Chemical Elements and the Quality of Mānuka (Leptospermum scoparium) Honey
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Honey Analysis
2.3. Soil and Plant Analysis
2.4. Quality Control
2.5. Statistical Analysis
3. Results and Discussion
3.1. Soil and Plants
3.2. Honey
3.3. Interactions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stephens, J.M.C.; Molan, P.C.; Clarkson, B.D. A review of Leptospermum scoparium (Myrtaceae) in New Zealand. N. Z. J. Bot. 2005, 43, 431–449. [Google Scholar] [CrossRef]
- Thompson, J.A. A revision of the genus Leptospermum (Myrtaceae). Telopea 1989, 3, 301–449. [Google Scholar] [CrossRef]
- MPI (Ministry for Primary Industries). Apiculture. Ministry for Primary Industries 2019 Apiculture Monitoring Programme; Ministry for Primary Industries: Wellington, New Zealand, 2019. [Google Scholar]
- Molan, P.C. The antibacterial activity of honey: 2. Variation in the potency of the antibacterial activity. N. Z. Bee World 1992, 73, 59–76. [Google Scholar] [CrossRef]
- Adams, C.J.; Boult, C.H.; Deadman, B.J.; Farr, J.M.; Grainger, M.N.; Manley-Harris, M.; Snow, M.J. Isolation by HPLC and characterisation of the bioactive fraction of New Zealand manuka (Leptospermum scoparium) honey. Carbohydr. Res. 2008, 343, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Mavric, E.; Wittmann, S.; Barth, G.; Henle, T. Identification and quantification of methylglyoxal as the dominant antibacterial constituent of manuka (Leptospermum scoparium) honeys from New Zealand. Mol. Nutr. Food Res. 2008, 52, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Carter, D.A.; Blair, S.E.; Cokcetin, N.N.; Bouzo, D.; Brooks, P.; Schothauer, R.; Harry, E.J. Therapeutic Manuka Honey: No Longer So Alternative. Front. Microbiol. 2016, 7, 569. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.J.; Manley-Harris, M.; Molan, P.C. The origin of methylglyoxal in New Zealand manuka (Leptospermum scoparium) honey. Carbohydr. Res. 2009, 344, 1050–1053. [Google Scholar] [CrossRef]
- Norton, A.M.; McKenzie, L.N.; Brooks, P.R.; Pappalardo, L.J. Quantitation of dihydroxyacetone in Australian Leptospermum nectar via High-Performance Liquid Chromatography. J. Agric. Food Chem. 2015, 63, 6513–6517. [Google Scholar] [CrossRef]
- Atrott, J.; Haberlau, S.; Henle, T. Studies on the formation of methylglyoxal from dihydroxyacetone in manuka (Leptospermum scoparium) honey. Carbohydr. Res. 2012, 361, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.E.L.; Brown, H.L.; Jenkins, R.E. On the antibacterial effects of manuka honey: Mechanistic insights. Res. Rep. Biol. 2015, 6, 215–224. [Google Scholar]
- Clearwater, M.J.; Revell, M.; Noe, S.; Manley-Harris, M. Influence of genotype, floral stage, and water stress on floral nectar yield and composition of mānuka (Leptospermum scoparium). Ann. Bot. 2018, 121, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Czipa, N.; Diosi, G.; Phillips, C.; Kovacs, B. Examination of honeys and flowers as soil element indicators. Environ. Monit. Assess 2017, 189, 412. [Google Scholar] [CrossRef]
- Xun, E.; Zhang, Y.; Zhao, J.; Guo, J. Translocation of heavy metals from soils into floral organs and rewards of Cucurbita pepo: Implications for plant reproductive fitness. Ecotoxicol. Environ. Saf. 2017, 145, 235–243. [Google Scholar] [CrossRef]
- Acquarone, C.; Buera, P.; Elizalde, B. Pattern of pH and electrical conductivity upon honey dilution as a complementary tool for discriminating geographical origin of honeys. Food Chem. 2007, 101, 695–703. [Google Scholar] [CrossRef]
- Baroni, M.V.; Podio, N.S.; Badini, R.G.; Inga, M.; Ostera, H.A.; Cagnoni, M.; Gautier, E.A.; Garcia, P.P.; Hoogewerff, J.; Wunderlin, D.A. Linking soil, water, and honey composition to assess the geographical origin of argentinean honey by multielemental and isotopic analyses. J. Agric. Food Chem. 2015, 63, 4638–4645. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Porto, A.V.; Martin Arroyo, T.; Bartolome Esteban, C. How soil type (gypsum or limestone) influences the properties and composition of thyme honey. Springerplus 2016, 5, 1663. [Google Scholar] [CrossRef][Green Version]
- Nickless, E.M.; Anderson, C.W.N.; Hamilton, G.; Stephens, J.M.; Wargent, J. Soil influences on plant growth, floral density and nectar yield in three cultivars of mānuka (Leptospermum scoparium). N. Z. J. Bot. 2017, 55, 100–117. [Google Scholar] [CrossRef]
- Williams, S.; King, J.; Revell, M.; Manley-Harris, M.; Balks, M.; Janusch, F.; Kiefer, M.; Clearwater, M.; Brooks, P.; Dawson, M. Regional, annual, and individual variations in the dihydroxyacetone content of the nectar of mānuka (Leptospermum scoparium) in New Zealand. J. Agric. Food Chem. 2014, 62, 10332–10340. [Google Scholar] [CrossRef]
- Hamilton, G.; Millner, J.P.; Robertson, A.; Stephens, J. Assessment of manuka provenances for production of high ‘unique manuka factor’ honey. Agron. N. Z. 2013, 43, 139–144. [Google Scholar]
- Millner, J.P.; Hamilton, G.; Ritchie, C.; Stephens, J. High UMF® honey production from manuka plantations. Hill Ctry. Grassl. Res. Pract. Ser. 2016, 16, 113–118. [Google Scholar]
- Noe, S.; Manley-Harris, M.; Clearwater, M.J. Floral nectar of wild mānuka (Leptospermum scoparium) varies more among plants than among sites. N. Z. J. Crop Hortic. Sci. 2019, 47, 282–296. [Google Scholar] [CrossRef]
- Stephens, J.M.C. The Factors Responsible for the Varying Levels of UMF® in Mānuka (Leptospermum scoparium) honey. Ph.D. Thesis, The University of Waikato, Hamilton, New Zealand, 2006. Available online: https://hdl.handle.net/10289/2655 (accessed on 1 May 2021).
- Esperschuetz, J.; Anderson, C.; Bulman, S.; Katamian, O.; Horswell, J.; Dickinson, N.M.; Robinson, B.H. Response of Leptospermum scoparium, Kunzea robusta and Pinus radiata to contrasting biowastes. Sci. Total Environ. 2017, 587–588, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.C. Investigation of Geographical Correlations between Honey Methylglyoxal Content and Dihydroxacetone in Manuka Floral Nectar. Master’s Thesis, The University of Auckland, Auckland, New Zealand, 2014. Available online: http://hdl.handle.net/2292/22629 (accessed on 3 May 2021).
- Williams, S. Mapping Current and Potential Honey Production from the Leptospermum genus in Australia. Ph.D. Thesis, University of the Sunshine Coast, Sippy Downs, QLD, Australia, 2019. Available online: https://research.usc.edu.au/permalink/61USC_INST/3k4ahb/alma99450654802621 (accessed on 18 April 2021).
- Pohl, P.; Sergiel, I.; Stecka, H. Determination and fractionation of metals in honey. Crit. Rev. Anal. Chem. 2009, 39, 276–288. [Google Scholar] [CrossRef]
- Windsor, S.; Pappalardo, M.; Brooks, P.; Williams, S.; Manley-Harris, M. A convenient new analysis of dihydroxyacetone and methylglyoxal applied to Australian Leptospermum honeys. J. Pharmacogn. Phytother. 2012, 4, 6–11. [Google Scholar] [CrossRef]
- Esperschuetz, J.; Bulman, S.; Anderson, C.; Lense, O.; Horswell, J.; Dickinson, N.; Robinson, B.H. Production of biomass crops using biowastes on low-fertility soil: 2. Effect of biowastes on nitrogen transformation processes. J. Environ. Qual. 2016, 45, 1970–1978. [Google Scholar] [CrossRef] [PubMed]
- Blakemore, L.C.; Searle, P.L.; Daly, B.K. Methods for Chemical Analysis of Soils; NZ Soil Bureau Scientific Report 80; Department of Scientific and Industrial Research: Lower Hutt, New Zealand, 1987. [Google Scholar]
- Reiser, R.; Simmler, M.; Portmann, D.; Clucas, L.; Schulin, R.; Robinson, B. Cadmium concentrations in New Zealand pastures: Relationships to soil and climate variables. J. Environ. Qual. 2014, 43, 917–925. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 1 April 2021).
- Cowie, J.D. Yellow-brown loams-definition and classification. In Soil Grups of New Zealand. Part 6, Yellow-Brown Loams; Neall, V.E., Ed.; New Zealand Society of Soil Science: Wellington, New Zealand, 1982. [Google Scholar]
- Berninger, E. Diversity of Tolerance to Soil pH in Leptospermum scoparium (J.R. and G.Forst) Populations from New Zealand. Acta Hortic. 1992, 320, 49–56. [Google Scholar] [CrossRef]
- Blume, H.-P.; Brümmer, G.W.; Fleige, H.; Horn, R.; Kandeler, E.; Kögel-Knabner, I.; Kretzschmar, R.; Stahr, K.; Wilke, B.-M. Scheffer/Schachtschabel Soil Science; Springer: Berlin/Heidelberg, Gernamy, 2016. [Google Scholar]
- Kirkby, E. Introduction, Definition and Classification of Nutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Academic Press: London, UK, 2012; pp. 3–5. [Google Scholar]
- Gutierrez-Gines, M.J.; Madejon, E.; Lehto, N.J.; McLenaghen, R.D.; Horswell, J.; Dickinson, N.; Robinson, B.H. Response of a pioneering species (Leptospermum scoparium J.R.Forst. & G.Forst.) to heterogeneity in a low-fertility soil. Front. Plant Sci. 2019, 10, 93. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.V.P.; Gutiérrez-Ginés, M.J.; Smith, C.M.S.; Lehto, N.J.; Robinson, B.H. Mānuka ( Leptospermum scoparium ) roots forage biosolids in low fertility soil. Environ. Exp. Bot. 2017, 133, 151–158. [Google Scholar] [CrossRef]
- Chaney, R.L. Toxic element accumulation in soils and crops: Protecting soil fertility and agricultural food-chains. In Inorganic Contaminants in the Vadose Zone; Bar-Yosef, B., Barrow, N.J., Goldshmid, J., Eds.; Ecological Studies; Springer: Berlin, Germany, 1989; pp. 140–158. [Google Scholar]
- Jarrell, W.; Beverly, R. The dilution effect in plant nutrition studies. Adv. Agron. 1981, 34, 197–224. [Google Scholar]
- Stephens, J.M.; Schlothauer, R.C.; Morris, B.D.; Yang, D.; Fearnley, L.; Greenwood, D.R.; Loomes, K.M. Phenolic compounds and methylglyoxal in some New Zealand manuka and kanuka honeys. Food Chem. 2010, 120, 78–86. [Google Scholar] [CrossRef]
- Vanhanen, L.P.; Emmertz, A.; Savage, G.P. Mineral analysis of mono-floral New Zealand honey. Food Chem. 2011, 128, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Alqarni, A.S.; Owayss, A.A.; Mahmoud, A.A.; Hannan, M.A. Mineral content and physical properties of local and imported honeys in Saudi Arabia. J. Saudi Chem. Soc. 2014, 18, 618–625. [Google Scholar] [CrossRef]
- Deng, J.; Liu, R.; Lu, Q.; Hao, P.; Xu, A.; Zhang, J.; Tan, J. Biochemical properties, antibacterial and cellular antioxidant activities of buckwheat honey in comparison to manuka honey. Food Chem. 2018, 252, 243–249. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Tan, S.W.; Yusof, Y.A.; Chua, L.S. Classification of honey from its bee origin via chemical profiles and mineral content. Food Anal. Methods 2016, 10, 19–30. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Chowdhury, M.A.; Rahman, M.A.; Sulaiman, S.A.; Gan, S.H. Determination of mineral, trace element, and pesticide levels in honey samples originating from different regions of Malaysia compared to manuka honey. Biomed. Res. Int. 2014, 2014, 359890. [Google Scholar] [CrossRef]
- Singh, I.; Singh, S. Honey moisture reduction and its quality. J. Food Sci. Technol. 2018, 55, 3861–3871. [Google Scholar] [CrossRef] [PubMed]
- Solayman, M.; Islam, M.A.; Paul, S.; Ali, Y.; Khalil, M.I.; Alam, N.; Gan, S.H. Physicochemical properties, minerals, trace elements, and heavy metals in honey of different origins: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 219–233. [Google Scholar] [CrossRef]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for nutrition and health: A review. J. Am. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef]
- González-Miret, M.L.; Terrab, A.; Hernanz, D.; Fernandéz-Recamales, M.Á.; Heredia, F.J. Multivariate correlation between color and mineral composition of honeys and by their botanical origin. J. Agric. Food Chem. 2005, 53, 2574–2580. [Google Scholar] [CrossRef]
- Machado De-Melo, A.A.; de Almeida-Muradian, L.B.; Sancho, M.T.; Pascual-Maté, A. Composition and properties of Apis mellifera honey: A review. J. Apic. Res. 2017, 57, 5–37. [Google Scholar] [CrossRef]
- Grainger, M.N.C.; Klaus, H.; Hewitt, N.; French, A.D. Investigation of inorganic elemental content of honey from regions of North Island, New Zealand. Food Chem. 2021, 361, 130110. [Google Scholar] [CrossRef] [PubMed]
- Klikocka, H.; Marks, M. Sulphur and Nitrogen Fertilization as a Potential Means of Agronomic Biofortification to Improve the Content and Uptake of Microelements in Spring Wheat Grain DM. J. Chem. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Hawkesford, M.; Horst, W.; Kichey, T.; Lambers, H.; Schjoerring, J.; Møller, I.S.; White, P. Functions of macronutrients. In Marschner’s Mineral Nutrition of Higher Plants; Marschner, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 135–189. [Google Scholar]
- Smallfield, B.M.; Joyce, N.I.; van Klink, J.W. Developmental and compositional changes in Leptospermum scoparium nectar and their relevance to mānuka honey bioactives and markers. N. Z. J. Bot. 2018, 56, 183–197. [Google Scholar] [CrossRef]
- Wenzler, M.; Holscher, D.; Oerther, T.; Schneider, B. Nectar formation and floral nectary anatomy of Anigozanthos flavidus: A combined magnetic resonance imaging and spectroscopy study. J. Exp. Bot. 2008, 59, 3425–3434. [Google Scholar] [CrossRef][Green Version]
- Liang, J.-Y.; Zhang, Y.; Huang, S.; Lipscomb, W.N. Allosteric transition of fructose-1, 6-bisphosphatase. Proc. Natl. Acad. Sci. USA 1993, 90, 2132–2136. [Google Scholar] [CrossRef]
- Lambeir, A.M.; Opperdoes, F.R.; Wierenga, R.K. Kinetic properties of triose—Phosphate isomerase from Trypanosoma brucei brucei: A comparison with the rabbit muscle and yeast enzymes. Eur. J. Biochem. 1987, 168, 69–74. [Google Scholar] [CrossRef]
- Carter, C.; Thornburg, R.W. Is the nectar redox cycle a floral defense against microbial attack? Trends Plant Sci. 2004, 9, 320–324. [Google Scholar] [CrossRef]
- Brudzynski, K.; Abubaker, K.; Castle, A. Re-Examining the Role of Hydrogen Peroxide in Bacteriostatic and Bactericidal Activities of Honey. Front. Microbiol. 2011, 2, 213. [Google Scholar] [CrossRef]
- Maksimovic, V.; Mojovic, M.; Vucinic, Z. Monosaccharide-H2O2 reactions as a source of glycolate and their stimulation by hydroxyl radicals. Carbohydr. Res. 2006, 341, 2360–2369. [Google Scholar] [CrossRef]
- Xun, E.; Zhang, Y.; Zhao, J.; Guo, J. Heavy metals in nectar modify behaviors of pollinators and nectar robbers: Consequences for plant fitness. Environ. Pollut. 2018, 242, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Meindl, G.A.; Bain, D.J.; Ashman, T.-L. Variation in nickel accumulation in leaves, reproductive organs and floral rewards in two hyperaccumulating Brassicaceae species. Plant Soil 2014, 383, 349–356. [Google Scholar] [CrossRef]
- Bogdanov, S.; Haldimann, M.; Luginbühl, W.; Gallmann, P. Minerals in honey: Environmental, geographical and botanical aspects. J. Apic. Res. Bee World 2007, 46, 269–275. [Google Scholar] [CrossRef]
- Porter, N.G.; Smale, P.E.; Nelson, M.A.; Hay, A.J.; Van Klink, J.W.; Dean, C.M. Variability in essential oil chemistry and plant morphology within a Leptospermum scoparium population. N. Z. J. Bot. 1998, 36, 125–133. [Google Scholar] [CrossRef]
- Prober, S.M.; Wiehl, G. Relationships among soil fertility, native plant diversity and exotic plant abundance inform restoration of forb-rich eucalypt woodlands. Divers. Distrib. 2012, 18, 795–807. [Google Scholar] [CrossRef]
- Porrini, C.; Ghini, S.; Girotti, S.; Sabatini, A.G.; Gattavecchia, E.; Celli, G. Use of honey bees as bioindicators of environmental pollution in Italy. In Honey Bees: Estimating the Environmental Impact of Chemicals; Devillers, J., Pham-Delegue, M.-H., Eds.; Taylor & Francis: London, UK, 2002. [Google Scholar]
- Scripca, L.A.; Amariei, S. The Influence of Chemical Contaminants on the Physicochemical Properties of Unifloral and Multifloral Honey. Foods 2021, 10, 1039. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Gasparrini, M.; Forbes-Hernandez, T.Y.; Mazzoni, L.; Giampieri, F. The Composition and Biological Activity of Honey: A Focus on Manuka Honey. Foods 2014, 3, 420–432. [Google Scholar] [CrossRef]
- Stoewsand, G.S.; Stamer, J.R.; Kosikowski, F.V.; Morse, R.A.; Bache, C.A.; Lisk, D.J. Chromium and nickel in acidic foods and by-products containing stainless steel during processing. Bull. Environ. Contam. Toxicol. 1979, 21, 600–603. [Google Scholar] [CrossRef]
- Tong, S.S.C.; Morse, R.A.; Bache, C.A.; Lisk, D.J. Elemental analysis of honey as an indicator of pollution. Arch. Environ. Health 1975, 30, 329–332. [Google Scholar] [CrossRef]
- Pita-Calvo, C.; Guerra-Rodriguez, M.E.; Vazquez, M. Analytical Methods Used in the Quality Control of Honey. J. Agric. Food Chem. 2017, 65, 690–703. [Google Scholar] [CrossRef] [PubMed]
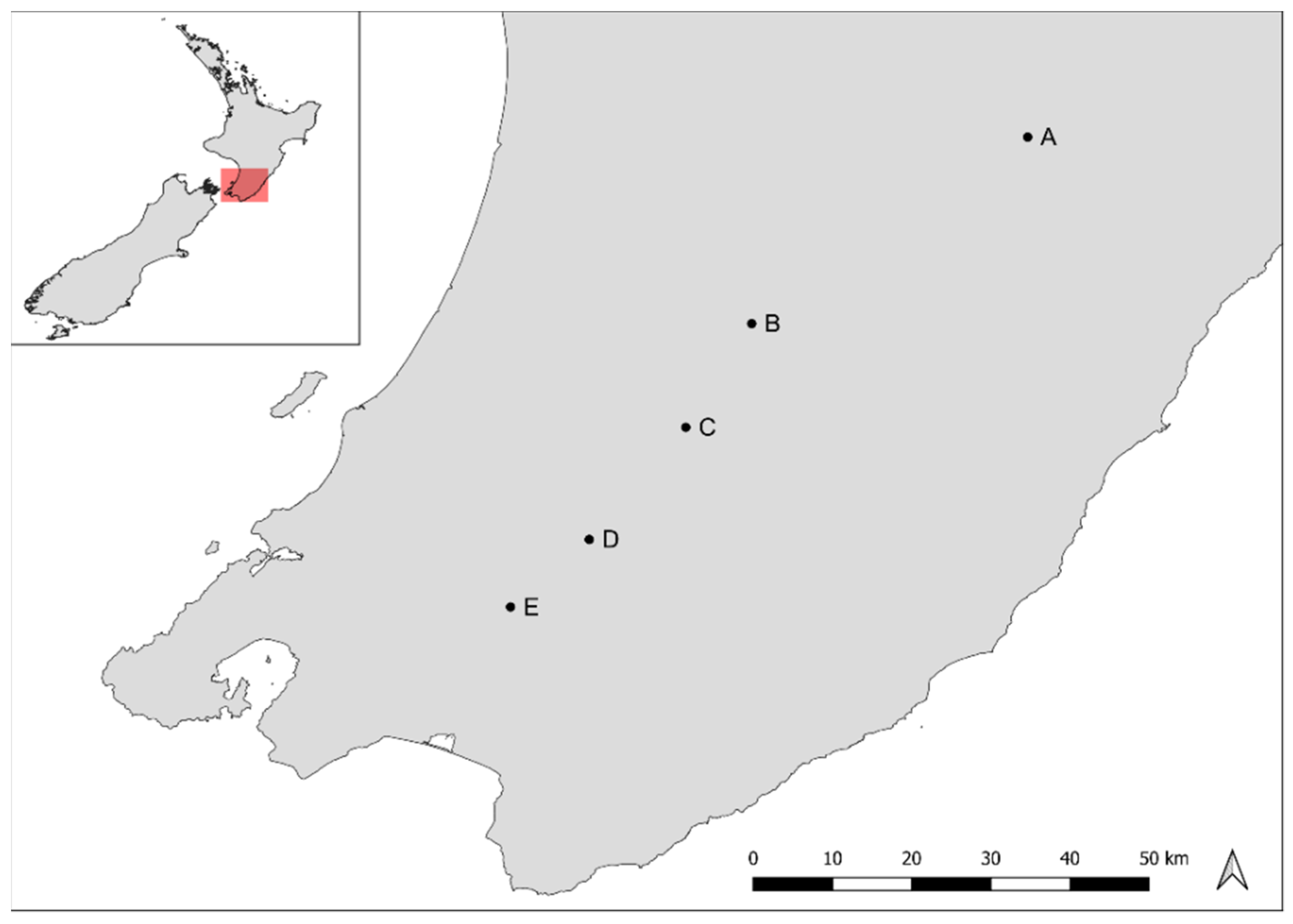

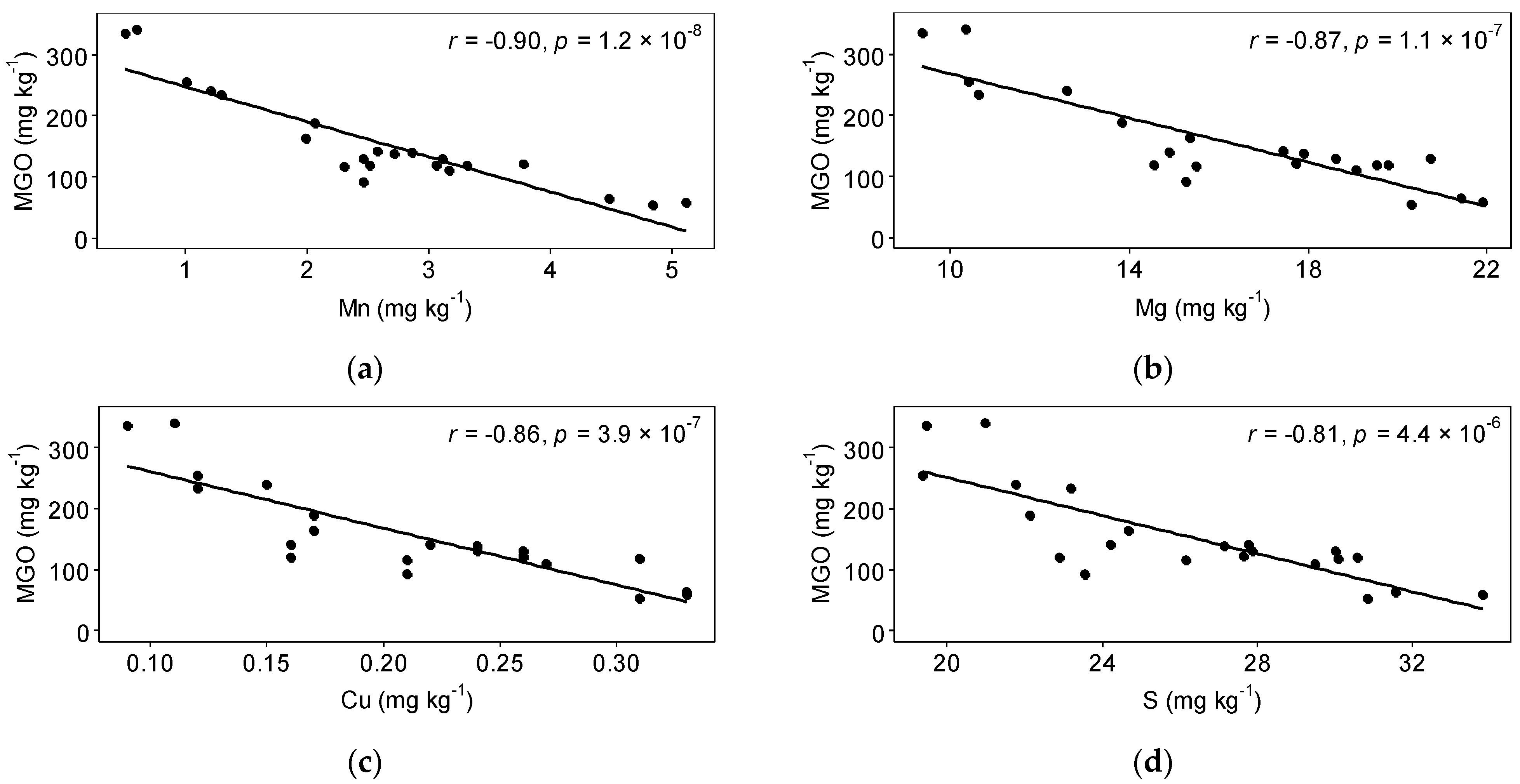
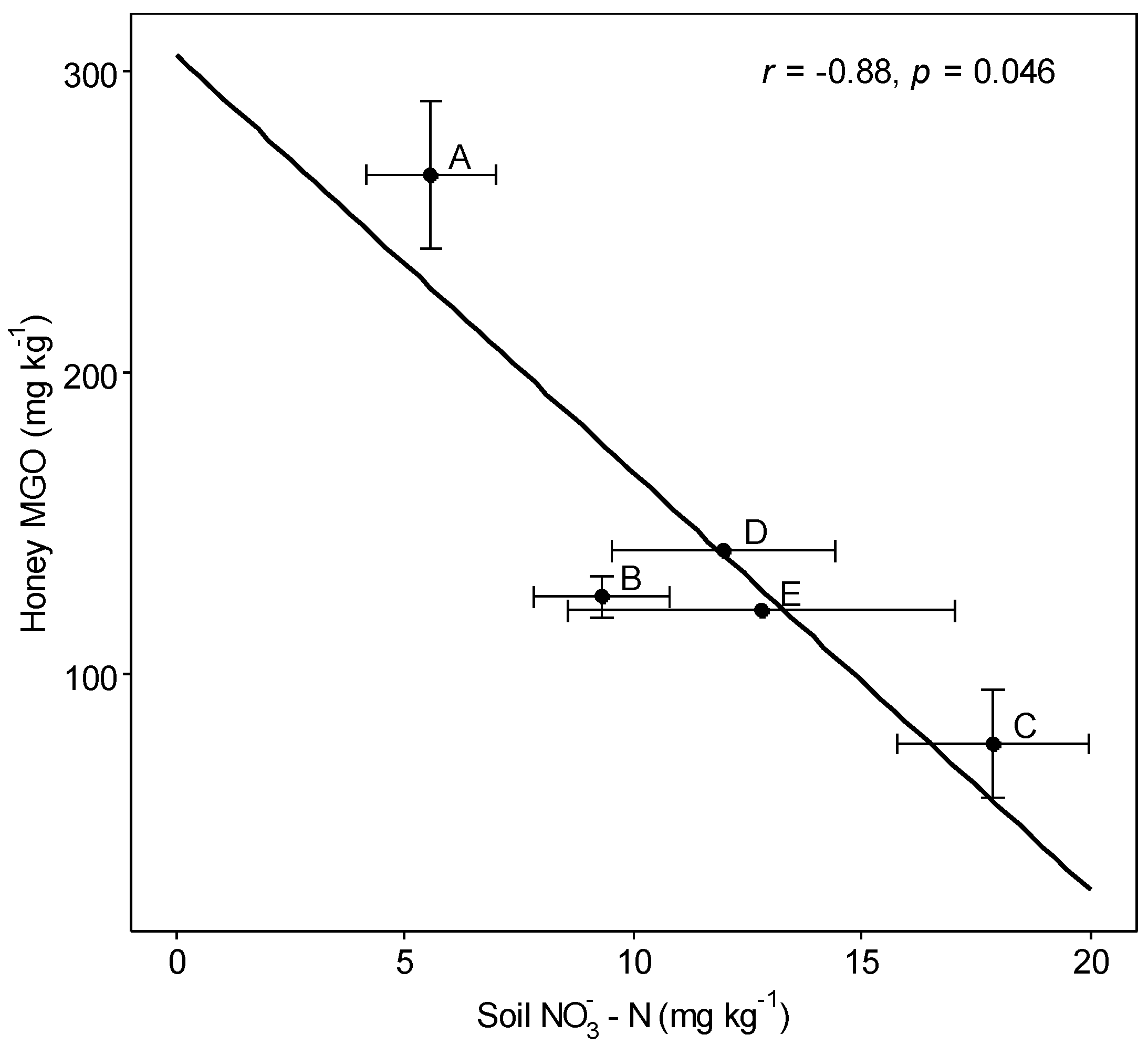
| Site | A | B | C | D | E |
|---|---|---|---|---|---|
| n = 6 | n = 9 | n = 4 | n = 1 | n = 2 | |
| MGO | 266 ± 25 a | 126 ± 6.9 b | 77 ± 18 b | 141 | 121 |
| DHA | 3246 ± 221 a | 1856 ± 71 b | 1293 ± 226 b | 1983 | 1642 |
| Al | 6.5 ± 0.91 a | 11 ± 0.60 b | 9.1 ± 0.34 ab | 5.1 | 5.5 |
| B | 2.8 ± 0.18 a | 2.8 ± 0.10 a | 2.5 ± 0.12 a | 2.8 | 2.8 |
| Ba | 0.08 ± 0.01 a | 0.09 ± 0.00 a | 0.11 ± 0.00 b | 0.09 | 0.09 |
| Ca | 61 ± 2.9 a | 60 ± 2.5 a | 61 ± 1.9 a | 62 | 68 |
| Cr | 0.02 ± 0.01 a | 0.02 ± 0.00 a | 0.02 ± 0.00 a | 0.02 | 0.03 |
| Cu | 0.13 ± 0.01 a | 0.24 ± 0.01 b | 0.30 ± 0.02 c | 0.16 | 0.21 |
| Fe | 1.2 ± 0.45 a | 1.1 ± 0.09 a | 0.91 ± 0.12 a | 0.72 | 0.95 |
| K | 487 ± 14 a | 671 ± 25 b | 1108 ± 55 c | 463 | 465 |
| Mg | 11 ± 0.68 a | 18 ± 0.61 b | 21 ± 0.35 c | 15 | 16 |
| Mn | 1.1 ± 0.23 a | 2.7 ± 0.15 b | 4.2 ± 0.60 c | 2.9 | 3.2 |
| Na | 27 ± 0.99 a | 34 ± 0.99 b | 47 ± 2.2 c | 38 | 40 |
| P | 54 ± 0.99 a | 50 ± 1.4 a | 65 ± 4.4 b | 62 | 64 |
| S | 21 ± 0.62 a | 28 ± 0.85 b | 31 ± 43 b | 24 | 25 |
| Zn | 0.32 ± 0.02 a | 0.36 ± 0.03 a | 0.47 ± 0.03 b | 0.37 | 0.47 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meister, A.; Gutierrez-Gines, M.J.; Maxfield, A.; Gaw, S.; Dickinson, N.; Horswell, J.; Robinson, B. Chemical Elements and the Quality of Mānuka (Leptospermum scoparium) Honey. Foods 2021, 10, 1670. https://doi.org/10.3390/foods10071670
Meister A, Gutierrez-Gines MJ, Maxfield A, Gaw S, Dickinson N, Horswell J, Robinson B. Chemical Elements and the Quality of Mānuka (Leptospermum scoparium) Honey. Foods. 2021; 10(7):1670. https://doi.org/10.3390/foods10071670
Chicago/Turabian StyleMeister, Alexandra, Maria Jesus Gutierrez-Gines, Aydin Maxfield, Sally Gaw, Nicholas Dickinson, Jacqui Horswell, and Brett Robinson. 2021. "Chemical Elements and the Quality of Mānuka (Leptospermum scoparium) Honey" Foods 10, no. 7: 1670. https://doi.org/10.3390/foods10071670
APA StyleMeister, A., Gutierrez-Gines, M. J., Maxfield, A., Gaw, S., Dickinson, N., Horswell, J., & Robinson, B. (2021). Chemical Elements and the Quality of Mānuka (Leptospermum scoparium) Honey. Foods, 10(7), 1670. https://doi.org/10.3390/foods10071670








