1-Methylcyclopropene Preserves the Quality of Chive (Allium schoenoprasum L.) by Enhancing Its Antioxidant Capacities and Organosulfur Profile during Storage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Treatments
2.2. Reference Compounds
2.3. Determination of Chlorophyll Content and Yellowing Rate
2.4. Determination of Respiration Rate, MDA and H2O2
2.5. Determination of Antioxidants
2.6. Determination of Antioxidant Enzymes
2.7. Determination of GTP and Allinase
2.8. Determination of Organosulfur Compounds and Amino Acids
2.9. Statistical Analysis
3. Results
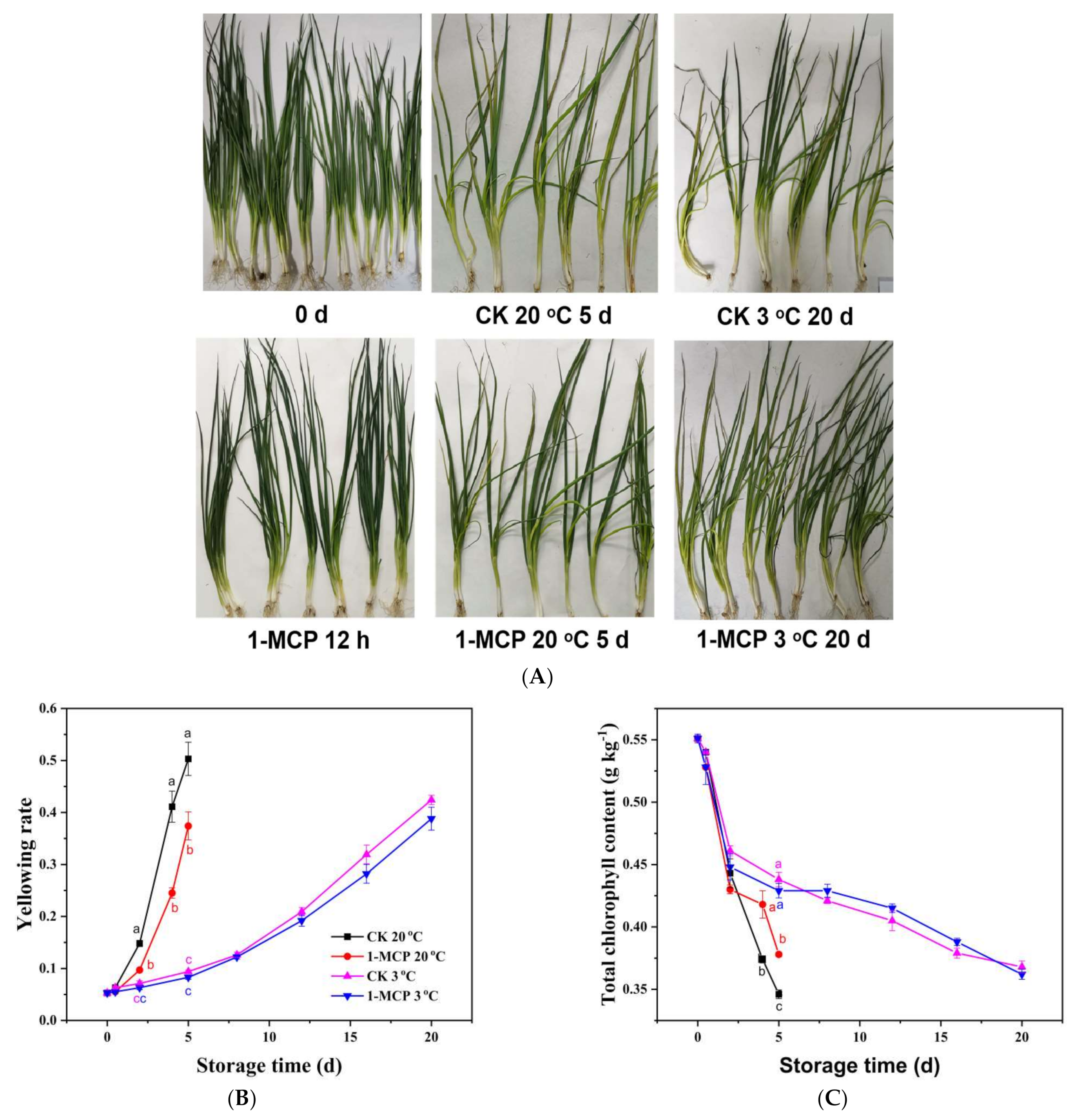
3.1. Yellowing Rate, Chlorophyll Content and Appearance Analysis
3.2. Respiration, H2O2 and MDA Analysis
3.3. Non-Enzymatic Antioxidants Analysis
3.4. Antioxidant Enzymes Analysis
3.5. Free Amino Acids Analysis
3.6. Organosulfur Compounds and Related Enzymes Analysis
4. Discussion
4.1. 1-MCP Enhances Antioxidant Capacity and Delays Senescence in Postharvest Chives
4.2. ACSOs Probably Act as Antioxidants Being Increased by 1-MCP in Postharvest Chives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, Z.; Ren, X.; Meng, X.; Zhang, Y.; Chen, D.; Tang, F. Novel Fluorescence Method for Detection of alpha-L-Fucosidase Based on CdTe Quantum Dots. Anal. Chem. 2012, 84, 4077–4082. [Google Scholar] [CrossRef]
- Fredotović, Ž.; Soldo, B.; Šprung, M.; Marijanović, Z.; Jerković, I.; Puizina, J. Comparison of Organosulfur and Amino Acid Composition between Triploid Onion Allium cornutum Clementi ex Visiani, 1842, and Common Onion Allium cepa L., and Evidences for Antiproliferative Activity of Their Extracts. Plants 2020, 9, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirschegger, P.; Jakše, J.; Trontelj, P.; Bohane, B. Origins of Allium ampeloprasum horticultural groups and a molecular phylogeny of the section Allium (Allium: Alliaceae). Mol. Phylogenet. Evol. 2010, 54, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Kamenetsky, R.; Rabinowitch, H.D. Physiology of Domesticated Alliums: Onions, Garlic, Leek, and Minor Crops. In Encyclopedia of Applied Plant Sciences, 2nd ed.; Thomas, B., Murray, B.G., Murphy, D.J., Eds.; Academic Press: Oxford, UK, 2017; pp. 255–261. [Google Scholar]
- Chen, H. Chives. In Handbook of Herbs and Spices; Woodhead Publishing: Sawston, UK, 2006; pp. 337–346. [Google Scholar]
- Yoshimoto, N.; Saito, K. S-Alk(en)ylcysteine sulfoxides in the genus Allium: Proposed biosynthesis, chemical conversion, and bioactivities. J. Exp. Bot. 2019, 70, 4123–4137. [Google Scholar] [CrossRef] [PubMed]
- Ougham, H.J.; Morris, P.; Thomas, H. The colors of autumn leaves as symptoms of cellular recycling and defenses against environmental stresses. Curr. Top. Dev. Biol. 2005, 66, 135–160. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Miller, G.; Morales, J.; Shulaev, V.; Angel Torres, M.; Mittler, R. Respiratory burst oxidases: The engines of ROS signaling. Curr. Opin. Plant Biol. 2011, 14, 691–699. [Google Scholar] [CrossRef]
- Tian, S.; Qin, G.; Li, B. Reactive oxygen species involved in regulating fruit senescence and fungal pathogenicity. Plant Mol. Biol. 2013, 82, 593–602. [Google Scholar] [CrossRef]
- Li, F.; Huang, H.; Ding, X.; Liu, J.; He, M.; Shan, Y.; Qu, H.; Jiang, Y. Effect of CPPU on postharvest attributes of Chinese flowering cabbage during storage. Postharvest Biol. Technol. 2021, 174, 111438. [Google Scholar] [CrossRef]
- Sun, B.; Yan, H.; Liu, N.; Wei, J.; Wang, Q. Effect of 1-MCP treatment on postharvest quality characters, antioxidants and glucosinolates of Chinese kale. Food Chem. 2012, 131, 519–526. [Google Scholar] [CrossRef]
- Xu, F.; Wang, H.; Tang, Y.; Dong, S.; Qiao, X.; Chen, X.; Zheng, Y. Effect of 1-methylcyclopropene on senescence and sugar metabolism in harvested broccoli florets. Postharvest Biol. Technol. 2016, 116, 45–49. [Google Scholar] [CrossRef] [Green Version]
- Al Ubeed, H.M.S.; Wills, R.B.H.; Bowyer, M.C.; Golding, J.B. Comparison of hydrogen sulphide with 1-methylcyclopropene (1-MCP) to inhibit senescence of the leafy vegetable, pak choy. Postharvest Biol. Technol. 2018, 137, 129–133. [Google Scholar] [CrossRef]
- Jiang, L.; Kang, R.; Zhang, L.; Jiang, J.; Yu, Z. Differential protein profiles of postharvest Gynura bicolor D.C leaf treated by 1-methylcyclopropene and ethephon. Food Chem. 2015, 176, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Hassan, F.A.S.; Mahfouz, S.A. Effect of 1-methylcyclopropene (1-MCP) on the postharvest senescence of coriander leaves during storage and its relation to antioxidant enzyme activity. Sci. Hortic. 2012, 141, 69–75. [Google Scholar] [CrossRef]
- Akan, S.; Tuna Gunes, N.; Yanmaz, R. Methyl jasmonate and low temperature can help for keeping some physicochemical quality parameters in garlic (Allium sativum L.) cloves. Food Chem. 2019, 270, 546–553. [Google Scholar] [CrossRef]
- Chope, G.A.; Cools, K.; Hammond, J.P.; Thompson, A.J.; Terry, L.A. Physiological, biochemical and transcriptional analysis of onion bulbs during storage. Ann. Bot. 2012, 109, 819–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamata, Y.; Aoyagi, M.; Sawada, Y.; Nakabayashi, R.; Hirai, M.Y.; Saito, K.; Imai, S. Changes in trans-S-1-Propenyl-L-cysteine Sulfoxide and Related Sulfur-Containing Amino Acids during Onion Storage. J. Agric. Food Chem. 2016, 64, 9063–9071. [Google Scholar] [CrossRef]
- Naheed, Z.; Cheng, Z.; Wu, C.; Wen, Y.; Ding, H. Total polyphenols, total flavonoids, allicin and antioxidant capacities in garlic scape cultivars during controlled atmosphere storage. Postharvest Biol. Technol. 2017, 131, 39–45. [Google Scholar] [CrossRef]
- Romo-Pérez, M.L.; Weinert, C.H.; Häußler, M.; Egert, B.; Frechen, M.A.; Trierweiler, B.; Kulling, S.E.; Zörb, C. Metabolite profiling of onion landraces and the cold storage effect. Plant Physiol. Biochem. 2020, 146, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Lande, N.V.; Subba, P.; Barua, P.; Gayen, D.; Prasad, T.S.K.; Chakraborty, S.; Chakraborty, N. Dissecting the chloroplast proteome of chickpea (Cicer arietinum L.) provides new insights into classical and non-classical functions. J. Proteom. 2017, 165, 11–20. [Google Scholar] [CrossRef]
- Du, Q.; Zhao, X.-H.; Xia, L.; Jiang, C.-J.; Wang, X.-G.; Han, Y.; Wang, J.; Yu, H.-Q. Effects of potassium deficiency on photosynthesis, chloroplast ultrastructure, ROS, and antioxidant activities in maize (Zea mays L.). J. Integr. Agric. 2019, 18, 395–406. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.; Lin, H.; Zhang, S.; Chen, Y.; Chen, M.; Lin, Y. The role of active oxygen metabolism in hydrogen peroxide-induced pericarp browning of harvested longan fruit. Postharvest Biol. Technol. 2014, 96, 42–48. [Google Scholar] [CrossRef]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Han, C.; Li, J.; Jin, P.; Li, X.; Wang, L.; Zheng, Y. The effect of temperature on phenolic content in wounded carrots. Food Chem. 2017, 215, 116–123. [Google Scholar] [CrossRef]
- Tareen, M.J.; Abbasi, N.A.; Hafiz, I.A. Postharvest application of salicylic acid enhanced antioxidant enzyme activity and maintained quality of peach cv. ’Flordaking’ fruit during storage. Sci. Hortic. 2012, 142, 221–228. [Google Scholar] [CrossRef]
- Razavi, F.; Hajilou, J. Enhancement of postharvest nutritional quality and antioxidant capacity of peach fruits by preharvest oxalic acid treatment. Sci. Hortic. 2016, 200, 95–101. [Google Scholar] [CrossRef]
- Zhao, F.; Qiao, X. Studies on the Purification and Partial Enzymology Characterization of γ-Glutamyl Transpeptidase in Garlic. J. Chin. Inst. Food Sci. Technol. 2009, 9, 41–45. [Google Scholar] [CrossRef]
- Hanum, T.; Sinha, N.K.; Cash, J.N. Characteristics of gamma-glutamyl-transpeptidase and alliinase of onion and their effects on the enhancement of pyruvate formation in onion macerates. J. Food Biochem. 1995, 19, 51–65. [Google Scholar] [CrossRef]
- Kubec, R.; Dadakova, E. Chromatographic methods for determination of S-substituted cysteine derivatives—A comparative study. J. Chromatogr. A 2009, 1216, 6957–6963. [Google Scholar] [CrossRef]
- Botton, A.; Tonutti, P.; Ruperti, B. Biology and Biochemistry of Ethylene. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Woodhead Publishing: Sawston, UK, 2019; pp. 93–112. [Google Scholar]
- Seymour, G.B.; Chapman, N.H.; Chew, B.L.; Rose, J.K.C. Regulation of ripening and opportunities for control in tomato and other fruits. Plant Biotechnol. J. 2013, 11, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Sohail, M.; Wills, R.B.H.; Bowyer, M.C.; Pristijono, P. Beneficial impact of exogenous arginine, cysteine and methionine on postharvest senescence of broccoli. Food Chem. 2021, 338, 128055. [Google Scholar] [CrossRef]
- Huan, C.; An, X.; Yu, M.; Jiang, L.; Ma, R.; Tu, M.; Yu, Z. Effect of combined heat and 1-MCP treatment on the quality and antioxidant level of peach fruit during storage. Postharvest Biol. Technol. 2018, 145, 193–202. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, C.; Chen, J.; Qiu, J.; Huang, Z.; Wang, Q.; Ye, Y. Cold acclimation improves photosynthesis by regulating the ascorbate-glutathione cycle in chloroplasts of Kandelia obovata. J. For. Res. 2019, 30, 755–765. [Google Scholar] [CrossRef]
- Yu, Z.; Zhang, W.; Yue, S.; Shen, C.; Yu, S. Effects of potassium nutrition on photosynthesis and senescence of winter wheat. Acta Agron. Sin. 1996, 305–312. [Google Scholar]
- Inestroza-Lizardo, C.; Mattiuz, B.-H.; da Silva, J.P.; Voigt, V.; Muniz, A.C.; Pinsetta, J.S. Effect of hyperbaric pressure on the activity of antioxidant enzymes and bioactive compounds of cv. ‘Débora’ tomatoes. Sci. Hortic. 2019, 249, 340–346. [Google Scholar] [CrossRef]
- Zhang, S.J.; Hu, T.T.; Liu, H.K.; Chen, Y.Y.; Pang, X.J.; Zheng, L.L.; Chang, S.M.; Kang, Y.F. Moderate vacuum packing and low temperature effects on qualities of harvested mung bean (Vigna radiata L.) sprouts. Postharvest Biol. Technol. 2018, 145, 83–92. [Google Scholar] [CrossRef]
- Song, L.; Yi, R.; Luo, H.; Jiang, L.; Gu, S.; Yu, Z. Postharvest 1-methylcyclopropene application delays leaf yellowing of pak choi (Brassica rapa subsp. chinensis) by improving chloroplast antioxidant capacity and maintaining chloroplast structural integrity during storage at 20 °C. Sci. Hortic. 2020, 270. [Google Scholar] [CrossRef]
- Palma, J.M.; Sandalio, L.M.; Corpas, F.J.; Romero-Puertas, M.C.; McCarthy, I.; del Rio, L.A. Plant proteases, protein degradation, and oxidative stress: Role of peroxisomes. Plant Physiol. Biochem. 2002, 40, 521–530. [Google Scholar] [CrossRef]
- Solomon, M.; Belenghi, B.; Delledonne, M.; Menachem, E.; Levine, A. The involvement of cysteine proteases and protease inhibitor genes in the regulation of programmed cell death in plants. Plant Cell 1999, 11, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Pu, Y.-Y.; Sun, D.-W. Recent advances in quality preservation of postharvest mushrooms (Agaricus bisporus): A review. Trends Food Sci. Technol. 2018, 78, 72–82. [Google Scholar] [CrossRef]
- Li, D.; Wang, D.; Fang, Y.; Belwal, T.; Li, L.; Lin, X.; Xu, Y.; Chen, H.; Zhu, M.; Luo, Z. Involvement of energy metabolism and amino acid metabolism in quality attributes of postharvest Pleurotus eryngii treated with a novel phase change material. Postharvest Biol. Technol. 2021, 173. [Google Scholar] [CrossRef]
- Chung, L.Y. The Antioxidant Properties of Garlic Compounds: Allyl Cysteine, Alliin, Allicin, and Allyl Disulfide. J Med. Food 2006, 9, 205–213. [Google Scholar] [CrossRef]
- Kim, J.-M.; Chang, H.J.; Kim, W.-K.; Chang, N.; Chun, H.S. Structure−Activity Relationship of Neuroprotective and Reactive Oxygen Species Scavenging Activities for Allium Organosulfur Compounds. J. Agric. Food Chem. 2006, 54, 6547–6553. [Google Scholar] [CrossRef]
- Kourounakis, P.N.; Rekka, E.A. Effect on active oxygen species of alliin and allium sativum (garlic) powder. Res. Commun. Chem. Pathol. Pharmacol. 1991, 74, 249–252. [Google Scholar]
- Avgeri, I.; Zeliou, K.; Petropoulos, S.A.; Bebeli, P.J.; Papasotiropoulos, V.; Lamari, F.N. Variability in Bulb Organosulfur Compounds, Sugars, Phenolics, and Pyruvate among Greek Garlic Genotypes: Association with Antioxidant Properties. Antioxidants 2020, 9, 967. [Google Scholar] [CrossRef]
- Fei, M.L.I.; Tong, L.I.; Wei, L.I.; De Yang, L. Changes in antioxidant capacity, levels of soluble sugar, total polyphenol, organosulfur compound and constituents in garlic clove during storage. Ind. Crop. Prod. 2015, 69, 137–142. [Google Scholar] [CrossRef]
- Hornickova, J.; Kubec, R.; Cejpek, K.; Velisek, J.; Ovesna, J.; Stavelikova, H. Profiles of S-Alk(en)ylcysteine Sulfoxides in Various Garlic Genotypes. Czech J. Food Sci. 2010, 28, 298–308. [Google Scholar] [CrossRef] [Green Version]
- Lancaster, J.E.; Shaw, M.L. Metabolism of γ-glutamyl peptides during development, storage and sprouting of onion bulbs. Phytochemistry 1991, 30, 2857–2859. [Google Scholar] [CrossRef]
- Hughes, J.; Tregova, A.; Tomsett, A.B.; Jones, M.G.; Cosstick, R.; Collin, H.A. Synthesis of the flavour precursor, alliin, in garlic tissue cultures. Phytochemistry 2005, 66, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Mitrova, K.; Svoboda, P.; Milella, L.; Ovesna, J. Alliinase and cysteine synthase transcription in developing garlic (Allium sativum L.) over time. Food Chem. 2018, 251, 103–109. [Google Scholar] [CrossRef] [PubMed]






| Group | Treatment | Storage Condition | Sample Time |
|---|---|---|---|
| CK 20 °C | No | 20 °C | Day 0, 0.5, 2, 4 and 5 |
| 1-MCP 20 °C | 1-MCP | 20 °C | Day 0, 0.5, 2, 4 and 5 |
| CK 3 °C | No | 3 °C | Day 0, 0.5, 2, 5, 8, 12, 16 and 20 |
| 1-MCP 3 °C | 1-MCP | 3 °C | Day 0, 0.5, 2, 5, 8, 12, 16 and 20 |
| Compounds Name | Storage Period (d) | 0 | 0.5 | 2 | 4 | 5 | 8 | 12 | 16 | 20 |
|---|---|---|---|---|---|---|---|---|---|---|
| MCSO (g kg−1) | CK 20 °C | 2.44 ± 0.08 a | 2.23 ± 0.00 b | 2.10 ± 0.02 c,C | 1.99 ± 0.09 d | 2.16 ± 0.07 b,B | nd | nd | nd | nd |
| 1-MCP 20 °C | 2.44 ± 0.08 b | 2.12 ± 0.10 c | 2.40 ± 0.08 b,A | 2.08 ± 0.08 c | 2.70 ± 0.12 a,A | nd | nd | nd | nd | |
| CK 3 °C | 2.44 ± 0.08 b | 2.23 ± 0.00 c,d | 2.25 ± 0.01 c,B | nd | 2.12 ± 0.14 d,e,B | 2.04 ± 0.10 e | 2.15 ± 0.13 c,d,e | 2.52 ± 0.02 b | 2.85 ± 0.11 a | |
| ACSO (g kg−1) | CK 20 °C | 0.35 ± 0.01 c | 0.62 ± 0.06 b,A | 0.57 ± 0.04 b | 0.57 ± 0.06 b | 1.01 ± 0.04 a,A | nd | nd | nd | nd |
| 1-MCP 20 °C | 0.35 ± 0.01 c | 0.41 ± 0.04 c,B | 0.56 ± 0.03 b | 0.62 ± 0.05 b | 1.06 ± 0.09 a,A | nd | nd | nd | nd | |
| CK 3 °C | 0.35 ± 0.01 e | 0.62 ± 0.06 d,A | 0.56 ± 0.03 d | nd | 0.72 ± 0.05 c,B | 0.75 ± 0.02 c | 0.82 ± 0.06 b | 0.83 ± 0.07 b | 1.07 ± 0.09 a | |
| PCSO (g kg−1) | CK 20 °C | 0.13 ± 0.00 a | 0.12 ± 0.00 b,B | 0.11 ± 0.00 d,B | 0.11 ± 0.01 d,A | 0.12 ± 0.00 c | nd | nd | nd | nd |
| 1-MCP 20 °C | 0.13 ± 0.00 b | 0.23 ± 0.01 a,A | 0.12 ± 0.01 c,A | 0.09 ± 0.01 d,B | 0.12 ± 0.01 b,c | nd | nd | nd | nd | |
| CK 3 °C | 0.13 ± 0.00 b | 0.12 ± 0.00 c,B | 0.10 ± 0.00 d,B | nd | 0.12 ± 0.00 c | 0.09 ± 0.01 e | 0.12 ± 0.00 c | 0.13 ± 0.00 b | 0.16 ± 0.01 a | |
| PeCSO (g kg−1) | CK 20 °C | 7.38 ± 0.17 a | 6.16 ± 0.01 c,B | 6.27 ± 0.06 c,B | 6.69 ± 0.32 b,B | 7.66 ± 0.26 a,B | nd | nd | nd | nd |
| 1-MCP 20 °C | 7.38 ± 0.17 b | 6.67 ± 0.27 c,A | 7.23 ± 0.22 b,A | 7.38 ± 0.25 b,A | 8.39 ± 0.29 a,A | nd | nd | nd | nd | |
| CK 3 °C | 7.38 ± 0.17 e | 6.16 ± 0.01 g,B | 7.02 ± 0.04 f,A | nd | 7.90 ± 0.16 c,B | 7.60 ± 0.26 d,e | 7.78 ± 0.17 c,d | 8.26 ± 0.07 b | 9.68 ± 0.33 a | |
| Total ACSOs (g kg−1) | CK 20 °C | 10.30 ± 0.03 b | 9.14 ± 0.07 c | 9.04 ± 0.12 c,B | 9.37 ± 0.48 c,B | 10.95 ± 0.37 a,B | nd | nd | nd | nd |
| 1-MCP 20 °C | 10.30 ± 0.03 b | 9.43 ± 0.58 c | 10.30 ± 0.39 b,A | 10.17 ± 0.42 b,A | 12.27 ± 0.69 a,A | nd | nd | nd | nd | |
| CK 3 °C | 10.30 ± 0.03 d,e | 9.14 ± 0.07 f | 9.93 ± 0.07 e,A | nd | 10.85 ± 0.38 c,B | 10.48 ± 0.47 c,d | 10.87 ± 0.40 c | 11.74 ± 0.16 b | 13.77 ± 0.53 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, X.; Lu, Y.; Yang, Y.; Yu, Z. 1-Methylcyclopropene Preserves the Quality of Chive (Allium schoenoprasum L.) by Enhancing Its Antioxidant Capacities and Organosulfur Profile during Storage. Foods 2021, 10, 1792. https://doi.org/10.3390/foods10081792
Dai X, Lu Y, Yang Y, Yu Z. 1-Methylcyclopropene Preserves the Quality of Chive (Allium schoenoprasum L.) by Enhancing Its Antioxidant Capacities and Organosulfur Profile during Storage. Foods. 2021; 10(8):1792. https://doi.org/10.3390/foods10081792
Chicago/Turabian StyleDai, Xiaomei, Yaping Lu, Yuan Yang, and Zhifang Yu. 2021. "1-Methylcyclopropene Preserves the Quality of Chive (Allium schoenoprasum L.) by Enhancing Its Antioxidant Capacities and Organosulfur Profile during Storage" Foods 10, no. 8: 1792. https://doi.org/10.3390/foods10081792
APA StyleDai, X., Lu, Y., Yang, Y., & Yu, Z. (2021). 1-Methylcyclopropene Preserves the Quality of Chive (Allium schoenoprasum L.) by Enhancing Its Antioxidant Capacities and Organosulfur Profile during Storage. Foods, 10(8), 1792. https://doi.org/10.3390/foods10081792






