Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease
Abstract
1. Introduction
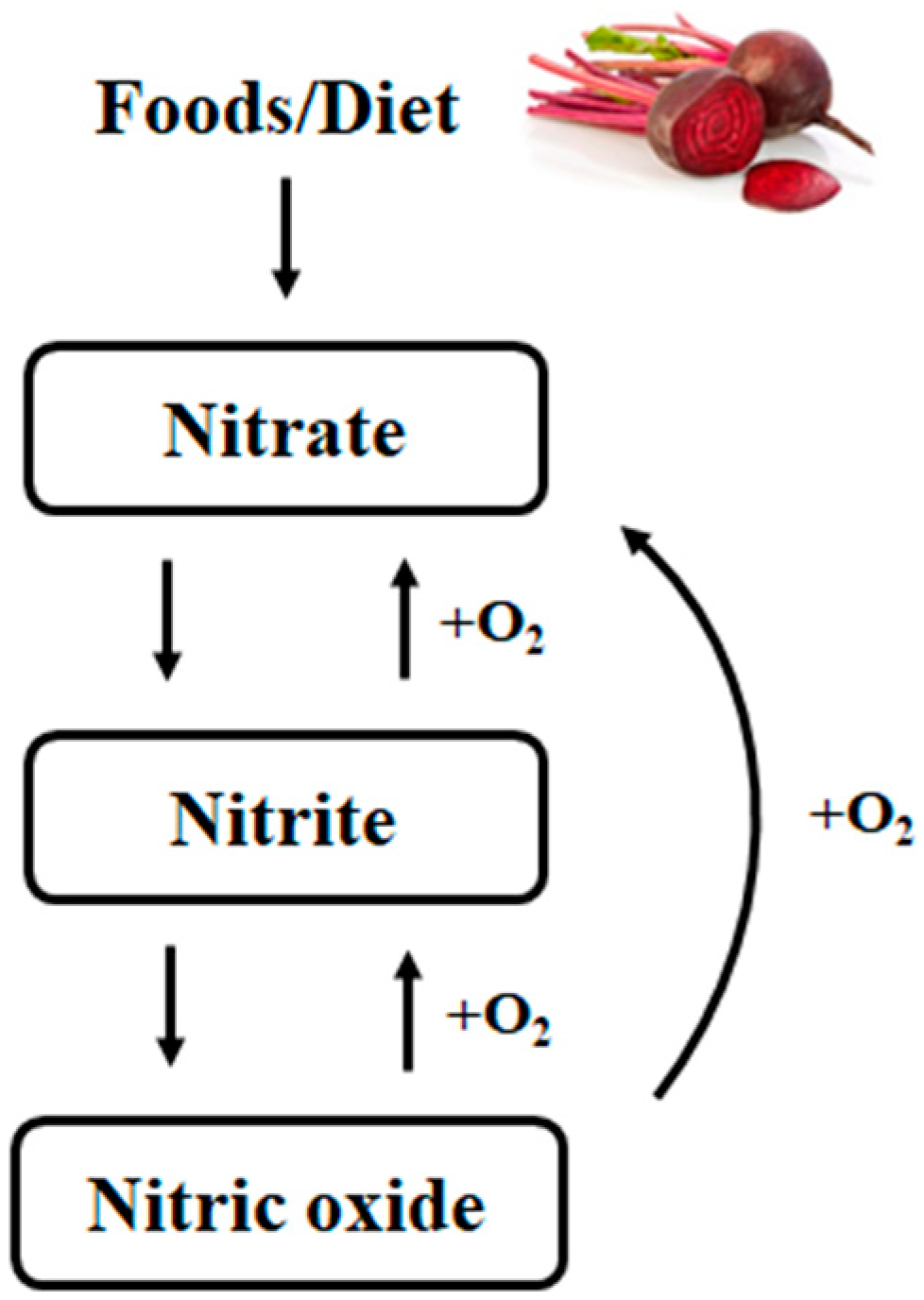
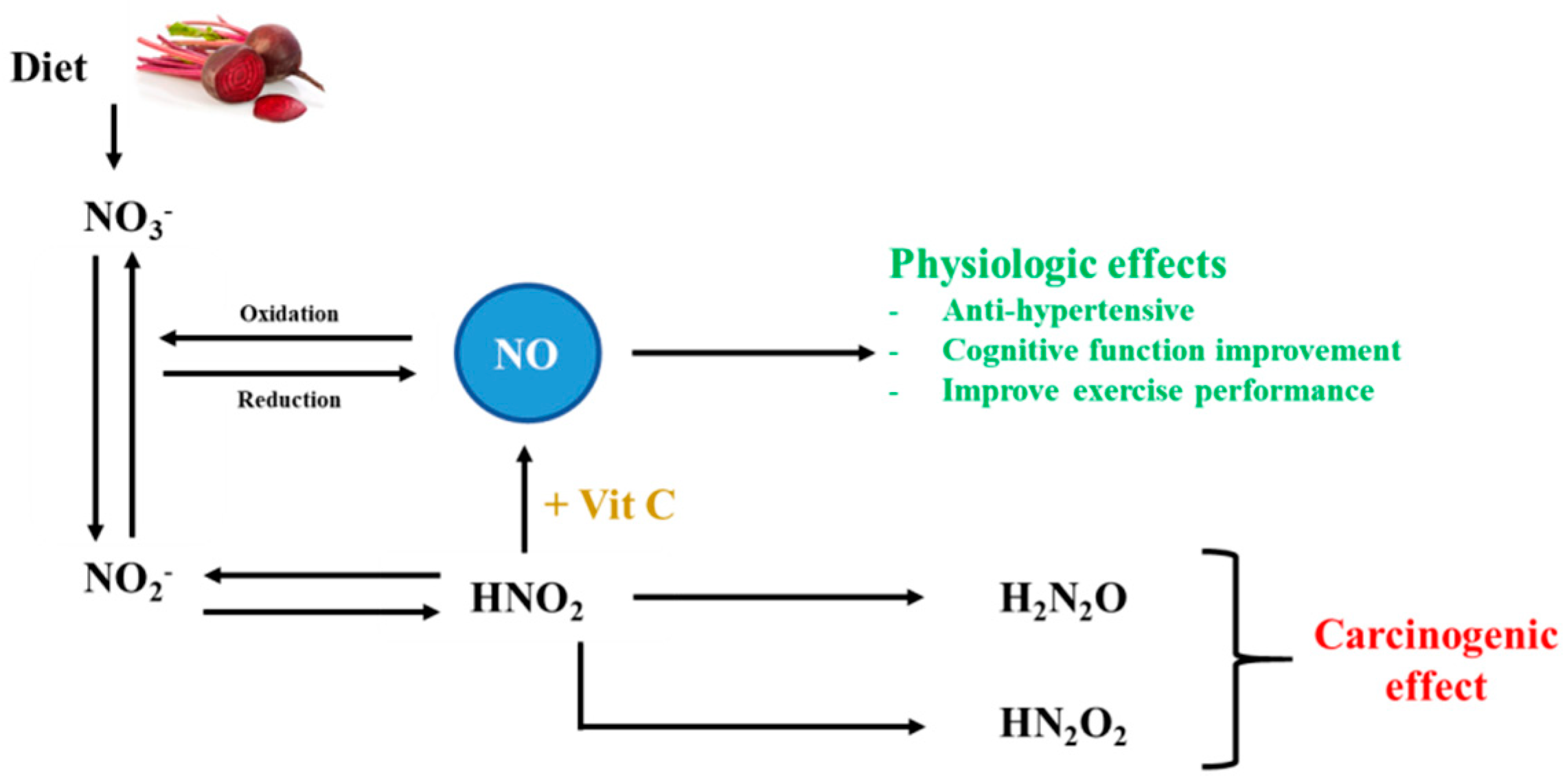
2. Nitrate
2.1. Bioavailability
2.2. Beneficial Health Effects of Nitrate
2.2.1. Antihypertensive Effects
2.2.2. Effects on Cognitive Function
2.2.3. Nitrate as an Ergogenic Aid to Improve Exercise Performance
2.3. Interactions and Adverse Effects of Nitrate Consumption
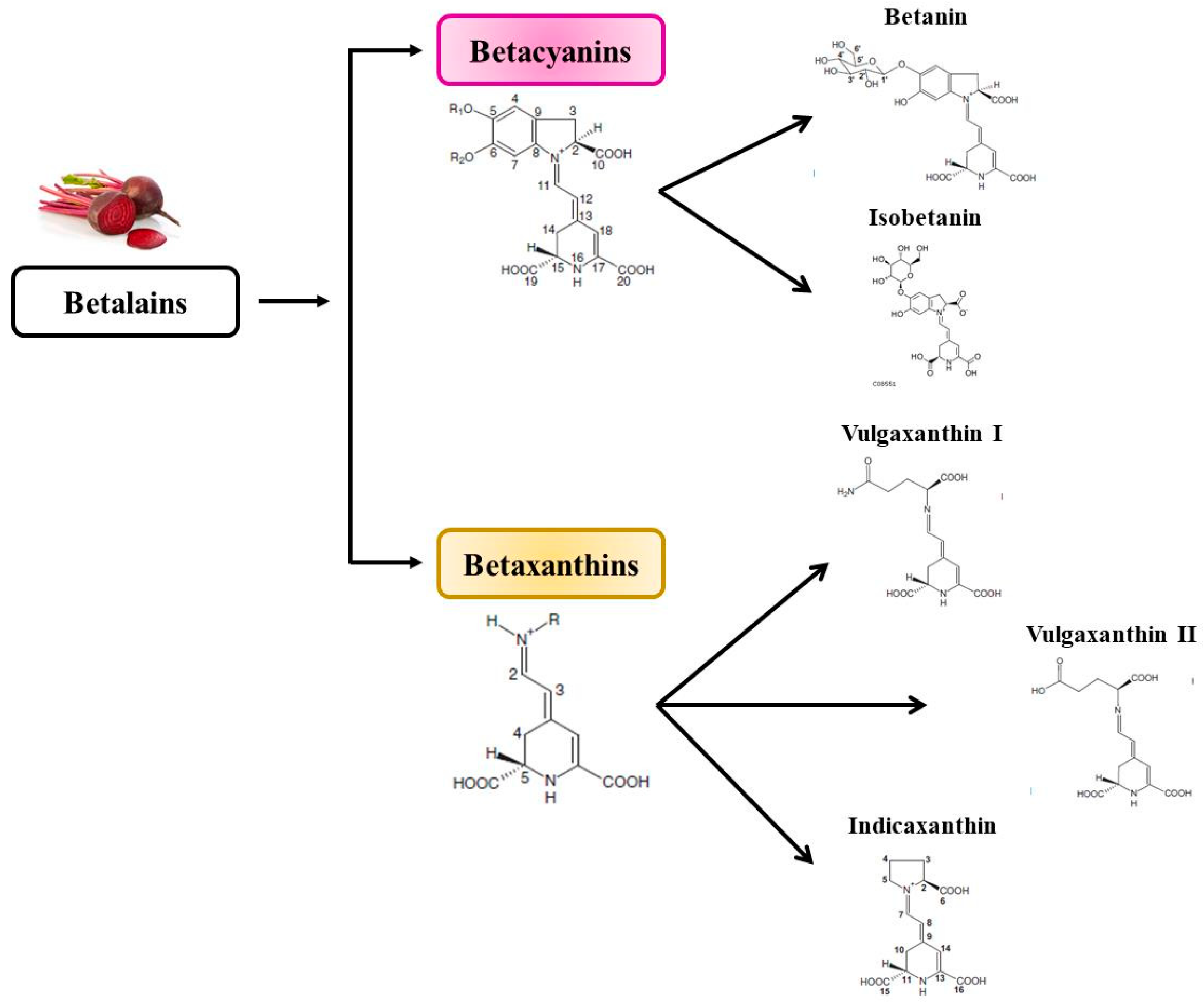
3. Betalains
3.1. Bioavailability
3.2. Beneficial Health Effects of Betalains
3.2.1. Effects on CVD
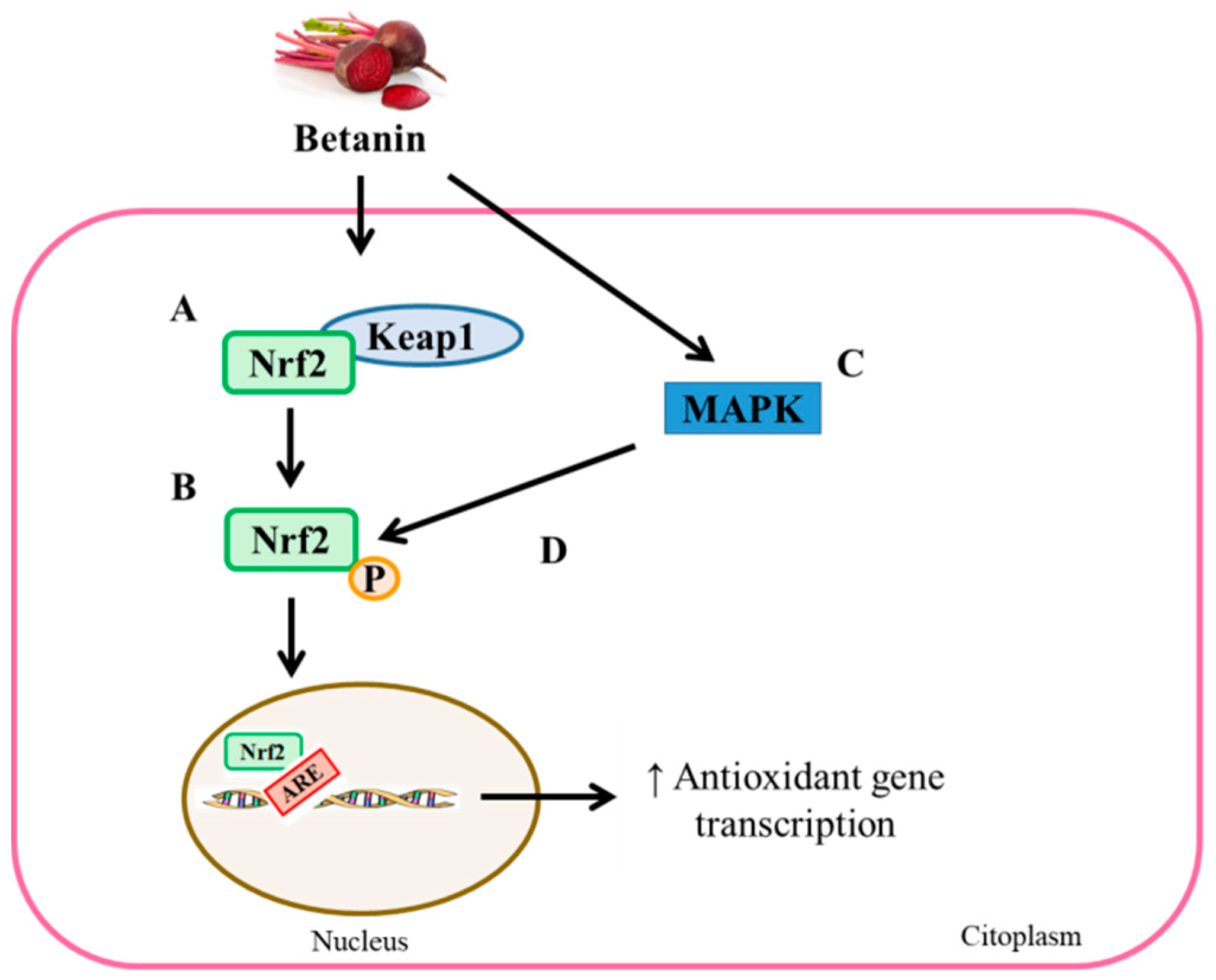
3.2.2. Antioxidant Effects
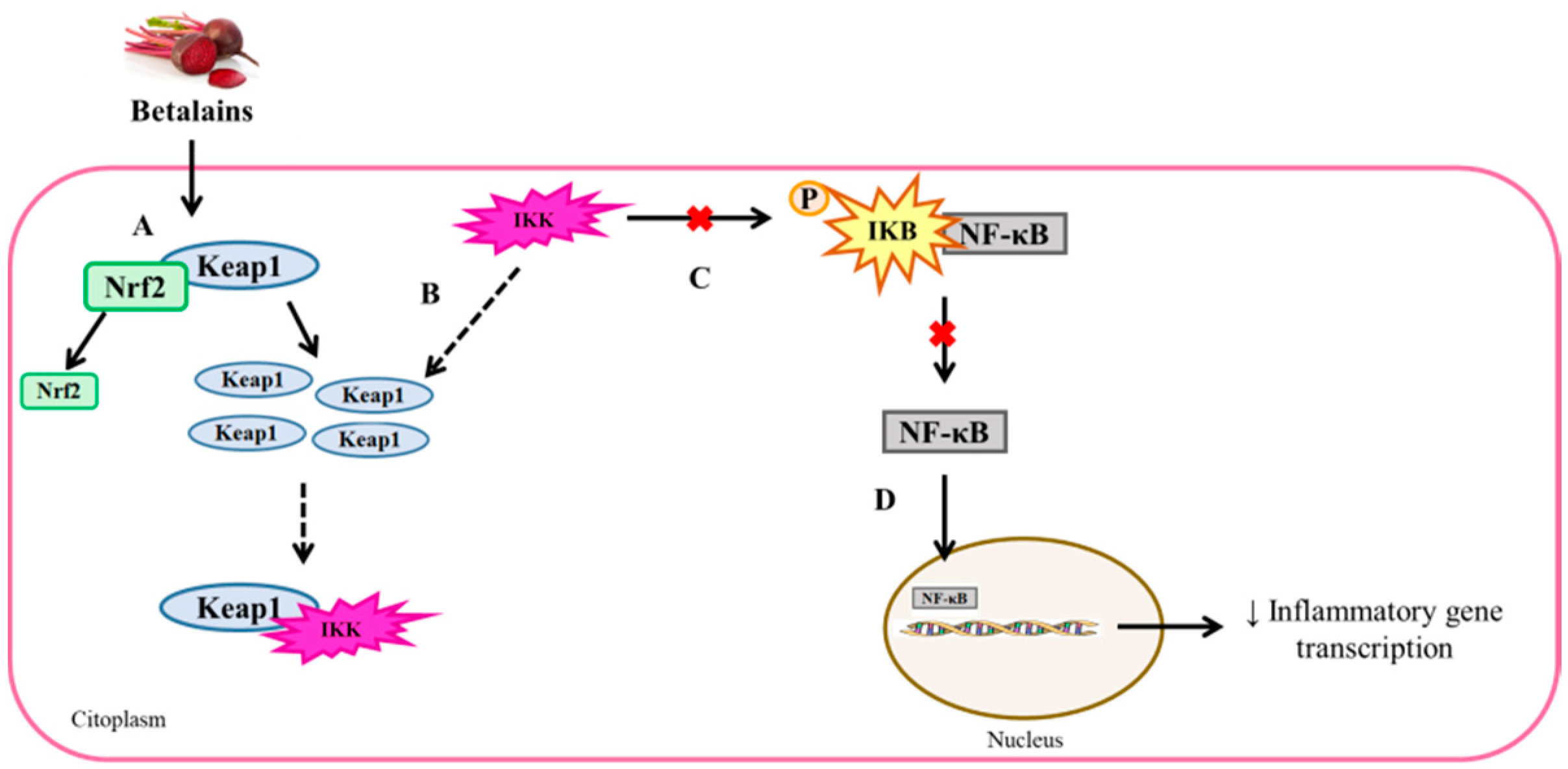
3.2.3. Anti-Inflammatory Effects
3.3. Interactions and Adverse Effects of Betalains
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef]
- Rees, K.; Hartley, L.; Flowers, N.; Clarke, A.; Hooper, L.; Thorogood, M.; Stranges, S.N. “Mediterranean” dietary pattern for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2013, 8, CD009825. [Google Scholar] [CrossRef]
- Gee, L.C.; Ahluwalia, A. Dietary Nitrate Lowers Blood Pressure: Epidemiological, Pre-clinical Experimental and Clinical Trial Evidence. Curr. Hypertens. Rep. 2016, 18, 17. [Google Scholar] [CrossRef]
- Papadaki, A.; Martínez-González, M.A.; Alonso-Gómez, A.; Rekondo, J.; Salas-Salvadó, J.; Corella, D.; Ros, E.; Fitó, M.; Estruch, R.; Lapetra, J.; et al. Mediterranean diet and risk of heart failure: Results from the PREDIMED randomized controlled trial. Eur. J. Heart Fail. 2017, 19, 1179–1185. [Google Scholar] [CrossRef]
- Clifford, T.; Howatson, G.; West, D.J.; Stevenson, E. The Potential Benefits of Red Beetroot Supplementation in Health and Disease. Nutrients 2015, 7, 2801–2822. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Carlström, M.; Weitzberg, E. Metabolic Effects of Dietary Nitrate in Health and Disease. Cell Metab. 2018, 28, 9–22. [Google Scholar] [CrossRef]
- Arciero, P.J.; Miller, V.J.; Ward, E. Performance enhancing diets and the PRISE Protocol to optimize athletic performance. J. Nutr. Metab. 2015, 2015, 715859. [Google Scholar] [CrossRef]
- Zamani, H.; de Joode, M.E.J.R.; Hossein, I.J.; Henckens, N.F.T.; Guggeis, M.A.; Berends, J.E.; de Kok, T.M.C.M.; van Breda, S.J.G. The benefits and risks of beetroot juice consumption: A systematic review. Crit. Rev. Food Sci. Nutr. 2021, 61, 788–804. [Google Scholar] [CrossRef]
- Lidder, S.; Webb, A.J. Vascular effects of dietary nitrate (as found in green leafy vegetables and beetroot) via the nitrate-nitrite-nitric oxide pathway. Br. J. Clin. Pharmacol. 2013, 75, 677–696. [Google Scholar] [CrossRef]
- Nyakayiru, J.; van Loon, L.J.C.; Verdijk, L.B. Could intramuscular storage of dietary nitrate contribute to its ergogenic effect? A mini-review. Free Radic. Biol. Med. 2020, 152, 295–300. [Google Scholar] [CrossRef]
- Hoon, M.W.; Johnson, N.A.; Chapman, P.G.; Burke, L.M. The effect of nitrate supplementation on exercise performance in healthy individuals: A systematic review and meta-analysis. Int. J. Sport Nutr. Exerc. Metab. 2013, 23, 522–532. [Google Scholar] [CrossRef]
- Van der Avoort, C.M.T.; Van Loon, L.J.C.; Hopman, M.T.E.; Verdijk, L.B. Increasing vegetable intake to obtain the health promoting and ergogenic effects of dietary nitrate. Eur. J. Clin. Nutr. 2018, 72, 1485–1489. [Google Scholar] [CrossRef]
- Moreno, B.; Soto, K.; González, D. Nitrate consumption and potential beneficial effect on cardiovascular health. Rev. Chil. Nutr. 2015, 42, 199–205. [Google Scholar]
- Weitzberg, E.; Lundberg, J.O. Novel aspects of dietary nitrate and human health. Annu. Rev. Nutr. 2013, 33, 129–159. [Google Scholar] [CrossRef]
- Gangolli, S.D.; van den Brandt, P.A.; Feron, V.J.; Janzowsky, C.; Koeman, J.H.; Speijers, G.J.; Spiegelhalder, B.; Walker, R.; Wisnok, J.S. Nitrate, nitrite and N-nitroso compounds. Eur. J. Pharmacol. 1994, 292, 1–38. [Google Scholar] [CrossRef]
- Walker, R. The metabolism of dietary nitrites and nitrates. Biochem. Soc. Trans. 1996, 24, 780–785. [Google Scholar] [CrossRef]
- Hezel, M.P.; Weitzberg, E. The oral microbiome and nitric oxide homoeostasis. Oral Dis. 2015, 21, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.A.; Nichols, A.L.; Honavar, J.; Dransfield, M.T.; Matalon, S.; Patel, R.K. Measuring nitrate reductase activity from human and rodent tongues. Nitric Oxide 2017, 66, 62–70. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Govoni, M. Inorganic nitrate is a possible source for systemic generation of nitric oxide. Free Radic. Biol. Med. 2004, 37, 395–400. [Google Scholar] [CrossRef]
- Piknova, B.; Park, J.W.; Swanson, K.M.; Dey, S.; Noguchi, C.T.; Schechter, A.N. Skeletal muscle as an endogenous nitrate reservoir. Nitric Oxide 2015, 47, 10–16. [Google Scholar] [CrossRef]
- Stamler, J.S.; Meissner, G. Physiology of nitric oxide in skeletal muscle. Physiol. Rev. 2001, 81, 209–237. [Google Scholar] [CrossRef]
- Wylie, L.J.; Park, J.W.; Vanhatalo, A.; Kadach, S.; Black, M.I.; Stoyanov, Z.; Schechter, A.N.; Jones, A.M.; Piknova, B. Human skeletal muscle nitrate store: Influence of dietary nitrate supplementation and exercise. J. Physiol. 2019, 597, 5565–5576. [Google Scholar] [CrossRef]
- Sobko, T.; Marcus, C.; Govoni, M.; Kamiya, S. Dietary nitrate in Japanese traditional foods lowers diastolic blood pressure in healthy volunteers. Nitric Oxide 2010, 22, 136–140. [Google Scholar] [CrossRef]
- Coles, L.T.; Clifton, P.M. Effect of beetroot juice on lowering blood pressure in free-living, disease-free adults: A randomized, placebo-controlled trial. Nutr. J. 2012, 11, 106. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, D.A.; Kaffa, N.; George, T.W.; Methven, L.; Lovegrove, J.A. Blood pressure-lowering effects of beetroot juice and novel beetroot-enriched bread products in normotensive male subjects. Br. J. Nutr. 2012, 108, 2066–2074. [Google Scholar] [CrossRef]
- Webb, A.J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; et al. Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension 2008, 51, 784–790. [Google Scholar] [CrossRef]
- Ocampo, D.A.B.; Paipilla, A.F.; Marín, E.; Vargas-Molina, S.; Petro, J.L.; Pérez-Idárraga, A. Dietary Nitrate from Beetroot Juice for Hypertension: A Systematic Review. Biomolecules 2018, 8, 134. [Google Scholar] [CrossRef]
- Kelly, J.; Fulford, J.; Vanhatalo, A.; Blackwell, J.R.; French, O.; Bailey, S.J.; Gilchrist, M.; Winyard, P.G.; Jones, A.M. Effects of short-term dietary nitrate supplementation on blood pressure, O2 uptake kinetics, and muscle and cognitive function in older adults. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 304, R73–R83. [Google Scholar] [CrossRef]
- Toledo, E.; Hu, F.B.; Estruch, R.; Buil-Cosiales, P.; Corella, D.; Salas-Salvadó, J.; Covas, M.I.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Effect of the Mediterranean diet on blood pressure in the PREDIMED trial: Results from a randomized controlled trial. BMC Med. 2013, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Poels, M.M.F.; Ikram, M.A.; Vernooij, M.W.; Krestin, G.P.; Hofman, A.; Niessen, W.J.; van der Lugt, A.; Breteler, M.B.M. Total cerebral blood flow in relation to cognitive function: The Rotterdam Scan Study. J. Cereb. Blood Flow Metab. 2008, 28, 1652–1655. [Google Scholar] [CrossRef]
- Presley, T.D.; Morgan, A.R.; Bechtold, E.; Clodfelter, W.; Dove, R.W.; Jennings, J.M.; Kraft, R.A.; King, S.B.; Laurienti, P.J.; Rejeski, W.J.; et al. Acute effect of a high nitrate diet on brain perfusion in older adults. Nitric Oxide 2011, 24, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Bond, V.; Curry, B.H.; Adams, R.G.; Asadi, M.S.; Millis, R.M.; Haddad, G.E. Effects of dietary nitrates on systemic and cerebrovascular hemodynamics. Cardiol. Res. Pract. 2013, 2013, 435629. [Google Scholar] [CrossRef]
- Wightman, E.L.; Haskell-Ramsay, C.F.; Thompson, K.G.; Blackwell, J.R.; Winyard, P.G.; Forster, J.; Jones, A.M.; Kennedy, D.O. Dietary nitrate modulates cerebral blood flow parameters and cognitive performance in humans: A double-blind, placebo-controlled, crossover investigation. Physiol. Behav. 2015, 149, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Joris, P.J.; Mensink, R.P.; Adam, T.C.; Liu, T.T. Cerebral Blood Flow Measurements in Adults: A Review on the Effects of Dietary Factors and Exercise. Nutrients 2018, 10, 530. [Google Scholar] [CrossRef]
- Domínguez, R.; Cuenca, E.; Maté-Muñoz, J.L.; García-Fernández, P.; Serra-Paya, N.; Lozano-Estevan, M.D.C.; Veiga-Herreros, P.; Garnacho-Castaño, M.V. Effects of Beetroot Juice Supplementation on Cardiorespiratory Endurance in Athletes. A Systematic Review. Nutrients 2017, 9, 43. [Google Scholar] [CrossRef]
- Cholewa, J.; Trexler, E.; Lima-Soares, F.; de Araújo Pessôa, K.; Sousa-Silva, R.; Moura Santos, A.; Zhi, X.; Nicastro, H.; Torres Cabido, C.E.; de Freitas, M.C.; et al. Effects of dietary sports supplements on metabolite accumulation, vasodilation and cellular swelling in relation to muscle hypertrophy: A focus on “secondary” physiological determinants. Nutrition 2019, 60, 241–251. [Google Scholar] [CrossRef]
- Porcelli, S.; Pugliese, L.; Rejc, E.; Pavei, G.; Bonato, M.; Montorsi, M.; La Torre, A.; Rasica, L.; Marzorati, M. Effects of a Short-Term High-Nitrate Diet on Exercise Performance. Nutrients 2016, 8, 534. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Paula, G.H.; Pinheiro, L.C.; Tanus-Santos, J.E. Mechanisms impairing blood pressure responses to nitrite and nitrate. Nitric Oxide 2019, 85, 35–43. [Google Scholar] [CrossRef]
- Berends, J.E.; van den Berg, L.M.M.; Guggeis, M.A.; Henckens, N.F.T.; Hossein, I.J.; de Joode, M.E.J.R.; Zamani, H.; van Pelt, K.A.A.J.; Beelen, N.A.; Kuhnle, G.G.; et al. Consumption of Nitrate-Rich Beetroot Juice with or without Vitamin C Supplementation Increases the Excretion of Urinary Nitrate, Nitrite, and N-nitroso Compounds in Humans. Int. J. Mol. Sci. 2019, 20, 2277. [Google Scholar] [CrossRef]
- Ashworth, A.; Bescos, R. Dietary nitrate and blood pressure: Evolution of a new nutrient? Nutr. Res. Rev. 2017, 30, 208–219. [Google Scholar] [CrossRef]
- Lee, C.H.; Wettasinghe, M.; Bolling, B.W.; Ji, L.L.; Parkin, K.L. Betalains, Phase II Enzyme-Inducing Components From Red Beetroot (Beta vulgaris L.) Extracts. Nutr. Cancer 2005, 53, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Sawicki, T.; Baczek, N.; Wiczkowski, W. Betalain profile, content and antioxidant capacity of red beetroot dependent on the genotype and root part. J. Funct. Foods 2016, 27, 249–261. [Google Scholar] [CrossRef]
- Akbar Hussain, E.; Sadiq, Z.; Zia-Ul-Haq, M. Bioavailability of Betalains. In Betalains: Biomolecular Aspects; Akbar Hussain, E., Sadiq, Z., Zia-Ul-Haq, M., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Volume 1, pp. 166–173. [Google Scholar]
- Lee, E.J.; An, D.; Nguyen, C.T.T.; Patil, B.S.; Kim, J.; Yoo, K.S. Betalain and Betaine Composition of Greenhouse- or Field-Produced Beetroot (Beta vulgaris L.) and Inhibition of HepG2 Cell Proliferation. J. Agric. Food Chem. 2014, 62, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Giridhar, P. Plant betalains: Chemistry and biochemistry. Phytochemistry 2015, 117, 267–295. [Google Scholar] [CrossRef]
- Tesoriere, L.; Allegra, M.; Butera, D.; Livrea, M.A. Absorption, excretion, and distribution of dietary antioxidant betalains in LDLs: Potential health effects of betalains in humans. Am. J. Clin. Nutr. 2004, 80, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Tesoriere, L.; Gentile, C.; Angileri, F.; Attanzio, A.; Tutone, M.; Allegra, M.; Livrea, M.A. Trans-epithelial transport of the betalain pigments indicaxanthin and betanin across Caco-2 cell monolayers and influence of food matrix. Eur. J. Nutr. 2013, 52, 1077–1087. [Google Scholar] [CrossRef]
- Kanner, J.; Harel, S.; Granit, R. Betalains—A New Class of Dietary Cationized Antioxidants. J. Agric. Food Chem. 2001, 49, 5178–5185. [Google Scholar] [CrossRef]
- Frank, T.; Stintzing, F.Z.; Carle, R.; Bitsch, I.; Quaas, D.; Strass, G.; Bitsch, R.; Netzel, M. Urinary pharmacokinetics of betalains following consumption of red beet juice in healthy humans. Pharmacol. Res. 2005, 52, 290–297. [Google Scholar] [CrossRef]
- Rahimi, M.; Mesbah-Namin, S.A.; Ostadrahimi, A.; Abedimanesh, S.; Separham, A.; Jafarabadi, M.A. Effects of betalains on atherogenic risk factors in patients with atherosclerotic cardiovascular disease. Food Funct. 2019, 10, 8286–8297. [Google Scholar] [CrossRef]
- Xie, R.; Jia, D.; Gao, C.; Zhou, J.; Sui, H.; Wei, X.; Zhang, T.; Han, Y.; Shi, J.; Bai, Y. Homocysteine induces procoagulant activity of red blood cells via phosphatidylserine exposure and microparticles generation. Amino Acids 2014, 46, 1997–2004. [Google Scholar] [CrossRef]
- Rahimi, M.; Abedimanesh, S.; Mesbah-Namin, S.A.; Ostadrahimi, A. Betalains, the nature-inspired pigments, in health and diseases. Crit. Rev. Food Sci. Nutr. 2019, 59, 2949–2978. [Google Scholar] [CrossRef]
- Reddy, M.K.; Alexander-Lindo, R.L.; Nair, M.G. Relative inhibition of lipid peroxidation, cyclooxygenase enzymes, and human tumor cell proliferation by natural food colors. J. Agric. Food Chem. 2005, 53, 9268–9273. [Google Scholar] [CrossRef]
- Pietrzkowski, Z.; Thresher, W.C. Solid betalain compositions and methods. U.S. Patent WO 2010/014839 A1, 25 March 2010. [Google Scholar]
- Esatbeyoglu, T.; Wagner, A.E.; Motafakkerazad, R.; Nakajima, Y.; Matsugo, S.; Rimbach, G. Free radical scavenging and antioxidant activity of betanin: Electron spin resonance spectroscopy studies and studies in cultured cells. Food Chem. Toxicol. 2014, 73, 119–126. [Google Scholar] [CrossRef]
- Szaefer, H.; Krajka-Kuźniak, V.; Ignatowicz, E.; Adamska, T.; Baer-Dubowska, W. Evaluation of the Effect of Beetroot Juice on DMBA-induced Damage in Liver and Mammary Gland of Female Sprague–Dawley Rats. Phytother. Res. 2014, 28, 55–61. [Google Scholar] [CrossRef]
- Han, J.; Zhang, Z.; Yang, S.; Wang, J.; Yang, X.; Tan, D. Betanin attenuates paraquat-induced liver toxicity through a mitochondrial pathway. Food Chem. Toxicol. 2014, 70, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Sutariya, B.; Saraf, M. Betanin, isolated from fruits of Opuntia elatior Mill attenuates renal fibrosis in diabetic rats through regulating oxidative stress and TGF-β pathway. J. Ethnopharmacol. 2017, 198, 432–443. [Google Scholar] [PubMed]
- Ahmadian, E.; Khosroushahi, A.Y.; Eghbal, M.A.; Eftekhari, A. Betanin reduces organophosphate induced cytotoxicity in primary hepatocyte via an anti-oxidative and mitochondrial dependent pathway. Pestic. Biochem. Physiol. 2018, 144, 71–78. [Google Scholar] [CrossRef]
- Motawi, T.K.; Ahmed, S.A.; El-Boghdady, N.A.; Metwally, N.S.; Nasr, N.N. Impact of betanin against paracetamol and diclofenac induced hepato-renal damage in rats. Biomarkers 2020, 25, 86–93. [Google Scholar] [PubMed]
- Krajka-Kuźniak, V.; Paluszczak, J.; Szaefer, H.; Baer-Dubowska, W. Betanin, a beetroot component, induces nuclear factor erythroid-2-related factor 2-mediated expression of detoxifying/antioxidant enzymes in human liver cell lines. Br. J. Nutr. 2013, 110, 2138–2149. [Google Scholar] [CrossRef] [PubMed]
- Silva, D.V.T.D.; Baião, D.D.S.; Ferreira, V.F.; Paschoalin, V.M.F. Betanin as a multipath oxidative stress and inflammation modulator: A beetroot pigment with protective effects on cardiovascular disease pathogenesis. Crit. Rev. Food Sci. Nutr. 2020, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gentile, C.; Tesoriere, L.; Allegra, M.; Livrea, M.A.; D’Alessio, P. Antioxidant betalains from cactus pear (Opuntia ficus-indica) inhibit endothelial ICAM-1 expression. Ann. N. Y. Acad. Sci. 2004, 1028, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I. Plant Betalains: Safety, Antioxidant Activity, Clinical Efficacy, and Bioavailability. Compr. Rev. Food Sci. Food Saf. 2016, 15, 316–330. [Google Scholar] [CrossRef]
- Vidal, P.J.; López-Nicolás, J.M.; Gandía-Herrero, F.; García-Carmona, F. Inactivation of lipoxygenase and cyclooxygenase by natural betalains and semi-synthetic analogues. Food Chem. 2014, 154, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Wang, Y.; Bai, B.; Yang, X.; Han, J. Betanin attenuates oxidative stress and inflammatory reaction in kidney of paraquat-treated rat. Food Chem. Toxicol. 2015, 78, 141–146. [Google Scholar] [CrossRef]
- El Gamal, A.A.; AlSaid, M.S.; Raish, M.; Al-Sohaibani, M.; Al-Massarani, S.M.; Ahmad, A.; Hefnawy, M.; Al-Yahya, M.; Basoudan, O.A.; Rafatullah, S. Beetroot (Beta vulgaris L.) extract ameliorates gentamicin-induced nephrotoxicity associated oxidative stress, inflammation, and apoptosis in rodent model. Mediat. Inflamm. 2014, 2014, 983952. [Google Scholar] [CrossRef]
- Pietrzkowski, Z.; Nemzer, B.; Spórna, A.; Stalica, P.; Tresher, W.; Keller, R.; Jiminez, R.; Michalowski, T.; Wybraniec, S. Influence of betalin-rich extracts on reduction of discomfort associated with osteoarthritis. NewMed 2010, 1, 12–17. [Google Scholar]
- Mitchell, S.C. Food idiosyncrasies: Beetroot and asparagus. Drug Metab. Dispos. 2001, 29, 539–543. [Google Scholar]
| Classification of Vegetables and Fruits According to Their NO3− Content (mg/kg Wet Weight) | ||||
|---|---|---|---|---|
| Very low (<200 mg/kg) | Low (200–500 mg/kg) | Medium (500–1000 mg/kg) | High (1000–2500 mg/kg) | Very high (>2500 mg/kg) |
| Garlic Artichoke Onion Melon Pear Potato Tomato | Chikory Broccoli Cauliflower Carrot Cucumber | Turnip Cabbage | Escarole Leek Parsley Celery-root | Beetroot Spinach Lettuce Radish Celery Chard |
| Reference | Population | Intervention | Comparison | Outcome |
|---|---|---|---|---|
| [24] | 25 healthy, physically active adults (15 m and 10 w): - mean age 36 ± 10 years - BMI < 18.5 | Consumption of a Japanese diet for 10 days (providing 18.8 mg/kg bw/day nitrate) | The controls received a non-Japanese diet (providing ≤3.7 mg/kg bw/day nitrate) for the same period | Increased plasma and saliva nitrate and nitrite levels. Significant decrease in DBP (4.5 mmHg). |
| [25] | 30 healthy adults (15 m and 15 w) with a SBP > 120 mmHg | Single intake of 500 g BJ (containing 15 mmol nitrate/L) | Single ingestion of 500 g PL (apple juice concentrate) | Significant reduction of SBP (4–5 mmHg) in men 6 h. |
| [26] | 18 normotensive healthy adults (18 m) | Single administration of 100, 250 or 500 g BJ diluted in mineral water (total weight of the mixture 500 g) | The controls were administered the same dose (500 g) of mineral water | SBP and DBP significantly reduced (dose dependently) over a period of 24 h. |
| [26] | 14 normotensive healthy adults (14 m) | Single ingestion of 200 g of bread enriched with red or white beetroot (50% of the total weight) | The controls received 200 g of white bread | Significant DBP reduction over a period of 24 h. |
| [29] | 12 old healthy adults (6 m, 6 w) | Prescription of 140 mL/d BJ (containing ≈ 9.6 mmol nitrate) during 2.5 days followed by a three-day washout period (this protocol was repeated during 6 weeks) | The controls received PL (nitrate depleted BJ) under the same conditions | A significant reduction in resting SBP, DBP and VO2 was found. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milton-Laskibar, I.; Martínez, J.A.; Portillo, M.P. Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease. Foods 2021, 10, 1314. https://doi.org/10.3390/foods10061314
Milton-Laskibar I, Martínez JA, Portillo MP. Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease. Foods. 2021; 10(6):1314. https://doi.org/10.3390/foods10061314
Chicago/Turabian StyleMilton-Laskibar, Iñaki, J. Alfredo Martínez, and María P. Portillo. 2021. "Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease" Foods 10, no. 6: 1314. https://doi.org/10.3390/foods10061314
APA StyleMilton-Laskibar, I., Martínez, J. A., & Portillo, M. P. (2021). Current Knowledge on Beetroot Bioactive Compounds: Role of Nitrate and Betalains in Health and Disease. Foods, 10(6), 1314. https://doi.org/10.3390/foods10061314









