Development of Desiccation-Tolerant Probiotic Biofilms Inhibitory for Growth of Foodborne Pathogens on Stainless Steel Surfaces
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of LAB
2.2. Screening of LAB Isolates for Antimicrobial Activity
2.3. Measurement of Cell Surface Hydrophobicity and Auto-Aggregation Ability of LAB
2.4. Biofilm Assay Using Crystal Violet Staining
2.5. Biofilm Formation Analysis by Surface Type and Incubation Temperature and Duration
2.6. Analysis of Antimicrobial Activity of LAB Biofilms against Pathogens on SSCs
2.7. Analysis of Desiccation Tolerance of LAB Biofilms
2.8. Statistical Analysis
3. Results
3.1. LAB Isolates Identified to Have Antibacterial Activity
3.2. Surface Hydrophobicity, Auto-Aggregation, and Biofilm Formation of LAB Isolates
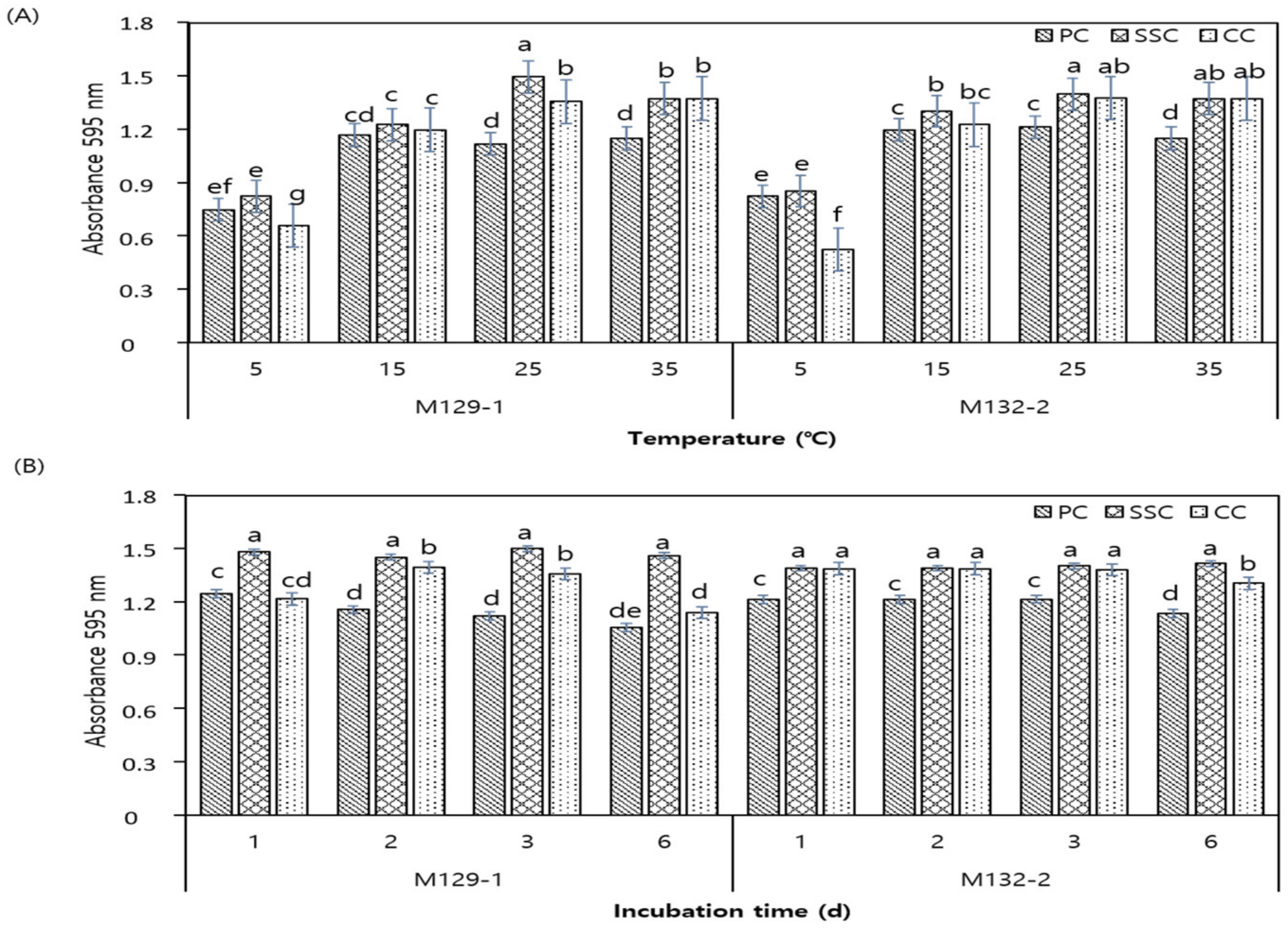
3.3. Biofilm Formation by LAB Isolates on Different Surfaces and Under Different Incubation Temperatures and Durations
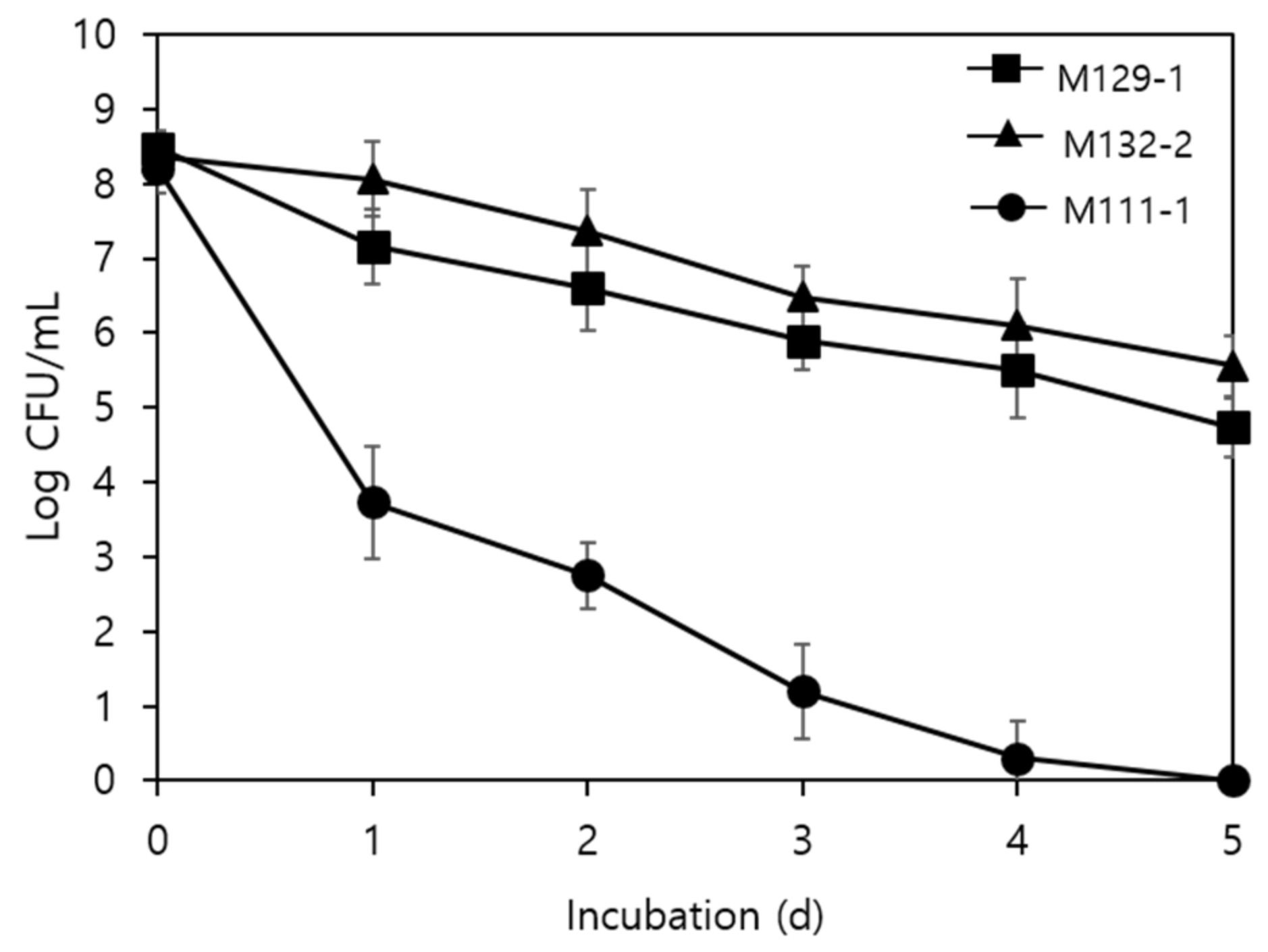
3.4. Survival of Antagonistic LAB Biofilms under Desiccated Conditions
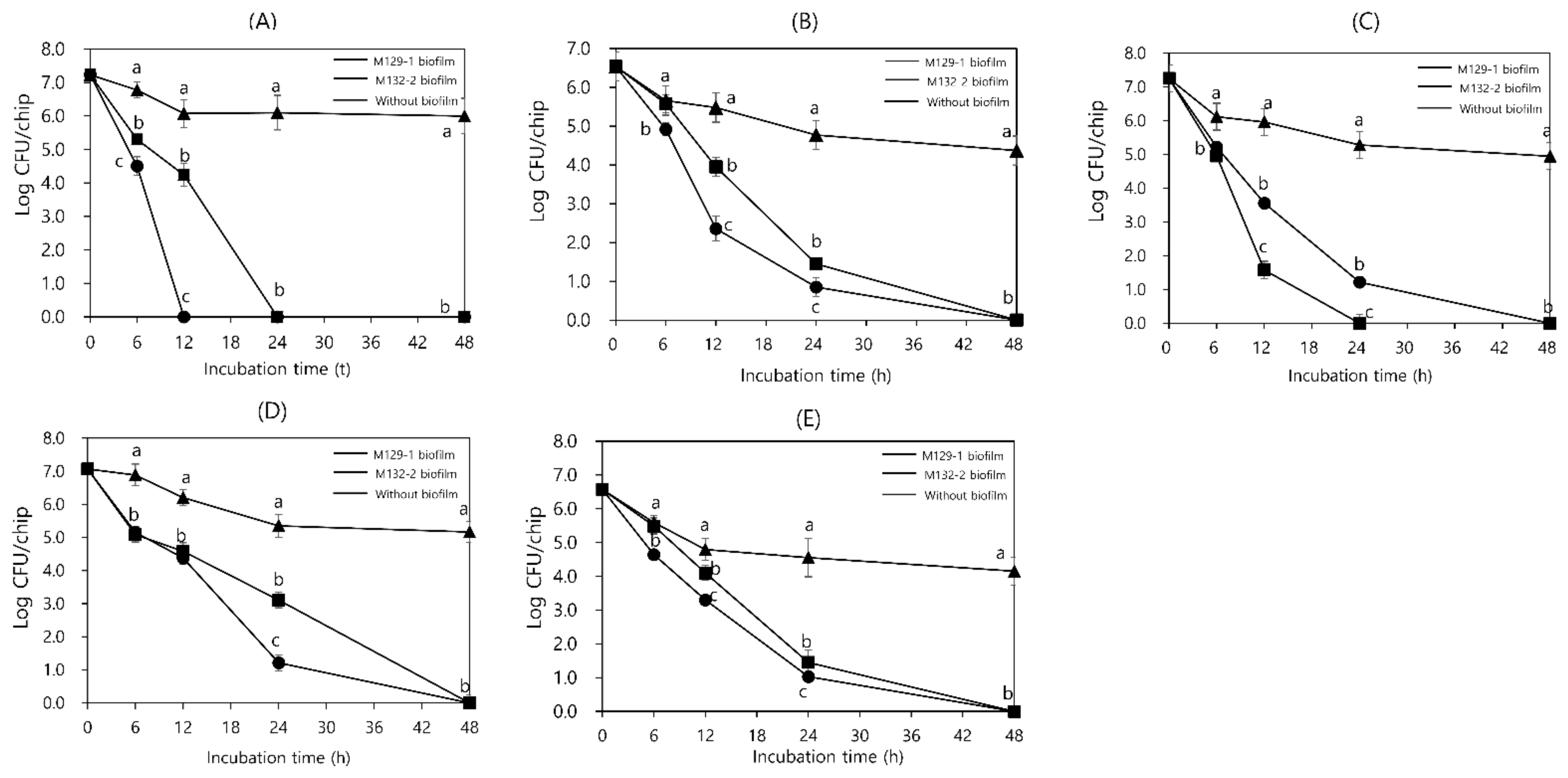
3.5. Antimicrobial Activity of LAB Biofilms on SSCs
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gizaw, Z. Public health risks related to food safety issues in the food market: A systematic literature review. Environ. Health Prev. Med. 2019, 24, 68. [Google Scholar] [CrossRef] [PubMed]
- Srey, S.; Jahid, I.K.; Ha, S.D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Food Safety; World Health Organization (WHO): Geneva, Switzerland, 2020; Available online: https://www.who.int/news-room/fact-sheets/detail/food-safety (accessed on 22 December 2021).
- Choi, J.; Nelson, D.; Almanza, B. Food safety risk for restaurant management: Use of restaurant health inspection report to predict consumers’ behavioral intention. J. Risk Res. 2019, 22, 1443–1457. [Google Scholar] [CrossRef]
- Vert, M.; Doi, Y.; Hellwich, K.H.; Hess, M.; Hodge, P.; Kubisa, P.; Rinaudo, M.; Schue, F. Terminology for biorelated polymers and applications (IUPAC Recommendations 2012). Pure Appl. Chem. 2012, 84, 377–410. [Google Scholar] [CrossRef]
- Jefferson, K.K. What drives bacteria to produce a biofilm? FEMS Microbiol. Lett. 2004, 236, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Health (US); Centers for Disease Control; Prevention (US); Human Services Dept (US) (Eds.) NIOSH Criteria for a Recommended Standard: Occupational Exposure to Heat and Hot Environments; National Institute on Drug Abuse: Rockville Pike Bethesda, MD, USA, 2018.
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol. 2007, 113, 1–15. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Beuchat, L.R.; Ryu, J.H. Inhibition of Listeria monocytogenes using biofilms of non-pathogenic soil bacteria (Streptomyces spp.) on stainless steel under desiccated condition. Food Microbiol. 2019, 79, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Jordan, K.; Dalmasso, M.; Zentek, J.; Mader, A.; Bruggeman, G.; Wallace, J.; Malakauskas, M. Microbes versus microbes: Control of pathogens in the food chain. J. Sci. Food Agric. 2014, 94, 3079–3089. [Google Scholar] [CrossRef]
- Castellano, P.; Pérez Ibarreche, M.; Blanco Massani, M.; Fontana, C.; Vignolo, G.M. Strategies for pathogen biocontrol using lactic acid bacteria and their metabolites: A focus on meat ecosystems and industrial environments. Microorganisms 2017, 5, 38. [Google Scholar] [CrossRef] [PubMed]
- Gómez, N.C.; Ramiro, J.M.; Quecan, B.X.; de Melo Franco, B.D. Use of potential probiotic lactic acid bacteria (LAB) biofilms for the control of Listeria monocytogenes, Salmonella Typhimurium, and Escherichia coli O157: H7 biofilms formation. Front. Microbiol. 2016, 7, 863. [Google Scholar] [CrossRef]
- Stein, T. Bacillus subtilis antibiotics: Structures, syntheses and specific functions. Mol. Microbiol. 2005, 56, 845–857. [Google Scholar] [CrossRef]
- Yan, L.; Boyd, K.G.; Burgess, J.G. Surface attachment induced production of antimicrobial compounds by marine epiphytic bacteria using modified roller bottle cultivation. J. Mar. Biotechnol. 2002, 4, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Guerrieri, E.; de Niederhäusern, S.; Messi, P.; Sabia, C.; Iseppi, R.; Anacarso, I.; Bondi, M. Use of lactic acid bacteria (LAB) biofilms for the control of Listeria monocytogenes in a small-scale model. Food Control 2009, 20, 861–865. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, E.S.; Kim, B.M.; Ham, J.S.; Oh, M.H. Broad-spectrum antimicrobial activity of cinnamoyl esterase-producing Lactobacilli and their application in fermented rice bran. J. Sci. Food Agric. 2022. [Google Scholar] [CrossRef]
- Kim, J.H.; Baik, S.H. Probiotic properties of Lactobacillus strains with high cinnamoyl esterase activity isolated from jeot-gal, a high-salt fermented seafood. Ann. Microbiol. 2019, 69, 407–417. [Google Scholar] [CrossRef]
- Merino, L.; Trejo, F.M.; De Antoni, G.; Golowczyc, M.A. Lactobacillus strains inhibit biofilm formation of Salmonella sp. isolates from poultry. Food Res. Int. 2019, 123, 258–265. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Beuchat, L.R.; Ryu, J.H. Development of non-pathogenic bacterial biofilms on the surface of stainless steel which are inhibitory to Salmonella enterica. Food Microbiol. 2018, 69, 136–142. [Google Scholar] [CrossRef]
- Oder, M.; Arlič, M.; Bohinc, K.; Fink, R. Escherichia coli biofilm formation and dispersion under hydrodynamic conditions on metal surfaces. Int. J. Environ. Health Res. 2018, 28, 55–63. [Google Scholar] [CrossRef]
- Todhanakasem, T.; Triwattana, K.; Pom, J.; Havanapan, P.O.; Koombhongse, P.; Thitisak, P. Physiological studies of the Pediococcus pentosaceus biofilm. Lett. Appl. Microbiol. 2021, 72, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ibarreche, M.; Mendoza, L.M.; Vignolo, G.; Fadda, S. Proteomic and genetics insights on the response of the bacteriocinogenic Lactobacillus sakei CRL1862 during biofilm formation on stainless steel surface at 10 °C. Int. J. Food Microbiol. 2017, 258, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- García-Cayuela, T.; Korany, A.M.; Bustos, I.; de Cadiñanos, L.P.G.; Requena, T.; Peláez, C.; Martínez-Cuesta, M.C. Adhesion abilities of dairy Lactobacillus plantarum strains showing an aggregation phenotype. Food Res. Int. 2014, 57, 44–50. [Google Scholar] [CrossRef]
- Ahmad, D.; van den Boogaert, I.; Miller, J.; Presswell, R.; Jouhara, H. Hydrophilic and hydrophobic materials and their applications. Energy Sour. A 2018, 40, 2686–2725. [Google Scholar] [CrossRef]
- Simões, M.; Simões, L.C.; Cleto, S.; Pereira, M.O.; Vieira, M.J. The effects of a biocide and a surfactant on the detachment of Pseudomonas fluorescens from glass surfaces. Int. J. Food Microbiol. 2008, 121, 335–341. [Google Scholar] [CrossRef][Green Version]
- Di Bonaventura, G.; Stepanović, S.; Picciani, C.; Pompilio, A.; Piccolomini, R. Effect of environmental factors on biofilm formation by clinical Stenotrophomonas maltophilia isolates. Folia Microbiol. 2007, 52, 86–90. [Google Scholar]
- Burgess, C.M.; Gianotti, A.; Gruzdev, N.; Holah, J.; Knøchel, S.; Lehner, A.; Tresse, O. The response of foodborne pathogens to osmotic and desiccation stresses in the food chain. Int. J. Food Microbiol. 2016, 221, 37–53. [Google Scholar] [CrossRef]
- Obana, N.; Nakamura, K.; Nomura, N. Temperature-regulated heterogeneous extracellular matrix gene expression defines biofilm morphology in Clostridium perfringens. NPJ Biofilms Microbiomes 2020, 6, 29. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J.; Szewzyk, U.; Steinberg, P.; Rice, S.A.; Kjelleberg, S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016, 14, 563–575. [Google Scholar] [PubMed]
- Coda, R.; Cassone, A.; Rizzello, C.G.; Nionelli, L.; Cardinali, G.; Gobbetti, M. Antifungal activity of Wickerhamomyces anomalus and Lactobacillus plantarum during sourdough fermentation: Identification of novel compounds and long-term effect during storage of wheat bread. Appl. Environ. Microbiol. 2011, 77, 3484–3492. [Google Scholar] [CrossRef] [PubMed]
- Scatassa, M.L.; Gaglio, R.; Macaluso, G.; Francesca, N.; Randazzo, W.; Cardamone, C.; Settanni, L. Transfer, composition and technological characterization of the lactic acid bacterial populations of the wooden vats used to produce traditional stretched cheeses. Food Microbiol. 2015, 52, 31–41. [Google Scholar] [PubMed]
- Jara, J.; Pérez-Ramos, A.; Del Solar, G.; Rodríguez, J.M.; Fernández, L.; Orgaz, B. Role of Lactobacillus biofilms in Listeria monocytogenes adhesion to glass surfaces. Int. J. Food Microbiol. 2020, 334, 108804. [Google Scholar] [CrossRef] [PubMed]
- Cruciata, M.; Gaglio, R.; Scatassa, M.L.; Sala, G.; Cardamone, C.; Palmeri, M.; Settanni, L. Formation and characterization of early bacterial biofilms on different wood typologies applied in dairy production. Appl. Environ. Microbiol. 2018, 84, e02107–e02117. [Google Scholar] [CrossRef]
| Escherichia coli O157:H7 | Staphylococcus aureus | Salmonella enterica | Listeria monocytogenes | Bacillus cereus | |
|---|---|---|---|---|---|
| M1-5 | 12.2 ± 0.3 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.6 ± 0.4 |
| M2-8 | 15.3 ± 0.5 | 17.9 ± 0.3 | 11.2 ± 0.5 | 15.4 ± 0.1 | 18.1 ± 0.2 |
| M7-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 15.5 ± 0.6 |
| M56 | 0 ± 0.0 | 13.5 ± 0.2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 |
| M57-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 12.0 ± 0.6 | 11.0 ± 0.8 |
| M102-3 | 24.5 ± 0.2 | 13.6 ± 0.6 | 22.9 ± 0.5 | 16.1 ± 0.4 | 15.6 ± 0.4 |
| M104-1 | 0 ± 0.0 | 23.4 ± 0.1 | 0 ± 0.0 | 13.6 ± 0.7 | 0 ± 0.0 |
| M111-1 | 16.7 ± 0.3 | 12.2 ± 0.4 | 17.8 ± 0.5 | 25.1 ± 0.9 | 16.2 ± 0.1 |
| M111-3 | 0 ± 0.0 | 24.4 ± 0.6 | 0 ± 0.0 | 17.3 ± 0.3 | 0 ± 0.0 |
| M113-5 | 12.3 ± 0.6 | 13.7 ± 0.5 | 15.6 ± 0.2 | 16.0 ± 0.4 | 18.6 ± 0.2 |
| M119-3 | 0 ± 0.0 | 22.2 ± 0.3 | 0 ± 0.0 | 15.2 ± 0.5 | 25.9 ± 0.6 |
| M123-3 | 0 ± 0.0 | 16.5 ± 0.6 | 0 ± 0.0 | 11.1 ± 0.7 | 15.2 ± 0.5 |
| M129-1 | 22.4 ± 0.5 | 16.8 ± 0.2 | 24.0 ± 0.8 | 25.7 ± 0.2 | 17.5 ± 0.1 |
| M132-2 | 20.9 ± 0.6 | 21.7 ± 0.3 | 23.2 ± 0.5 | 25.2 ± 0.2 | 18.1 ± 0.1 |
| M177-4 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.1 ± 0.4 | 16.6 ± 0.8 |
| M182-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 23.5 ± 0.7 | 16.2 ± 0.2 |
| M183-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 15.9 ± 0.5 | 15.0 ± 0.4 |
| M187-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 17.1 ± 0.2 | 16.9 ± 0.6 |
| M188-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 12.5 ± 0.6 | 24.3 ± 0.5 |
| M190-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.9 ± 0.4 | 22.3 ± 0.1 |
| M193-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 17.4 ± 0.9 | 23.6 ± 0.7 |
| M194-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 17.0 ± 0.6 | 24.8 ± 0.1 |
| M194-4 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 18.1 ± 0.2 | 17.7 ± 0.5 |
| M204-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 25.6 ± 0.1 | 23.3 ± 0.6 |
| M205-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.4 ± 0.2 | 24.8 ± 0.5 |
| Y101-5 | 0 ± 0.0 | 13.2 ± 0.1 | 0 ± 0.0 | 11.6 ± 0.6 | 17.2 ± 0.1 |
| Y103-13 | 0 ± 0.0 | 22.0 ± 0.2 | 0 ± 0.0 | 16.4 ± 0.2 | 12.0 ± 0.7 |
| Y105-25 | 0 ± 0.0 | 21.4 ± 0.3 | 0 ± 0.0 | 16.1 ± 0.3 | 13.3 ± 0.2 |
| Y105-27 | 0 ± 0.0 | 11.5 ± 0.2 | 0 ± 0.0 | 11.5 ± 0.9 | 0 ± 0.0 |
| Y105-3 | 0 ± 0.0 | 16.7 ± 0.7 | 0 ± 0.0 | 12.8 ± 0.1 | 0 ± 0.0 |
| Y108-14 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.1 ± 0.5 | 15.5 ± 0.3 |
| Y108-18 | 0 ± 0.0 | 20.9 ± 0.3 | 0 ± 0.0 | 15.2 ± 0.6 | 0 ± 0.0 |
| Y109-10 | 0 ± 0.0 | 21.1 ± 0.8 | 0 ± 0.0 | 13.9 ± 0.2 | 0 ± 0.0 |
| Y110-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 13.2 ± 0.1 | 15.4 ± 0.5 |
| Y110-20 | 0 ± 0.0 | 22.4 ± 0.6 | 0 ± 0.0 | 12.5 ± 0.3 | 0 ± 0.0 |
| Y143-5 | 0 ± 0.0 | 20.7 ± 0.3 | 0 ± 0.0 | 0 ± 0.0 | 22.4 ± 0.1 |
| Y148-5 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 12.3 ± 0.1 | 13.0 ± 0.2 |
| Y149-5 | 0 ± 0.0 | 22.5 ± 0.1 | 0 ± 0.0 | 15.3 ± 0.3 | 0 ± 0.0 |
| Y149-6 | 0 ± 0.0 | 22.1 ± 0.3 | 0 ± 0.0 | 15.8 ± 0.5 | 0 ± 0.0 |
| Y154-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.6 ± 0.2 | 15.3 ± 0.3 |
| Y154-4 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 17.4 ± 0.9 | 17.7 ± 0.5 |
| Y155-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 21.3 ± 0.4 | 14.8 ± 0.1 |
| Y156-3 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 20.5 ± 0.6 | 13.2 ± 0.3 |
| Y205-3 | 0 ± 0.0 | 15.5 ± 0.9 | 0 ± 0.0 | 12.8 ± 0.6 | 0 ± 0.0 |
| Y21-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 16.2 ± 0.7 | 11.3 ± 0.5 |
| Y24-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 11.4 ± 0.9 | 12.0 ± 0.4 |
| Y28-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 11.7 ± 0.2 | 12.2 ± 0.5 |
| Y30-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 14.4 ± 0.5 | 16.8 ± 0.1 |
| Y69-1 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 15.5 ± 0.4 | 12.7 ± 0.2 |
| Y69-3 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 17.1 ± 0.3 | 13.6 ± 0.9 |
| Y83-2 | 0 ± 0.0 | 0 ± 0.0 | 0 ± 0.0 | 11.5 ± 0.7 | 15.1 ± 0.6 |
| Isolates | Hydrophobicity (%) | Auto-Aggregation (%) | Biofilm Formation (Crystal Violet Assay) |
|---|---|---|---|
| Leuconostoc mesenteroides M2-8 | 38.40 ± 1.06 c,b | 20.53 ± 2.19 c | 0.59 ± 0.09 c |
| Lactobacillus sakei M102-3 | 43.70 ± 2.41 b | 22.10 ± 2.56 c | 0.89 ± 0.13 b |
| Leuconostoc lactis M111-1 | 34.76 ± 2.98 c | 24.56 ± 3.16 c | 0.57 ± 0.10 c |
| Lactobacillus curvatus M113-5 | 71.21 ± 3.35 a | 45.98 ± 3.05 b | 0.85 ± 0.08 b |
| Lactobacillus sakei M129-1 | 69.73 ± 6.11 a | 47.53 ± 3.33 a,b | 1.48 ± 0.15 a |
| Pediococcus pentosaceus M132-2 | 68.90 ± 3.53 a | 52.28 ± 2.11 a | 1.59 ± 0.14 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.-H.; Lee, E.-S.; Song, K.-J.; Kim, B.-M.; Ham, J.-S.; Oh, M.-H. Development of Desiccation-Tolerant Probiotic Biofilms Inhibitory for Growth of Foodborne Pathogens on Stainless Steel Surfaces. Foods 2022, 11, 831. https://doi.org/10.3390/foods11060831
Kim J-H, Lee E-S, Song K-J, Kim B-M, Ham J-S, Oh M-H. Development of Desiccation-Tolerant Probiotic Biofilms Inhibitory for Growth of Foodborne Pathogens on Stainless Steel Surfaces. Foods. 2022; 11(6):831. https://doi.org/10.3390/foods11060831
Chicago/Turabian StyleKim, Jong-Hui, Eun-Seon Lee, Kyoung-Ja Song, Bu-Min Kim, Jun-Sang Ham, and Mi-Hwa Oh. 2022. "Development of Desiccation-Tolerant Probiotic Biofilms Inhibitory for Growth of Foodborne Pathogens on Stainless Steel Surfaces" Foods 11, no. 6: 831. https://doi.org/10.3390/foods11060831
APA StyleKim, J.-H., Lee, E.-S., Song, K.-J., Kim, B.-M., Ham, J.-S., & Oh, M.-H. (2022). Development of Desiccation-Tolerant Probiotic Biofilms Inhibitory for Growth of Foodborne Pathogens on Stainless Steel Surfaces. Foods, 11(6), 831. https://doi.org/10.3390/foods11060831








