Vitamin B12 Status and Gut Microbiota among Saudi Females with Obesity
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Collection
2.2.1. Anthropometric Measurements
2.2.2. Biochemical Measurements
2.2.3. Stool Analysis
DNA Extraction
Library Preparation and Sequencing
Identification of the Microbial Composition
2.2.4. Dietary Intake
2.3. Statistical Analysis
3. Results
3.1. Anthropometric and Biochemical Analysis
3.2. Dietary Intake and Lifestyle Characteristics
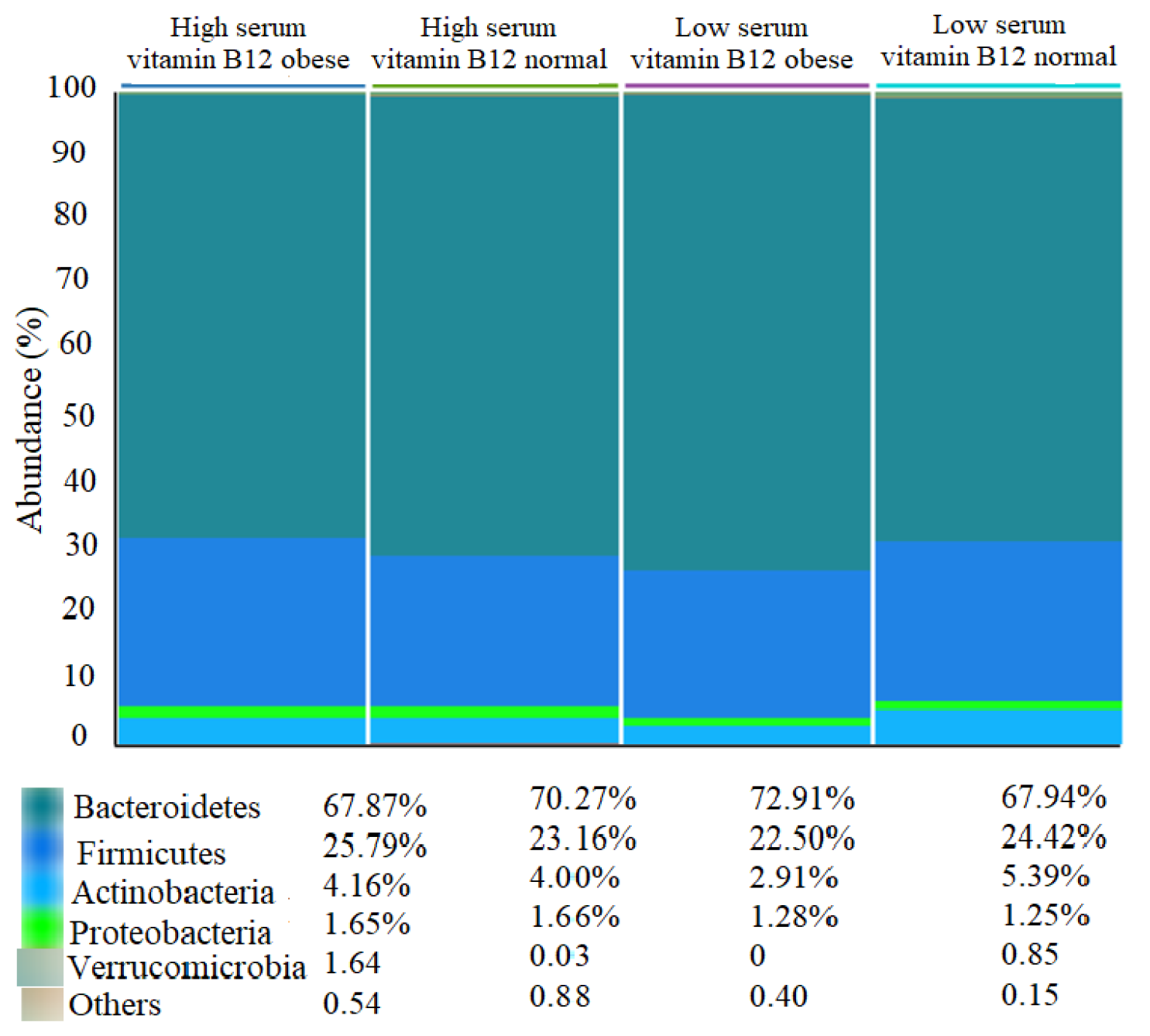
3.3. Differences in Gut Microbiota Composition across Subgroups
3.3.1. Correlation between Vitamin B12 (Serum and Dietary) and Gut Microbiota Composition
3.3.2. Abundance and Diversity
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Obesity and Overweight. Available online: https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 23 October 2021).
- World Health Survey—Saudi Arabia (KSAWHS) 2019. Ministry of Health. 2019. Available online: https://www.moh.gov.sa/en/Ministry/Statistics/Population-Health-Indicators/Documents/World-Health-Survey-Saudi-Arabia.pdf (accessed on 17 April 2022).
- Westerman, R.; Kuhnt, A.K. Metabolic risk factors and fertility disorders: A narrative review of the female perspective. Reprod. Biomed. Soc. Online 2022, 14, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.A.; Gluckman, P.D. Early developmental conditioning of later health and disease: Physiology or pathophysiology? Physiol. Rev. 2014, 94, 1027–1076. [Google Scholar] [CrossRef] [PubMed]
- Endalifer, M.L.; Diress, G. Epidemiology, Predisposing Factors, Biomarkers, and Prevention Mechanism of Obesity: A Systematic Review. J. Obes. 2020, 2020, 6134362. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Jiang, W.; Huang, W.; Lin, Y.; Chan, F.K.L.; Ng, S.C. Gut microbiota in patients with obesity and metabolic disorders—A systematic review. Genes Nutr. 2022, 17, 2. [Google Scholar] [CrossRef]
- Altveş, S.; Yildiz, H.K.; Vural, H.C. Interaction of the microbiota with the human body in health and diseases. Biosci. Microbiota Food Health 2020, 39, 23–32. [Google Scholar] [CrossRef]
- Tokarek, J.; Gadzinowska, J.; Młynarska, E.; Franczyk, B.; Rysz, J. What Is the Role of Gut Microbiota in Obesity Prevalence? A Few Words about Gut Microbiota and Its Association with Obesity and Related Diseases. Microorganisms 2022, 10, 52. [Google Scholar] [CrossRef]
- Yang, Q.; Liang, Q.; Balakrishnan, B.; Belobrajdic, D.P.; Feng, Q.J.; Zhang, W. Role of Dietary Nutrients in the Modulation of Gut Microbiota: A Narrative Review. Nutrients 2020, 12, 381. [Google Scholar] [CrossRef]
- Singh, P.; Rawat, A.; Alwakeel, M.; Sharif, E.; Al Khodor, S. The potential role of vitamin D supplementation as a gut microbiota modifier in healthy individuals. Sci. Rep. 2020, 10, 21641. [Google Scholar] [CrossRef]
- Deledda, A.; Annunziata, G.; Tenore, G.C.; Palmas, V.; Manzin, A.; Velluzzi, F. Diet-Derived Antioxidants and Their Role in Inflammation, Obesity and Gut Microbiota Modulation. Antioxidants 2021, 10, 708. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.L.; Brito, A.; Guéant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.-H.; et al. Vitamin B 12 deficiency. Nat. Rev. Dis. Prim. 2017, 3, 17040. [Google Scholar] [CrossRef]
- Bailey, R.L.; Carmel, R.; Green, R.; Pfeiffer, C.M.; Cogswell, M.E.; Osterloh, J.D.; Sempos, C.T.; Yetley, E.A. Monitoring of vitamin B-12 nutritional status in the United States by using plasma methylmalonic acid and serum vitamin B-12. Am. J. Clin. Nutr. 2011, 94, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Al-Musharaf, S.; McTernan, P.G.; Hussain, S.D.; Aleisa, K.A.; Alnaami, A.M.; Wani, K.; Saravanan, P.; Al-Daghri, N. Prevalence and Indicators of Vitamin B12 Insufficiency among Young Women of Childbearing Age. Int. J. Environ. Res. Public Health 2021, 18, 1. [Google Scholar] [CrossRef] [PubMed]
- Boachie, J.; Adaikalakoteswari, A.; Samavat, J.; Saravanan, P. Low Vitamin B12 and Lipid Metabolism: Evidence from Pre-Clinical and Clinical Studies. Nutrients 2020, 12, 1925. [Google Scholar] [CrossRef] [PubMed]
- Al-Musharaf, S.; Aljuraiban, G.S.; Hussain, S.D.; Alnaami, A.M.; Saravanan, P.; Al-Daghri, N. Low Serum Vitamin B12 Levels Are Associated with Adverse Lipid Profiles in Apparently Healthy Young Saudi Women. Nutrients 2020, 12, 2395. [Google Scholar] [CrossRef]
- Wiebe, N.; Field, C.J.; Tonelli, M. A systematic review of the vitamin B12, folate and homocysteine triad across body mass index. Obes. Rev. 2018, 19, 1608–1618. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, VitaminB₆, Folate, Vitamin B₁₂, Pantothenic Acid, Biotin, and Choline; Institute of Medicine: Washington, DC, USA, 1998; pp. 306–356. [Google Scholar]
- Guetterman, H.M.; Huey, S.L.; Knight, R.; Fox, A.M.; Mehta, S.; Finkelstein, J.L. Vitamin B-12 and the Gastrointestinal Microbiome: A Systematic Review. Adv. Nutr. 2021, 13, 530–558. [Google Scholar] [CrossRef]
- Sharma, V.; Rodionov, D.A.; Leyn, S.A.; Tran, D.; Iablokov, S.N.; Ding, H.; Peterson, D.A.; Osterman, A.L.; Peterson, S.N. B-Vitamin sharing promotes stability of gut microbial communities. Front. Microbiol. 2019, 10, 1485. [Google Scholar] [CrossRef]
- Voland, L.; Le Roy, T.; Debédat, J.; Clément, K. Gut microbiota and vitamin status in persons with obesity: A key interplay. Obes. Rev. 2022, 23, e13377. [Google Scholar] [CrossRef]
- Crovesy, L.; Masterson, D.; Rosado, E.L. Profile of the gut microbiota of adults with obesity: A systematic review. Eur. J. Clin. Nutr. 2020, 74, 1251–1262. [Google Scholar] [CrossRef]
- DeNicola, E.; Aburizaiza, O.S.; Siddique, A.; Khwaja, H.; Carpenter, D.O. Obesity and public health in the Kingdom of Saudi Arabia. Rev. Environ. Health 2015, 30, 191–205. [Google Scholar] [CrossRef]
- Aljazairy, E.A.; Al-Musharaf, S.; Abudawood, M.; Almaarik, B.; Hussain, S.D.; Alnaami, A.M.; Sabico, S.; Al-Daghri, N.M.; Clerici, M.; Aljuraiban, G.S. Influence of Adiposity on the Gut Microbiota Composition of Arab Women: A Case-Control Study. Biology 2022, 28, 1586. [Google Scholar] [CrossRef] [PubMed]
- Nishida, C.; Ko, G.T.; Kumanyika, S. Body fat distribution and noncommunicable diseases in populations: Overview of the 2008 WHO Expert Consultation on Waist Circumference and Waist-Hip Ratio. Eur. J. Clin. Nutr. 2010, 64, 2–5. [Google Scholar] [CrossRef]
- So, W.Y.; Swearingin, B.; Crooms, B.; Lee, R.; Choi, Y.; Dail, T.K.; Melton, D.; Fuller, T.M.; Ha, C.H. Body composition measurements determined by air displacement plethysmography and eight-polar bioelectrical impedance analysis are equivalent in African American college students. HealthMED 2012, 6, 1896–1899. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, Without Use of the Preparative Ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Salgado, A.L.F.D.A.; De Carvalho, L.; Oliveira, A.C.; Dos Santos, V.N.; Vieira, J.G.; Parise, E.R. Insulin resistance index (HOMA-IR) in the differentiation of patients with non-alcoholic fatty liver disease and healthy individuals. Arq. Gastroenterol. 2010, 47, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Byrd, D.A.; Sinha, R.; Hoffman, K.L.; Chen, J.; Hua, X.; Shi, J.; Chia, N.; Petrosino, J.; Vogtmann, E. Comparison of Methods To Collect Fecal Samples for Microbiome Studies Using Whole-Genome Shotgun Metagenomic Sequencing. MSphere 2020, 26, e00827-19. [Google Scholar] [CrossRef] [PubMed]
- Alkhalaf, M.M.; Edwards, C.A.; Combet, E. Validation of a food frequency questionnaire specific for salt intake in Saudi Arabian adults using urinary biomarker and repeated multiple pass 24-hour dietary recall. Proc. Nutr. Soc. 2015, 74, E337. [Google Scholar] [CrossRef]
- Mearns, G.J.; Rush, E.C. Screening for inadequate dietary vitamin B-12 intake in South Asian women using a nutrient-specific, semi-quantitative food frequency questionnaire. Asia Pac. J. Clin. Nutr. 2017, 26, 1119–1124. [Google Scholar] [CrossRef]
- Waljee, A.K.; Mukherjee, A.; Singal, A.G.; Zhang, Y.; Warren, J.; Balis, U.; Marrero, J.; Zhu, J.; Higgins, P. Comparison of imputation methods for missing laboratory data in medicine. BMJ Open 2013, 3, e002847. [Google Scholar] [CrossRef]
- Jiang, S.; Zhu, Q.; Mai, M.; Yang, W.; Du, G. Vitamin B and vitamin D as modulators of gut microbiota in overweight individuals. Int. J. Food Sci. Nutr. 2020, 71, 1001–1009. [Google Scholar] [CrossRef]
- Lurz, E.; Horne, R.G.; Määttänen, P.; Wu, R.Y.; Botts, S.R.; Li, B.; Rossi, L.; Johnson-Henry, K.C.; Pierro, A.; Surette, M.G. Vitamin B12 Deficiency Alters the Gut Microbiota in a Murine Model of Colitis. Front. Nutr. 2020, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Alexeev, E.E.; Farb, L.; Vickery, T.W.; Zheng, L.; Eric, L.C.; Kitzenberg, D.; Battista, K.D.; Kominsky, D.J.; Robertson, C.E. Oral vitamin B12 supplement is delivered to the distal gut, altering the corrinoid profile and selectively depleting Bacteroides in C57BL/6 mice. Gut Microbes 2019, 10, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Shou, Y.; Zhu, X.; Xu, Y.; Shi, L.; Xiang, S.; Feng, X.; Han, J. Stability of vitamin B12 with the protection of whey proteins and their effects on the gut microbiome. Food Chem. 2019, 276, 298–306. [Google Scholar] [CrossRef]
- Boran, P.; Baris, H.E.; Kepenekli, E.; Erzik, C.; Soysal, A.; Dinh, D.M. The impact of vitamin B12 deficiency on infant gut microbiota. Eur. J. Pediatr. 2020, 179, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Gurwara, S.; Ajami, N.J.; Jang, A.; Hessel, F.C.; Chen, L.; Plew, S.; Wang, Z.; Graham, D.Y.; Hair, C.; White, D.L. Dietary Nutrients Involved in One-Carbon Metabolism and Colonic Mucosa-Associated Gut Microbiome in Individuals with an Endoscopically Normal Colon. Nutrients 2019, 11, 613. [Google Scholar] [CrossRef] [PubMed]
- Palmas, V.; Pisanu, S.; Madau, V.; Casula, E.; Deledda, A.; Cusano, R.; Uva, P.; Vascellari, S.; Loviselli, A.; Manzin, A. Gut microbiota markers associated with obesity and overweight in Italian adults. Sci. Rep. 2021, 11, 5532. [Google Scholar] [CrossRef] [PubMed]
- Stanislawski, M.A.; Dabelea, D.; Lange, L.A.; Wagner, B.D.; Lozupone, C.A. Gut microbiota phenotypes of obesity. Npj Biofilms Microbiomes 2019, 5, 18. [Google Scholar] [CrossRef]
- Harakeh, S.; Angelakis, E.; Karamitros, T.; Bachar, D.; Bahijri, S.; Ajabnoor, G.; Alfadul, S.M.; Farraj, S.A.; Al Amri, T.; Al-Hejin, A. Impact of smoking cessation, coffee and bread consumption on the intestinal microbial composition among Saudis: A cross-sectional study. PLoS ONE 2020, 15, e0230895. [Google Scholar] [CrossRef]
- Yasir, M.; Angelakis, E.; Bibi, F.; Azhar, E.I.; Bachar, D.; Lagier, J.C.; Gaborit, B.; Hassan, A.M.; Jiman-Fatani, A.A.; Alshali, K.Z.; et al. Comparison of the gut microbiota of people in France and Saudi Arabia. Nutr. Diabetes 2015, 5, e153. [Google Scholar] [CrossRef]
- Valentini, L.; Pinto, A.; Bourdel-Marchasson, I.; Ostan, R.; Brigidi, P.; Turroni, S.; Hrelia, S.; Hrelia, P.; Bereswill, S.; Fischer, A.; et al. Impact of personalized diet and probiotic supplementation on inflammation, nutritional parameters and intestinal microbiota—The “RISTOMED project”: Randomized controlled trial in healthy older people. Clin. Nutr. 2015, 34, 593–602. [Google Scholar] [CrossRef]
- Mosca, A.; Leclerc, M.; Hugot, J.P. Gut Microbiota Diversity and Human Diseases: Should We Reintroduce Key Predators in Our Ecosystem? Front. Microbiol. 2016, 7, 455. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, N.; Tan, H.Y.; Li, S.; Zhang, C.; Feng, Y. Function of Akkermansia muciniphila in Obesity: Interactions With Lipid Metabolism, Immune Response and Gut Systems. Front. Microbiol. 2020, 11, 219. [Google Scholar] [CrossRef] [PubMed]
- Putnam, E.E.; Goodman, A.L. B vitamin acquisition by gut commensal bacteria. PLoS Pathog. 2020, 16, e1008208. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.H.; Stabler, S.P. Identification and quantitation of cobalamin and cobalamin analogues in human feces. Am. J. Clin. Nutr. 2008, 87, 1324–1335. [Google Scholar] [CrossRef] [PubMed]
- Degnan, P.H.; Taga, M.E.; Goodman, A.L. Vitamin B12 as a modulator of gut microbial ecology. Cell Metab. 2014, 20, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Vandana, U.K.; Barlaskar, N.H.; Gulzar, A.B.M.; Laskar, I.H.; Kumar, D.; Paul, P.; Pandey, P.; Mazumder, P.B. Linking gut microbiota with the human diseases. Bioinformation 2020, 16, 196–208. [Google Scholar] [CrossRef]


| Variables | Normal | Obese | ||||
|---|---|---|---|---|---|---|
| High Serum Vitamin B12 (>404.0 pg/mL) | Low Serum Vitamin B12 (≤404.0 pg/mL) | p-Value | High Serum Vitamin B12 (>404.0 pg/mL) | Low Serum Vitamin B12 (≤404.0 pg/mL) | p-Value | |
| Anthropometric Measurements | ||||||
| BMI (kg/m2) | 21.8 ± 1.9 | 22.1 ± 1.9 | 0.68 | 35.9 ± 4.2 | 36.6 ± 5.2 | 0.68 |
| WHR (ratio) | 0.7 ± 0.0 | 0.7 ± 0.1 | 0.32 | 0.8 ± 0.1 | 0.8 ± 0.1 | 0.74 |
| Body Fat (%) | 35.2 ± 5.5 | 34.8 ± 5.6 | 0.83 | 51.7 ± 2.4 | 51.1 ± 3.5 | 0.56 |
| Muscle mass (%) | 31.8 ± 6.7 | 25.4 ± 11.8 | 0.03 | 26.2 ± 1.4 | 26.6 ± 2.1 | 0.51 |
| Blood Analysis | ||||||
| Total cholesterol (mmol/L) | 3.4 ± 1.7 | 4.0 ± 1.7 | 0.29 | 3.9 ± 1.0 | 4.8 ± 1.0 | 0.02 |
| HDL-cholesterol (mmol/L) | 0.9 ± 0.4 | 1.1 ± 0.4 | 0.19 | 1.0 ± 0.2 | 1.0 ± 0.3 | 0.82 |
| LDL-cholesterol (mmol/L) | 2.5 ± 1.5 | 2.8 ± 1.5 | 0.53 | 2.8 ± 0.7 | 3.6 ± 1.0 | 0.02 |
| Total cholesterol/HDL ratio | 3.8 ± 1.4 | 3.8 ± 1.9 | 0.88 | 3.9 ± 0.5 | 5.0 ± 1.9 | 0.07 |
| Triglyceride (mmol/L) # | 0.5 (0.4–0.6) | 0.7 (0.4–0.8) | 0.18 | 0.8 (0.6–1.0) | 1.0 (0.9–1.2) | 0.03 |
| FBG (mmol/L) | 4.5 ± 0.9 | 4.5 ± 0.5 | 0.79 | 4.8 ± 0.8 | 4.8 ± 0.4 | 0.92 |
| Insulin (µIU/mL) # | 6.3 (5.0–9.3) | 7.8 (5.1–9.5) | 0.72 | 14.3 (12.5–19.5) | 16.1 (12.2–20.5) | 0.31 |
| HOMA-IR # | 1.2 (0.9–1.5) | 1.4 (0.9–1.8) | 0.59 | 3.4 (2.6–4.2) | 3.7 (2.5–4.4) | 0.57 |
| HOMA-β # | 112.0 (66.8–145.1) | 127.1 (80.1–154.5) | 0.36 | 210.0 (168.6–224.5) | 252.8 (183.1–322.2) | 0.36 |
| Vitamin B12 (pg/mL) | 620.4 (474.3–688.7) | 334.7 (295.0–362.7) | <0.001 | 632.8 (427.7–781.6) | 305.6 (236.9–354.6) | <0.001 |
| Dietary Intake | ||||||
| Energy intake (kcal/day) | 3534.5 (3056.1–4454.9) | 4236.5 (3351.1–5881.4) | 0.29 | 3079.5 (1966.6–4611.7) | 3041.0 (2637.3–4252.6) | 0.95 |
| Fat intake (%) | 36.8 (33.6–42.1) | 40.2 (34.8–45.7) | 0.34 | 35.1 (25.6–41.2) | 42.0 (34.0–47.2) | 0.03 |
| Vitamin B12 (mg/day) | 8.9 (6.4–12.4) | 8.9 (5.1–17.4) | 0.94 | 6.9 (4.1–15.5) | 6.9 (4.7–12.3) | 0.90 |
| Dairy (gm/1000 kcal) | 81.5 (53.1–132.1) | 75.7 (39.8–93.9) | 0.29 | 115.3 (32.7–201.2) | 99.3 (57.0–129.8) | 0.91 |
| Fruit (gm/1000 kcal) | 124.3 (72.4–197.2) | 98.9 (67.6–202.0) | 0.56 | 164.0 (45.9–231.8) | 70.2 (47.1–127.3) | 0.18 |
| Vegetables (gm/1000 kcal) | 194.9 (134.0–220.6) | 286.0 (177.4–310.5) | 0.05 | 132.7 (89.7–271.1) | 164.3 (124.6–241.3) | 0.51 |
| Egg (gm/day) | 34.9 (10.0–72.8) | 24.4 (12.0–42.5) | 0.43 | 25.4 (4.0–68.2) | 51.6 (8.4–94.3) | 0.25 |
| Chicken (gm/day) | 101.4 (56.7–132.8) | 64.3 (33.7–119.8) | 0.50 | 70.1 (39.1–114.0) | 50.0 (10.8–104.8) | 0.15 |
| Red meat (gm/day) | 51.7 (34.2–72.7) | 64.3 (40.9–96.4) | 0.27 | 26.0 (18.5–37.2) | 29.6 (6.8–52.9) | 0.53 |
| Variables | Normal | Obese | ||||
|---|---|---|---|---|---|---|
| High Serum Vitamin B12 (>404.0 pg/mL) | Low Serum Vitamin B12 (<404.0 pg/mL) | p-Value | High Serum Vitamin B12 (>404.0 pg/mL) | Low Serum Vitamin B12 (<404.0 pg/mL) | p-Value | |
| Firmicutes | 0.21 (0.17–0.30) | 0.20 (0.16–0.22) | 0.59 | 0.21 (0.16–0.27) | 0.20 (0.13–0.30) | 0.70 |
| Blautia wexlerae | 0.01 (0.002–0.01) | 0.01 (0.002–0.01) | 0.38 | 0.01 (0.003–0.01) | 0.01 (0.004–0.01) | 0.78 |
| Flavonifractor plautii | 0.001 (0.0003–0.001) | 0.001 (0.001–0.001) | 0.59 | 0.001 (0.0003–0.001) | 0.001 (0.0004–0.002) | 0.43 |
| Clostridium bolteae | 0.001 (0.0000–0.0006) | 0.0004 (0.00–0.001) | 0.80 | 0.00 (0.00–0.0008) | 0.0002 (0.00–0.001) | 0.82 |
| Faecalibacterium prausnitzii | 0.02 (0.01–0.03) | 0.02 (0.01–0.03) | 0.63 | 0.018 (0.02–0.03) | 0.016 (0.01–0.03) | 0.19 |
| Lactobacillus acidophilus | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.71 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.73 |
| Clostridioides difficile | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.40 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.95 |
| Bacteroidetes | 0.73 (0.55–0.79) | 0.74 (0.69–0.77) | 0.95 | 0.75 (0.66–0.81) | 0.74 (0.67–0.83) | 0.78 |
| Bacteroides “unspecified species” | 0.001 (0.0003–0.003) | 0.002 (0.0004–0.005) | 0.31 | 0.004 (0.001–0.004) | 0.002 (0.0003–0.01) | 0.57 |
| Bacteroides faecichinchillae | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.91 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.73 |
| Bacteroides thetaiotaomicron | 0.01 (0.004–0.01) | 0.01 (0.003–0.02) | 0.74 | 0.01 (0.004–0.01) | 0.01 (0.003–0.01) | 0.82 |
| Bacteroides uniformis | 0.07 (0.03–0.09) | 0.08 (0.05–0.08) | 0.59 | 0.08 (0.04–0.13) | 0.07 (0.04–0.09) | 0.78 |
| Actinobacteria | 0.03 (0.02–0.06) | 0.05 (0.03–0.07) | 0.28 | 0.02 (0.003–0.05) | 0.02 (0.02–0.04) | 0.87 |
| Bifidobacterium adolescentis | 0.005 (0.00–0.02) | 0.001 (0.00–0.02) | 0.56 | 0.003 (0.0003–0.01) | 0.001 (0.00–0.01) | 0.70 |
| Bifidobacterium kashiwanohense | 0.00 (0.00–0.0003) | 0.00 (0.00–0.003) | 0.47 | 0.00 (0.00–0.001) | 0.00 (0.00–0.0004) | 0.95 |
| Bifidobacterium longum | 0.007 (0.002–0.01) | 0.01 (0.001–0.01) | 0.97 | 0.003 (0.00–0.009) | 0.01 (0.001–0.01) | 0.63 |
| Bifidobacterium merycicum | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.44 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.82 |
| Bifidobacterium pseudocatenulatum | 0.0002 (0.00–0.002) | 0.001 (0.00–0.005) | 0.22 | 0.0003 (0.00–0.001) | 0.0003 (0.00–0.003) | 0.66 |
| Verrucomicrobia | 0.001 (0.00–0.0069) | 0.0002 (0.00–0.001) | 0.16 | 0.001 (0.00–0.01) | 0.0002 (0.00–0.001) | 0.80 |
| Akkermansia muciniphila | 0.002 (0.00–0.008) | 0.0002 (0.00–0.001) | 0.12 | 0.001 (0.00–0.01) | 0.0002 (0.00–0.001) | 0.51 |
| Proteobacteria | 0.02 (0.01–0.02) | 0.01 (0.01–0.02) | 0.23 | 0.01 (0.01–0.02) | 0.10 (0.01–0.02) | 0.43 |
| Synergistetes | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.99 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.68 |
| Fusobacteria | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 1.00 | 0.00 (0.00–0.00) | 0.00 (0.00–0.00) | 0.73 |
| Bacteria “unspecified phylum” | 0.00 (0.00–0.0004) | 0.0002 (0.00–0.001) | 0.51 | 0.0004 (0.00–0.001) | 0.0002 (0.00–0.001) | 0.75 |
| F/B (ratio) | 0.28 (0.21–0.57) | 0.27 (0.20–0.31) | 0.67 | 0.27 (0.20–0.41) | 0.25 (0.16–0.45) | 0.73 |
| Variables | Vitamin B12 Intake | R | p-Value * | |
|---|---|---|---|---|
| Gut Microbiota | Total participants | Bifidobacterium kashiwanohense species | −0.23 | 0.03 |
| Blautia wexlerae | −0.24 | 0.02 | ||
| Obese participants | Akkermansia muciniphila species | −0.32 | 0.03 | |
| Verrucomicrobia phylum | −0.30 | 0.04 | ||
| Bacteroides “unspecified species” | 0.30 | 0.04 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Musharaf, S.; Aljuraiban, G.S.; Al-Ajllan, L.; Al-Khaldi, N.; Aljazairy, E.A.; Hussain, S.D.; Alnaami, A.M.; Sabico, S.; Al-Daghri, N. Vitamin B12 Status and Gut Microbiota among Saudi Females with Obesity. Foods 2022, 11, 4007. https://doi.org/10.3390/foods11244007
Al-Musharaf S, Aljuraiban GS, Al-Ajllan L, Al-Khaldi N, Aljazairy EA, Hussain SD, Alnaami AM, Sabico S, Al-Daghri N. Vitamin B12 Status and Gut Microbiota among Saudi Females with Obesity. Foods. 2022; 11(24):4007. https://doi.org/10.3390/foods11244007
Chicago/Turabian StyleAl-Musharaf, Sara, Ghadeer S. Aljuraiban, Lama Al-Ajllan, Noura Al-Khaldi, Esra’a A. Aljazairy, Syed Danish Hussain, Abdullah M. Alnaami, Shaun Sabico, and Nasser Al-Daghri. 2022. "Vitamin B12 Status and Gut Microbiota among Saudi Females with Obesity" Foods 11, no. 24: 4007. https://doi.org/10.3390/foods11244007
APA StyleAl-Musharaf, S., Aljuraiban, G. S., Al-Ajllan, L., Al-Khaldi, N., Aljazairy, E. A., Hussain, S. D., Alnaami, A. M., Sabico, S., & Al-Daghri, N. (2022). Vitamin B12 Status and Gut Microbiota among Saudi Females with Obesity. Foods, 11(24), 4007. https://doi.org/10.3390/foods11244007








