Multiple Breeds and Countries’ Predictions of Mineral Contents in Milk from Milk Mid-Infrared Spectrometry
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data
2.2. Reference Analysis
2.3. Modelling
2.4. Relationships with Traits Related to Animal Health and Milk Technological Properties
2.5. Large-Scale Phenotyping
3. Results and Discussion
3.1. Reference Dataset
3.2. Creation of Models Predicting Mineral Content in Milk
3.3. Correlation between Mineral Traits and Traits Related to Animal Health and Milk Technological Properties
3.4. Descriptive Statistics Applied on the Walloon Database
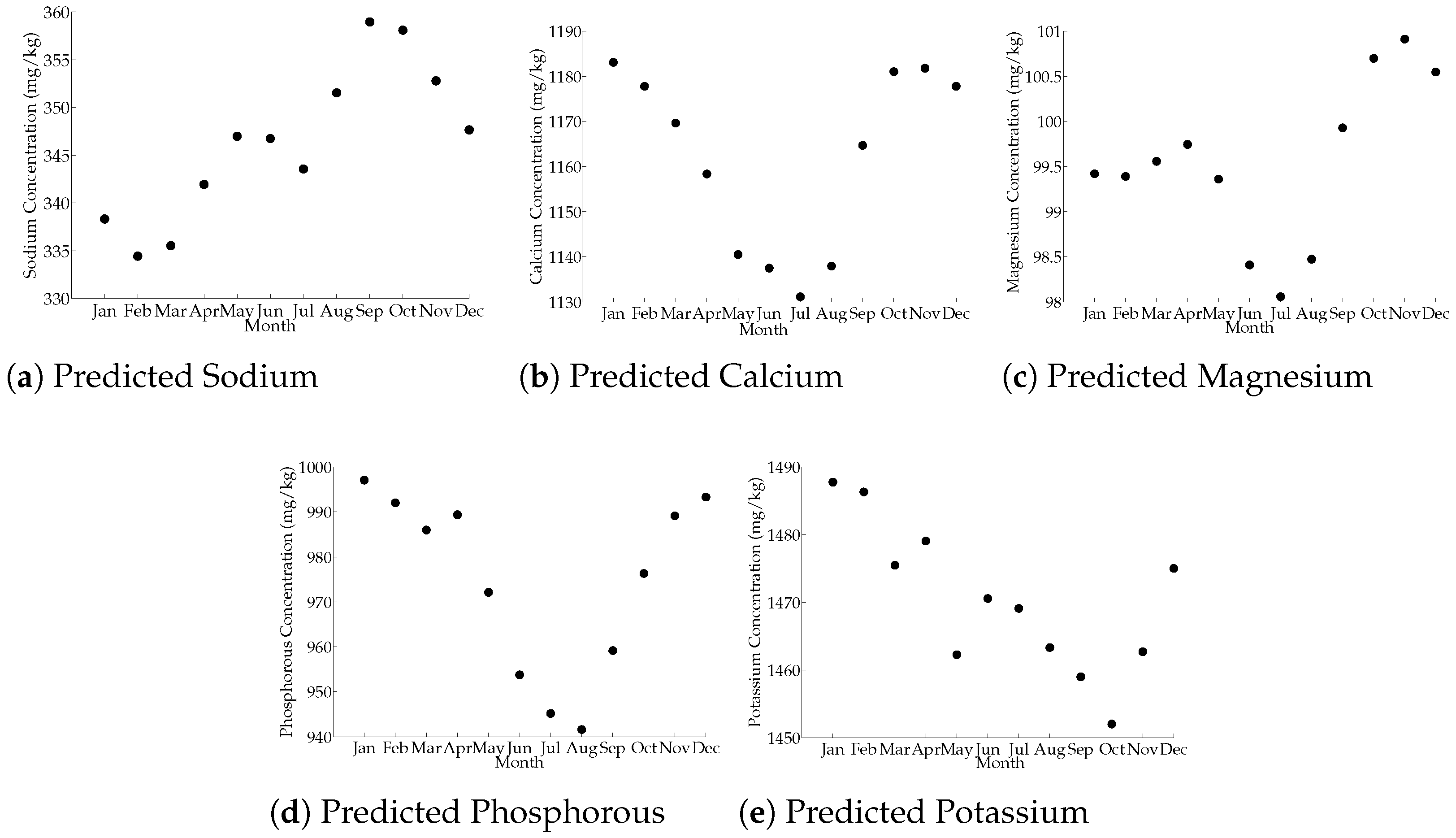
3.5. Month Influence on Milk-Predicted Mineral Composition
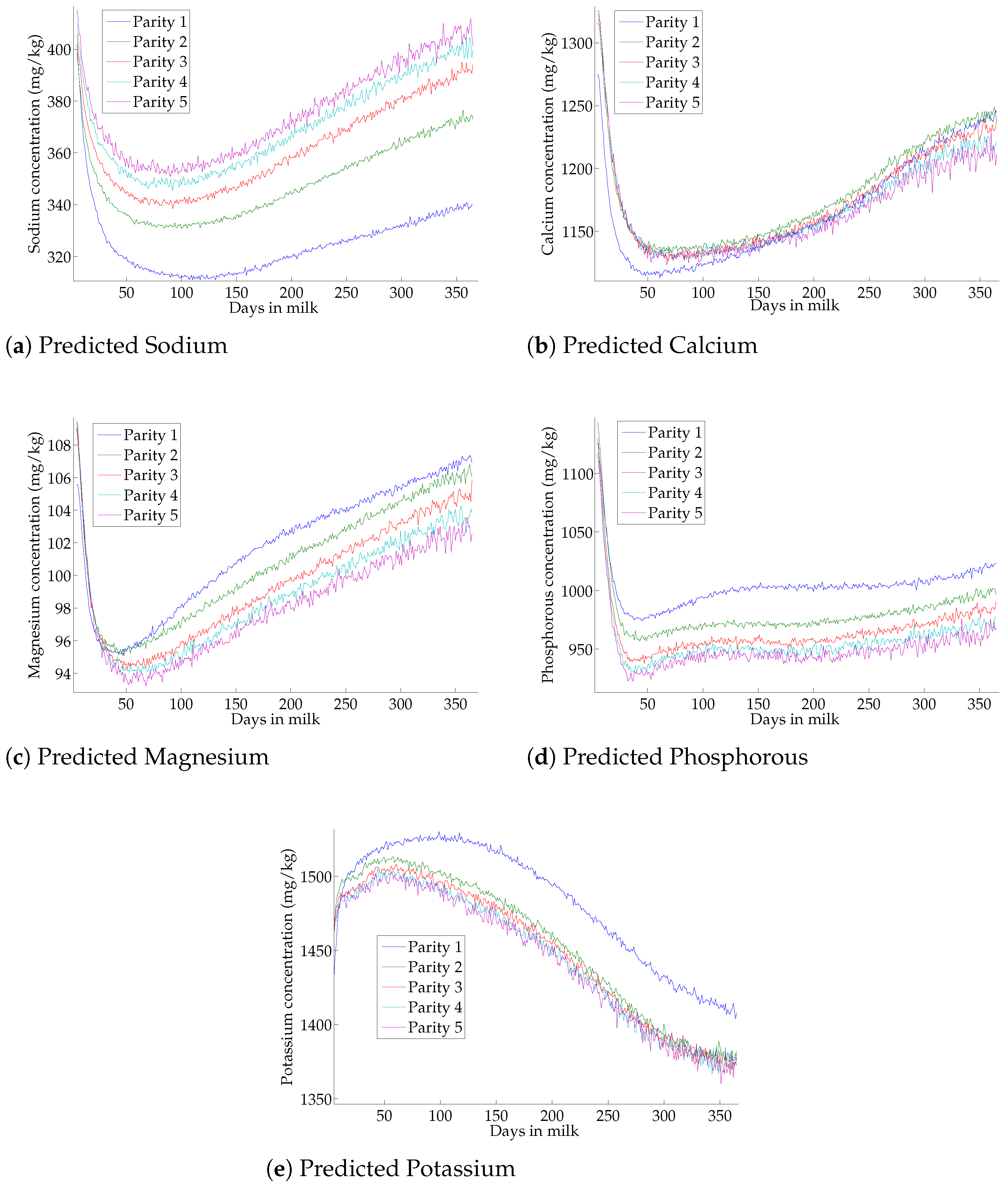
3.6. Effect of Lactation on Milk-Predicted Minerals
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DHI | Dairy Herd Improvement |
| ICP-AES | Inductively Coupled Plasma—Atomic Emission Spectroscopy |
| GH | Malabonis distance |
| NH | Neighboring H distance |
| RMSE | Root Mean Square Error |
| MIR | Mid-infrared |
| PDS | Piece-wise standardized method |
| PLS | Partial least square regression |
| RMSEcv | Root Mean Square Error of cross-validation |
| RMSEP | Root Mean Square Error of prediction |
| RPD | Ratio of prediction to deviation |
| DIM | Days in milk |
| pX | Predicted traits (X = Na, Ca, Mg, P, K, etc.) |
| NAGase | N-acetyl--D-glucosaminidase |
| LDH | Lactase Lactate dehydrogenase |
| RCT | Rennet coagulation time |
| k20 | Curd-firming time |
| a30 | Curd firmness 30 min after rennet addition |
| ILCY | Individual laboratory cheese yield |
References
- Zamberlin, Š.; Antunac, N.; Havranek, J.; Samaržija, D. Mineral elements in milk and dairy products. Mljekarstvo 2012, 62, 111–125. [Google Scholar]
- FAO. The State of Food and Agriculture 2001; Number 33; FAO: Rome, Italy, 2001. [Google Scholar]
- Guéguen, L.; Pointillart, A. The Bioavailability of Dietary Calcium. J. Am. Coll. Nutr. 2000, 19, 119S–136S. [Google Scholar] [CrossRef] [PubMed]
- Huth, P.J.; DiRienzo, D.B.; Miller, G.D. Major Scientific Advances with Dairy Foods in Nutrition and Health. J. Dairy Sci. 2006, 89, 1207–1221. [Google Scholar] [CrossRef]
- Lanou, A.J.; Berkow, S.E.; Barnard, N.D. Calcium, Dairy Products, and Bone Health in Children and Young Adults: A Reevaluation of the Evidence. Pediatrics 2005, 115, 736–743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, F.J.; MacGregor, G.A. Beneficial effects of potassium on human health. Physiol. Plant. 2008, 133, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; He, J. Health effects of sodium and potassium in humans. Curr. Opin. Lipidol. 2014, 25, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Uribarri, J.; Calvo, M.S. Dietary phosphorus intake and health. Am. J. Clin. Nutr. 2014, 99, 247–248. [Google Scholar] [CrossRef] [Green Version]
- Thachepan, S.; Li, M.; Mann, S. Mesoscale crystallization of calcium phosphate nanostructures in protein (casein) micelles. Nanoscale 2010, 2, 2400–2405. [Google Scholar] [CrossRef]
- Zhang, Z.P.; Aoki, T. Behaviour of calcium and phosphate in bovine casein micelles. Int. Dairy J. 1996, 6, 769–780. [Google Scholar] [CrossRef]
- Fossa, E.; Pecorari, M.; Sandri, S.; Tosi, F.C.L.C.; Mariani, P.P.U.I.I.d.Z. The Role of Milk Casein Content in the Parmigiano-Reggiano Cheese Production: Chemical Composition—Rennet Coagulation Properties and Dairy-Technological Behaviour of Milk; Scienza e Tecnica Lattiero-Caseari: Parma, Italy, 1994; ISSN 0390-6361. [Google Scholar]
- Auldist, M.; Hubble, I. Effects of mastitis on raw milk and dairy products. Aust. J. Dairy Technol. 1998, 53, 28. [Google Scholar]
- Hamann, J.; Krömker, V. Potential of specific milk composition variables for cow health management. Livest. Prod. Sci. 1997, 48, 201–208. [Google Scholar] [CrossRef]
- Qayyum, A.; Khan, J.A.; Hussain, R.; Avais, M.; Ahmad, N.; Khan, M.S. Investigation of Milk and Blood Serum Biochemical Profile as an Indicator of Sub-Clinical Mastitis in Cholistani Cattle. Pak. Vet. J. 2016, 36, 275–279. [Google Scholar]
- Bastin, C.; Théron, L.; Lainé, A.; Gengler, N. On the role of mid-infrared predicted phenotypes in fertility and health dairy breeding programs. J. Dairy Sci. 2016, 99, 4080–4094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarkhot, D.V.; Ghezzehei, T.A.; Berhe, A.A. Effectiveness of Biochar for Sorption of Ammonium and Phosphate from Dairy Effluent. J. Environ. Qual. 2013, 42, 1545–1554. [Google Scholar] [CrossRef]
- Kurzbaum, E.; Bar Shalom, O. The potential of phosphate removal from dairy wastewater and municipal wastewater effluents using a lanthanum-modified bentonite. Appl. Clay Sci. 2016, 123, 182–186. [Google Scholar] [CrossRef]
- CBS. Phosphate Output from Livestock Manure Down Again. 2019. Available online: https://www.cbs.nl/en-gb/news/2019/07/phosphate-output-from-livestock-manure-down-again (accessed on 2 June 2021).
- Soyeurt, H.; Bruwier, D.; Romnee, J.M.; Gengler, N.; Bertozzi, C.; Veselko, D.; Dardenne, P. Potential estimation of major mineral contents in cow milk using mid-infrared spectrometry. J. Dairy Sci. 2009, 92, 2444–2454. [Google Scholar] [CrossRef] [Green Version]
- Toffanin, V.; De Marchi, M.; Lopez-Villalobos, N.; Cassandro, M. Effectiveness of mid-infrared spectroscopy for prediction of the contents of calcium and phosphorus, and titratable acidity of milk and their relationship with milk quality and coagulation properties. Int. Dairy J. 2015, 41, 68–73. [Google Scholar] [CrossRef]
- Gottardo, P.; De Marchi, M.; Cassandro, M.; Penasa, M. Technical note: Improving the accuracy of mid-infrared prediction models by selecting the most informative wavelengths. J. Dairy Sci. 2015, 98, 4168–4173. [Google Scholar] [CrossRef] [Green Version]
- Bonfatti, V.; Degano, L.; Menegoz, A.; Carnier, P. Short communication: Mid-infrared spectroscopy prediction of fine milk composition and technological properties in Italian Simmental. J. Dairy Sci. 2016, 99, 8216–8221. [Google Scholar] [CrossRef] [Green Version]
- Visentin, G.; Penasa, M.; Gottardo, P.; Cassandro, M.; De Marchi, M. Predictive ability of mid-infrared spectroscopy for major mineral composition and coagulation traits of bovine milk by using the uninformative variable selection algorithm. J. Dairy Sci. 2016, 99, 8137–8145. [Google Scholar] [CrossRef]
- Franzoi, M.; Niero, G.; Penasa, M.; De Marchi, M. Development of Infrared Prediction Models for Diffusible and Micellar Minerals in Bovine Milk. Animals 2019, 9, 430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fleming, A.; Schenkel, F.S.; Ali, R.A.; Corredig, M.; Carta, S.; Gregu, C.M.; Malchiodi, F.; Macciotta, N.P.P.; Miglior, F. Phenotypic investigation of fine milk components in bovine milk and their prediction using mid-infrared spectroscopy. Can. J. Anim. Sci. 2018, 99, 218–227. [Google Scholar] [CrossRef]
- Zaalberg, R.; Poulsen, N.; Bovenhuis, H.; Sehested, J.; Larsen, L.; Buitenhuis, A. Genetic analysis on infrared-predicted milk minerals for Danish dairy cattle. J. Dairy Sci. 2021, 104, 8947–8958. [Google Scholar] [CrossRef]
- Grelet, C.; Dardenne, P.; Soyeurt, H.; Fernandez, J.A.; Vanlierde, A.; Stevens, F.; Gengler, N.; Dehareng, F. Large-scale phenotyping in dairy sector using milk MIR spectra: Key factors affecting the quality of predictions. Methods 2020, 186, 97–111. [Google Scholar] [CrossRef]
- Zhang, L.; Li, C.; Dehareng, F.; Grelet, C.; Colinet, F.; Gengler, N.; Brostaux, Y.; Soyeurt, H. Appropriate Data Quality Checks Improve the Reliability of Values Predicted from Milk Mid-Infrared Spectra. Animals 2021, 11, 533. [Google Scholar] [CrossRef] [PubMed]
- Grelet, C.; Fernández Pierna, J.A.; Dardenne, P.; Baeten, V.; Dehareng, F. Standardization of milk mid-infrared spectra from a European dairy network. J. Dairy Sci. 2015, 98, 2150–2160. [Google Scholar] [CrossRef]
- Murcia, M.A.; Vera, A.; Martinez-Tome, M.; Munoz, A.; Hernandez-Cordoba, M.; Ortiz-Gonzalez, R. Fast Determination of the Ca, Mg, K, Na and Zn Contents in Milk and Nondairy Imitation Milk Using ICP-AES without Mineralization Stage. Food Sci. Technol. 1999, 32, 175–179. [Google Scholar] [CrossRef]
- Rousseeuw, P.J.; Debruyne, M.; Engelen, S.; Hubert, M. Robustness and outlier detection in chemometrics. Crit. Rev. Anal. Chem. 2006, 36, 221–242. [Google Scholar] [CrossRef]
- Grelet, C.; Bastin, C.; Gelé, M.; Davière, J.B.; Johan, M.; Werner, A.; Reding, R.; Pierna, J.F.; Colinet, F.; Dardenne, P.; et al. Development of Fourier transform mid-infrared calibrations to predict acetone, β-hydroxybutyrate, and citrate contents in bovine milk through a European dairy network. J. Dairy Sci. 2016, 99, 4816–4825. [Google Scholar] [CrossRef] [Green Version]
- van der Voet, H. Comparing the predictive accuracy of models using a simple randomization test. Chemom. Intell. Lab. Syst. 1994, 25, 313–323. [Google Scholar] [CrossRef]
- Soyeurt, H.; Grelet, C.; McParland, S.; Calmels, M.; Coffey, M.; Tedde, A.; Delhez, P.; Dehareng, F.; Gengler, N. A comparison of 4 different machine learning algorithms to predict lactoferrin content in bovine milk from mid-infrared spectra. J. Dairy Sci. 2020, 103, 11585–11596. [Google Scholar] [CrossRef]
- Colinet, F.; Troch, T.; Baeten, V.; Dehareng, F.; Dardenne, P.; Sindic, M.; Gengler, N. Genetic variability of MIR predicted milk technological properties in Walloon dairy cattle. In Book of Abstracts of the 66th Annual Meeting of the European Federation of Animal Science; Wageningen Academic Publishers: Wageningen, The Netherlands, 2015; p. 400. [Google Scholar]
- Saeys, W.; Mouazen, A.; Ramon, H. Potential for Onsite and Online Analysis of Pig Manure using Visible and Near Infrared Reflectance Spectroscopy. Biosyst. Eng. 2005, 91, 393–402. [Google Scholar] [CrossRef]
- Fox, P.; Uniacke-Lowe, T.; McSweeney, P.; O’Mahony, J. Milk proteins. In Dairy Chemistry and Biochemistry; Springer: Cham, Switzerland, 2015; pp. 145–239. [Google Scholar]
- Oh, H.E.; Deeth, H.C. Magnesium in milk. Int. Dairy J. 2017, 71, 89–97. [Google Scholar] [CrossRef] [Green Version]
- Fox, P.; Uniacke-Lowe, T.; McSweeney, P.; O’Mahony, J. Salts of milk. In Dairy Chemistry and Biochemistry; Springer: Cham, Switzerland, 2015; pp. 241–270. [Google Scholar]
- Franzoi, M.; Niero, G.; Penasa, M.; Cassandro, M.; De Marchi, M. Technical note: Development and validation of a new method for the quantification of soluble and micellar calcium, magnesium, and potassium in milk. J. Dairy Sci. 2018, 101, 1883–1888. [Google Scholar] [CrossRef] [PubMed]
- Batavani, R.; Asri, S.; Naebzadeh, H. The effect of subclinical mastitis on milk composition in dairy cows. Iran. J. Vet. Res. 2007, 8, 205–211. [Google Scholar]
- Pyörälä, S. Indicators of inflammation in the diagnosis of mastitis. J. Vet. Res. 2003, 34, 565–578. [Google Scholar] [CrossRef] [Green Version]
- Kitchen, B.J.; Kwee, W.S.; Middleton, G.; Andrews, R.J. Relationship between the level of N-acetyl-β-D-glucosaminidase (NAGase) in bovine milk and the presence of mastitis pathogens. J. Dairy Res. 1984, 51, 11–16. [Google Scholar] [CrossRef]
- Hovinen, M.; Simojoki, H.; Pösö, R.; Suolaniemi, J.; Kalmus, P.; Suojala, L.; Pyörälä, S. N-acetyl-[...]-D-glucosaminidase activity in cow milk as an indicator of mastitis. J. Dairy Res. 2016, 83, 219. [Google Scholar] [CrossRef] [PubMed]
- Soyeurt, H.; Bastin, C.; Colinet, F.; Arnould, V.R.; Berry, D.P.; Wall, E.; Dehareng, F.; Nguyen, H.; Dardenne, P.; Schefers, J.; et al. Mid-infrared prediction of lactoferrin content in bovine milk: Potential indicator of mastitis. Animal 2012, 6, 1830–1838. [Google Scholar] [CrossRef] [Green Version]
- Norberg, E. Electrical conductivity of milk as a phenotypic and genetic indicator of bovine mastitis: A review. Livest. Prod. Sci. 2005, 96, 129–139. [Google Scholar] [CrossRef]
- Summer, A.; Franceschi, P.; Malacarne, M.; Formaggioni, P.; Tosi, F.; Tedeschi, G.; Mariani, P. Influence of somatic cell count on mineral content and salt equilibria of milk. Ital. J. Anim. Sci. 2010, 8, 435–437. [Google Scholar] [CrossRef]
- Lucey, J.; Fox, P. Importance of Calcium and Phosphate in Cheese Manufacture: A Review. J. Dairy Sci. 1993, 76, 1714–1724. [Google Scholar] [CrossRef]
- Sanchez, M.; El Jabri, M.; Minéry, S.; Wolf, V.; Beuvier, E.; Laithier, C.; Delacroix-Buchet, A.; Brochard, M.; Boichard, D. Genetic parameters for cheese-making properties and milk composition predicted from mid-infrared spectra in a large data set of Montbéliarde cows. J. Dairy Sci. 2018, 101, 10048–10061. [Google Scholar] [CrossRef]
- Zhang, L.; Gengler, N.; Dehareng, F.; Colinet, F.; Froidmont, E.; Soyeurt, H. Can We Observe Expected Behaviors at Large and Individual Scales for Feed Efficiency-Related Traits Predicted Partly from Milk Mid-Infrared Spectra? Animals 2020, 10, 873. [Google Scholar] [CrossRef]
- Bittante, G.; Cipolat-Gotet, C.; Malchiodi, F.; Sturaro, E.; Tagliapietra, F.; Schiavon, S.; Cecchinato, A. Effect of dairy farming system, herd, season, parity, and days in milk on modeling of the coagulation, curd firming, and syneresis of bovine milk. J. Dairy Sci. 2015, 98, 2759–2774. [Google Scholar] [CrossRef]
- Cuvelier, C.; Dufrasne, I. L’alimentation de la vache laitiere aliments, calculs de ration, indicateurs d’évaluation des déséquilibres de la ration et pathologies d’origine. Livret Agric. 2005, 22, 1–100. [Google Scholar]
- Visentin, G.; Penasa, M.; Niero, G.; Cassandro, M.; De Marchi, M. Phenotypic characterisation of major mineral composition predicted by mid-infrared spectroscopy in cow milk. Ital. J. Anim. Sci. 2018, 17, 549–556. [Google Scholar] [CrossRef] [Green Version]
- Manuelian, C.L.; Penasa, M.; Visentin, G.; Zidi, A.; Cassandro, M.; De Marchi, M. Mineral composition of cow milk from multibreed herds. Anim. Sci. J. 2018, 89, 1622–1627. [Google Scholar] [CrossRef]
- Holt, C.; Carver, J.; Ecroyd, H.; Thorn, D. Invited review: Caseins and the casein micelle: Their biological functions, structures, and behavior in foods. J. Dairy Sci. 2013, 96, 6127–6146. [Google Scholar] [CrossRef]
- Kume, S.I.; Tanabe, S. Effect of Parity on Colostral Mineral Concentrations of Holstein Cows and Value of Colostrum as a Mineral Source for Newborn Calves. J. Dairy Sci. 1993, 76, 1654–1660. [Google Scholar] [CrossRef]
- Beckely, M.; Johnson, T. Five year study of California mastitis test testing on a commercial dairy herd. J. Dairy Sci. 1966, 49, 746. [Google Scholar]
- Blackburn, P. The variation in the cell count of cow’s milk throughout lactation and from one lactation to the next. J. Dairy Res. 1966, 33, 193–198. [Google Scholar] [CrossRef]
- Sharma, N.; Singh, N.; Bhadwal, M. Relationship of somatic cell count and mastitis: An overview. Asian Australas. J. Anim. Sci. 2011, 24, 429–438. [Google Scholar] [CrossRef]
| Traits | Related Parameter | N Records | Country | RPDcv | Author |
|---|---|---|---|---|---|
| Casein (g/100 g) | Cheese process | 996 | 1 | 4.46 | Unpublished |
| Citrate (mmol/L) | Energy balanced | 566 | 4 | 3.21 | Grelet et al. (2016) [32] |
| NAGase (Units/L) | Mastitis | 377 | 6 | 1.41 | Unpublished |
| LDH (Units/L) | Mastitis | 305 | 6 | 1.21 | Unpublished |
| Lactoferrin (mg/L) | Mastitis | 2654 | 4 | 1.43 | Soyeurt et al. (2020) [34] |
| RCT (s) | Cheese process | 337 | 1 | 1.54 | Colinet et al. (2015) [35] |
| k20 (s) | Cheese process | 337 | 1 | 1.24 | Colinet et al. (2015) [35] |
| a30 (s) | Cheese process | 337 | 1 | 1.54 | Colinet et al. (2015) [35] |
| Fresh ILCY (g curd/100 g) | Cheese process | 337 | 1 | 1.91 | Colinet et al. (2015) [35] |
| Mineral | Nsamples | Mean | SD | CV | Min | Max |
|---|---|---|---|---|---|---|
| Na | 1145 | 354.73 | 90.09 | 25.40 | 234.00 | 1273.00 |
| Ca | 1251 | 1148.40 | 132.96 | 11.58 | 593.40 | 1743.00 |
| Mg | 1281 | 99.38 | 13.04 | 13.12 | 60.82 | 156.60 |
| P | 1238 | 990.91 | 126.31 | 12.75 | 508.52 | 1447.00 |
| K | 1246 | 1513.83 | 148.88 | 9.83 | 819.15 | 1984.60 |
| 10-Fold Cross-Validation | Validation | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Subset | Mineral | Nsamples | Noutliers | LV | RMSEcv | R²cv | RPDcv | CCC | RMSEP | R²p | RPDp | CCCp | |
| Na | 1019 | 26 | 13 | 6.41 | 50.98 | 0.44 | 1.34 | 0.63 | 55.19 | 0.36 | 1.24 | 0.58 | |
| Without | Ca | 1094 | 57 | 10 | 46.22 | 53.38 | 0.82 | 2.34 | 0.90 | 71.74 | 0.78 | 1.74 | 0.81 |
| Austrian | Mg | 1124 | 57 | 11 | 4.28 | 6.53 | 0.72 | 1.88 | 0.85 | 8.11 | 0.51 | 1.51 | 0.68 |
| samples | P | 1083 | 55 | 10 | 22.72 | 58.71 | 0.75 | 1.99 | 0.86 | 81.31 | 0.83 | 1.44 | 0.75 |
| K | 1090 | 56 | 12 | 9.23 | 88.14 | 0.55 | 1.48 | 0.74 | 181.48 | 0.24 | 0.72 | 0.20 | |
| With 30 | Na | 997 | 78 | 13 | 7.65 | 49.05 | 0.43 | 1.16 | 0.58 | 54.56 | 0.33 | 1.04 | 0.54 |
| Austrian | Ca | 1106 | 75 | 10 | 33.04 | 53.52 | 0.81 | 2.31 | 0.90 | 63.63 | 0.77 | 1.94 | 0.84 |
| samples | Mg | 1164 | 47 | 11 | 13.81 | 6.87 | 0.69 | 1.79 | 0.83 | 7.30 | 0.55 | 1.69 | 0.72 |
| included | P | 1126 | 42 | 10 | 27.44 | 62.70 | 0.72 | 1.88 | 0.85 | 59.47 | 0.84 | 2.00 | 0.84 |
| K | 1120 | 56 | 12 | 8.35 | 91.30 | 0.54 | 1.46 | 0.73 | 152.89 | 0.26 | 0.87 | 0.29 | |
| 80% of | Na | 886 | 31 | 13 | 17.73 | 54.39 | 0.39 | 1.15 | 0.58 | 62.24 | 0.49 | 1.00 | 0.58 |
| randomly | Ca | 961 | 41 | 10 | 29.18 | 58.30 | 0.80 | 2.20 | 0.89 | 69.98 | 0.67 | 1.83 | 0.80 |
| selected | Mg | 998 | 27 | 11 | 8.51 | 7.18 | 0.66 | 1.70 | 0.81 | 7.78 | 0.69 | 1.57 | 0.79 |
| samples | P | 965 | 30 | 10 | 6.49 | 65.04 | 0.71 | 1.85 | 0.84 | 66.97 | 0.72 | 1.79 | 0.84 |
| K | 965 | 33 | 12 | 14.27 | 100.17 | 0.48 | 1.36 | 0.74 | 106.69 | 0.52 | 1.28 | 0.66 | |
| Reference Minerals | Predicted Minerals | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Traits | Na | Ca | Mg | P | K | pNa | pCa | pMg | pP | pK |
| pNa | 0.68 | 0.08 | 0.12 | 0.04 * | −0.15 | 0.02 | 0.06 | −0.16 | −0.27 | |
| pCa | 0.00 * | 0.86 | 0.58 | 0.42 | −0.28 | 0.06 * | 0.62 | 0.51 | −0.36 | |
| pMg | 0.06 * | 0.61 | 0.81 | 0.53 | 0.03 * | 0.16 * | 0.74 | 0.62 | −0.15 | |
| pP | 0.00 * | 0.44 | 0.53 | 0.82 | −0.18 | 0.03 * | 0.54 | 0.66 | 0.13 | |
| pK | −0.08 | −0.29 | −0.16 | 0.03 * | 0.67 | −0.14 | −0.40 | −0.24 | 0.03 * | |
| Milk Yields | −0.13 | −0.19 | −0.22 | −0.10 | 0.28 | −0.15 | −0.21 | −0.27 | −0.12 | 0.32 |
| pFat | −0.04 * | 0.27 | 0.27 | 0.11 | −0.21 | −0.09 | 0.39 | 0.34 | 0.11 | −0.41 |
| pProteins | 0.14 | 0.58 | 0.56 | 0.50 | −0.30 | 0.26 | 0.68 | 0.72 | 0.63 | −0.40 |
| pLactose | −0.41 | 0.07 * | −0.08 * | 0.07 | −0.06 * | −0.69 | 0.073 | −0.09 | 0.10 | −0.11 |
| pCasein | 0.09 * | 0.55 | 0.56 | 0.51 | −0.24 | 0.17 | 0.66 | 0.71 | 0.64 | −0.34 |
| pCitrate | −0.04 * | 0.43 | 0.48 | 0.12 | −0.06 * | −0.02 * | 0.54 | 0.59 | 0.19 | −0.07 * |
| pNAGase | 0.46 | 0.19 | 0.32 | 0.03 * | −0.15 | 0.74 | 0.21 | 0.42 | 0.06 * | −0.15 |
| pLDH | 0.32 | 0.11 | 0.24 | 0.08 * | −0.12 | 0.54 | 0.15 | 0.34 | 0.00 * | −0.15 |
| pLactoferrin | 0.39 | 0.17 | 0.08 * | 0.03 * | −0.25 | 0.62 | 0.18 | 0.13 | 0.04 | −0.29 |
| pRCT | 0.21 | 0.01 * | −0.01 * | −0.01 * | 0.07 * | 0.323 | −0.06 * | −0.07 * | −0.02 * | 0.09 * |
| pk20 | 0.06 * | −0.21 | −0.25 | −0.37 | 0.09 * | 0.098 | −0.30 | −0.46 | −0.28 | 0.15 |
| pa30 | 0.04 * | 0.23 | 0.25 | 0.35 | −0.10 | 0.07 * | 0.29 | 0.45 | 0.29 | −0.14 |
| pFresh ILCY | −0.02 * | 0.49 | 0.49 | 0.38 | −0.25 | −0.02 * | 0.61 | 0.58 | 0.41 | −0.43 |
| Mineral | N | Mean | SD | Min | Max |
|---|---|---|---|---|---|
| Na | 3,510,077 | 346 | 43 | 200 | 500 |
| Ca | 3,510,077 | 1163 | 102 | 900 | 1500 |
| Mg | 3,510,077 | 100 | 8 | 80 | 130 |
| P | 3,510,077 | 976 | 91 | 700 | 1300 |
| K | 3,510,077 | 1471 | 98 | 1100 | 1700 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christophe, O.S.; Grelet, C.; Bertozzi, C.; Veselko, D.; Lecomte, C.; Höeckels, P.; Werner, A.; Auer, F.-J.; Gengler, N.; Dehareng, F.; et al. Multiple Breeds and Countries’ Predictions of Mineral Contents in Milk from Milk Mid-Infrared Spectrometry. Foods 2021, 10, 2235. https://doi.org/10.3390/foods10092235
Christophe OS, Grelet C, Bertozzi C, Veselko D, Lecomte C, Höeckels P, Werner A, Auer F-J, Gengler N, Dehareng F, et al. Multiple Breeds and Countries’ Predictions of Mineral Contents in Milk from Milk Mid-Infrared Spectrometry. Foods. 2021; 10(9):2235. https://doi.org/10.3390/foods10092235
Chicago/Turabian StyleChristophe, Octave S., Clément Grelet, Carlo Bertozzi, Didier Veselko, Christophe Lecomte, Peter Höeckels, Andreas Werner, Franz-Josef Auer, Nicolas Gengler, Frédéric Dehareng, and et al. 2021. "Multiple Breeds and Countries’ Predictions of Mineral Contents in Milk from Milk Mid-Infrared Spectrometry" Foods 10, no. 9: 2235. https://doi.org/10.3390/foods10092235
APA StyleChristophe, O. S., Grelet, C., Bertozzi, C., Veselko, D., Lecomte, C., Höeckels, P., Werner, A., Auer, F.-J., Gengler, N., Dehareng, F., & Soyeurt, H. (2021). Multiple Breeds and Countries’ Predictions of Mineral Contents in Milk from Milk Mid-Infrared Spectrometry. Foods, 10(9), 2235. https://doi.org/10.3390/foods10092235






