1. Introduction
Salmonella is one of the most important foodborne pathogens and a leading cause of foodborne illness in humans in the EU [
1]. The source of the infection is usually contaminated food products of animal origin. Continuous surveillance of the occurrence of this pathogen in foods is required to ensure public health. Therefore, official food testing methodology according to European and national food legislation is necessary. Moreover, the crucial issues are the rapidity, efficiency, and accuracy of these methods. Traditionally, bacteriological culture methods have been used for isolation and identification of
Salmonella spp. Colonies with a morphology indicative of
Salmonella spp. are then selected for further studies, and identification is based on the determination of biochemical features and completed by serotyping. The specificity of meat samples makes it a challenging material for routine bacteriological testing as it contains a high number of indigenous microorganisms. Another complication may be the low
Salmonella number compared to other bacteria in the sample. Enrichment culture procedures are aimed at increasing the salmonellae population but, at the same time, the number of background bacteria also increases. The reliable identification of
Salmonella spp. is essential to ensure food safety. However, the presence of similarities in phenotypic characteristics between closely related bacteria may lead to incorrect identification. Bacteria belonging to the genus
Citrobacter are particularly often incorrectly identified as
Salmonella spp.
This study aimed to compare the identification of Salmonella spp. isolates obtained from meat samples using routine bacteriological methods and molecular biology methods.
2. Materials and Methods
2.1. Sampling
A total of 180 raw meat samples (60 beef, 60 pork, and 60 poultry) were obtained from meat processing plants cutting beef, pork, and poultry carcasses in central Poland. All samples were derived from carcass parts classified by official veterinary inspections as fit for human consumption. All samples collected as a single sample weighed at least 200 g for each type of meat (from parts of carcasses for culinary use, i.e., roast beef, entrecote, boneless ribs, neck, breast, and thigh). The meat samples were collected randomly using the aseptic technique and were packed in separate sterile bags, which were then labeled. All samples were transported to the laboratory in refrigerated containers at a temperature of 4 °C within one hour of collection.
2.2. Salmonella spp. Isolation and Identification
Salmonella spp. from all samples were isolated in accordance with PN-EN ISO 6579-1:2017-04 Microbiology of the food chain—Horizontal method for the detection, enumeration and serotyping of
Salmonella—Part 1: Detection of
Salmonella spp. [
2].
Samples were pre-enriched: for pork and beef samples, 10 g of each sample was mixed with 90 mL buffered peptone water (BPW GRASO, Starogard, Poland), and 25 g of each poultry sample was mixed with 225 mL BPW at a temperature of 25 °C (±3 °C) in sterile stomacher bags (Whirl-Pak, NAsco, Madison, WI, USA), placed in a stomacher and crushed for 2 min. The selective proliferation of Salmonella spp. was carried out using modified semi-solid Rappaport-Vassiliadis (MSRV) agar (GRASO, Starogard, Poland) and Muller–Kauffmann tetrathionate-novobiocin (MKTTn) broth (GRASO, Starogard, Poland). Two selective enrichment media, xylose lysine deoxycholate agar (XLD; GRASO, Starogard, Poland) and brilliant green agar (BGA; OXOID, Hampshire, United Kingdom), were used. Salmonella suspect colonies were transferred to a non-selective nutrient agar (GRASO, Starogard, Poland) to obtain the pure culture for further testing and a semi-solid medium by Garda for testing flagellar antigens. Serotyping was performed by slide agglutination with commercial H poly antisera for verification of the genus Salmonella enterica (IBSS Biomed, Kraków, Poland), O group antisera to determine O group (IBSS Biomed, Kraków, Poland), and H phase and H factor antisera to determine H phase and H factor (IBSS Biomed, Kraków, Poland, Bio-Rad, Hercules, CA, USA), according to the White–Kauffmann–Le Minor scheme.
2.2.1. Biochemical Strain Identification
Colonies showing morphology typical for Salmonella spp. on selective agars were subjected to biochemical identification using two commercially available tests: API 20E (BioMérieux, Craponne, France) and a VITEK2 COMPACT automated system for bacterial identification. VITEK® 2 GN cards (BioMérieux, Craponne, France) with reference strains for E. coli ATCC 25922, Salmonella Typhimurium ATCC 14028, Salmonella Enteritidis ATCC 1307,6 and Pseudomonas aeruginosa ATCC 27853 served as a quality check. Both tests were used according to the manufacturer’s instructions.
2.2.2. Confirmation of Salmonella Identification with Molecular Biology Methods
A real-time PCR method based on the detection of genes specific for Salmonella spp. was used to confirm presumptive identification. DNA for real-time PCR was extracted from bacterial cells using a commercial Kylt® DNA Extraction-Mix II (Anicon, Emstek, Germany). For detection of Salmonella spp., a commercial Kylt® Salmonella spp. (Anicon, Emstek, Germany) kit was used, and, for simultaneous detection of Salmonella Enteritidis and Salmonella Typhimurium, a commercial Spp-Se-St PCR (BioChek, Reeuwijk, The Netherland) kits was used. Both real-time PCR tests to detect Salmonella were performed according to the manufacturer’s instructions using an Applied Biosystems 7500 Fast Real-Time PCR System (Thermo, Waltham, MA, USA).
2.3. Antibiotic Resistance Test
Antimicrobial susceptibility was assessed by determining the MIC values using a VITEK® 2 System and an AST-GN96 card for Gram-negative bacteria (BioMérieux).
To analyze MIC patterns and detect phenotypes of Citrobacter braakii, an Advanced Expert System (AES, BioMérieux, Craponne, France) and VITEK® 2 GN 96 cards (BioMérieux, Craponne, France) were used. The MICs were interpreted according to the Clinical and Laboratory Standards Institute (CLSI) and FDA breakpoints (CLSI M100-ED28, 2018).
2.4. Statistical Assessment
Statistical testing was performed with a Statistica 13.1 software package (StatSoft, Kraków, Poland). Descriptive statistics were computed to determine the proportions of isolates resistant to different antimicrobial agents. The chi-square test was adopted for the determination of the statistical significance of differences between the proportions.
3. Results
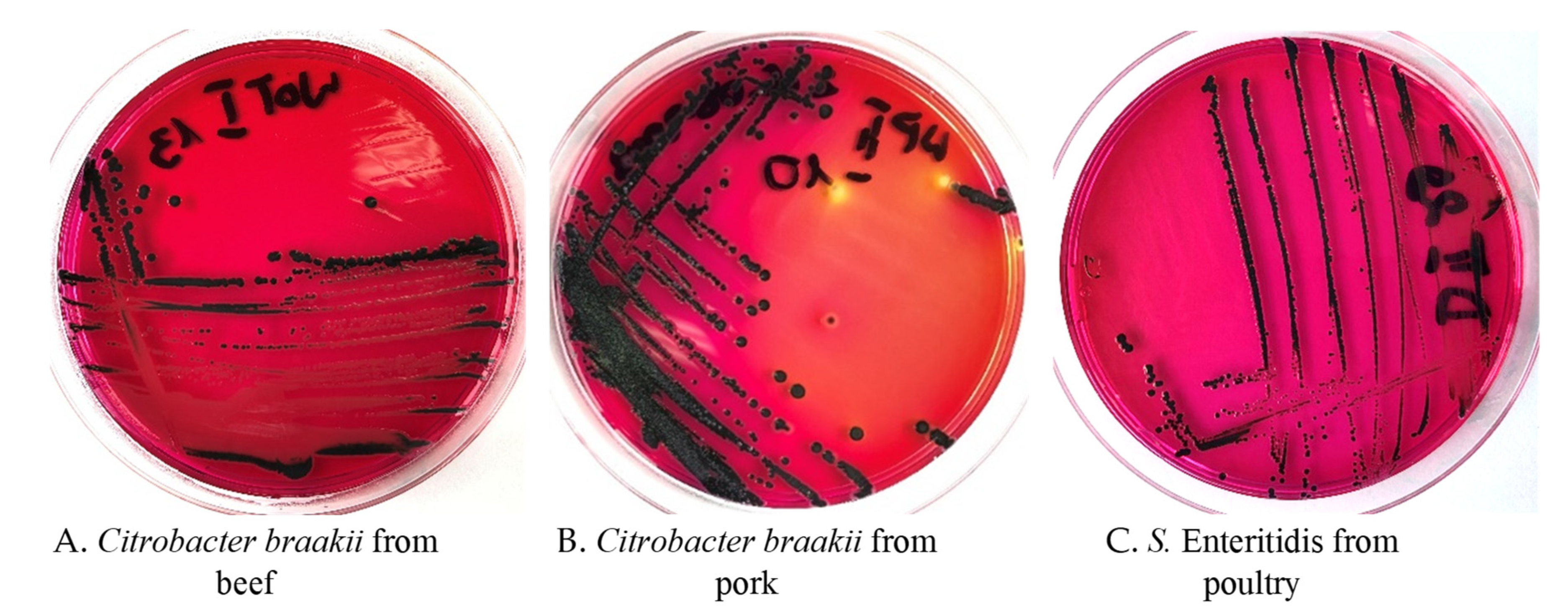
The accuracy of identification obtained with the first biochemical test, API 20E (BioMérieux, Craponne, France), for some of the isolates of
Salmonella like colonies on selective agars (
Figure 1) was unsatisfactory, e.g.,
Salmonella spp. 71.9% and
Citrobacter freundii 25% [
3] Sero-diagnosis was difficult because it showed autoagglutinations or the test with the group sera was positive (especially DO), which could cause presumptive
Salmonella diagnosis. Out of 180 meat samples, 23.33% indicated
Salmonella spp. or presumptive strains of
Salmonella. After obtaining the biochemical pattern from all 42 strains, 35 were confirmed as belonging to
Salmonella enterica spp.
enterica, 6 proved to be
Citrobacter braakii, and 1
Proteus mirabilis. The results of the occurrence of
Salmonella spp.,
C. braakii, and
Proteus mirabilis in the meat samples tested are presented in
Table 1.
The accuracy of identification obtained with the first biochemical test, API 20E (BioMérieux, Craponne, France), for some of the isolates of
Salmonella like colonies on selective agars (
Figure 1) was unsatisfactory, e.g.,
Salmonella spp. 71.9% and
Citrobacter freundii 25% [
3] Sero-diagnosis was difficult because it showed autoagglutinations or the test with the group sera was positive (especially DO), which could cause presumptive
Salmonella diagnosis. Out of 180 meat samples, 23.33% indicated
Salmonella spp. or presumptive strains of
Salmonella. After obtaining the biochemical pattern from all 42 strains, 35 were confirmed as belonging to
Salmonella enterica spp.
enterica, 6 proved to be
Citrobacter braakii, and 1
Proteus mirabilis. The results of the occurrence of
Salmonella spp.,
C. braakii, and
Proteus mirabilis in the meat samples tested are presented in
Table 1.
General results based on colony morphology on selective agars, biochemical properties, and the PCR technique are presented in
Table 2.
No Salmonella spp. were isolated from beef meat samples but three Citrobacter braakii were isolated. From pork meat samples, 1.67% (n = 1) was Salmonella spp. positive, Citrobacter braakii were isolated from three samples, and Proteus mirabilis from one. In poultry, 56.67% of samples (n = 35) isolated were Salmonella spp. Amongst isolated species, Salmonella enterica subsp. enterica was detected. The most common serovars were S. Enteritidis (55.88%, n = 19), S. Derby (14.71%, n = 5), and S. Newport (14.71%, n = 5); nd less frequently isolated were S. Infantis (5.88%, n = 2), S. Kentucky (5.88%, n = 2), and S. Mbandaka (2.94%, n = 1).
The colonies formed by
Citrobacter braakii and
Salmonella Enteritidis on XLD selective media look almost identical (
Figure 1).
The antibiotic resistance studies showed that all strains of
Citrobacter braakii were susceptible to ampicillin, cephalosporins (III generation cefoperazone, ceftiofur, and IV generation cefquinome), aminoglycosides (gentamicin, neomycin), enrofloxacin, and trimethoprim/sulfamethoxazole, while resistance to other antibiotics was variable. For individual strains of
Citrobacter braakii isolated from pork and beef samples,
Table 3 presents several multi-drug resistance patterns.
4. Discussion
The phenotypic heterogeneity amongst bacteria is well-known, especially amongst closely related microorganisms. However, in the case of pathogens such as
Salmonella, the misidentification may lead to a serious public health threat. In 2000, Manafi noticed that conventional media for the detection of
Salmonella have a very poor specificity, creating an abundance of false positives such as
Citrobacter or
Proteus among the rare real positive
Salmonella [
4]. Twenty years later, despite many modifications and the development of further enriched media for the detection of
Salmonella, there are still problems with the rapid identification of
Salmonella spp. in meat.
There is no information in the world literature about difficulties in interpreting the results obtained when the result is “presumably
Salmonella”. However, it should be remembered that the experience of a laboratory technician and their proficiency in conducting research are crucial for the correctness of the conducted research. At the same time, the necessity to introduce molecular diagnostics for the final confirmation of samples with the result “presumably
Salmonella” should be obligatory, not only to protect public health, but also in connection with the handling of meat on the EU market. Mistakenly qualifying
Citrobacter braakii as
Salmonella spp. may lead to the withdrawal from the market of meat or meat products and may cause risk of economic loss for farmers and producers. The requirements of the PN-EN ISO 6579-1: 2017-04 standard indicate that the result should specify the serotype of
Salmonella spp. with the provisions of the Commission Regulation (EC) No/ 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs [
2,
5]. However, in the Rapid Alert System for Food and Feed (RASFF) system, sometimes an alert notification occurs with a risk description of “serious” resulting in withdrawal from the customer because apathogenic microorganisms identified as “no definition–
Salmonella” in beef was detected [
6].
Reliable identification of
Salmonella is crucial for ensuring public health security. Culture-based methods, biochemical identification, and serotyping are traditionally used and recommended by relevant standards. However, considerable variability is observed in the biochemical properties of some
Salmonella, Proteus, and
Citrobacter isolates. The consequence of this is that some “presumptive
Salmonella” isolates cannot be identified by subsequent serotyping. Previous studies on the reliability of the API 20E test showed both good and inaccurate results. The presumptive
Salmonella identification was confirmed by API 20E, but 24% of the isolates were recognized as
Citrobacter spp. and 16.4% as
Proetus spp. [
7]. The results presented in this study are in accordance with the above observations. We found that 16.7% of 42 isolates recognized as presumptive
Salmonella were confirmed as
Citrobacter braakii.
Recently, many alternative non-culture methods have been described for
Salmonella detection. These methods are based on DNA analysis or immunological reactions. A significant disadvantage of traditional methods is the time required for all culture procedures, which may take up to a few days for a presumptive identification. The available literature data indicate that the evident superiority of DNA-based methods is their high specificity and that results can be obtained after 24 h [
8,
9].
Bacteria belonging to the
Citrobacter genus are closely related to
Salmonella; thus, some similarities in cell surface antigens and biochemical properties occur between them. Pilar et al. proved that approximately one-third of
Citrobacter and
Salmonella genes are composed of core genes, confirming their close relationships [
10]. This surprisingly high genotypic similarity can be explained by their common evolutionary history and genetic exchange [
11]. All the properties of
Citrobacter spp. listed above may result in false identification of these bacteria as
Salmonella. It is also important to mention that it takes another day or two to confirm questionable identifications and, therefore, it takes longer to obtain the results and increases the costs of analysis.
Our results suggest that DNA-based methods should be used in cases of questionable results of Salmonella identification based on phenotypic properties.
On the other hand, further research on C. braakii is desirable in order to determine the current potential pathogenicity for humans, especially in view of the developing antibiotic resistance of C. braakii, which may contribute to the spread of resistance genes in the environment.