The Use of Prebiotics from Pregnancy and Its Complications: Health for Mother and Offspring—A Narrative Review
Abstract
1. Introduction
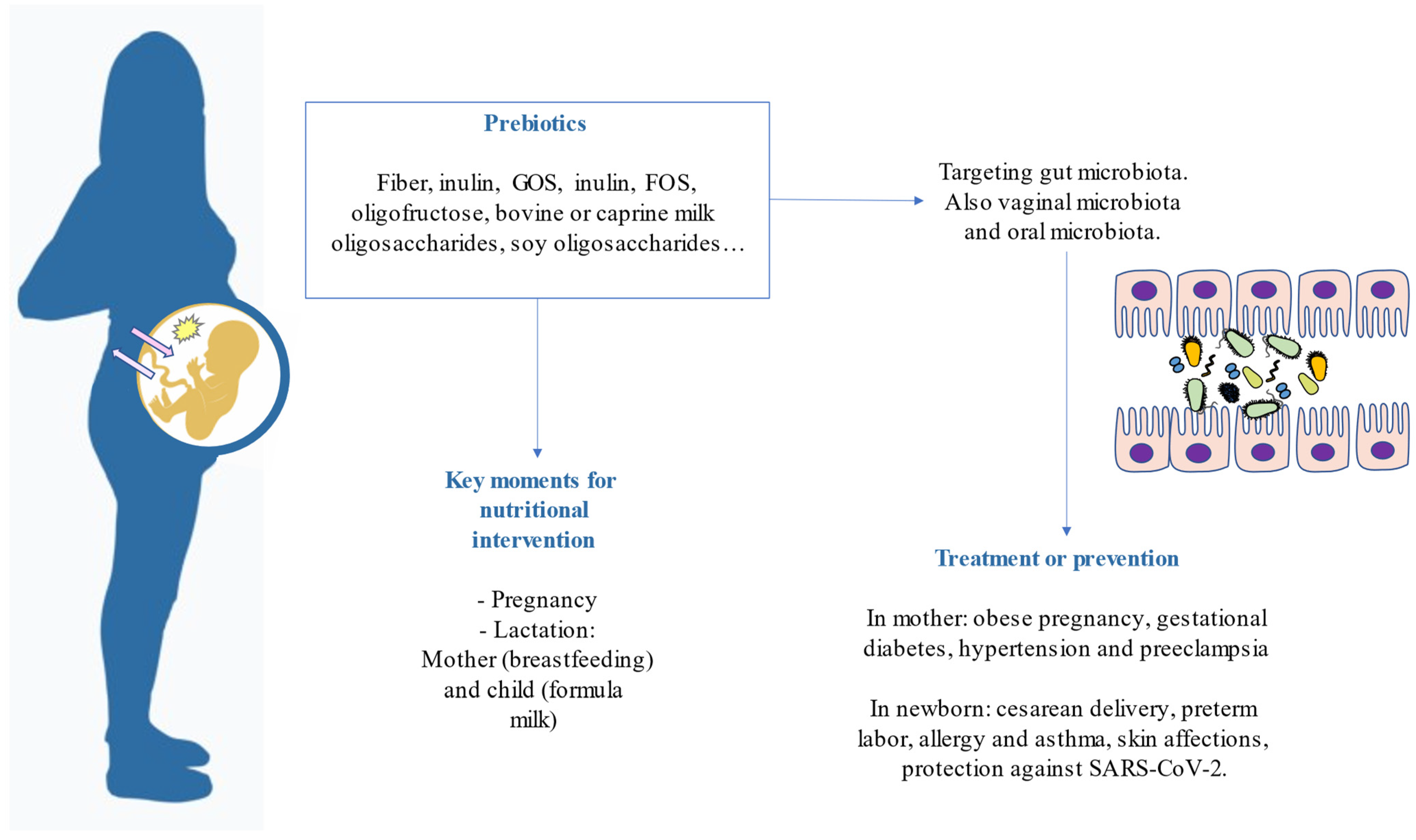
2. What Are Prebiotics and What Types Are There?
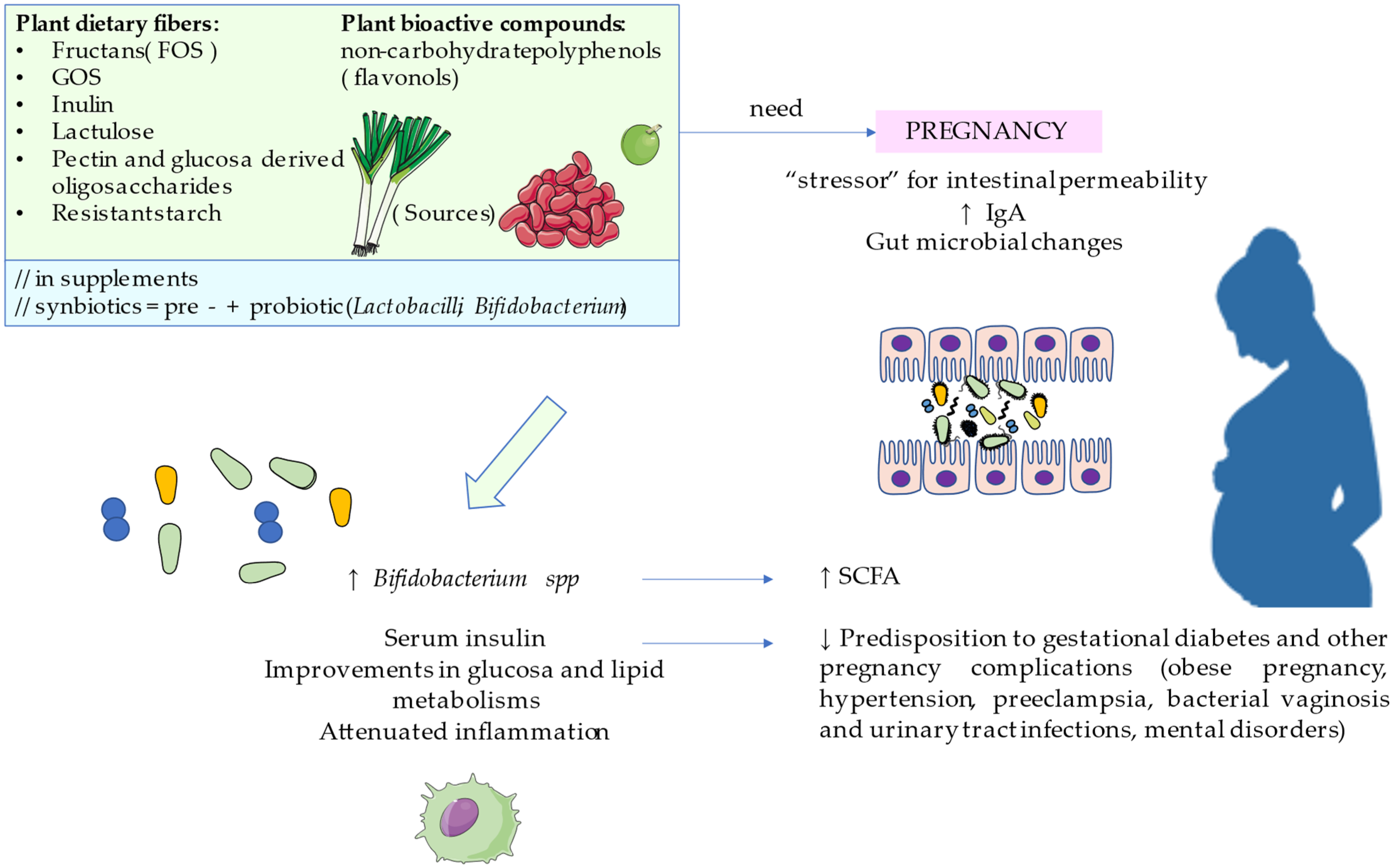
3. Prebiotics for Healthy Pregnant Women
4. Prebiotics for Pregnancy Complications
4.1. Obese Pregnancy
4.2. Gestational Diabetes
4.3. Hypertension and Preeclampsia
4.4. Bacterial Vaginosis
4.5. Perinatal Mental Health
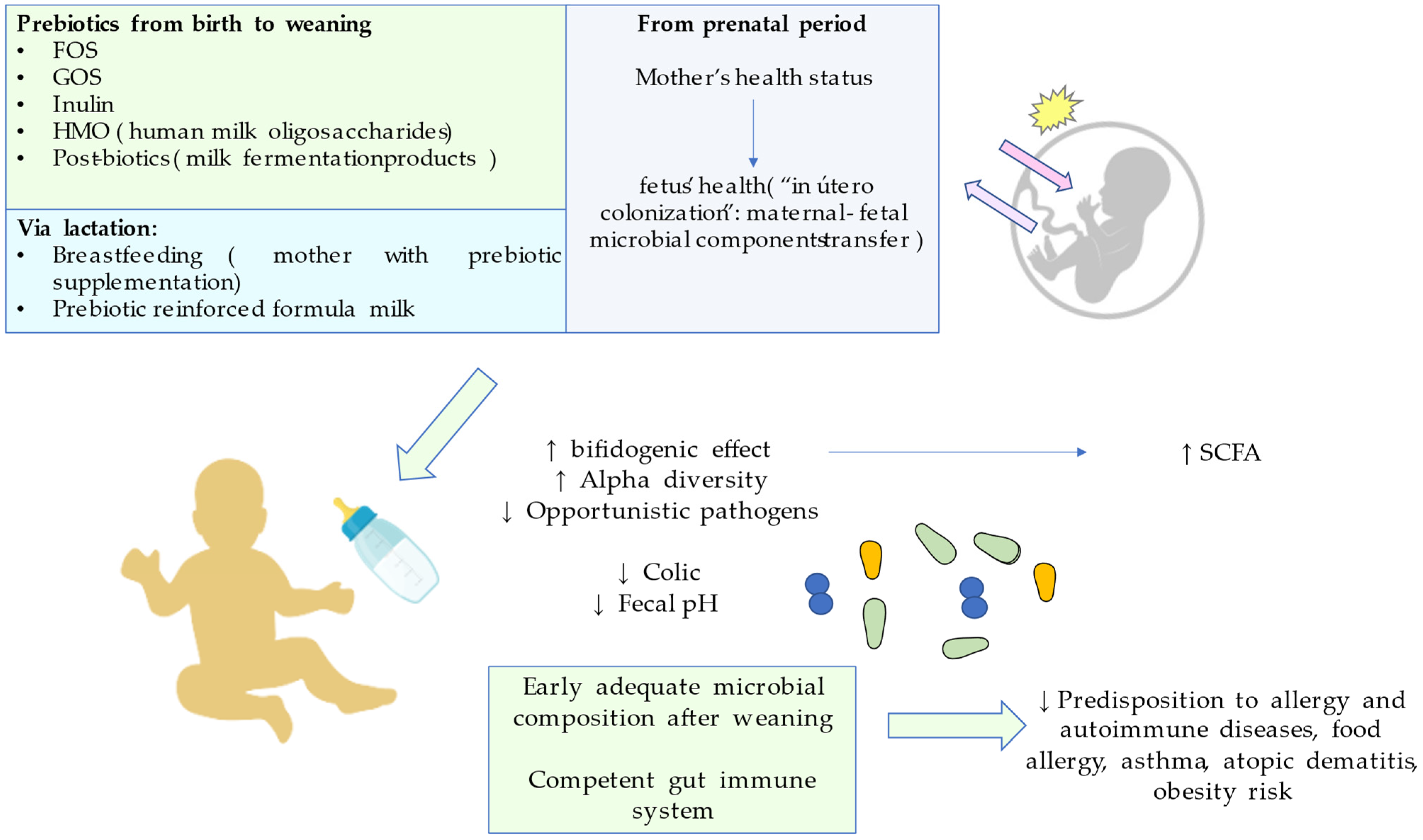
5. Prevention of Pediatric Disorders
5.1. Prebiotics in the Window of Opportunity
5.2. Caesarean Delivery
5.3. Preterm Labor
5.4. Asthma and Allergy
5.5. Skin Maladies
5.6. Protection against SARS-CoV-2
6. Conclusions, Limitations, and Reflections
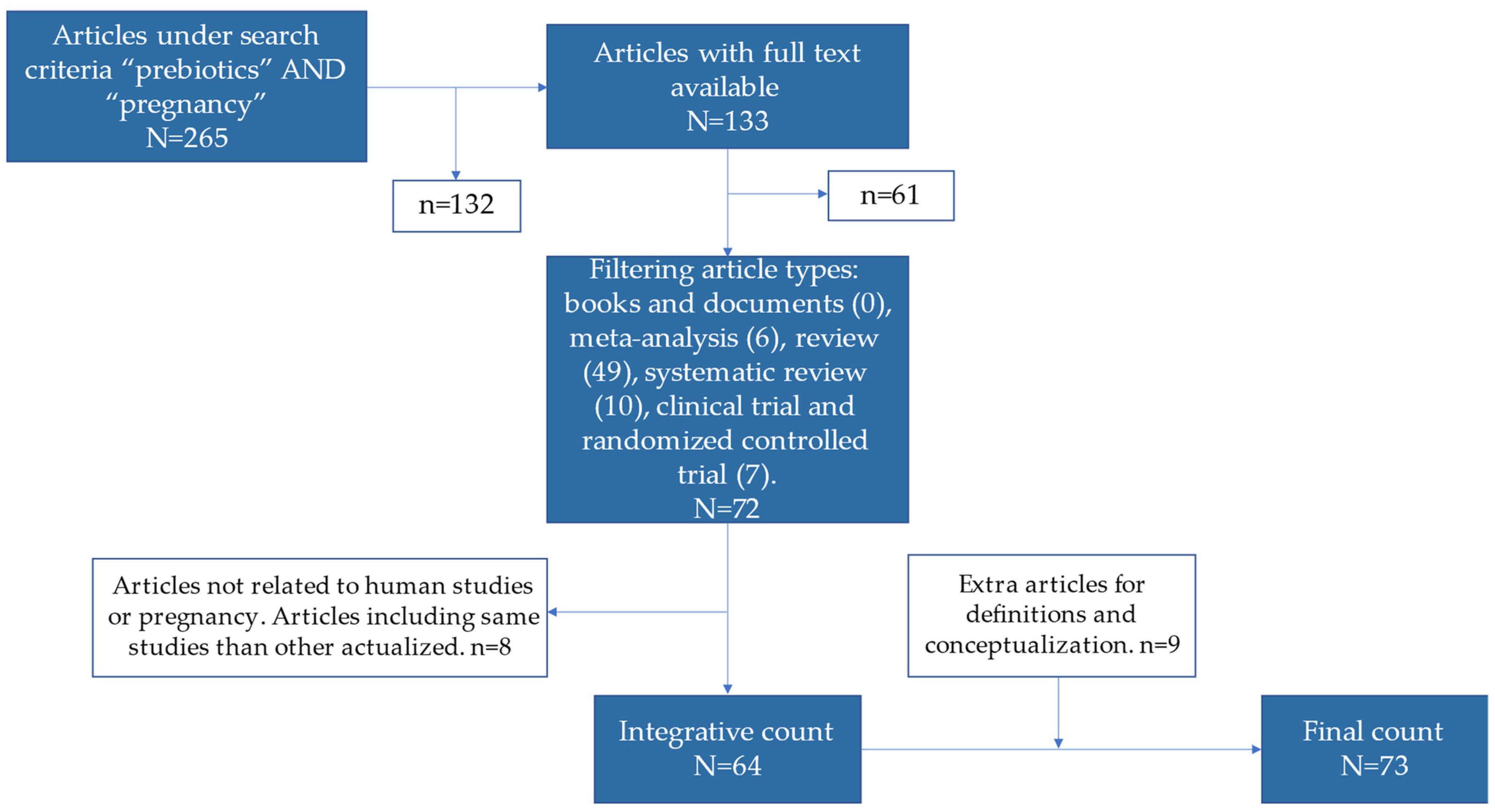
7. Methods of Searching
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Plećaš, D.; Plešinac, S.; Vučinić, O.K. Nutrition in pregnancy: Basic principles and recommendations. Srp. Arh. Celok. Lek. 2014, 142, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Gresham, E.; Bisquera, A.; Byles, J.E.; Hure, A.J. Effects of dietary interventions on pregnancy outcomes: A systematic review and meta-analysis. Matern. Child Nutr. 2016, 12, 5–23. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Tan, X.; Arnold, C.D.; Sitthideth, D.; Kounnavong, S.; Hess, S.Y. Traditional prenatal and postpartum food restrictions among women in northern Lao PDR. Matern. Child Nutr. 2022, 18, e13273. [Google Scholar] [CrossRef]
- Teh, K.; Quek, I.P.; Tang, W.E. Postpartum dietary and physical activity-related beliefs and behaviors among women with recent gestational diabetes mellitus: A qualitative study from Singapore. BMC Pregnancy Childbirth 2021, 21, 612. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Charlton, K.E.; Yeatman, H. Nutrition advice during pregnancy: Do women receive it and can health professionals provide it? Matern. Child Health J. 2014, 18, 2465–2478. [Google Scholar] [CrossRef]
- Homayouni, A.; Bagheri, N.; Mohammad-Alizadeh-Charandabi, S.; Kashani, N.; Mobaraki-Asl, N.; Mirghafurvand, M.; Asgharian, H.; Ansari, F.; Pourjafar, H. Prevention of Gestational Diabetes Mellitus (GDM) and Probiotics: Mechanism of Action: A Review. Curr. Diabetes Rev. 2020, 16, 538–545. [Google Scholar] [CrossRef]
- Venter, C.; Agostoni, C.; Arshad, S.H.; Ben-Abdallah, M.; Du Toit, G.; Fleischer, D.M.; Greenhawt, M.; Glueck, D.H.; Groetch, M.; Lunjani, N.; et al. Dietary factors during pregnancy and atopic outcomes in childhood: A systematic review from the European Academy of Allergy and Clinical Immunology. Pediatr. Allergy Immunol. 2020, 31, 889–912. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Gibson, G.R.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar] [CrossRef]
- Gibson, G.R.; Scott, K.P.; Rastall, R.A.; Tuohy, K.M.; Hotchkiss, A.; Dubert-Ferrandon, A.; Gareau, M.; Murphy, E.F.; Saulnier, D.; Loh, G.; et al. Dietary prebiotics: Current status and new definition. Food Sci. Technol. Bull. Funct. Foods 2010, 7, 1–19. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Jarde, A.; Lewis-Mikhael, A.M.; Moayyedi, P.; Stearns, J.C.; Collins, S.M.; Beyene, J.; McDonald, S.D. Pregnancy outcomes in women taking probiotics or prebiotics: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2018, 18, 14. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, P.O.; Bindels, L.B.; Saulnier, D.M.; Reid, G.; Nova, E.; Holmgren, K.; O’Toole, P.W.; Bunn, J.; Delzenne, N.; Scott, K.P. Can prebiotics and probiotics improve therapeutic outcomes for undernourished individuals? Gut Microbes 2014, 5, 74–82. [Google Scholar] [CrossRef]
- Camilleri, M. Human Intestinal Barrier: Effects of Stressors, Diet, Prebiotics, and Probiotics. Clin. Transl. Gastroenterol. 2021, 12, e00308. [Google Scholar] [CrossRef]
- Suárez, J.E. [Autochthonous microbiota, probiotics and prebiotics]. Nutr. Hosp. 2015, 31 (Suppl. 1), 3–9. [Google Scholar] [CrossRef]
- Jinno, S.; Toshimitsu, T.; Nakamura, Y.; Kubota, T.; Igoshi, Y.; Ozawa, N.; Suzuki, S.; Nakano, T.; Morita, Y.; Arima, T.; et al. Maternal Prebiotic Ingestion Increased the Number of Fecal Bifidobacteria in Pregnant Women but Not in Their Neonates Aged One Month. Nutrients 2017, 9, 196. [Google Scholar] [CrossRef]
- Wibowo, N.; Bardosono, S.; Irwinda, R. Effects of Bifidobacterium animalis lactis HN019 (DR10TM), inulin, and micronutrient fortified milk on faecal DR10TM, immune markers, and maternal micronutrients among Indonesian pregnant women. Asia Pac. J. Clin. Nutr. 2016, 25, S102–S110. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Asemi, Z. Effects of synbiotic food consumption on glycemic status and serum hs-CRP in pregnant women: A randomized controlled clinical trial. Hormones 2014, 13, 398–406. [Google Scholar] [CrossRef]
- Sheyholislami, H.; Connor, K.L. Are Probiotics and Prebiotics Safe for Use during Pregnancy and Lactation? A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 2382. [Google Scholar] [CrossRef] [PubMed]
- Sohn, K.; Underwood, M.A. Prenatal and postnatal administration of prebiotics and probiotics. Semin. Fetal Neonatal Med. 2017, 22, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Wiedmer, E.B.; Herter-Aeberli, I. The Potential of Prebiotic and Probiotic Supplementation During Obese Pregnancy to Improve Maternal and Offspring’s Metabolic Health and Reduce Obesity Risk-A Narrative Review. Front. Nutr. 2022, 9, 819882. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Xiao, X. The role of gut microbiota in the effects of maternal obesity during pregnancy on offspring metabolism. Biosci. Rep. 2018, 38, BSR20171234. [Google Scholar] [CrossRef]
- Rubini, E.; Schenkelaars, N.; Rousian, M.; Sinclair, K.D.; Wekema, L.; Faas, M.M.; Steegers-Theunissen, R.P.M.; Schoenmakers, S. Maternal obesity during pregnancy leads to derangements in one-carbon metabolism and the gut microbiota: Implications for fetal development and offspring wellbeing. Am. J. Obstet. Gynecol. 2022, 227, 392–400. [Google Scholar] [CrossRef]
- Plows, J.F.; Stanley, J.L.; Baker, P.N.; Reynolds, C.M.; Vickers, M.H. The Pathophysiology of Gestational Diabetes Mellitus. Int. J. Mol. Sci. 2018, 19, 3342. [Google Scholar] [CrossRef]
- Fei, B.B.; Ling, L.; Hua, C.; Ren, S.Y. Effects of soybean oligosaccharides on antioxidant enzyme activities and insulin resistance in pregnant women with gestational diabetes mellitus. Food Chem. 2014, 158, 429–432. [Google Scholar] [CrossRef]
- De Mendonça, E.L.S.S.; Fragoso, M.B.T.; de Oliveira, J.M.; Xavier, J.A.; Goulart, M.O.F.; de Oliveira, A.C.M. Gestational Diabetes Mellitus: The Crosslink among Inflammation, Nitroxidative Stress, Intestinal Microbiota and Alternative Therapies. Antioxidants 2022, 11, 129. [Google Scholar] [CrossRef]
- Zhou, L.; Ding, C.; Wu, J.; Chen, X.; Ng, D.M.; Wang, H.; Zhang, Y.; Shi, N. Probiotics and synbiotics show clinical efficacy in treating gestational diabetes mellitus: A meta-analysis. Prim. Care Diabetes 2021, 15, 937–947. [Google Scholar] [CrossRef]
- Çetinkaya Özdemir, S.; Küçüktürkmen Paşa, B.; Metin, T.; Dinçer, B.; Sert, H. The effect of probiotic and synbiotic use on glycemic control in women with gestational diabetes: A systematic review and meta-analysis. Diabetes Res. Clin. Pract. 2022, 194, 110162. [Google Scholar] [CrossRef]
- Łagowska, K.; Malinowska, A.M.; Zawieja, B.; Zawieja, E. Improvement of glucose metabolism in pregnant women through probiotic supplementation depends on gestational diabetes status: Meta-analysis. Sci. Rep. 2020, 10, 17796. [Google Scholar] [CrossRef] [PubMed]
- Ishimwe, J.A. Maternal microbiome in preeclampsia pathophysiology and implications on offspring health. Physiol. Rep. 2021, 9, e14875. [Google Scholar] [CrossRef] [PubMed]
- Willmott, T.; McBain, A.J.; Humphreys, G.J.; Myers, J.; Cottrell, E. Does the Oral Microbiome Play a Role in Hypertensive Pregnancies? Front. Cell. Infect. Microbiol. 2020, 10, 389. [Google Scholar] [CrossRef] [PubMed]
- Movaghar, R.; Farshbaf-Khalili, A.; Hajizade, K.; MirzaRezaei, M.E.; Shahnazi, M. The Effect of Probiotics or Synbiotics on the Hypertensive Disorders of Pregnant Women with Gestational Diabetes: A Systematic Review and Meta-analysis. J. Caring Sci. 2022, 11, 94–104. [Google Scholar] [CrossRef]
- Bustamante, M.; Oomah, B.D.; Oliveira, W.P.; Burgos-Díaz, C.; Rubilar, M.; Shene, C. Probiotics and prebiotics potential for the care of skin, female urogenital tract, and respiratory tract. Folia Microbiol. 2020, 65, 245–264. [Google Scholar] [CrossRef]
- Afifirad, R.; Darb Emamie, A.; Golmoradi Zadeh, R.; Asadollahi, P.; Ghanavati, R.; Darbandi, A. Effects of Pro/Prebiotics Alone over Pro/Prebiotics Combined with Conventional Antibiotic Therapy to Treat Bacterial Vaginosis: A Systematic Review. Int. J. Clin. Pract. 2022, 2022, 4774783. [Google Scholar] [CrossRef]
- Desai, V.; Kozyrskyj, A.L.; Lau, S.; Sanni, O.; Dennett, L.; Walter, J.; Ospina, M.B. Effectiveness of Probiotic, Prebiotic, and Synbiotic Supplementation to Improve Perinatal Mental Health in Mothers: A Systematic Review and Meta-Analysis. Front. Psychiatry 2021, 12, 622181. [Google Scholar] [CrossRef]
- Rackers, H.S.; Thomas, S.; Williamson, K.; Posey, R.; Kimmel, M.C. Emerging literature in the Microbiota-Brain Axis and Perinatal Mood and Anxiety Disorders. Psychoneuroendocrinology 2018, 95, 86–96. [Google Scholar] [CrossRef]
- Beluska-Turkan, K.; Korczak, R.; Hartell, B.; Moskal, K.; Maukonen, J.; Alexander, D.E.; Salem, N.; Harkness, L.; Ayad, W.; Szaro, J.; et al. Nutritional Gaps and Supplementation in the First 1000 Days. Nutrients 2019, 11, 2891. [Google Scholar] [CrossRef]
- Firmansyah, A.; Chongviriyaphan, N.; Dillon, D.H.S.; Khan, N.C.; Morita, T.; Tontisirin, K.; Tuyen, L.D.; Wang, W.; Bindels, J.; Deurenberg, P.; et al. Fructans in the first 1000 days of life and beyond, and for pregnancy. Asia Pac. J. Clin. Nutr. 2016, 25, 652–675. [Google Scholar] [CrossRef]
- Zaidi, A.Z.; Moore, S.E.; Okala, S.G. Impact of Maternal Nutritional Supplementation during Pregnancy and Lactation on the Infant Gut or Breastmilk Microbiota: A Systematic Review. Nutrients 2021, 13, 1137. [Google Scholar] [CrossRef] [PubMed]
- Castanys-Muñoz, E.; Martin, M.J.; Vazquez, E. Building a beneficial microbiome from birth. Adv. Nutr. 2016, 7, 323–330. [Google Scholar] [CrossRef]
- Ong, T.G.; Gordon, M.; Banks, S.S.C.; Thomas, M.R.; Akobeng, A.K. Probiotics to prevent infantile colic. Cochrane Database Syst. Rev. 2019, 2019, CD012473. [Google Scholar] [CrossRef] [PubMed]
- Sprockett, D.; Fukami, T.; Relman, D.A. Role of priority effects in the early-life assembly of the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, V.; Indrio, F.; Verduci, E.; Calcaterra, V.; Pop, T.L.; Mari, A.; Zuccotti, G.V.; Cokugras, F.C.; Pettoello-Mantovani, M.; Goulet, O. Term Infant Formulas Influencing Gut Microbiota: An Overview. Nutrients 2021, 13, 4200. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.C.; Dinsmoor, A.M.; Wang, M.; Donovan, S.M. Microbiome Composition in Pediatric Populations from Birth to Adolescence: Impact of Diet and Prebiotic and Probiotic Interventions. Dig. Dis. Sci. 2020, 65, 706–722. [Google Scholar] [CrossRef] [PubMed]
- Allan Walker, W. Initial intestinal colonization in the human infant and immune homeostasis. Ann. Nutr. Metab. 2013, 63 (Suppl. 2), 8–15. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.J.; Yeung, C.Y. Recent advance in infant nutrition: Human milk oligosaccharides. Pediatr. Neonatol. 2021, 62, 347–353. [Google Scholar] [CrossRef]
- Salvini, F.; Riva, E.; Salvatici, E.; Boehm, G.; Jelinek, J.; Banderali, G.; Giovannini, M. A specific prebiotic mixture added to starting infant formula has long-lasting bifidogenic effects. J. Nutr. 2011, 141, 1335–1339. [Google Scholar] [CrossRef]
- Del Toca, M.C.; Burgos, F.; Fernández, A.; Giglio, N.; Orsi, M.; Sosa, P.; Tabacco, O.; Ursino, F.; Ussher, F.; Vinderola, G. Gut ecosystem during infancy: The role of “biotics”. Arch. Argent. Pediatr. 2020, 118, 278–285. [Google Scholar] [CrossRef]
- Luoto, R.; Collado, M.C.; Salminen, S.; Isolauri, E. Reshaping the gut microbiota at an early age: Functional impact on obesity risk? Ann. Nutr. Metab. 2013, 63 (Suppl. 2), 17–26. [Google Scholar] [CrossRef]
- Arboleya, S.; Suárez, M.; Fernández, N.; Mantecón, L.; Solís, G.; Gueimonde, M.; De Los Reyes-Gavilán, C.G. C-section and the Neonatal Gut Microbiome Acquisition: Consequences for Future Health. Ann. Nutr. Metab. 2018, 73 (Suppl. 3), 17–23. [Google Scholar] [CrossRef] [PubMed]
- Pinto Coelho, G.D.; Arial Ayres, L.F.; Barreto, D.S.; Henriques, B.D.; Cardoso Prado, M.R.M.; Dos Passos, C.M. Acquisition of microbiota according to the type of birth: An integrative review. Rev. Lat. Am. Enfermagem 2021, 29, e3446. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Forster, S.C.; Tsaliki, E.; Vervier, K.; Strang, A.; Simpson, N.; Kumar, N.; Stares, M.D.; Rodger, A.; Brocklehurst, P.; et al. Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature 2019, 574, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Bello, M.G.; De Jesus-Laboy, K.M.; Shen, N.; Cox, L.M.; Amir, A.; Gonzalez, A.; Bokulich, N.A.; Song, S.J.; Hoashi, M.; Rivera-Vinas, J.I.; et al. Partial restoration of the microbiota of cesarean-born infants via vaginal microbial transfer. Nat. Med. 2016, 22, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Francavilla, R.; Cristofori, F.; Tripaldi, M.E.; Indrio, F. Intervention for Dysbiosis in Children Born by C-Section. Ann. Nutr. Metab. 2018, 73 (Suppl. 3), 33–39. [Google Scholar] [CrossRef]
- Lista, G.; Meneghin, F.; Bresesti, I.; Cavigioli, F. Functional nutrients in infants born by vaginal delivery or Cesarean section. Pediatr. Med. Chir. 2017, 39, 124–127. [Google Scholar] [CrossRef]
- Moya-Pérez, A.; Luczynski, P.; Renes, I.B.; Wang, S.; Borre, Y.; Ryan, C.A.; Knol, J.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Intervention strategies for cesarean section-induced alterations in the microbiota-gut-brain axis. Nutr. Rev. 2017, 75, 225–240. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Cano-Ibáñez, N.; Pinto-Gallardo, M.; Amezcua-Prieto, C. The Impact of Probiotics, Prebiotics, and Synbiotics during Pregnancy or Lactation on the Intestinal Microbiota of Children Born by Cesarean Section: A Systematic Review. Nutrients 2022, 14, 341. [Google Scholar] [CrossRef]
- Buffet-Bataillon, S.; Bellanger, A.; Boudry, G.; Gangneux, J.P.; Yverneau, M.; Beuchée, A.; Blat, S.; Le Huërou-Luron, I. New Insights Into Microbiota Modulation-Based Nutritional Interventions for Neurodevelopmental Outcomes in Preterm Infants. Front. Microbiol. 2021, 12, 676622. [Google Scholar] [CrossRef]
- Gray, L.E.K.; O’Hely, M.; Ranganathan, S.; Sly, P.D.; Vuillermin, P. The Maternal Diet, Gut Bacteria, and Bacterial Metabolites during Pregnancy Influence Offspring Asthma. Front. Immunol. 2017, 8, 365. [Google Scholar] [CrossRef] [PubMed]
- Van Esch, B.C.A.M.; Porbahaie, M.; Abbring, S.; Garssen, J.; Potaczek, D.P.; Savelkoul, H.F.J.; van Neerven, R.J.J. The Impact of Milk and Its Components on Epigenetic Programming of Immune Function in Early Life and Beyond: Implications for Allergy and Asthma. Front. Immunol. 2020, 11, 2141. [Google Scholar] [CrossRef] [PubMed]
- Trambusti, I.; Nuzzi, G.; Costagliola, G.; Verduci, E.; D’Auria, E.; Peroni, D.G.; Comberiati, P. Dietary Interventions and Nutritional Factors in the Prevention of Pediatric Asthma. Front. Pediatr. 2020, 8, 480. [Google Scholar] [CrossRef]
- Forsberg, A.; West, C.E.; Prescott, S.L.; Jenmalm, M.C. Pre- and probiotics for allergy prevention: Time to revisit recommendations? Clin. Exp. Allergy 2016, 46, 1506–1521. [Google Scholar] [CrossRef]
- Gern, J.E. Promising candidates for allergy prevention. J. Allergy Clin. Immunol. 2015, 136, 23–28. [Google Scholar] [CrossRef]
- D’Auria, E.; Peroni, D.G.; Sartorio, M.U.A.; Verduci, E.; Zuccotti, G.V.; Venter, C. The Role of Diet Diversity and Diet Indices on Allergy Outcomes. Front. Pediatr. 2020, 8, 545. [Google Scholar] [CrossRef] [PubMed]
- Zepeda-Ortega, B.; Goh, A.; Xepapadaki, P.; Sprikkelman, A.; Nicolaou, N.; Hernandez, R.E.H.; Latiff, A.H.A.; Yat, M.T.; Diab, M.; Al Hussaini, B.; et al. Strategies and Future Opportunities for the Prevention, Diagnosis, and Management of Cow Milk Allergy. Front. Immunol. 2021, 12, 608372. [Google Scholar] [CrossRef] [PubMed]
- Trikamjee, T.; Comberiati, P.; D’Auria, E.; Peroni, D.; Zuccotti, G.V. Nutritional Factors in the Prevention of Atopic Dermatitis in Children. Front. Pediatr. 2021, 8, 577413. [Google Scholar] [CrossRef]
- Kantor, R.; Silverberg, J.I. Environmental risk factors and their role in the management of atopic dermatitis. Expert Rev. Clin. Immunol. 2017, 13, 15–26. [Google Scholar] [CrossRef]
- Dang, D.; Zhou, W.; Lun, Z.J.; Mu, X.; Wang, D.X.; Wu, H. Meta-analysis of probiotics and/or prebiotics for the prevention of eczema. J. Int. Med. Res. 2013, 41, 1426–1436. [Google Scholar] [CrossRef]
- Rusu, E.; Enache, G.; Cursaru, R.; Alexescu, A.; Radu, R.; Onila, O.; Cavallioti, T.; Rusu, F.; Posea, M.; Jinga, M.; et al. Prebiotics and probiotics in atopic dermatitis. Exp. Ther. Med. 2019, 18, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Larsen, V.; Ierodiakonou, D.; Jarrold, K.; Cunha, S.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Ruparelia, A.; Devani, P.; Trivella, M.; et al. Diet during pregnancy and infancy and risk of allergic or autoimmune disease: A systematic review and meta-analysis. PLoS Med. 2018, 15, e1002507. [Google Scholar] [CrossRef] [PubMed]
- Grot, M.; Krupa-Kotara, K.; Wypych-ślusarska, A.; Grajek, M.; Białek-Dratwa, A. The Concept of Intrauterine Programming and the Development of the Neonatal Microbiome in the Prevention of SARS-CoV-2 Infection. Nutrients 2022, 14, 1702. [Google Scholar] [CrossRef] [PubMed]
| Condition | Evidence from Clinical Trials | References | |
|---|---|---|---|
| Pregnancy and its complications | Healthy pregnancy | In maternal intake of FOS during the third trimester, there was a significant increase in fecal Bifidobacterium spp. and Bifidobacterium longum in the intervention group at the end of pregnancy. Administration of milk fortified with prebiotic, probiotic, DHA and micronutrients, observing notable increases in fecal concentration of the organism used as probiotic, alleging that probiotic colonization is successful when combined with prebiotics and other micronutrients. Comparing pregnant women for 9 weeks, synbiotics containing Lactobacillus sporogenes and inulin showed significant differences with lower serum insulin levels in the intervention group versus the control group. Regarding safety of pro- and prebiotics use in pregnancy and lactation, although only in some cases were changes in stool consistency noticed, but did not have serious effects for the mother or the infant’s health. | [18,19,20,21] |
| Gestational diabetes (GDM) | Intervention with synbiotics has shown significant improvements on glucose and lipid metabolism, insulin resistance, as well as anti-inflammatory and antioxidant ability in diet controlled GDM patients, reducing the risk of fetal hyperbilirubinemia, fetal macrosomia, and limiting newborn weight. However, to optimize the results, more characterization about combinations with certain probiotics strains should be considered. | [29,30,31] | |
| Hypertension and preeclampsia | The safety of pro- and prebiotic use stimulates gut-derived metabolites, such as butyrate, that attenuate inflammation. There were no notable benefits from the administration of probiotics or synbiotics in women with hypertensive disorders or GDM, although due to the limited number of studies available, additional efforts are required. | [32,33] | |
| Bacterial vaginosis | Synbiotics consumption seems to effectively prevent recurrent urinary tract infections in women. In the case of bacterial vaginosis, they suggest that a combo of probiotics and prebiotics should be applied instead of using antibiotics, which is risky for a pregnant woman. Furthermore, pre/probiotic regimens seem to have even higher cure rates than antibiotics. | [35,36] | |
| Perinatal mental health | Limited evidence of a lower incidence of anxiety and depressive symptoms in the perinatal period has been reported when supplementing with pro-, pre- and synbiotics during pregnancy. | [37,38] | |
| Fortified formula milk and breastfeeding) | Prevention in the window of opportunity | There are significant increases in fecal Bifidobacterium spp. when treating mothers with FOS, but no difference was observed in neonates aged 1 month. Longer term studies need to be undertaken. Short-term studies have observed reduced daily crying time when administering probiotics compared to placebo. The same studies defend the application of oligosaccharide prebiotics to promote the growth of beneficial bacteria as treatments for allergy or intolerance and for crying in babies with colic that are on formula. A premise from ecological theory says that microbial community development is affected by priority effects that determine species arrival and their behavior, which can be favorably modulated by pro- and prebiotics, so therapies targeting the gut microbiota are a potential choice. Different formulas of milk have reported an increase in bifidogenic bacteria and a decrease in opportunistic pathogens, in addition to a reduction of fecal pH, an increase in alpha diversity, and optimized calcium absorption. Human milk composition in prebiotics stimulate Bifidobacteria and Lactobacilli as well. However, when breast milk is not available or adequate, HMO fortified formulas seem to be the best option. A randomized controlled trial studied the effect of a specific prebiotic mixture administered from the day of birth on bifidobacterial and lactobacilli counts. An examination was performed at 3, 6 and 12 months of age, finding that the supplemented group had more fecal bifidobacterial and lactobacilli compared to placebo, these differences being maintained six months later without further supplementation. This kind of explorative study shows that prebiotics, even in the first days of life, are effective in establishing a competent gut immune system. | [18,43,44,46,47,48] |
| Pediatric disorders | Caesarean delivery | Stool samples from supplemented babies are softer and seem to be related to a lower level of pathogens. Nevertheless, the duration of supplementation to ensure a lasting beneficial effect is yet unknown, and another gap to close in clinical studies. A recent systematic review noted that the sooner the intervention during pregnancy and lactation after cesarean delivery, the better the effects that are achieved, especially Bifidobacterium colonization. Results were even more favorable in breastfed infants from supplemented mothers. The utilized formulas included GOS, FOS, or bovine milk-derived oligosaccharides, combined with probiotics from the genera Lactobacillus, Bifidobacterium, Propionibacterium, and Streptococcus. | [56,59] |
| Asthma and allergy | The use of a formula milk containing a mixture of prebiotic oligosaccharides was associated with the lower incidence for recurrent wheezing in 132 infants at risk of atopy, although supplementation of non-human neutral and acidic oligosaccharides during the neonatal period did not reduce the incidence of allergies, bronchial hyper-reactivity, or respiratory infections in 113 preterm infants. They found a meta-analysis of two studies with 249 infants, reporting a reduction in infant asthma or recurrent wheezing in prebiotic-treated infants, and a single study reporting a significant reduction of the risk of developing food allergies by the use of prebiotics. These results are due to their ability to induce the production of short-chain fatty acids, especially dietary fiber and oligosaccharides, leading to the activation of T regulatory cells and tolerance mechanisms. Despite these potential benefits, the available literature seems to indicate that the evidence for the supplementation of prebiotics for the prevention of allergies are not yet strong enough to make any clear recommendations. | [64,65,66] | |
| Skin maladies | Synbiotics consumption during pregnancy and lactation seems to decrease eczema incidence in offspring. Few clinical trials have noticed lower eczema and atopic dermatitis severity in unweaned babies. Studies related to maternal supplement intake while lactating compared to using prebiotic-enriched formula milk, seem to be a more proper approach to reduce eczema in ≤4 year-aged babies. | [70,71,72] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Montero, C.; Fraile-Martinez, O.; Rodriguez-Martín, S.; Saz, J.V.; Rodriguez, R.A.; Moreno, J.M.P.; Labarta, J.R.; García-Honduvilla, N.; Alvarez-Mon, M.; Bravo, C.; et al. The Use of Prebiotics from Pregnancy and Its Complications: Health for Mother and Offspring—A Narrative Review. Foods 2023, 12, 1148. https://doi.org/10.3390/foods12061148
García-Montero C, Fraile-Martinez O, Rodriguez-Martín S, Saz JV, Rodriguez RA, Moreno JMP, Labarta JR, García-Honduvilla N, Alvarez-Mon M, Bravo C, et al. The Use of Prebiotics from Pregnancy and Its Complications: Health for Mother and Offspring—A Narrative Review. Foods. 2023; 12(6):1148. https://doi.org/10.3390/foods12061148
Chicago/Turabian StyleGarcía-Montero, Cielo, Oscar Fraile-Martinez, Sonia Rodriguez-Martín, Jose V. Saz, Rocio Aracil Rodriguez, Juan Manuel Pina Moreno, Javier Ruiz Labarta, Natalio García-Honduvilla, Melchor Alvarez-Mon, Coral Bravo, and et al. 2023. "The Use of Prebiotics from Pregnancy and Its Complications: Health for Mother and Offspring—A Narrative Review" Foods 12, no. 6: 1148. https://doi.org/10.3390/foods12061148
APA StyleGarcía-Montero, C., Fraile-Martinez, O., Rodriguez-Martín, S., Saz, J. V., Rodriguez, R. A., Moreno, J. M. P., Labarta, J. R., García-Honduvilla, N., Alvarez-Mon, M., Bravo, C., De Leon-Luis, J. A., & Ortega, M. A. (2023). The Use of Prebiotics from Pregnancy and Its Complications: Health for Mother and Offspring—A Narrative Review. Foods, 12(6), 1148. https://doi.org/10.3390/foods12061148











