Mutation of Signal Transducer and Activator of Transcription 5 (STAT5) Binding Sites Decreases Milk Allergen αS1-Casein Content in Goat Mammary Epithelial Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Goat CSN1S1 Promoter Region Cloning and Bioinformatics Analysis
2.3. Vector Construction
2.4. Cell Culture
2.5. Cell Treatment
2.6. Luciferase Assays
2.7. RNA Extraction and Quantitative Real-Time PCR
2.8. Western Blot
2.9. Chromatin Immunoprecipitation (ChIP) Assays
2.10. Statistical Analysis
3. Results
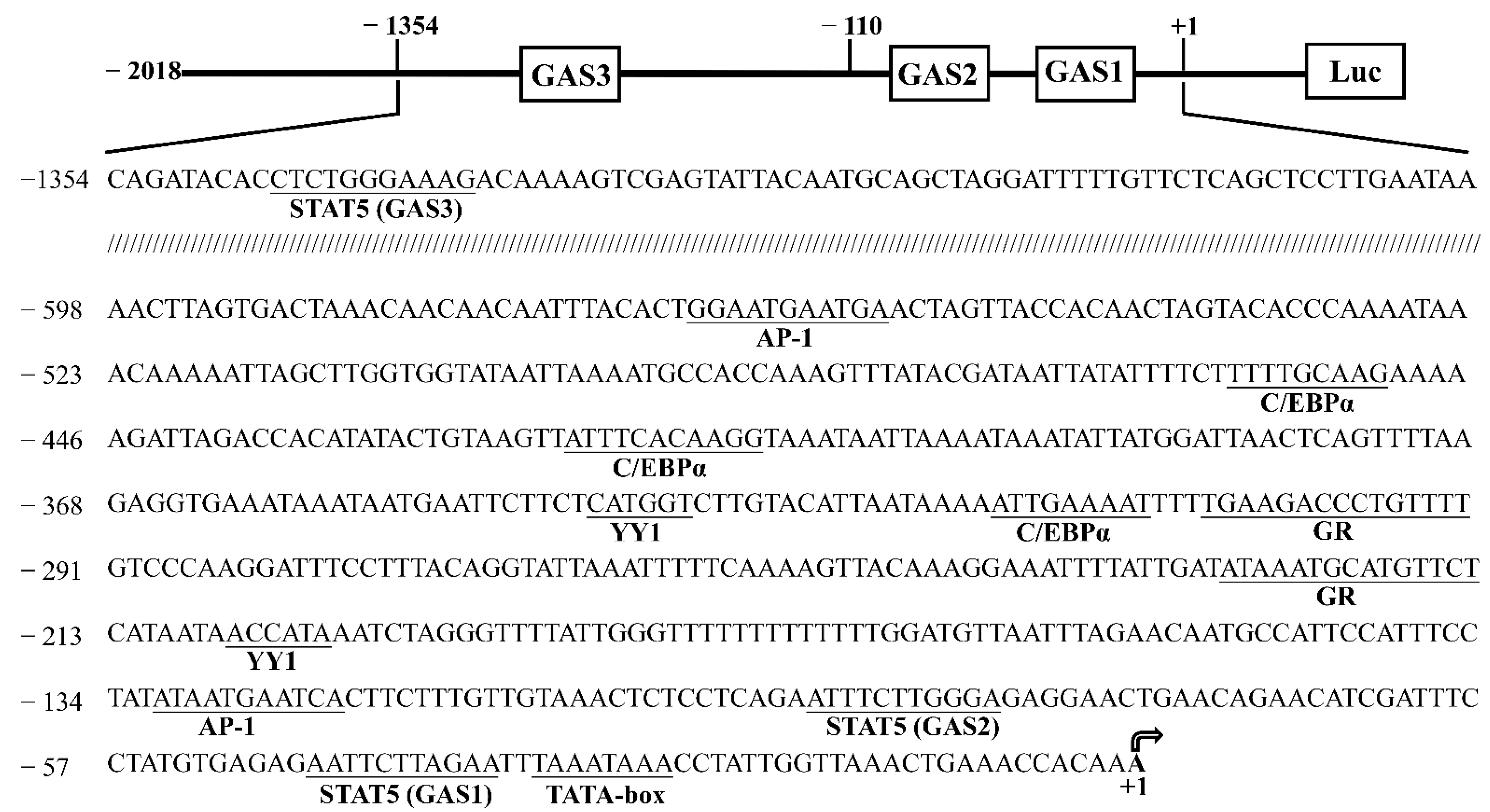
3.1. Cloning and Characterization of the Goat CSN1S1 Promoter
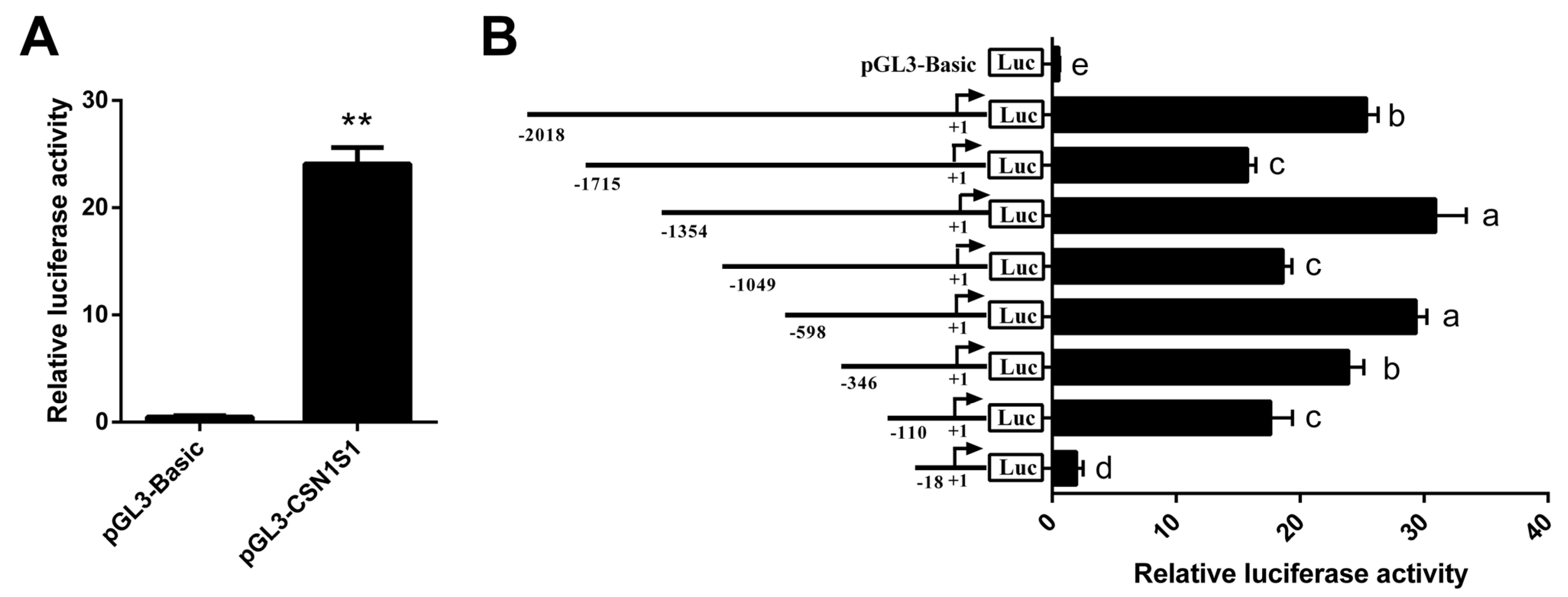
3.2. Core Promoter Region Identification of CSN1S1 Gene
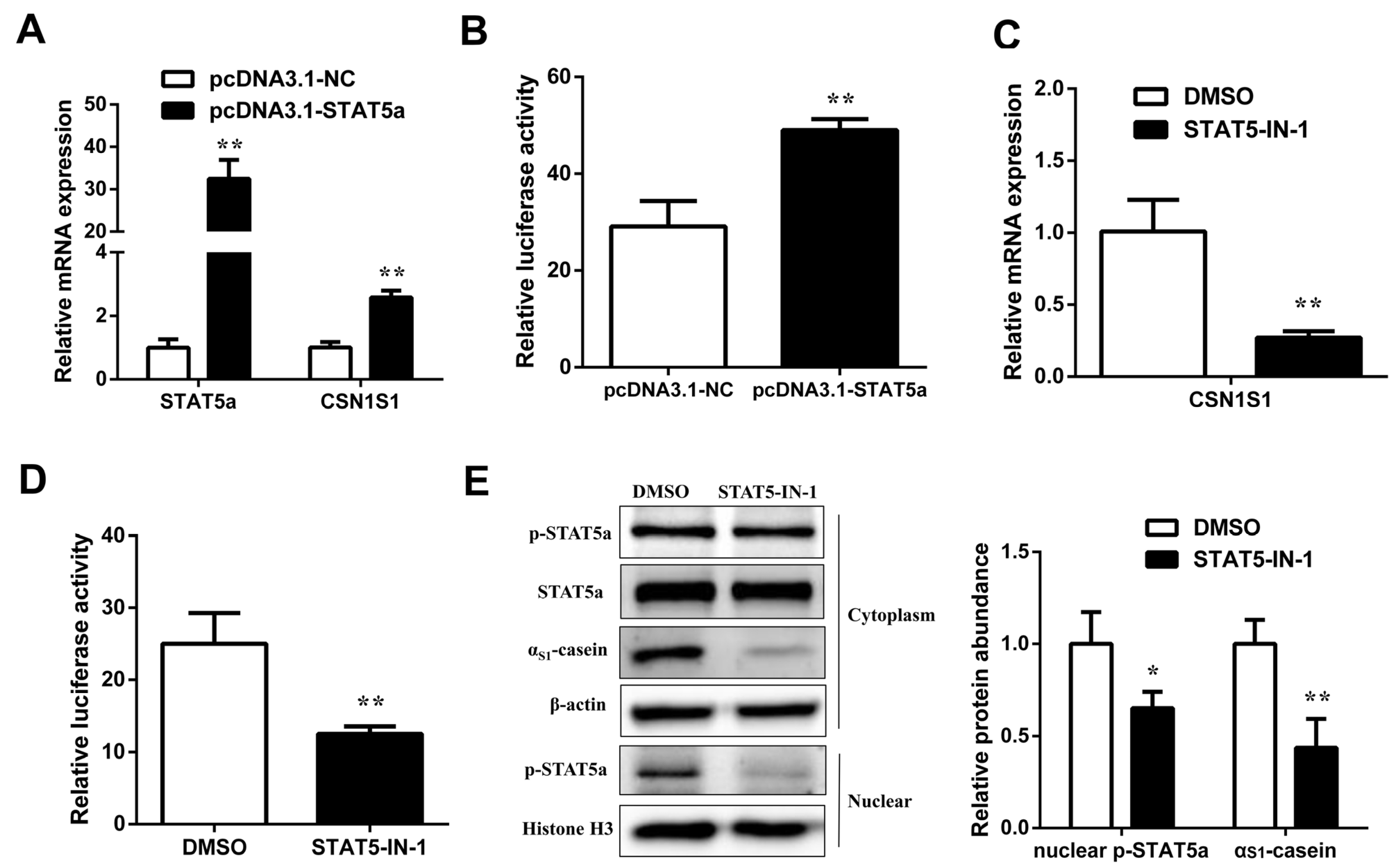
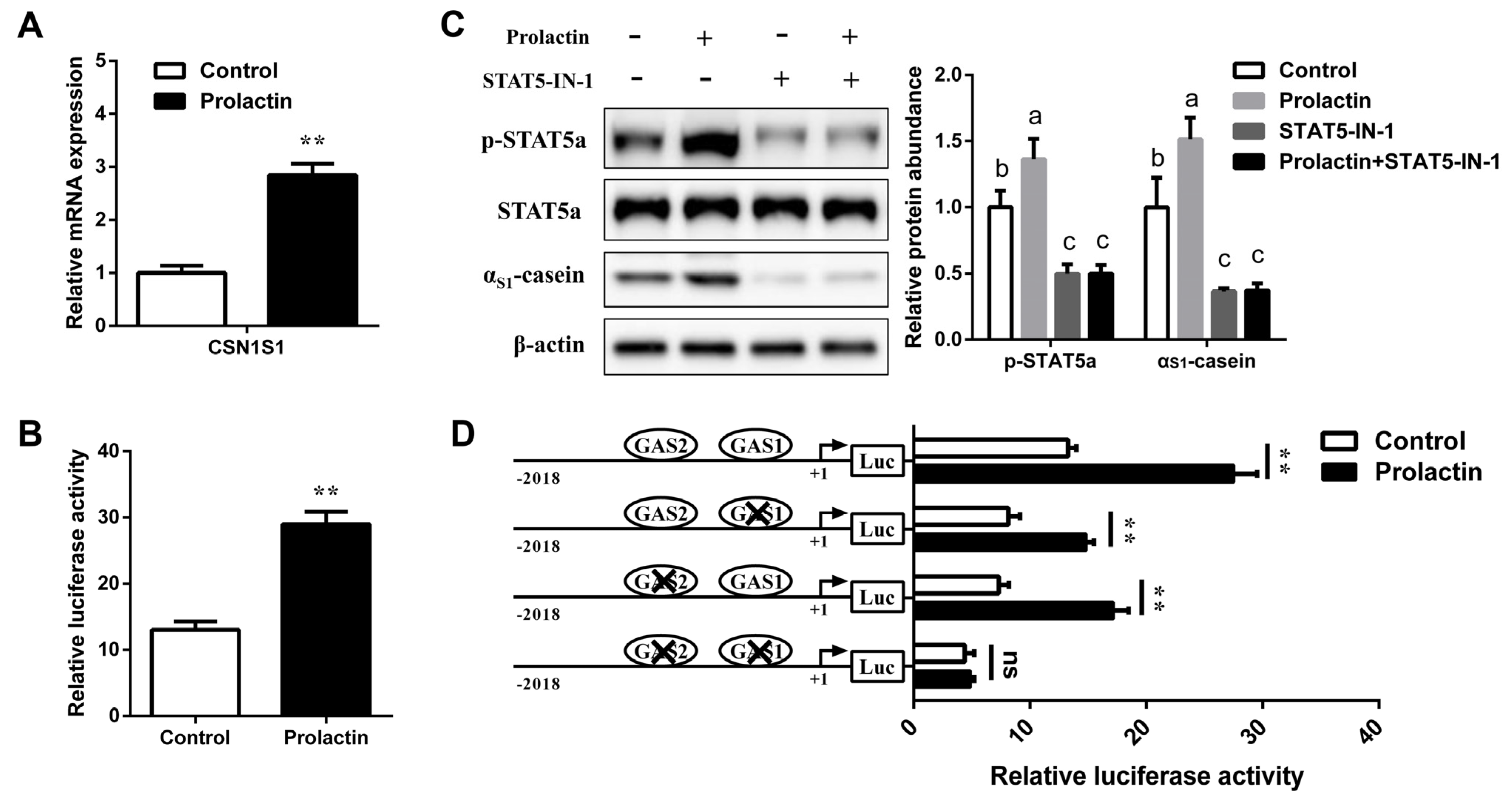
3.3. STAT5 Upregulates CSN1S1 Expression
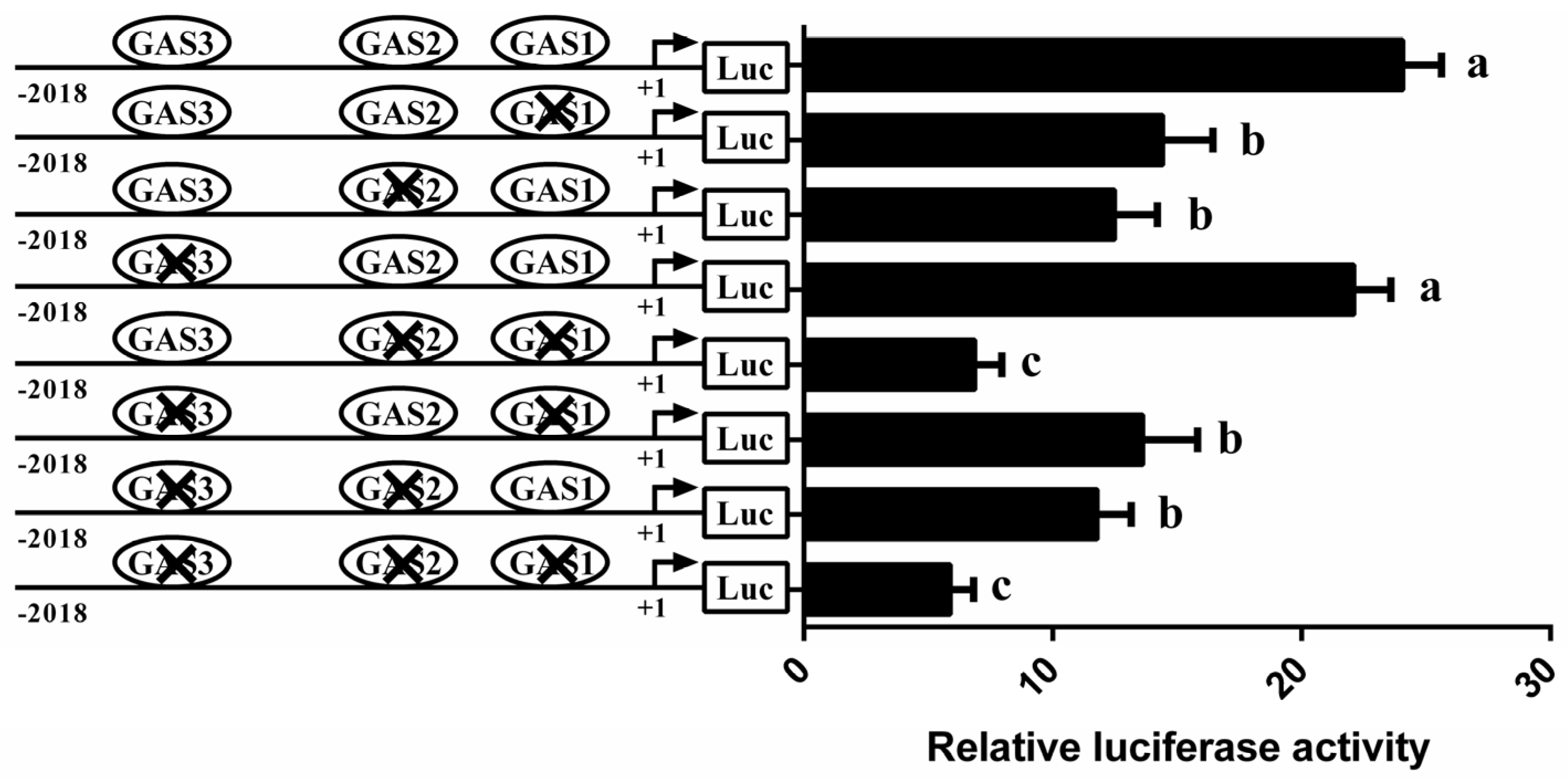
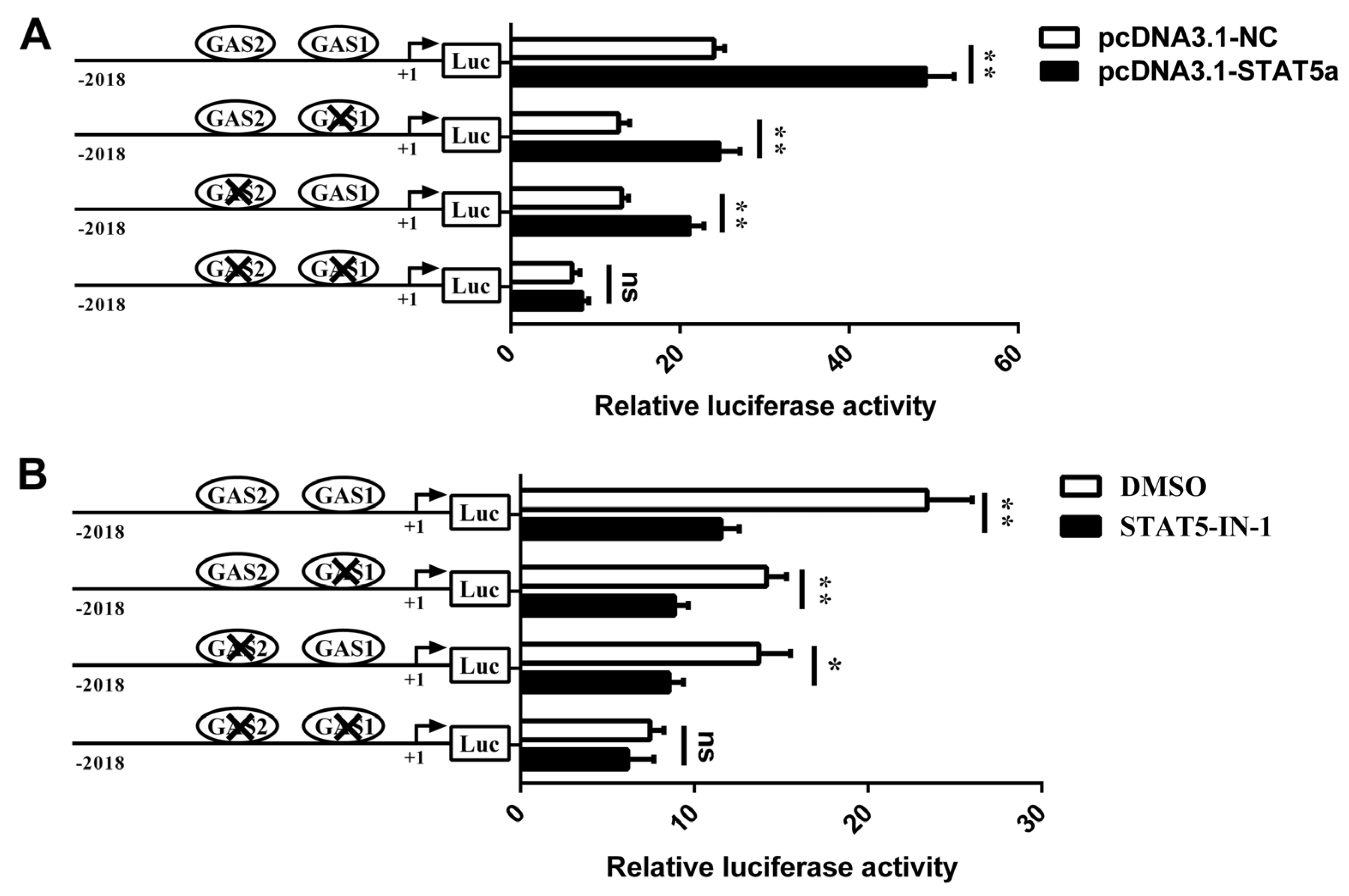
3.4. Involvement of STAT5 in Transcriptional Regulation of CSN1S1
3.5. Prolactin Increases CSN1S1 Promoter Activity via STAT5
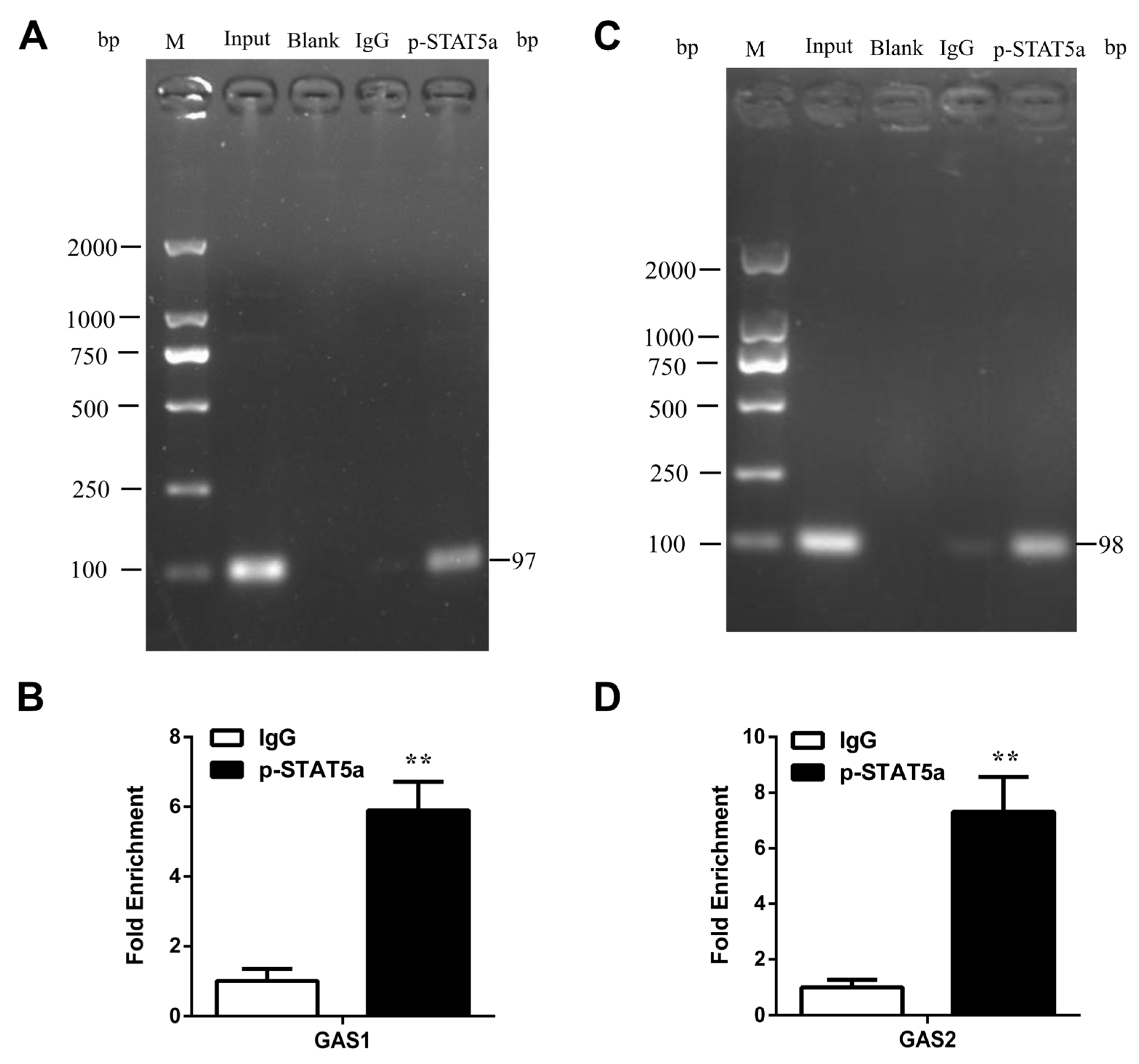
3.6. STAT5 Binds to the GAS Sites of CSN1S1 Promoter Region
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gillespie, A.L.; Green, B.D. The bioactive effects of casein proteins on enteroendocrine cell health, proliferation and incretin hormone secretion. Food Chem. 2016, 211, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.W.; Juarez, M.; Ramos, M.; Haenlein, G.F.W. Physico-chemical characteristics of goat and sheep milk. Small Rumin. Res. 2007, 68, 88–113. [Google Scholar] [CrossRef]
- Hochwallner, H.; Schulmeister, U.; Swoboda, I.; Focke-Tejkl, M.; Reininger, R.; Civaj, V.; Campana, R.; Thalhamer, J.; Scheiblhofer, S.; Balic, N. Infant milk formulas differ regarding their allergenic activity and induction of T cell and cytokine responses. Allergy 2016, 72, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; Garcia, M.B.M. A 100-Year Review: Advances in goat milk research. J. Dairy Sci. 2017, 100, 10026–10044. [Google Scholar] [CrossRef]
- Fiocchi, A.; Schuenemann, H.J.; Brozek, J.; Restani, P.; Beyer, K.; Troncone, R.; Martelli, A.; Terracciano, L.; Bahna, S.L.; Rance, F.; et al. Diagnosis and Rationale for Action against Cow’s Milk Allergy (DRACMA): A summary report. J. Allergy Clin. Immun. 2010, 126, 1119–1128.e12. [Google Scholar] [CrossRef]
- Hochwallner, H.; Schulmeister, U.; Swoboda, I.; Spitzauer, S.; Valenta, R. Cow’s milk allergy: From allergens to new forms of diagnosis, therapy and prevention. Methods 2014, 66, 22–33. [Google Scholar] [CrossRef]
- Caubet, J.C.; Lin, J.; Ahrens, B.; Gimenez, G.; Bardina, L.; Niggemann, B.; Sampson, H.A.; Beyer, K. Natural tolerance development in cow’s milk allergic children: IgE and IgG4 epitope binding. Allergy 2017, 72, 1677–1685. [Google Scholar] [CrossRef]
- Schulmeister, U.; Hochwallner, H.; Swoboda, I.; Focketejkl, M.; Geller, B.; Nystrand, M.; Härlin, A.; Thalhamer, J.; Scheiblhofer, S.; Keller, W. Cloning, Expression, and Mapping of Allergenic Determinants of alpha S1-Casein, a Major Cow’s Milk Allergen. J. Immunol. 2009, 182, 7019–7029. [Google Scholar] [CrossRef]
- Clark, S.; Sherbon, J.W. Genetic variants of alpha(s1)-CN in goat milk: Breed distribution and associations with milk composition and coagulation properties. Small Rumin. Res. 2000, 38, 135–143. [Google Scholar] [CrossRef]
- Carillier-Jacquin, C.; Larroque, H.; Robert-Granie, C. Including a(s1) casein gene information in genomic evaluations of French dairy goats. Genet. Sel. Evol. 2016, 48, 54. [Google Scholar] [CrossRef]
- Shin, H.Y.; Hennighausen, L.; Yoo, K.H. STAT5-Driven Enhancers Tightly Control Temporal Expression of Mammary-Specific Genes. J. Mammary Gland Biol. 2019, 24, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Radler, P.D.; Wehde, B.L.; Wagner, K. Crosstalk between STAT5 activation and PI3K/AKT functions in normal and transformed mammary epithelial cells. Mol. Cell. Endocrinol. 2017, 451, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Maurer, B.; Kollmann, S.; Pickem, J.; Hoelbl-Kovacic, A.; Sexl, V. STAT5a and STAT5B-Twins with Different Personalities in Hematopoiesis and Leukemia. Cancers 2019, 11, 1726. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Huang, S.; Qiao, S.; Jiang, J.; Shi, D.; Li, X. Cloning and functional characterization of STAT5a and STAT5b genes in buffalo mammary epithelial cells. Anim. Biotechnol. 2020, 31, 59–66. [Google Scholar] [CrossRef]
- Reichenstein, M.; Rauner, G.; Barash, I. Conditional repression of STAT5 expression during lactation reveals its exclusive roles in mammary gland morphology, milk-protein gene expression, and neonate growth. Mol. Reprod. Dev. 2011, 78, 585–596. [Google Scholar] [CrossRef]
- Zhong, W.; Shen, J.; Liao, X.; Liu, X.; Zhang, J.; Zhou, C.; Jin, Y. Camellia (Camellia oleifera Abel.) seed oil promotes milk fat and protein synthesis-related gene expression in bovine mammary epithelial cells. Food Sci. Nutr. 2019, 8, 419–427. [Google Scholar] [CrossRef]
- Kung, M.H.; Lee, Y.J.; Hsu, J.T.; Huang, M.C.; Ju, Y.T. A functional study of proximal goat beta-casein promoter and intron 1 in immortalized goat mammary epithelial cells. J. Dairy Sci. 2015, 98, 3859–3875. [Google Scholar] [CrossRef]
- Song, N.; Luo, J.; Huang, L.; Tian, H.; Chen, Y.; He, Q. miR-204-5p and miR-211 synergistically downregulate the alpha(S1)-casein content and contribute to the lower allergy of goat milk. J. Agric. Food Chem. 2021, 69, 5353–5362. [Google Scholar] [CrossRef]
- Shi, H.; Shi, H.; Luo, J.; Wang, W.; Haile, A.B.; Xu, H.; Li, J. Establishment and characterization of a dairy goat mammary epithelial cell line with human telomerase (hT-MECs). Anim. Sci. J. 2014, 85, 735–743. [Google Scholar] [CrossRef]
- Song, N.; Chen, Y.; Luo, J.; Huang, L.; Tian, H.; Li, C.; Loor, J.J. Negative regulation of alpha(S1)-casein (CSN1S1) improves beta-casein content and reduces allergy potential in goat milk. J. Dairy Sci. 2020, 103, 9561–9572. [Google Scholar] [CrossRef]
- Yao, D.; Luo, J.; He, Q.; Shi, H.; Li, J.; Wang, H.; Xu, H.; Chen, Z.; Yi, Y.; Loor, J.J. SCD1 alters long-chain fatty acid (LCFA) composition and its expression is directly regulated by SREBP-1 and PPAR 1 in dairy goat mammary cells. J. Cell. Physiol. 2017, 232, 635–649. [Google Scholar] [CrossRef]
- Bionaz, M.; Loor, J.J. Identification of reference genes for quantitative real-time PCR in the bovine mammary gland during the lactation cycle. Physiol. Genom. 2007, 29, 312–319. [Google Scholar] [CrossRef]
- Archila, L.D.; Khan, F.S.; Bhatnagar, N.; Robinson, D.; Farrington, M.L.; Kwok, W.W. Alpha(S1)-Casein elucidate major T-cell responses in cow’s milk allergy. J. Allergy Clin. Immun. 2017, 140, 854–857. [Google Scholar] [CrossRef]
- Dong, X.; Zhou, Z.; Wang, L.; Saremi, B.; Helmbrecht, A.; Wang, Z.; Loor, J.J. Increasing the availability of threonine, isoleucine, valine, and leucine relative to lysine while maintaining an ideal ratio of lysine: Methionine alters mammary cellular metabolites, mammalian target of rapamycin signaling, and gene transcription. J. Dairy Sci. 2018, 101, 5502–5514. [Google Scholar] [CrossRef]
- Ahmed, A.; Rahmatalla, S.; Bortfeldt, R.; Arends, D.; Reissmann, M.; Brockmann, G. Milk protein polymorphisms and casein haplotypes in Butana cattle. J. Appl. Genet. 2017, 58, 261–271. [Google Scholar] [CrossRef]
- Noce, A.; Pazzola, M.; Dettori, M.L.; Amills, M.; Castello, A.; Cecchinato, A.; Bittante, G.; Vacca, G.M. Variations at regulatory regions of the milk protein genes are associated with milk traits and coagulation properties in the Sarda sheep. Anim. Genet. 2016, 47, 717–726. [Google Scholar] [CrossRef]
- Bonfatti, V.; Giantin, M.; Gervaso, M.; Coletta, A.; Dacasto, M.; Carnier, P. Effect of CSN1S1-CSN3 (alpha(s1)-kappa-casein) composite genotype on milk production traits and milk coagulation properties in Mediterranean water buffalo. J. Dairy Sci. 2012, 95, 3435–3443. [Google Scholar] [CrossRef]
- Cieslak, J.; Wodas, L.; Borowska, A.; Pawlak, P.; Czyzak-Runowska, G.; Wojtowski, J.; Puppel, K.; Kuczynska, B.; Mackowski, M. 5′-flanking variants of equine casein genes (CSN1S1, CSN1S2, CSN2, CSN3) and their relationship with gene expression and milk composition. J. Appl. Genet. 2019, 60, 71–78. [Google Scholar] [CrossRef]
- Ramos Matosinho, C.G.; Rosse, I.C.; Souza Fonseca, P.A.; de Oliveira, F.S.; Dos Santos, F.G.; Gomes Araujo, F.M.; de Matos Salim, A.C.; Lopes, B.C.; Arbex, W.A.; Machado, M.A.; et al. Identification and in silico characterization of structural and functional impacts of genetic variants in milk protein genes in the Zebu breeds Guzerat and Gyr. Trop. Anim. Health Prod. 2021, 53, 524. [Google Scholar] [CrossRef]
- Wal, J.M. Structure and function of milk allergens. Allergy 2001, 56, 35–58. [Google Scholar] [CrossRef]
- Bai, W.; Yin, R.; Dou, Q.; Yang, J.; Zhao, S.; Ma, Z.; Yin, R.; Luo, G.; Zhao, Z. A single nucleotide polymorphism and sequence analysis of CSN1S1 gene promoter region in Chinese BOS Grunniens (YAK). Anim. Biotechnol. 2010, 21, 36–41. [Google Scholar] [CrossRef]
- Ramunno, L.; Cosenza, G.; Rando, A.; Illario, R.; Gallo, D.; Berardino, D.D.; Masina, P. The goat alpha s1-casein gene: Gene structure and promoter analysis. Gene 2004, 334, 105–111. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, J.; Sun, Y.; Dong, X.; Wang, Z.; Chen, J.; Dong, G. PGN and LTA from staphylococcus aureus induced inflammation and decreased lactation through regulating DNA methylation and histone H3 acetylation in bovine mammary epithelial cells. Toxins 2020, 12, 238. [Google Scholar] [CrossRef]
- Montazer-Torbati, M.B.; Hue-Beauvais, C.; Droineau, S.; Ballester, M.; Coant, N.; Aujean, E.; Petitbarat, M.; Rijnkels, M.; Devinoy, E. Epigenetic modifications and chromatin loop organization explain the different expression profiles of the Tbrg4, WAP and Ramp3 genes. Exp. Cell Res. 2008, 314, 975–987. [Google Scholar] [CrossRef]
- Nguyen, M.; Boutinaud, M.; Petridou, B.; Gabory, A.; Pannetier, M.; Chat, S.; Bouet, S.; Jouneau, L.; Jaffrezic, F.; Lalo, D. DNA methylation and transcription in a distal region upstream from the bovine alphaS1 casein gene after once or twice daily milking. PLoS ONE 2014, 9, e111556. [Google Scholar] [CrossRef]
- Buser, A.C.; Obr, A.E.; Kabotyanski, E.B.; Grimm, S.L.; Rosen, J.M.; Edwards, D.P. Progesterone receptor directly inhibits beta-casein gene transcription in mammary epithelial cells through promoting promoter and enhancer repressive chromatin modifications. Mol. Endocrinol. 2011, 25, 955–968. [Google Scholar] [CrossRef][Green Version]
- Yao, D.; Luo, J.; He, Q.; Xu, H.; Li, J.; Shi, H.; Wang, H.; Chen, Z.; Loor, J.J. Liver X receptor alpha promotes the synthesis of monounsaturated fatty acids in goat mammary epithelial cells via the control of stearoyl-coenzyme A desaturase 1 in an SREBP-1-dependent manner. J. Dairy Sci. 2016, 99, 6391–6402. [Google Scholar] [CrossRef]
- Qian, X.; Zhao, F. Regulatory roles of Oct proteins in the mammary gland. Biochim. Biophys. Acta 2016, 1859, 812–819. [Google Scholar] [CrossRef]
- Li, S.; Khan, R.; Raza, S.H.A.; Jieyun, H.; Chugang, M.; Kaster, N.; Cheng, G.; Zhao, C.; Schreurs, N.M.; Zan, L. Function and characterization of the promoter region of perilipin 1 (PLIN1): Roles of E2F1, PLAG1, C/EBP beta, and SMAD3 in bovine adipocytes. Genomics 2020, 112, 2400–2409. [Google Scholar] [CrossRef]
- Xu, H.; Luo, J.; Ma, G.; Zhang, X.; Yao, D.; Li, M.; Loor, J.J. Acyl-CoA synthetase short-chain family member 2 (ACSS2) is regulated by SREBP-1 and plays a role in fatty acid synthesis in caprine mammary epithelial cells. J. Cell. Physiol. 2018, 233, 1005–1016. [Google Scholar] [CrossRef]
- Xu, H.F.; Luo, J.; Wang, H.P.; Wang, H.; Zhang, T.Y.; Tian, H.B.; Yao, D.W.; Loor, J.J. Sterol regulatory element binding protein-1 (SREBP-1)c promoter: Characterization and transcriptional regulation by mature SREBP-1 and liver X receptor a in goat mammary epithelial cells. J. Dairy Sci. 2016, 99, 1595–1604. [Google Scholar] [CrossRef]
- He, Q.; Luo, J.; Wu, J.; Li, Z.; Yao, W.; Zang, S.; Niu, H. ELOVL6 promoter binding sites directly targeted by sterol regulatory element binding protein 1 in fatty acid synthesis of goat mammary epithelial cells. J. Dairy Sci. 2021, 104, 6253–6266. [Google Scholar] [CrossRef]
- Geng, Z.; Shan, X.; Lian, S.; Wang, J.; Wu, R. LPS-induced SOCS3 antagonizes the JAK2-STAT5 pathway and inhibits beta-casein synthesis in bovine mammary epithelial cells. Life Sci. 2021, 278, 119547. [Google Scholar] [CrossRef]
- Cai, J.; Wang, D.; Zhao, F.; Liang, S.; Liu, J. AMPK-mTOR pathway is involved in glucose-modulated amino acid sensing and utilization in the mammary glands of lactating goats. J. Anim. Sci. Biotechnol. 2020, 11, 32. [Google Scholar] [CrossRef]
- Luo, C.; Zhao, S.; Zhang, M.; Gao, Y.; Wang, J.; Hanigan, M.D.; Zheng, N. SESN2 negatively regulates cell proliferation and casein synthesis by inhibition the amino acid-mediated mTORC1 pathway in cow mammary epithelial cells. Sci. Rep.-UK 2018, 8, 3912. [Google Scholar] [CrossRef]
- Khan, M.Z.; Khan, A.; Xiao, J.; Ma, Y.; Ma, J.; Gao, J.; Cao, Z. Role of the JAK-STAT pathway in bovine mastitis and milk production. Animals 2020, 10, 2107. [Google Scholar] [CrossRef]
- Huang, Y.; Zhao, F.; Luo, C.; Zhang, X.; Si, Y.; Sun, Z.; Zhang, L.; Li, Q.; Gao, X. SOCS3-mediated blockade reveals major contribution of JAK2/STAT5 signaling pathway to lactation and proliferation of dairy cow mammary epithelial cells in vitro. Molecules 2013, 18, 12987–13002. [Google Scholar] [CrossRef]
- Ghosh, A.; Radhakrishnan, R. Time-dependent antagonist-agonist switching in receptor tyrosine kinase-mediated signaling. BMC Bioinform. 2019, 20, 242. [Google Scholar] [CrossRef]
- Tian, M.; Qi, Y.; Zhang, X.; Wu, Z.; Chen, J.; Chen, F.; Guan, W.; Zhang, S. Regulation of the JAK2-STAT5 Pathway by Signaling Molecules in the Mammary Gland. Front. Cell Dev. Biol. 2020, 8, 604896. [Google Scholar] [CrossRef]
- Zhou, J.; Jiang, M.; Shi, Y.; Song, S.; Hou, X.; Lin, Y. Prolactin regulates LAT1 expression via STAT5 (signal transducer and activator of transcription 5) signaling in mammary epithelial cells of dairy cows. J. Dairy Sci. 2020, 103, 6627–6634. [Google Scholar] [CrossRef]
- Wang, B.; Men, J.; Wang, C.; Hou, X.; Zhao, F. Laminin-dependent integrin beta 1 signaling regulates milk protein synthesis via prolactin/STAT5 pathway in bovine mammary epithelial cells. Biochem. Bioph. Res. Commun. 2020, 524, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Oyama, S.; Kuki, C.; Tsugami, Y.; Matsunaga, K.; Suzuki, T.; Nishimura, T. Distinct roles of prolactin, epidermal growth factor, and glucocorticoids in beta-casein secretion pathway in lactating mammary epithelial cells. Mol. Cell. Endocrinol. 2017, 440, 16–24. [Google Scholar] [CrossRef] [PubMed]
| Primers | Primer Sequence † (5′–3′) | Binding Region |
|---|---|---|
| Cloning and 5′ deletion primers | ||
| Forward 1 | CGGGGTACCGGTGGCAAGAATAGTATTACAG | −2018 |
| Forward 2 | CGGGGTACCGACATCACTTTACTGATAAG | −1715 |
| Forward 3 | CGGGGTACCCAGATACACCTCTGGGAAAG | −1354 |
| Forward 4 | CGGGGTACCGATTCCTTTCTTATAAACAA | −1049 |
| Forward 5 | CGGGGTACCAACTTAGTGACTAAACAACA | −598 |
| Forward 6 | CGGGGTACCCTTCTCATGGTCTTGTACAT | −346 |
| Forward 7 | CGGGGTACCGTAAACTCTCCTCAGAATTTC | −110 |
| Forward 8 | CGGGGTACCGTTAAACTGAAACCACAAAAT | −18 |
| Reverse | CCCAAGCTTCAACTGCGTATTAGTGAAGA | +183 |
| Site-directed mutagenesis primers ‡ | ||
| GAS1-mut | CCTATGTGAGAGAAcgtTTAtgcTTTAAA | −46 |
| GAS1-anti-mut | TAGGTTTATTTAAAgcaTAAacgTTCTCT | −46 |
| GAS2-mut | ACTCTCCTCAGAATcgtTTGtgcGAGGAA | −92 |
| GAS2-anti-mut | CTGTTCAGTTCCTCgcaCAAacgATTCTG | −92 |
| GAS3-mut | AAGTCCAGATACACgcaTGGacgAGACAAAA | −1345 |
| GAS3-anti-mut | TACTCGACTTTTGTCTcgtCCAtgcGTGTAT | −1345 |
| GenBank ID | Gene | Primer Sequence (5′–3′) | Length (bp) |
|---|---|---|---|
| XM_018049127.1 | CSN1S1 | F, TCCACTAGGCACACAATACACTGA | 61 |
| R, GCCAATGGGATTAGGGATGTC | |||
| XM_018065112.1 | STAT5a | F, CCATCGACCTGGACAATCCC | 96 |
| R, CGACTTGGTGCTCTGCCTTCTT | |||
| XM_005700842.2 | UXT | F, CAGCTGGCCAAATACCTTCAA | 125 |
| R, GTGTCTGGGACCACTGTGTCAA | |||
| XM_005709411.1 | RPS9 | F, CCTCGACCAAGAGCTGAAG | 64 |
| R, CCTCCAGACCTCACGTTTGTTC | |||
| ChIP primers | GAS1 | F, GAGGAACTGAACAGAACATC | 97 |
| R, GTAAAATGCTAATTTTGTGG | |||
| GAS2 | F, ACAATGCCATTCCATTTCCTAT | 98 | |
| R, AGGAAATCGATGTTCTGTTCAG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, N.; Luo, J.; Huang, L.; Zang, S.; He, Q.; Wu, J.; Huang, J. Mutation of Signal Transducer and Activator of Transcription 5 (STAT5) Binding Sites Decreases Milk Allergen αS1-Casein Content in Goat Mammary Epithelial Cells. Foods 2022, 11, 346. https://doi.org/10.3390/foods11030346
Song N, Luo J, Huang L, Zang S, He Q, Wu J, Huang J. Mutation of Signal Transducer and Activator of Transcription 5 (STAT5) Binding Sites Decreases Milk Allergen αS1-Casein Content in Goat Mammary Epithelial Cells. Foods. 2022; 11(3):346. https://doi.org/10.3390/foods11030346
Chicago/Turabian StyleSong, Ning, Jun Luo, Lian Huang, Saige Zang, Qiuya He, Jiao Wu, and Jiangtao Huang. 2022. "Mutation of Signal Transducer and Activator of Transcription 5 (STAT5) Binding Sites Decreases Milk Allergen αS1-Casein Content in Goat Mammary Epithelial Cells" Foods 11, no. 3: 346. https://doi.org/10.3390/foods11030346
APA StyleSong, N., Luo, J., Huang, L., Zang, S., He, Q., Wu, J., & Huang, J. (2022). Mutation of Signal Transducer and Activator of Transcription 5 (STAT5) Binding Sites Decreases Milk Allergen αS1-Casein Content in Goat Mammary Epithelial Cells. Foods, 11(3), 346. https://doi.org/10.3390/foods11030346






