Chitosan–Sodium Caseinate Composite Edible Film Incorporated with Probiotic Limosilactobacillus fermentum: Physical Properties, Viability, and Antibacterial Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Culture
2.2. Preparation of the Bioactive Film
2.3. Film Analysis
2.3.1. Thickness
2.3.2. Moisture Content
2.3.3. Water Solubility
2.3.4. Opacity
2.3.5. Color
2.3.6. Mechanical Properties
2.3.7. Viability of Lb. fermentum
2.3.8. Antibacterial Activity
2.4. Statistical Analysis
3. Results and Discussion
3.1. Film Analysis
3.1.1. Thickness, Moisture Content, and Water Solubility
3.1.2. Opacity and Color
3.1.3. Mechanical Analysis
3.2. Optimization of the Edible Film Characteristics
3.3. Addition of Lb. fermentum into the Edible Film
3.3.1. Physical Properties
3.3.2. Mechanical Properties
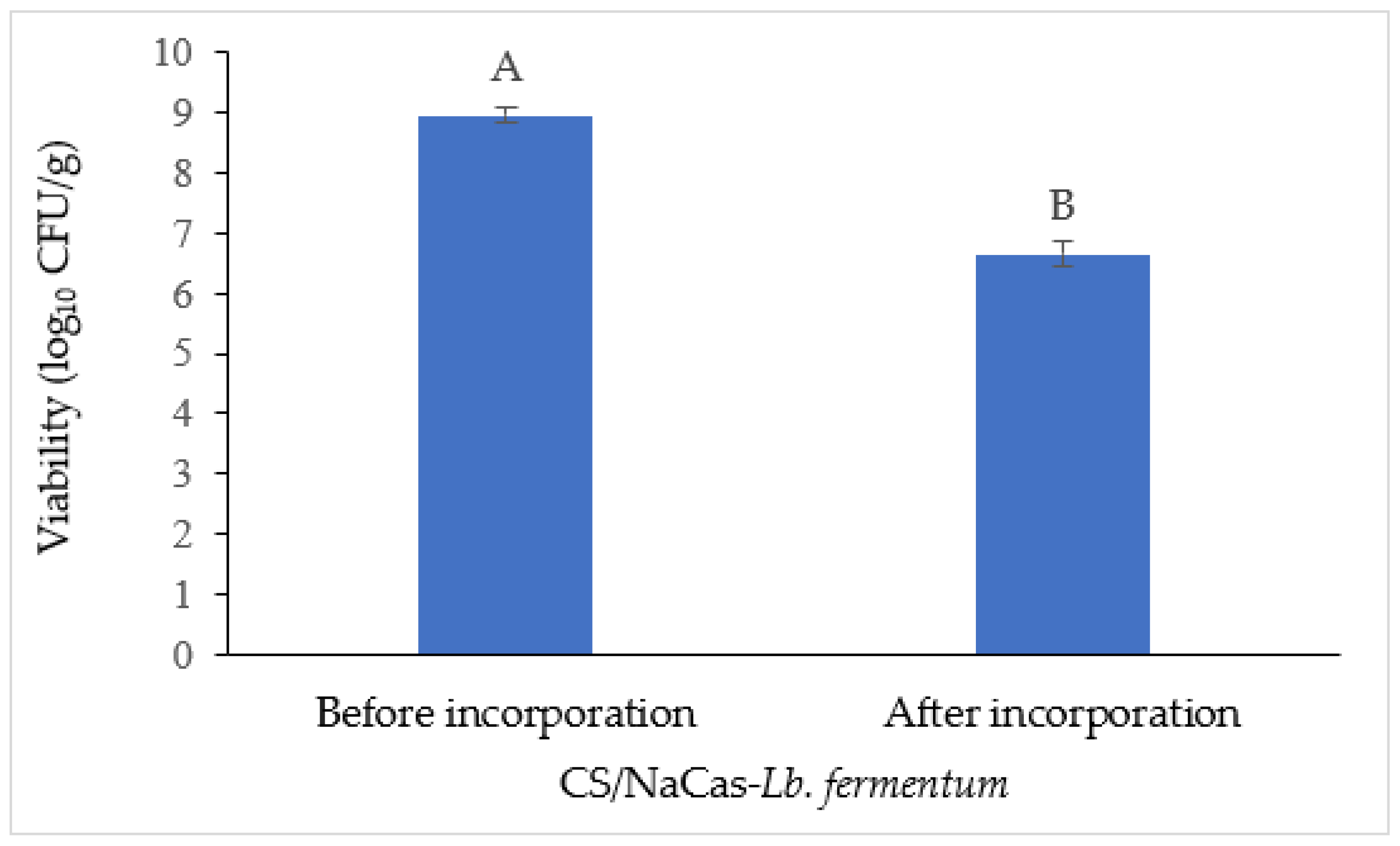
3.4. Survivability of Probiotics after Film Drying
3.5. Antibacterial Test
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adeyanju, G.C.; Augustine, T.M.; Volkmann, S.; Oyebamiji, U.A.; Ran, S.; Osobajo, O.A.; Otitoju, A. Effectiveness of Intervention on Behaviour Change against Use of Non-Biodegradable Plastic Bags: A Systematic Review. Discov. Sustain. 2021, 2, 13. [Google Scholar] [CrossRef]
- Rhim, J.W.; Park, H.M.; Ha, C.S. Bio-Nanocomposites for Food Packaging Applications. Prog. Polym. Sci. 2013, 38, 1629–1652. [Google Scholar] [CrossRef]
- Almasi, H.; Jahanbakhsh Oskouie, M.; Saleh, A. A Review on Techniques Utilized for Design of Controlled Release Food Active Packaging. Crit. Rev. Food Sci. Nutr. 2021, 61, 2601–2621. [Google Scholar] [CrossRef] [PubMed]
- Abdollahzadeh, E.; Ojagh, S.M.; Fooladi, A.A.I.; Shabanpour, B.; Gharahei, M. Effects of Probiotic Cells on the Mechanical and Antibacterial Properties of Sodium-Caseinate Films. Appl. Food Biotechnol. 2018, 5, 155–162. [Google Scholar] [CrossRef]
- Lin, H.C.; Wang, B.J.; Weng, Y.M. Development and Characterization of Sodium Caseinate Edible Films Cross-Linked with Genipin. LWT Food Sci. Technol. 2020, 118, 108813. [Google Scholar] [CrossRef]
- El Knidri, H.; Belaabed, R.; Addaou, A.; Laajeb, A.; Lahsini, A. Extraction, Chemical Modification and Characterization of Chitin and Chitosan. Int. J. Biol. Macromol. 2018, 120, 1181–1189. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial Properties of Chitosan and Mode of Action: A State of the Art Review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef]
- Morales, R.; Martinez, M.J.; Pilosof, A.M.R. Impact of Casein Glycomacropeptide on Sodium Caseinate Self-Assembly and Gelation. Int. Dairy J. 2015, 49, 30–36. [Google Scholar] [CrossRef]
- Fabra, M.J.; Hambleton, A.; Talens, P.; Debeaufort, F.; Chiralt, A. Effect of Ferulic Acid and α-Tocopherol Antioxidants on Properties of Sodium Caseinate Edible Films. Food Hydrocoll. 2011, 25, 1441–1447. [Google Scholar] [CrossRef]
- Dhumal, C.V.; Sarkar, P. Composite Edible Films and Coatings from Food-Grade Biopolymers. J. Food Sci. Technol. 2018, 55, 4369–4383. [Google Scholar] [CrossRef]
- Mohamed, S.A.A.; El-Sakhawy, M.; El-Sakhawy, M.A.M. Polysaccharides, Protein and Lipid -Based Natural Edible Films in Food Packaging: A Review. Carbohydr. Polym. 2020, 238, 116178. [Google Scholar] [CrossRef]
- Wu, J.; Zhong, F.; Li, Y.; Shoemaker, C.F.; Xia, W. Preparation and Characterization of Pullulan-Chitosan and Pullulan –Carboxymethyl Chitosan Blended Films. Food Hydrocoll. 2012, 30, 82–91. [Google Scholar] [CrossRef]
- Kurek, M.; Galus, S.; Debeaufort, F. Surface, Mechanical and Barrier Properties of Bio-Based Composite Films Based on Chitosan and Whey Protein. Food Packag. Shelf Life 2014, 1, 56–67. [Google Scholar] [CrossRef]
- Volpe, S.; Cavella, S.; Masi, P.; Torrieri, E. Effect of Solid Concentration on Structure and Properties of Chitosan-Caseinate Blend Films. Food Packag. Shelf Life 2017, 13, 76–84. [Google Scholar] [CrossRef]
- Pereda, M.; Aranguren, M.I.; Marcovich, N.E. Characterization of Chitosan/Caseinate Films. J. Appl. Polym. Sci. 2008, 107, 1080–1090. [Google Scholar] [CrossRef]
- Soukoulis, C.; Behboudi-Jobbehdar, S.; Yonekura, L.; Parmenter, C.; Fisk, I.D. Stability of Lactobacillus rhamnosus GG in Prebiotic Edible Films. Food Chem. 2014, 159, 302–308. [Google Scholar] [CrossRef]
- Gialamas, H.; Zinoviadou, K.G.; Biliaderis, C.G.; Koutsoumanis, K.P. Development of a Novel Bioactive Packaging Based on the Incorporation of Lactobacillus sakei into Sodium-Caseinate Films for Controlling Listeria Monocytogenes in Foods. Food Res. Int. 2010, 43, 2402–2408. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Quintero Saavedra, J.I.; Chiralt, A. Physical Properties and Antilisterial Activity of Bioactive Edible Films Containing Lactobacillus plantarum. Food Hydrocoll. 2013, 33, 92–98. [Google Scholar] [CrossRef]
- Silva, D.R.; Sardi, J.d.C.O.; de Souza Pitangui, N.; Roque, S.M.; da Silva, A.C.B.; Rosalen, P.L. Probiotics as an Alternative Antimicrobial Therapy: Current Reality and Future Directions. J. Funct. Foods 2020, 73, 104080. [Google Scholar] [CrossRef]
- Pop, O.L.; Pop, C.R.; Dufrechou, M.; Vodnar, D.C.; Socaci, S.A.; Dulf, F.V.; Minervini, F.; Suharoschi, R. Edible Films and Coatings Functionalization by Probiotic Incorporation: A Review. Polymers 2020, 12, 12. [Google Scholar] [CrossRef]
- Pereira, J.O.; Soares, J.; Costa, E.; Silva, S.; Gomes, A.; Pintado, M. Characterization of Edible Films Based on Alginate or Whey Protein Incorporated with Bifidobacterium animalis subsp. lactis BB-12 and Prebiotics. Coatings 2019, 9, 493. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Jafarpour, D. Bioactive Edible Film Based on Konjac Glucomannan and Probiotic Lactobacillus plantarum Strains: Physicochemical Properties and Shelf Life of Fresh-Cut Kiwis. J. Food Sci. 2021, 86, 513–522. [Google Scholar] [CrossRef]
- Soukoulis, C.; Singh, P.; Macnaughtan, W.; Parmenter, C.; Fisk, I.D. Compositional and Physicochemical Factors Governing the Viability of Lactobacillus rhamnosus GG Embedded in Starch-Protein Based Edible Films. Food Hydrocoll. 2016, 52, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Davachi, S.M.; Pottackal, N.; Torabi, H.; Abbaspourrad, A. Development and Characterization of Probiotic Mucilage Based Edible Films for the Preservation of Fruits and Vegetables. Sci. Rep. 2021, 11, 16608. [Google Scholar] [CrossRef] [PubMed]
- Concha-Meyer, A.; Schöbitz, R.; Brito, C.; Fuentes, R. Lactic Acid Bacteria in an Alginate Film Inhibit Listeria monocytogenes Growth on Smoked Salmon. Food Control 2011, 22, 485–489. [Google Scholar] [CrossRef]
- Soukoulis, C.; Yonekura, L.; Gan, H.H.; Behboudi-Jobbehdar, S.; Parmenter, C.; Fisk, I. Probiotic Edible Films as a New Strategy for Developing Functional Bakery Products: The Case of Pan Bread. Food Hydrocoll. 2014, 39, 231–242. [Google Scholar] [CrossRef]
- López De Lacey, A.M.; López-Caballero, M.E.; Gómez-Estaca, J.; Gómez-Guillén, M.C.; Montero, P. Functionality of Lactobacillus acidophilus and Bifidobacterium bifidum Incorporated to Edible Coatings and Films. Innov. Food Sci. Emerg. Technol. 2012, 16, 277–282. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Batista, R.A.; Azeredo, H.M.C.; Otoni, C.G. Probiotics and Their Potential Applications in Active Edible Films and Coatings. Food Res. Int. 2016, 90, 42–52. [Google Scholar] [CrossRef]
- Léonard, L.; Beji, O.; Arnould, C.; Noirot, E.; Bonnotte, A.; Gharsallaoui, A.; Degraeve, P.; Lherminier, J.; Saurel, R.; Oulahal, N. Preservation of Viability and Anti-Listeria Activity of Lactic Acid Bacteria, Lactococcus lactis and Lactobacillus paracasei, Entrapped in Gelling Matrices of Alginate or Alginate/Caseinate. Food Control 2015, 47, 7–19. [Google Scholar] [CrossRef]
- Chee, W.J.Y.; Chew, S.Y.; Than, L.T.L. Vaginal Microbiota and the Potential of Lactobacillus Derivatives in Maintaining Vaginal Health. Microb. Cell Fact. 2020, 19, 203. [Google Scholar] [CrossRef]
- Garcia-Castillo, V.; Komatsu, R.; Clua, P.; Indo, Y.; Takagi, M.; Salva, S.; Islam, M.A.; Alvarez, S.; Takahashi, H.; Garcia-Cancino, A.; et al. Evaluation of the Immunomodulatory Activities of the Probiotic Strain Lactobacillus fermentum UCO-979C. Front. Immunol. 2019, 10, 1376. [Google Scholar] [CrossRef]
- Arian, S.; Kaboosi, H.; Heshmatipour, Z.; Khazaei Koohpar, Z.; Peyravii, F. Anti-Proliferative Effects of Two New Lactobacillus Strains of Human Origin on Caco-2 Cell Line. Iran. Red Crescent Med. J. 2019, 21, e84683. [Google Scholar] [CrossRef]
- Ayyanna, R.; Ankaiah, D.; Arul, V. Anti-Inflammatory and Antioxidant Properties of Probiotic Bacterium Lactobacillus mucosae AN1 and Lactobacillus fermentum SNR1 in Wistar Albino Rats. Front. Microbiol. 2018, 9, 3063. [Google Scholar] [CrossRef]
- García, A.; Navarro, K.; Sanhueza, E.; Pineda, S.; Pastene, E.; Quezada, M.; Henríquez, K.; Karlyshev, A.; Villena, J.; González, C. Characterization of Lactobacillus fermentum UCO-979C, a Probiotic Strain with a Potent Anti-Helicobacter pylori Activity. Electron. J. Biotechnol. 2017, 25, 75–83. [Google Scholar] [CrossRef]
- Shieh, M.J.; Shang, H.f.; Liao, F.H.; Zhu, J.S.; Chien, Y.W. Lactobacillus Fermentum Improved Intestinal Bacteria Flora by Reducing Clostridium perfringens. e-SPEN Eur. e-J. Clin. Nutr. Metab. 2011, 6, e59–e63. [Google Scholar] [CrossRef][Green Version]
- Rodrigues, J.Z.d.S.; Passos, M.R.; Silva de Macêdo Neres, N.; Almeida, R.S.; Pita, L.S.; Santos, I.A.; Santana Silveira, P.H.; Reis, M.M.; Santos, I.P.; de Oliveira Negrão Ricardo, L.; et al. Antimicrobial Activity of Lactobacillus fermentum TcUESC01 against Streptococcus mutans UA159. Microb. Pathog. 2020, 142, 104063. [Google Scholar] [CrossRef]
- Mohanty, J.N.; Das, P.K.; Nanda, S.; Nayak, P.; Pradhan, P. Comparative Analysis of Crude and Pure Lactic Acid Produced by Lactobacillus fermentum and Its Inhibitory Effects on Spoilage Bacteria. Pharma Innov. J. 2015, 38, 38–42. [Google Scholar]
- Naghmouchi, K.; Belguesmia, Y.; Bendali, F.; Spano, G.; Seal, B.S.; Drider, D. Lactobacillus Fermentum: A Bacterial Species with Potential for Food Preservation and Biomedical Applications. Crit. Rev. Food Sci. Nutr. 2020, 60, 3387–3399. [Google Scholar] [CrossRef]
- Lee, Y.Y.; Elaine; Yusof, Y.A.; Pui, L.P. Development of Milk Protein Edible Films Incorporated with Lactobacillus rhamnosus GG. BioResources 2020, 15, 6960–6973. [Google Scholar] [CrossRef]
- Kuan, Y.L.; Navin Sivanasvaran, S.; Pui, L.P.; Yusof, Y.A.; Senphan, T. Physicochemical Properties of Sodium Alginate Edible Film Incorporated with Mulberry (Morus australis) Leaf Extract. Pertanika J. Trop. Agric. Sci. 2020, 43, 357–376. [Google Scholar]
- Siah, W.M.; Aminah, A.; Ishak, A. Edible Films from Seaweed (Kappaphycus alvarezii). Int. Food Res. J. 2015, 22, 2230–2236. [Google Scholar]
- Choong, K.W.; Tee, Y.B.; Nyam, K.L.; Pui, L.P. Antibacterial Properties of Chitosan Edible Films Incorporated with Musk Lime Extracts for the Preservation of Squids. Malays. J. Anal. Sci. 2019, 23, 914–925. [Google Scholar] [CrossRef]
- Zaman, N.B.K.; Lin, N.K.; Phing, P.L. Chitosan Film Incorporated with Garcinia Atroviridis for the Packaging of Indian Mackerel (Rastrelliger kanagurta). Cienc. Agrotecnol. 2018, 42, 666–675. [Google Scholar] [CrossRef]
- Champagne, C.P.; Ross, R.P.; Saarela, M.; Hansen, K.F.; Charalampopoulos, D. Recommendations for the Viability Assessment of Probiotics as Concentrated Cultures and in Food Matrices. Int. J. Food Microbiol. 2011, 149, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Kristo, E.; Koutsoumanis, K.P.; Biliaderis, C.G. Thermal, Mechanical and Water Vapor Barrier Properties of Sodium Caseinate Films Containing Antimicrobials and Their Inhibitory Action on Listeria monocytogenes. Food Hydrocoll. 2008, 22, 373–386. [Google Scholar] [CrossRef]
- Kanmani, P.; Lim, S.T. Development and Characterization of Novel Probiotic-Residing Pullulan/Starch Edible Films. Food Chem. 2013, 141, 1041–1049. [Google Scholar] [CrossRef]
- Santoso, R.A.; Atma, Y. Physical Properties of Edible Films from Pangasius Catfish Bone Gelatin-Breadfruits Strach with Different Formulations. Indones. Food Sci. Technol. J. 2020, 3, 42–47. [Google Scholar] [CrossRef]
- Jiang, S.; Qiao, C.; Wang, X.; Li, Z.; Yang, G. Structure and Properties of Chitosan/Sodium Dodecyl Sulfate Composite Films. RSC Adv. 2022, 12, 3969–3978. [Google Scholar] [CrossRef]
- Apriliyani, M.W.; Andriani, R.D.; Rahayu, P.P.; Purwadi, P.; Manab, A. Mechanical, Chemical and Microstructure Properties of Composite Edible Film Added with Modified Casein. J. Ilmu Prod. Teknol. Has. Peternak. 2020, 15, 162–171. [Google Scholar] [CrossRef]
- Huang, X.; Luo, X.; Liu, L.; Dong, K.; Yang, R.; Lin, C.; Song, H.; Li, S.; Huang, Q. Formation Mechanism of Egg White Protein/κ-Carrageenan Composite Film and Its Application to Oil Packaging. Food Hydrocoll. 2020, 105, 105780. [Google Scholar] [CrossRef]
- Shahrampour, D.; Khomeiri, M.; Razavi, S.M.A.; Kashiri, M. Development and Characterization of Alginate/Pectin Edible Films Containing Lactobacillus plantarum KMC 45. LWT Food Sci. Technol. 2020, 118, 108758. [Google Scholar] [CrossRef]
- Jiménez-Gómez, C.P.; Cecilia, J.A. Chitosan: A Natural Biopolymer with a Wide and Varied Range of Applications. Molecules 2020, 25, 3981. [Google Scholar] [CrossRef] [PubMed]
- Pérez Córdoba, L.J.; Sobral, P.J.A. Physical and Antioxidant Properties of Films Based on Gelatin, Gelatin-Chitosan or Gelatin-Sodium Caseinate Blends Loaded with Nanoemulsified Active Compounds. J. Food Eng. 2017, 213, 47–53. [Google Scholar] [CrossRef]
- Chakravartula, S.S.N.; Soccio, M.; Lotti, N.; Balestra, F.; Dalla Rosa, M.; Siracusa, V. Characterization of Composite Edible Films Based on Pectin/Alginate/Whey Protein Concentrate. Materials 2019, 12, 2454. [Google Scholar] [CrossRef] [PubMed]
- Azaza, Y.B.; Hamdi, M.; Charmette, C.; Jridi, M.; Li, S.; Nasri, M.; Nasri, R. Development and Characterization of Active Packaging Films Based on Chitosan and Sardinella Protein Isolate: Effects on the Quality and the Shelf Life of Shrimps. Food Packag. Shelf Life 2022, 31, 100796. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Ghaderi, J.; Gómez-Guillén, M.C. Trans-Cinnamaldehyde-Doped Quadripartite Biopolymeric Films: Rheological Behavior of Film-Forming Solutions and Biofunctional Performance of Films. Food Hydrocoll. 2021, 112, 106339. [Google Scholar] [CrossRef]
- Hedayatnia, S.; Tan, C.P.; Joanne Kam, W.L.; Tan, T.B.; Mirhosseini, H. Modification of Physicochemical and Mechanical Properties of a New Bio-Based Gelatin Composite Films through Composition Adjustment and Instantizing Process. LWT Food Sci. Technol. 2019, 116, 108575. [Google Scholar] [CrossRef]
- Lyu, J.S.; Lee, J.S.; Han, J. Development of a Biodegradable Polycaprolactone Film Incorporated with an Antimicrobial Agent via an Extrusion Process. Sci. Rep. 2019, 9, 20236. [Google Scholar] [CrossRef]
- Yoo, S.; Krochta, J.M. Whey Protein-Polysaccharide Blended Edible Film Formation and Barrier, Tensile, Thermal and Transparency Properties. J. Sci. Food Agric. 2011, 91, 2628–2636. [Google Scholar] [CrossRef]
- Othman, S.H.; Kechik, N.R.A.; Shapi’i, R.A.; Talib, R.A.; Tawakkal, I.S.M.A. Water Sorption and Mechanical Properties of Starch/Chitosan Nanoparticle Films. J. Nanomater. 2019, 2019, 3843949. [Google Scholar] [CrossRef]
- Putri, R.D.A.; Fitrianto, R. Edible Film Innovation from Jackfruit Seed Starch (Artocarpus hetrophyllus) with the Addition of Sorbitol and Carrageenan. Int. J. Res. Innov. Entrep. 2020, 1, 14–21. [Google Scholar]
- Dalimunthe, N.F.; Kusumastuti, Y.; Wirawan, S.K. Effect of Carbonate Hydroxyapatite (CHA) on the Properties of Pectin Edible Films. Key Eng. Mater. 2020, 840, 87–92. [Google Scholar] [CrossRef]
- Rahmiatiningrum, N.; Sukardi, S.; Warkoyo, W. Study of Physical Characteristic, Water Vapor Transmission Rate and Inhibition Zones of Edible Films from Aloe Vera (Aloe barbadensis) Incorporated with Yellow Sweet Potato Starch and Glycerol. Food Technol. Halal Sci. J. 2019, 2, 195. [Google Scholar] [CrossRef]
- Ribeiro, A.M.; Estevinho, B.N.; Rocha, F. Preparation and Incorporation of Functional Ingredients in Edible Films and Coatings. Food Bioprocess Technol. 2021, 14, 209–231. [Google Scholar] [CrossRef]
- Apriliyani, M.W.; Purwadi, P.; Manab, A.; Apriliyanti, M.W.; Ikhwan, A.D. Characteristics of Moisture Content, Swelling, Opacity and Transparency with Addition Chitosan as Edible Films/Coating Base on Casein. Adv. J. Food Sci. Technol. 2020, 18, 9–14. [Google Scholar] [CrossRef]
- Fematt-Flores, G.E.; Aguiló-aguayo, I.; Marcos, B.; Camargo-olivas, B.A.; Sánchez-vega, R.; Soto-caballero, M.C.; Salas-salazar, N.A.; Flores-córdova, M.A.; Rodríguez-roque, M.J. Milk Protein-based Edible Films: Influence on Mechanical, Hydrodynamic, Optical and Antioxidant Properties. Coatings 2022, 12, 196. [Google Scholar] [CrossRef]
- Nisar, T.; Alim, A.; Iqbal, M.; Tehseen, S.; Zi-Chao, W.; Guo, Y. Functionality of Different Probiotic Strains Embedded in Citrus Pectin Based Edible Films. Int. J. Food Sci. Technol. 2021, 57, 1005–1015. [Google Scholar] [CrossRef]
- Akman, P.K.; Bozkurt, F.; Dogan, K.; Tornuk, F.; Tamturk, F. Fabrication and Characterization of Probiotic Lactobacillus plantarum Loaded Sodium Alginate Edible Films. J. Food Meas. Charact. 2021, 15, 84–92. [Google Scholar] [CrossRef]
- Ebrahimi, B.; Mohammadi, R.; Rouhi, M.; Mortazavian, A.M.; Shojaee-Aliabadi, S.; Koushki, M.R. Survival of Probiotic Bacteria in Carboxymethyl Cellulose-Based Edible Film and Assessment of Quality Parameters. LWT Food Sci. Technol. 2018, 87, 54–60. [Google Scholar] [CrossRef]
- Quintanilla, P.; Beltrán, M.C.; Molina, A.; Escriche, I.; Molina, M.P. Characteristics of Ripened Tronchón Cheese from Raw Goat Milk Containing Legally Admissible Amounts of Antibiotics. J. Dairy Sci. 2019, 102, 2941–2953. [Google Scholar] [CrossRef]
- Namratha, S.; Sreejit, V.; Preetha, R. Fabrication and Evaluation of Physicochemical Properties of Probiotic Edible Film Based on Pectin–Alginate–Casein Composite. Int. J. Food Sci. Technol. 2020, 55, 1497–1505. [Google Scholar] [CrossRef]
- Odila Pereira, J.; Soares, J.; Sousa, S.; Madureira, A.R.; Gomes, A.; Pintado, M. Edible Films as Carrier for Lactic Acid Bacteria. LWT Food Sci. Technol. 2016, 73, 543–550. [Google Scholar] [CrossRef]
- Garciía-Argueta, I.; Dublaán-Garciía, O.; Quintero-Salazar, B.; Dominguez-Lopez, A.; Goómez-Olivaán, L.M.; Salem, A.-F.Z. Effect of Lactic Acid Bacteria on the Textural Properties of an Edible Film Based on Whey, Inulin and Gelatin. Afr. J. Biotechnol. 2016, 12, 2659–2669. [Google Scholar] [CrossRef]
- Ma, D.; Jiang, Y.; Ahmed, S.; Qin, W.; Liu, Y. Physical and Antimicrobial Properties of Edible Films Containing Lactococcus lactis. Int. J. Biol. Macromol. 2019, 141, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Mbye, M.; Baig, M.A.; AbuQamar, S.F.; El-Tarabily, K.A.; Obaid, R.S.; Osaili, T.M.; Al-Nabulsi, A.A.; Turner, M.S.; Shah, N.P.; Ayyash, M.M. Updates on Understanding of Probiotic Lactic Acid Bacteria Responses to Environmental Stresses and Highlights on Proteomic Analyses. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1110–1124. [Google Scholar] [CrossRef] [PubMed]
- Akinlaja, J.; Sachs, F. The Breakdown of Cell Membranes by Electrical and Mechanical Stress. Biophys. J. 1998, 75, 247–254. [Google Scholar] [CrossRef]
- Iaconelli, C.; Lemetais, G.; Kechaou, N.; Chain, F.; Bermúdez-Humarán, L.G.; Langella, P.; Gervais, P.; Beney, L. Drying Process Strongly Affects Probiotics Viability and Functionalities. J. Biotechnol. 2015, 214, 17–26. [Google Scholar] [CrossRef]
- Pereda, M.; Ponce, A.G.; Marcovich, N.E.; Ruseckaite, R.A.; Martucci, J.F. Chitosan-Gelatin Composites and Bi-Layer Films with Potential Antimicrobial Activity. Food Hydrocoll. 2011, 25, 1372–1381. [Google Scholar] [CrossRef]
- Goy, R.C.; Morais, S.T.B.; Assis, O.B.G. Evaluation of the Antimicrobial Activity of Chitosan and Its Quaternized Derivative on E. coli and S. aureus Growth. Rev. Bras. Farmacogn. 2016, 26, 122–127. [Google Scholar] [CrossRef]
- Heredia-Castro, P.Y.; Reyes-Díaz, R.; Rendón-Rosales, M.Á.; Beltrán-Barrientos, L.M.; Torres-Llanez, M.J.; Estrada-Montoya, M.C.; Hernández-Mendoza, A.; González-Córdova, A.F.; Vallejo-Cordoba, B. Novel Bacteriocins Produced by Lactobacillus fermentum Strains with Bacteriostatic Effects in Milk against Selected Indicator Microorganisms. J. Dairy Sci. 2021, 104, 4033–4043. [Google Scholar] [CrossRef]
- Gao, Z.; Daliri, E.B.M.; Wang, J.U.N.; Liu, D.; Chen, S.; Ye, X.; Ding, T. Inhibitory Effect of Lactic Acid Bacteria on Foodborne Pathogens: A Review. J. Food Prot. 2019, 82, 441–453. [Google Scholar] [CrossRef]
- Mohanty, D.; Misra, S.; Mohapatra, S.; Sahu, P.S. Prebiotics and Synbiotics: Recent Concepts in Nutrition. Food Biosci. 2018, 26, 152–160. [Google Scholar] [CrossRef]
- Fooks, L.J.; Gibson, G.R. In Vitro Investigations of the Effect of Probiotics and Prebiotics on Selected Human Intestinal Pathogens. FEMS Microbiol. Ecol. 2002, 39, 67–75. [Google Scholar] [CrossRef]

| Edible Films | Thickness (mm) | Moisture Content (%) | Water Solubility (%) |
|---|---|---|---|
| CS | 0.09 | 24.8 ± 1.2 A | 32.9 ± 1.9 B |
| NaCas | 0.14 | 10.0 ± 0.8 C | 55.5 ± 1.9 A |
| CS/NaCas | 0.12 ± 0.01 B | 17.2 ± 0.4 B | 27.6 ± 3.9 B |
| Edible Films | Opacity (A/mm) | L* | a* | b* | ΔE |
|---|---|---|---|---|---|
| CS | 2.22 ± 0.01 B | 8.53 ± 1.02 C | −0.65 ± 0.06 A | −0.97 ± 0.08 B | 8.62 ± 1.01 C |
| NaCas | 1.65 ± 0.16 B | 13.41 ± 0.95 B | −0.90 ± 0.12 B | −1.85 ± 0.24 C | 13.58 ± 0.90 B |
| CS/NaCas | 7.40 ± 0.65 A | 21.20 ± 1.12 A | −0.50 ± 0.07 A | −0.42 ± 0.06 A | 21.25 ± 1.14 A |
| Edible Films | Tensile Strength (MPa) | Elongation at Break (%) | Young’s Modulus (MPa) |
|---|---|---|---|
| CS | 41.5 ± 3.0 B | 96.9 ± 3.1 A | 0.43 ± 0.04 B |
| NaCas | 57.8 ± 5.1 A | 8.4 ± 0.4 B | 6.92 ± 0.66 A |
| CS/NaCas | 28.5 ± 2.5 C | 106.6 ± 7.5 A | 0.27 ± 0.01 B |
| Edible Films | Thickness (mm) | Moisture Content (%) | Water Solubility (%) |
|---|---|---|---|
| CS/NaCas | 0.12 ± 0.01 A | 17.23 ± 0.41 A | 27.59 ± 3.86 A |
| CS/NaCas–Lb. fermentum | 0.11 ± 0.01 A | 17.94 ± 2.18 A | 30.80 ± 3.02 A |
| Edible Films | Opacity (A/mm) | L* | a* | b* | ΔE |
|---|---|---|---|---|---|
| CS/NaCas | 7.4 ± 0.6 A | 21.2 ± 1.1 A | −0.50 ± 0.07 A | −0.42 ± 0.06 A | 21.2 ± 1.1 A |
| CS/NaCas–Lb. fermentum | 8.7 ± 0.9 A | 21.7 ± 0.9 A | −0.37 ± 0.04 A | −0.31 ± 0.05 A | 21.7 ± 0.9 A |
| Edible Films | Tensile Strength (MPa) | Elongation at Break (%) | Young’s Modulus (MPa) |
|---|---|---|---|
| CS/NaCas | 28.46 ± 2.45 A | 106.57 ± 7.51 A | 0.27 ± 0.01 A |
| CS/NaCas–Lb. fermentum | 20.94 ± 2.48 B | 88.80 ± 9.12 A | 0.24 ± 0.01 B |
| Edible Films | Escherichia coli | Staphylococcus aureus |
|---|---|---|
| CS/NaCas | 0.37 ± 0.06 Bb | 0.47 ± 0.06 Bb |
| CS/NaCas–Lb. fermentum | 0.67 ± 0.06 Aa | 0.80 ± 0.05 Aa |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wai, S.N.; How, Y.H.; Saleena, L.A.K.; Degraeve, P.; Oulahal, N.; Pui, L.P. Chitosan–Sodium Caseinate Composite Edible Film Incorporated with Probiotic Limosilactobacillus fermentum: Physical Properties, Viability, and Antibacterial Properties. Foods 2022, 11, 3583. https://doi.org/10.3390/foods11223583
Wai SN, How YH, Saleena LAK, Degraeve P, Oulahal N, Pui LP. Chitosan–Sodium Caseinate Composite Edible Film Incorporated with Probiotic Limosilactobacillus fermentum: Physical Properties, Viability, and Antibacterial Properties. Foods. 2022; 11(22):3583. https://doi.org/10.3390/foods11223583
Chicago/Turabian StyleWai, Seat Ni, Yu Hsuan How, Lejaniya Abdul Kalam Saleena, Pascal Degraeve, Nadia Oulahal, and Liew Phing Pui. 2022. "Chitosan–Sodium Caseinate Composite Edible Film Incorporated with Probiotic Limosilactobacillus fermentum: Physical Properties, Viability, and Antibacterial Properties" Foods 11, no. 22: 3583. https://doi.org/10.3390/foods11223583
APA StyleWai, S. N., How, Y. H., Saleena, L. A. K., Degraeve, P., Oulahal, N., & Pui, L. P. (2022). Chitosan–Sodium Caseinate Composite Edible Film Incorporated with Probiotic Limosilactobacillus fermentum: Physical Properties, Viability, and Antibacterial Properties. Foods, 11(22), 3583. https://doi.org/10.3390/foods11223583





