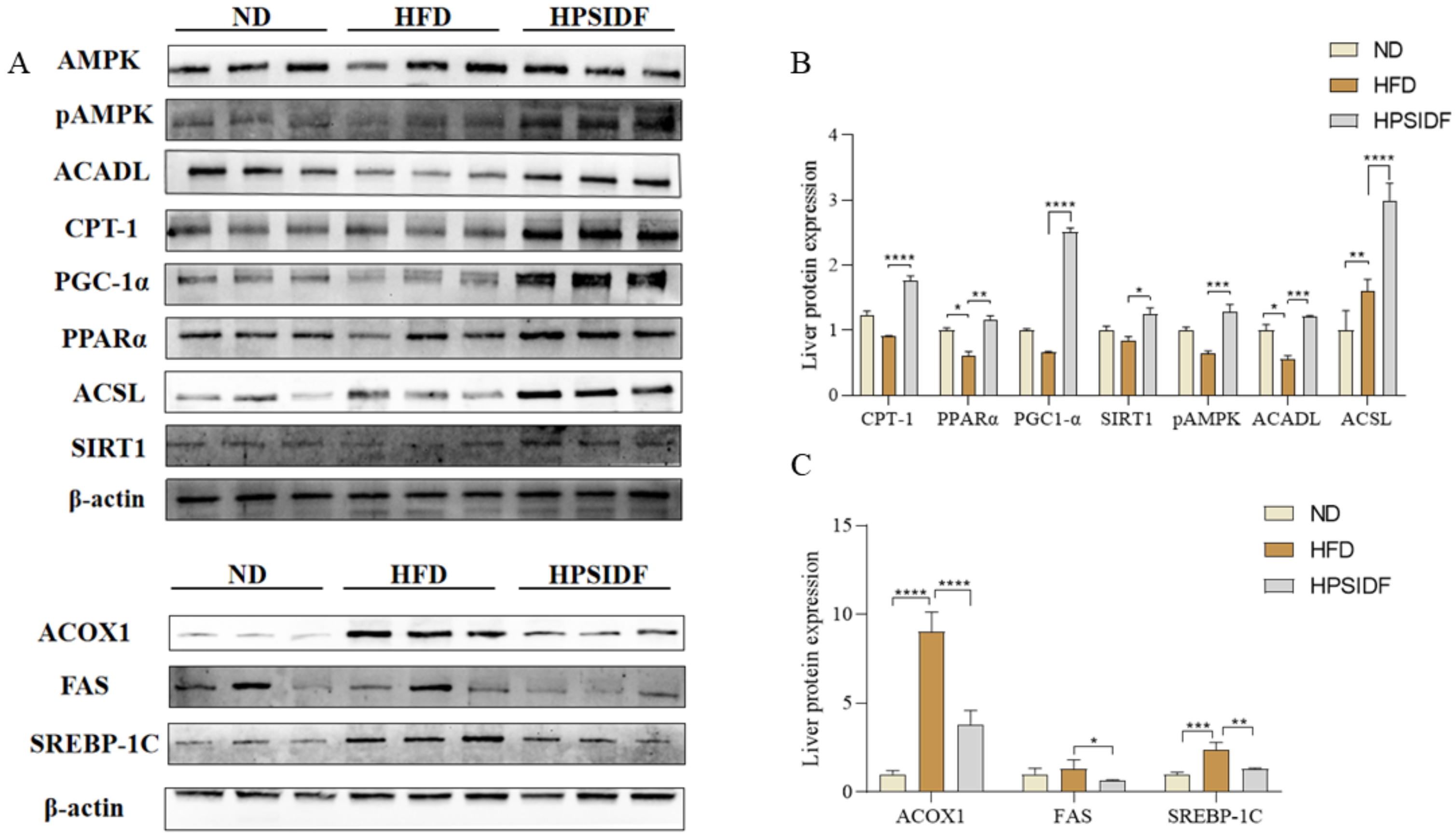
3.6. Effects of HPSIDF on the Expression Levels of Hepatic Fatty Acid β-Oxidation-Associated Proteins in HFD-Fed Mice
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Mattson, M.P. Roles of the Lipid Peroxidation Product 4-Hydroxynonenal in Obesity, the Metabolic Syndrome, and Associated Vascular and Neurodegenerative Disorders. Exp. Gerontol. 2009, 44, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; McPherson, K.; Marsh, T.; Gortmaker, S.L.; Brown, M. Obesity 2 Health and Economic Burden of the Projected Obesity Trends in the USA and the UK. Lancet 2011, 378, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Gauvreau, D.; Villeneuve, N.; Deshaies, Y.; Cianflone, K. Novel adipokines: Links between obesity and atherosclerosis. Ann. Endocrinol. 2011, 72, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Korenblat, K.M.; Fabbrini, E.; Mohammed, B.S.; Klein, S. Liver, Muscle, and Adipose Tissue Insulin Action Is Directly Related to Intrahepatic Triglyceride Content in Obese Subjects. Gastroenterology 2008, 134, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Kotronen, A.; Yki-Jarvinen, H.; Sevastianova, K.; Bergholm, R.; Hakkarainen, A.; Pietilainen, K.H.; Juurinen, L.; Lundbom, N.; Sorensen, T.I.A. Comparison of the Relative Contributions of Intra-Abdominal and Liver Fat to Components of the Metabolic Syndrome. Obesity 2011, 19, 23–28. [Google Scholar] [CrossRef]
- Harwood, J. Fatty-Acid Metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1988, 39, 101–138. [Google Scholar] [CrossRef]
- Habinowski, S.A.; Witters, L.A. The Effects of AICAR on Adipocyte Differentiation of 3T3-L1 Cells. Biochem. Biophys. Res. Commun. 2001, 286, 852–856. [Google Scholar] [CrossRef]
- Lage, R.; Dieguez, C.; Vidal-Puig, A.; Lopez, M. AMPK: A Metabolic Gauge Regulating Whole-Body Energy Homeostasis. Trends Mol. Med. 2008, 14, 539–549. [Google Scholar] [CrossRef]
- Lim, C.T.; Kola, B.; Korbonits, M. AMPK as a Mediator of Hormonal Signalling. J. Mol. Endocrinol. 2010, 44, 87–97. [Google Scholar] [CrossRef]
- Peng, I.-C.; Chen, Z.; Sun, W.; Li, Y.-S.; Marin, T.L.; Hsu, P.-H.; Su, M.-I.; Cui, X.; Pan, S.; Lytle, C.Y.; et al. Glucagon Regulates ACC Activity in Adipocytes through the CAMKK Beta/AMPK Pathway. Am. J. Physiol.-Endocrinol. Metab. 2012, 302, E1560–E1568. [Google Scholar] [CrossRef]
- Ducharme, N.A.; Bickel, P.E. Minireview: Lipid Droplets in Lipogenesis and Lipolysis. Endocrinology 2008, 149, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Szkudelski, T.; Szkudelska, K. Effects of AMPK Activation on Lipolysis in Primary Rat Adipocytes: Studies at Different Glucose Concentrations. Arch. Physiol. Biochem. 2017, 123, 43–49. [Google Scholar] [CrossRef]
- Elgebaly, A.; Radwan, I.A.I.; AboElnas, M.M.; Ibrahim, H.H.; Eltoomy, M.F.M.; Atta, A.A.; Mesalam, H.A.; Sayed, A.A.; Othman, A.A. Resveratrol Supplementation in Patients with Non-Alcoholic Fatty Liver Disease: Systematic Review and Meta-Analysis. J. Gastrointestin. Liver Dis. 2017, 26, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Isken, F.; Klaus, S.; Osterhoff, M.; Pfeiffer, A.F.H.; Weickert, M.O. Effects of Long-Term Soluble vs. Insoluble Dietary Fiber Intake on High-Fat Diet-Induced Obesity in C57BL/6J Mice. J. Nutr. Biochem. 2010, 21, 278–284. [Google Scholar] [CrossRef]
- Han, X.; Yang, D.; Zhang, S.; Liu, X.; Zhao, Y.; Song, C.; Sun, Q. Characterization of Insoluble Dietary Fiber from Pleurotus Eryngii and Evaluation of Its Effects on Obesity-Preventing or Relieving Effects via Modulation of Gut Microbiota. J. Future Foods 2023, 3, 55–66. [Google Scholar] [CrossRef]
- Wang, S.; Sun, W.; Swallah, M.S.; Amin, K.; Lyu, B.; Fan, H.; Zhang, Z.; Yu, H. Preparation and Characterization of Soybean Insoluble Dietary Fiber and Its Prebiotic Effect on Dyslipidemia and Hepatic Steatosis in High Fat-Fed C57BL/6J Mice. Food Funct. 2021, 12, 8760–8773. [Google Scholar] [CrossRef] [PubMed]
- Agostoni, C.; Bresson, J.-L.; Fairweather-Tait, S.; Flynn, A.; Golly, I.; Korhonen, H.; Lagiou, P.; Lovik, M.; Marchelli, R.; Martin, A.; et al. Scientific Opinion on Dietary Reference Values for Carbohydrates and Dietary Fibre. Efsa J. 2010, 8, 1462. [Google Scholar] [CrossRef]
- Esmail, M.; Anwar, S.; Kandeil, M.; El-Zanaty, A.M.; Abdel-Gabbar, M. Effect of Nigella Sativa, Atorvastatin, or L-Carnitine on High Fat Diet-Induced Obesity in Adult Male Albino Rats. Biomed. Pharmacother. 2021, 141, 111818. [Google Scholar] [CrossRef]
- Alhasaniah, A.H. L-Carnitine: Nutrition, Pathology, and Health Benefits. Saudi J. Biol. Sci. 2023, 30, 103555. [Google Scholar] [CrossRef]
- Nishida, C.; Uauy, R.; Kumanyika, S.; Shetty, P. The Joint WHO/FAO Expert Consultation on Diet, Nutrition and the Prevention of Chronic Diseases: Process, Product and Policy Implications. Public Health Nutr. 2004, 7, 245–250. [Google Scholar] [CrossRef]
- Ye, S.; Shah, B.R.; Li, J.; Liang, H.; Zhan, F.; Geng, F.; Li, B. A Critical Review on Interplay between Dietary Fibers and Gut Microbiota. Trends Food Sci. Technol. 2022, 124, 237–249. [Google Scholar] [CrossRef]
- Zhao, G.; Zhang, R.; Dong, L.; Huang, F.; Tang, X.; Wei, Z.; Zhang, M. Particle Size of Insoluble Dietary Fiber from Rice Bran Affects Its Phenolic Profile, Bioaccessibility and Functional Properties. Lwt-Food Sci. Technol. 2018, 87, 450–456. [Google Scholar] [CrossRef]
- Kapravelou, G.; Martinez, R.; Andrade, A.M.; Sanchez, C.; Lopez Chaves, C.; Lopez-Jurado, M.; Aranda, P.; Cantarero, S.; Arrebola, F.; Fernandez-Segura, E.; et al. Health Promoting Effects of Lupin (Lupinus albus Var. Multolupa) Protein Hydrolyzate and Insoluble Fiber in a Diet-Induced Animal Experimental Model of Hypercholesterolemia. Food Res. Int. 2013, 54, 1471–1481. [Google Scholar] [CrossRef]
- Ren, L.; Sun, D.; Zhou, X.; Yang, Y.; Huang, X.; Li, Y.; Wang, C.; Li, Y. Chronic Treatment with the Modified Longdan Xiegan Tang Attenuates Olanzapine-Induced Fatty Liver in Rats by Regulating Hepatic de Novo Lipogenesis and Fatty Acid Beta-Oxidation-Associated Gene Expression Mediated by SREBP-1c, PPAR-Alpha and AMPK-Alpha. J. Ethnopharmacol. 2019, 232, 176–187. [Google Scholar] [CrossRef]
- Song, Y.-B.; An, Y.R.; Kim, S.J.; Park, H.-W.; Jung, J.-W.; Kyung, J.-S.; Hwang, S.Y.; Kim, Y.-S. Lipid Metabolic Effect of Korean Red Ginseng Extract in Mice Fed on a High-Fat Diet. J. Sci. Food Agric. 2012, 92, 388–396. [Google Scholar] [CrossRef]
- Smith, U.; Kahn, B.B. Adipose Tissue Regulates Insulin Sensitivity: Role of Adipogenesis, de Novo Lipogenesis and Novel Lipids. J. Intern. Med. 2016, 280, 465–475. [Google Scholar] [CrossRef]
- Smith, B.K.; Marcinko, K.; Desjardins, E.M.; Lally, J.S.; Ford, R.J.; Steinberg, G.R. Treatment of Nonalcoholic Fatty Liver Disease: Role of AMPK. Am. J. Physiol.-Endocrinol. Metab. 2016, 311, E730–E740. [Google Scholar] [CrossRef]
- Huang, X.; Zhou, Y.; Sun, Y.; Wang, Q. Intestinal Fatty Acid Binding Protein: A Rising Therapeutic Target in Lipid Metabolism. Prog. Lipid Res. 2022, 87, 101178. [Google Scholar] [CrossRef]
- Kim, J.J.P.; Battaile, K.P. Burning Fat: The Structural Basis of Fatty Acid Beta-Oxidation. Curr. Opin. Struct. Biol. 2002, 12, 721–728. [Google Scholar] [CrossRef]
- Reddy, J.K.; Rao, M.S. Lipid Metabolism and Liver Inflammation. II. Fatty Liver Disease and Fatty Acid Oxidation. Am. J. Physiol.-Gastrointest. Liver Physiol. 2006, 290, G852–G858. [Google Scholar] [CrossRef]
- Wanders, R.J.A.; Ferdinandusse, S.; Brites, P.; Kemp, S. Peroxisomes, Lipid Metabolism and Lipotoxicity. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2010, 1801, 272–280. [Google Scholar] [CrossRef]
- Adeva-Andany, M.M.; Carneiro-Freire, N.; Seco-Filgueira, M.; Fernandez-Fernandez, C.; Mourino-Bayolo, D. Mitochondrial Beta-Oxidation of Saturated Fatty Acids in Humans. Mitochondrion 2019, 46, 73–90. [Google Scholar] [CrossRef]
- Osmundsen, H.; Bremer, J.; Pedersen, J. Metabolic Aspects of Peroxisomal Beta-Oxidation. Biochim. Biophys. Acta 1991, 1085, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Wang, Y.; Zhang, X.; Li, P.; Shang, L.; Chen, X.; Zeng, J. Targeting Peroxisomal Fatty Acid Oxidation Improves Hepatic Steatosis and Insulin Resistance in Obese Mice. J. Biol. Chem. 2023, 299, 102845. [Google Scholar] [CrossRef]
- Mannaerts, G.; Van veldhoven, P.; Van broekhoven, A.; Vandebroek, G.; Debeer, L. Evidence That Peroxisomal Acyl-Coa Synthetase Is Located at the Cytoplasmic Side of the Peroxisomal Membrane. Biochem. J. 1982, 204, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shang, L.; Deng, S.; Li, P.; Chen, K.; Gao, T.; Zhang, X.; Chen, Z.; Zeng, J. Peroxisomal Oxidation of Erucic Acid Suppresses Mitochondrial Fatty Acid Oxidation by Stimulating Malonyl-CoA Formation in the Rat Liver. J. Biol. Chem. 2020, 295, 10168–10179. [Google Scholar] [CrossRef] [PubMed]
- Burri, L.; Thoresen, G.H.; Berge, R.K. The Role of PPAR Alpha Activation in Liver and Muscle. PPAR Res. 2010, 2010, 542359. [Google Scholar] [CrossRef] [PubMed]
- Glosli, H.; Gudbrandsen, O.A.; Mullen, A.J.; Halvorsen, B.; Rost, T.H.; Wergedahl, H.; Prydz, H.; Aukrust, P.; Berge, R.K. Down-Regulated Expression of PPAR Alpha Target Genes, Reduced Fatty Acid Oxidation and Altered Fatty Acid Composition in the Liver of Mice Transgenic for HTNF Alpha. Biochim. Biophys. Acta-Mol. Cell Biol. Lipids 2005, 1734, 235–246. [Google Scholar] [CrossRef]
- Evans, R.M.; Barish, G.D.; Wang, Y.X. PPARs and the Complex Journey to Obesity. Nat. Med. 2004, 10, 355–361. [Google Scholar] [CrossRef]
- Day, E.A.; Ford, R.J.; Steinberg, G.R. AMPK as a Therapeutic Target for Treating Metabolic Diseases. Trends Endocrinol. Metab. 2017, 28, 545–560. [Google Scholar] [CrossRef]
- Singh, A.B.; Kan, C.F.K.; Dong, B.; Liu, J. SREBP2 Activation Induces Hepatic Long-Chain Acyl-CoA Synthetase 1 (ACSL1) Expression in Vivo and in Vitro through a Sterol Regulatory Element (SRE) Motif of the ACSL1 C-Promoter. J. Biol. Chem. 2016, 291, 5373–5384. [Google Scholar] [CrossRef]
- Gao, J.; Gu, X.; Zhang, M.; Zu, X.; Shen, F.; Hou, X.; Hao, E.; Bai, G. Ferulic Acid Targets ACSL1 to Ameliorate Lipid Metabolic Disorders in Db/Db Mice. J. Funct. Foods 2022, 91, 105009. [Google Scholar] [CrossRef]
- Cohen, H.Y.; Miller, C.; Bitterman, K.J.; Wall, N.R.; Hekking, B.; Kessler, B.; Howitz, K.T.; Gorospe, M.; de Cabo, R.; Sinclair, D.A. Calorie Restriction Promotes Mammalian Cell Survival by Inducing the SIRT1 Deacetylase. Science 2004, 305, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, C.; Li, N.; Hao, T.; Han, T.; Hill, D.E.; Vidal, M.; Lin, J.D. Genome-Wide Coactivation Analysis of PGC-1 Alpha Identifies BAF60a as a Regulator of Hepatic Lipid Metabolism. Cell Metab. 2008, 8, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, J.T.; Lerin, C.; Haas, W.; Gygi, S.P.; Spiegelman, B.M.; Puigserver, P. Nutrient Control of Glucose Homeostasis through a Complex of PGC-1 Alpha and SIRT1. Nature 2005, 434, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Purushotham, A.; Schug, T.T.; Xu, Q.; Surapureddi, S.; Guo, X.; Li, X. Hepatocyte-Specific Deletion of SIRT1 Alters Fatty Acid Metabolism and Results in Hepatic Steatosis and Inflammation. Cell Metab. 2009, 9, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Abelson, P.; Kennedy, D. The Obesity Epidemic. Science 2004, 304, 1413. [Google Scholar] [CrossRef]
- Stein, C.J.; Colditz, G.A. The Epidemic of Obesity. J. Clin. Endocrinol. Metab. 2004, 89, 2522–2525. [Google Scholar] [CrossRef] [PubMed]
- Maes, H.H.M.; Neale, M.C.; Eaves, L.J. Genetic and Environmental Factors in Relative Body Weight and Human Adiposity. Behav. Genet. 1997, 27, 325–351. [Google Scholar] [CrossRef]
- Brownlee, I.A. The Physiological Roles of Dietary Fibre. Food Hydrocoll. 2011, 25, 238–250. [Google Scholar] [CrossRef]
- Li, L.; Pan, M.; Pan, S.; Li, W.; Zhong, Y.; Hu, J.; Nie, S. Effects of Insoluble and Soluble Fibers Isolated from Barley on Blood Glucose, Serum Lipids, Liver Function and Caecal Short-Chain Fatty Acids in Type 2 Diabetic and Normal Rats. Food Chem. Toxicol. 2020, 135, 110937. [Google Scholar] [CrossRef] [PubMed]
- Gadde, K.M.; Martin, C.K.; Berthoud, H.-R.; Heymsfield, S.B. Obesity Pathophysiology and Management. J. Am. Coll. Cardiol. 2018, 71, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Muniesa, P.; Martinez-Gonzalez, M.-A.; Hu, F.B.; Despres, J.-P.; Matsuzawa, Y.; Loos, R.J.F.; Moreno, L.A.; Bray, G.; Alfredo Martinez, J. Obesity. Nat. Rev. Dis. Primer 2017, 3, 17034. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, T.I.A. From Fat Cells through an Obesity Theory. Eur. J. Clin. Nutr. 2018, 72, 1329–1335. [Google Scholar] [CrossRef] [PubMed]
- Weickert, M.O.; Mohlig, M.; Koebnick, C.; Holst, J.J.; Namsolleck, P.; Ristow, M.; Osterhoff, M.; Rochlitz, H.; Rudovich, N.; Spranger, J.; et al. Impact of Cereal Fibre on Glucose-Regulating Factors. Diabetologia 2005, 48, 2343–2353. [Google Scholar] [CrossRef]

| Items | ND | HFD | HPSIDF | PC |
|---|---|---|---|---|
| Initial body weight (g) | 20.02 ± 0.93 | 20.80 ± 1.19 | 21.10 ± 1.04 | 21.45 ± 0.63 |
| Final body weight (g) | 27.49 ± 2.09 b | 31.78 ± 2.77 a | 26.90 ± 2.79 b | 28.96 ± 2.24 b |
| Body weight gain (g) | 7.47 ± 1.40 b | 10.98 ± 2.94 a | 5.99 ± 2.40 c | 7.50 ± 2.57 b |
| Food intake (g) | 2.13 ± 0.10 b | 2.47 ± 0.04 a | 2.08 ± 0.07 b | 2.15 ± 0.13 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Wang, S.; Wang, J.; Liu, W.; Gong, H.; Zhang, Z.; Lyu, B.; Yu, H. Insoluble Dietary Fiber from Soybean Residue (Okara) Exerts Anti-Obesity Effects by Promoting Hepatic Mitochondrial Fatty Acid Oxidation. Foods 2023, 12, 2081. https://doi.org/10.3390/foods12102081
Zhang J, Wang S, Wang J, Liu W, Gong H, Zhang Z, Lyu B, Yu H. Insoluble Dietary Fiber from Soybean Residue (Okara) Exerts Anti-Obesity Effects by Promoting Hepatic Mitochondrial Fatty Acid Oxidation. Foods. 2023; 12(10):2081. https://doi.org/10.3390/foods12102081
Chicago/Turabian StyleZhang, Jiarui, Sainan Wang, Junyao Wang, Wenhao Liu, Hao Gong, Zhao Zhang, Bo Lyu, and Hansong Yu. 2023. "Insoluble Dietary Fiber from Soybean Residue (Okara) Exerts Anti-Obesity Effects by Promoting Hepatic Mitochondrial Fatty Acid Oxidation" Foods 12, no. 10: 2081. https://doi.org/10.3390/foods12102081
APA StyleZhang, J., Wang, S., Wang, J., Liu, W., Gong, H., Zhang, Z., Lyu, B., & Yu, H. (2023). Insoluble Dietary Fiber from Soybean Residue (Okara) Exerts Anti-Obesity Effects by Promoting Hepatic Mitochondrial Fatty Acid Oxidation. Foods, 12(10), 2081. https://doi.org/10.3390/foods12102081





