Dynamic Changes of Bacterial Communities and Microbial Association Networks in Ready-to-Eat Chicken Meat during Storage
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Processing
2.2. DNA Extraction
2.3. PCR Amplification and Sequencing
2.4. Sequence Data Analysis
2.5. Statistical Analysis
3. Results
3.1. Characteristics of OTUs in RTE Chicken Samples during Storage
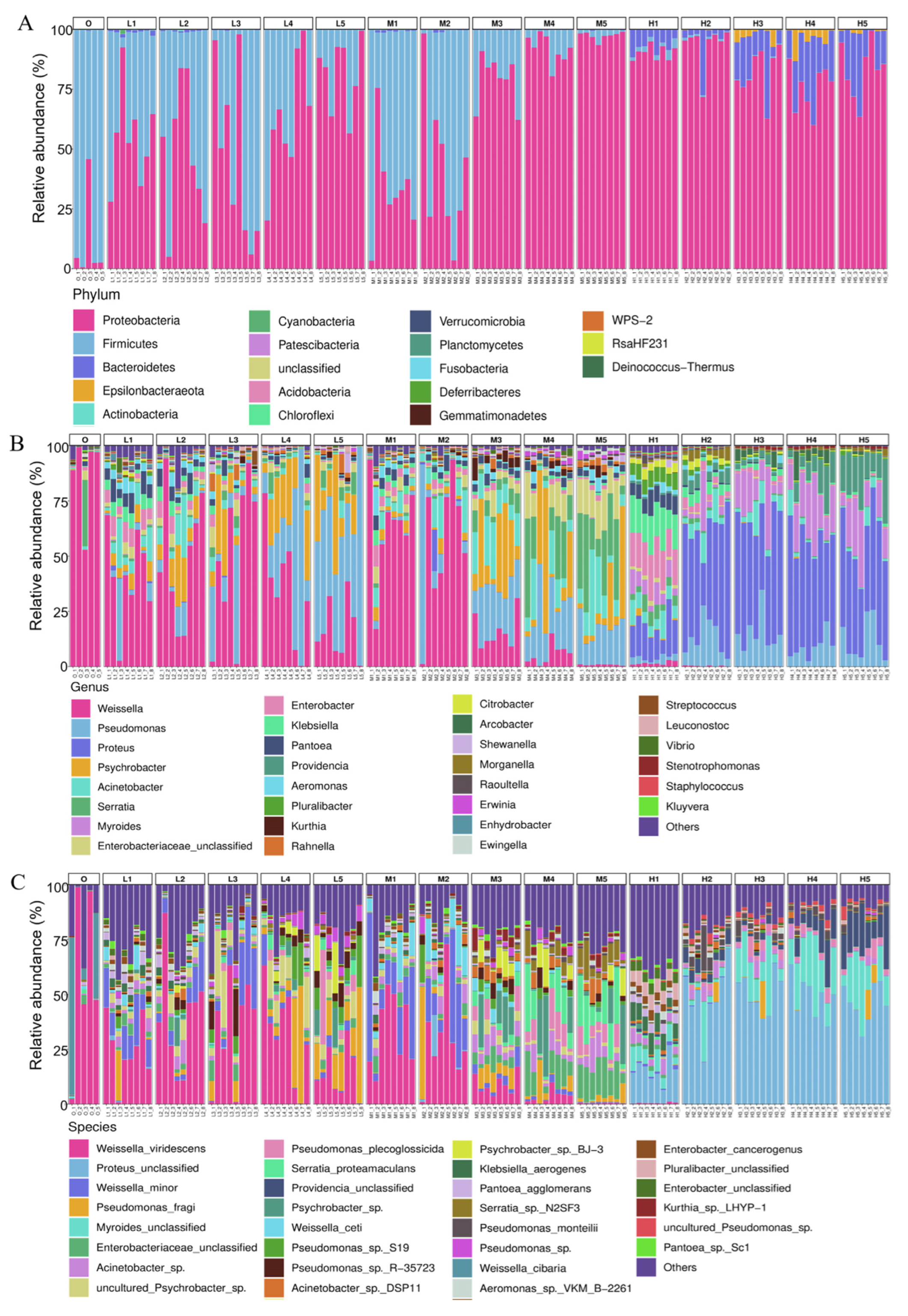
3.2. Taxonomic Composition of Bacterial Community
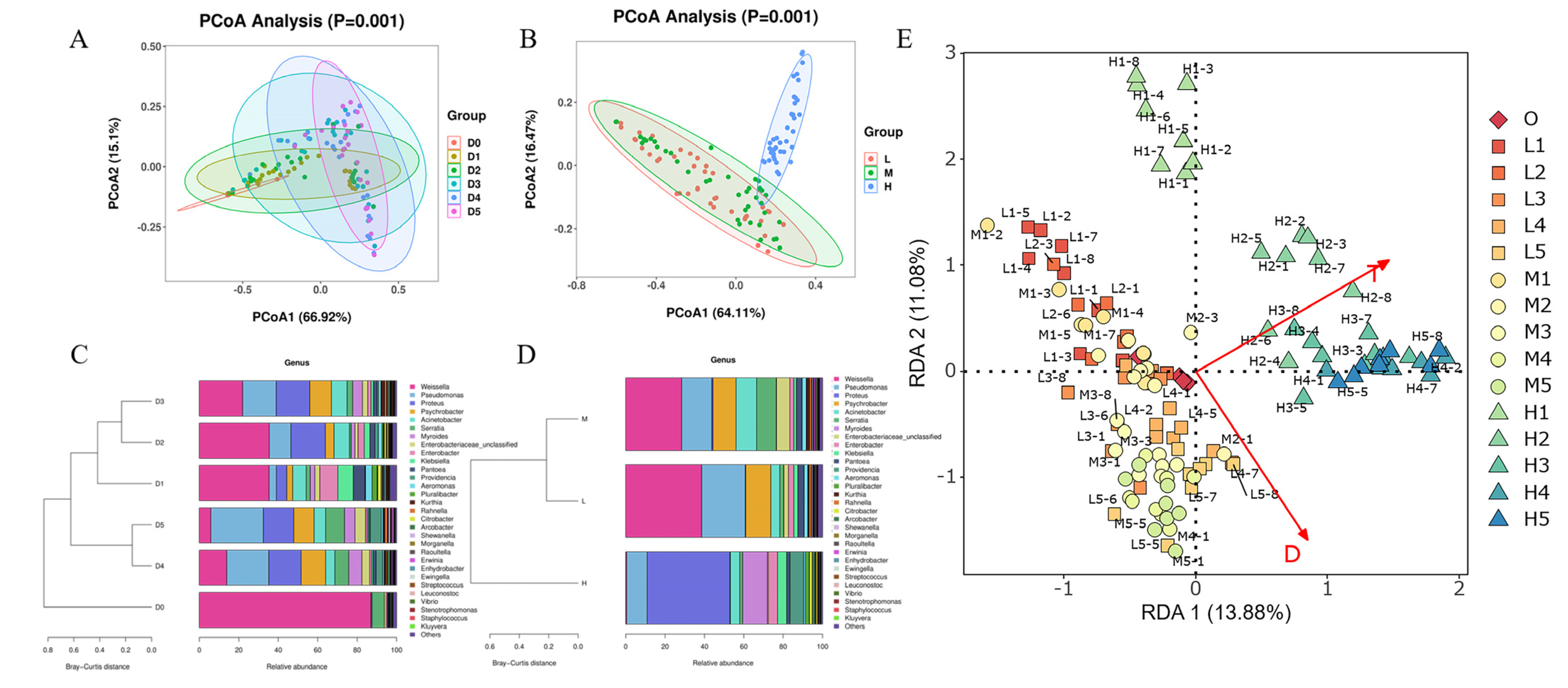
3.3. Microbiota on RTE Chicken Meat Varies over Time during Storage at Different Temperatures
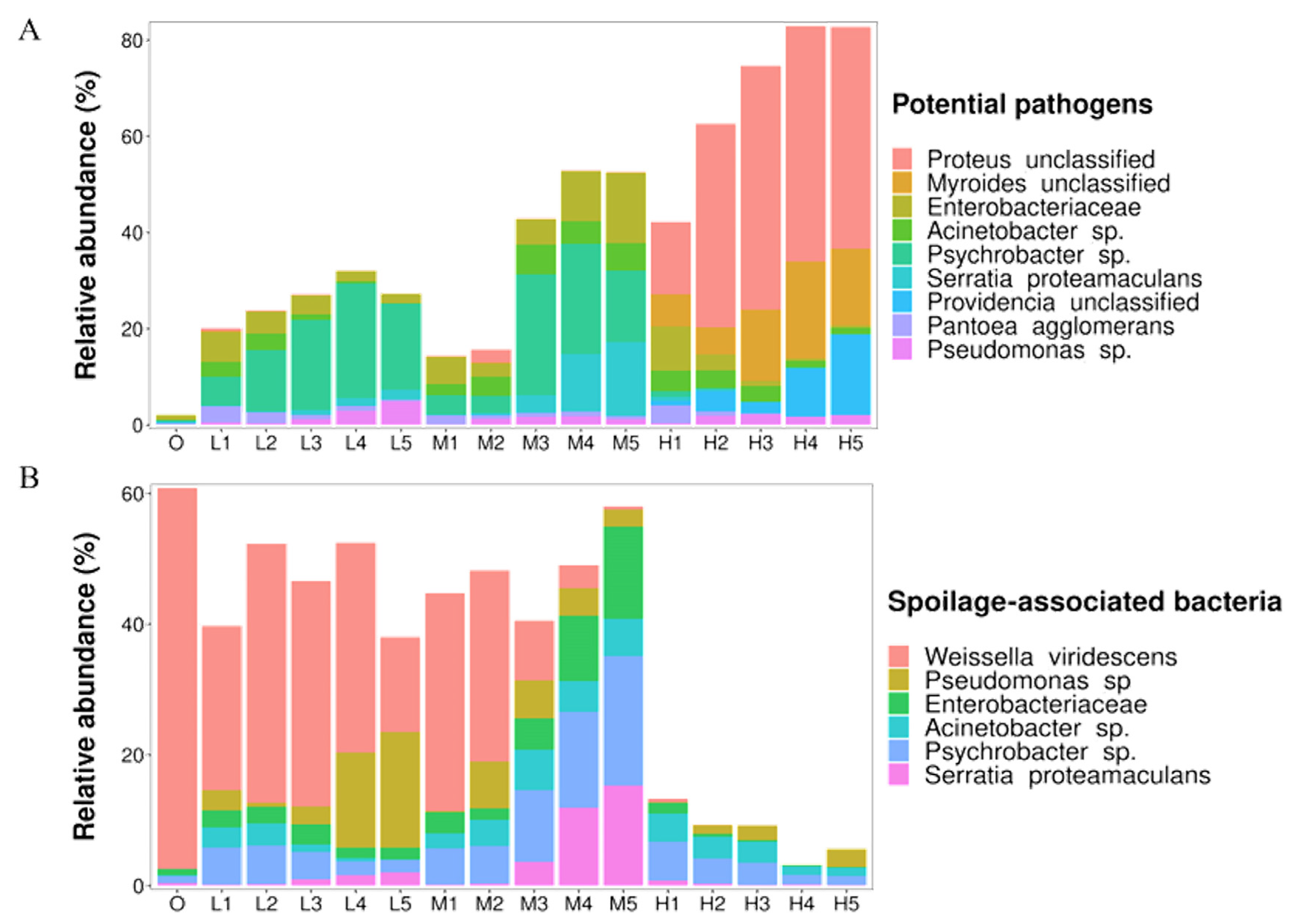
3.4. Potentially Pathogenic and Spoilage-Associated Species
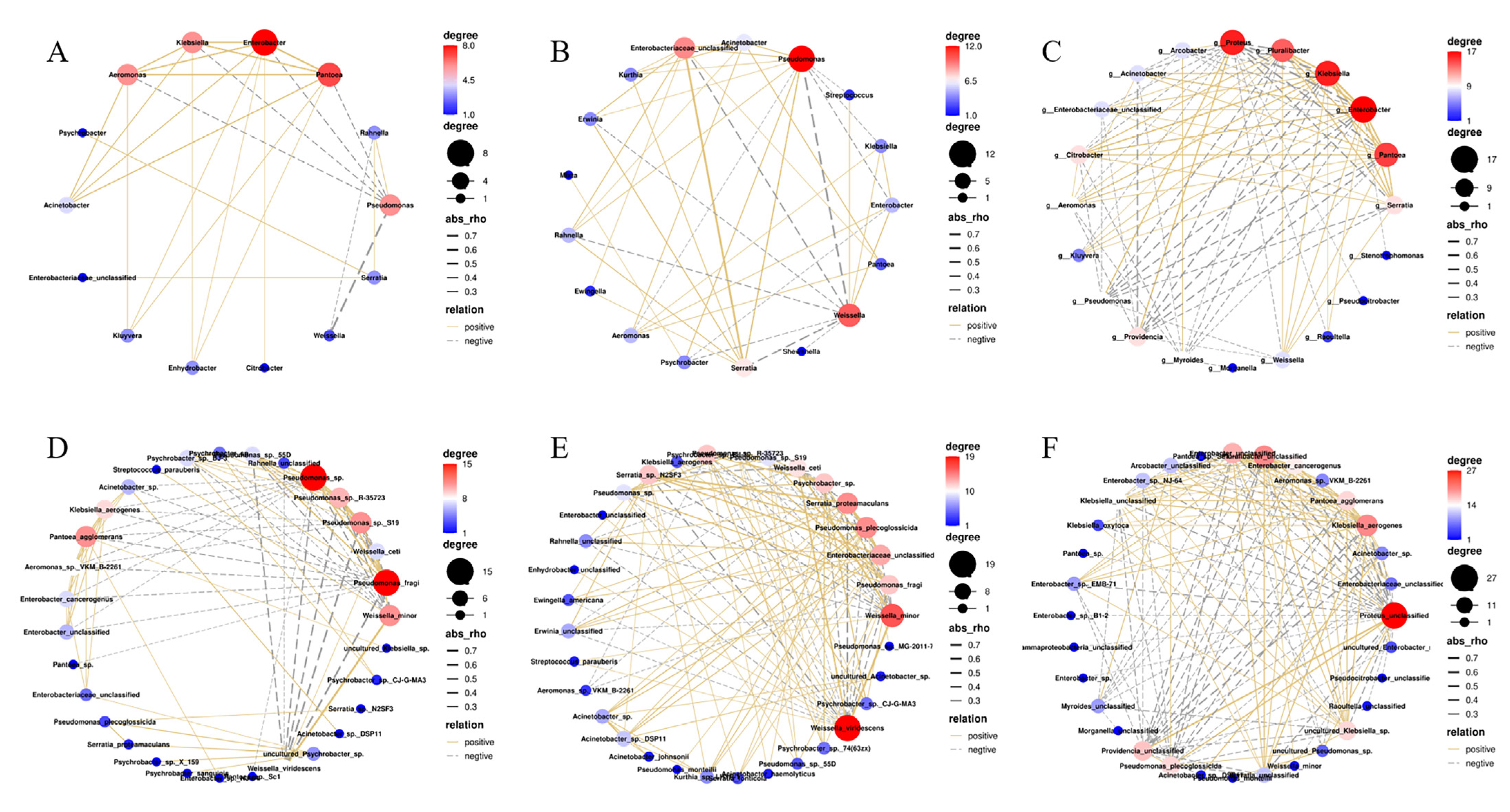
3.5. Microbial Association Networks during Storage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Cui, B.; Wang, L.D.; Chen, X.; Xu, M.Y.; Ke, J.; Tian, Y. Chicken meat taste preferences, perceived risk of human infection with avian influenza virus, and self-reported chicken meat consumption in China. Prev. Vet. Med. 2022, 203, 105658. [Google Scholar] [CrossRef]
- Li, J.; Zhang, D.; Yin, L.; Li, Z.; Yu, C.; Du, H.; Jiang, X.; Yang, C.; Liu, Y. Integration analysis of metabolome and transcriptome profiles revealed the age-dependent dynamic change in chicken meat. Food Res. Int. 2022, 156, 111171. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhao, D.; Xiang, Q.; Li, K.; Bai, Y.J. Effect of cinnamon essential oil nanoemulsions on microbiological safety and quality properties of chicken breast fillets during refrigerated storage. LWT 2021, 152, 112376. [Google Scholar] [CrossRef]
- González-González, C.; Labo-Popoola, O.; Delgado-Pando, G.; Theodoridou, K.; Doran, O.; Stratakos, A.C. The effect of cold atmospheric plasma and linalool nanoemulsions against Escherichia coli O157:H7 and Salmonella on ready-to-eat chicken meat. LWT 2021, 149, 111898. [Google Scholar] [CrossRef]
- Doulgeraki, A.I.; Ercolini, D.; Villani, F.; Nychas, G.J. Spoilage microbiota associated to the storage of raw meat in different conditions. Int. J. Food Microbiol. 2012, 157, 130–141. [Google Scholar] [CrossRef] [PubMed]
- İncili, G.K.; Akgöl, M.; Aydemir, M.E.; Alan, S.; Mutlu, M.; İlhak, O.İ.; Öksüztepe, G. Fate of Listeria monocytogenes and Salmonella Typhimurium in homemade marinade and on marinated chicken drumsticks, wings and breast meat. LWT 2020, 134, 110231. [Google Scholar] [CrossRef]
- Chen, X.; Chen, W.; Lu, X.; Mao, Y.; Luo, X.; Liu, G.; Zhu, L.; Zhang, Y. Effect of chitosan coating incorporated with oregano or cinnamon essential oil on the bacterial diversity and shelf life of roast duck in modified atmosphere packaging. Food Res. Int. 2021, 147, 110491. [Google Scholar] [CrossRef]
- Tsaloumi, S.; Aspridou, Z.; Tsigarida, E.; Gaitis, F.; Garofalakis, G.; Barberis, K.; Tzoumanika, F.; Dandoulaki, M.; Skiadas, R.; Koutsoumanis, K. Quantitative risk assessment of Listeria monocytogenes in ready-to-eat (RTE) cooked meat products sliced at retail stores in Greece. Food Microbiol. 2021, 99, 103800. [Google Scholar] [CrossRef]
- Wang, P.; Hu, A.; Fan, X.; Zhao, X.; Ge, Y.; Chen, Y. Bacterial communities in prepared foods available at supermarkets in Beijing, China. Food Res. Int. 2019, 120, 668–678. [Google Scholar] [CrossRef]
- Ezeokoli, O.T.; Gupta, A.K.; Mienie, C.; Popoola, T.O.S.; Bezuidenhout, C.C. PCR-denaturing gradient gel electrophoresis analysis of microbial community in soy-daddawa, a Nigerian fermented soybean (Glycine max (L.) Merr.) condiment. Int. J. Food Microbiol. 2016, 220, 58–62. [Google Scholar] [CrossRef]
- White, R.A.; Callister, S.J.; Moore, R.J.; Baker, E.S.; Jansson, J.K. The past, present and future of microbiome analyses. Nat. Protoc. 2016, 11, 2049–2053. [Google Scholar] [CrossRef]
- Mirhosseini, S.Z.; Seidavi, A.R.; Shivazad, M.; Chamani, M.; Sadeghi, A.A.; Pourseify, R. Detection of Clostridium spp. and its relation to different ages and gastrointestinal segments as measured by molecular analysis of 16S rRNA genes. Braz. Arch. Biol. Technol. 2010, 53, 69–76. [Google Scholar] [CrossRef]
- Ahir, V.B.; Singh, K.M.; Tripathi, A.K.; Mathakiya, R.A.; Jakhesara, S.J.; Koringa, P.G.; Rank, D.N.; Jhala, M.K.; Joshi, C.G. Study of bacterial diversity in poultry gut using denaturing gradient gel electrophoresis. Iranian J. Appl. Anim. Sci. 2012, 2, 227–232. [Google Scholar]
- Zotta, T.; Parente, E.; Ianniello, R.G.; De Filippis, F.; Ricciardi, A. Dynamics of bacterial communities and interaction networks in thawed fish fillets during chilled storage in air. Int. J. Food Microbiol. 2019, 293, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Faust, K.; Raes, J. CoNet app: Inference of biological association networks using Cytoscape. F1000Research 2016, 5, 1519. [Google Scholar] [CrossRef]
- Friedman, J.; Alm, E.J. Inferring correlation networks from genomic survey data. PLoS Comput. Biol. 2012, 8, e1002687. [Google Scholar] [CrossRef]
- Kurtz, Z.D.; Muller, C.L.; Miraldi, E.R.; Littman, D.R.; Blaser, M.J.; Bonneau, R.A. Sparse and compositionally robust inference of microbial ecological networks. PLoS Comput. Biol. 2015, 11, e1004226. [Google Scholar] [CrossRef]
- Tsilimigras, M.C.; Fodor, A.A. Compositional data analysis of the microbiome: Fundamentals, tools, and challenges. Ann. Epidemiol. 2016, 26, 330–335. [Google Scholar] [CrossRef]
- Parente, E.; Zotta, T.; Faust, K.; De Filippis, F.; Ercolini, D. Structure of association networks in food bacterial communities. Food Microbiol. 2018, 73, 49–60. [Google Scholar] [CrossRef]
- Weiss, S.; Van Treuren, W.; Lozupone, C.; Faust, K.; Friedman, J.; Deng, Y.; Xia, L.C.; Xu, Z.Z.; Ursell, L.; Alm, E.J.; et al. Correlation detection strategies in microbial data sets vary widely in sensitivity and precision. ISME J. 2016, 10, 1669–1681. [Google Scholar] [CrossRef]
- Logue, J.B.; Stedmon, C.A.; Kellerman, A.M.; Nielsen, N.J.; Andersson, A.F.; Laudon, H.; Lindstrom, E.S.; Kritzberg, E.S. Experimental insights into the importance of aquatic bacterial community composition to the degradation of dissolved organic matter. ISME J. 2016, 10, 533–545. [Google Scholar] [CrossRef]
- Magoc, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–5963. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Yu, G.; Smith, D.K.; Zhu, H.; Guan, Y.; Lam, T.T.Y. ggtree: An r package for visualization and annotation of phylogenetic trees with their covariates and other associated data. Methods Ecol. Evol. 2016, 8, 28–36. [Google Scholar] [CrossRef]
- Wei, Z.Y.; Rao, J.H.; Tang, M.T.; Zhao, G.A.; Li, Q.C.; Wu, L.M.; Liu, S.Q.; Li, B.H.; Xiao, B.Q.; Liu, X.Y. Characterization of changes and driver microbes in gut microbiota during healthy aging using a captive monkey model. Genom. Proteom. Bioinform. 2021, 20, 350–365. [Google Scholar] [CrossRef]
- Woolhouse, M.E.J.; Gowtage-Sequeria, S.; Evans, B. T16: Quantitative analysis of the characteristics of emerging and re-emerging human pathogens. In The UK Government’s Foresight Project Infectious Diseases: Preparing for the Future; University of Edinburgh: Edinburgh, UK, 2016. [Google Scholar]
- TRBA 466 Technical Rules for Biological Agents 466. Classification of Prokaryotes (Bacteria and 825 Archaea) into Risk Groups, December 2010, ed.; Federal Institute for Occupational Safety and Health: Dortmund, Germany, 2010. (In Germany)
- Amato, K.R.; Yeoman, C.J.; Kent, A.; Righini, N.; Carbonero, F.; Estrada, A.; Gaskins, H.R.; Stumpf, R.M.; Yildirim, S.; Torralba, M.; et al. Habitat degradation impacts black howler monkey (Alouatta pigra) gastrointestinal microbiomes. ISME J. 2013, 7, 1344–1353. [Google Scholar] [CrossRef]
- Liang, T.; Xie, X.; Zhang, J.; Ding, Y.; Wu, Q. Bacterial community and composition of different traditional fermented dairy products in China, South Africa, and Sri Lanka by high-throughput sequencing of 16S rRNA genes. LWT 2021, 144, 111209. [Google Scholar] [CrossRef]
- Yong, W.; Pan, X.J. Pan, Bacteria: Proteus. Encycl. Food Saf. 2014, 1, 486–489. [Google Scholar]
- Bux, A.; Mustafa, A.; Niazi, M.; Manchandani, U.; Mobarakai, N.; Lafferty, J.; DeChavez, V. Multivalvular infective endocarditis with Proteus mirabilis. IDCases 2022, 27, e01429. [Google Scholar] [CrossRef]
- Pérez-Lazo, G.; Morales-Moreno, A.; Soto-Febres, F.; Jove-Químper, H.; Silva-Caso, W.J.I. First report of Myroides phaeus bacteraemia identified by Polymerase chain reaction and genetic sequencing. IDCases 2020, 19, e00695. [Google Scholar] [CrossRef]
- Díaz-Jiménez, D.; García-Menio, I.; Fernández, J.; García, V.; Mora, A. Chicken and turkey meat: Consumer exposure to multidrug-resistant Enterobacteriaceae including mcr-carriers, uropathogenic E. coli and high-risk lineages such as ST131. Int. J. Food Microbiol. 2020, 331, 108750. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Peters, B.M.; Li, B.; Li, L.; Yu, G.; Xu, Z.; Shirtliff, M.E.J.M.P. Clinical features and antimicrobial resistance profiles of important Enterobacteriaceae pathogens in Guangzhou representative of Southern China, 2001–2015. Microb. Pathog. 2017, 107, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Carvalheira, A.; Silva, J.; Teixeira, P.J.F.M. Acinetobacter spp. in food and drinking water—A review. Microbial. Pathogenesis 2020, 95, 103675. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.; Lopes, M.S.; Carvalheira, A.; Barbosa, J.; Teixeira, P.J.F.M. Survival of clinical and food Acinetobacter spp. isolates exposed to different stress conditions. Food Microbiol. 2019, 77, 202–207. [Google Scholar] [CrossRef]
- García-López, M.L.; Santos, J.A.; Otero, A.; Rodríguez-Calleja, J.M. Psychrobacter. In Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014; pp. 261–268. [Google Scholar]
- Rafii, F. Serratia. In Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014; pp. 371–375. [Google Scholar]
- Galac, M.R.; Lazzaro, B.P.J.M. Comparative pathology of bacteria in the genus Providencia to a natural host, Drosophila melanogaster. Microbes Infect. 2011, 13, 673–683. [Google Scholar] [CrossRef]
- Ashoka Mahapatraa, S.; Pragnya, P.J.; Asutosh, M.; Debabrata, D.; Ambareesh, P. Neonatal septicaemia due to a rare bacterium: Pantoea agglomerans (case series). Pediatr. Infect. Dis. J. 2014, 6, 102–104. [Google Scholar]
- Korcova, J.; Koprnova, J.; Krcmery, V.; Krcmery, V. Bacteraemia due to Pseudomonas putida and other Pseudomonas non-aeruginosa in children. J. Infect. 2005, 51, 81. [Google Scholar] [CrossRef]
- Martins, W.F.; Longhi, D.; Menezes, N.; Camargo, A.; Laurindo, J.B.; AragO, G. Predicting growth of Weissella viridescens in culture medium under dynamic temperature conditions. Procedia Food Sci. 2016, 7, 37–40. [Google Scholar] [CrossRef][Green Version]
- Kavitake, D.; Devi, P.B.; Shetty, P.H. Overview of exopolysaccharides produced by Weissella genus—A review. Int. J. Biol. Macromol. 2020, 164, 2964–2973. [Google Scholar] [CrossRef]
- Wickramasinghe, N.N.; Ravensdale, J.; Coorey, R.; Dykes, G.A.; Chandry, P.S. Transcriptional profiling of biofilms formed on chilled beef by psychrotrophic meat spoilage bacterium, Pseudomonas fragi 1793. Biofilm 2021, 3, 100045. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Qi, J.; Dong, Y.; Li, Y.; Xu, X.; Zhou, G.J. Characterization of attachment and biofilm formation by meat-borne Enterobacteriaceae strains associated with spoilage. LWT 2017, 86, 399–407. [Google Scholar] [CrossRef]
- Kämpfer, P. Acinetobacter. In Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014; pp. 11–17. [Google Scholar]
- Gennari, M.; Parini, M.; Volpon, D.; Serio, M.J.I.J.o.F.M. Isolation and characterization by conventional methods and genetic transformation of Psychrobacter and Acinetobacter from fresh and spoiled meat, milk and cheese. Int. J. Food Microbiol. 1992, 15, 61–75. [Google Scholar] [CrossRef]
- Begrem, S.; Jerme, M.; Leroi, F.; Delbarre-Ladrat, C.; Grovel, O.; Passerini, D. Genomic diversity of Serratia proteamaculans and Serratia liquefaciens predominant in seafood products and spoilage potential analyses. Int. J. Food Microbiol. 2021, 354, 109326. [Google Scholar] [CrossRef] [PubMed]
- Faust, K.; Raes, J. Microbial interactions: From networks to models. Nat. Rev. Microbiol. 2012, 10, 538–550. [Google Scholar] [CrossRef] [PubMed]
- Feng, K.; Zhang, Z.; Cai, W.; Liu, W.; Xu, M.; Yin, H.; Wang, A.; He, Z.; Deng, Y. Biodiversity and species competition regulate the resilience of microbial biofilm community. Mol. Ecol. 2017, 26, 6170–6182. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Guo, Y.; Hou, Q.; Huang, M.; Zhou, X. Dynamic changes of the bacterial communities in roast chicken stored under normal and modified atmosphere packaging. J. Food Sci. 2020, 85, 1231–1239. [Google Scholar] [CrossRef]
- Li, X.; Xiong, Q.; Xu, B.; Wang, H.; Zhou, H.; Sun, Y. Bacterial community dynamics during different stages of processing of smoked bacon using the 16S rRNA gene amplicon analysis. Int. J. Food Microbiol. 2021, 351, 109076. [Google Scholar] [CrossRef]
- Kaur, M.; Williams, M.; Bissett, A.; Ross, T.; Bowman, J.P. Effect of abattoir, livestock species and storage temperature on bacterial community dynamics and sensory properties of vacuum packaged red meat. Food Microbiol. 2021, 94, 103648. [Google Scholar] [CrossRef]
- Wei, Z.; Chu, R.; Li, L.; Zhang, J.; Zhang, H.; Pan, X.; Dong, Y.; Liu, G. Study on microbial community succession and protein hydrolysis of donkey meat during refrigerated storage based on Illumina NOVA sequencing technology. Food Sci. Anim. Resour. 2021, 41, 701–714. [Google Scholar] [CrossRef]
- Manthou, E.; Coeuret, G.; Chaillou, S.; Nychas, G.E. Metagenetic characterization of bacterial communities associated with ready-to-eat leafy vegetables and study of temperature effect on their composition during storage. Korean J. Food Res. Int. 2022, 158, 111563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Ding, H.; Chen, L.; Zhang, S.; Wu, P.; Xie, K.; Pan, Z.; Zhang, G.; Dai, G.; Wu, H.; et al. Characterization of chilled chicken spoilage using an integrated microbiome and metabolomics analysis. Food Res. Int. 2021, 144, 110328. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, K.; Babu, D.; Juneja, V.K. Proteus. In Encyclopedia of Food Microbiology; Academic Press: Cambridge, MA, USA, 2014; pp. 238–243. [Google Scholar]
- Gram, L.; Ravn, L.; Rasch, M.; Bruhn, J.B.; Christensen, A.B.; Givskov, M. Food spoilage—Interactions between food spoilage bacteria. Int. J. Food Microbiol. 2002, 78, 79–97. [Google Scholar] [CrossRef]
- Ma, B.; Wang, Y.; Ye, S.; Liu, S.; Stirling, E.; Gilbert, J.A.; Faust, K.; Knight, R.; Jansson, J.K.; Cardona, C.; et al. Earth microbial co-occurrence network reveals interconnection pattern across microbiomes. Microbiome 2020, 8, 82. [Google Scholar] [CrossRef]
- Layeghifard, M.; Hwang, D.M.; Guttman, D.S. Disentangling Interactions in the microbiome: A network perspective. Trends Microbiol. 2017, 25, 217–228. [Google Scholar] [CrossRef]
- Gram, L. Inhibitory effect against pathogenic and spoilage bacteria of Pseudomonas strains isolated from spoiled and fresh fish. Appl. Environ. Microbiol. 1993, 59, 2197–2203. [Google Scholar] [CrossRef]
- MCKELLAR, R.C. Role of Nutrient Limitation in the Competition between Pseudomonas fluorescens and Escherichia coli O157:H7. J. Food Prot. 2007, 70, 1739–1743. [Google Scholar] [CrossRef]
- Collazo, C.; Abadías, M.; Colás-Medà, P.; Iglesias, M.B.; Granado-Serrano, A.B.; Serrano, J.; Viñas, I. Effect of Pseudomonas graminis strain CPA-7 on the ability of Listeria monocytogenes and Salmonella enterica subsp. enterica to colonize Caco-2 cells after pre-incubation on fresh-cut pear. Int. J. Food Microbiol. 2017, 4, 55–62. [Google Scholar] [CrossRef]
- Tsafrakidou, P.; Sameli, N.; Bosnea, L.; Chorianopoulos, N.; Samelis, J. Assessment of the spoilage microbiota in minced free-range chicken meat during storage at 4 °C in retail modified atmosphere packages. Food Microbiol. 2021, 99, 103822. [Google Scholar] [CrossRef]
- Doulgeraki, A.I.; Paramithiotis, S.; Nychas, G. Characterization of the Enterobacteriaceae community that developed during storage of minced beef under aerobic or modified atmosphere packaging conditions. Int. J. Food Microbiol. 2011, 145, 77–83. [Google Scholar] [CrossRef]
- Yu, Y.C.; Yum, S.J.; Jeon, D.Y.; Jeong, H. Analysis of the microbiota on lettuce (Lactuca sativa L.) cultivated in South Korea to identify foodborne pathogens. J. Microbiol. Biotechnol. 2018, 28, 1318–1331. [Google Scholar] [CrossRef]
- Chen, X.; Zhu, L.; Liang, R.; Mao, Y.; Hopkins, D.L.; Li, K.; Dong, P.; Yang, X.; Niu, L.; Zhang, Y.; et al. Shelf-life and bacterial community dynamics of vacuum packaged beef during long-term super-chilled storage sourced from two Chinese abattoirs. Food Res. Int. 2020, 130, 108937. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.F.; Mckay, A.M.; Connolly, M.; Linton, M.J.F.M. Effect of high pressure on the microbiological quality of cooked chicken during storage at normal and abuse refrigeration temperatures. Food Microbiol. 2010, 27, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Bassey, A.P.; Chen, Y.; Zhu, Z.; Odeyemi, O.A.; Frimpong, E.B.; Ye, K.; Li, C.; Zhou, G. Assessment of quality characteristics and bacterial community of modified atmosphere packaged chilled pork loins using 16S rRNA amplicon sequencing analysis. Food Res. Int. 2021, 145, 110412. [Google Scholar] [CrossRef] [PubMed]
- Min, J.J.; Kim, S.Y.; Ricke, S.C.; Min, S.R.; Sun, A.K.J. Microbial ecology of alfalfa, radish, and rapeseed sprouts based on culture methods and 16S rRNA microbiome sequencing. Food Res. Int. 2021, 144, 110316. [Google Scholar]
- Mladenovic, K.G.; Grujovic, M.Z.; Kis, M.; Furmeg, S.; Tkalec, V.J.; Stefanovic, O.D.; Kocic-Tanackov, S.D. Enterobacteriaceae in food safety with an emphasis on raw milk and meat. Appl. Microbiol. Biotechnol. 2021, 105, 8615–8627. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Wang, L. Evaluation of the bacterial populations present in Spring Mix salad and their impact on the behavior of Escherichia coli O157:H7. Food Control 2021, 124, 107865. [Google Scholar] [CrossRef]
- Greay, T.L.; Gofton, A.W.; Zahedi, A.; Paparini, A.; Linge, K.L.; Joll, C.A.; Ryan, U.M. Evaluation of 16S next-generation sequencing of hypervariable region 4 in wastewater samples: An unsuitable approach for bacterial enteric pathogen identification. Sci. Total Environ. 2019, 670, 1111–1124. [Google Scholar] [CrossRef]
- Jameson, J.E. A discussion of the dynamics of Salmonella enrichment. J. Hyg. 1962, 60, 193–207. [Google Scholar] [CrossRef]
- Dens, E.J.; Impe, J. 21-Modelling applied to foods: Predictive microbiology for solid food systems. In Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2003; pp. 472–506. [Google Scholar]

| Sample | Observed OTU | Shannon | Simpson | Chao1 | Good’s Coverage |
|---|---|---|---|---|---|
| O | 116 ± 61 | 2.12 ± 1.45 | 0.49 ± 0.32 | 117.47 ± 61.65 | 0.9998 |
| L1 | 365 ± 62 | 5.41 ± 0.76 | 0.90 ± 0.07 | 373.24 ± 60.49 | 0.9996 |
| L2 | 334 ± 95 | 4.62 ± 1.75 | 0.78 ± 0.24 | 337.67 ± 94.39 | 0.9997 |
| L3 | 248 ± 68 | 3.99 ± 1.35 | 0.78 ± 0.13 | 250.68 ± 68.57 | 0.9998 |
| L4 | 208 ± 66 | 3.94 ± 0.77 | 0.80 ± 0.10 | 209.78 ± 65.85 | 0.9999 |
| L5 | 179 ± 51 | 4.37 ± 0.78 | 0.88 ± 0.06 | 180.21 ± 50.95 | 0.9999 |
| M1 | 292 ± 85 | 4.32 ± 1.22 | 0.79 ± 0.12 | 296.57 ± 84.04 | 0.9997 |
| M2 | 242 ± 80 | 4.23 ± 1.19 | 0.81 ± 0.15 | 245.80 ± 78.77 | 0.9998 |
| M3 | 253 ± 30 | 5.54 ± 0.33 | 0.95 ± 0.02 | 254.55 ± 30.50 | 0.9999 |
| M4 | 225 ± 29 | 5.04 ± 0.61 | 0.91 ± 0.05 | 227.37 ± 29.61 | 0.9999 |
| M5 | 237 ± 29 | 5.18 ± 0.57 | 0.92 ± 0.03 | 238.79 ± 29.06 | 0.9999 |
| H1 | 289 ± 43 | 6.44 ± 0.14 | 0.97 ± 0.01 | 291.01 ± 43.59 | 0.9999 |
| H2 | 235 ± 39 | 4.92 ± 0.36 | 0.90 ± 0.02 | 236.52 ± 38.55 | 0.9999 |
| H3 | 228 ± 35 | 4.68 ± 0.46 | 0.90 ± 0.02 | 228.64 ± 34.72 | 0.9999 |
| H4 | 177 ± 26 | 4.22 ± 0.23 | 0.89 ± 0.03 | 177.89 ± 25.79 | 0.9999 |
| H5 | 189 ± 28 | 4.23 ± 0.37 | 0.89 ± 0.02 | 189.92 ± 27.73 | 0.9999 |
| Species | CardinalSymptom/CorruptionPhenomenon | Reference | |
|---|---|---|---|
| Potential pathogens | Proteus unclassified | Urinary tract infections, gastroenteritis and wound infections | [31,32] |
| Myroides unclassified | Urinary tract infections, skin and soft tissue infections, bacteremia, pneumonia and intra-abdominal infections | [33] | |
| Enterobacteriaceae | Urinary tract infections, septicemia, pneumonia, peritonitis, meningitis and intra-abdominal infections | [34,35] | |
| Acinetobacter sp. | Urinary tract infections, skin infections, bacteremia, pneumonia, meningitis and endocarditis | [36,37] | |
| Psychrobacter sp. | Conjunctivitis, endocarditis, peritonitis, bacteremia, infant meningitis, arthritis and surgical wound infections | [38] | |
| Serratia proteamaculans | Pneumonia | [39] | |
| Providencia unclassified | Diarrhea | [40] | |
| Pantoea agglomerans | Septicemia | [41] | |
| Pseudomonas sp. | Bacteraemia | [42] | |
| Spoilage-associated bacteria | Weissella viridescens | Produces peroxide which reacts with meat pigment and forms a green-colored | [43,44] |
| Pseudomonas fragi | slime formation | [45] | |
| Enterobacteriaceae | Forms biofilm and produces gas; putrescine and cadaverine | [46] | |
| Acinetobacter sp. | Produces some volatile spoilage compounds and sulfurous, rancid and fishy off-odors | [47] | |
| Psychrobacter sp. | Produces some volatile spoilage compounds and musty off-odors | [38,48] | |
| Serratia proteamaculans | Produces trimethylamine, putrescine, cadaverine and off-odors | [49] |
| Temperature (°C) | Interaction | Pearson Correlation † | Relation |
|---|---|---|---|
| 4 | Enterobacter ↔ Pantoea | 0.583 | Copresence |
| Klebsiella ↔ Pantoea | 0.524 | Copresence | |
| Aeromonas ↔ Pantoea | 0.515 | Copresence | |
| Enterobacter ↔ Klebsiella | 0.498 | Copresence | |
| Aeromonas ↔ Enterobacter | 0.485 | Copresence | |
| Aeromonas ↔ Klebsiella | 0.464 | Copresence | |
| Acinetobacter ↔ Pantoea | 0.420 | Copresence | |
| Pseudomonas ↔ Weissella | −0.702 | Mutual exclusion | |
| 8 | Enterobacteriaceae_unclassified ↔ Serratia | 0.740 | Copresence |
| Pseudomonas ↔ Serratia | 0.469 | Copresence | |
| Enterobacteriaceae_unclassified ↔ Weissella | −0.510 | Mutual exclusion | |
| Serratia ↔ Weissella | −0.563 | Mutual exclusion | |
| Pseudomonas ↔ Weissella | −0.677 | Mutual exclusion | |
| 22 | Enterobacter ↔ Klebsiella | 0.664 | Copresence |
| Klebsiella ↔ Pluralibacter | 0.556 | Copresence | |
| Enterobacter ↔ Pantoea | 0.548 | Copresence | |
| Klebsiella ↔ Pantoea | 0.539 | Copresence | |
| Enterobacter ↔ Pluralibacter | 0.538 | Copresence | |
| Enterobacter ↔ Serratia | 0.479 | Copresence | |
| Proteus ↔ Providencia | 0.462 | Copresence | |
| Citrobacter ↔ Klebsiella | 0.457 | Copresence | |
| Klebsiella ↔ Serratia | 0.455 | Copresence | |
| Pantoea ↔ Pluralibacter | 0.439 | Copresence | |
| Pantoea ↔ Serratia | 0.438 | Copresence | |
| Proteus ↔ Pseudomonas | 0.421 | Copresence | |
| Citrobacter ↔ Enterobacter | 0.403 | Copresence | |
| Pantoea ↔ Pseudomonas | −0.425 | Mutual exclusion | |
| Klebsiella ↔ Pseudomonas | −0.444 | Mutual exclusion | |
| Proteus ↔ Serratia | −0.445 | Mutual exclusion | |
| Enterobacter ↔ Pseudomonas | −0.445 | Mutual exclusion | |
| Pantoea ↔ Providencia | −0.467 | Mutual exclusion | |
| Klebsiella ↔ Providencia | −0.476 | Mutual exclusion | |
| Pluralibacter ↔ Proteus | −0.489 | Mutual exclusion | |
| Acinetobacter ↔ Providencia | −0.502 | Mutual exclusion | |
| Enterobacter ↔ Providencia | −0.503 | Mutual exclusion | |
| Pantoea ↔ Proteus | −0.530 | Mutual exclusion | |
| Klebsiella ↔ Proteus | −0.572 | Mutual exclusion | |
| Enterobacter ↔ Proteus | −0.578 | Mutual exclusion |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, M.; Xiao, X.; Xiao, Y.; Ma, J.; Yang, H.; Jiang, H.; Dong, Q.; Wang, W. Dynamic Changes of Bacterial Communities and Microbial Association Networks in Ready-to-Eat Chicken Meat during Storage. Foods 2022, 11, 3733. https://doi.org/10.3390/foods11223733
Qiu M, Xiao X, Xiao Y, Ma J, Yang H, Jiang H, Dong Q, Wang W. Dynamic Changes of Bacterial Communities and Microbial Association Networks in Ready-to-Eat Chicken Meat during Storage. Foods. 2022; 11(22):3733. https://doi.org/10.3390/foods11223733
Chicago/Turabian StyleQiu, Mengjia, Xingning Xiao, Yingping Xiao, Jiele Ma, Hua Yang, Han Jiang, Qingli Dong, and Wen Wang. 2022. "Dynamic Changes of Bacterial Communities and Microbial Association Networks in Ready-to-Eat Chicken Meat during Storage" Foods 11, no. 22: 3733. https://doi.org/10.3390/foods11223733
APA StyleQiu, M., Xiao, X., Xiao, Y., Ma, J., Yang, H., Jiang, H., Dong, Q., & Wang, W. (2022). Dynamic Changes of Bacterial Communities and Microbial Association Networks in Ready-to-Eat Chicken Meat during Storage. Foods, 11(22), 3733. https://doi.org/10.3390/foods11223733








