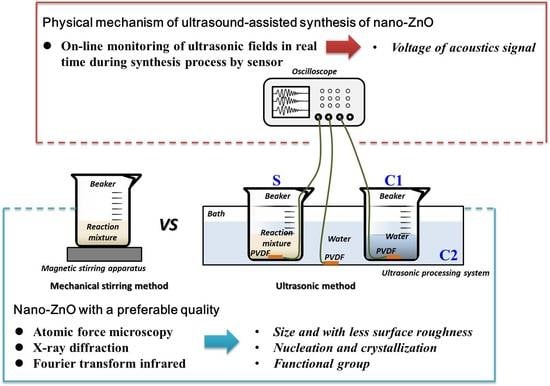
Ultrasound-Assisted Synthesis of Potentially Food-Grade Nano-Zinc Oxide in Ionic Liquids: A Safe, Green, Efficient Approach and Its Acoustics Mechanism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Materials and Apparatus
2.2. Synthesis of Nano-ZnO by Mechanical Stirring Method
2.3. Synthesis of Nano-ZnO by Ultrasonic Method
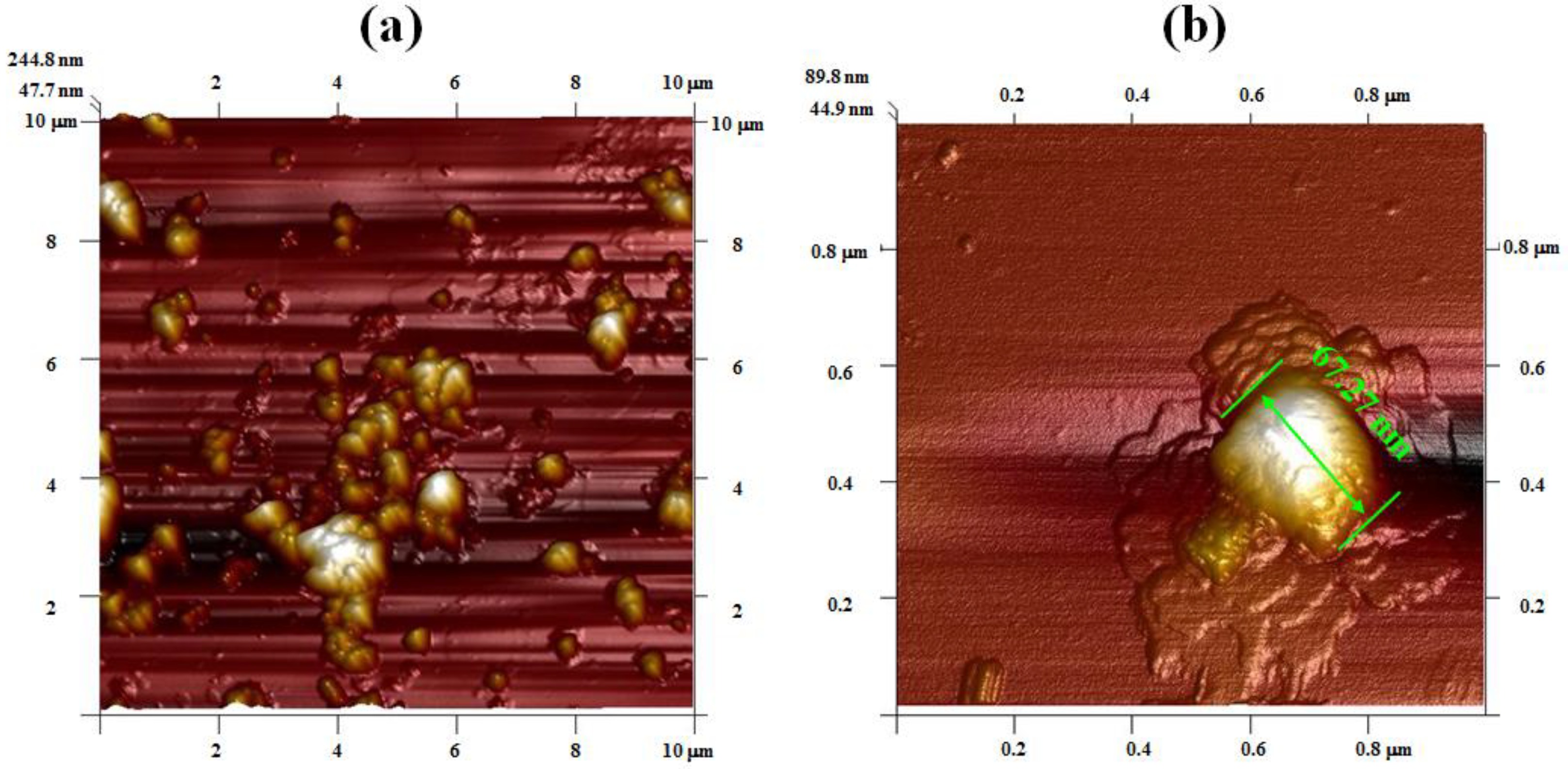
2.4. Determination of Nano-ZnO Micromorphology by Atomic Force Microscopy (AFM)
2.5. Determination of Nano-ZnO Structure by X-ray Diffraction (XRD)
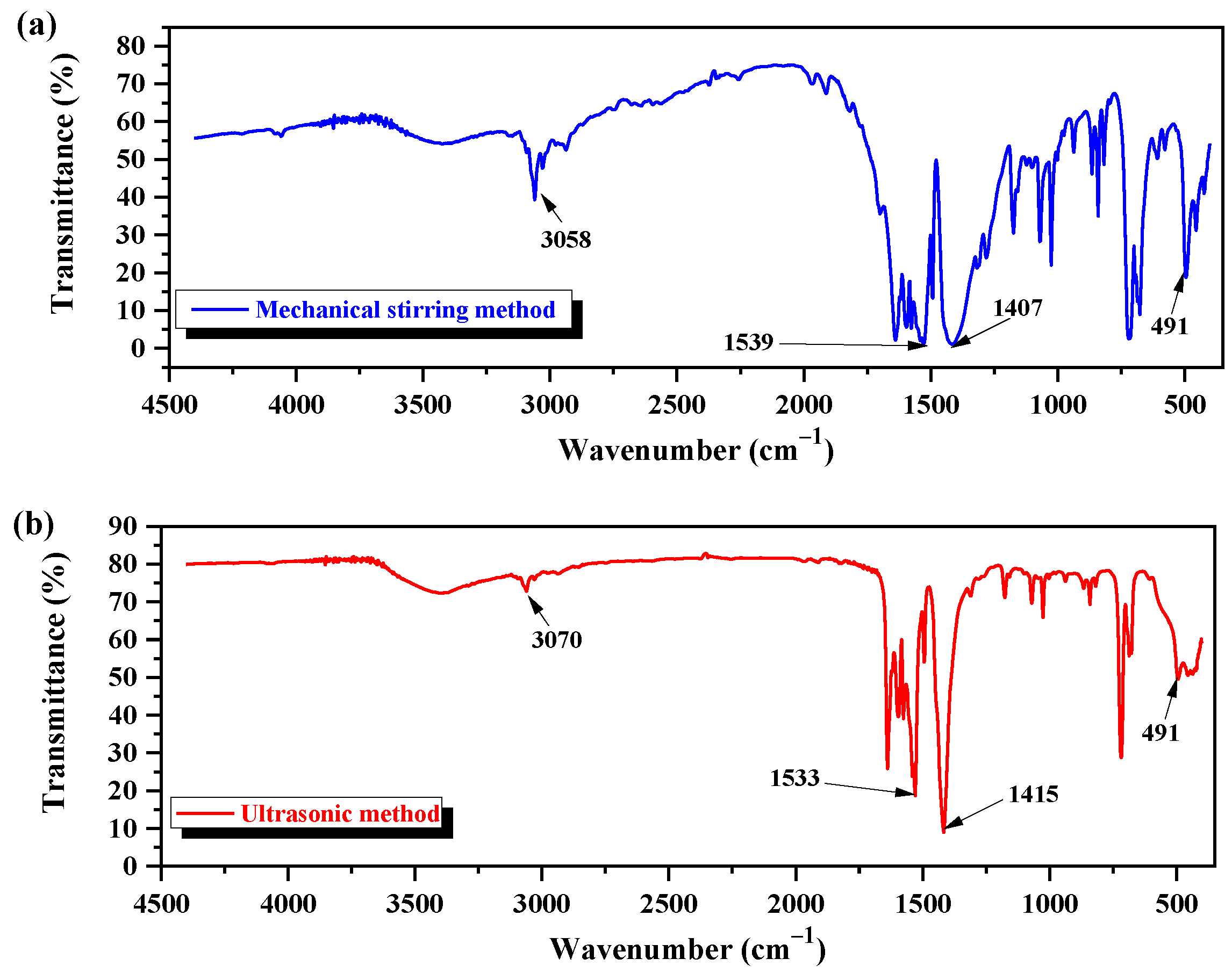
2.6. Determination of Functional Groups of Nano-ZnO by FOURIER Transform Infrared (FTIR) Spectroscopy
2.7. Monitoring of Ultrasonic Fields during Nano-ZnO Synthesis Process by Ultrasonic Method
3. Results
3.1. Characterization of Nano-ZnO Micromorphology by AFM
3.2. Characterization of Nano-ZnO Crystal Structure by XRD
3.3. Characterization of Nano-ZnO Structure by FTIR
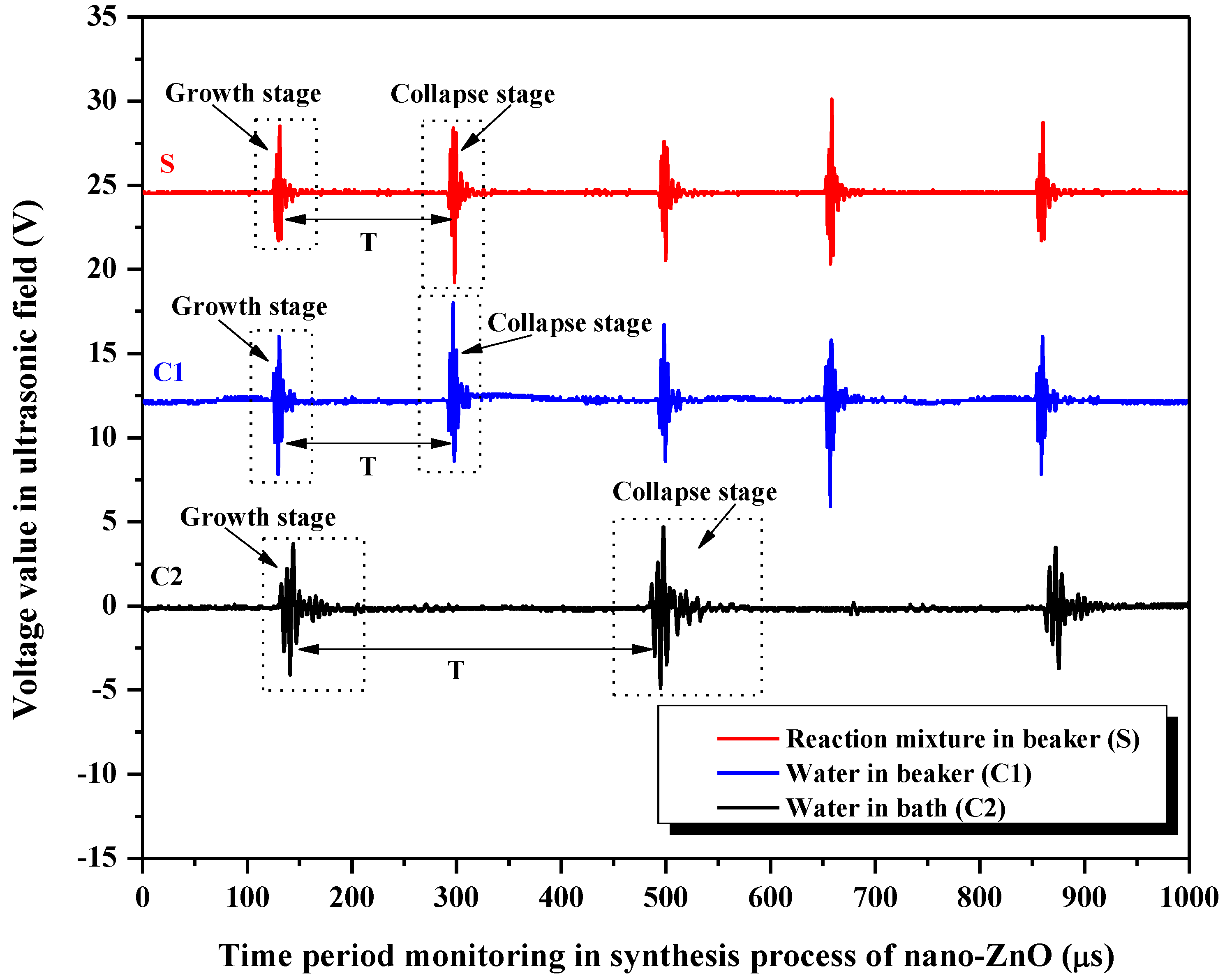
3.4. Analysis of Ultrasonic Fields during Synthesis Process of Nano-ZnO
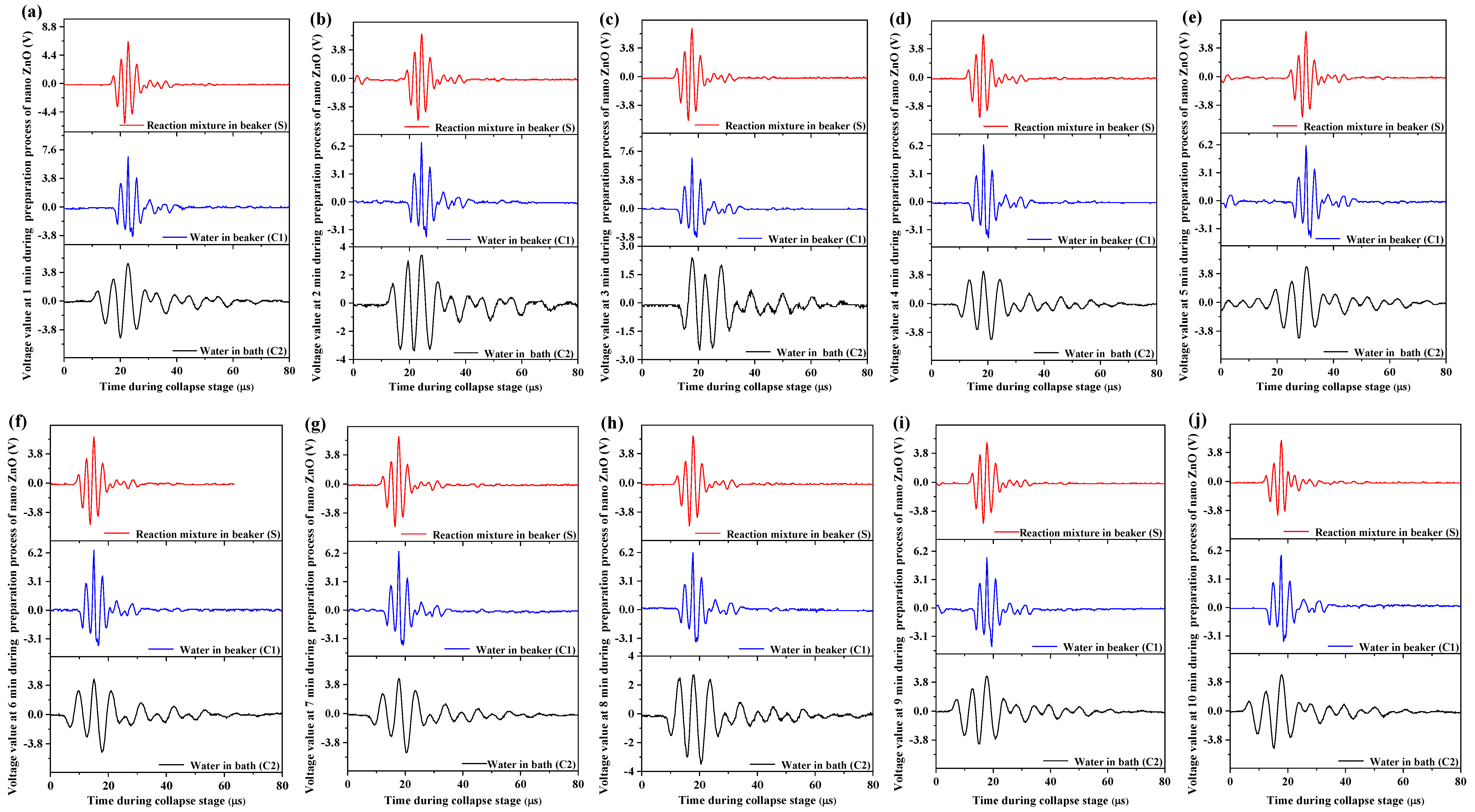
3.4.1. Monitoring and Analysis of Voltage Waveforms in Different Ultrasonic Fields
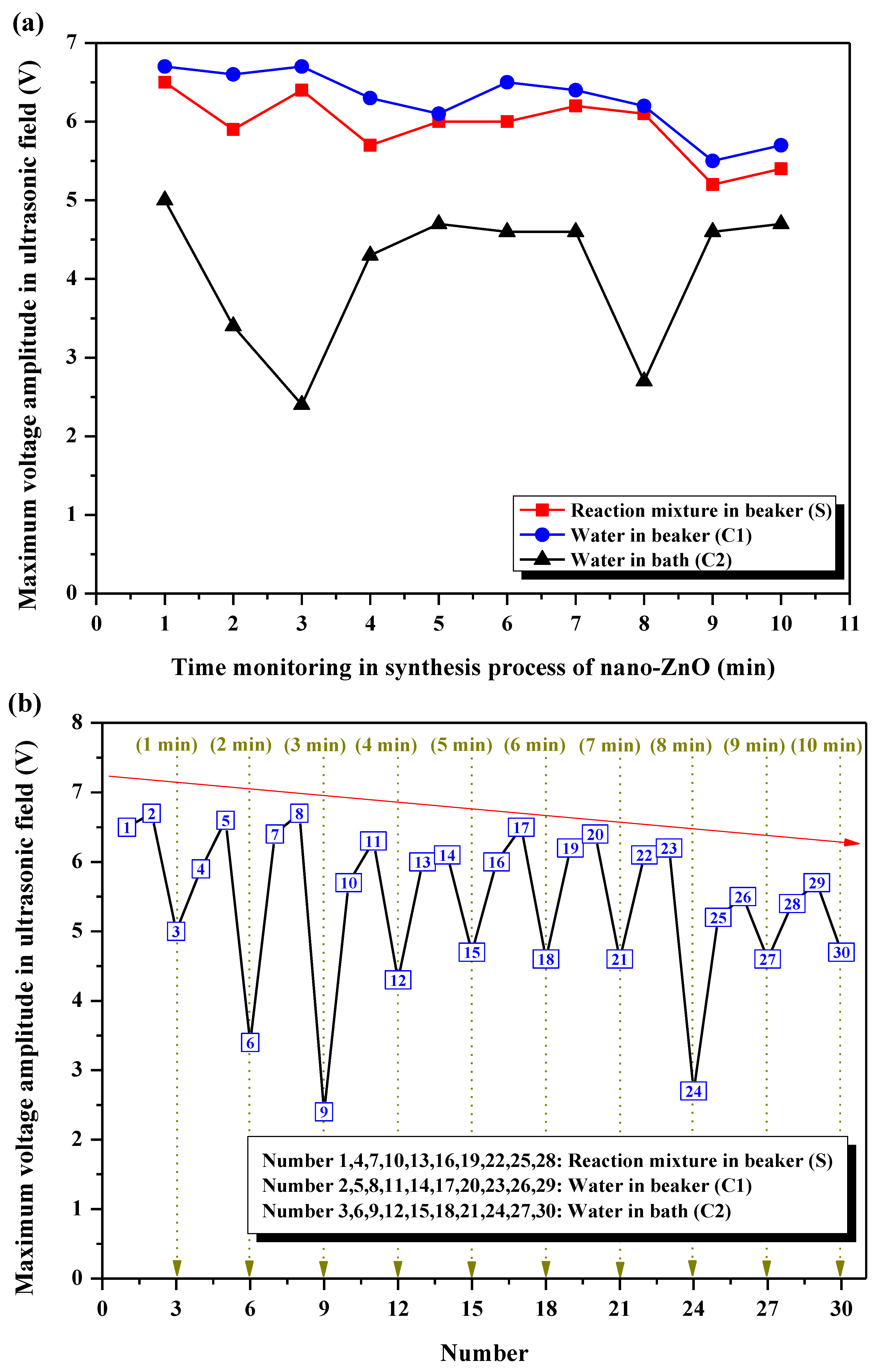
3.4.2. Analysis of the Maximum Voltage Amplitude
3.4.3. Analysis of Space Peak Temporal Peak Acoustic Intensity and Space Peak Temporal Average Acoustic Intensity
4. Conclusions
- (1)
- The AFM analysis indicated that nano-ZnO synthesized by the ultrasonic method was characterized by a smaller structure and surface roughness, and better dispersity, compared with that by the mechanical stirring method. According to the XRD analysis, the nucleation and crystallization process was controlling effectively in the ultrasonic field, resulting in nano-ZnO with a better crystal structure. FTIR spectral analysis proved the occurrence of some differences in the ZnO nano-structure by ultrasonic and mechanical stirring methods.
- (2)
- Under the ultrasonic field, the reaction mixture during the synthesis of nano-ZnO S and the control group C1 showed shorter life cycles of cavitation bubble oscillation compared with C2. The cycle of collapse stage in S and C1 was almost the same, but it was longer in C2. The maximum voltage amplitudes in C1 were generally higher than that in S, and were the lowest in C2. A general trend in the maximum voltage amplitudes in S, C1, and C2 was to decrease as the action time prolonged. ISPTP in S was lower than that in C1, and ISPTP in C2 was the lowest. ISPTA in C1 was stronger than that in S due to differences in the properties between ILs and water. ISPTA in C2 was the strongest.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ngom, B.D.; Mpahane, T.; Manikandan, E.; Maaza, M. ZnO nano-discs by lyophilization process: Size effects on their intrinsic luminescence. J. Alloys Compd. 2016, 656, 758–763. [Google Scholar] [CrossRef]
- Hilal, M.; Han, J.I. Bi-functional carbon doped and decorated ZnO nanorods for enhanced pH monitoring of dairy milk and adsorption of hazardous dyes. J. Ind. Eng. Chem. 2022, 110, 520–528. [Google Scholar] [CrossRef]
- Bigdeli, F.; Ghasempour, H.; Tehrani, A.A.; Morsali, A.; Hosseini-Monfared, H. Ultrasound-assisted synthesis of nano-structured Zinc (II)-based metal-organic frameworks as precursors for the synthesis of ZnO nano-structures. Ultrason. Sonochem. 2017, 37, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Kumar, G.; Akhtar, M.S.; Umar, A. Sonophotocatalytic degradation of methyl orange using ZnO nano-aggregates. J. Alloys Compd. 2015, 629, 167–172. [Google Scholar] [CrossRef]
- V, L.P.; Vijayaraghavan, R. Chemical manipulation of oxygen vacancy and antibacterial activity in ZnO. Mater. Sci. Eng. C 2017, 77, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- He, X.P.; Yang, P.F.; Kuang, H.J.; Xu, H.Y. Application and security of nano zinc oxide in food field. J. Food Saf. Qual. 2016, 7, 4525–4530. [Google Scholar] [CrossRef]
- Wang, C.S.; Zhang, J.; Chen, J.H.; Shi, J.W.; Zhao, Y.T.; He, M.; Ding, L. Bio-polyols based waterborne polyurethane coatings reinforced with chitosan-modified ZnO nanoparticles. Int. J. Biol. Macromol. 2022, 208, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.W.; Xue, X.Y.; Xu, X.F.; Zhou, H.; Li, W.J.; Yang, G.L.; Xie, P.J. Detoxified and antimicrobial-enhanced olive mill wastewater phenols capping ZnO nanoparticles incorporated with carboxymethyl cellulose for fresh strawberry preservation. Postharvest Biol. Technol. 2022, 188, 111891. [Google Scholar] [CrossRef]
- Phothisarattana, D.; Wongphan, P.; Promhuad, K.; Promsorn, J.; Harnkarnsujarit, N. Blown film extrusion of PBAT/TPS/ZnO nanocomposites for shelf-life extension of meat packaging. Colloids Surf. B 2022, 214, 112472. [Google Scholar] [CrossRef]
- Emamifar, A.; Kadivar, M.; Shahedi, M.; Soleimanian-Zad, S. Evaluation of nanocomposite packaging containing Ag and ZnO on shelf life of fresh orange juice. Innov. Food Sci. Emerg. Technol. 2010, 11, 742–748. [Google Scholar] [CrossRef]
- Raghupathi, K.R.; Koodali, R.T.; Manna, A.C. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2021, 27, 4020–4028. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Gao, W.; Wang, B.; Kang, X.M.; Liu, P.F.; Cui, B.; EI-Aty, A.M.A. Preparation and evaluation of starch-based extrusion-blown nanocomposite films incorporated with nano-ZnO and nano-SiO2. Int. J. Biol. Macromol. 2021, 183, 1371–1378. [Google Scholar] [CrossRef]
- Shahmohammadi, J.F.; Almasi, H. Morphological, physical, antimicrobial and release properties of ZnO nanoparticles-loaded bacterial cellulose films. Carbohyd. Polym. 2016, 149, 8–19. [Google Scholar] [CrossRef]
- Zamiri, R.; Singh, B.; Bdikin, I.; Rebelo, A.; Belsley, M.S.; Ferreira, J.M.F. Influence of Mg doping on dielectric and optical properties of ZnO nano-plates prepared by wet chemical method. Solid State Commun. 2014, 195, 74–79. [Google Scholar] [CrossRef]
- Vafaee, M.; Ghamsari, M.S. Preparation and characterization of ZnO nanoparticles by a novel sol-gel route. Mater. Lett. 2007, 61, 3265–3268. [Google Scholar] [CrossRef]
- Wang, Y.G.; Lau, S.P.; Zhang, X.H.; Lee, H.W.; Yu, S.F.; Tay, B.K.; Hng, H.H. Evolution of visible luminescence in ZnO by thermal oxidation of zinc films. Chem. Phys. Lett. 2003, 375, 113–118. [Google Scholar] [CrossRef]
- Kang, S.H.; Zhang, J.; Guo, X.B.; Lei, Y.D.; Yang, M. Effects of ultrasonic treatment on the structure, functional properties of chickpea protein isolate and its digestibility in vitro. Foods 2022, 11, 880. [Google Scholar] [CrossRef]
- Salarbashi, D.; Mortazavi, S.A.; Noghabi, M.S.; Bazzaz, B.S.F.; Sedaghat, N.; Ramezani, M.; Shahabi-Ghahfarrokhi, I. Development of new active packaging film made from a soluble soybean polysaccharide incorporating ZnO nanoparticles. Carbohyd. Polym. 2016, 140, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Ameer, K.; Ameer, S.; Kim, Y.M.; Nadeem, M.; Park, M.K.; Murtaza, M.A.; Khan, M.A.; Nasir, M.A.; Mueen-ud-Din, G.; Mahmood, S.; et al. A hybrid RSM-ANN-GA approach on optimization of ultrasound-assisted extraction conditions for bioactive component-rich Stevia rebaudiana (Bertoni) leaves extract. Foods 2022, 11, 883. [Google Scholar] [CrossRef]
- Daryabor, M.; Ahmadi, A.; Zilouei, H. Solvent extraction of cadmium and zinc from sulphate solutions: Comparison of mechanical agitation and ultrasonic irradiation. Ultrason. Sonochem. 2017, 34, 931–937. [Google Scholar] [CrossRef]
- Baidukova, O.; Skorb, E.V. Ultrasound-assisted synthesis of magnesium hydroxide nanoparticles from magnesium. Ultrason. Sonochem. 2016, 31, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Panahi-Kalamuei, M.; Mousavi-Kamazani, M.; Salavati-Niasari, M.; Hosseinpour-Mashkani, S.M. A simple sonochemical approach for synthesis of selenium nanostructures and investigation of its light harvesting application. Ultrason. Sonochem. 2015, 23, 246–256. [Google Scholar] [CrossRef] [PubMed]
- He, Z.Q.; Alexandridis, P. Ionic liquid and nanoparticle hybrid systems: Emerging applications. Adv. Colloid Interface Sci. 2017, 244, 54–70. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.M.; Zhou, F.; Sun, Y.B.; Liu, W.M. Ultrasound-assisted synthesis of dentritic ZnO nanostructure in ionic liquid. Mater. Lett. 2007, 61, 1789–1792. [Google Scholar] [CrossRef]
- Goharshadi, E.K.; Ding, Y.L.; Jorabchi, M.N.; Nancarrow, P. Ultrasound-assisted green synthesis of nanocrystalline ZnO in the ionic liquid [hmim][NTf2]. Ultrason. Sonochem. 2009, 16, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Alammar, T.; Mudring, A.V. Facile ultrasound-assisted synthesis of ZnO nanorods in an ionic liquid. Mater. Lett. 2009, 63, 732–735. [Google Scholar] [CrossRef]
- Sabbaghan, M.; Beheshtian, J.; Mirsaeidi, S.A.M. Preparation of uniform 2D ZnO nanostructures by the ionic liquid-assisted sonochemical method and their optical properties. Ceram. Int. 2014, 40, 7769–7774. [Google Scholar] [CrossRef]
- Hosni, M.; Farhat, S.; Schoenstein, F.; Karmous, F.; Jouini, N.; Viana, B.; Mgaidi, A. Ultrasound assisted synthesis of nanocrystalline zinc oxide: Experiments and modelling. J. Alloys Compd. 2014, 615, S472–S475. [Google Scholar] [CrossRef]
- Chen, C.Y.; Li, Q.; Nie, M.; Lin, H.; Li, Y.; Wu, H.J.; Wang, Y.Y. An efficient room-temperature route to uniform ZnO nanorods with an ionic liquid. Mater. Res. Bull. 2011, 46, 888–893. [Google Scholar] [CrossRef]
- Merouani, S.; Hamdaoui, O.; Rezgui, Y.; Guemini, M. Effects of ultrasound frequency and acoustic amplitude on the size of sonochemically active bubbles—Theoretical study. Ultrason. Sonochem. 2013, 20, 815–819. [Google Scholar] [CrossRef]
- Duan, B.R.; Shao, X.F.; Han, Y.; Li, Y.; Zhao, Y.J. Mechanism and application of ultrasound-enhanced bacteriostasis. J. Clean. Prod. 2021, 290, 125750. [Google Scholar] [CrossRef]
- Bui, T.T.; Shin, M.K.; Jee, S.Y.; Long, D.X.; Hong, J.; Kim, M.G. Ferroelectric PVDF nanofiber membrane for high-efficiency PM0.3 air filtration with low air flow resistance. Colloids Surf. A 2022, 640, 128418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, X.; Hu, Y.; Fakayode, O.A.; Ma, H.L.; Zhou, C.S.; Hu, Z.Y.; Xia, A.M.; Li, Q. Dual-frequency multi-angle ultrasonic processing technology and its real-time monitoring on physicochemical properties of raw soymilk and soybean protein. Ultrason. Sonochem. 2021, 80, 105803. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Ma, H.L.; Wang, B.; Yagoub, A.E.G.A.; Wang, K.; He, R.H.; Zhou, C.S. Effects and mechanism of dual-frequency power ultrasound on the molecular weight distribution of corn gluten meal hydrolysates. Ultrason. Sonochem. 2016, 30, 44–51. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Wang, B.; Zhou, C.S.; Atungulu, G.G.; Xu, K.K.; Ma, H.L.; Ye, X.F.; Abdualrahman, M.A.Y. Surface topography, nano-mechanics and secondary structure of wheat gluten pretreated by alternate dual-frequency ultrasound and the correlation to enzymolysis. Ultrason. Sonochem. 2016, 31, 267–275. [Google Scholar] [CrossRef]
- Shou, W.D.; Qian, D.C. Measurement of pulsed sound intensity in ultrasonic diagnosis and its significance. Chin. J. Biomed. Eng. 1987, 6, 125–131. [Google Scholar]
- Lin, M.H.; Wang, Y.H.; Kuo, C.H.; Ou, S.F.; Huang, P.Z.; Song, T.Y.; Chen, Y.C.; Chen, S.T.; Wu, C.H.; Hsueh, Y.H.; et al. Hybrid ZnO/chitosan antimicrobial coatings with enhanced mechanical and bioactive properties for titanium implants. Carbohyd. Polym. 2021, 257, 117639. [Google Scholar] [CrossRef]
- Król, A.; Pomastowski, P.; Rafińska, K.; Railean-Plugaru, V.; Buszewski, B. Zinc Oxide nanoparticles: Synthesis, antiseptic activity and toxicity mechanism. Adv. Colloid Interface Sci. 2017, 245, 100. [Google Scholar] [CrossRef] [PubMed]
- Nii, S.; Takayanagi, S. Growth and size control in anti-solvent crystallization of glycine with high frequency ultrasound. Ultrason. Sonochem. 2014, 21, 1182–1186. [Google Scholar] [CrossRef]
- Horn, E.J.; Rosen, B.R.; Chen, Y.; Tang, J.Z.; Chen, K.; Eastgate, M.D.; Baran, P.S. Scalable and sustainable electrochemical allylic C-H oxidation. Nature 2016, 533, 77–81. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.Y.; Zhang, M.; Yang, C.H. ZnO nanoparticles combined low frequency microwave on the quality and shelf life of Hongsu chicken dishes. J. Food Sci. Biotechnol. 2020, 39, 76–83. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, Y.; Wang, X.; Fakayode, O.A.; Ma, H.L.; Zhou, C.S.; Xia, A.M.; Li, Q. Improving soaking efficiency of soybeans through sweeping frequency ultrasound assisted by parameters optimization. Ultrason. Sonochem. 2021, 79, 105794. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, C.S.; Wang, B.; Yagoub, A.E.G.A.; Ma, H.L.; Zhang, X.; Wu, M. Study of ultrasonic cavitation during extraction of the peanut oil at varying frequencies. Ultrason. Sonochem. 2017, 37, 106–113. [Google Scholar] [CrossRef]
- Wijngaarden, L.V. Mechanics of collapsing cavitation bubbles. Ultrason. Sonochem. 2016, 29, 524–527. [Google Scholar] [CrossRef] [Green Version]
- Fan, D.M.; Huang, L.L.; Li, B.; Huang, J.L.; Zhao, J.X.; Yan, B.W.; Zhou, W.G.; Zhang, W.H.; Zhang, H. Acoustic intensity in ultrasound field and ultrasound-assisted gelling of surimi. LWT-Food Sci. Technol. 2017, 75, 497–504. [Google Scholar] [CrossRef]



| Surface Roughness (nm) | ||
|---|---|---|
| Ra | Rq | |
| Mechanical stirring method | 25.6 | 43.1 |
| Ultrasonic method | 4.2 | 10.5 |
| Synthesis Time | Space Peak Temporal Peak Acoustic Intensity ISPTP (W/m2) × 1010 | ||
|---|---|---|---|
| Reaction Mixture (S) | Water in Beaker (C1) | Water in Bath (C2) | |
| 1 min | 4.6 | 7.5 | 4.2 |
| 2 min | 3.8 | 7.3 | 1.9 |
| 3 min | 4.4 | 7.5 | 1.0 |
| 4 min | 3.5 | 6.6 | 3.1 |
| 5 min | 3.9 | 6.2 | 3.7 |
| 6 min | 3.9 | 7.0 | 3.5 |
| 7 min | 4.2 | 6.8 | 3.5 |
| 8 min | 4.0 | 6.4 | 1.2 |
| 9 min | 2.9 | 5.0 | 3.5 |
| 10 min | 3.2 | 5.4 | 3.7 |
| Liquid | Space Peak Temporal Average Acoustic Intensity ISPTA (W/m2) × 107 |
|---|---|
| Reaction mixture (S) | 6.4 |
| Water in beaker (C1) | 9.9 |
| Water in bath (C2) | 33.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, L.; Hu, Y.; Wang, X.; Zhang, A.; Gao, X.; Yagoub, A.E.-G.A.; Ma, H.; Zhou, C. Ultrasound-Assisted Synthesis of Potentially Food-Grade Nano-Zinc Oxide in Ionic Liquids: A Safe, Green, Efficient Approach and Its Acoustics Mechanism. Foods 2022, 11, 1656. https://doi.org/10.3390/foods11111656
Zhang L, Hu Y, Wang X, Zhang A, Gao X, Yagoub AE-GA, Ma H, Zhou C. Ultrasound-Assisted Synthesis of Potentially Food-Grade Nano-Zinc Oxide in Ionic Liquids: A Safe, Green, Efficient Approach and Its Acoustics Mechanism. Foods. 2022; 11(11):1656. https://doi.org/10.3390/foods11111656
Chicago/Turabian StyleZhang, Lei, Yang Hu, Xue Wang, Ao Zhang, Xianli Gao, Abu El-Gasim A. Yagoub, Haile Ma, and Cunshan Zhou. 2022. "Ultrasound-Assisted Synthesis of Potentially Food-Grade Nano-Zinc Oxide in Ionic Liquids: A Safe, Green, Efficient Approach and Its Acoustics Mechanism" Foods 11, no. 11: 1656. https://doi.org/10.3390/foods11111656
APA StyleZhang, L., Hu, Y., Wang, X., Zhang, A., Gao, X., Yagoub, A. E.-G. A., Ma, H., & Zhou, C. (2022). Ultrasound-Assisted Synthesis of Potentially Food-Grade Nano-Zinc Oxide in Ionic Liquids: A Safe, Green, Efficient Approach and Its Acoustics Mechanism. Foods, 11(11), 1656. https://doi.org/10.3390/foods11111656