In Vitro Protein Digestion of Cooked Spent Commercial Laying Hen and Commercial Broilers Breast Meat
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design
2.3. In Vitro Enzymatic Digestion
2.4. Bicinchoninic Acid (BCA) Assay Quantitative Analysis
2.5. Trinitrobezenesulfonic (TNBS) Acid Method
2.6. Gas Chromatography-Mass Spectrometry (GC-MS)
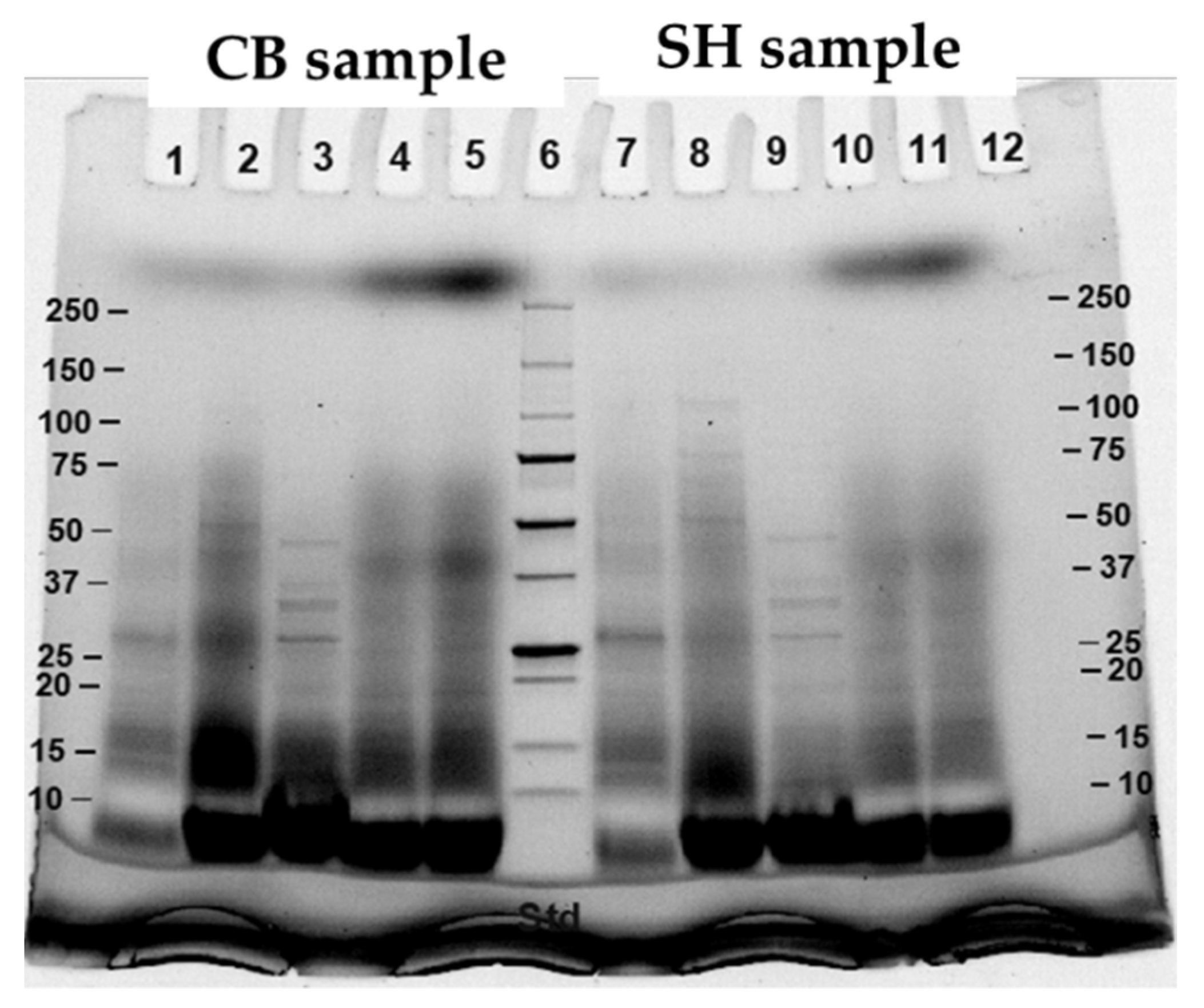
2.7. Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE)
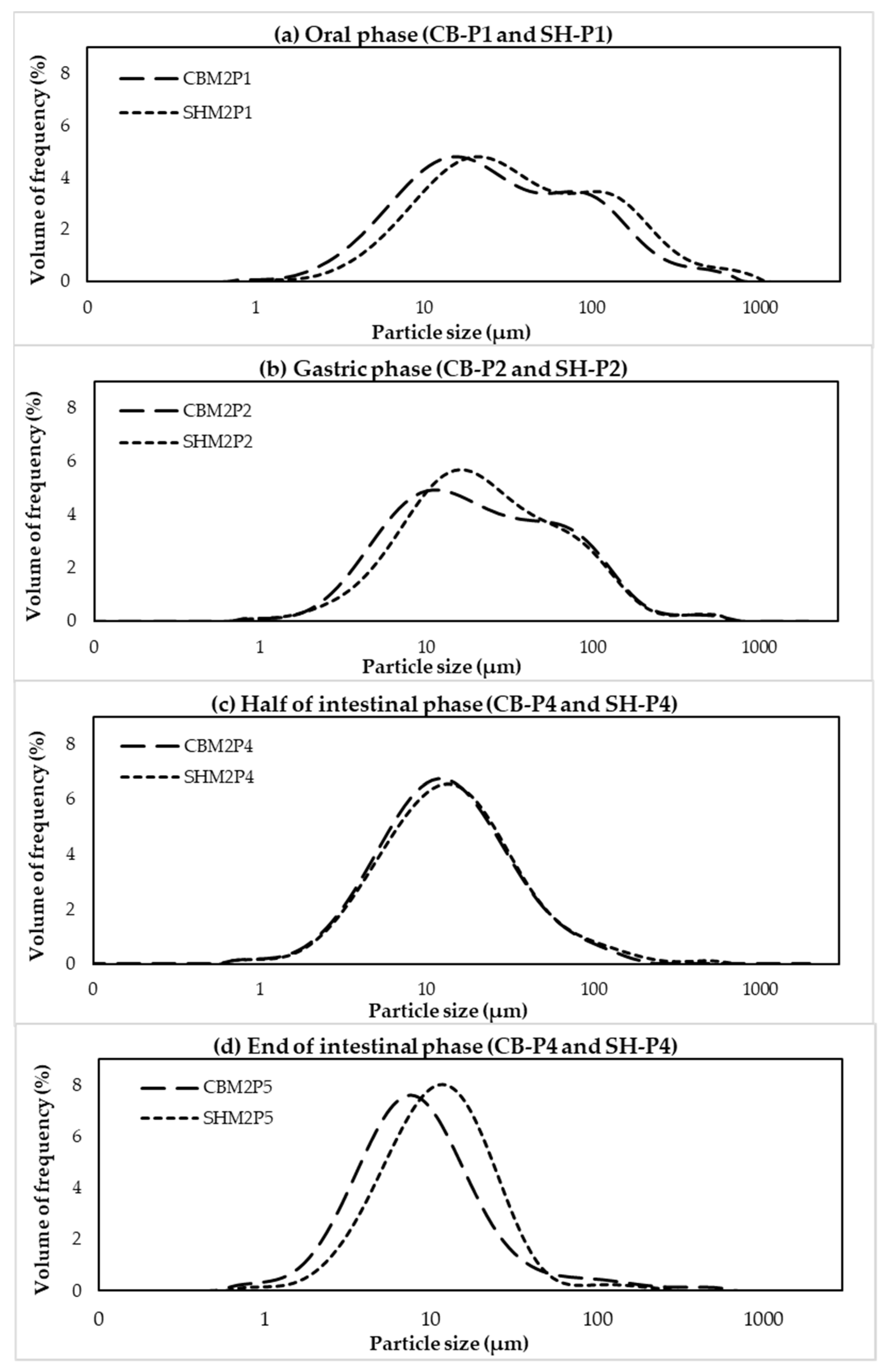
2.8. Particle Size Distribution
2.9. Statistical Analysis
3. Results and Discussion
3.1. Effects of Different In Vitro Digestion Methods on Protein Digestibility of Cooked CB Breast Meat
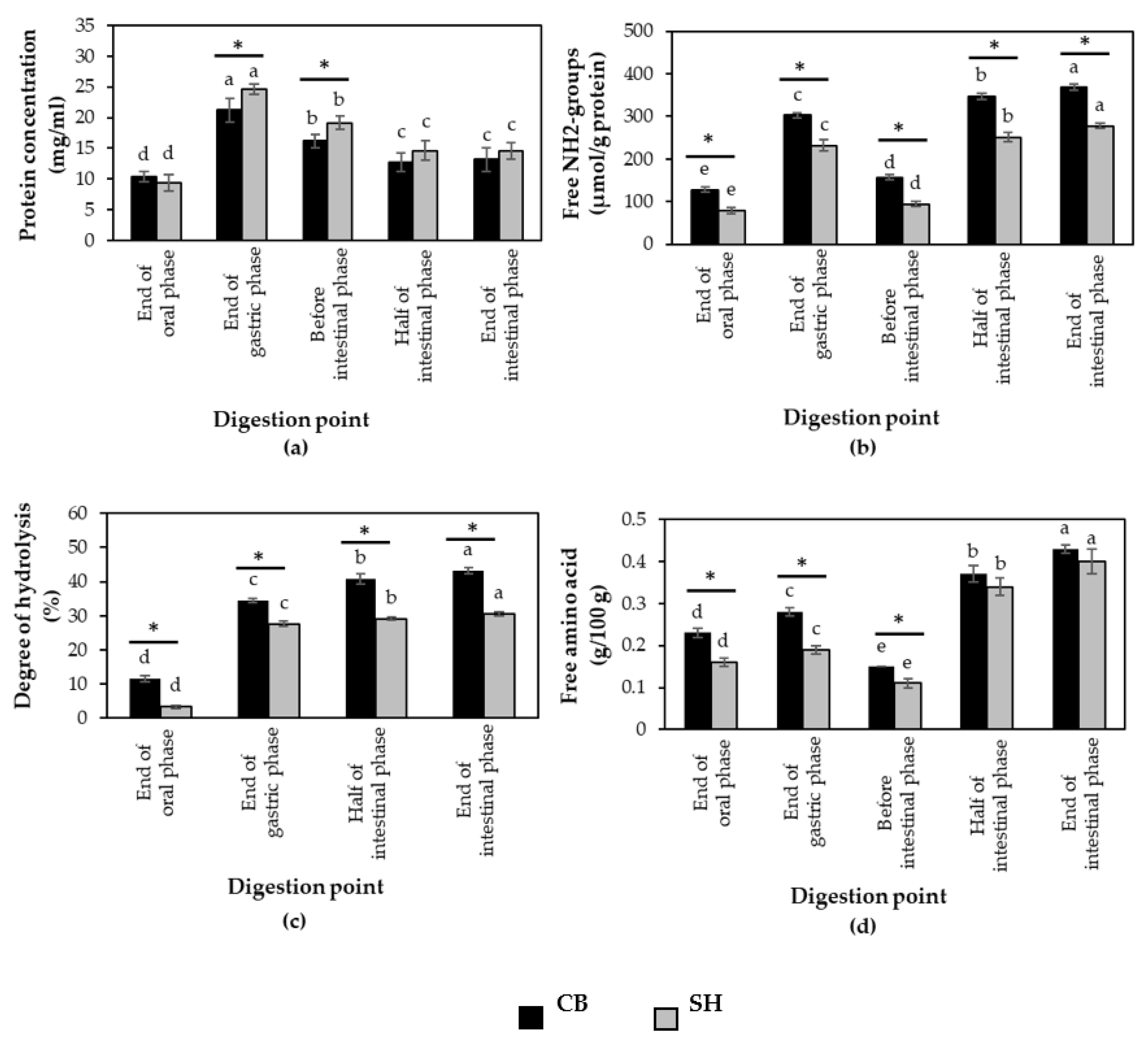
3.2. Protein Digestibility of Cooked SH and CB Breast Samples during In Vitro Digestion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Population Prospects: The 2019 Revision. Available online: https://population.un.org/wpp/ (accessed on 1 April 2022).
- Jach, M.E.; Malm, A. Yarrowia lipolytica as an Alternative and Valuable Source of Nutritional and Bioactive Compounds for Humans. Molecules 2022, 27, 2300. [Google Scholar] [CrossRef] [PubMed]
- Akande, O.A.; Oluwamukomi, M.; Osundahunsi, O.F.; Ijarotimi, O.S.; Mukisa, I.M. Evaluating the potential for utilising migratory locust powder (Locusta migratoria) as an alternative protein source in peanut-based ready-to-use therapeutic foods. Food Sci. Techn. Int. 2022, 10820132211069773. [Google Scholar] [CrossRef] [PubMed]
- Malla, N.; Nørgaard, J.V.; Lærke, H.N.; Heckmann, L.-H.L.; Roos, N. Some Insect Species Are Good-Quality Protein Sources for Children and Adults: Digestible Indispensable Amino Acid Score (DIAAS) Determined in Growing Pigs. J. Nutr. 2022, 152, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Onwezen, M.C.; Bouwman, E.P.; Reinders, M.J.; Dagevos, H. A systematic review on consumer acceptance of alternative proteins: Pulses, algae, insects, plant-based meat alternatives, and cultured meat. Appetite 2021, 159, 105058. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K. Nutritional value of proteins and protein requirements of people with special reference to meat proteins. Mitteilungsbl. Bundesanst. Fleischforsch. 1993, 32, 422–429. [Google Scholar]
- Escudero, E.; Sentandreu, M.A.; Toldra, F. Characterization of peptides released by in vitro digestion of pork meat. J. Agric. Food Chem. 2010, 58, 5160–5165. [Google Scholar] [CrossRef]
- Lakshani, P.; Jayasena, D.D.; Jo, C. Comparison of quality traits of breast meat from commercial broilers and spent hens in Sri Lanka. Korean J. Poult. Sci. 2016, 43, 55–61. [Google Scholar] [CrossRef][Green Version]
- Suriani, N.W.; Purnomo, H.; Estiasih, T.; Suwetja, I.K. Physicochemical properties, fatty acids profile and cholesterol content of indigenous manado chicken, broiler and spent hen meat. Int. J. ChemTech Res. 2014, 6, 3896–3902. [Google Scholar]
- Abdalla, H.; Ali, N.; Siddig, F.; Ali, S. Improving tenderness of spent layer hens meat using papaya leaves (Carica papaya). Pak. Vet. J. 2013, 33, 73–76. [Google Scholar]
- Navid, S.; Sheikhlar, A.; Kaveh, K. Influence of the combination of vitamin D3 and papaya leaf on meat quality of spent layer hen. Agric. J. 2011, 6, 197–200. [Google Scholar] [CrossRef][Green Version]
- Limpisophon, K.; Koeipudsa, C.; Charoensuk, D.; Malila, Y. Characterization of breast meat collected from spent lohmann brown layers in comparison to commercial ross broilers. Braz. J. Poult. Sci. 2019, 21. [Google Scholar] [CrossRef]
- Savoie, L.; Agudelo, R.A.; Gauthier, S.F.; Marin, J.; Pouliot, Y. In vitro determination of the release kinetics of peptides and free amino acids during the digestion of food proteins. J. AOAC Int. 2005, 88, 935–948. [Google Scholar] [CrossRef] [PubMed]
- Freeman, H.J.; Kim, Y.S.; Sleisenger, M.H. Protein digestion and absorption in man: Normal mechanisms and protein-energy malnutrition. Am. J. Med. 1979, 67, 1030–1036. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D. A standardised static in vitro digestion method suitable for food–an international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef]
- Egger, L.; Ménard, O.; Delgado-Andrade, C.; Alvito, P.; Assunção, R.; Balance, S.; Barberá, R.; Brodkorb, A.; Cattenoz, T.; Clemente, A. The harmonized INFOGEST in vitro digestion method: From knowledge to action. Food Res. Int. 2016, 88, 217–225. [Google Scholar] [CrossRef]
- Bordoni, A.; Laghi, L.; Babini, E.; Di Nunzio, M.; Picone, G.; Ciampa, A.; Valli, V.; Danesi, F.; Capozzi, F. The foodomics approach for the evaluation of protein bioaccessibility in processed meat upon in vitro digestion. Electrophoresis 2014, 35, 1607–1614. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of the AOAC International, 17th ed.; Gaithersburg AOAC International: Gaithersburg, MD, USA, 2000; pp. 2201–3301. [Google Scholar]
- He, F. BCA (bicinchoninic acid) protein assay. Bio-Protocol 2011, 1, e44. [Google Scholar] [CrossRef]
- Kristoffersen, K.A.; Afseth, N.K.; Böcker, U.; Lindberg, D.; de Vogel-van den Bosch, H.; Ruud, M.L.; Wubshet, S.G. Average molecular weight, degree of hydrolysis and dry-film FTIR fingerprint of milk protein hydrolysates: Intercorrelation and application in process monitoring. Food Chem. 2020, 310, 125800. [Google Scholar] [CrossRef]
- Adler-Nissen, J. Enzymatic hydrolysis of food proteins. Process Biochem. 1977, 12, 18–23. [Google Scholar]
- Nielsen, P.; Petersen, D.; Dambmann, C. Improved method for determining food protein degree of hydrolysis. J. Food Sci. 2001, 66, 642–646. [Google Scholar] [CrossRef]
- Jiménez-Martín, E.; Ruiz, J.; Pérez-Palacios, T.; Silva, A.; Antequera, T. Gas chromatography–mass spectrometry method for the determination of free amino acids as their dimethyl-tert-butylsilyl (TBDMS) derivatives in animal source food. J. Agric. Food Chem. 2012, 60, 2456–2463. [Google Scholar] [CrossRef] [PubMed]
- Grabski, A.; Burgess, R. Preparation of protein samples for SDS-polyacrylamide gel electrophoresis: Procedures and tips. inNovations 2001, 13, 10–12. [Google Scholar]
- Palka, K. Heat effects on meat | Physics and Chemistry. In Encyclopedia of Meat Sciences; Jensen, W.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2004; pp. 567–570. [Google Scholar]
- Chelh, I.; Gatellier, P.; Santé-Lhoutellier, V. A simplified procedure for myofibril hydrophobicity determination. Meat Sci. 2006, 74, 681–683. [Google Scholar] [CrossRef]
- Santé-Lhoutellier, V.; Astruc, T.; Marinova, P.; Greve, E.; Gatellier, P. Effect of meat cooking on physicochemical state and in vitro digestibility of myofibrillar proteins. J. Agric. Food Chem. 2008, 56, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Palka, K.; Wesierska, E. Cooking of meat | Physics and Chemistry. In Encyclopedia of Meat Sciences; Jensen, W.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 2, pp. 404–409. [Google Scholar]
- Hsu, H.; Vavak, D.; Satterlee, L.; Miller, G. A multienzyme technique for estimating protein digestibility. J. Food Sci. 1977, 42, 1269–1273. [Google Scholar] [CrossRef]
- Liu, B.-Y.; Zhu, K.-X.; Peng, W.; Guo, X.-N.; Zhou, H.-M. Effect of sequential hydrolysis with endo-and exo-peptidase on bitterness properties of wheat gluten hydrolysates. RSC Adv. 2016, 6, 27659–27668. [Google Scholar] [CrossRef]
- Lengkidworraphiphat, P.; Wongpoomchai, R.; Bunmee, T.; Chariyakornkul, A.; Chaiwang, N.; Jaturasitha, S. Taste-Active and Nutritional Components of Thai Native Chicken Meat: A Perspective of Consumer Satisfaction. Food Sci. Anim. Resour. 2021, 41, 237–246. [Google Scholar] [CrossRef]
- Wu, H.-C.; Shiau, C.-Y. Proximate composition, free amino acids and peptides contents in commercial chicken and other meat essences. J. Food Drug Anal. 2002, 10, 170–177. [Google Scholar] [CrossRef]
- Rikimaru, K.; Takahashi, H. Evaluation of the meat from Hinai-jidori chickens and broilers: Analysis of general biochemical components, free amino acids, inosine 5’-monophosphate, and fatty acids. J. Appl. Poult. Res. 2010, 19, 327–333. [Google Scholar] [CrossRef]
- Ali, M.; Lee, S.-Y.; Park, J.-Y.; Jung, S.; Jo, C.; Nam, K.-C. Comparison of functional compounds and micronutrients of chicken breast meat by breeds. Food Sci. Anim. Resour. 2019, 39, 632. [Google Scholar] [CrossRef]
- Smith-Mungo, L.I.; Kagan, H.M. Lysyl oxidase: Properties, regulation and multiple functions in biology. Matrix Biol. 1998, 16, 387–398. [Google Scholar] [CrossRef]
- Depalle, B.; Qin, Z.; Shefelbine, S.J.; Buehler, M.J. Influence of cross-link structure, density and mechanical properties in the mesoscale deformation mechanisms of collagen fibrils. J. Mech. Behav. Biomed. Mater. 2015, 52, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.J.; Paul, R.G.; Knott, L. Mechanisms of maturation and ageing of collagen. Mech. Ageing Dev. 1998, 106, 1–56. [Google Scholar] [CrossRef]
- van der Slot-Verhoeven, A.J.; van Dura, E.A.; Attema, J.; Blauw, B.; DeGroot, J.; Huizinga, T.W.; Zuurmond, A.-M.; Bank, R.A. The type of collagen cross-link determines the reversibility of experimental skin fibrosis. Biochim. Biophys. Acta-Mol. Basis Dis. 2005, 1740, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Chaplot, S.; Chalamaiah, M.; Roy, B.C.; Bruce, H.L.; Wu, J. Removing cross-linked telopeptides enhances the production of low-molecular-weight collagen peptides from spent hens. J. Agric. Food Chem. 2017, 65, 7491–7499. [Google Scholar] [CrossRef] [PubMed]
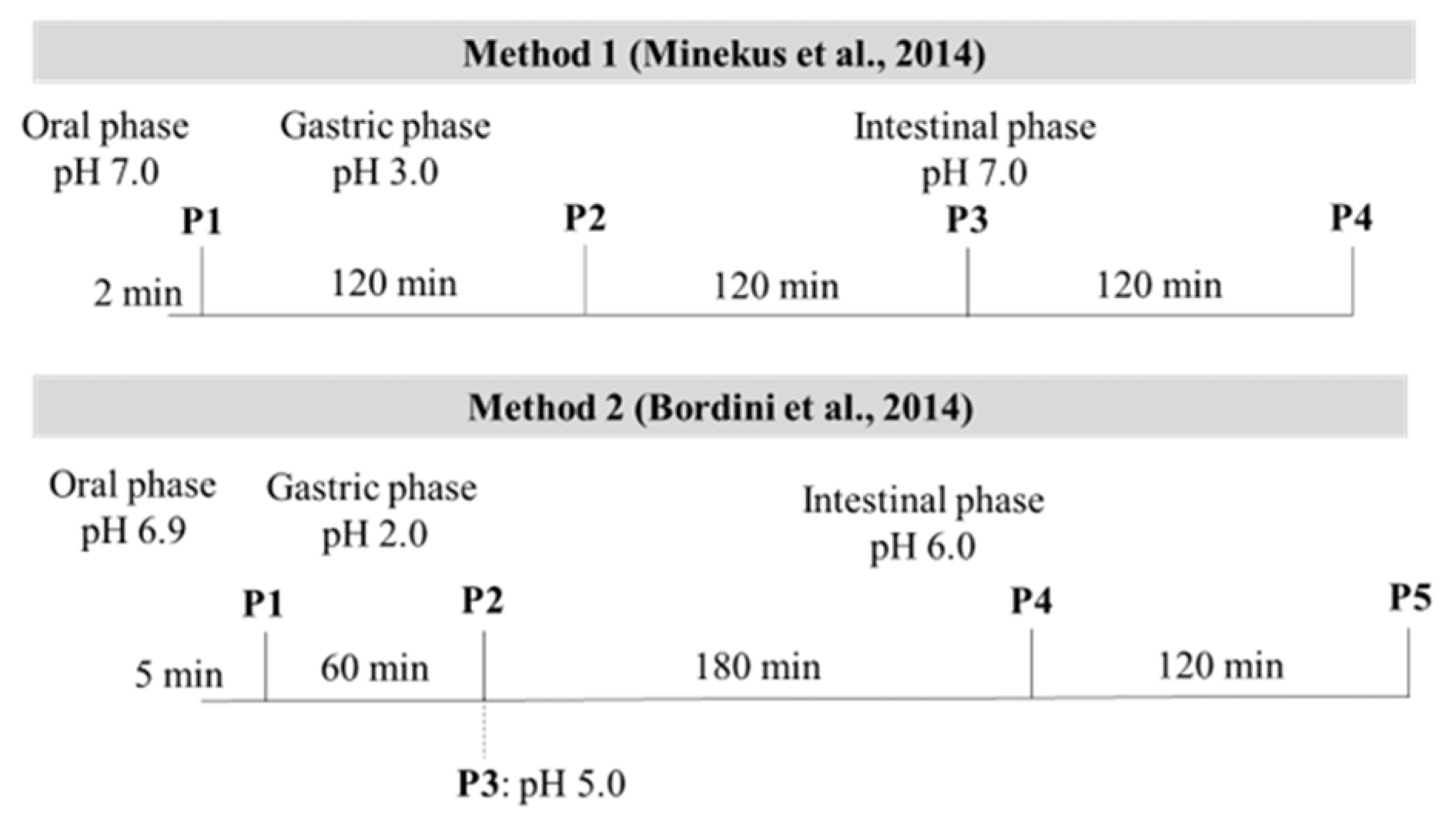
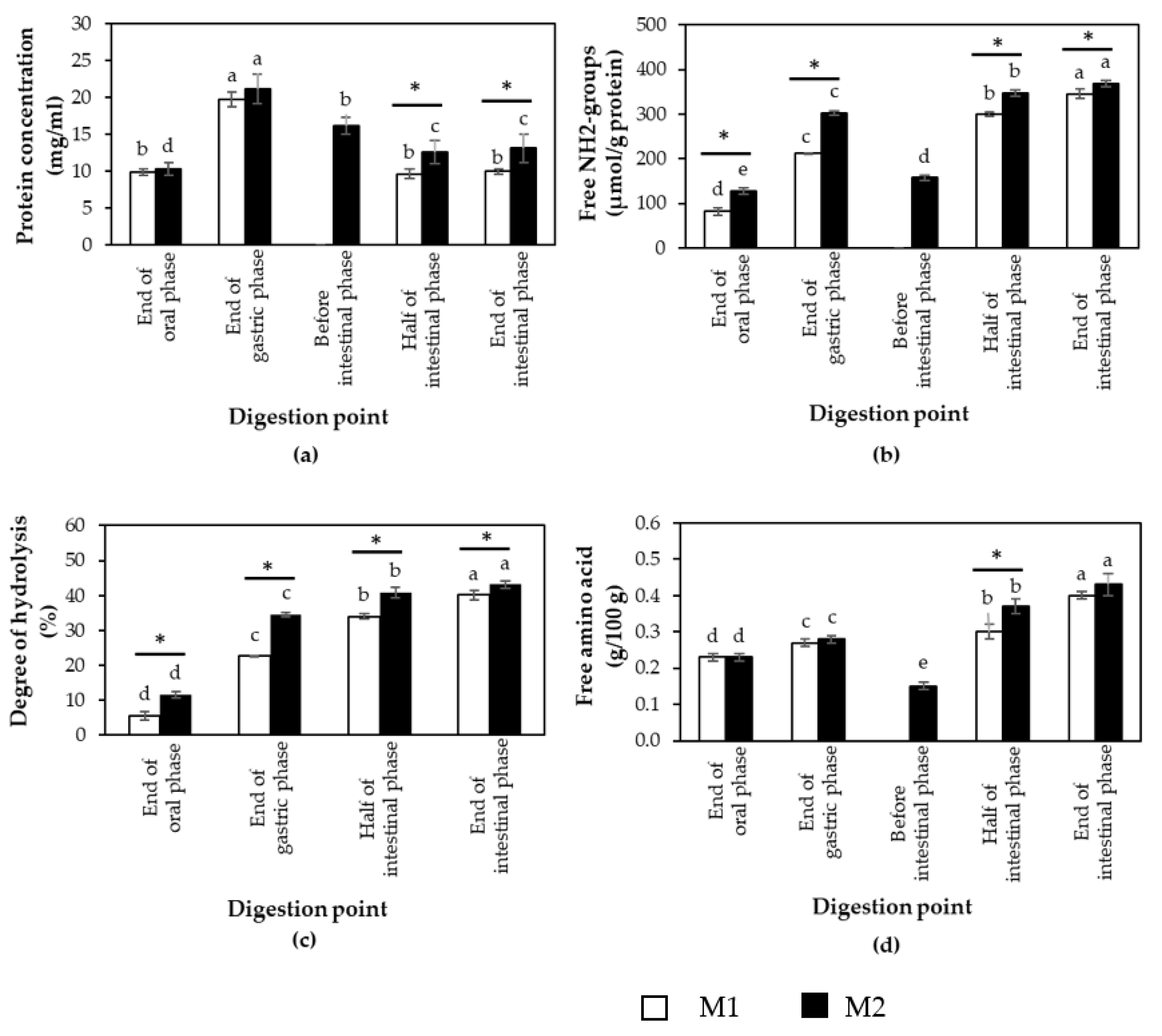
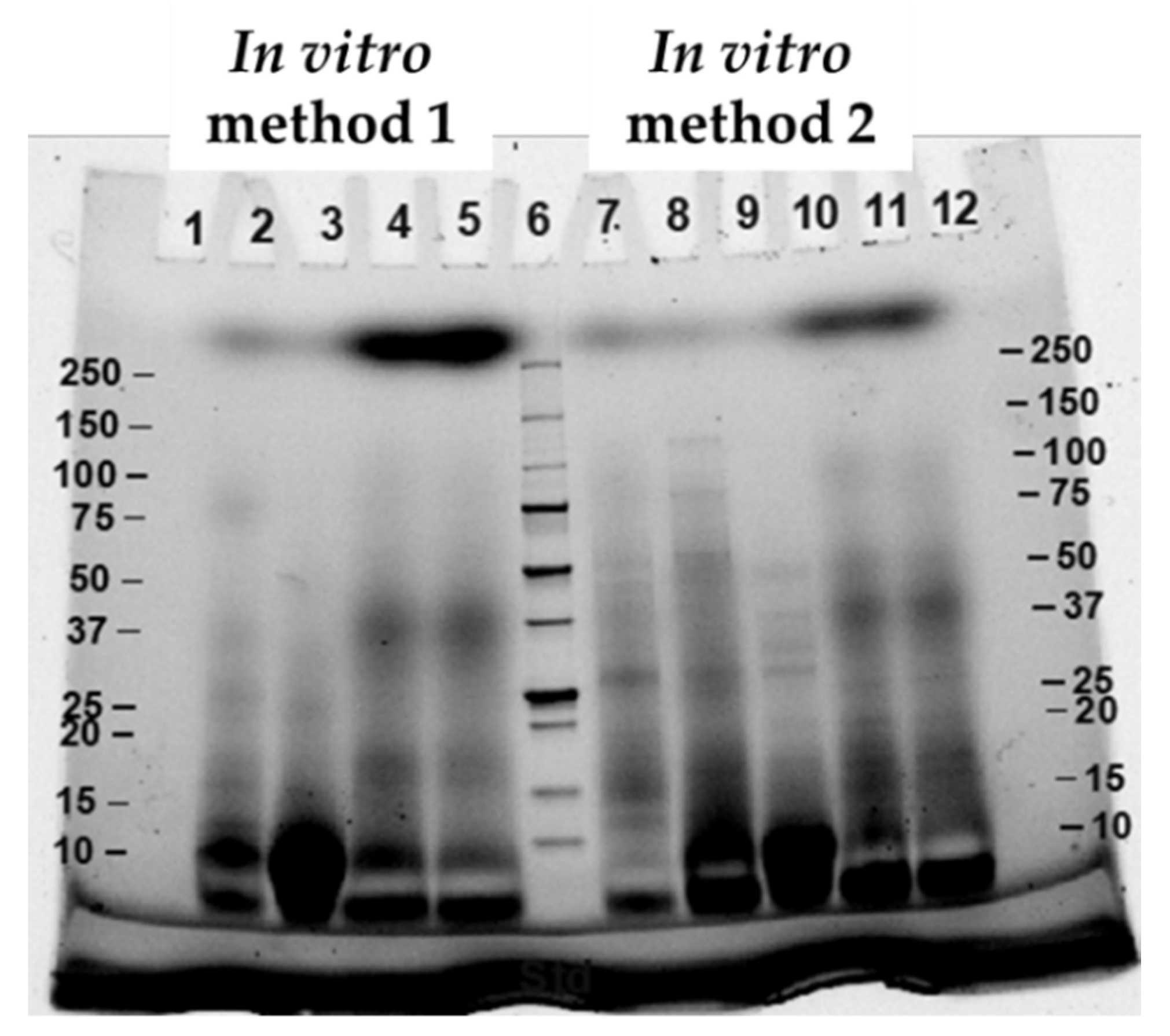
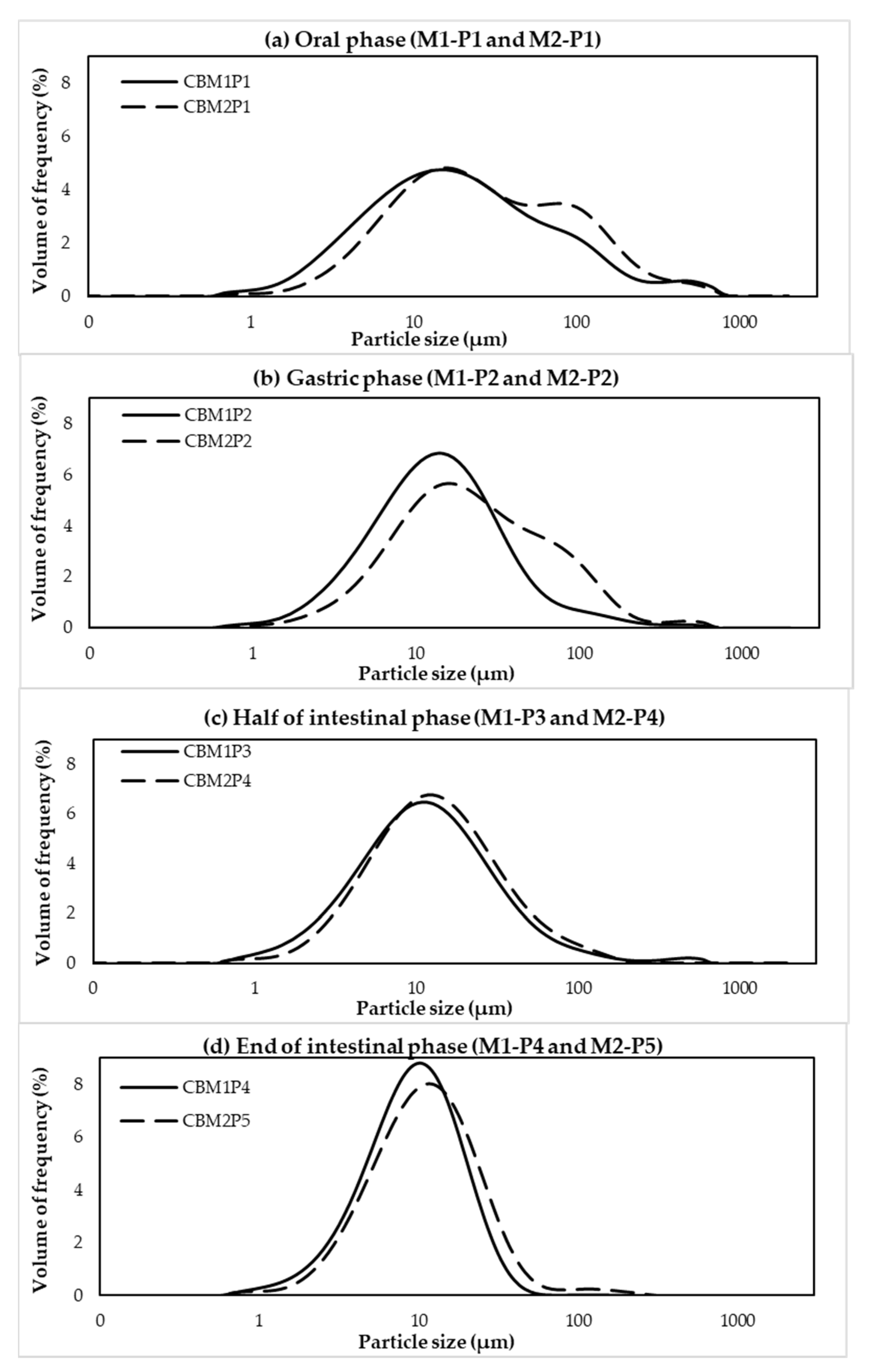
| Constituent | Stock Concentration (mol/L) | SSF (pH 7) (mL) | SGF (pH 3) (mL) | SIF (pH 7) (mL) |
|---|---|---|---|---|
| KCl | 0.5 | 15.1 | 6.9 | 6.8 |
| KH2PO4 | 0.5 | 3.7 | 0.9 | 0.8 |
| NaHCO3 | 1.0 | 6.8 | 12.5 | 42.5 |
| NaCl | 2.0 | - | 11.8 | 9.6 |
| MgCl2(H2O)6 | 0.15 | 0.5 | 0.4 | 1.1 |
| (NH4)2CO3 | 0.5 | 0.06 | 0.5 | - |
| CaCl2(H2O)2 | 0.3 | 0.09 | 1.3 | 0.7 |
| Digestion Point | D[4,3] (µm) 1,2 | Effect of Different In Vitro Methods within Each Digestion Point 3 | |
|---|---|---|---|
| M1 | M2 | ||
| End of oral phase | 45.50 a ± 0.04 | 53.28 a ± 1.23 | * |
| End of gastric phase | 22.96 b ± 0.96 | 35.59 b ± 1.19 | * |
| Before intestinal phase | - | 51.68 a ± 1.25 | - |
| Half of intestinal phase | 22.03 b ± 1.75 | 20.20 c ± 3.38 | ns |
| End of intestinal phase | 19.59 b ± 2.19 | 17.19 c ± 1.69 | ns |
| Amino Acid (mg/100 g) | CBP1 1 | SHP1 1 | Significant Difference 2 | CBP5 1 | SHP5 1 | Significant Difference 2 |
|---|---|---|---|---|---|---|
| Essential amino acids | ||||||
| Valine | 6.1 ± 0.5 | 5.3 ± 1.4 | ns | 5.2 ± 0.1 | 4.5 ± 0.5 | ns |
| Leucine | 8.6 ± 0.3 | 7.8 ± 0.8 | ns | 25.8 ± 0.0 | 27.0 ± 1.3 | ns |
| Isoleucine | 4.7 ± 0.3 | 3.9 ± 0.2 | * | 5.4 ± 0.2 | 5.1 ± 0.3 | ns |
| Methionine | 3.7 ± 0.1 | 3.5 ± 0.5 | ns | 6.4 ± 0.4 | 6.6 ± 0.4 | ns |
| Threonine | 8.2 ± 0.8 | 5.5 ± 0.5 | * | 5.7 ± 0.5 | 3.7 ± 0.3 | * |
| Phenylalanine | 5.8 ± 0.5 | 5.2 ± 0.6 | ns | 22.6 ± 4.9 | 28.7 ± 1.7 | ns |
| Lysine | 10.2 ± 0.3 | 7.2 ± 1.1 | * | 32.1 ± 2.4 | 33.0 ± 2.9 | ns |
| Histidine | 7.7 ± 0.3 | 7.1 ± 1.3 | ns | 5.9 ± 0.2 | 5.4 ± 0.4 | * |
| Tryptophan | 11.4 ± 0.6 | 9.3 ± 0.3 | * | 25.8 ± 1.1 | 19.2 ± 3.2 | * |
| Non-essential amino acids | ||||||
| Alanine | 10.8 ± 1.5 | 6.4 ± 1.2 | * | 7.2 ± 0.8 | 4.4 ± 0.4 | * |
| Glycine | 8.1 ± 1.1 | 3.5 ± 0.2 | * | 11.0 ± 0.6 | 9.1 ± 0.3 | * |
| Proline | 26.9 ± 0.8 | 16.2 ± 0.8 | * | 31.3 ± 2.1 | 26.5 ± 1.3 | * |
| Serine | 7.3 ± 0.7 | 4.8 ± 0.9 | * | 4.8 ± 0.5 | 3.1 ± 0.3 | * |
| Aspartic acid | 6.7 ± 1.0 | 3.5 ± 1.1 | * | 4.0 ± 0.7 | 2.0 ± 0.4 | * |
| Hydroxyproline | 1.1 ± 0.1 | 0.5 ± 0.0 | * | 0.8 ± 0.1 | 0.4 ± 0.0 | * |
| Cysteine | 2.4 ± 0.1 | 2.2 ± 0.1 | * | 3.2 ± 0.2 | 3.0 ± 0.2 | ns |
| Glutamic acid | 32.6 ± 2.4 | 21.5 ± 5.9 | * | 15.0 ± 1.4 | 10.7 ± 2.3 | * |
| Arginine | 3.2 ± 0.5 | 1.9 ± 0.5 | * | 11.4 ± 0.9 | 10.6 ± 0.6 | ns |
| Tyrosine | 10.3 ± 0.3 | 8.4 ± 0.9 | * | 37.3 ± 1.9 | 37.9 ± 2.4 | ns |
| Digestion Point | D[4,3] (µm) 1,2 | Effect of Different Chicken Types within Each Digestion Point 3 | |
|---|---|---|---|
| CB | SH | ||
| End of oral phase | 53.28 a ± 1.23 | 133.17 a ± 2.16 | * |
| End of gastric phase | 35.59 b ± 1.19 | 46.52 c ± 2.20 | * |
| Before intestinal phase | 51.68 a ± 1.25 | 112.96 b ± 3.63 | * |
| Half of intestinal phase | 20.20 c ± 3.38 | 22.45 d ± 2.74 | ns |
| End of intestinal phase | 17.19 c ± 1.69 | 17.52 d ± 2.46 | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trithavisup, T.; Sanpinit, P.; Sakulwech, S.; Klamchuen, A.; Malila, Y. In Vitro Protein Digestion of Cooked Spent Commercial Laying Hen and Commercial Broilers Breast Meat. Foods 2022, 11, 1853. https://doi.org/10.3390/foods11131853
Trithavisup T, Sanpinit P, Sakulwech S, Klamchuen A, Malila Y. In Vitro Protein Digestion of Cooked Spent Commercial Laying Hen and Commercial Broilers Breast Meat. Foods. 2022; 11(13):1853. https://doi.org/10.3390/foods11131853
Chicago/Turabian StyleTrithavisup, Thanatorn, Pornnicha Sanpinit, Sakhiran Sakulwech, Annop Klamchuen, and Yuwares Malila. 2022. "In Vitro Protein Digestion of Cooked Spent Commercial Laying Hen and Commercial Broilers Breast Meat" Foods 11, no. 13: 1853. https://doi.org/10.3390/foods11131853
APA StyleTrithavisup, T., Sanpinit, P., Sakulwech, S., Klamchuen, A., & Malila, Y. (2022). In Vitro Protein Digestion of Cooked Spent Commercial Laying Hen and Commercial Broilers Breast Meat. Foods, 11(13), 1853. https://doi.org/10.3390/foods11131853







